Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
Introduction
Material and Methods
Study Area
Source of Data
Genetic Analysis
Results and Discussion
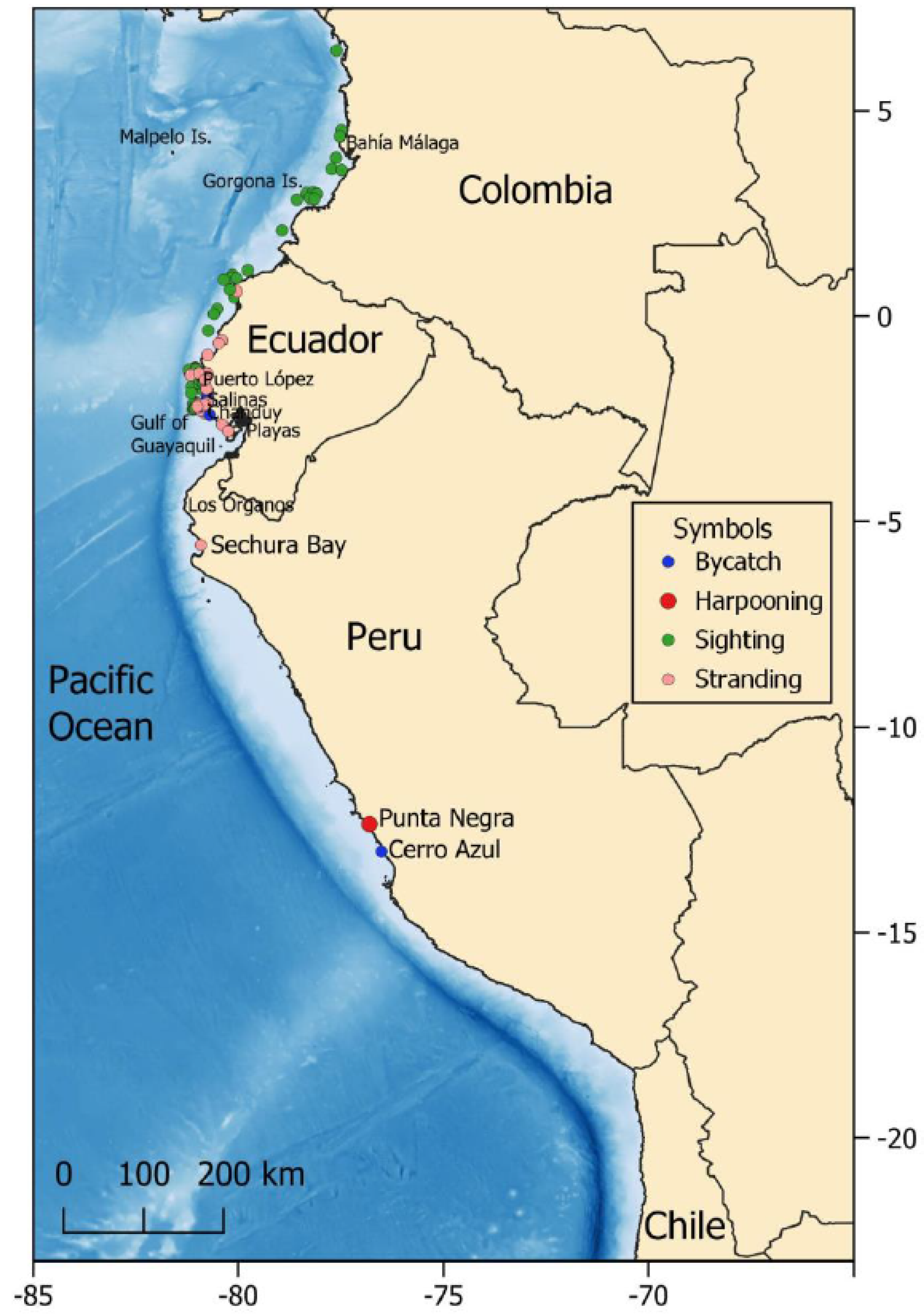
Distribution Off Western South America
Colombia
Ecuador
Peru
Chile
Life History
Pigmentation, External Morphology
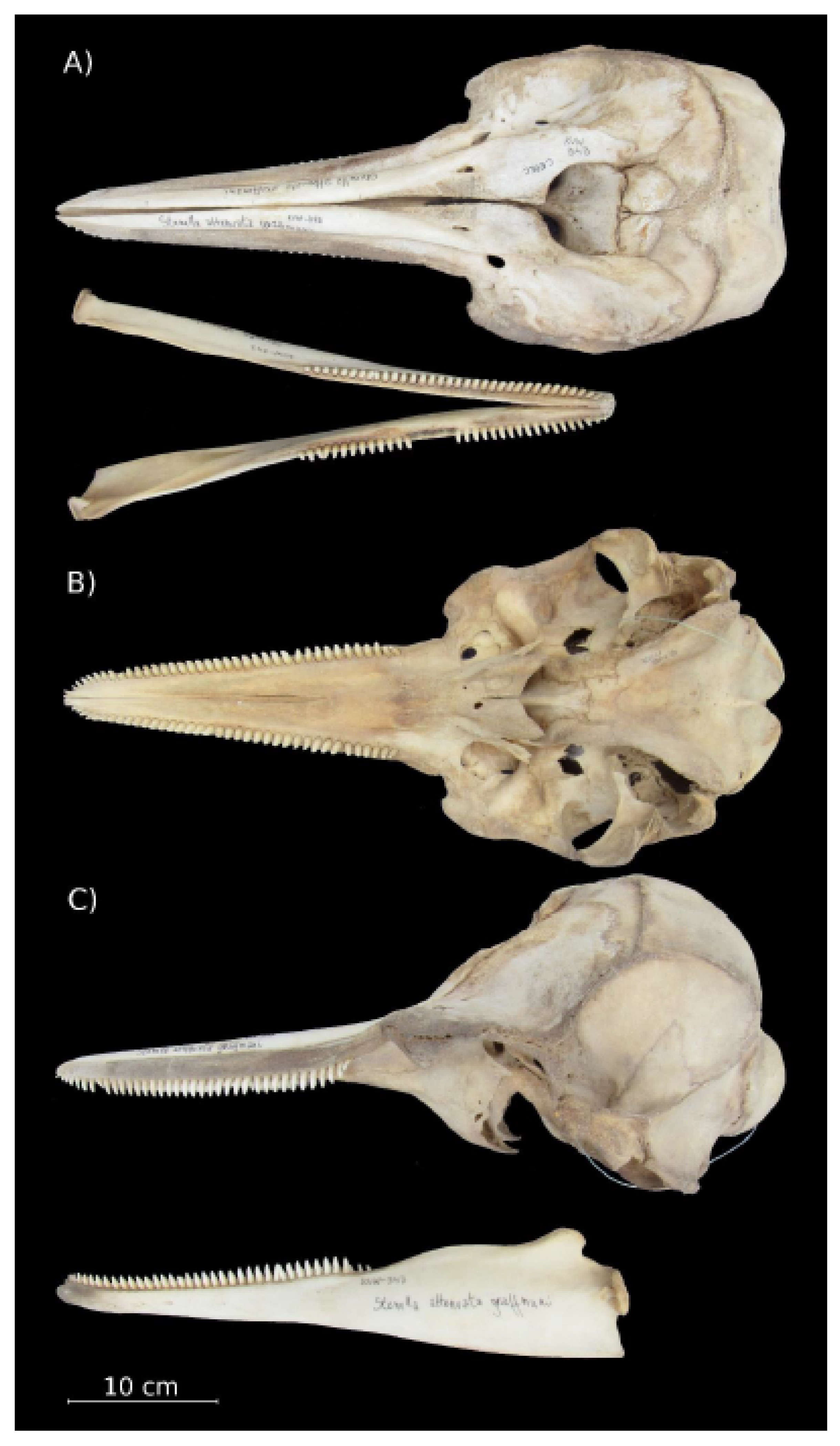
Cranial Characteristics
Reproduction and Growth
Feeding
Helminth Parasites and Pathology
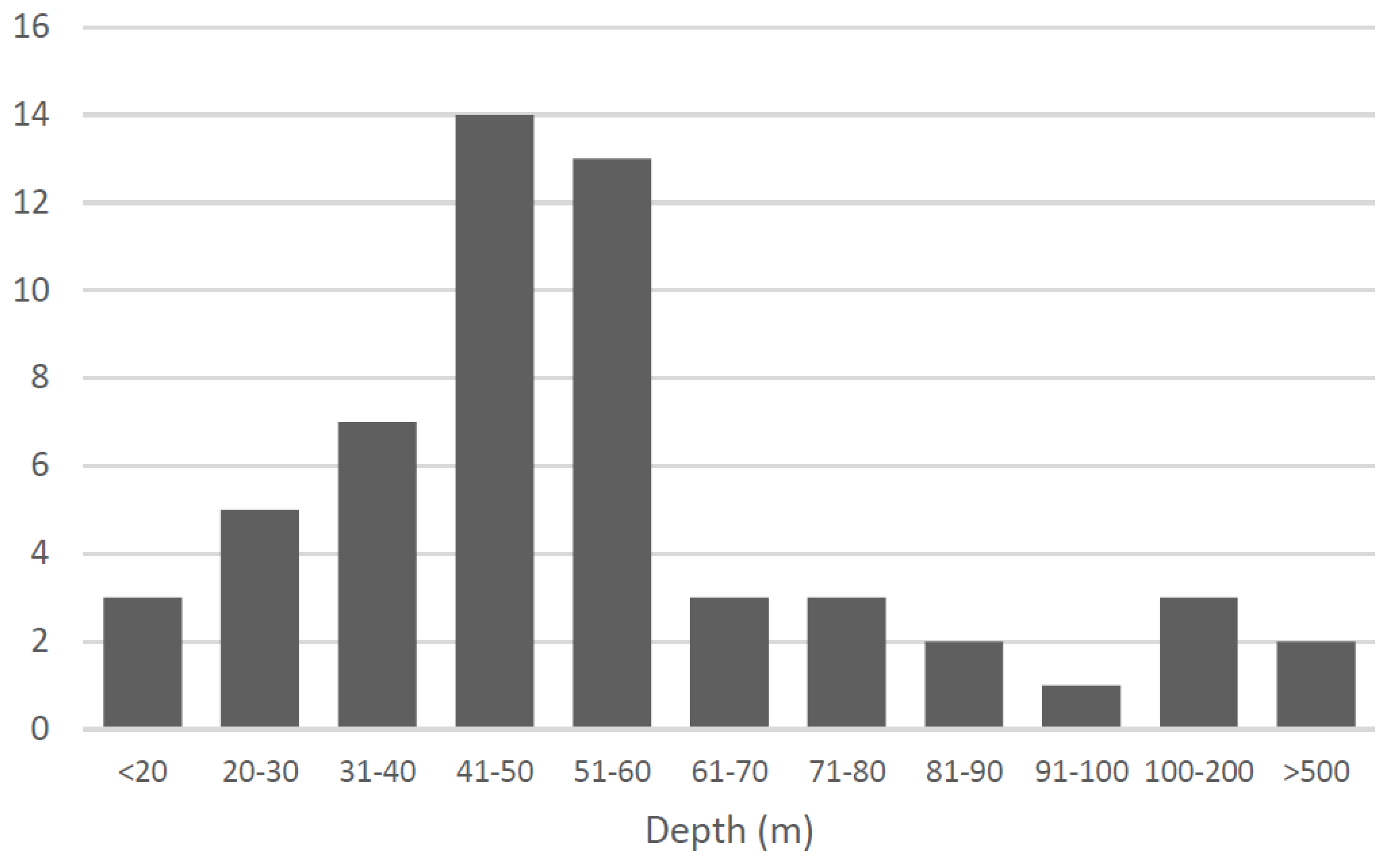
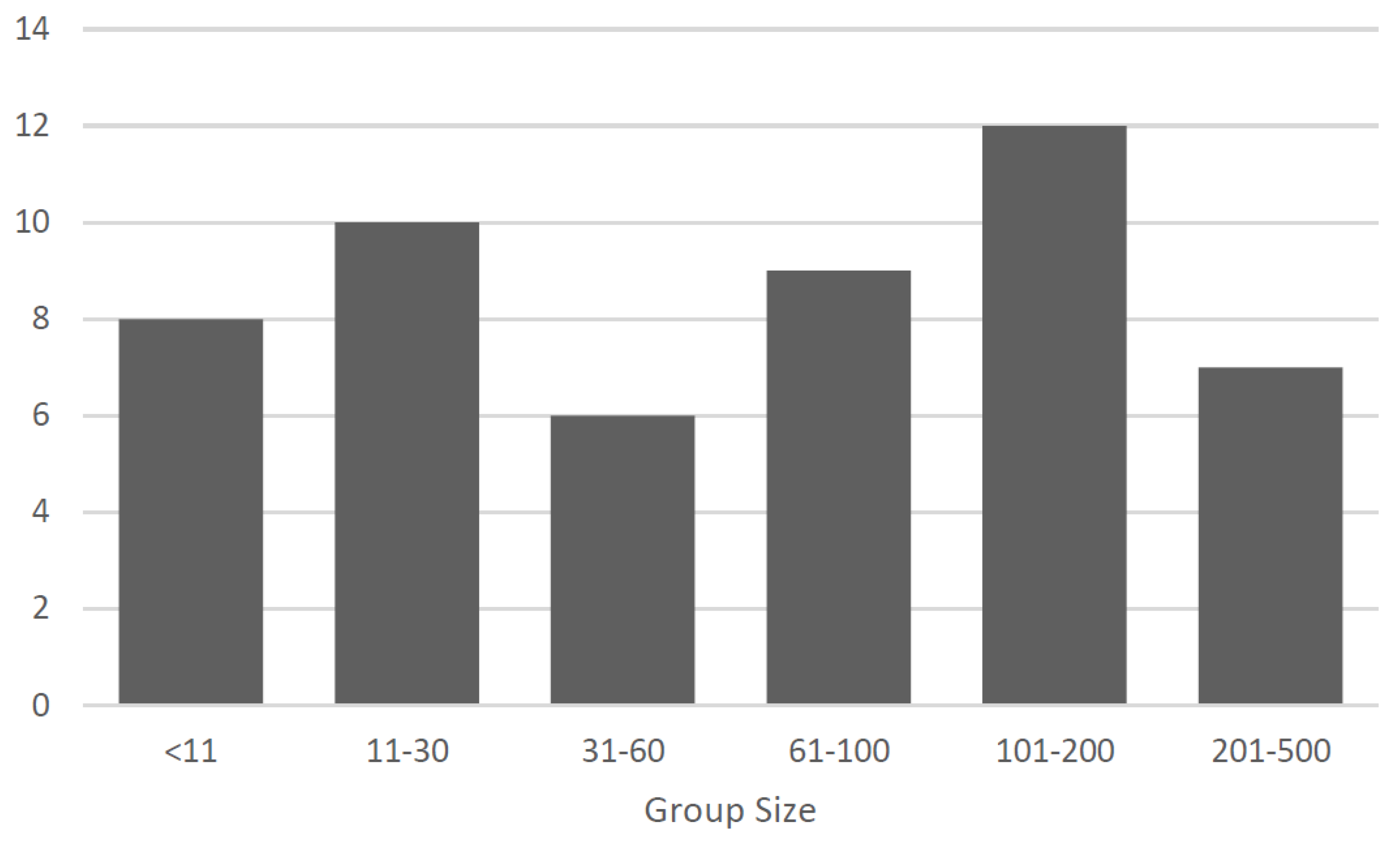
Behavioral Ecology
Genetic Diversity and Population Structure
Fisheries Interactions and Other Threats
Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Aguayo, A. Progress report on small cetacean research in Chile. Journal Fisheries Research Board of Canada 1975, 32, 1123–1143. [Google Scholar] [CrossRef]
- Aguayo-Lobo, A.; Torres Navarro, D.; Acevedo Ramírez, J. Los mamíferos marinos de Chile: I. Cetacea. Ser. Cient. INACH 1998a, 48, 19–159. [Google Scholar]
- Aguayo, A.; Bernal, R.; Olavarria, C.; Vallejos, V.; Hucke, R. Observaciones de cetáceos realizadas entre Valparaiso e isla de Pascua, Chile, durante los inviernos de 1993, 1994 y 1995. Revista de Biología Marina y Oceanografía 1998b, 33(1), 101–123. [Google Scholar]
- Aguilar, A.; Forcada, J.; Gazo, M.; Badosa, E. Los Cetáceos del Parque Nacional de Coiba (Panamá). In Flora y Fauna del Parque Nacional de Coiba (Panamá); Castroviejo, S., Velayos, M., Eds.; Agencia Española de Cooperación Internacional (AECI): Madrid, 1997; pp. 75–102. [Google Scholar]
- Altschul, S.F.; Madden, T.L.; Schäffer, A.A.; Zhang, J.; Zhang, Z.; Miller, W.; Lipman, D.J. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997, 25, 3389–3402. [Google Scholar] [CrossRef] [PubMed]
- Avila, I.C.; Garcia, C.; Bastidas, J.C. A note on the use of dolphins as bait in the artisanal fisheries off Bahía Solano, Chocó, Colombia. J. Cetacean Res. Manag. 2008, 10, 179–182. [Google Scholar] [CrossRef]
- Bandelt, H.J.; Forster, P.; Rohl, A. Median-joining networks for inferring intraspecific phylogenies. Mol. Biol. Evol. 1999, 16, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Barber, R. T.; Chavez, F. P. Biological consequences of El Niño. Science 1983, 222, 1203–1210. [Google Scholar] [CrossRef]
- Bayas-Rea, R.d.L.Á.; Félix, F.; Montufar, R. Genetic divergence and fine scale population structure of the common bottlenose dolphin (Tursiops truncatus, Montagu) found in the Gulf of Guayaquil, Ecuador. PeerJ 2018, 6, e4589. [Google Scholar] [CrossRef] [PubMed]
- Capella, A.J.; Flórez González, L.; Falk, P. E.; Herrera, J.C.; Tobón, I.C.; Hernández, E.; Recalde, A. Plan básico para el manejo de los mamíferos marinos en el PNN Gorgona, Pacífico colombiano; Parques Nacionales Naturales y WWF-Colombia: Cali, Colombia, 2014; p. 72 p. [Google Scholar]
- Cárdenas, J. C.; Stutzin, M. E.; Oporto, J. A.; Cabello, C.; Torres, D. Manual de identificación de los cetáceos chilenos. Proyecto WH-445 Cetáceos-Chile, WWF and CODEFF, Santiago; 1986. [Google Scholar]
- Carwardine, M. Handbook of Whales, dolphins and Porpoises; Bloomsbury Wildlife, London, 2020; p. 528pp. [Google Scholar]
- Castro, C.; Rosero, P. Small cetacean interaction with fishing gear in Machalilla National Park, Ecuador. Document SC/62/BC3, 2010; presented to the Scientific Committee of the International Whaling Commission. [Google Scholar]
