Submitted:
28 January 2026
Posted:
05 February 2026
You are already at the latest version
Abstract
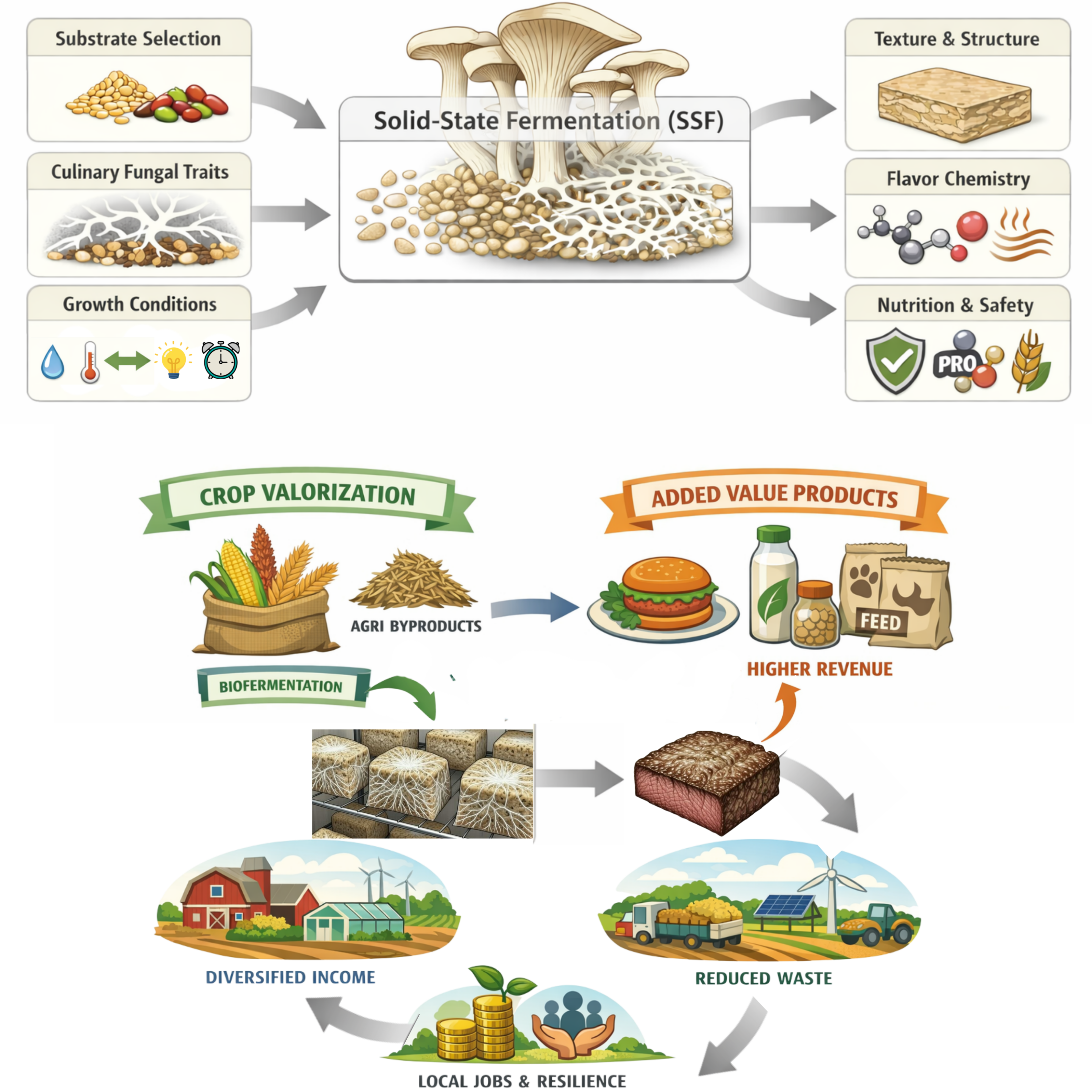
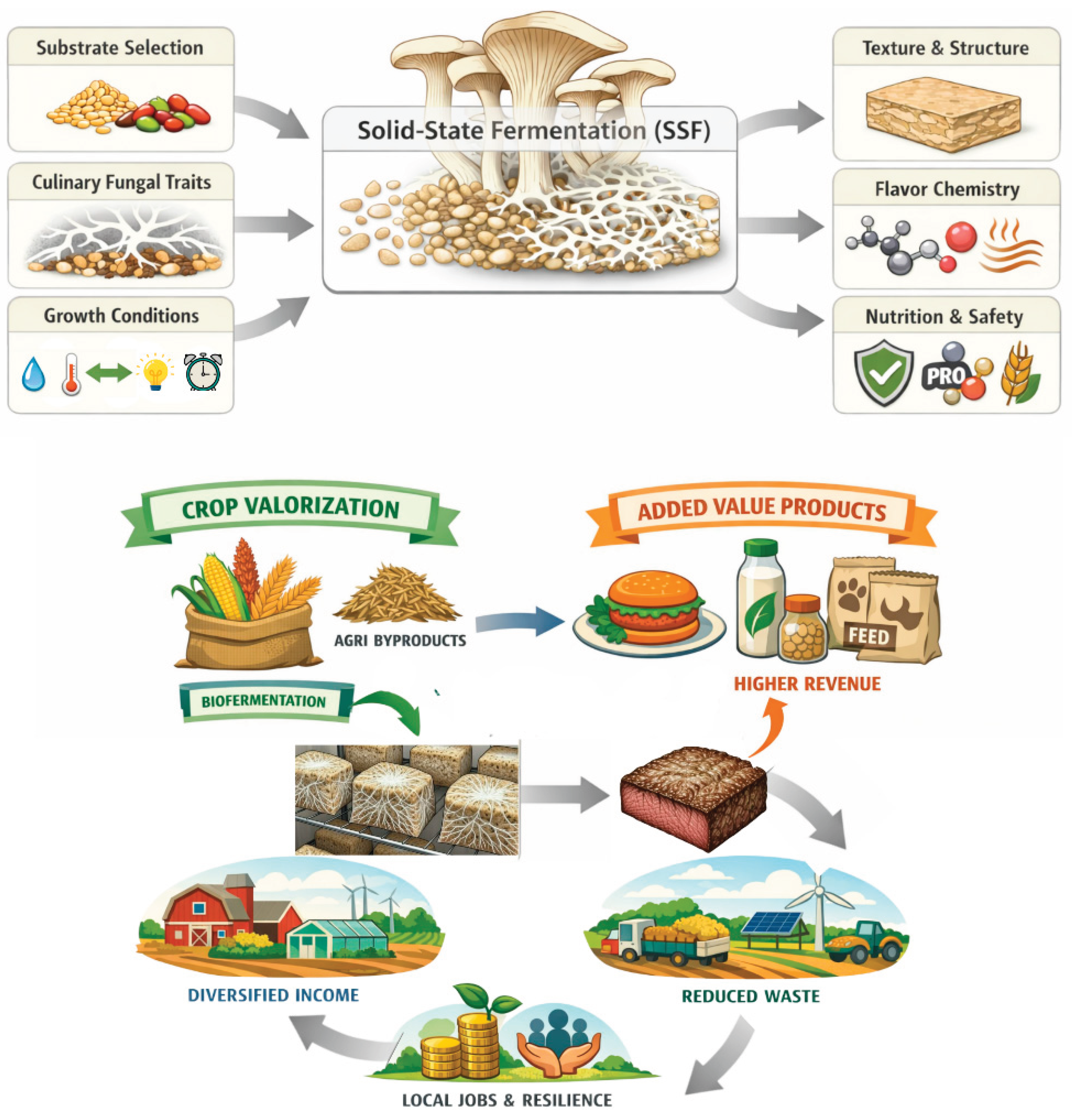
Keywords:
1. Introduction
2. Discussion and the Future of SSF
3. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hölker U, Höfer M, Lenz J. Biotechnological advantages of laboratory-scale solid-state fermentation with fungi. Appl Microbiol Biotechnol. 2004;64(2):175-186. [CrossRef]
- Thomas L, Larroche C, Pandey A. Current developments in solid-state fermentation. Biochemical Engineering Journal. 2013;81:146-161. [CrossRef]
- Wijaya CH, Prabawati PI, et al. Oncom: A Nutritive Functional Fermented Food Made from Food Process Solid Residue. Applied Sciences. 2024;14(22):10702. [CrossRef]
- Hölker U, Lenz J. Solid-state fermentation--are there any biotechnological advantages? Curr Opin Microbiol. 2005;8(3):301-306. [CrossRef]
- Milcarz A, Harasym J. Solid State Fermentation—A Promising Approach to Produce Meat Analogues. Foods. 2025;14(10). [CrossRef]
- Elhalis H, See XY, Osen R, Chin XH, Chow Y. Significance of Fermentation in Plant-Based Meat Analogs: A Critical Review of Nutrition, and Safety-Related Aspects. Foods. 2023;12(17):3222. [CrossRef]
- Pandey A. Solid-state fermentation. Biochemical Engineering Journal. 2003;13(2):81-84. [CrossRef]
- Elhalis H, See XY, Osen R, Chin XH, Chow Y. The potentials and challenges of using fermentation to improve the sensory quality of plant-based meat analogs. (1664-302X (Print)). [CrossRef]
- Eklund-Jonsson C, Sandberg A-S, Hulthén L. Tempe Fermentation of Whole Grain Barley Increased Human Iron Absorption and In Vitro Iron Availability. The Open Nutrition Journal. 2008;2:42-47. [CrossRef]
- Ahnan-Winarno AD, Cordeiro L, Winarno FG, Gibbons J, Xiao H. Tempeh: A semicentennial review on its health benefits, fermentation, safety, and sustainability. Comprehensive Reviews in Food Science and Food Safety. 2021;20(2):1717-1767. [CrossRef]
- Holt RR, Munafo JP, Jr., Salmen J, et al. Mycelium: A Nutrient-Dense Food To Help Address World Hunger, Promote Health, and Support a Regenerative Food System. J Agric Food Chem. 2024;72(5):2697-2707. [CrossRef]
- Garrido-Galand S, Asensio-Grau A, Calvo-Lerma J, Heredia A, Andrés A. The potential of fermentation on nutritional and technological improvement of cereal and legume flours: A review. Food Res Int. 2021;145:110398. [CrossRef]
- GFI. Deep dive: Fermentation upstream bioprocess design. Good Food Institute2025.
