Introduction
The COVID-19 pandemic, caused by SARS-CoV-2, profoundly impacted the management of acute respiratory distress syndrome (ARDS) in the ICU [
1]. Among its complications, COVID-19-associated pulmonary aspergillosis (CAPA) emerged as a significant threat, affecting up to 30% of ventilated patients [
2]. Multiple risk factors predispose COVID-19 patients to CAPA, including direct viral damage to the respiratory epithelium, immune dysregulation, and prolonged mechanical ventilation [
3]. Additionally, immunomodulatory treatments such as corticosteroids and IL-6 inhibitors further increase susceptibility [
4,
5,
6].
Before COVID-19, the EORTC/MSG definitions were the reference for diagnosing invasive aspergillosis, primarily in immunocompromised patients [
7]. However, these criteria are not well suited for ICU patients, who often present atypical host factors and less specific radiological findings [
2,
8,
9]. To address this gap, the AspICU algorithm was developed before the pandemic to facilitate the diagnosis of pulmonary aspergillosis in critically ill patients, allowing the definition of “putative” invasive disease based on positive respiratory cultures even in the absence of classical risk factors [
10]. Following the pandemic, the ECMM/ISHAM consensus proposed adapted definitions for CAPA, with a stronger emphasis on mycological criteria [
11].
Recent work, notably by Dellière et al. [13], provided additional insights into the diagnostic limitations of current definitions. In a retrospective cohort of 176 mechanically ventilated COVID-19 patients (350 respiratory samples analyzed), the authors systematically evaluated seven mycological criteria and highlighted that isolated positive results -such as a single BAL galactomannan or culture-were often non-reproducible and weakly predictive of outcomes. Conversely, the combination of multiple complementary tests, particularly those reflecting high fungal burden (direct microscopy, serum galactomannan, plasma Aspergillus qPCR), offered a more reliable approach for identifying clinically relevant infections. These findings underscore the current absence of a gold standard and the risk of misclassifying colonization as invasive disease.
Given this diagnostic uncertainty, it is crucial to evaluate the practical utility and prognostic relevance of existing CAPA definitions. Our objective was to assess and compare the EORTC/MSG, AspICU, and ISHAM classifications for CAPA in severe COVID-19 patients admitted to the ICU.
Methods
Study Design
We conducted a retrospective, observational, monocentric study including all COVID-19 patients admitted to the intensive care units (ICUs) of Centre Hospitalier Universitaire de Charleroi, Marie Curie and Vesale hospitals, Belgium between March 2020 and December 2021. Approval was obtained from the local ethics committee (OM008) who waived the signed inform consent due to the retrospective design of the study.
Study Population
Inclusion criteria were age (equal or more than 18 years), a positive nasopharyngeal SARS-CoV-2 PCR test, and the requirement for invasive mechanical ventilation.Patients were excluded if they were transferred from another hospital (due to incomplete data) or presented a positive PCR without clinical signs of lower respiratory tract infection (absence of fever, dyspnea, hypoxemia, and/or radiographic pulmonary infiltrates).
Data collected were the demographic variables (age, sex, body mass index), APACHE II and SOFA scores at ICU admission, comorbidities (hypertension, diabetes mellitus, chronic obstructive pulmonary disease/asthma, active cancer, chronic immunosuppression, chronic kidney disease), as well as delays between symptom onset, and ICU admission. Management characteristics such as extra corporeal membrane oxygenation (ECMO) support, use of corticosteroids, and immunomodulators including convalescent plasma from the CONFIDENT trial [
9] were also recorded. Outcomes included length of ICU stay, ICU mortality, 28-day mortality and hospital mortality.
Diagnosis of CAPA
For microbiological investigations, screening for COVID-19-associated pulmonary aspergillosis (CAPA) included: serum and bronchoalveolar lavage (BAL) galactomannan detection, Aspergillus PCR, direct microscopic examination, and fungal culture from respiratory specimens.Investigations were prompted either by clinical/radiological deterioration or performed as part of routine screening protocols in ventilated patients.
