Submitted:
04 February 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
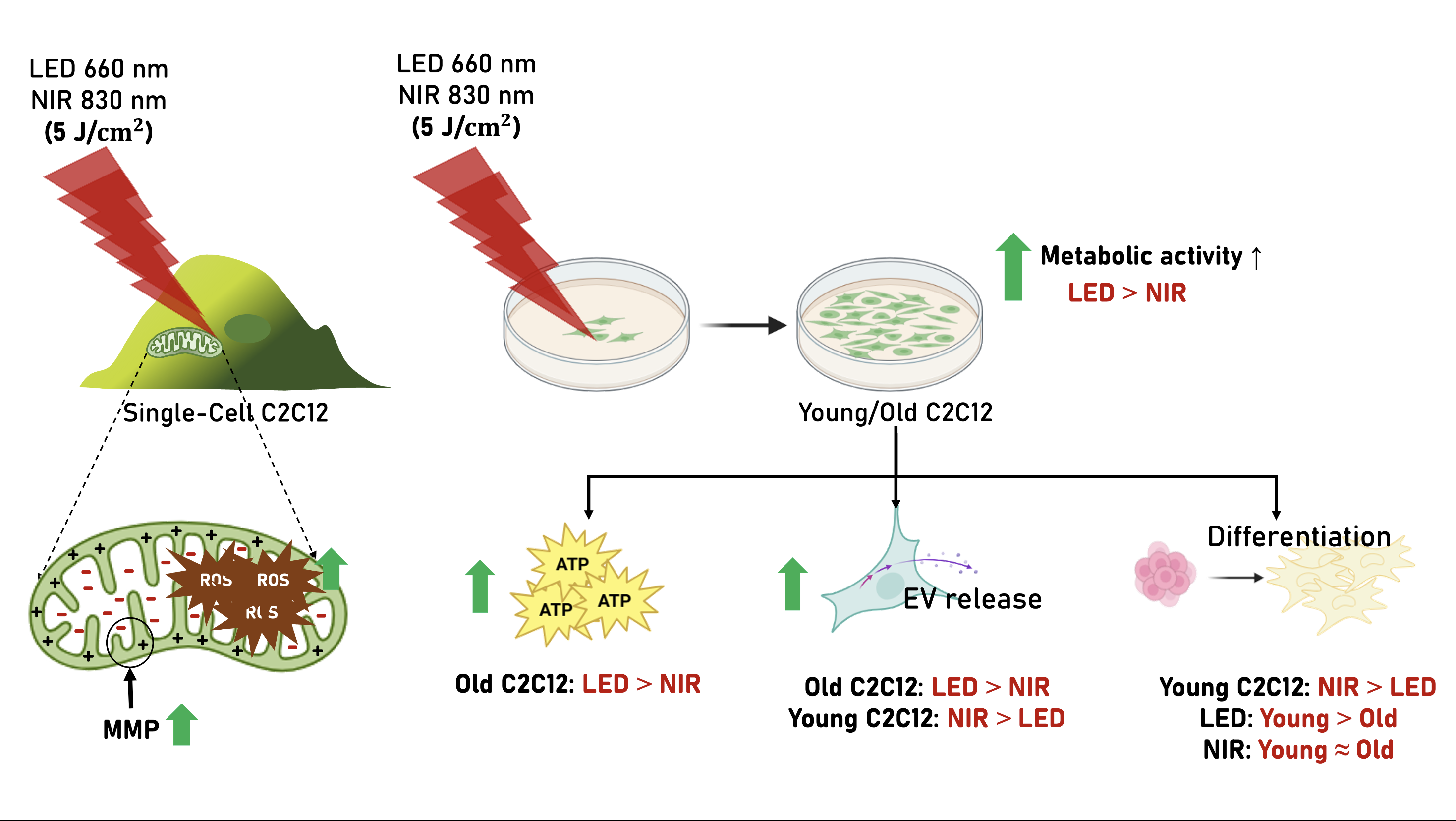
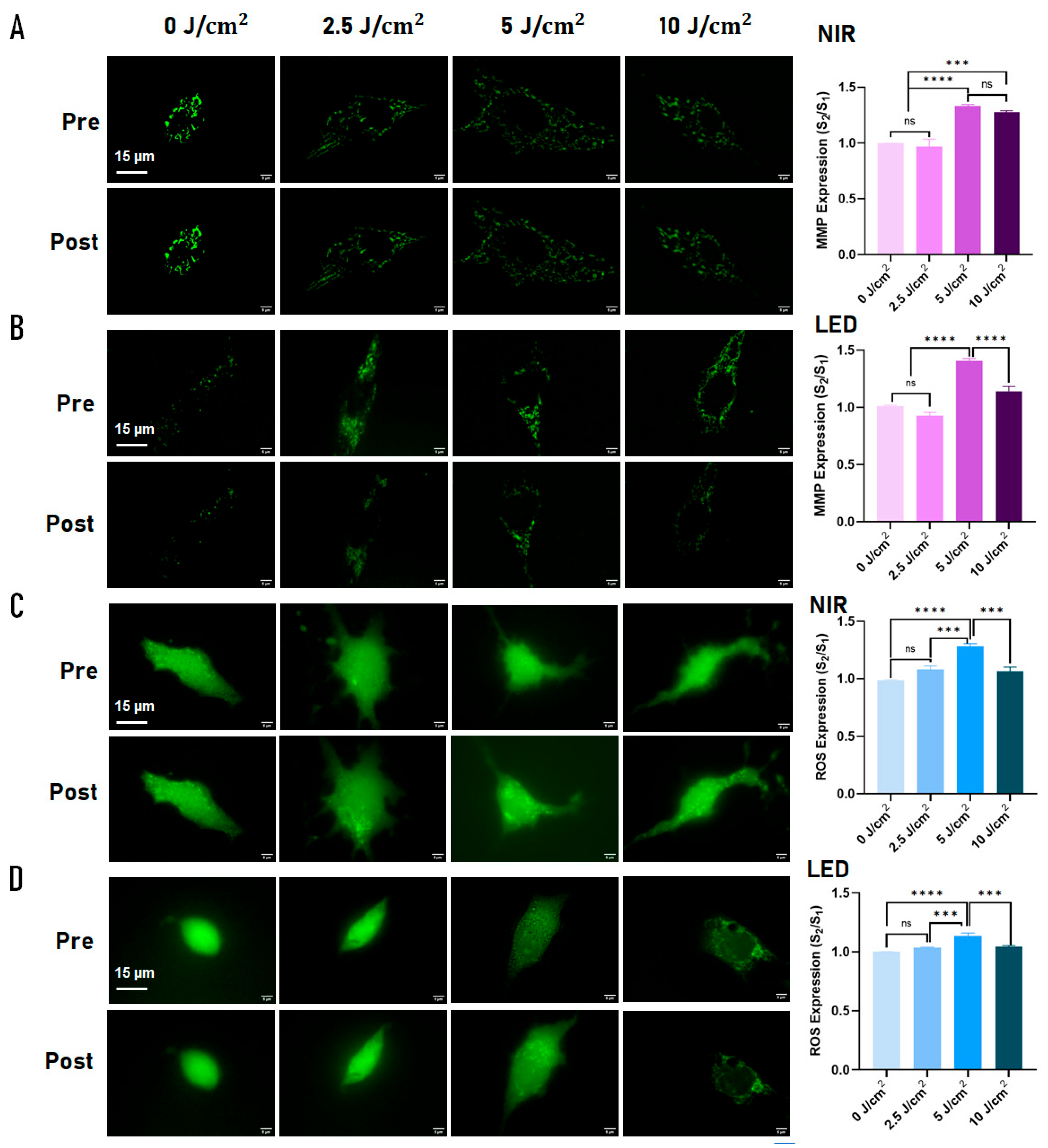
2.1. Effects of PBM on Mitochondrial Membrane Potential and ROS at the Single-Cell Level
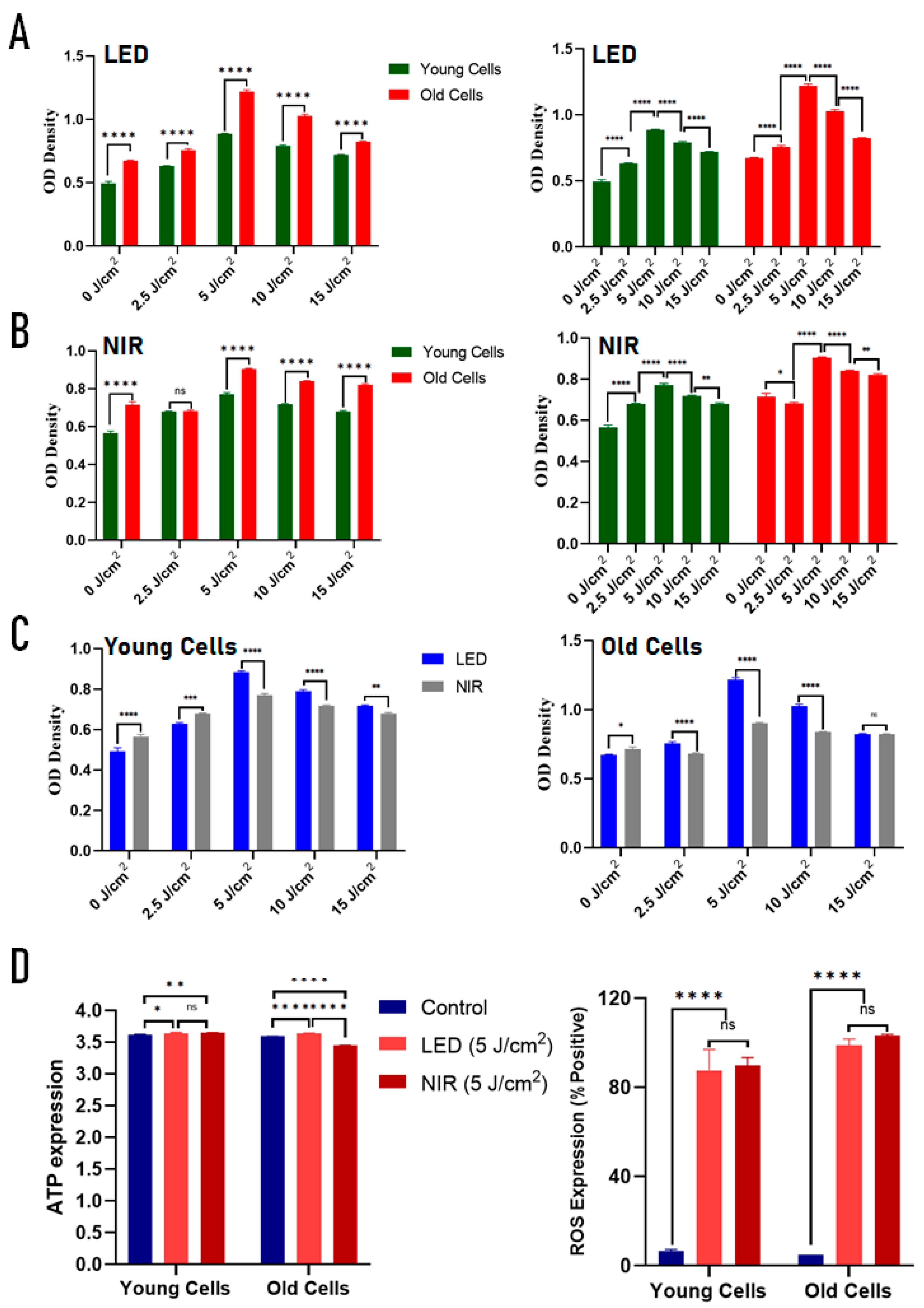
2.2. Effects of PBM on Metabolic Activity, ATP Production, and ROS Signaling in Young and Replicatively Aged C2C12 Cells
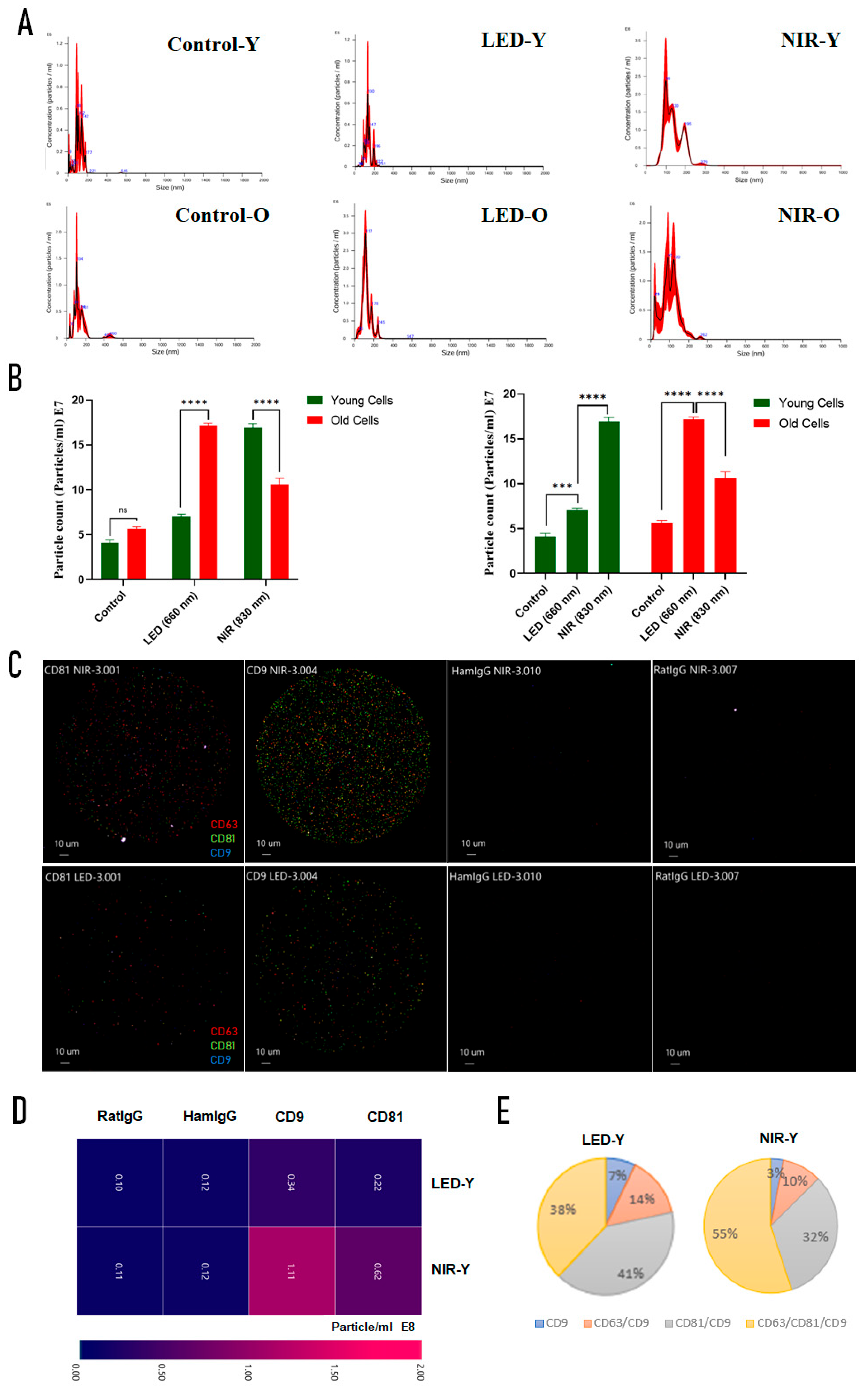
2.3. Extracellular Vesicle Release and Characterization Following PBM
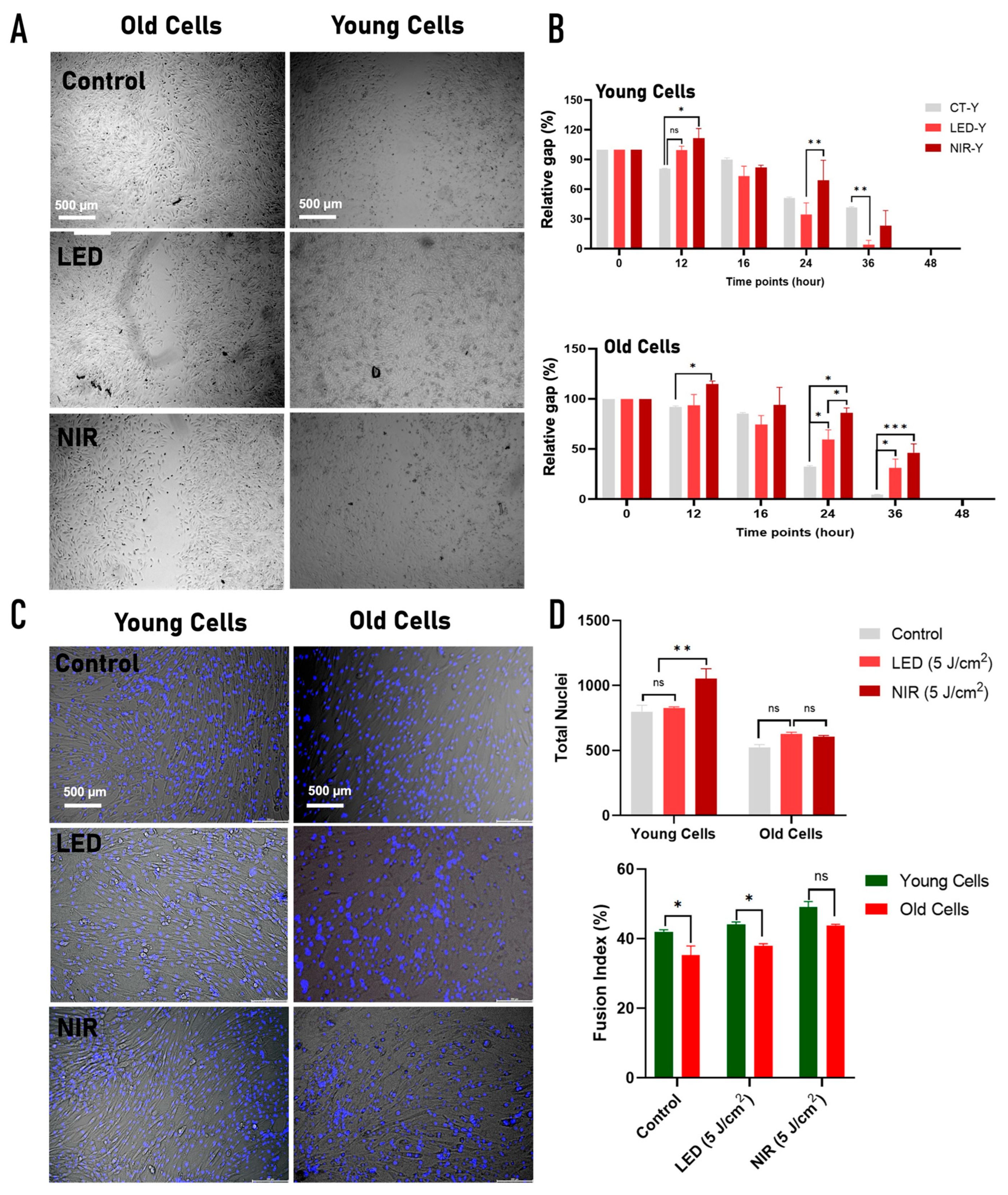
2.4. Effects of PBM on Migration and Differentiation of Young and Old C2C12 Cells
2.5. Integrated Summary of PBM Responses Across Cellular Outputs
3. Discussion
3.1. Dose-Dependent Mitochondrial Responses to PBM
3.2. Age-Dependent Differences in Mitochondrial and Redox Responses
3.3. Functional Outcomes: Migration and Differentiation
3.4. PBM-Induced Extracellular Vesicle Release
3.5. Wavelength and Source Considerations
3.6. Translational Relevance for Age-Related Progressive Muscle Conditions
4. Materials and Methods
4.1. Cell Culture
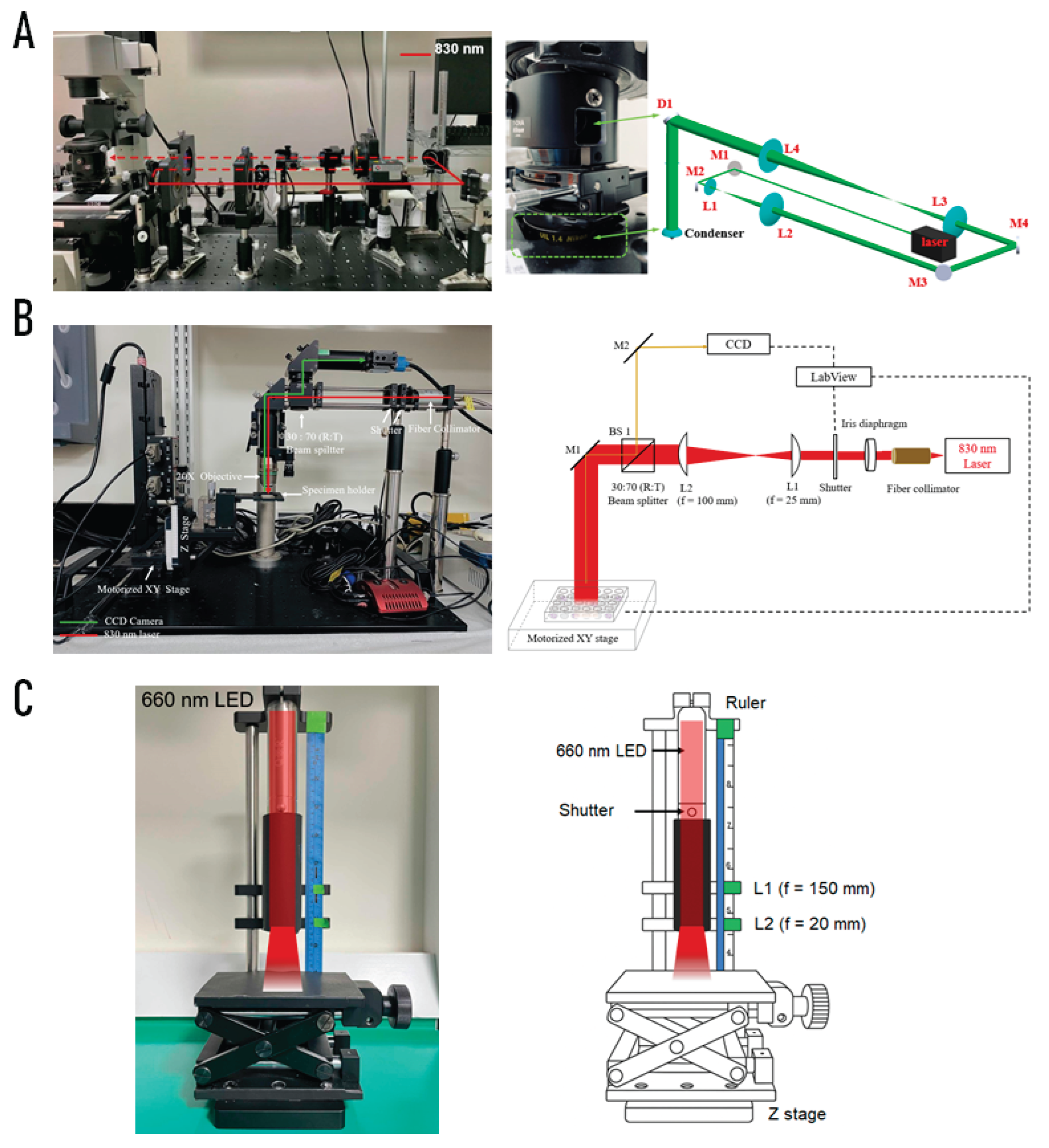
4.2. Photobiomodulation (PBM) Irradiation Systems
4.2.1. PBM Dose Selection and Experimental Design
- Cell viability screening (96-well format). To more fully characterize biphasic dose–response behavior, bulk viability assays were performed using 2.5, 5.0, 10.0, and 15.0 J/cm² for both LED and NIR platforms.
-
Mechanistic and functional assays (96- and 24-well formats).Based on concordant findings from single-cell mitochondrial assays and bulk viability screening, 5.0 J/cm² was identified as the most consistently effective fluence across both light sources. This dose was therefore selected for all subsequent functional studies, including ATP production (96-well), ROS signaling, migration, differentiation, and extracellular vesicle (EV) release (24-well format).