- Castro, C.; Van Waerebeek, K. Strandings and mortality of cetaceans due to interactions with fishing nets in Ecuador, 2001-2017. Document SC/68A/HIM/17 presented to the IWC Scientific Committee Meeting, Nairobi, May 2019; 2019; p. 11 pp. [Google Scholar]
- Chiluiza, D.; Aguirre, W.; Félix, F.; Haase, B. Varamientos de mamíferos marinos en la costa continental ecuatoriana período 1987-1995. Acta Oceanográfica del Pacífico. INOCAR 1998, 9(1), 209–217. [Google Scholar]
- Chinacalle-Martínez, N.; García-Rada, E.; López-Macías, J.; Pinoargote, S.; Loor, G.; Zevallos-Rosado, J.; Cruz, P.; Pablo, D.; Andrade, B.; Robalino-Mejía, C.; et al. Oceanic primary production trend patterns along coast of Ecuador. Neotropical Biodivers. 2021, 7, 379–391. [Google Scholar] [CrossRef]
- Cortez-Casamayor, S.; Rodriguez, K.A.; Gubbins, S.; Cortés, D.; Gutierrez, C.; Silva, S.; Alcorta, B.; Pacheco, A.S. Occurrence of pantropical spotted dolphin (Stenella attenuata) in northern Peru: an El Niño effect? Mammalia 2024, 89, 182–188. [Google Scholar] [CrossRef]
- Courbis, S.; Baird, R.W.; Cipriano, F.; Duffield, D. Multiple Populations of Pantropical Spotted Dolphins in Hawaiian Waters. J. Hered. 2014, 105, 627–641. [Google Scholar] [CrossRef]
- Darriba, D.; Taboada, G.L.; Doallo, R.; Posada, D. jModelTest 2: more models, new heuristics and parallel computing. Nat. Methods 2012, 9, 772. [Google Scholar] [CrossRef] [PubMed]
- Dalebout, M.L.; VAN Helden, A.; VAN Waerebeek, K.; Baker, C.S. Molecular genetic identification of southern hemisphere beaked whales (Cetacea: Ziphiidae). Mol. Ecol. 1998, 7, 687–694. [Google Scholar] [CrossRef]
- Day, D. List of cetaceans seen in Galápagos. Noticias de Galápagos 1994, 53, 5–6. [Google Scholar]
- De Weerdt, J.; Ramos, E.A.; Pouplard, E.; Kochzius, M.; Clapham, P. Cetacean strandings along the Pacific and Caribbean coasts of Nicaragua from 2014 to 2021. Mar. Biodivers. Rec. 2021, 14, 1–9. [Google Scholar] [CrossRef]
- Denkinger, J.; Oña, J.; Alarcón, D.; Merlen, G.; Salazar, S.; Palacios. From whaling to whalewatching: cetacean presence and species diversity in the Galapagos Marine Reserve. Science and Conservation in the Galapagos Islands: Frameworks & Perspectives, Social and Ecological Interactions in the Galapagos Islands 2013. [Google Scholar] [CrossRef]
- Dizon, A. E.; Perrin, W. F.; Akin, P. A. Stocks of dolphins (Stenella spp. and Delphinus delphis) in the eastern tropical Pacific: a phylogeographic classification. NOAA Technical Report NMFS 1994, 119, 1–20. [Google Scholar]
- Douglas, M.E.; Schnell, G.D.; Hough, D.J. Differentiation between Inshore and Offshore Spotted Dolphins in the Eastern Tropical Pacific Ocean. J. Mammal. 1984, 65, 375–387. [Google Scholar] [CrossRef]
- Escorza-Treviño, S.; Archer, F.I.; Rosales, M.; Lang, A.; Dizon, A.E. Genetic differentiation and intraspecific structure of Eastern Tropical Pacific spotted dolphins, Stenella attenuata, revealed by DNA analyses. Conserv. Genet. 2005, 6, 587–600. [Google Scholar] [CrossRef]
- Excoffier, L.; Lischer, H.E.L. Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol. Ecol. Resour. 2010, 10, 564–567. [Google Scholar] [CrossRef] [PubMed]
- Félix, F. Organization and social structure of the bottlenose dolphin Tursiops truncatus in the Gulf of Guayaquil, Ecuador. Aquatic Mammals 1997, 23(1), 1–16. [Google Scholar]