- Chaffee O, Ardoin R. Consumer perceptions of plant-based and mushroom-based jerky: A focus on texture, main ingredient and protein information, and willingness to pay. Current Research in Food Science. 2025;10:101058. [CrossRef]
- Carrasco-González JA, Serna-Saldívar SO, Gutiérrez-Uribe JA. Nutritional composition and nutraceutical properties of the Pleurotus fruiting bodies: Potential use as food ingredient. Journal of Food Composition and Analysis. 2017;58:69-81. [CrossRef]
- Grabarczyk M, Mączka W, Wińska K, Uklańska-Pusz C. Mushrooms of the Pleurotus genus—properties and application. Biotechnology and Food Science. 2019;83(1):13-30.
- Yin Z, Sun-Waterhouse D, Wang J, Ma C, Waterhouse GIN, Kang W. Polysaccharides from edible fungi Pleurotus spp.: advances and perspectives. Journal of Future Foods. 2021;1(2):128-140. [CrossRef]
- Sabu A, Sarita S, Pandey A, Bogar B, Szakacs G, Soccol CR. Solid-state fermentation for production of phytase by Rhizopus oligosporus. Appl Biochem Biotechnol. 2002;102-103(1-6):251-260. [CrossRef]
- Manikandan A, Muthusamy S, Wang ES, et al. Breeding and biotechnology approaches to enhance the nutritional quality of rapeseed byproducts for sustainable alternative protein sources- a critical review. Frontiers in Plant Science. 2024;Volume 15 - 2024.
- Jatuwong K, Suwannarach N, Kumla J, Penkhrue W, Kakumyan P, Lumyong S. Bioprocess for Production, Characteristics, and Biotechnological Applications of Fungal Phytases. Frontiers in Microbiology. 2020;Volume 11 - 2020.
- Ritota M, Manzi P. Edible mushrooms: Functional foods or functional ingredients? A focus on Pleurotus spp. AIMS Agriculture and Food. 2023;8(2):391-439. [CrossRef]
- Rodrigues BJ, dos Santos Freitas MM, da Silva Martins LH, de Carvalho RN. Polysaccharides of mushroom Pleurotus spp.: New extraction techniques, biological activities and development of new technologies. Carbohydrate Polymers. 2020;229:115550. [CrossRef]
- Torres-Martínez BM, Vargas-Sánchez RD, Torrescano-Urrutia GR, et al. Pleurotus genus as a potential ingredient for meat products. Foods. 2022;11. [CrossRef]
- Heidari F, Øverland M, Hansen JØ, et al. Solid-state fermentation of Pleurotus ostreatus to improve the nutritional profile of mechanically-fractionated canola meal. Biochemical Engineering Journal. 2022;187:108591. [CrossRef]
- Eliopoulos C, Langousi I, Kougia E, et al. Solid-State Fermentation Initiated by Pleurotus ostreatus of a Cottonseed Cake and Lathyrus clymenum Pericarp Mixture: Impact on Nutritional Profile and Gossypol Content. Applied Sciences. 2024;14(12). [CrossRef]
- Lee I-H, Hung Y-H, Chou C-C. Solid-state fermentation with fungi to enhance the antioxidative activity, total phenolic and anthocyanin contents of black bean. International Journal of Food Microbiology. 2008;121(2):150-156. [CrossRef]
- Lee AML, Chin CFS, Seelan JSS, Chye FY, Lee HH, Rakib MRM. Metabolites profiling of protein enriched oyster mushroom (Pleurotus ostreatus (Jacq.) P. Kumm.) grown on oil palm empty fruit bunch substrate. LWT. 2023;181:114731. [CrossRef]
- Adebo OA, Gabriela Medina-Meza I. Impact of Fermentation on the Phenolic Compounds and Antioxidant Activity of Whole Cereal Grains: A Mini Review. Molecules. 2020;25(4). [CrossRef]
- Pascual MM, Herbert LT, Campos M, Jurski V, Painefilú JC, Luquet CM. Nutritional improvement of wheat grains and soybeans by solid-state fermentation with Pleurotus ostreatus mycelium. Innovative Food Science & Emerging Technologies. 2025;102:104021. [CrossRef]
- Kaneko K, Aoyagi Y, Fukuuchi T, Inazawa K, Yamaoka N. Total purine and purine base content of common foodstuffs for facilitating nutritional therapy for gout and hyperuricemia. Biol Pharm Bull. 2014;37(5):709-721. [CrossRef]
- Żuchowski J, Pecio Ł, Jaszek M, Stochmal A. Solid-state fermentation of rapeseed meal with the white-rot fungi trametes versicolor and Pleurotus ostreatus. Appl Biochem Biotechnol. 2013;171(8):2075-2081. [CrossRef]
- Niu Y, Jiang M, Guo M, et al. Characterization of the Factors that Influence Sinapine Concentration in Rapeseed Meal during Fermentation. PLOS ONE. 2015;10(1):e0116470. [CrossRef]
- Zhang WJ, Xu ZR, Zhao SH, Jiang JF, Wang YB, Yan XH. Optimization of process parameters for reduction of gossypol levels in cottonseed meal by Candida tropicalis ZD-3 during solid substrate fermentation. Toxicon. 2006;48(2):221-226. [CrossRef]
- Diamantopoulou P, Fourtaka K, Melanouri EM, et al. Examining the Impact of Substrate Composition on the Biochemical Properties and Antioxidant Activity of Pleurotus and Agaricus Mushrooms. Fermentation. 2023;9(7). [CrossRef]
- Ritota M, Manzi P. Pleurotus spp. cultivation on different agri-food by-products: Example of biotechnological application. Sustainability. 2019;11(18). [CrossRef]
- Boin E, Cláudia A, João N, Guerra MM. Consumer Acceptability and Descriptive Characterization of Fresh and Dried King Oyster (Pleurotus eryngii) and Hedgehog (Hydnum repandum) Mushrooms. Journal of Food Research. 2016;5:55-55. [CrossRef]
- Eklund-Jonsson C, Sandberg A-S, Alminger M. Reduction of phytate content while preserving minerals during whole grain cereal tempe fermentation. Journal of Cereal Science. 2006;44(2):154-160. [CrossRef]
- He M, Peng Q, Xu X, Shi B, Qiao Y. Antioxidant capacities and non-volatile metabolites changes after solid-state fermentation of soybean using oyster mushroom (Pleurotus ostreatus) mycelium. Frontiers in Nutrition. 2024;Volume 11 - 2024.
