Submitted:
03 February 2026
Posted:
06 February 2026
You are already at the latest version
Abstract
The utilization of unconventional feed resources offers a sustainable strategy to mitigate feed shortages particularly in tropical and subtropical regions where access to conventional feeds is often limited. Therefore, among these, water hyacinth (Eichhornia crassipes) has drawn attention due to its dual role as a problematic invasive weed and a potential livestock feed resource. As one of the world's most aggressive aquatic weeds, water hyacinth poses significant environmental and agricultural challenges by reducing water quality and quantity through excessive evapotranspiration, while also contributing to biodiversity loss and economic damage in farming systems. At the same time, its high capacity for nutrient-absorption makes it a viable source of protein and energy for ruminants when properly harvested and processed into forms such as hay, dried leaves, and silage. However, its utilization requires caution, as the plant can accumulate toxins and heavy metals from polluted water, which may harm animal health if unprocessed. Addressing global feed shortages particularly in the tropics during dry seasons requires innovative solutions. This review aims to synthesize the current knowledge on the potential of water hyacinth to enhance ruminant performance while contributing to sustainable weed management and improved agricultural resilience.
Keywords:
1. Introduction
2. Methodology
3. Ecology of Water Hyacinth
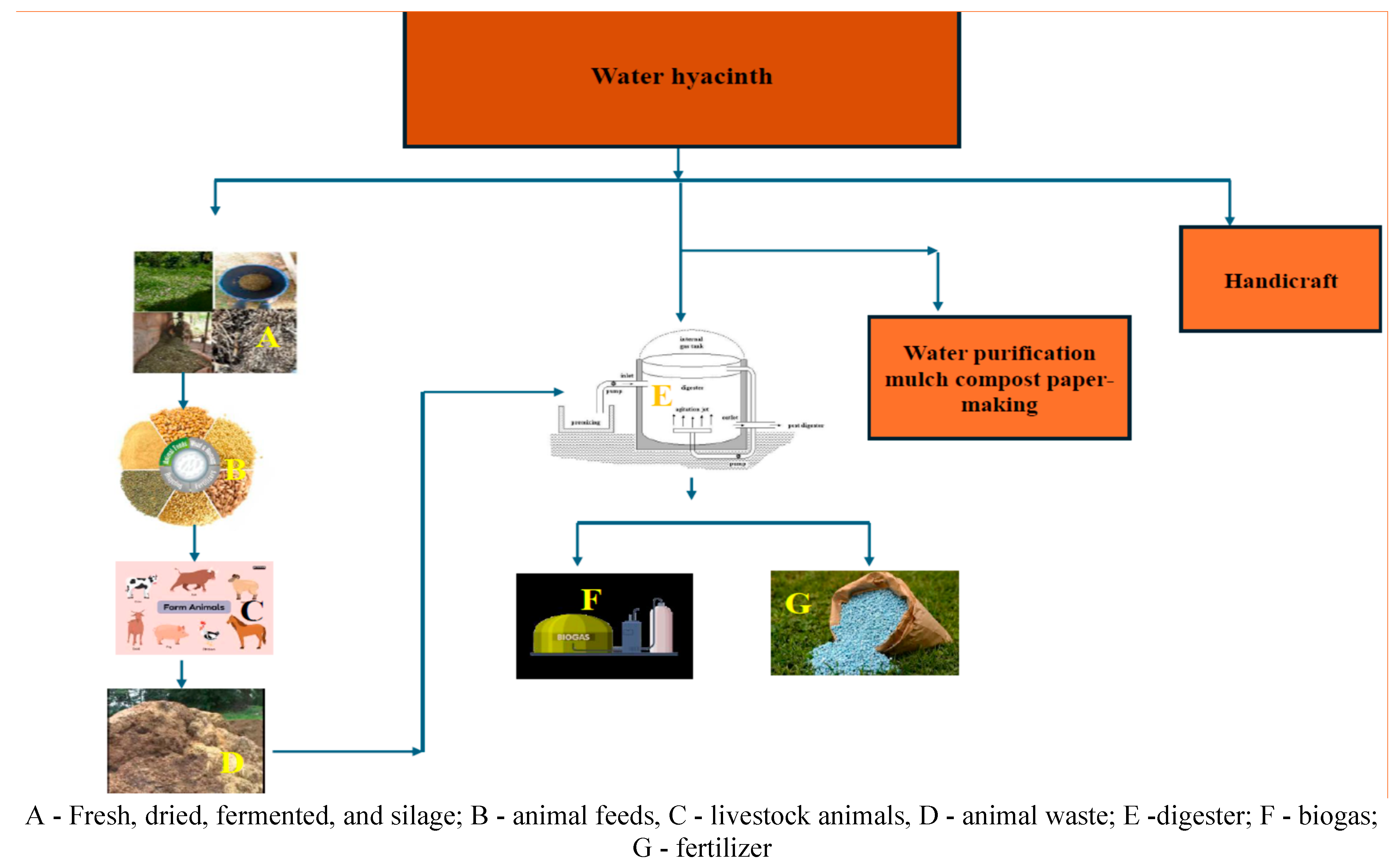
4. The Uses of Water Hyacinth
5. Nutritive Value
5.1. Proximate Composition of Water Hyacinth
| By-product | DM | CP | CF | EE | Ash | Moisture | Carbohydrates | References |
|---|---|---|---|---|---|---|---|---|
| Fermented | - | 200.4 | - | 47.6 | 105.6 | - | - | [33] |
| Dried | - | 214.4 | 376.6 | 2.3 | 111.1 | 117.0 | 178.6 | [34] |
| Dried | - | 90.4 | 579.9 | 2.2 | 180.0 | 30.0 | 117.5 | [34] |
| Forage | 186.1 | 135.1 | - | - | 33.1 | 966.9 | 284.5 | [35] |
| Dried leaves | 94.0 | 240 | 115.0 | 17.0 | 129.0 | - | - | [36] |
| Sun dried | - | 105.1 | 226.6 | 114.5 | 97.5 | - | - | [2] |
| Fermented | 915.2 | 145.6 | 259.1 | 43.5 | 191.2 | - | - | [37] |
| Silage | 84.5 | 123.4 | - | - | 146.4 | 853.6 | 355.6 | [38] |
| Dried | - | 137.3 | 96.0 | 23.7 | 66.3 | - | - | [39] |
| Fodder | 923.4 | 109.1 | - | 54.2 | 108.8 | - | 71.65 | [40] |
5.2. Amino Acid Profile of Water Hyacinth
5.3. Anti-Nutritional Factors of Water Hyacinth
6. Processing and Pre-Treatment Methods for Water Hyacinth
7. Effect of Water Hyacinth When Fed to Ruminants
7.1. Growth Performance of Ruminants
7.2. Nutrient Digestibility
7.3. Nitrogen Utilisation
7.4. Rumen Fermentation
7.5. Blood Metabolites of Ruminants