Patients were classified according to three diagnostic systems: the 2021 updated EORTC/MSG criteria [
10] 15, the AspICU algorithm [
11], and the 2020 ECMM/ISHAM consensus definitions for CAPA [
12]. To better compare the three diagnostic systems, we defined five internal clinical groups based on both microbiological findings and clinical context. Definition of each group is showed in
Table 1.
Statistical Analysis
Descriptive statistics were computed. Quantitative variables are expressed as medians with interquartile ranges (IQR) or means ± standard deviation (SD), as appropriate. Qualitative variables are presented as absolute and relative frequencies. In comparative analyses on differences across groups and waves of COVID-19, categorical variables were compared using chi-squared tests or Fisher’s exacts tests, and quantitative variables were compared using Mann-Whitney U tests or Kruskal Wallis tests as appropriate. Modified scores were computed to facilitate two-by-two comparisons as explained before. The agreement between modified scores was displayed in bivariate scatterplots using disk areas proportional to the number of patients entering each combination. The degree of agreement between modified ISHAM score and modified EORTC score (whose response modalities were identical) was also computed using an unweighted kappa coefficient. Statistical analyses were performed on R version 4.4.2 (R Core Team (2024). _R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria).
Results
Study Population
Among the 500 patients initially screened, 405 fulfilled the inclusion criteria. Demographic datas are shown in
Table 2. The median age was 63 years [IQR 54.8–71.0], with a predominance of male patients (65.2%). The main comorbidities were hypertension (57.3%), diabetes mellitus (40.2%), chronic respiratory disease (20.7%), active malignancy (7.6%), and chronic immunosuppression (12.8%). The median body mass index was 29.1 [IQR 26.0–32.7] kg/m
2. The median APACHE II score was 13 [IQR 10–17].
Respiratory Management and Outcomes
All 405 patients underwent invasive mechanical ventilation. Prone positioning was performed in 76% of patients, and ECMO support was used in 15,5% of cases. (
Table 3)
ICU mortality for the overall cohort was 47.2%, mainly due to refractory hypoxemia and alveolar hemorrhage. (
Table 4)
CAPA Classification According to Diagnostic Criteria
According to EORTC/MSG criteria, probable CAPA was diagnosed in 4.9% of patients (n=20), possible CAPA in 1.2% (n=5), and CAPA was ruled out in 93.8% (n=380).
According to AspICU algorithm, probable/putative CAPA was identified in 8.5% (n=34), colonization in 2.1% (n=9), and CAPA ruled out in 89.3% (n=362).
ECMM/ISHAM criteria, probable CAPA was found in 12.3% (n=50), possible CAPA in 2.8% (n=11), and CAPA ruled out in 84.9% (n=344).
Detailed distribution of CAPA classification across COVID-19 waves is presented in Tables 6 and 7.
Table 5 summarizes the use and results of mycological investigations across the entire cohort and according to the four COVID-19 waves. Overall, bronchoalveolar (BAL) alactomannan (GM) testing was performed in 218 of 328 patients (66.5%), with an increasing testing rate over successive waves, reflecting evolving diagnostic practices. Among tested patients, serum GM positivity was observed at 14.7%, with a marked increase from the first wave (8.3%) to the fourth wave (21.7%). Median BAL GM index values also varied between waves, remaining generally low, with higher values observed during later waves.
Lower respiratory tract sampling was widely performed, with bronchoalveolar lavage (BAL) available in nearly all patients who underwent GM testing (99.0%). GM testing on sputum was rarely performed and showed very few positive results. Serum GM testing was less obtained, with a very low positivity rate overall (0.5%), reflecting its limited use and diagnostic yield in this population.
Aspergillus cultures from respiratory samples were positive in 9.8% of patients overall, with higher positivity rates observed during the third and fourth waves compared to earlier periods.