Dosimetry and Irradiation Control
4.2.1. Single-Cell NIR Laser System (830 nm)
4.2.2. Bulk-Cell NIR Laser System (830 nm)
4.2.3. Customized LED System (660 nm)
4.3. Single-Cell Assays of Mitochondrial Function and ROS
4.3.1. Mitochondrial Membrane Potential (ΔΨm)
4.3.2. Intracellular ROS
4.4. Population Level Experiments
4.4.1. Cell Viability
4.4.2. Bulk ROS Measurement
4.4.3. Bulk ATP Measurement
4.4.4. Migration Assay
Wound Closure Quantification
- Relative gap (%):
- Relative closure rate (%/h):
- Interval-specific closure rate (%/h):
4.4.5. Differentiation Assay
4.4.5. Extracellular Vesicle Isolation and Characterization
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of Variance |
| ATP | Adenosine Triphosphate |
| CCK-8 | Cell Counting Kit-8 |
| CcO | Cytochrome c Oxidase |
| CO₂ | Carbon Dioxide |
| DMEM | Dulbecco’s Modified Eagle Medium |
| DMSO | Dimethyl Sulfoxide |
| ΔΨm | Mitochondrial Membrane Potential |
| EMCCD | Electron-Multiplying Charge-Coupled Device |
| ESCRT | Endosomal Sorting Complex Required for Transport |
| EV | Extracellular Vesicle |
| FACS | Fluorescence-Activated Cell Sorting |
| FBS | Fetal Bovine Serum |
| FI | Fusion Index |
| LED | Light-Emitting Diode |
| MAPK | Mitogen-Activated Protein Kinase |
| MMP | Mitochondrial Membrane Potential |
| NIR | Near-Infrared |
| NTA | Nanoparticle Tracking Analysis |
| PBM | Photobiomodulation |
| Rh123 | Rhodamine 123 |
| ROS | Reactive Oxygen Species |
| SEM | Standard Error of the Mean |
References
- Anders, J.J.; Lanzafame, R.J.; Arany, P.R. Low-level light/laser therapy versus photobiomodulation therapy. Photomed. Laser Surg. 2015, 33, 183–184. [Google Scholar] [CrossRef]
- Hamblin, M.R. Mechanisms and mitochondrial redox signaling in photobiomodulation. Photochem. Photobiol. 2018, 94, 199–212. [Google Scholar] [CrossRef]