7.6. Reproduction Performance in Male Ruminants
7.7. Milk Production
7.8. Carcass Characteristics and Meat Production
8. Challenges of Using Water Hyacinth as Feed for Ruminants
9. Future Research Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yusuf, A.O.; Mlambo, V.; Iposu, S.O. A nutritional and economic evaluation of Moringa oleifera leaf meal as a dietary supplement in West African Dwarf goats. S. Afr. J. Anim. Sci. 2018, 48, 81. [Google Scholar] [CrossRef]
- Yusuf, A.O.; Owolabi, A.J.; Adebayo, K.O.; Aina, A.B.J.; Sowande, O.S.; Ajayi, T.O. Growth performance of goats fed diets containing varying levels of water hyacinth. Agric. Trop. Subtrop. 2021, 54, 228–237. [Google Scholar] [CrossRef]
- Lamidi, A.A.; Ologbose, F.I. Dry season feeds and feeding: A threat to sustainable ruminant animal production in Nigeria. J. Agric. Soc. Res. 2014, 14, 17–30. [Google Scholar]
- Su, W.; Sun, Q.; Xia, M.; Wen, Z.; Yao, Z. The resource utilization of water hyacinth (Eichhornia crassipes [Mart.] Solms) and its challenges. Res. 2018, 7, 46. [Google Scholar] [CrossRef]
- Sadique, J.; Pandey, A.; Kumar, N. Effect of molasses-fermented water hyacinth feed on growth and body composition of common carp, Cyprinus carpio. J. Entomol. Zool. Stud. 2018, 6, 1161–65. [Google Scholar]
- Hossain, M.E.; Hillol Sikder, H.S.; Kabir, M.H.; Sarma, S.M. Nutritive value of water hyacinth (Eichhornia crassipes). online J. Anim. Feed Res. 2015, 40–44. [Google Scholar]
- Tham; Ho Thanh, T.; Van Man, N.; Pauly, T. Fermentation quality of ensiled water hyacinth (Eichhornia Crassipes) as affected by additives. Asian-Australas. J. Anim Sci. 2013, 26, 195. [Google Scholar] [CrossRef]
- Gunnarsson, C.C.; Petersen, C.M. Water hyacinths as a resource in agriculture and energy production: A literature review. Waste Manag. 2007, 27, 117–29. [Google Scholar] [CrossRef]
- Valk, V.A. Valorization of water hyacinth as a renewable source of animal feed and biogas: A business case for Lake Victoria, Kenya. 2015. [Google Scholar]
- Pérez, E.A.; Coetzee, J.A.; Téllez, T.R.; Hill, M.P. A first report of water hyacinth (Eichhornia crassipes) soil seed banks in South Africa. S. Afri. J. Bot. 2011, 77, 795–800. [Google Scholar] [CrossRef]
- Patel, S. Threats, management and envisaged utilizations of aquatic weed Eichhornia crassipes: An overview. Rev. Environ. Sci. Biotechnol. 2012, 11, 249–59. [Google Scholar] [CrossRef]
- Penfound, W.T.; Earle, T.T. The biology of the water hyacinth. Ecol. Monogra 1948, 447–472. [Google Scholar] [CrossRef]
- Aquatic Ecosystem Restoration Foundation (AERF) 2005. Best Management Practices Handbook for Aquatic Plant Management in Support of Fish and Wildlife Habitat. Aquatic Ecosystem Restoration Foundation, Flint, MI.