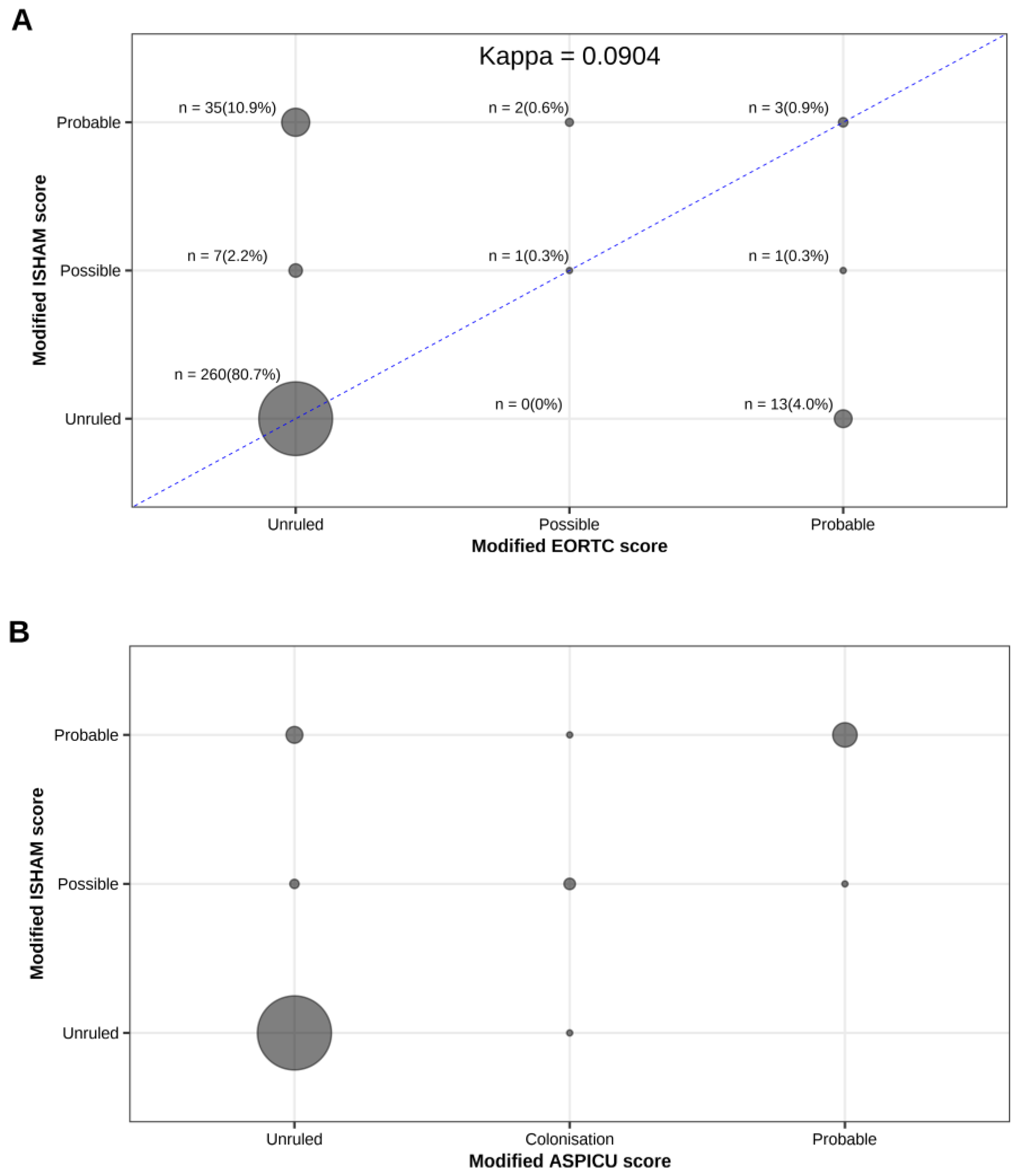
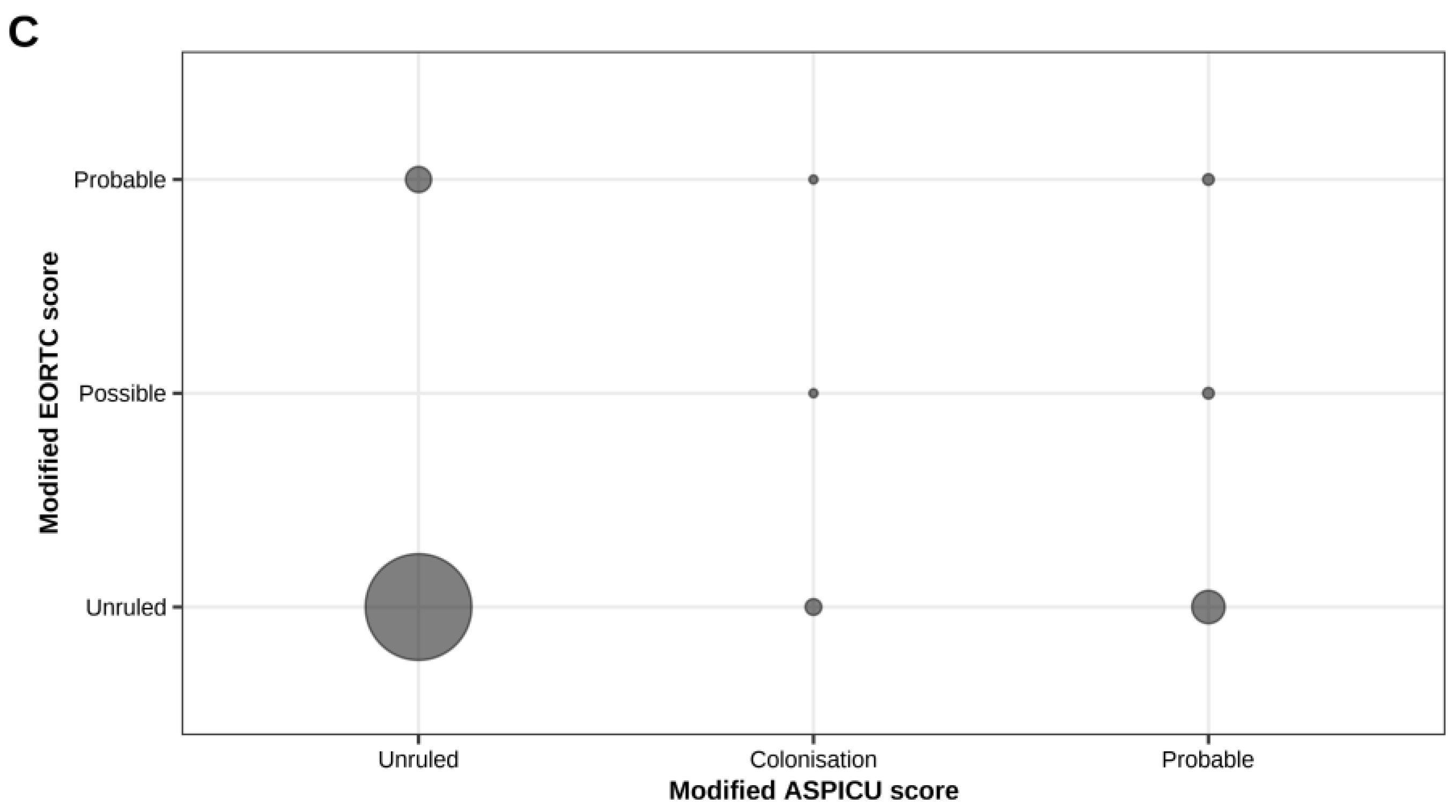
Figure 1 illustrates the concordance between the different classification systems used to diagnose CAPA, demonstrating a poor overall concordance between the three classification systems, particularly for intermediate and high-probability categories.