- Mumpuni A, Ekowati N, Purnomowati P, Purwati E. Growth and Protein Content Establishment of Pleurotus ostreatus on Liquid and Solid Medium. Biosaintifika: Journal of Biology & Biology Education. 2017;9:572. [CrossRef]
- Reis FS, Barros L, Martins A, Ferreira ICFR. Chemical composition and nutritional value of the most widely appreciated cultivated mushrooms: An inter-species comparative study. Food and Chemical Toxicology. 2012;50(2):191-197. [CrossRef]
- Wang X-M, Zhang J, Wu L-H, et al. A mini-review of chemical composition and nutritional value of edible wild-grown mushroom from China. Food Chemistry. 2014;151:279-285. [CrossRef]
- Meyer F, Hutmacher A, Lu B, Steiger N, Nyström L, Narciso JO. Vegan shrimp alternative made with pink oyster and lion's mane mushrooms: Nutritional profiles, presence of conjugated phenolic acids, and prototyping. Curr Res Food Sci. 2023;7:100572. [CrossRef]
- Srivastava A, Attri BL, Bijla S, Arjun AD, Sharma P. Utilization of Lion’s Mane mushroom (Hericium erinaceus) for vegan crab analogue: effects of pre-treatments on nutritional, sensory, and physicochemical properties. Journal of Food Science and Technology. 2025. [CrossRef]
- Gonkhom D, Luangharn T, Raghoonundon B, Hyde K, Stadler M, Thongklang N. Hericium: A review of the cultivation, health-enhancing applications, economic importance, industrial, and pharmaceutical applications. Fungal Biotec. 2021;1:115-127. [CrossRef]
- Ge F, Chen Y, Wang B, Zhou W, Du B, Hou L. Bioactive Polysaccharides from Hericium erinaceus: Extraction, Structure, Bioactivities, and Applications. Molecules. 2025;30(8):1850. [CrossRef]
- Górska N, Włodarczyk K, Różańska A. Tempeh and Fermentation—Innovative Substrates, Co-Cultures, and Nutritional Potential: A Review. Applied Sciences. 2025;15(16):8888. [CrossRef]
- Song W, Li M, Yue X, Zheng Y. Effect of extraction and purification on the structure and activity of Flammulina velutipes polysaccharides: a review. International Journal of Food Science and Technology. 2024;59(12):9571-9591. [CrossRef]
- Bandara AR. A review of the polysaccharide, protein and selected nutrient content of Auricularia, and their potential pharmacological value. Mycosphere. 2019;10(1):579-607. [CrossRef]
- Sone Y, Kakuta M, Misaki A. Isolation and Characterization of Polysaccharides of “Kikurage,” Fruit Body of Auricularia auricula-judae. Agricultural and Biological Chemistry. 1978;42(2):417-425. [CrossRef]
- Ma Z, Wang J, Zhang L. Structure and chain conformation of β-glucan isolated from Auricularia auricula-judae. Biopolymers. 2008;89(7):614-622. [CrossRef]
- Liu M, Jiang W, Huang K, et al. Sustainable Cultivation and Functional Bioactive Compounds of Auricularia Mushrooms: Advances, Challenges, and Future Prospects. Biology. 2025;14(11):1555. [CrossRef]
- Regis M, Geösel A. Cultivation of Auricularia species: a review of the history, health benefits, principles, practices, environmental conditions, research methods, and recent trends. Sydowia. 2023;76.
- Geng J, Wang G, Guo J, et al. Preparation and structural analysis of fucomannogalactan and β-1,6-glucan from Grifola frondosa mycelium. Frontiers in Chemistry. 2023;Volume 11 - 2023.
- Yang Y-M, Fu X, Cui F-J, Sun L, Zan X-Y, Sun W-J. Biochemical and structural characterization of a glucan synthase GFGLS2 from edible fungus Grifola frondosa to synthesize β-1, 3-glucan. Biotechnology for Biofuels and Bioproducts. 2023;16(1):163. [CrossRef]
- Cerletti C, Esposito S, Iacoviello L. Edible Mushrooms and Beta-Glucans: Impact on Human Health. Nutrients. 2021;13(7). [CrossRef]
- Miles PG, Chang S-T. Mushrooms: cultivation, nutritional value, medicinal effect, and environmental impact. CRC press; 2004.
- Lee DJ, Kang AN, Lee J, et al. Molecular characterization of Fusarium venenatum-based microbial protein in animal models of obesity using multi-omics analysis. Communications Biology. 2024;7(1):133. [CrossRef]
- Boye J, Wijesinha-Bettoni R, Burlingame B. Protein quality evaluation twenty years after the introduction of the protein digestibility corrected amino acid score method. Br J Nutr. 2012;108 Suppl 2:S183-211. [CrossRef]
- Wolf WJ. Soybean proteins. Their functional, chemical, and physical properties. Journal of Agricultural and Food Chemistry. 1970;18(6):969-976. [CrossRef]
- Friedman M, Brandon DL. Nutritional and Health Benefits of Soy Proteins. Journal of Agricultural and Food Chemistry. 2001;49(3):1069-1086. [CrossRef]
- Chen G, Liu Y, Zeng J, Tian X, Bei Q, Wu Z. Enhancing three phenolic fractions of oats (Avena sativa L.) and their antioxidant activities by solid-state fermentation with Monascus anka and Bacillus subtilis. Journal of Cereal Science. 2020;93:102940. [CrossRef]
- Queiroz Santos VA, Nascimento CG, Schmidt CAP, Mantovani D, Dekker RFH, da Cunha MAA. Solid-state fermentation of soybean okara: Isoflavones biotransformation, antioxidant activity and enhancement of nutritional quality. LWT. 2018;92:509-515. [CrossRef]
- Verduzco-Oliva R, Gutierrez-Uribe JA. Beyond Enzyme Production: Solid State Fermentation (SSF) as an Alternative Approach to Produce Antioxidant Polysaccharides. Sustainability. 2020;12(2):495. [CrossRef]
- Mukherjee R, Chakraborty R, Dutta A. Role of Fermentation in Improving Nutritional Quality of Soybean Meal - A Review. Asian-Australas J Anim Sci. 2016;29(11):1523-1529. [CrossRef]
- Bekirian C, Valsecchi I, Bachellier-Bassi S, et al. β-1,6-Glucan plays a central role in the structure and remodeling of the bilaminate fungal cell wall. Elife. 2024;13. [CrossRef]
- Colosimo R, Mulet-Cabero A-I, Warren FJ, Edwards CH, Finnigan TJA, Wilde PJ. Mycoprotein ingredient structure reduces lipolysis and binds bile salts during simulated gastrointestinal digestion. Food & Function. 2020;11(12):10896-10906. [CrossRef]
- Jiang Q, Zhang M, Mujumdar AS. UV induced conversion during drying of ergosterol to vitamin D in various mushrooms: Effect of different drying conditions. Trends Food Sci Technol. 2020;105:200-210. [CrossRef]
- Cardwell G, Bornman JF, James AP, Black LJ. A Review of Mushrooms as a Potential Source of Dietary Vitamin D. Nutrients. 2018;10(10). [CrossRef]
- Bashir KMI, Choi J-S. Clinical and Physiological Perspectives of β-Glucans: The Past, Present, and Future. International Journal of Molecular Sciences. 2017;18(9). [CrossRef]
- Kumari R, Koul B, Kumar V, Kumar A, Somal MK, Sachan RSK. Protease and chitinase activity of Trichoderma isolates and their synergy with biochar in enhancing chickpea defense related enzymes. Frontiers in Microbiology. 2025;Volume 16 - 2025.
- El-Shora HM, El-Sharkawy RM, Khateb AM, Darwish DB. Production and immobilization of β-glucanase from Aspergillus niger with its applications in bioethanol production and biocontrol of phytopathogenic fungi. Sci Rep. 2021;11(1):21000. [CrossRef]
- Scholtmeijer K, van den Broek LAM, Fischer ARH, van Peer A. Potential Protein Production from Lignocellulosic Materials Using Edible Mushroom Forming Fungi. J Agric Food Chem. 2023;71(11):4450-4457. [CrossRef]
- Gardeli C, Mela N, Dedousi M, et al. The Influence of Substrate and Strain on Protein Quality of Pleurotus ostreatus. Applied Sciences. 2024;14(10). [CrossRef]
- Ng T-B. Protease Inhibitors, Lectins, Antifungal Protein and Saponins in Soybean. In: Ng T-B, ed. Soybean - Biochemistry, Chemistry and Physiology. London: IntechOpen; 2011.
- Sharma NK, Beniwal V, Kumar N, Kumar S, Pathera AK, Ray A. Production of tannase under solid-state fermentation and its application in detannification of guava juice. Prep Biochem Biotechnol. 2014;44(3):281-290. [CrossRef]
- van de Lagemaat J, Pyle DL. Solid-state fermentation: a continuous process for fungal tannase production. Biotechnol Bioeng. 2004;87(7):924-929. [CrossRef]
- Asghar A, Afzaal M, Saeed F, et al. Valorization and food applications of okara (soybean residue): A concurrent review. Food Sci Nutr. 2023;11(7):3631-3640. [CrossRef]
- Terefe ZK, Omwamba MN, Nduko JM. Effect of solid state fermentation on proximate composition, antinutritional factors and in vitro protein digestibility of maize flour. Food Sci Nutr. 2021;9(11):6343-6352. [CrossRef]
- Heidari F. Fungal treatment to enhance the nutritional value of canola meal for animal feed [Thesis]. Minneapolis, MN, University of Minnesota; 2023.
- Rozan P, Villaum C, Bau HM, Schwertz A, Nicolas JP, Méjean L. Detoxication of rapeseed meal by Rhizopus Oligosporus sp-T3: A first step towards rapeseed protein concentrate. International Journal of Food Science & Technology. 1996;31(1):85-90.