- Downing-Kunz, M.A.; Stacey, M.T. Observations of mean and turbulent flow structure in a free-floating macrophyte root canopy. Limnol. Oceanogr. Fluids Environ. 2012, 2, 67–79. [Google Scholar] [CrossRef]
- El-Sayed, Abdel Fattah M. Effects of fermentation methods on the nutritive value of water hyacinth for Nile Tilapia Oreochromis Niloticus (L.) Fingerlings. Aquac. 2003, 218, 471–78. [Google Scholar] [CrossRef]
- Bote, M.A.; Naik, V.R.; Jagdeeshgouda, K.B. Production of biogas with aquatic weed water hyacinth and development of briquette making machine. Mater. Sci. Energy Technol. 2020, 3, 64–71. [Google Scholar] [CrossRef]
- Zarkami, R.; Esfandi, J.; Sadeghi, R. Modelling occurrence of invasive water hyacinth (Eichhornia Crassipes) in wetlands. Wetlands 2021, 41, 1–13. [Google Scholar] [CrossRef]
- Harun, I.; Pushiri, H.; Amirul-Aiman, A.J.; Zulkeflee, Z. Invasive water hyacinth: Ecology, impacts and prospects for the rural economy. Plants 2021, 10, 1613. [Google Scholar] [CrossRef]
- Bick, E.; de Lange, E.S.; Kron, C.R.; da Silva Soler, L.; Liu, J.; Nguyen, H.D. Effects of salinity and nutrients on water hyacinth and its biological control agent, Neochetina Bruchi. Hydrobiologia 2020, 847, 3213–24. [Google Scholar] [CrossRef]
- Wang, Y., 2022. The environmental impacts and high-effective solutions of invasion of water hyacinth. In IOP Conference Series: Earth. Environ. Sci. 1011, 012045.
- Villamagna, A.M.; Murphy, B.R. Ecological and socio-economic impact of invasive water hyacinth (Eichhornia crassipes): a review. Freshwater biology 2010, 55, 282–298. [Google Scholar] [CrossRef]
- Alagu, K.; Venu, H.; Jayaraman, J.; Raju, V.D.; Subramani, L.; Appavu, P.; Dhanasekar, S. Novel water hyacinth biodiesel as a potential alternative fuel for existing unmodified diesel engine: Performance, combustion and emission characteristics. Energy 2019, 179, 295–305. [Google Scholar] [CrossRef]
- Sharma, A.K.; Sharma, V.; Sharma, V.; Sharma, J.K.; Singh, R. Multifaceted potential of Eichhornia crassipes (Water Hyacinth) ladened with numerous value aided and therapeutic properties. Plant Arch. 2020, 20, 2059–2065. [Google Scholar]
- Islam, M.N.; Rahman, F.; Papri, S.A.; Faruk, M.O.; Das, A. K.; Adhikary, N.; Debrot, A.O.; Ahsan, M.N. Water hyacinth (Eichhornia crassipes (Mart.) Solms.) as an alternative raw material for the production of bio-compost and handmade paper. J. Environ. Manag. 2021, 294, 113036. [Google Scholar] [CrossRef]
- Charoensopa, K.; Ploysri, W. Making paper from water hyacinth for products and home decorations. J. Posit. Sch. Psychol. 2022, 6, 5916–5921. [Google Scholar]
- Zheng, J.C.; Liu, H.Q.; Feng, H.M.; Li, W.W.; Lam, M.H.W.; Lam, P.K.S.; Yu, H.Q. Competitive absorption of heavy metals by water hyacinth roots. Environ. Pollut. 2016, 219, 837–845. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Huang, G.; An, C.; Song, P.; Xin, X.; Yao, Y.; Zheng, R. Biophysiological and factorial analyses in the treatment of rural domestic wastewater using multi-soil-layering systems. J. Environ. Manag. 2018, 226, 83–94. [Google Scholar] [CrossRef]
- Du, Y.; Wu, Q.; Kong, D.; Shi, Y.; Huang, X.; Luo, D.; Chen, Z.; Xiao, T.; Leung, J. Y.S. Accumulation and translocation of heavy metals in water hyacinth: Maximising the use of green resources to remediate sites impacted by e-waste recycling activities. Ecol. Indic. 2020, 115, 106384. [Google Scholar] [CrossRef]
- Tham, H.T., 2012. Water Hyacinth (Eichhornia crassipes) Biomass Production, Ensilability and Feeding Value to Growing Cattle. Faculty of Veterinary Medicine and Animal Science Department of Animal Nutrition and Management Uppsala. Doctoral Thesis Swedish University of Agricultural Sciences Uppsala 2012.