Mortality According to CAPA Classification
ICU mortality varied substantially according to aspergillosis diagnosis group. Patients classified as probable CAPA (Group 3) showed a mortality of approximately 76%, compared with around 43% in patients without evidence of aspergillosis (Groups 0–1).
Evolution Across COVID-19 Waves
The incidence of CAPA increased progressively over successive waves, particularly when using the ECMM/ISHAM criteria. This trend correlated with changes in COVID-19 management practices, notably the introduction and generalization of corticosteroids following the RECOVERY trial results (16). During the first wave, we observed a low incidence of CAPA, which coincided with the limited use of corticosteroids. The second wave was marked by a moderate increase in CAPA diagnoses, while the third and fourth waves showed a significant rise in probable CAPA cases, particularly according to the ECMM/ISHAM criteria.
Table 5.
diagnosis of CAPA according to EORTC, ISHAM and ASPICU criterias through waves and overall.
Table 5.
diagnosis of CAPA according to EORTC, ISHAM and ASPICU criterias through waves and overall.
| EORTC |
Overall |
Wave 1 |
Wave 2 |
Wave 3 |
Wave 4 |
| Ruled out |
304/324 (93.8%) |
23/25 (92.0%) |
98/104 (94.2%) |
132/135 (97.8%) |
51/60 (85.0%) |
| Possible aspergillosis |
16/324 (4.9%) |
2/25 (8.0%) |
6/104 (5.8%) |
2/135 (1.5%) |
6/60 (10.0%) |
| Probable aspergillosis |
4/324 (1.2%) |
0/25 (0.0%) |
0/104 (0.0%) |
1/135 (0.7%) |
3/60 (5.0%) |
| ASPICU |
|
|
|
|
|
| Ruled out |
293/328 (89.3%) |
24/25 (96.0%) |
103/105 (98.1%) |
114/135 (84.4%) |
52/63 (82.5%) |
| Colonization |
7/328 (2.1%) |
0/25 (0.0%) |
1/105 (1.0%) |
5/135 (3.7%) |
1/63 (1.6%) |
| Putative |
25/328 (7.6%) |
1/25 (4.0%) |
1/105 (1.0%) |
16/135 (11.9%) |
7/63 (11.1%) |
| Probable |
3/328 (0.9%) |
0/25 (0.0%) |
0/105 (0.0%) |
0/135 (0.0%) |
3/63 (4.8%) |
| ISHAM |
|
|
|
|
|
| Ruled out |
275/324 (84.9%) |
24/25 (96.0%) |
96/105 (91.4%) |
110/135 (81.5%) |
45/59 (76.3%) |
| Possible |
9/324 (2.8%) |
0/25 (0.0%) |
2/105 (1.9%) |
4/135 (3.0%) |
3/59 (5.1%) |
| Probable |
40/324 (12.3%) |
1/25 (4.0%) |
7/105 (6.7%) |
21/135 (15.6%) |
11/59 (18.6%) |
Table 6.
diagnosis of CAPA according to our recoding of EORTC, ISHAM and ASPICU criterias through waves and overall.
Table 6.
diagnosis of CAPA according to our recoding of EORTC, ISHAM and ASPICU criterias through waves and overall.