- Karu, T.I. Primary and secondary mechanisms of action of visible to near-IR radiation on cells. J. Photochem. Photobiol. B 1999, 49, 1–17. [Google Scholar] [CrossRef]
- Karu, T.I.; Kolyakov, S.F. Exact action spectra for cellular responses relevant to phototherapy. Photomed. Laser Surg. 2005, 23, 355–361. [Google Scholar] [CrossRef]
- Sarti, P.; Forte, E.; Mastronicola, D.; Giuffrè, A.; Arese, M. Cytochrome c oxidase and nitric oxide in action: Molecular mechanisms and pathophysiological implications. Biochim. Biophys. Acta Bioenerg. 2012, 1817, 610–619. [Google Scholar] [CrossRef]
- Mochizuki-Oda, N.; Kataoka, Y.; Cui, Y.; Yamada, H.; Heya, M. Effects of near-infrared laser irradiation on adenosine triphosphate and adenosine diphosphate contents of rat brain tissue. Neurosci. Lett. 2002, 323, 207–209. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Jones, D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef]
- Zorov, D.B.; Juhaszova, M.; Sollott, S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol. Rev. 2014, 94, 909–950. [Google Scholar] [CrossRef]
- Turrens, J.F. Mitochondrial formation of reactive oxygen species. J. Physiol. 2003, 552, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009, 417, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Divakaruni, A.S.; Brand, M.D. The regulation and physiology of mitochondrial proton leak. Physiol. Rev. 2011, 91, 793–829. [Google Scholar] [CrossRef]
- Korshunov, S.S.; Skulachev, V.P.; Starkov, A.A. High protonic potential actuates a mechanism of production of reactive oxygen species in mitochondria. FEBS Lett. 1997, 416, 15–18. [Google Scholar] [CrossRef]
- Ferraresi, C.; Hamblin, M.R.; Parizotto, N.A. Low-level laser (light) therapy on muscle tissue: Performance, fatigue and repair benefited by the power of light. Photonics Lasers Med. 2012, 5, 247–261. [Google Scholar] [CrossRef]