- Félix, F.; Samaniego, J. Incidental catches of small cetaceans in the artisanal fisheries of Ecuador. Report International Whaling Commission (Special Issue 15) 1994, 475–480. [Google Scholar]
- Félix, F.; Castro, C. Occurrence, abundance and some ecological aspects of the offshore bottlenose dolphin off Ecuador’s central coast. Lat. Am. J. Aquat. Mamm. 2023. [Google Scholar] [CrossRef]
- Félix, F.; Haase, B.; Denkinger, J.; Falconí, J. Varamientos de mamíferos marinos registrados en la costa continental de Ecuador entre 1996 y 2009. Acta Oceano Del. Pac 2011, 16, 61–73. [Google Scholar]
- Félix, F.; Castro, C.; Barragán-Tabares, L.; Orellana-Vásquez, H.; Platt, M. THE BRYDE’S WHALE ON THE CENTRAL COAST OF ECUADOR, DISTRIBUTION, BEHAVIOR AND ECOLOGICAL INSIGHTS. J. Cetacean Res. Manag. 2025, 26, 87–100. [Google Scholar] [CrossRef]
- Fiedler, P. Environmental change in the eastern tropical Pacific Ocean: review of ENSO and decadal variability. Mar. Ecol. Prog. Ser. 2002, 244, 265–283. [Google Scholar] [CrossRef]
- Findlay, K.; Pitman, R.; Tsurui, T.; Sakai, K.; Ensor, P.; Iwakami, H.; Ljungblad, D.; Shimada, H.; Thiele, D.; Van Waerebeek, K.; Hucke-Gaete, R.; Sanino-Vattier, G.P. 1997/1998 IWC-Southern Ocean whale and ecosystem research (IWC-SOWER) blue whale cruise, Chile. Document SC/50/Rep2 presented to the IWC Scientific Committee, Muscat, Oman, May 1998; 1998. [Google Scholar]
- Flórez-González, L.; Capella, J.; Falk-Fernández, P. Guia de campo de los mamíferos acuaticos de Colombia; Editorial Sepia Ltda: Santiago de Cali, Colombia, 2004; p. 124 pp. [Google Scholar]
- Fu, Y.-X. Statistical Tests of Neutrality of Mutations Against Population Growth, Hitchhiking and Background Selection. Genetics 1997, 147, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Garcia, C.; Dawson, S.M. Distribution of pantropical spotted dolphins in Pacific coastal waters of Panama. Lat. Am. J. Aquat. Mamm. 2003, 2, 29–38. [Google Scholar] [CrossRef]
- García, C.; Soler, G.; Avila, I.C.; Bessudo, S. Observaciones preliminares de la presencia y distribución estacional de Mamíferos Marinos del Pacífico colombiano. p. 123. Resúmenes Primera Reunión Internacional sobre el Estudio de los Mamíferos Acuáticos SOMEMMA – SOLAMAC, Mérida, México, 2006. [Google Scholar]
- García, C.; Avila, I.C.; Palacios, D.; Gerrodette, T.; Suárez, M.; Soler, G.; Rasmussen, K.; May-Collado, L.; Parson, C.; Trujillo, F.; Bessudo, S. Presence, distribution and threats of Stenella attenuata and Tursiops truncatus in the Pacific waters of Panamá and Colombia. In Resumen 310 en Memorias XIII Reunión de Trabajo de Especialistas en Mamíferos Acuáticos de América del Sur y 7° Congreso SOLAMAC; Montevideo, Uruguay, 2008. [Google Scholar]
- Garcia-Cegarra, A.M.; Toro, F.; Gonzalez-Borasca, V. Citizen science as a tool to assess cetacean diversity in the Atacama Desert coast. Ocean Coast. Manag. 2021, 213. [Google Scholar] [CrossRef]
- Gerrodette, T.; Forcada, J. Non-recovery of two spotted and spinner dolphin populations in the eastern tropical Pacific Ocean. Mar. Ecol. Prog. Ser. 2005, 291, 1–21. [Google Scholar] [CrossRef]
- Zamorano-Abramson, J.; Gibbons, J.; Capella, J. DIVERSITY AND SUMMER DISTRIBUTION OF CETACEANS IN INLET WATERS OF NORTHERN AISÉN, CHILE. An. del Inst. de la Patagon. 2010, 38, 151–157. [Google Scholar] [CrossRef]
- Gibbons, J. A.; Waring, G. T. Observations of coastal and offshore dolphins in Chilean waters. 2013. [Google Scholar]
- Marine Ecology Progress Series 493, 283–295.