- Mageshwaran V, Satankar V, Paul S. Solid-state fermentation for gossypol detoxification and nutritive enrichment of cottonseed cake: A scale-up of batch fermentation process. BioResources. 2024;19(1):1107-1118. [CrossRef]
- Bhanja Dey T, Kuhad RC. Enhanced production and extraction of phenolic compounds from wheat by solid-state fermentation with Rhizopus oryzae RCK2012. Biotechnology Reports. 2014;4:120-127. [CrossRef]
- Wu JY, Wee S, Ler SG, Henry CJ, Gunaratne J. Unraveling the impact of tempeh fermentation on protein nutrients: An in vitro proteomics and peptidomics approach. Food Chemistry. 2025;474:143154. [CrossRef]
- Wiebe MG. Myco-protein from Fusarium venenatum: a well-established product for human consumption. Appl Microbiol Biotechnol. 2002;58(4):421-427. [CrossRef]
- Lonchamp J, Akintoye M, Clegg PS, Euston SR. Sonicated extracts from the Quorn fermentation co-product as oil-lowering emulsifiers and foaming agents. European Food Research and Technology. 2020;246(4):767-780. [CrossRef]
- Yegin S. Solid-state fermentation as a strategy for improvement of bioactive properties of the plant-based food resources. Bioresour Bioprocess. 2025;12(1):140. [CrossRef]
- Petraglia T, Latronico T, Liuzzi GM, Fanigliulo A, Crescenzi A, Rossano R. Hydrolytic Enzymes in the Secretome of the Mushrooms P. eryngii and P. ostreatus: A Comparison Between the Two Species. Molecules. 2025;30(12). [CrossRef]
- El-Gendi H, Saleh AK, Badierah R, Redwan EM, El-Maradny YA, El-Fakharany EM. A Comprehensive Insight into Fungal Enzymes: Structure, Classification, and Their Role in Mankind's Challenges. J Fungi (Basel). 2021;8(1). [CrossRef]
- Alvarez-Cervantes J, Sanchez C, Diaz R, Diaz-Godinez G. Characterization of production of laccases, cellulases and xylanases of Pleurotus ostreatus grown on solid-state fermentation using an inert support. Revista Mexicana de Ingeniería Química. 2016;15(2):323-331. [CrossRef]
- Xu LN, Guo S, Zhang S. Effects of solid-state fermentation with three higher fungi on the total phenol contents and antioxidant properties of diverse cereal grains. FEMS Microbiol Lett. 2018;365(16). [CrossRef]
- Li M, Wang H, Ng TB. Isolation of a phytase with distinctive characteristics from an edible mushroom, Pleurotus eryngii. Protein Pept Lett. 2013;20(4):459-466.
- Hoa HT, Wang CL. The Effects of Temperature and Nutritional Conditions on Mycelium Growth of Two Oyster Mushrooms (Pleurotus ostreatus and Pleurotus cystidiosus). Mycobiology. 2015;43(1):14-23. [CrossRef]
- Aditya, Neeraj, Jarial RS, Jarial K, Bhatia JN. Comprehensive review on oyster mushroom species (Agaricomycetes): Morphology, nutrition, cultivation and future aspects. Heliyon. 2024;10(5):e26539. [CrossRef]
- Rathore KS, Pandeya D, Campbell LM, et al. Ultra-Low Gossypol Cottonseed: Selective Gene Silencing Opens Up a Vast Resource of Plant-Based Protein to Improve Human Nutrition. Critical Reviews in Plant Sciences. 2020;39(1):1-29. [CrossRef]
- Zhang Y, Wei R, Azi F, et al. Solid-state fermentation with Rhizopus oligosporus RT-3 enhanced the nutritional properties of soybeans. Frontiers in Nutrition. 2022;Volume 9 - 2022.
- Stodolak B, StarzySka-Janiszewska A, Mickowska B. Effect of Flaxseed Oil-Cake Addition on the Nutritional Value of Grass Pea Tempeh. Food Science and Technology Research. 2013;19(6):1107-1114. [CrossRef]
- Chen L, Song P, Jia F, Wang JS. Reducing the Allergenicity from Food by Microbial Fermentation. Advanced Materials Research. 2012;524-527:2302-2305. [CrossRef]
- Tahmasian A, Drew R, Broadbent JA, Juhász A, Nye-Wood M, Colgrave ML. Conventional solid-state fermentation impacts the white lupin proteome reducing the abundance of allergenic peptides. Food Chemistry. 2023;426:136622. [CrossRef]
- Mattison CP, Dupre RA, Clermont K, Gibbons JG, Yu JH. Proteomic characterization of peanut flour fermented by Rhizopus oryzae. Heliyon. 2024;10(15):e34793. [CrossRef]
- Verhoeckx KCM, Vissers YM, Baumert JL, et al. Food processing and allergenicity. Food Chem Toxicol. 2015;80:223-240. [CrossRef]
- Schmidberger L, Schieberle P. Changes in the Key Aroma Compounds of Raw Shiitake Mushrooms (Lentinula edodes) Induced by Pan-Frying as well as by Rehydration of Dry Mushrooms. Journal of Agricultural and Food Chemistry. 2020. [CrossRef]
- Rahardjo YS, Tramper J, Rinzema A. Modeling conversion and transport phenomena in solid-state fermentation: a review and perspectives. Biotechnol Adv. 2006;24(2):161-179. [CrossRef]
- Handoyo T, Morita N. Hypoallergenic buckwheat flour preparation by Rhizopus oligosporus and its application to soba noodle. Food Research International. 2006;39(5):598-605. [CrossRef]
- Feng XM, Passoth V, Eklund-Jonsson C, Alminger ML, Schnürer J. Rhizopus oligosporus and yeast co-cultivation during barley tempeh fermentation—Nutritional impact and real-time PCR quantification of fungal growth dynamics. Food Microbiology. 2007;24:393-402. [CrossRef]
- Jeleń H, Majcher M, Ginja A, Kuligowski M. Determination of compounds responsible for tempeh aroma. Food chemistry. 2013;141(1):459-465. [CrossRef]
- Amin MNG, Kusnadi J, Hsu J-L, Doerksen RJ, Huang T-C. Identification of a novel umami peptide in tempeh (Indonesian fermented soybean) and its binding mechanism to the umami receptor T1R. Food Chemistry. 2020;333:127411. [CrossRef]
- Zhang Y, Venkitasamy C, Pan Z, Wang W. Recent developments on umami ingredients of edible mushrooms – A review. Trends in Food Science & Technology. 2013;33(2):78-92. [CrossRef]
- Godschalk-Broers L, Sala G, Scholten E. Meat Analogues: Relating Structure to Texture and Sensory Perception. Foods. 2022;11(15). [CrossRef]
- Tao A, Zhang H, Duan J, et al. Mechanism and application of fermentation to remove beany flavor from plant-based meat analogs: A mini review. Frontiers in Microbiology. 2022;13:1070773. [CrossRef]
- Appiani M, Cattaneo C, Laureati M. Sensory properties and consumer acceptance of plant-based meat, dairy, fish and eggs analogs: a systematic review. Frontiers in Sustainable Food Systems. 2023;Volume 7 - 2023.
- Aguilar CN, Rodríguez R, Gutiérrez-Sánchez G, et al. Microbial tannases: advances and perspectives. Appl Microbiol Biotechnol. 2007;76(1):47-59. [CrossRef]
- Saad MM, Saad AM, Hassan HM, Ibrahim EI, Hassabo AA, Ali BA. Bioremoval of tannins and heavy metals using immobilized tannase and biomass of Aspergillus glaucus. Microb Cell Fact. 2024;23(1):209. [CrossRef]
- Cai S, Gao F, Zhang X, et al. Evaluation of γ- aminobutyric acid, phytate and antioxidant activity of tempeh-like fermented oats (Avena sativa L.) prepared with different filamentous fungi. J Food Sci Technol. 2014;51(10):2544-2551. [CrossRef]
- Dekkers BL, Boom RM, van der Goot AJ. Structuring Processes for Meat Analogues. Trends in Food Science & Technology. 2018;81:25-36. [CrossRef]
- Wang Y, Tuccillo F, Lampi AM, et al. Flavor challenges in extruded plant-based meat alternatives: A review. Compr Rev Food Sci Food Saf. 2022;21(3):2898-2929. [CrossRef]
- Landaud S, Helinck S, Bonnarme P. Formation of volatile sulfur compounds by microorganisms in fermented foods: mechanisms and control. Food Microbiology. 2008.