- Leal-Junior, E.C.P.; Vanin, A.A.; Miranda, E.F.; de Carvalho, P.D.T.C.; Dal Corso, S. Effect of phototherapy (low-level laser therapy and light-emitting diode therapy) on exercise performance and markers of exercise recovery: A systematic review with meta-analysis. Lasers Med. Sci. 2015, 30, 925–939. [Google Scholar] [CrossRef]
- Huang, Y.Y.; Sharma, S.K.; Carroll, J.; Hamblin, M.R. Biphasic dose response in low level light therapy - an update. Dose Response 2011, 9, 602–618. [Google Scholar] [CrossRef]
- Silveira, P.C.L.; Streck, E.L.; Pinho, R.A. Evaluation of mitochondrial respiratory chain activity in muscle healing by low-level laser therapy. J. Photochem. Photobiol. B 2009, 95, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Gholami, L.; Khorsandi, K.; Nooshabadi, V.T.; Shahabi, S.; Jazaeri, M.; Esfahani, H.; Faradonbeh, D.R.; Malekshahi, Z.V.; Afsartala, Z.; Mostafa, N. Effect of photobiomodulation on structure and function of extracellular vesicle secreted from mesenchymal stem cells. Photochem. Photobiol. 2022, 98, 1447–1458. [Google Scholar] [CrossRef]
- Lovisetto, R.; Malavazzi, T.C.S.; Andreo, L.; Rodrigues, M.F.S.D.; Bussadori, S.K.; Fernandes, K.P.S.; Mesquita-Ferrari, R.A. Photobiomodulation using different infrared light sources promotes muscle precursor cells migration and proliferation. Photonics 2022, 9, 469. [Google Scholar] [CrossRef]
- Vechetti, I.J., Jr.; Valentino, T.; Mobley, C.B.; McCarthy, J.J. The role of extracellular vesicles in skeletal muscle and systemic adaptation to exercise. J. Physiol. 2021, 599, 845–861. [Google Scholar] [CrossRef]