- Gray, J.E. The Zoology of the Voyage of H.M.S. Erebus and Terror; Richardson, J., Gray, J.E., Eds.; London, 1846. [Google Scholar]
- Gerrodette, T.; Palacios, D. Estimates of cetacean abundance in EEZ. Waters of the Eastern Tropical Pacific. Administrative Report LJ 96-10; NMFS - SWFSC: La Jolla, CA, 1996. [Google Scholar]
- Guerra-Correa, C.; Van Waerebeek, K.; Portflitt, G.; Luna, G. Presencia de cetáceos frente a la segúnda región de Chile. Estudios Oceanológicos 1987, 6, 87–96. [Google Scholar]
- Hamilton, T.A.; Redfern, J.; Barlow, J.; Balance, L.T.; Gerrodette, T.; Holt, R.S.; Forney, K.A.; Taylor, B.L. NOAA-TM-NMFS-SWFSC-440; Atlas of cetacean sightings for Southwest Fisheries Science Center cetacean and ecosystem surveys: 1986-2005. 2009.
- Herrera, J.C.; Ávila, I.; Falk, P.; Soler, G.; García, C.; Tobón, I.; Capella, J. Los Mamíferos Marinos en el Santuario de Fauna y Flora Malpelo y Aguas hacia el Continente, Pacífico Colombiano. DIMAR-CCCP y UAESPNN. Santuario de Fauna y Flora Malpelo: descubrimiento en marcha, (Ed.) DIMAR 2007, 113–130. [Google Scholar]
- Herrera, J. C.; Flórez-González, L.; Giraldo, A. Mamíferos Acuáticos. In Colombia Pacífico: Una visión sobre su biodiversidad marina; Cantera, J., Londoño, E., Eds.; Editorial Universidad del Valle: Cali, 2011a; pp. 393–402. [Google Scholar]
- Carmona, J.C.H.; Alzueta, J.J.C.; Soler, G.A.; Bessudo, S.; García, C.; González, L.F. OCURRENCIA Y TASAS DE ENCUENTRO DE MAMÍFEROS MARINOS EN LAS AGUAS DE LA ISLA MALPELO Y HACIA EL CONTINENTE. Bull. Mar. Coast. Res. 2016, 40. [Google Scholar] [CrossRef]
- Herrera, J.C.; Flórez-González, L. Registro de cetáceos durante los cruceros de evaluación pesquera de pequeños pelágicos sobre la plataforma continental del Pacífico de Colombia. In Cruceros de evaluación de pequeños pelágicos en el Pacífico colombiano; Zapata, L.A., Herrera, J.C., Eds.; Ministerio de Agricultura y Desarrollo Rural, Parques Nacionales Naturales, AUNAP, Universidad del Valle, HARMAR S.A., y WWF-Colombia: Cali, 2018; pp. 141–149. [Google Scholar]
- Hohn, A.A.; Chivers, S.J.; Barlow, J. REPRODUCTIVE MATURITY AND SEASONALITY OF MALE SPOTTED DOLPHINS, STENELLA ATTENUATA, IN THE EASTERN TROPICAL PACIFIC. Mar. Mammal Sci. 1985, 1, 273–293. [Google Scholar] [CrossRef]
- Hohn, A. A.; Hammond, P. S. Early postnatal growth of the spotted dolphin, Stenella attenuata, in the offshore eastern tropical Pacific. Fishery Bulletin 1985, 83(4), 553–566. [Google Scholar]
- Ibarra-Portillo, R.; Barraza, J.E.; Pineda, L. Registros de varamientos de cetáceos en El Salvador entre 1995-2019. Real. Y Reflex. 2020, 20, 210–231. [Google Scholar]
- Jefferson, T.A.; Webber, M.A.; Pitman, R.L. Marine Mammals of the World: A Comprehensive Guide to Their Identification, 2nd Edition ed; Elsevier: San Diego, CA, 2015. [Google Scholar]
- Justines, G. Métodos de monitoreo para estimar la mortalidad incidental de mamíferos marinos en el Pacífico de Panamá. Comisión Permanente del Pacífico Sur, Quito; (unpublished); 1997; p. 100pp. [Google Scholar]
- Kellar, N.M.; Trego, M.L.; Chivers, S.J.; Archer, F.I. Pregnancy patterns of pantropical spotted dolphins (Stenella attenuata) in the eastern tropical Pacific determined from hormonal analysis of blubber biopsies and correlations with the purse-seine tuna fishery. Mar. Biol. 2013, 160, 3113–3124. [Google Scholar] [CrossRef]
- Leslie, M. S.; Archer, F. I.; Morin, P. A. Mitogenome and nuclear DNA differentiation in spinner (Stenella longirostris) and pantropical spotted dolphins (S. attenuata) from the Eastern Tropical Pacific Ocean. bioRxiv 2016, 091215. [Google Scholar]
- Londoño, C. Ecología del delfín Tursiops truncatus y Stenella attenuata en el sector de Bahía Málaga, Pacífico colombiano; Instituto de Biología, Facultad de Ciencias Exactas y Naturales, Universidad de Antioquia: Medellín, Colombia, 2005. [Google Scholar]
- Lönnberg, E. Prodelphinus graffmani n.sp. a new dolphin from the Pacific coast of Mexico. Arkiv för Zoologi 1934, 26A(19), 1–11. [Google Scholar]
- Mangel, J.C.; Alfaro-Shigueto, J.; Van Waerebeek, K.; Cáceres, C.; Bearhop, S.; Witt, M.J.; Godley, B.J. Small cetacean captures in Peruvian artisanal fisheries: High despite protective legislation. Biol. Conserv. 2010, 143, 136–143. [Google Scholar] [CrossRef]
- Martínez, L.; Silva, S.; Alcorta, B.; Pacheco, A.S. First records of the pantropical spotted dolphin Stenella attenuata, within groups of common dolphins Delphinus delphis in northern Peru. Rev. De Biol. Mar. Y Oceanogr. 2021, 56, 78–82. [Google Scholar] [CrossRef]
- Myrick, A.C., Jr.; Hohn, A. A.; Barlow, J.; Sloan, P. A. Reproductive biology of female spotted dolphins, Stenella attenuata, from the Eastern Tropical Pacific. Fishery Bulletin 1986, 84(2), 247–259. [Google Scholar]
- NOAA. Climate Prediction Service. 2025. [Google Scholar]
- 10 December 2025. Available online: https://www.cpc.ncep.noaa.gov/products/analysis_monitoring/ensostuff/ONI_v5.php.