- Mattheis JP, Roberts RG. Identification of geosmin as a volatile metabolite of Penicillium expansum. Appl Environ Microbiol. 1992;58(9):3170-3172. [CrossRef]
- Espinosa-Páez E, Hernández-Luna CE, Longoria-García S, et al. Pleurotus ostreatus: A potential concurrent biotransformation agent/ingredient on development of functional foods (cookies). LWT. 2021;148:111727. [CrossRef]
- Losoya-Sifuentes C, Simões LS, Cruz M, et al. Development and characterization of Pleurotus ostreatus Mushroom—Wheat bread. Starch - Stärke. 2022;74:2100126. [CrossRef]
- Aoyama Y, Kawahara S, Miyake Y. Characterization of kokumi gamma-glutamyl peptides and volatile compounds in misos. Food Research International. 2023;170:112986. [CrossRef]
- Yamaguchi S, Ninomiya K. Umami and Food Palatability. The Journal of Nutrition. 2000;130(4S):921S-926S.
- Ohsu T, Amino Y, Nagasaki H, et al. Involvement of the Calcium-Sensing Receptor in Human Taste Perception of Kokumi Substances. Journal of Agricultural and Food Chemistry. 2010.
- Li Q, Zhang L, Lametsch R. Current progress in kokumi-active peptides, evaluation and preparation methods: a review. Critical Reviews in Food Science and Nutrition. 2022;62(5):1230-1241. [CrossRef]
- Bryant C, Szejda K, Parekh N, Deshpande V, Tse B. A Survey of Consumer Perceptions of Plant-Based and Clean Meat in the USA, India, and China. Frontiers in Sustainable Food Systems. 2019;Volume 3 - 2019.
- Bryant C, Barnett J. Consumer acceptance of cultured meat: A systematic review. Meat Sci. 2018;143:8-17. [CrossRef]
- Siegrist M, Hartmann C. Consumer acceptance of novel food technologies. Nat Food. 2020;1(6):343-350. [CrossRef]
- Siegrist M, Hartmann C. Impact of sustainability perception on consumption of organic meat and meat substitutes. Appetite. 2019;132:196-202. [CrossRef]
- Aaslyng MD, Højer R. Introducing Tempeh as a New Plant-Based Protein Food Item on the Danish Market. Foods. 2021;10(11). [CrossRef]
- Szenderák J, Fróna D, Rákos M. Consumer Acceptance of Plant-Based Meat Substitutes: A Narrative Review. Foods. 2022;11(9). [CrossRef]
- Jang J, Lee D-W. Advancements in plant based meat analogs enhancing sensory and nutritional attributes. npj Science of Food. 2024;8(1):50. [CrossRef]
- da Silva BD, da Costa Marques JM, Conte-Junior CA. The meat mimicry dilemma in the sensory acceptance of plant-based meat analogue: A systematic review. Food and Humanity. 2025;5:100734. [CrossRef]
- Ng GCF, Choy MJY, Tan VWK, et al. Comparative analysis of sensory, textural, microstructural, amino acids and protein digestibility properties of animal and alternative meat products in the Asian market. International Journal of Food Science & Technology. 2024;59:5837-5858.
- Kim A, Öström Å, Mihnea M, Niimi J. Consumers’ Attachment to Meat: Association between Sensory Properties and Preferences for Plant-Based Meat Alternatives. Food Quality and Preference. 2024;116:105134. [CrossRef]
- Cirlincione F, Venturella G, Gargano ML, et al. Functional bread supplemented with Pleurotus eryngii powder: A potential new food for human health. International Journal of Gastronomy and Food Science. 2022;27:100449. [CrossRef]
- Sławińska A, Sołowiej BG, Radzki W, Fornal E. Wheat bread supplemented with Agaricus bisporus powder: Effect on bioactive substances content and technological quality. Foods. 2022;11(23):3786. [CrossRef]
- Ishak W, Azman W, Abu Bakar N. Does oyster mushroom (Pleurotus sajor-caju) powder addition improve nutrient composition, sensory acceptability, and glycaemic index (GI) of flatbread (Tortilla)? Kuwait Journal of Science. 2021;48(2):1-13. [CrossRef]
- Sitanggang AB, Sinaga WSL, Wie F, Fernando F, Krusong W. Enhanced antioxidant activity of okara through solid-state fermentation of GRAS Fungi. Food Science and Technology (Campinas). 2019;40(4). [CrossRef]
- Yasar S, Tosun R, Sonmez Z. Fungal fermentation inducing improved nutritional qualities associated with altered secondary protein structure of soybean meal determined by FTIR spectroscopy. Measurement. 2020;161:107895. [CrossRef]
- Nout MJR, de Dreu MA, Zuurbier AM, Bonants-van Laarhoven TMG. Ecology of controlled soyabean acidification for tempe manufacture. Food Microbiology. 1987;4(2):165-172. [CrossRef]
- Zhang W, Deng Z, Liu T, Liang J, Liu J. Fermentation with edible mushroom mycelia improves flavor characteristics and techno-functionalities of soybean protein. Food Bioscience. 2024;59:104123. [CrossRef]
- Gbenle J, Mert M, Phasha NN, et al. Fungal-mediated solid-state fermentation ameliorates antinutritional factors but does not improve in vitro digestibility of marama (Tylosema esculentum) beans. Future Foods. 2025;11:100664. [CrossRef]
- Pandey A, Soccol CR, Larroche C. Current Developments in Solid-State Fermentation. Berlin, Heidelberg: Springer; 2008.
- Artola A, Font X, Moral-Vico J, Sánchez A. The role of solid-state fermentation to transform existing waste treatment plants based on composting and anaerobic digestion into modern organic waste-based biorefineries, in the framework of circular bioeconomy. Frontiers in Chemical Engineering. 2024;6:1463785. [CrossRef]
- Sánchez C. Reactive oxygen species and antioxidant properties from mushrooms. Synthetic and Systems Biotechnology. 2017;2(1):13-22. [CrossRef]
- Lonnie M, Hooker E, Brunstrom JM, et al. Protein for Life: Review of Optimal Protein Intake, Sustainable Dietary Sources and the Effect on Appetite in Ageing Adults. Nutrients. 2018;10(3). [CrossRef]
- Yuwanita R, Isnansetyo A, Budhiyanti SA, Istiqomah I. Nutritional improvement of Spirulina (Arthrospira platensis) by-product through simultaneous fermentation with Bacillus subtilis (T2A) & (T3P1), and Lactococcus formosensis (JAL 11). Discover Food. 2025;6(1):27.
- Ayllón-Parra N, Castellari M, Gou P, Ribas-Agustí A. Effects of solid-state fermentation with Pleurotus ostreatus on the nutritional and techno-functional properties of alternative protein ingredients. Food Chemistry. 2025;490:145090. [CrossRef]
- Bonilla Loaiza AM, Rodríguez-Jasso RM, Belmares R, et al. Fungal Proteins from Sargassum spp. Using Solid-State Fermentation as a Green Bioprocess Strategy. Molecules. 2022;27(12). [CrossRef]
- Gervais P, Molin P. The role of water in solid-state fermentation. Biochemical Engineering Journal. 2003;13(2):85-101. [CrossRef]
- Mitchell DA, Ruiz HA, Krieger N. A Critical Evaluation of Recent Studies on Packed-Bed Bioreactors for Solid-State Fermentation. Processes. 2023;11(3):872. [CrossRef]
- Soccol CR, Costa ESFd, Letti LAJ, Karp SG, Woiciechowski AL, Vandenberghe LPdS. Recent developments and innovations in solid state fermentation. Biotechnology Research and Innovation. 2017;1(1):52-71. [CrossRef]
- Roenneberg T, Dragovic Z, Merrow M. Demasking biological oscillators: Properties and principles of entrainment exemplified by the Neurospora circadian clock. Proceedings of the National Academy of Sciences. 2005;102(21):7742-7747. [CrossRef]
- Merrow M, Roenneberg T. The circadian cycle: is the whole greater than the sum of its parts? Trends in Genetics. 2001;17(1):4-7. [CrossRef]
- Baker CL, Loros JJ, Dunlap JC. The circadian clock of Neurospora crassa. FEMS Microbiol Rev. 2012;36(1):95-110. [CrossRef]
- Corrochano LM. Light in the Fungal World: From Photoreception to Gene Transcription and Beyond. Annual Review of Genetics. 2019;53(Volume 53, 2019):149-170. [CrossRef]
- Bayram Ö S, Bayram Ö. An Anatomy of Fungal Eye: Fungal Photoreceptors and Signalling Mechanisms. J Fungi (Basel). 2023;9(5). [CrossRef]
- Qi Y, Sun X, Ma L, Wen Q, Qiu L, Shen J. Identification of two Pleurotus ostreatus blue light receptor genes (PoWC-1 and PoWC-2) and in vivo confirmation of complex PoWC-12 formation through yeast two hybrid system. Fungal Biol. 2020;124(1):8-14. [CrossRef]
- Ye D, Du F, Hu Q, Zou Y, Bai X. Transcriptome Analysis Reveals Candidate Genes Involved in Light-Induced Primordium Differentiation in Pleurotus eryngii. International Journal of Molecular Sciences. 2021;23(1):435. [CrossRef]
- Andayani SN, Lioe HN, Wijaya CH, Ogawa M. Umami fractions obtained from water-soluble extracts of red oncom and black oncom—Indonesian fermented soybean and peanut products. Journal of Food Science. 2020;85(3):657-665. [CrossRef]
- Dissasa G. Cultivation of different oyster mushroom (Pleurotus species) on coffee waste and determination of their relative biological efficiency and pectinase enzyme production, Ethiopia. International Journal of Microbiology. 2022. [CrossRef]
- Hultberg M, Asp H, Bergstrand KJ, Golovko O. Production of oyster mushroom (Pleurotus ostreatus) on sawdust supplemented with anaerobic digestate. Waste Management. 2023;155:1-7. [CrossRef]
- Gerber S, Roberts SB. Peanut hulls, an underutilized nutritious culinary ingredient: valorizing food waste for global food, health, and farm economies—a narrative review. Frontiers in Nutrition. 2024;Volume 11 - 2024.