- da Silva, E.M.; Mesquita-Ferrari, R.A.; Rodrigues, M.F.S.D.; Magalhães, E.M.R.; Bussadori, S.K.; de Brito, A.; Messias, F.D.M.; Souza, N.H.C.; Alves, A.N.; Fernandes, K.P.S. The effects of photobiomodulation on inflammatory infiltrate during muscle repair in advanced-age rats. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 437–441. [Google Scholar] [CrossRef] [PubMed]
- Coen, P.M.; Musci, R.V.; Hinkley, J.M.; Miller, B.F. Mitochondria as a target for mitigating sarcopenia. Front. Physiol. 2019, 9, 1883. [Google Scholar] [CrossRef] [PubMed]
- Short, K.R.; Bigelow, M.L.; Kahl, J.; Singh, R.; Coenen-Schimke, J.; Raghavakaimal, S.; Nair, K.S. Decline in skeletal muscle mitochondrial function with aging in humans. Proc. Natl. Acad. Sci. USA 2005, 102, 5618–5623. [Google Scholar] [CrossRef]
- Rygiel, K.A.; Picard, M.; Turnbull, D.M. The ageing neuromuscular system and sarcopenia: A mitochondrial perspective. J. Physiol. 2016, 594, 4499–4512. [Google Scholar] [CrossRef]
- Chen, L.K.; Woo, J.; Assantachai, P.; et al. Asian Working Group for Sarcopenia: 2019 consensus update on sarcopenia diagnosis and treatment. J. Am. Med. Dir. Assoc. 2020, 21, 300–307.e2. [Google Scholar] [CrossRef] [PubMed]
- Girasol, C.E.; Moraes, J.M.A.F.; Bachmann, L.; Alfredo, D.M.N.; Barbosa, R.I.; Guirro, E.C.O.; Guirro, R.R.J. In vivo attenuation profile of 660 nm and 830 nm wavelengths on human elbow skin and calcaneus tendon of different phototypes. Lasers Med. Sci. 2024, 39, 24. [Google Scholar] [CrossRef] [PubMed]
- de Lima, E.V.; Pacheco-Soares, C.; da Silva, N.S. Photobiomodulation assay of muscle cells C2C12 after irradiation with LED device. Res. Soc. Dev. 2022, 11, e41511628884. [Google Scholar] [CrossRef]
- Oyebode, O.A.; Houreld, N.N. A comparison between photobiomodulation at 830 nm and 660 nm on differentiation in diabetic human skin fibroblast cells. In Proceedings of the SAIP2021 Conference, South Africa, 2021; pp. 1–6. https://saip.org.za/Proceedings/Track%20C/41.pdf.