| Recoded EORTC |
Overall |
Wave 1 |
Wave 2 |
Wave 3 |
Wave 4 |
| 0 |
305/325 (93.8%) |
23/25 (92.0%) |
99/105 (94.3%) |
132/135 (97.8%) |
51/60 (85.0%) |
| 2 |
3/325 (0.9%) |
0/25 (0.0%) |
0/105 (0.0%) |
0/135 (0.0%) |
3/60 (5.0%) |
| 3 |
17/325 (5.2%) |
2/25 (8.0%) |
6/105 (5.7%) |
3/135 (2.2%) |
6/60 (10.0%) |
| Recoded Aspicu criterias |
|
|
|
|
|
| 0 |
293/328 (89.3%) |
24/25 (96.0%) |
103/105 (98.1%) |
114/135 (84.4%) |
52/63 (82.5%) |
| 1 |
7/328 (2.1%) |
0/25 (0.0%) |
1/105 (1.0%) |
5/135 (3.7%) |
1/63 (1.6%) |
| 3 |
28/328 (8.5%) |
1/25 (4.0%) |
1/105 (1.0%) |
16/135 (11.9%) |
10/63 (15.9%) |
| Recoded ISHAM criterias |
|
|
|
|
|
| 0 |
275/324 (84.9%) |
24/25 (96.0%) |
96/105 (91.4%) |
110/135 (81.5%) |
45/59 (76.3%) |
| 2 |
9/324 (2.8%) |
0/25 (0.0%) |
2/105 (1.9%) |
4/135 (3.0%) |
3/59 (5.1%) |
| 3 |
40/324 (12.3%) |
1/25 (4.0%) |
7/105 (6.7%) |
21/135 (15.6%) |
11/59 (18.6%) |
Figure 1.
Concordance between the different classification systems. (A) Concordance between modified EORTC and modified ISHAM criteria. Circle size is proportional to the number of patients in each cell, with absolute numbers and percentages indicated. The diagonal line represents perfect agreement. The kappa coefficient is 0.09, indicating a low level of agreement between the two classifications. (B) Concordance between modified ISHAM and modified AspICU criteria. Circle size is proportional to the number of patients per cell. Most patients were classified as “unruled”, with some overlap observed in the “probable” and “possible” categories. (C) Concordance between modified EORTC and modified AspICU criteria. Circle size reflects the number of patients per cell. Most patients clustered in the “unruled” category, with some overlap in the “probable” and “possible” categories.
Figure 1.
Concordance between the different classification systems. (A) Concordance between modified EORTC and modified ISHAM criteria. Circle size is proportional to the number of patients in each cell, with absolute numbers and percentages indicated. The diagonal line represents perfect agreement. The kappa coefficient is 0.09, indicating a low level of agreement between the two classifications. (B) Concordance between modified ISHAM and modified AspICU criteria. Circle size is proportional to the number of patients per cell. Most patients were classified as “unruled”, with some overlap observed in the “probable” and “possible” categories. (C) Concordance between modified EORTC and modified AspICU criteria. Circle size reflects the number of patients per cell. Most patients clustered in the “unruled” category, with some overlap in the “probable” and “possible” categories.
Discussion
In this large cohort of critically ill COVID-19 patients requiring mechanical ventilation, we observed that the diagnosis of COVID-19-associated pulmonary aspergillosis (CAPA) remains highly dependent on the diagnostic criteria applied. The incidence of probable or possible CAPA ranged from 6.1% to 15.1% depending on whether EORTC/MSG, AspICU, or ISHAM criteria were used.
The EORTC/MSG criteria (15) , historically developed for classical immunocompromised patients (such as those with neutropenia or hematologic malignancies), appeared too restrictive for critically ill COVID-19 patients. Many cases of CAPA likely remained unrecognized because these patients often lack the traditional host factors required for EORTC diagnosis. As a result, using EORTC criteria may lead to underdiagnosis of CAPA in ICU settings.
Conversely, the ISHAM consensus definitions (12) permitted a broader diagnosis by accepting isolated mycological criteria (such as a single positive BAL galactomannan or a positive culture) without mandatory host factors. While this approach increases sensitivity, it also risks overdiagnosing CAPA, including cases of simple Aspergillus colonization rather than true invasive disease. This highlights a major limitation of applying ISHAM definitions without integrating clinical context and additional microbiological corroboration.
The AspICU algorithm (11), although developed prior to the COVID-19 pandemic, may be more suitable for critically ill patients without classical immunosuppression. It aims to balance sensitivity and specificity by combining clinical deterioration with mycological and radiological findings (11). In our cohort, AspICU definitions tended to identify patients with a clinically meaningful infection burden, although some uncertainty remains regarding its ability to fully distinguish invasive disease from colonization in COVID-19 patients.