- Oremus, M.; Leqata, J.; Baker, C.S. Resumption of traditional drive hunting of dolphins in the Solomon Islands in 2013. R. Soc. Open Sci. 2015, 2, 140524. [Google Scholar] [CrossRef]
- Ortiz-Wolford, J.; Corona-Figueroa, M.F.; Dávila, V.; Cabrera, A.A. Cetacean stranding records along the Pacific coastline of Guatemala, 2007–2021: Implications for management, conservation and research. Mar. Policy 2021, 134. [Google Scholar] [CrossRef]
- Pacheco, A.S.; Silva, S.; Alcorta, B.; Gubbins, S.; Guidino, C.; Sanchez-Salazar, F.; Petit, A.; Llapapasca, M.A.; Balducci, N.; Larrañaga, E.; et al. Cetacean Diversity Revealed from Whale-Watching Observations in Northern Peru. Aquat. Mamm. 2019, 45, 116–122. [Google Scholar] [CrossRef]
- Palacios, D. M.; Salazar, S. Cetáceos. En: Reserva Marina de Galápagos. In Línea Base de la Biodiversidad; Danulat, E., Edgar, G.J., Eds.; Fundación Charles Darwin/Servicio Parque Nacional Galápagos: Santa Cruz, Galápagos, Ecuador, 2002; pp. pp 291–304. [Google Scholar]
- Palacios, D.M.; Herrera, J.C.; Gerrodette, T.; Garcia, C.; Soler, G.A.; Avila, I.C.; Bessudo, S.; Hernandez, E.; Trujillo, F.; Forez-Gonzalez, L.; et al. Cetacean distribution and relative abundance in Colombia’s Pacific EEZ from survey cruises and platforms of opportunity. J. Cetacean Res. Manag. 2012, 12, 45–60. [Google Scholar] [CrossRef]
- Perrin, W.F. Color pattern of the Eastern Pacific spotted porpoise Stenella graffmani Lönnberg (Cetacea, Delphinidae). Sci. Contrib. New York Zoöl. Soc. 1970, 54, 135–149. [Google Scholar] [CrossRef]
- Perrin, W.F. Variation of spotted and spinner dolphin (genus Stenella) in the eastern tropical Pacific and Hawaii. Bulletin Scripps Institute of Oceanography 1975, 21, 1–206. [Google Scholar]
- Perrin, W.F. Stenella attenuata. Mamm. Species 2001, 1–8. [Google Scholar] [CrossRef]
- Perrin, W.F.; Würsig, B.; Thewissen, J. Encyclopedia of Marine Mammals; Elsevier: Amsterdam, NX, Netherlands; ISBN, 2009. [Google Scholar]
- Perrin, W.F.; Mitchell, E.D.; Mead, J.G.; Caldwell, D.K.; Caldwell, M.C.; van Bree, P.J.H.; Dawbin, W.H. REVISION OF THE SPOTTED DOLPHINS, STENELLA SPP. Mar. Mammal Sci. 1987, 3, 99–170. [Google Scholar] [CrossRef]
- Perrin, W.F.; Hohn, A.A. Pantropical spotted dolphin. In Handbook of Marine Mammals; Ridgway, S.H., Harrison, R., Eds.; Academic Press: London, 1994; Volume 5, p. pp. 71-98 71-98. [Google Scholar]
- Perrin, W.F.; Schnell, G. D.; Hough, D. J.; Gilpatrick, J., Jr.; Kashiwada, J. V. Reexamination of geographic variation in canial morphology of the pantropical spotted dolphin, Stenella attenuata, in the eastern Pacific. Fishery Bulletin 1994, 92, 324–346. [Google Scholar]
- Pizarro-Neyra, J. Varamiento de cetáceos en Tacna, Perú (2002-2010). Revista peru Biología 2010, 17(2), 253–255. [Google Scholar] [CrossRef]
- Pizarro-Neyra, J.; Mamani-Fernández, J.; Vizcarra, J.K. Varamiento de Cetáceos en la costa sur del Perú (2016-2021). Cienc. Desarro. 2021, 20, 29–40. [Google Scholar] [CrossRef]
- Psarakos, S.; Herzing, D.L.; Marten, K. Mixed-species associations between Pantropical spotted dolphins (Stenella attenuata) and Hawaiian spinner dolphins (Stenella longirostris) off Oahu, Hawaii. Aquat. Mamm. 2003, 29, 390–395. [Google Scholar] [CrossRef]
- Rambaut, A.; Drummond, A.J.; Xie, D.; Baele, G.; Suchard, M.A. Posterior Summarization in Bayesian Phylogenetics Using Tracer 1.7. Syst. Biol. 2018, 67, 901–904. [Google Scholar] [CrossRef]
- Rambaut, A. FigTree v1.4.4, a graphical viewer of phylogenetic trees; Institute of Evolutionary Biology: University of Edinburgh, 2018. [Google Scholar]
- Ramírez, P.; Urquizo, W. Los cetáceos mayores y el fenómeno El Niño 1982-1983. In Vol. Extraordinario. El Niño. Su impacto en la Fauna Marina. Boletín IMARPE; Arntz, W., Landa, A., Tarazona, J., Eds.; Callao, Peru, 1985; pp. 201–206. [Google Scholar]
- Read, A.J.; Van Waerebeek, K.; Reyes, J.C.; McKinnon, J.S.; Lehman, L.C. The exploitation of small cetaceans in Coastal Peru. Biol. Conserv. 1988, 46, 53–70. [Google Scholar] [CrossRef]
- Reyes, J. C. Ballenas, delfines y otros cetáceos del Perú; Squema-Ediciones: Lima, 2009; p. 150pp. [Google Scholar]
- Reyes, J.C.; Van Waerebeek, K. Aspects of the Biology of Burmeister's porpoise from Peru. In Report of the International Whaling Commission (Special Issue 16); 1995; pp. 349–364. [Google Scholar]
- Rice, D. W. Marine Mammals of the World. In Systematics and Distribution; The Society for Marine Mammalogy, 1998; Volume Special Publication no. 4. [Google Scholar]
- Rodríguez-Fonseca, J.; Cubero-Pardo, P. Cetacean strandings in Costa Rica (1966-1999). 2001, 49, 667–72. [Google Scholar]
- Rodríguez-Rubio, E.; Schneider, W.; Abarca del Rio, R. On the seasonal circulation within the Panama Bight derived from satellite observations of wind, altimetry and sea surface temperature. Geophysical Research Letters 2003, 30(7), 1410. [Google Scholar] [CrossRef]