- Desmet, K.D.; Paz, D.A.; Corry, J.J.; et al. Clinical and experimental applications of NIR-LED photobiomodulation. Photomed. Laser Surg. 2006, 24, 121–128. [Google Scholar] [CrossRef]
- Ferro, A.P.; de Jesus Guirro, R.R.; Orellana, M.D.; De Santis, G.C.; Farina, J.A., Jr.; de Oliveira Guirro, E.C. Photobiomodulation with laser and LED on mesenchymal stem cells viability and wound closure in vitro. Lasers Med. Sci. 2024, 39, 205. [Google Scholar] [CrossRef]
- Heiskanen, V.; Hamblin, M.R. Photobiomodulation: Lasers vs. light-emitting diodes? Photochem. Photobiol. Sci. 2018, 17, 1003–1017. [Google Scholar] [CrossRef]
- Yaffe, D.; Saxel, O. Serial passaging and differentiation of myogenic cells isolated from dystrophic mouse muscle. Nature 1977, 270, 725–727. [Google Scholar] [CrossRef]
- Burattini, S.; Ferri, P.; Battistelli, M.; Curci, R.; Luchetti, F.; Falcieri, E. C2C12 murine myoblasts as a model of skeletal muscle development: Morpho-functional characterization. Eur. J. Histochem. 2004, 48, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Potes, Y.; Bermejo-Millo, J.C.; Mendes, C.; Castelão-Baptista, J.P.; Díaz-Luis, A.; Pérez-Martínez, Z.; Solano, J.J.; Sardão, V.A.; Oliveira, P.J.; Caballero, B.; Coto-Montes, A. p66Shc signaling and autophagy impact on C2C12 myoblast differentiation during senescence. Cell Death Dis. 2024, 15, 200. [Google Scholar] [CrossRef]
- Rahman, F.A.; Hian-Cheong, D.J.; Boonstra, K.; Ma, A.; Thoms, J.P.; Zago, A.S.; Quadrilatero, J. Augmented mitochondrial apoptotic signaling impairs C2C12 myoblast differentiation following cellular aging through sequential passaging. J. Cell. Physiol. 2024, 239, e31155. [Google Scholar] [CrossRef]
- Pan, L.C.; Hang, N.L.T.; Colley, M.M.S.; Chang, J.; Hsiao, Y.C.; Lu, L.S.; Li, B.S.; Chang, C.J.; Yang, T.S. Single-cell effects of photobiomodulation on mitochondrial membrane potential and reactive oxygen species production in human adipose mesenchymal stem cells. Cells 2022, 11, 972. [Google Scholar] [CrossRef]
- Baldassarro, V.A.; Alastra, G.; Lorenzini, L.; Giardino, L.; Calzà, L. Photobiomodulation at defined wavelengths regulates mitochondrial membrane potential and redox balance in skin fibroblasts. Oxid. Med. Cell. Longev. 2023, 2023, 7638223. [Google Scholar] [CrossRef]
- Ristow, M.; Schmeisser, K. Mitohormesis: Promoting health and lifespan by increased levels of reactive oxygen species (ROS). Dose Response 2014, 12, 288–341. [Google Scholar] [CrossRef]
- Chang, C.Y.; Aviña, A.E.; Chang, C.J.; Lu, L.S.; Chong, Y.Y.; Ho, T.Y.; Yang, T.S. Exploring the biphasic dose–response effects of photobiomodulation on the viability, migration, and extracellular vesicle secretion of human adipose mesenchymal stem cells. J. Photochem. Photobiol. B 2024, 256, 112940. [Google Scholar] [CrossRef]
- Zorova, L.D.; Popkov, V.A.; Plotnikov, E.Y.; et al. Mitochondrial membrane potential. Cell Death Dis. 2018, 9, 950. [Google Scholar] [CrossRef]
- Suski, J.M.; Lebiedzinska, M.; Bonora, M.; et al. Relation between mitochondrial membrane potential and ROS formation. Methods Mol. Biol. 2012, 810, 183–205. [Google Scholar] [CrossRef] [PubMed]
- Scaduto, R.C.; Grotyohann, L.W. Measurement of mitochondrial membrane potential using fluorescent rhodamine derivatives. Biophys. J. 1999, 76, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.D.; Stewart, C.E.; Burniston, J.G. Degradation of ribosomal and chaperone proteins is attenuated during the differentiation of replicatively aged C2C12 myoblasts. J. Cachexia Sarcopenia Muscle 2022, 13, 2562–2575. [Google Scholar] [CrossRef] [PubMed]
- Shintani-Ishida, K.; Tsurumi, R.; Ikegaya, H. Decrease in the expression of muscle-specific miRNAs, miR-133a and miR-1, in myoblasts with replicative senescence. PLoS ONE 2023, 18, e0280527. [Google Scholar] [CrossRef]
- Hawke, T.J.; Garry, D.J. Myogenic satellite cells: Physiology to molecular biology. J. Appl. Physiol. 2001, 91, 534–551. [Google Scholar] [CrossRef] [PubMed]
- Barbieri, E.; Sestili, P. Reactive oxygen species in skeletal muscle signaling. J. Signal Transduct. 2012, 2012, 982794. [Google Scholar] [CrossRef] [PubMed]
- Lian, D.; Chen, M.M.; Wu, H.; Deng, S.; Hu, X. The role of oxidative stress in skeletal muscle myogenesis and muscle disease. Antioxidants 2022, 11, 755. [Google Scholar] [CrossRef]