Some differences between classification systems come from how they define host risk factors. For example, immunosuppressed patients are considered as having probable aspergillosis under the EORTC/MSG criteria. But if these same patients don’t have any positive mycological results, they wouldn’t be classified as probable cases under the AspICU or ISHAM definitions. This explains why the same patient might be classified as Group 0 in one system and Group 3 in another.
In our pragmatic classification, colonization (Group 1) refers to cases with a positive Aspergillus culture from the respiratory tract, but without any signs of respiratory worsening. Although this group included only a small number of patients, we believe it represents a clinically meaningful distinction.
Dellière et al. (13) conducted a one-year retrospective cohort study including 176 mechanically ventilated COVID-19 patients, with 350 respiratory samples analyzed to systematically assess seven mycological criteria for CAPA. They demonstrated that isolated positive findings -such as a single BAL galactomannan or a solitary positive culture - were often poorly reproducible and only weakly correlated with clinical outcomes, whereas combining several criteria, particularly those reflecting a high fungal burden (direct microscopy, serum galactomannan, or plasma Aspergillus qPCR), offered a more robust diagnostic approach and clearer prognostic value. This work underlines the importance of integrating multiple complementary tests rather than relying on a single positive result to enhance diagnostic precision and minimize the risk of misclassifying colonization as invasive disease.
Importantly, patients classified as probable CAPA - regardless of the definition used - had significantly higher ICU mortality. This observation reinforces the clinical relevance of CAPA and suggests that at least a proportion of diagnosed cases reflect true invasive infection rather than colonization. However, these findings do not necessarily imply a causal relationship between excess mortality and CAPA diagnosis, as we know that mortality in severe COVID-19 disease is multifactorial. CAPA could increase the risk of poor outcome or represent a marker of disease severity, rather than being the direct cause of death.
Our study has several strengths, including a large cohort size, systematic application of three major diagnostic definitions, and analysis across successive COVID-19 waves. However, some limitations must be acknowledged. The retrospective, monocentric design may limit external generalizability. Serum galactomannan testing was not routinely performed outside immunocompromised patients, which might have underestimated CAPA diagnoses by ISHAM criteria. Radiological assessment was not standardized due to logistical challenges during the pandemic. Finally, our adaptation of CAPA classifications into a five-group system (from 0 to 4) was an arbitrary simplification intended to facilitate comparison across criteria. Although useful for standardization, this approach may not perfectly reflect the nuances of each original definition.
Overall, our findings highlight the urgent need for a pragmatic, harmonized approach to CAPA diagnosis, tailored to the ICU setting. Prospective validation of combined diagnostic criteria could help optimize antifungal therapy decisions and improve patient outcomes.
Conclusions
Diagnosing COVID-19-associated pulmonary aspergillosis (CAPA) in critically ill patients requiring mechanical ventilation remains a major challenge, particularly in the absence of a clear diagnostic gold standard. Our study highlights the significant variability in CAPA incidence depending on the diagnostic criteria applied. While EORTC/MSG criteria appear too restrictive for ICU patients, ISHAM definitions may lead to overdiagnosis. The AspICU algorithm offers a pragmatic alternative better suited to critically ill, non-classically immunocompromised patients. Given the high mortality associated with probable CAPA cases, future prospective studies should aim to refine CAPA definitions and integrate multiple mycological criteria to improve diagnostic accuracy.
References
- Grasselli G, Pesenti A, Cecconi M. Critical Care Utilization for the COVID-19 Outbreak in Lombardy, Italy: Early Experience and Forecast During an Emergency Response. JAMA. 2020 Apr 28;323(16):1545-1546.