- Chilakamarry CR, Mimi Sakinah AM, Zularisam AW, et al. Advances in solid-state fermentation for bioconversion of agricultural wastes to value-added products: Opportunities and challenges. Bioresource Technology. 2022;343:126065. [CrossRef]
- Erismann Y, Brück WM, Andlauer W. Solid-State Fermentation of Agro-Industrial By-Products. Nutraceuticals. 2025;5(2):11. [CrossRef]
- Navarro ME, Brizuela NS, Flores NE, et al. Preservation of Malolactic Starters of Lactiplantibacillus plantarum Strains Obtained by Solid-State Fermentation on Apple Pomace. Beverages. 2024;10(3):52. [CrossRef]
- FAO. The Impact of Disasters on Agriculture and Food Security 2025. Rome: Food Agriculture Organization of the United, Nations;2025.
- Grzegorczyk M, Kancelista A, Laba W, Piegza M, Witkowska D. The effect of lyophilization and storage time on the survival rate and hydrolytic activity of Trichoderma strains. Folia Microbiol (Praha). 2018;63(4):433-441. [CrossRef]
- Coblentz M, Evans JD, Kothe CI, et al. Food fermentation in space: Opportunities and challenges. iScience. 2025;28(4):112189. [CrossRef]
- Mycsology Foods I, National A, Space A. STTR Phase I: A Discovery Platform to Optimize the Production of Nutrient-Rich Food via Solid-State Fermentation of Feedstocks Relevant to Spaceflight. Washington, DC: NASA Shared Services Center; 2025-09-10 2025.
- Shahid M, Shah P, Mach K, et al. The environmental impact of mycoprotein-based meat alternatives compared to plant-based meat alternatives: A systematic review. Future Foods. 2024;10:100410. [CrossRef]
- Purwandari FA, Fogliano V, Capuano E. Tempeh fermentation improves the nutritional and functional characteristics of Jack beans (Canavalia ensiformis (L.) DC). Food & Function. 2024;15(7):3680-3691. [CrossRef]
- Olagunju LK, Isikhuemhen OS, Dele PA, et al. Effects of the Incubation Period of Pleurotus ostreatus on the Chemical Composition and Nutrient Availability of Solid-State-Fermented Corn Stover. Animals (Basel). 2023;13(16). [CrossRef]
- Watson E. Atlast Food Co: Mycelium scaffold can unlock the ‘Holy Grail’ of plant-based meat: whole cuts. FoodNavigator-USA. 2019.
- FAO. Sustainability Pathways: Livestock and Landscapes. Food Agriculture Organization of the United, Nations;2012.
- Ritchie H, Roser M. Half of the world’s habitable land is used for agriculture. Our World in Data. 2019.
- Bond JK. Corn and Other Feed Grains - Feed Grains Sector at a Glance. US Department of Agriculture, Economic Research Service. 2025.
- Cassidy ES, West PC, Gerber JS, Foley JA. Redefining agricultural yields: from tonnes to people nourished per hectare. Environmental Research Letters. 2013;8(3):034015. [CrossRef]
- Marbaniang EK. Cultivation of Oyster Mushroom: Training Manual. Meghalaya, India: Krishi Vigyan Kendra, West Khasi Hills (ICAR-ATARI Zone VII);2024.
- FAO. Indoor oyster mushroom (Pleurotus spp.) cultivation for livelihood diversification and resilience: Uganda field practice note. Rome: Food Agriculture Organization of the United, Nations;2023.
- FAO. Make money by growing mushrooms. Rome: Food Agriculture Organization of the United, Nations;2004.
- Clark AJ, Soni BK, Sharkey B, et al. Shiitake mycelium fermentation improves digestibility, nutritional value, flavor and functionality of plant proteins. LWT. 2022;156:113065. [CrossRef]
- Machida M, Yamada O, Gomi K. Genomics of Aspergillus oryzae: learning from the history of Koji mold and exploration of its future. DNA Res. 2008;15(4):173-183. [CrossRef]
- Steinkraus K. Industrialization of Indigenous Fermented Foods, Revised and Expanded (2nd ed.). . CRC Press; 2004.
- Center BC. Fermentos: Ciencia y técnica de la fermentación. Planeta Gastro2026.
- Maseko KH, Regnier T, Bartels P, Meiring B. Mushroom mycelia as sustainable alternative proteins for the production of hybrid cell-cultured meat: A review. J Food Sci. 2025;90(2):e70060. [CrossRef]
- Ogawa M, Kermani AS, Huynh MJ, Baar K, Leach JK, Block DE. Edible mycelium as proliferation and differentiation support for anchorage-dependent animal cells in cultivated meat production. npj Science of Food. 2024;8(1):23. [CrossRef]
- Gmoser R, Ferreira JA, Lennartsson PR, Taherzadeh MJ. Filamentous ascomycetes fungi as a source of natural pigments. Fungal Biol Biotechnol. 2017;4:4. [CrossRef]
- Wu H, Sakai K, Zhang J, McClements DJ. Plant-based meat analogs: color challenges and coloring agents. Food, Nutrition and Health. 2024;1(1):4. [CrossRef]
- Chaudhary V, Katyal P, Poonia AK, Kaur J, Puniya AK, Panwar H. Natural pigment from Monascus: The production and therapeutic significance. Journal of Applied Microbiology. 2022;133(1):18-38. [CrossRef]
- Future Market I. Mycoprotein Market Outlook (2024 to 2034): Demand for Sustainable Protein Sources Drives Growth. Future Market Insights;2024.
- McIntyre L. Section 3.9 Tempeh. In: McIntyre L, Fermented Foods working g, eds. Safety of fermented foods: Assessing risks in fermented food processing practices and advice on how to mitigate them. Environmental Health Services, BC Centre for Disease Control; 2024:5-13.
- Gibbons John G, Salichos L, Slot Jason C, et al. The Evolutionary Imprint of Domestication on Genome Variation and Function of the Filamentous Fungus Aspergillus oryzae. Current Biology. 2012;22(15):1403-1409. [CrossRef]
- Jeong E, Kwon YJ, Seo J-A. Distinction Between Aspergillus oryzae and Aflatoxigenic Aspergillus flavus by Rapid PCR Method Based on the Comparative Sequence Analysis of the Aflatoxin Biosynthesis Gene Cluster. Journal of Fungi. 2026;12(1):10. [CrossRef]
- Yue X, Ren X, Fu J, et al. Characterization and mechanism of aflatoxin degradation by a novel strain of Trichoderma reesei CGMCC3.5218. Front Microbiol. 2022;13:1003039. [CrossRef]
- EFSA Panel on Food Contact Materials E, Processing A, Silano V, et al. Safety evaluation of the food enzyme α-amylase from Aspergillus oryzae (strain DP-Bzb41). EFSA Journal. 2019;17(11):e05899.
- EPA. Framework for Risk Characterization of Environmental Problems. U.S. Environmental Protection Agency; 1997/09/01 1997.
- FDA. FDA Food Ingredient and Packaging Inventories 2025.
- Jia M, Yu X, Xu K, et al. The High-Efficiency Degradation of Multiple Mycotoxins by Lac-W Laccase in the Presence of Mediators. Toxins (Basel). 2024;16(11). [CrossRef]
- Niyigaba T, Küçükgöz K, Kołożyn-Krajewska D, Królikowski T, Trząskowska M. Advances in Fermentation Technology: A Focus on Health and Safety. Applied Sciences. 2025;15(6). [CrossRef]
- Dwipa RP, Maneeboon T, Chuaysrinule C, Mahakarnchanakul W. Interactive Inhibition of Aflatoxigenic Aspergillus flavus and Ochratoxigenic Aspergillus carbonarius by Aspergillus oryzae under Fluctuating Temperatures. Microbiology Research. 2022;13(1):114-127. [CrossRef]
- Watson E. There's more than one way to make meat from mycelium, says MyForest CEO: 'What we're doing is far cheaper than building fermenters'. FoodNavigator-USA. 2022.
- FDA. GRAS Notice No. GRN 000945: Agency Response Letter—Protein from mycelial biomass of Fusarium venenatum. College Park, MD: Center for Food Safety and Applied Nutrition, Office of Food Additive Safety; 2022/04/22 2022.