- Hang, N.L.T.; Chuang, A.E.Y.; Chang, C.J.; Yen, Y.; Wong, C.C.; Yang, T.S. Photobiomodulation associated with alginate-based engineered tissue on promoting chondrocytes-derived biological responses for cartilage regeneration. Int. J. Biol. Macromol. 2024, 280, 135982. [Google Scholar] [CrossRef]
- Murata, A.; Yamaguchi, Y.; Ueda, Y.; et al. Electrical pulse stimulation-induced tetanic exercise simulation increases the secretion of extracellular vesicles from C2C12 myotubes. Biochem. Biophys. Res. Commun. 2023, 672, 177–184. [Google Scholar] [CrossRef]
- de Freitas, L.F.; Hamblin, M.R. Proposed mechanisms of photobiomodulation or low-level light therapy. IEEE J. Sel. Top. Quantum Electron. 2016, 22, 348–364. [Google Scholar] [CrossRef] [PubMed]
- Silveira, P.C.L.; Ferreira, G.K.; Zaccaron, R.P.; Remor, A.P.; da Silva, L.A.; Pinho, R.A. Effects of photobiomodulation on mitochondria of brain, muscle, and C6 astroglioma cells. Med. Eng. Phys. 2019, 71, 108–113. [Google Scholar] [CrossRef]
- Frangini, M.; Franzolin, E.; Chemello, F.; Laveder, P.; Romualdi, C.; Bianchi, V.; Rampazzo, C. Synthesis of mitochondrial DNA precursors during myogenesis: An analysis in purified C2C12 myotubes. J. Biol. Chem. 2013, 288, 5624–5635. [Google Scholar] [CrossRef]
- Parisi, B.; Sünnen, M.; Chippalkatti, R.; Abankwa, D.K. A flow-cytometry-based pipeline for the rapid quantification of C2C12 cell differentiation. STAR Protoc. 2023, 4, 102637. [Google Scholar] [CrossRef]
- Tannu, N.S.; Rao, V.K.; Chaudhary, R.M.; Giorgianni, F.; Saeed, A.E.; Gao, Y.; Raghow, R. Comparative proteomes of the proliferating C2C12 myoblasts and fully differentiated myotubes reveal the complexity of the skeletal muscle differentiation program. Mol. Cell. Proteomics 2004, 3, 1065–1082. [Google Scholar] [CrossRef]
- Timmons, J.A.; Larsson, O.; Jansson, E.; Fischer, H.; Gustafsson, T.; Greenhaff, P.L.; Ridden, J.; Rachman, J.; Peyrard-Janvid, M.; Wahlestedt, C.; Sundberg, C.J. Human muscle gene expression responses to endurance training provide a novel perspective on Duchenne muscular dystrophy. FASEB J. 2005, 19, 750–760. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, S.; Yan, Y.; Li, S.; Tong, H. SPARCL1 promotes C2C12 cell differentiation via BMP7-mediated BMP/TGF-β cell signaling pathway. Cell Death Dis. 2019, 10, 852. [Google Scholar] [CrossRef]
- Bortoletto, R.; Silva, N.S.; Zangaro, R.A.; Pacheco, M.T.T.; Da Matta, R.A.; Pacheco-Soares, C. Mitochondrial membrane potential after low-power laser irradiation. Lasers Med. Sci. 2004, 18, 204–206. [Google Scholar] [CrossRef] [PubMed]
- Groeger, G.; Quiney, C.; Cotter, T.G. Hydrogen peroxide as a cell-survival signaling molecule. Antioxid. Redox Signal. 2009, 11, 2655–2671. [Google Scholar] [CrossRef]
- Zhao, R.Z.; Jiang, S.; Zhang, L.; Yu, Z.B. Mitochondrial electron transport chain, ROS generation and uncoupling. Int. J. Mol. Med. 2019, 44, 3–15. [Google Scholar] [CrossRef]
- Aviña, A.E.; Chen, E.Y.H.; Chuang, K.M.Y.; Chang, C.Y.; Chang, C.J.; Yang, T.S. Safe Mitochondrial Activation through Photobiomodulation: Distinct Red and Near-Infrared Responses in Normal and Malignant Cells. J. Biophotonics 2025, e202500555. [Google Scholar] [CrossRef] [PubMed]
- Imamura, H.; Huynh Nhat, K.P.; Togawa, H.; Saito, K.; Iino, R.; Kato-Yamada, Y.; Nagai, T.; Noji, H. Visualization of ATP levels inside single living cells with fluorescence resonance energy transfer-based genetically encoded indicators. Proc. Natl. Acad. Sci. USA 2009, 106, 15651–15656. [Google Scholar] [CrossRef]
- van Hameren, G.; Campbell, G.; Deck, M.; Berthelot, J.; Gautier, B.; Quintana, P.; Chrast, R.; Tricaud, N. In vivo real-time dynamics of ATP and ROS production in axonal mitochondria show decoupling in mouse models of peripheral neuropathies. Acta Neuropathol. Commun. 2019, 7, 86. [Google Scholar] [CrossRef] [PubMed]
- Hour, T.C.; Vo, T.C.T.; Chuu, C.P.; Chang, H.W.; Su, Y.F.; Chen, C.H.; Chen, Y.K. The promotion of migration and myogenic differentiation in skeletal muscle cells by quercetin and underlying mechanisms. Nutrients 2022, 14, 4106. [Google Scholar] [CrossRef] [PubMed]