- Romero, A.; Ponce, G.; Hurtado, M.; Prado, J.; Armijos, G.; Vilela, K.; González, N.; Aguilar, F.; Muñoz, A.; Jurado, V. Estimación hidroacústica de los principales peces pelágicos pequeños en el Ecuador y su distribución geoespacial, durante diciembre de 2019 (INP 2019-12-02V). Instituto Nacional de Pesca, Ecuador. 2020. Available online: https://www.institutopesca.gob.ec/wp-content/uploads/2018/01/Estimaci%C3%B3n-Hidroac%C3%BAstica-de-los-principales-peces-pel%C3%A1gicos-peque%C3%B1os-en-el-Ecuador-y-su-distribuci%C3%B3n-geoespacial-durante-diciembre-de-2019.pdf.
- Ronquist, F.; Teslenko, M.; van der Mark, P.; Ayres, D.L.; Darling, A.; Höhna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian Phylogenetic Inference and Model Choice across a Large Model Space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef]
- Rosel, P.E.; Dizon, A.E.; Heyning, J.E. Genetic analysis of sympatric morphotypes of common dolphins (genus Delphinus). Mar. Biol. 1994, 119, 159–167. [Google Scholar] [CrossRef]
- Rosel, P.E.; France, S.C.; Wang, J.Y.; Kocher, T.D. Genetic structure of harbour porpoisePhocoena phocoenapopulations in the northwest Atlantic based on mitochondrial and nuclear markers. Mol. Ecol. 1999, 8, S41–54. [Google Scholar] [CrossRef] [PubMed]
- Rossi-Santos, M.R.; Santos-Neto, E.; Baracho, C.G. Interspecific cetacean interactions during the breeding season of humpback whale (Megaptera novaeangliae) on the north coast of Bahia State, Brazil. J. Mar. Biol. Assoc. United Kingd. 2009, 89, 961–966. [Google Scholar] [CrossRef]
- Rozas, J.; Ferrer-Mata, A.; Sánchez-DelBarrio, J.C.; Guirao-Rico, S.; Librado, P.; Ramos-Onsins, S.E.; Sánchez-Gracia, A. DnaSP 6: DNA Sequence Polymorphism Analysis of Large Data Sets. Mol. Biol. Evol. 2017, 34, 3299–3302. [Google Scholar] [CrossRef] [PubMed]
- Corrales, L.A.S. Cetáceos del sur del Perú. Cienc. Desarro. 2021, 20, 3–11. [Google Scholar] [CrossRef]
- Siciliano, S.; Higuera, D. Distribution and ecology of dolphins off the southern Chilean coast: implications for conservation. Marine Mammal Science 2015, 31(4), 1378–1391. [Google Scholar]
- Schweber, C.; Thomas, P. O. Cetacean distribution in Chilean coastal waters: a baseline for conservation efforts. Ocean & Coastal Management 2010, 53(12), 750–759. [Google Scholar]
- Sielfeld, W. Mamíferos marinos en colecciones y museos de Chile. Anales del Instituto de la Patagonía 1980, 11, 273–280. [Google Scholar]
- Sielfeld, W. Mamíferos marinos de Chile. Ediciones de la Universidad de Chile, Santiago; 1983. [Google Scholar]
- Silva-Buse, S. Gran diversidad. In (ed.) Fondo Editorial, Universidad Científica del Sur). Ballenas en el Norte del Perú; Industria Gráfica Cimagraf S.A.C.: Lima, Peru, 2016; pp. 138–171. [Google Scholar]
- Suárez, M. Aspectos ecológicos y del compotrtamiento de los delfines Tursiops truncatus (Montagu, 1821) y Stenella attenuata (Gray, 1846) en el Parque Nacional Utría, Chocó, Colombia. Tesis de grado en biología Universidad Nacional de Colombia, (unpublished). Santa Fe de Bogotá, 1994; p. 107 pp. [Google Scholar]
- Tajima, F. Statistical method for testing the neutral mutation hypothesis by DNA polymorphism. Genetics 1989, 123, 585–595. [Google Scholar] [CrossRef] [PubMed]
- Tamura, K.; Stecher, G.; Kumar, S. MEGA11: Molecular Evolutionary Genetics Analysis Version 11. Mol. Biol. Evol. 2021, 38, 3022–3027. [Google Scholar] [CrossRef]
- Tzika, A.C.; D’aMico, E.; Alfaro-Shigueto, J.; Mangel, J.C.; Van Waerebeek, K.; Milinkovitch, M.C. Molecular identification of small cetacean samples from Peruvian fish markets. Conserv. Genet. 2010, 11, 2207–2218. [Google Scholar] [CrossRef]
- Valencia, B. Presencia y comportamiento del delfín moteado pantropical (Stenella attenuata) en el Parque Nacional Natural Gorgona, Pacífico Colombiano. Tesis de grado, (unpublished). Universidad del Valle, Cali, Colombia, 2006. [Google Scholar]
- Van Bressem, M.-F.; Reyes, J.C.; Félix, F.; Echegaray, M.; Siciliano, S.; Di Beneditto, A.P.; Flach, L.; Viddi, F.; Avila, I.C.; Herrera, J.C.; et al. A preliminary overview of skin and skeletal diseases and traumata in small cetaceans from South American waters. Lat. Am. J. Aquat. Mamm. 2007, 6, 7–42. [Google Scholar] [CrossRef]
- Van Bressem, M.; Van Waerebeek, K.; Aznar, F.; Raga, J.; Jepson, P.; Duignan, P.; Deaville, R.; Flach, L.; Viddi, F.; Baker, J.; et al. Epidemiological pattern of tattoo skin disease: a potential general health indicator for cetaceans. Dis. Aquat. Org. 2009, 85, 225–237. [Google Scholar] [CrossRef]
- Van Waerebeek, K.; Reyes, J.C.; Luscombe, B.A. Revisión de la distribución de pequeños cetáceos frente al Perú. In Recursos y Dinámica del Ecosistema de Afloramiento Peruano. Boletín del Instituto del Mar del Perú, Callao; Salzwedel, H., Landa, A., Eds.; 1988; Volumen extraordinario, pp. 345–351. [Google Scholar]
- Van Waerebeek, K.; Reyes, J.C.; Read, A.J.; McKinnon. Preliminary observations of bottlenose dolphins from the Pacific coast of South America. The Bottlenose Dolphin. [CrossRef]
- Van Waerebeek, K.; Reyes, J.C.; Alfaro, J.O. Helminth parasites and phoronts of dusky dolphins Lagenorhynchus obscurus (Gray 1828) from Peru. Aquatic Mammals 1993, 19(30), 159–169. [Google Scholar]
- Van Waerebeek, K.; Reyes, J.C. Post-ban small cetacean takes off Peru: a review. Report of the International Whaling Commission (Special Issue 15) 1994, 503–520. [Google Scholar]