- Xiao, D.; Meng, T. Nutritional value evaluation and processing technology of feed and nutrition regulation measures for ruminants. Anim. 2024, 14, 3153. [Google Scholar] [CrossRef]
- Ilo, O.P.; Simatele, M.D.; Nkomo, S.P.L.; Mkhize, N.M.; Prabhu, N.G. The benefits of water hyacinth (Eichhornia crassipes) for Southern Africa: A review. Sustain. 2020, 12, 9222. [Google Scholar] [CrossRef]
- Chaiwarit, T.; Chanabodeechalermrung, B.; Kantrong, N.; Chittasupho, C.; Jantrawut, P. Fabrication and evaluation of water hyacinth cellulose-composited hydrogel containing quercetin for topical antibacterial applications. Gels 2022, 8, 767. [Google Scholar] [CrossRef] [PubMed]
- Sayed-Lafi, R.M.; Al-Tameemi, R.A.; Gowdet, A.I. Evaluation of raw and fermented water hyacinth (Eichhornia Crassipes) incorporated diets on growth and feed efficiency of young Grass Carp (Ctenopharyngodon Idella). Basrah J. Agric. Sci. 2018, 31, 31–39. [Google Scholar] [CrossRef]
- Enyi, C.N.; Uwakwe, A.A.; Wegwu, M.O. Nutritional potentials of water hyacinth (Eichhornia crassipes). Direct Res. J. Public Health Environ. Technol. 2020, 5, 14–18. [Google Scholar]
- Tiwari, M.R.; Karki, M.; Pandey, L.N.; Poudel, N. Replacement of concentrate mixture with different levels of water hyacinth (Eichhornia Crassipes) in basal diet on feed intake and production performance of piglets. J. Agric. Nat. Res. 2020, 3, 205–17. [Google Scholar] [CrossRef]
- Rufchaei, R.; Abbas-Mohammadi, M.; Mirzajani, A.; Nedaei, S. Evaluation of the chemical compounds and antioxidant and antimicrobial activities of the leaves of Eichhornia crassipes (water hyacinth). J. Nat. Pharm. Prod. 2022, 17, 101436. [Google Scholar] [CrossRef]
- Emshaw, Y.; Getahun, A.; Geremew, A. The effect of Aspergillus niger on the nutritive value of water hyacinth as fish feed using solid state fermentation. Ethiop. J. Biol. Sci. 2021, 20, 143–154. [Google Scholar]
- Dong, N.T K.; Van Thu, N. Effects of using water hyacinth (Eichhornia crassipes L.) in the diet of Swamp Buffaloes on nutrient digestibility, rumen environment, purine derivatives, and nitrogen retention. J. Buffalo Sci. 2023, 12, 21–27. [Google Scholar] [CrossRef]
- Al-Aboudi, A.M.; Hamodi, S.J. Improving the nutritional value of water hyacinth leaves (WHL) and adding it to broiler diets and its effect on the productive performance. Iraqi J. Agric. Sci. 2023, 54, 1497–1508. [Google Scholar] [CrossRef]
- Fanta, Y.U., Kebede, Y.K., Asfaw, N.Y., 2024. Effects of feeding water hyacinth (Eichhornia crassipes) fodder with or without commercial concentrate on zoo-technical performance and profitability in tropical goats and sheep. bioRxiv, 2024-10.