- FDA. GRN. 1152: Phoenix oyster mushroom (Pleurotus pulmonarius) mycelia biomass. In: FDA, ed2024.
- FDA. Recently Published GRAS Notices and FDA Letters. FDA. 2025.
- FDA. GRAS Notice No. GRN 001152: Agency Response Letter—Pleurotus mycelium biomass. College Park, MD: Center for Food Safety and Applied Nutrition, Office of Food Additive Safety.
- Henchion M, Hayes M, Mullen AM, Fenelon M, Tiwari B. Future Protein Supply and Demand: Strategies and Factors Influencing a Sustainable Equilibrium. Foods. 2017;6(7). [CrossRef]
- Qin J, Kurt E, LBassi T, Sa L, Xie D. Biotechnological production of omega-3 fatty acids: current status and future perspectives. Frontiers in Microbiology. 2023;Volume 14 - 2023.
| Outcome target | Representative SSF system(s) | Quantitative effect examples | Notes/implications | Key sources (year) |
| Protein content and bioavailability Increases | Canola meal × Pleurotus ostreatus (≈12 d); Soybeans × P. ostreatus (long run); Peanut press cake (oncom) × Neurospora sitophila/Rhizopus oligosporus; Okara × R. oligosporus/A. oryzae; Tempeh soy × Rhizopus | Content: +11–18% (canola); +27–28% (soy, 31 d); 52.6–55.35% (peanut press cake, dry basis); Essential AAs of > 12.06 g/100 g; Digestibility increased quality; >50% of soy protein became free amino acids (tempeh) | Mix of concentration effect + de novo fungal protein; enables protein-dense composite foods. Fungal amino acid spectrum complements legumes/cereals Protease/peptidase action. | Heidari 2022; He 2024; Nuramlia 2024; Wijaya 2024 ; Sitanggang 2020; van Veen & Sohaefer 1950; Steinkraus 2004; Wu 2025; Yegin 2025; Kumar 2025 |
| β-glucan Increases | Cottonseed cake + Lathyrus (80:20) × P. ostreatus (11 d); Fusarium venenatum mycoprotein and binding of bile salts | 5× increase in Beta-glucan content; Mycoprotein fiber 24% (DW), 2/3 β-glucans and 1/3 chitin | Adds soluble fiber with immune-metabolic potential55; texture contribution. In vitro digestion of mycoprotein reduced lipolysis and bound bile salts, a mechanism associated with reduced blood cholesterol in humans | Eliopoulos 2024; Ritota & Manzi 2023; Colosimo 2020 |
| Reduced Anti-Nutrients (phytates, glucosinolates, sinapine, gossypol) | Canola meal × P. ostreatus; Oats/Barley × Rhizopus (tempeh-style); Cottonseed + Lathyrus × P. ostreatus | Phytates: −55–76% (canola); oats −74%, barley -89%; Glucosinolates : -98.8%; Sinapine:99.8%; Gossypol: total reduction 60-80%, with some reporting up to −89%, and free gossypol −12.45% | Increases Fe/Zn accessibility; supports bioavailability gains; Major removal of pungent/bitterness drivers; flavor improvement; increased protein digestibility | Heidari 2022; Cai 2014; Handoyo & Morita 2006; Ahnan-Winarno 2021; Eklund-Jonsson 2006; Eklund-Jonsson 2008; Niu 2015; Gbenle 2025; Eliopoulos 2024 |
| Aflatoxin Reduction (contaminated inputs) | Peanut press cake (black oncom) × Neurospora/Rhizopus | −50–70% | Requires tight process control and clean sourcing | Wijaya 2024 |
| Increased antioxidants and phenolics | Soy × P. ostreatus (long SSF); Okara × R. oligosporus/A. oryzae; Oats × Rhizopus; Oats × R. oryzae; Okara × Rh. oligosporus/Asp. oryzae; Soybeans × P. ostreatus | Soy phenolics 4.47×, DPPH 3.92×; Okara +260–550% antioxidant; Oats total phenolics increase | Liberation of bound phenolics; flavor co-benefits of reduced bitterness and astringency; longer shelf-stability | He et al 2024; Sitanggang 2020; Cai 2014; Ahnan-Winarno 2021; Verduzco-Oliva 2020; Chen 2020; Quieroz Santos 2018 |
| Omega fatty acids (ω-3) profile | Grass pea + flax press cake × Rhizopus (tempeh) | ALA increases by >10×; ratio of ω6:ω3 from 11:1 to 0.5–2.5:1 | Co-substrate strategy; retains PUFA while carbs fuel growth | Stodolak 2013; Ahnan-Winarno 2021 |
| Vitamin & Mineral content and absorption increases | Okara (red oncom) × Neurospora intermedia; Barley meal × Rhizopus (tempeh-style) vs. boiled barley | Ca 215 mg/100 g; P 66 mg/100 g; Fe 12.5 mg/100 g; non-heme Fe absorption 5.5% vs. 3.0% (+83% relative); Vitamin B1 raised to 150 µg/100 g | Demonstrates micronutrient density in fermented matrices | Adebo 2020; Asghar 2023; Wijaya 2024; Eklund-Jonsson et al. 2008; van Veen & Sohaefer 1950; Anhan-Winarno 2021; Yegin 2025 |
| Flavor balance improvements | Barley/oats × Rhizopus (with/without yeasts); SSF soy/cereal × Pleurotus/Rhizopus; Brassica meals × Pleurotus; cereal/legume tempeh × Rhizopus | Reduced grassy, beany flavor ratings; increased glutamate, 5′-GMP/IM savory flavor volatiles; increased pyrazines, Strecker aldehydes, 2-acetylpyrrole for cooking step flavor expression; Sensory bitterness falls as sinapine/IP6 decline | Sensory lift via proteolysis + ribonucleotides + Maillard on cooking | Reis 2012; Carrasco-Gonzalez 2017; Feng 2007; Jelen 2013; Handoyo and Morita 2006; Amin 2020; Tao 2022; Zhang 2024; He 2024; Heidari 2022; Eklund-Jonsson 2006 |
| Texture, moisture and cohesion improvements | Cereal/legume SSF flours; Pleurotus (esp. P. eryngii) | higher water holding capacity (WHC); better juice release; elastic, sliceable networks (hyphal entanglement); Dense, anisotropic mycelial mats; Moisture 55–65%; 2–4 mm particle size; O2 diffusion | Enables anisotropic, meat-like bite with minimal texturizing | Godschalk-Broers 2022; Dekkers 2018; Carrasco-González 2017; Ritota & Manzi 2023; He 2024 |
| Allergenicity | Buckwheat soba (tempeh-processing) × Rhizopus; (lupin proteomics in SSF, various); Lupin × Rhizopus; peanut press cake oncom × Neurospora/Rhizopus | Loss of allergen bands; reductions in IgE not always reported; Lupin: β-conglutin peptides reduce; Peanut: IgE binding slightly lower. | Promising but awaiting more research to be clinically validated | Handoyo & Morita 2006; Ahnan-Winarno et al (2021); Tahmasian 2023 |
| Compound | Foodstuff Source Examples | Effects on Consumption | Example Impacts of Combining Through SSF | References |
| Phytate | Brassica meals, cereals, okara, brans, spent grains, pomace, pseudocereals | Chelates Fe, Zn, Ca, Mg, lowering bioavailability | Fungal phytases (e.g., Rhizopus, Aspergillus, Pleurotus) produce phytases that hydrolyze phytate releasing bound minerals | Pandey 2008; Heidari 2022; Eklund-Jonsson 2006; Eklund-Jonsson 2008; Cai 2014; Sabu 2002; Manikandan 2024; Jatuwong 2020; Zhang 2022 |
| Glucosinolates | Brassica meals (canola, mustard), cruciferous vegetables | Goitrogenic effects, thyroid dysfunction, bitter taste, reduced animal performance | Fermentation with Rhizopus oligosporus, Lactobacillus spp., or Bacillus spp. to hydrolyze glucosinolates into less harmful compounds | Adebo 2022; Heidari 2022; Traka 2016; Zuchowski 2013 |
| Sinapine | Brassicas (Rapeseed, mustard seed, and others) | Bitter, astringent taste; reduced protein digestion; may cause allergic response | Solid-state fermentation with Trametes sp., Rhizopus oligosporus, or Bacillus subtilis degrades sinapine via laccase and other enzymes | Heidari 2022; Ahnan-Winarno 2021; Adebo 2024; Manikandan 2024; Niu 2015 |
| Bound phenolics | Legumes, cereals (wheat), rapeseed, canola, quinoa, fruits, vegetables | Reduced protein/mineral bioavailability, astringency, reduced digestibility | Fermentation with Rhizopus oligosporus, Aspergillus niger, Pleurotus ostreatus, or Lactobacillus plantarum to release bound phenolics | Cai 2014; Sitanggang 2020; Anhan-Winarno 2021; Yasar 2020; Pascual 2025; Verduzco-Oliva 2020; Bhanja Dey 2014; Yegin 2025 |
| Tannins | Legumes, cereals, nuts, tea, fruits, vegetables | Reduced protein digestibility, astringency, toxicity at high intake | Fermentation with tannase-producing fungi (e.g Penicillium glabrum, Aspergillus glaucus, A. niger, Rhizopus sppand others) to hydrolyze tannins | Aguilar 2002; Saad 2024; Górska 2025; Traka, 2016; Górska 2025; Sharma 2014; van de Lagemaat 2004 |