- Van Waerebeek, K.; Van Bressem, M. F.; Félix, F.; Alfaro, J.; García-Godos, A.; Chávez, L.; Ontón, K.; Montes, D.; Bello, R. Mortality of dolphins and porpoises in coastal fisheries off Peru and southern Ecuador in 1994. Biological Conservation 1997, 81, 43–49. [Google Scholar] [CrossRef]
- Van Waerebeek, K.; Félix, F.; Haase, B.; Palacios, D. M.; Mora-Pinto, D. M.; Muñoz-Hincapié, M. Inshore records of the striped dolphin, Stenella coeruleoalba, from the Pacific coast of South America. Report of the International Whaling Commission 1998, 48, 525–532. Available online: https://www.researchgate.net/publication/230630858.
- Van Waerebeek, K.; Reyes, J.C.; Sanino, G.; Félix, F.; Van Bressem, M-F.; Santillán, L.; Montes, D.; García-Godos, I.; Echegaray, M.; Venegas-Abad, A. Common bottlenose dolphins Tursiops truncatus of Pacific South America, a synoptic review of population identification data. Document SC/67A/SM/10, IWC Scientific Committee Meeting, Bled, Slovenia, May 2017; 2017. [Google Scholar]
- Van Waerebeek, K.; Apaza, M.; Reyes, J.C.; Alfaro-Shigueto, J.; Santillán, L.; Barreda, E.; Altamirano-Sierra, A.; Astohuaman-Uribe, J.; Ortiz-Alvarez, C.; Mangel, J. Beach cast small cetaceans bear evidence of continued catches and utilisation in coastal Peru. Document SC/67B/HIM/01 presented to IWC Scientific Committee Meeting, Bled, Slovenia, April 2018; 2018. [Google Scholar]
- Vidal, O.; Findley, L. T.; Leatherwood, S. Annotated checklist of the marine mammals of the Gulf of California. Proceedings of the San Diego Society of Natural History 1993, 28, 1–16. [Google Scholar]
- Walker, W.A. Geographical variation in morphology and biology of bottlenose dolphins (Tursiops) in the eastern north Pacific. In Southwest Fisheries Center (U.S.) Administrative report LJ-81-03C; 1981; p. 54 pp. [Google Scholar]
- Available online: https://repository.library.noaa.gov/view/noaa/22873.
- Wyrtki, K. Oceanography of the eastern Equatorial Pacific Ocean. Oceanography and Marine Biology Annual Review 1966, 4, 33–68. [Google Scholar]







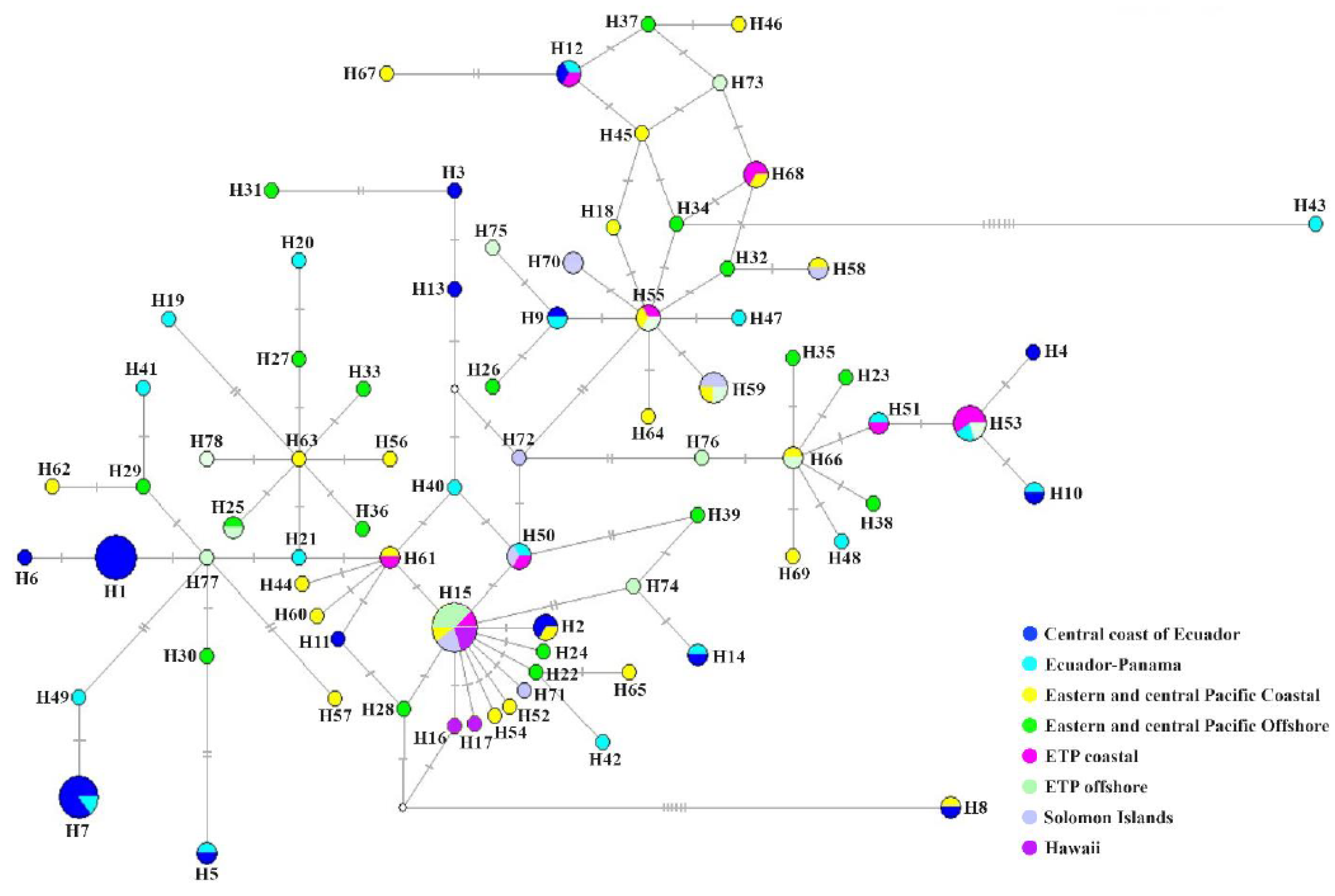
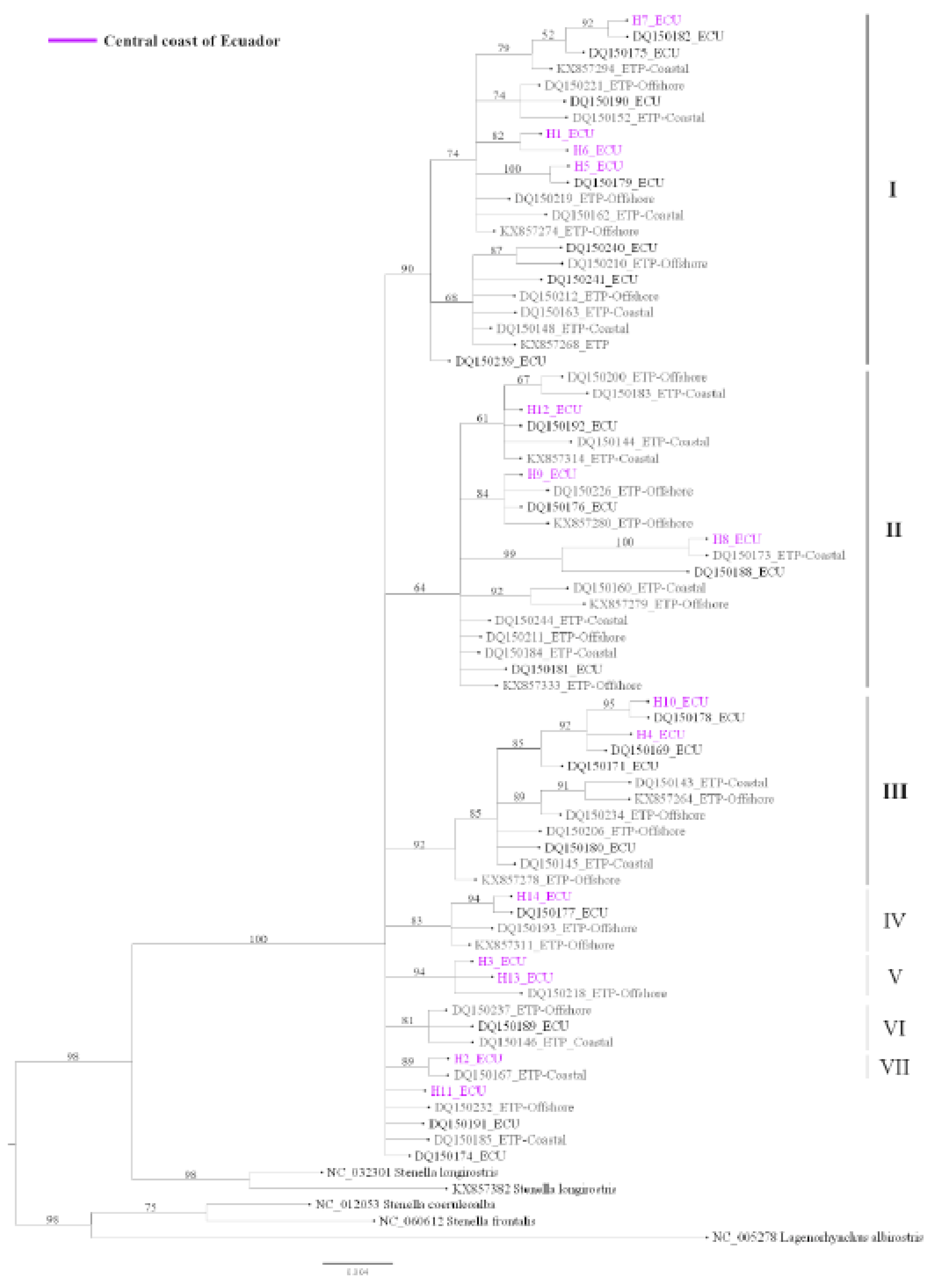

| Date | Site | Position | Sex | Length (cm) | Source |
|---|---|---|---|---|---|
| 5/10/1993 | San Vicente | 3º35'S. 80º24'W | 212 | Chiluiza et. al. (1998) | |
| 21/11/1993 | Anconcito | 2º22'S, 80º47'W | Chiluiza et. al. (1998) | ||
| 5/4/1994 | Montañita | 1º49'S 80º45'W | 204 | Chiluiza et. al. (1998) | |
| 30/4/1995 | Libertador Bolívar | 1º56'S, 80º44'W | Chiluiza et. al. (1998) | ||
| 12/7/1995 | Olón | 1º50'S, 80º34'W | 191 | Chiluiza et. al. (1998) | |
| 2/9/1995 | Playas | 2º37'S, 80º23'W | Chiluiza et. al. (1998) | ||
| 3/9/1995 | Monte Verde | 2º03'S, 80º44'W | Chiluiza et. al. (1998) | ||
| 25/5/2007 | Ayangue | 1º58'S, 80º45'W | M | » 200 | Félix et al. (2011) |
| 14/4/2006 | Mar Bravo | 2º15'S, 80º56'W | 190 | Félix et al. (2011) | |
| 23/12/2002 | San José | 1º46'S, 80º46'W | M | 193 | Félix et al. (2011) |
| 21/3/2004 | Mar Bravo | 2º13'S, 80º57'W | »120 | Félix et al. (2011) | |
| 14/9/2004 | Punta Carnero | 2º17'S, 80º54'W | Félix et al. (2011) | ||
| 19/9/2004 | Mar Bravo | 2º14'S, 80º56'W | F | 228 | Félix et al. (2011) |
| 11/11/2005 | Playa FAE | 2º11'S, 80º59'W | >200 | Félix et al. (2011) | |
| 16/2/2006 | Punta Carnero | 2º17'S, 80º54'W | M | 92 | Félix et al. (2011) |
| 18/11/2006 | Mar Bravo | 2º16'S, 80º55'W | M | 181 | Félix et al. (2011) |
| 2/12/2006 | Mar Bravo | 2º15'S, 80º55'W | F | 218 | Félix et al. (2011) |
| 13/5/2007 | Diablica | 2º18'S, 80º54'W | Félix et al. (2011) | ||
| 13/1/2008 | Mar Bravo | 2º14'S, 80º57'W | F | 186 | Félix et al. (2011) |
| 31/10/2009 | Punta Carnero | 2º17'S, 80º54'W | F | 179 | Félix et al. (2011) |
| 2/1/2010 | Mar Bravo | 2º15'S, 80º56W | 195 | This report | |
| 1/5/2015 | Diablica | 2º17'S, 80º54W | » 200 | This report | |
| 31/8/2017 | San Pablo | 2º06'S, 80º44W | F | 190 | This report |
| 27/6/2018 | Cauchiche | 2º48'S, 80º14'W | This report | ||
| 10/7/2021 | Mar Bravo | 2º14'S, 80º57'W | Chocho et al. (2023) |
| Date | Position | Group size | Remarks |
|---|---|---|---|
| 23/7/2000 | 1°18'S, 81°02'W | 6-10 | With humpback whales |
| 2/7/2002 | 2º09'S, 81º02'W | With humpback whales | |
| 1/8/2003 | 2º16'S, 81º06'W | 30-40 | Feeding |
| 23/9/2004 | 2º15'S, 81º04'W | ~100 | With humpback whales |
| 2/10/2004 | 2º06'S, 81º00'W | 100-200 | Several subgroups |
| 14/8/2005 | 2°08'S, 80°53'W | 60-80 | |
| 18/8/2005 | 2°11'S, 81°01'W | ~80 | |
| 21/8/2005 | 2°10'S, 80°58'W | ~60 | Feeding |
| 9/7/2007 | 2°08'S, 81°00´W | 3 | Feeding |
| 15/7/2008 | 2°10'S, 81°00´W | ~30 | |
| 11/8/2008 | 2°06'S, 81°03'W | With humpback whales | |
| 11/7/2010 | 2°09'S, 80°59’W | 50-100 | |
| 28/7/2010 | 80-100 | ||
| 29/7/2010 | 2°08'S, 81°01'W | ~50 | |
| 4/8/2010 | 2°11'S, 80°58'W | ~60 | |
| 5/8/2010 | ~40 | ||
| 12/8/2010 | 2°07.'S, 81°05'W | With humpback whales and common bottlenose dolphins | |
| 13/8/2010 | 2°12'S, 81°03'W | 300-400 | |
| 14/8/2010 | 60-80 | ||