- Huang SF, Ying-Jung Wu A, Shin-Jung Lee S, Huang YS, Et al. COVID-19 associated mold infections: Review of COVID-19 associated pulmonary aspergillosis and mucormycosis. J Microbiol Immunol Infect. 2023 Jun;56(3):442-454. [CrossRef]
- Rouzé A, Lemaitre E, Martin-Loeches I, et al. coVAPid study group. Invasive pulmonary aspergillosis among intubated patients with SARS-CoV-2 or influenza pneumonia: a European multicenter comparative cohort study. Crit Care. 2022 Jan 4;26(1):11. [CrossRef]
- Schauwvlieghe AFAD, Rijnders BJA, Philips N, et al. Invasive aspergillosis in patients admitted to the intensive care unit with severe influenza: a retrospective cohort study. Lancet Respir Med. 2018;6(10):782-792.
- Gangneux JP, Dannaoui E, Fekkar A, et al. Fungal infections in mechanically ventilated patients with COVID-19 during the first wave: the French multicentre MYCOVID study. Lancet Respir Med. 2022;10(2):180-190. [CrossRef]
- Prattes J, Wauters J, Giacobbe DR, et al. Risk factors and outcome of pulmonary aspergillosis in critically ill coronavirus disease 2019 patients-a multinational observational study by the European Confederation of Medical Mycology. Clin Microbiol Infect. 2022;28(4):580-587.
- Apostolopoulou A, Esquer Garrigos Z, Vijayvargiya P, Lerner AH, Farmakiotis D. Invasive Pulmonary Aspergillosis in Patients with SARS-CoV-2 Infection: A Systematic Review of the Literature. Diagnostics (Basel). 2020;10(10):807. [CrossRef]
- Verweij PE, Rijnders BJA, Brüggemann RJM, et al. Review of influenza-associated pulmonary aspergillosis in ICU patients and proposal for a case definition: an expert opinion. Intensive Care Med. 2020;46(8):1524-1535.
- Haydour Q, Hage CA, Carmona EM, et al. Diagnosis of Fungal Infections. A Systematic Review and Meta-Analysis Supporting American Thoracic Society Practice Guideline. Ann Am Thorac Soc. 2019;16(9):1179-1188. [CrossRef]
- Misset B, Piagnerelli M, Hoste E, Dardenne N, Grimaldi D, Michaux I, et al. Convalescent plasma for Covid-19–induced ARDS in mechanically ventilated patients.N Engl J Med. 2023;389. [CrossRef]
- Blot SI, Taccone FS, Van den Abeele AM, et al. A clinical algorithm to diagnose invasive pulmonary aspergillosis in critically ill patients. Am J Respir Crit Care Med. 2012;186(1):56-64. [CrossRef]
- Koehler P, Bassetti M, Chakrabarti A, et al. Defining and managing COVID-19-associated pulmonary aspergillosis: the 2020 ECMM/ISHAM consensus criteria for research and clinical guidance. Lancet Infect Dis. 2021;21(6):e149-e162.
- Dellière S, Dudoignon E, Voicu S, et al. Combination of Mycological Criteria: a Better Surrogate to Identify COVID-19-Associated Pulmonary Aspergillosis Patients and Evaluate Prognosis?. J Clin Microbiol. 2022;60(3):e0216921. [CrossRef]
- Fortarezza F, Boscolo A, Pezzuto F, et al. Proven COVID-19-associated pulmonary aspergillosis in patients with severe respiratory failure. Mycoses. 2021;64(10):1223-1229. [CrossRef]
- Bassetti M, Azoulay E, Kullberg BJ, et al. EORTC/MSGERC Definitions of Invasive Fungal Diseases: Summary of Activities of the Intensive Care Unit Working Group. Clin Infect Dis. 2021;72(Suppl 2):S121-S127. [CrossRef]
- RECOVERY Collaborative Group; Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, Staplin N, Brightling C, Ustianowski A, Elmahi E, Prudon B, Green C, Felton T, Chadwick D, Rege K, Fegan C, Chappell LC, Faust SN, Jaki T, Jeffery K, Montgomery A, Rowan K, Juszczak E, Baillie JK, Haynes R, Landray MJ. Dexamethasone in Hospitalized Patients with Covid-19. N Engl J Med. 2021 Feb 25;384(8):693-704.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).