- Grada, A.; Otero-Viñas, M.; Prieto-Castrillo, F.; Obagi, Z.; Falanga, V. Research techniques made simple: Analysis of collective cell migration using the wound healing assay. J. Investig. Dermatol. 2017, 137, e11–e16. [Google Scholar] [CrossRef]
- Wang, Z.; Ongkosuwito, E.M.; Von den Hoff, J.W.; Wagener, F.A. The effects of TGF-β receptor I inhibitors on myofibroblast differentiation and myotube formation. Front. Cell Dev. Biol. 2025, 13, 1636884. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, M.T.; Ly, Q.K.; Ngo, T.H.P.; Lee, W. Calponin 3 regulates myoblast proliferation and differentiation through actin cytoskeleton remodeling and YAP1-mediated signaling in myoblasts. Cells 2025, 14, 142. [Google Scholar] [CrossRef]
- Breitwieser, K.; Voglstaetter, M.; Stockner, T.; Boeck, T.; Scheiflinger, F.; Binder, C.J. Detailed characterization of small extracellular vesicles from different cell types based on tetraspanin composition by ExoView R100 platform. Int. J. Mol. Sci. 2022, 23, 8544. [Google Scholar] [CrossRef]
- Debbi, L.; Zohar, R.; Shilo, S.; et al. Boosting extracellular vesicle secretion. Trends Biotechnol. 2022, 40, 1239–1257. [Google Scholar] [CrossRef]
- Guo, S.C.; Tao, S.C.; Yin, W.J.; Qi, X.; Yuan, T.; Zhang, C.Q. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics 2017, 7, 81. [Google Scholar] [CrossRef] [PubMed]
- Fukuta, T.; Nishikawa, A.; Kogure, K. Low level electricity increases the secretion of extracellular vesicles from cultured cells. Biochem. Biophys. Rep. 2020, 21, 100713. [Google Scholar] [CrossRef]
- Dompe, C.; Moncrieff, L.; Matys, J.; Grzech-Leśniak, K.; Kocherova, I.; Bryja, A.; Bruska, M.; Dominiak, M.; Mozdziak, P.; Skiba, T.H.I. Photobiomodulation: Underlying mechanisms and clinical applications. J. Clin. Med. 2020, 9, 1724. [Google Scholar] [CrossRef]
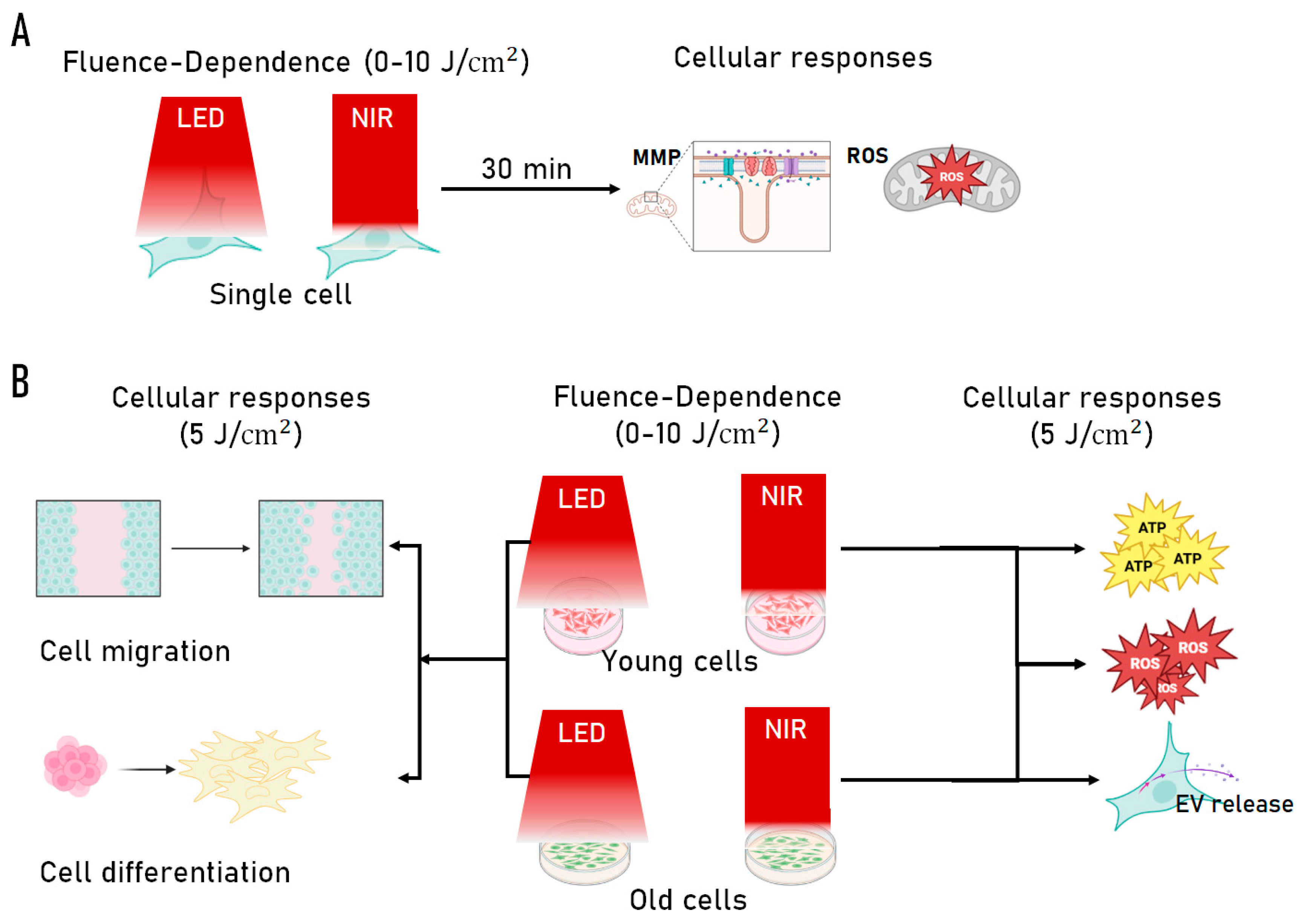
| Output (Single-cell) |
LED (660 nm) |
NIR (830 nm) |
Dose Dependence / Interpretation |
|
|---|---|---|---|---|
|
Mitochondrial membrane potential (ΔΨm, Rh123) |
↑ at 5 J/cm² | ↑ at 5 J/cm² | Biphasic, plateau ≥10 J/cm² | Both sources enhance mitochondrial polarization; optimal response at 5 J/cm² |
| Intracellular ROS (H₂DCFDA) | ↑ at 5 J/cm² | ↑ at 5 J/cm² | Biphasic | Moderate ROS signaling consistent with mitohormetic activation |
| Source comparison | Comparable to NIR | Comparable to LED | No strong wavelength-specific differences at single-cell level | |
| Dose selection outcome | 5 J/cm² selected | 5 J/cm² selected | Guided dose for population-level assays | |
| Output / Assay | Young Cells LED (660 nm) | Young Cells NIR (830 nm) | Old Cells LED (660 nm) |
Old Cells NIR (830 nm) |
Key Biological Trend |
|---|---|---|---|---|---|
| Metabolic activity (CCK-8) | ↑↑ | ↑ | ↑↑ | ↑ | LED produces stronger metabolic activation |
| ATP production | ↑ (≈ NIR) | ↑ (≈ LED) | ↑↑ (LED > NIR) | ↓ | Aged cells show greater ATP response to LED |
| Bulk ROS (% positive cells) | ↑↑ | ↑↑ | ↑↑ | ↑↑ | Strong redox activation independent of irradiation modality |
|
EV particle release (NTA) [Phenotype in early-passage] |
↑ [↑ CD9/CD81] |
↑↑ [↑↑ triple-positive EVs] |
↑↑ | ↑ | Source sensitivity shifts with age [NIR favors canonical small EV populations] |
| Migration (wound healing) | ↑↑ sustained (24–36 h) | ↑ transient / delayed | ↑ early only (16 h) | No sustained effect | Migration weak and strongly age-dependent; LED modestly improves young cell closure |
| Myogenic fusion (fusion index) | Trend ↑ | Significant ↑↑ | NS | Significant ↑↑ | NIR strongly promotes differentiation |
| System & Light Source | Fluence (J/cm²) | Time (s) | Power (W) | Area (cm²) |
|---|---|---|---|---|
|
Microchamber NIR 830 nm |
2.5 | 27 | 0.0073 | 0.000008 |
| 5.0 | 54 | |||
| 10.0 | 108 | |||
|
Microchamber LED 660 nm (5 mm pinhole before L1) |
2.5 | 27 | 0.0181 | 0.196 |
| 5.0 | 54 | |||
| 10.0 | 108 | |||
|
96-well NIR 830 nm |
2.5 | 130 | 0.0073 | 0.38 |
| 5.0 | 260 | |||
| 10.0 | 520 | |||
| 15.0 | 780 | |||
|
96-well LED 660 nm (5 mm pinhole before L2) |
2.5 | 28 | 0.034 | 0.38 |
| 5.0 | 56 | |||
| 10.0 | 112 | |||
| 15.0 | 168 | |||
|
24-well NIR 830 nm |
5.0 | 32 | 0.311 | 2.01 |
|
24-well LED 660 nm |
5.0 | 75 | 0.127 | 1.90 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