| 23/8/2010 | 2°13'S, 81°03'W | 6-8 | With humpback whales |
| 19/9/2010 | 2°08'S, 81°00’W | 15-20 | |
| 2021-03-05 | 1°36'S, 81°00'W | 15-20 | |
| 2022-03-25 | 1°47'S, 80°51'W | ~100 | |
| 2022-12-20 | 1°26'S, 80°56'W | 4 | |
| 2023-01-22 | 1°23'S, 81°04'W | 18-20 | |
| 2023-02-25 | 1°27'S, 80°59'W | ~200 | Feeding |
| 7/3/2023 | 1°19'S, 80°58'W | ~200 | |
| 8/3/2023 | 1°24'S, 80°58'W | ~100 | Feeding |
| 26/2/2024 | 1°25'S, 81°07'W | ~500 | |
| 28/2/2024 | 1°25'S, 81°08'W | ~500 | |
| 14/3/2024 | 1°26'S, 80°59'W | ~200 | Feeding , with Bryde's whales |
| 14/3/2024 | 1°33'S, 81°11'W | ~500 | Feeding |
| 17/3/2024 | 1°19'S, 80°59'W | ~200 | Feeding |
| 3/4/2024 | 1°24'S, 81°11'W | ~50 | |
| 11/4/2024 | 1°21'S, 80°57'W | ~20 | |
| 11/4/2024 | 1°16'S, 81°04'W | ~50 | |
| 18/4/2024 | 1°25'S, 80°57'W | ~200 | |
| 18/4/2024 | 1°35'S, 80°53'W | 5 | With Bryde's whales |
| 1/5/2024 | 1°25'S, 80°56'W | ~300 | |
| 2/5/2024 | 1°25'S, 80°55'W | ~300 | Feeding |
| 4/6/2024 | 1°26'S, 80°54'W | 10 | With Bryde's whales |
| 27/7/2024 | 1°33'S, 80°56'W | ~300 | With humpback whales |
| 15/8/2024 | 1°19'S, 81°00'W | ~20 | With humpback whales |
| 17/8/2024 | 1°24'S, 80°57'W | ~20 | |
| 19/8/2024 | 1°21'S, 80°59'W | ~200 | |
| 24/8/2024 | 1°16'S, 81°02'W | ~200 | |
| 26/8/2024 | 1°25'S, 80°56'W | ~20 | With Bryde's whales |
| 3/6/2024 | 1°35'S, 80°52'W | ~200 | Feeding |
| 8/9/2024 | 1°25'S, 80°56'W | ~100 | |
| 11/9/2024 | 1°24'S, 80°54'W | ~200 | |
| 20/9/2024 | 1°26'S, 80°54'W | ~20 | |
| 20/9/2024 | 1°26'S, 80°55'W | ~20 | |
| 21/9/2024 | 1°23'S, 80°58'W | ~200 | |
| 21/9/2024 | 1°24'S, 80°57'W | ~200 | Feeding |
| 22/9/2024 | 1°25'S, 80°56'W | ~100 | Feeding |
| 1/10/2024 | 1°28'S, 80°52'W | ~200 | With humpback whales |
| 19/10/2024 | 1°17'S, 81°1'W | 10 | |
| 26/10/2024 | 1°24'S, 80°55'W | 3 | |
| 28/10/2024 | 1°26'S, 80°54'W | 5 |
| Specimen | Date | Location | Coord. | Sex, maturity | SL (cm) | Circumstances | Voucher, Museo de Delfines, CEPEC |
|---|---|---|---|---|---|---|---|
| KVW-343 | 20/04/1985 | Landed at Cerro Azul port, Lima Department | 13°02'S, 76°29'W | Adult female (lactating) | 211 | Gillnet bycatch | skull |
| KVW-344 | 20/04/1985 | Landed at Cerro Azul port, Lima Dept. | 13°02'S, 76°29'W | Female. Small calf. Milk in stomach. | 106 | Gillnet bycatch, landed with KVW-343 | skull |
| AJR-036 | 24/02/1986 | Taken at Punta Negra, landed at Pucusana port, Lima Dept. | 12°22'S, 76°48'W | Male. Sexually immature; cranially subadult | 188 | Harpooned; collected by A.J. Read | skull |
| KVW-1999 | 28/12/1989 | Playa Michallo, Bahía Sechura (sur), Piura Dept. | 05°35'S, 80°57'W | Sex unknown; cranially mature | NA | Beach pick-up (Santiago Zambrano) | skull |
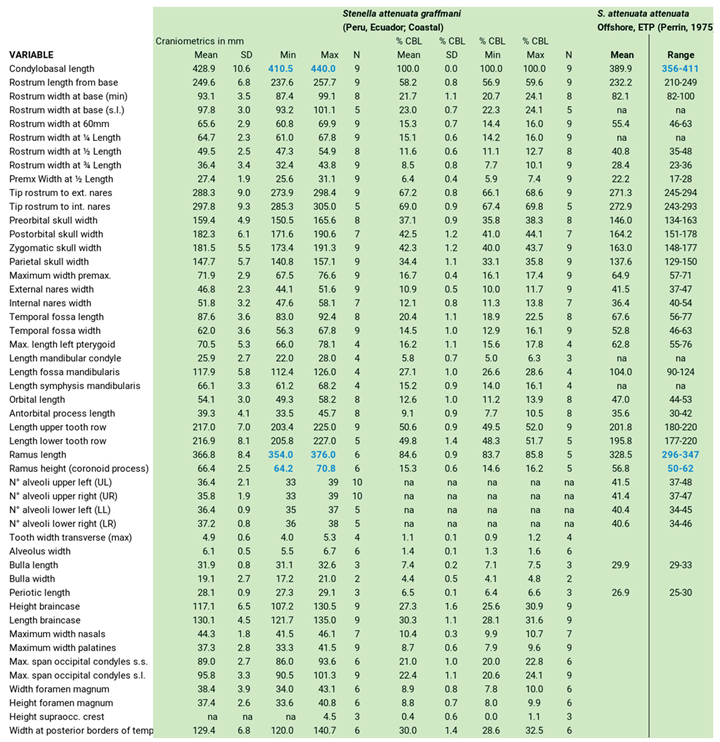 |
| Haplotype | Polymorphic sites | Haplotype frequency | |||||||||||||||||||||||||
| 1 | 2 | 5 | 7 | 8 | 1 | 1 | 1 | 1 | 1 | 1 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 3 | 3 | 3 | 3 | 3 | 3 | ||
| 6 | 6 | 1 | 9 | 8 | 0 | 3 | 3 | 4 | 6 | 7 | 2 | 3 | 4 | 4 | 5 | 5 | 6 | 6 | 7 | 1 | 5 | 5 | 7 | 7 | 9 | ||
| 4 | 4 | 6 | 9 | 5 | 5 | 7 | 0 | 1 | 7 | 1 | 9 | 4 | 5 | 2 | 1 | 2 | 5 | 2 | 3 | 3 | |||||||
| Hap_R | C | T | T | T | G | T | G | A | T | T | A | T | A | T | A | C | T | T | T | A | C | C | T | A | C | A | |
| Hap_1 | . | . | . | . | A | . | . | . | . | . | . | . | . | . | . | . | . | . | C | . | . | . | C | . | . | G | 8 |
| Hap_2 | . | . | . | . | . | . | . | . | . | . | . | . | . | . | . | . | . | . | C | . | T | . | . | . | T | . | 2 |
| Hap_3 | . | . | C | . | . | . | . | . | . | . | . | . | . | . | . | . | . | C | C | . | . | . | . | . | . | . | 1 |
| Hap_4 | . | . | . | . | . | C | . | . | . | . | . | C | . | . | . | T | . | . | C | . | T | T | . | G | T | . | 1 |
| Hap_5 | . | . | . | . | A | . | . | . | . | . | . | . | . | . | . | T | . | . | C | . | . | T | . | . | T | G | 1 |
| Hap_6 | . | . | . | . | A | . | . | C | . | . | . | . | . | . | . | . | . | . | C | . | . | . | C | . | . | G | 1 |
| Hap_7 | . | . | . | . | A | . | . | . | . | . | . | . | . | C | . | . | . | . | . | . | . | T | . | . | . | G | 6 |
| Hap_8 | T | C | . | . | . | . | A | . | . | . | . | . | G | . | T | . | C | . | . | G | . | . | . | . | T | . | 1 |
| Hap_9 | . | C | . | . | . | . | A | . | . | . | G | . | . | . | . | . | . | C | C | . | . | T | . | . | T | . | 1 |
| Hap_10 | . | . | . | . | . | C | . | . | C | . | . | C | . | . | . | T | . | C | C | . | T | T | . | G | T | . | 1 |
| Hap_11 | . | . | . | . | . | . | . | . | . | . | . | . | G | . | . | . | . | . | C | . | . | . | . | . | . | . | 1 |
| Hap_12 | . | C | . | . | . | . | A | . | . | C | . | C | G | . | . | . | . | C | C | . | . | T | . | . | T | . | 1 |
| Hap_13 | . | . | C | . | . | . | . | . | . | . | . | . | . | . | . | . | . | C | C | . | . | T | . | . | . | . | 1 |
| Hap_14 | . | . | . | C | . | . | . | . | . | . | . | . | . | . | . | . | . | . | . | G | . | . | . | . | T | . | 1 |
| 1 | Published in Arkiv för Zoologi which has ceased to exist. It was a Swedish scientific journal on zoology published between 1903 and 1974. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