| Protease inhibitors/lectins | Legumes (soybeans, kidney beans, chickpeas), cereals | Inhibit digestive enzymes, reduce protein utilization, cause pancreatic hypertrophy | Fermentation with Rhizopus oligosporus, Aspergillus oryzae, Bacillus subtilis, or protease-secreting fungi to degrade inhibitors/lectins | Friedman 2001; Wolf 1970; Ng 2011; Górska 2025; Adebo 2022; Yasar 2020; Mukherjee 2016; Gbenle 2025 |
| Allergenic proteins (β-conglutin) | Lupin seeds (Lupinus angustifolius, L. mutabilis), fungal/yeast biomass | IgE binding in sensitized individuals and allergenic reactions, immune hypersensitivity, reduce nutrient intake through increased excretion | Proteolysis during SSF reduces specific peptides; Fermentation with Rhizopus oligosporus, Propionibacterium spp., or protease-producing fungi to hydrolyze allergenic proteins (should be validated with immunoassays) | Tahmasian 2023; Verhoeckx 2015; Mattison 2024; Wijaya 2024 |
| Gossypol | Cottonseed meal or cake | Toxicity (liver, reproductive, cardiac), binds lysine | Solid-state fermentation with Candida tropicalis, Saccharomyces cerevisiae, Aspergillus niger, Pleurotus spp., or Paecilomyces variotii | Mageshwaran 2024; Adebo 2022; Eliopoulos 2024; Zhang 2006 |
| Saponins | Legumes (soybeans, chickpeas), quinoa, green microalgae | Bitter taste, hemolytic activity, reduced nutrient absorption | Fermentation with Rhizopus oligosporus, Aspergillus spp., Saccharomyces cerevisiae, or lactic acid bacteria to degrade saponins. SSF can alter saponin profile (sometimes reducing, sometimes increasing saponins); pre-washing/dehulling and post-processing are often required for reliable debittering. | Górska et al., 2025; Traka, 2016; Gautheron 2024 |
| Purines (nucleic acids) | Yeast-rich biomass, fungi, legumes | Uric acid load (hyperuricemia) for gout | Fermentation with Aspergillus oryzae, Blastobotrys adeninivorans, Candida utilis, or low-purine yeast strains; enzymatic degradation. Downstream RNA reduction is described by commercial operators using heat-shock to activate endogenous RNases as a standard mitigation step for fungal biomass intended for high intake. | Kaneko 2014; Adebo 2022; |
| Chitin/Beta-glucans | Fungal biomass, mushrooms, yeasts, microalgae | Reduces apparent protein digestibility in vitro; Associated with indigestion; tough chewing texture; potential allergenicity | Fermentation with chitinase/β-glucanase-producing fungi (Mucor rouxii, Aspergillus terreus, Trichoderma spp.). Milling, alkaline/thermal pretreatments, or targeted enzymatic hydrolysis can further break down to improve digestion. | Yang 2023; Bekirian 2024; Zhang 2013; Kumar 2025; El-Shora 2021 |
| Mycotoxins | Contaminated grains, nuts, by products | Toxicity (carcinogenicity, nephrotoxicity, immunosuppression, reproductive toxicity); carcinogens, hepatotoxicity, immunosuppression | Fermentation with mycotoxin-degrading fungi (Rhizopus spp., a-toxigenic strains of Aspergillus spp.); laccase/peroxidase enzymatic detoxification (lactonases, peroxidases, laccases); Aflatoxin-degrading fungi (Trichoderma reesei, Aspergillus niger, Rhizopus spp.) | Adebo 2022; Wijaya 2024; Yue 2022; Jia 2024 |
| Substrate | Typical examples | Baseline nutrition | SSF Outcomes | Formulation notes | Applied examples |
| Whole cereal grains & pseudocereals | Wheat, rice, maize, barley, oats, millet; buckwheat, quinoa | High starch, moderate protein; bound phenolics & phytate common | Increase protein concentration and/or digestibility; Increase free phenolics/antioxidants; Decrease phytate; added umami/volatiles and other sensory gains via fermentation metabolites | Steam/gelatinize grains or hydrate to support colonization; adjust moisture (commonly ~60–65% wb); control particle size and aeration to modulate moisture; inoculate warm; mill post-SSF for high-protein flour | Whole-grain cereal tempeh fermentation reduced phytate (oats/barley; Rhizopus) (Eklund-Jonsson 2006); barley tempeh increased Fe absorption vs. boiled barley (Eklund-Jonsson 2008); Diverse cereal grains show increased phenolics/antioxidant under (Xu 2018); more grain-fermentation phenolics effects (Adebo 2020). phenolics effects (Adebo 2020). |
| Legumes | Soybeans, chickpeas, lupin, black bean; legume flours | High protein (often 30–50% db), PUFA, protease inhibitors, lectins, phytates; beany notes (bitter phenolics) | Increase protein quality & digestibility (proteolysis);increased free amino acids; decrease in many anti-nutrients; EAA profile improved; increased phenolics and antioxidants including phenol pigments | Balance moisture and oxygen; consider dehulling/cooking to reduce flatulence factors and improve texture; co-blend with cereals when additional carbon is needed | Soybeans × Rhizopus oligosporus SSF (Zhang 2022). Black bean SSF with fungi increased antioxidative activity and phenolics (Lee 2008). White lupin SSF reduced allergenic peptides (proteomics) (Tahmasian 2023). |
| Oilseed, meals and press cakes | Canola/rapeseed meal, cottonseed cake, sunflower, sesame, coconut; okara and other protein-rich byproducts | High protein (~30–50% db), variable lipids; constraints include sinapine/glucosinolates (Brassica) or gossypol (cottonseed); bitterness/astringency | Increased protein concentration and digestibility; reduced brassica anti-nutrients; detoxification of gossypol | Co-ferment with a cereal fraction when C:N is limiting; manage residual oil to avoid hydrophobic zones; verify food-grade sourcing (esp. cottonseed) | Canola meal × Pleurotus ostreatus SSF degraded sinapine and glucosinolates (Heidari M. 2022,). Canola-meal upgrading (Heidari F. 2022). Cottonseed cake + Lathyrus pericarp x P. ostreatus increased β-glucans and decreased gossypol (Eliopoulos 2024). Wheat grains and soybeans × P. ostreatus nutritional improvements (Pascual 2025). |
| Fiber- & bran-rich byproducts | Wheat/rice bran, corn fiber; fruit/veg pomace (apple, grape, pumpkin); spent brewers’ grains | High insoluble fiber, micronutrients; low protein; bound phenols | Increase extractable phenols and antioxidants; partial fiber depolymerization softening fiber matrix; increased moisture holding capacity | Use as a fraction (e.g., 10–40%) blended into cereal/legume/oilseed bases to maintain growth; fine grinding improves uniform colonization; ensure low contaminant load and safe water activity | Wheat SSF × Rhizopus oryzae increased production of phenols (Bhanja Dey 2014). SSF as platform to produce antioxidant polysaccharides (Verduzco-Oliva & Gutierrez-Uribe 2020); cereal-grain antioxidants via SSF across fungi (Xu 2018). |
| Omega-rich seeds & lipid sources | Flaxseed, chia seed; defatted seed meals; oil press cakes | ALA/PUFA, lignans | Can improve ω-3 content and ω6:ω3 ratio in the composite when lipid fraction is blended into a carbohydrate-supporting matrix; retains PUFA while fungus consumes carbs | Pre-mix finely; excess free oil can inhibit if pooled, limit oil to avoid hydrophobic zones; combine with cereal/legume | Grass pea tempeh + flaxseed oil-cake improved nutritional value (including lipid profile) (Stodolak et al. 2013). |
| Microalgae and Aquatic biomass | Chlorella vulgaris (typically blended with cereals); Arthrospira/Spirulina (often with stabilization) | High protein and pigments; bound phenols; sensory off-notes or fishy odors; benefits from a carbohydrate carrier | Increased water retention and gellification/emulsification; broader micronutrient enhancements | Use as minor fraction (e.g., 5–30%) in cereal/legume composite; pre-treatments help support integration | P. ostreatus SSF on oat and Chlorella + oat improved protein solubility (Ayllón-Parra et al., 2025). Arthrospira SSF (Yuwanita 2025). SSF of Sargassum macroalgae with Aspergillus oryzae (Bonilla Loaiza 2022) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