- Wu, G.; Bazer, F.W.; Dai, Z.; Li, D.; Wang, J.; Wu, Z. Amino acid nutrition in animals: protein synthesis and beyond. Annu. Rev. Anim. Biosci. 2014, 2, 387–417. [Google Scholar] [CrossRef]
- Lopez, M.J.; Mohiuddin, S.S. Biochemistry, essential amino acids. In StatPearls [Internet]; StatPearls Publishing, 2024. [Google Scholar]
- Gilbreath, K.R., Bazer, F.W., Satterfield, M.C., Wu, G., 2021. Amino acid nutrition and reproductive performance in ruminants. Amino Acids in Nutrition and Health: Amino Acids in the Nutrition of Companion, Zoo and Farm Animals. Cham: Springer International Publishing. 43-61.
- Cao, Y., Yao, J., Sun, X., Liu, S., Martin, G.B., 2021. Amino acids in the nutrition and production of sheep and goats. Amino Acids in Nutrition and Health: Amino Acids in the Nutrition of Companion, Zoo and Farm Animals. 63-79.
- Adeyemi, O.; Osubor, C.C. Assessment of nutritional quality of water hyacinth leaf protein concentrate. Egypt. J. Aquat. Res. 2016, 42, 269–72. [Google Scholar] [CrossRef]
- Hontiveros, G.J.S.; Serrano, A.E. Nutritional value of water hyacinth (Eichhornia crassipes) leaf protein concentrate for aquafeeds. Aquacult. Aquarium Conserv. Legis. 2015, 8, 26–33. [Google Scholar]
- Thakur, A.; Sharma, V.; Thakur, A. An overview of anti-nutritional factors in food. Int. J. Chem. Stud. 2019, 7, 2472–2479. [Google Scholar]
- Moses, T.; Barku, V.Y.A.; Kyereme, C.; Odoi, F.N.A. Composition of water hyacinth (Eichhornia crassipes) plant harvested from the Volta lake in Ghana and its potential value as a feed ingredient in rabbit rations. Adv. Anim. Vet. Sci. 2021, 9, 230–237. [Google Scholar] [CrossRef]
- Duraiswamy, A.; Sneha A, N.M.; Jebakani K, S.; Selvaraj, S.; Pramitha J, L.; Selvaraj, R.; Kumar P, R. Genetic manipulation of anti-nutritional factors in major crops for a sustainable diet in future. Front. Plant Sci. 2023, 13, 1070398. [Google Scholar] [CrossRef] [PubMed]
- Okaiyeto, S.A., Liu, D., Zhang, C., Bai, J.W., Chen, C., Sharma, P., Venugopal, A.P., Asiamah, E., Ketemepi, H.K., Imadegbor, F.A. and Gabriel, O.T., 2025. Anti-nutrients of plant-based food: physicochemical properties, effects on health and degradation techniques-a comprehensive review. J. Future Foods.
- Suleiman, M.; Khadija, A.Y.; Nasiru, Y.; Garba, A.A.; Alhassan, M.; Bello, H. Proximate, minerals and anti-nutritional composition of water hyacinth (Eichhornia Crassipes) grass. Earthline J. Chem. Sci. 2020, 3, 51–59. [Google Scholar] [CrossRef]
- Adelakun, K.M.; Mustapha, M.K.; Muazu, M.M.; Omotayo, O.L.; Olaoye, O. Phytochemical screening and antibacterial activities of crude extract of Nymphaea lotus (water lily) against fish pathogens. J. Biomed. Sci. 2015, 2, 38–42. [Google Scholar] [CrossRef]
- Saha, S.; Ray, A.K. Evaluation of nutritive value of water hyacinth (Eichhornia crassipes) leaf meal in compound diets for rohu, Labeo rohita (Hamilton, 1822) fingerlings after fermentation with two bacterial strains isolated from fish gut. Turk. J. Fish. Aquat. Sci. 2011, 11. [Google Scholar] [CrossRef]
- Dharmawati, S.; Hartutik, H.; Sjofjan, O.; Natsir, M.H. Effect of the level freshwater snail flesh (Pomacea paludosa) Soaked in biochar in feed on the performance and digestives tract of male Alabio Ducks (Anas Plathyrhyncos Borneo). Iran. J. Appl. Anim. Sci. 2023, 13. [Google Scholar]
- Saputra, A.H.; Putri, R.A. The determination of optimum condition in water hyacinth drying process by mixed adsorption drying method and modified fly ash as an adsorbent. AIP Conference Proceedings 2017, 1840, 100005. [Google Scholar]
- Priya, P.; Nikhitha, S.O.; Anand, C.; Nath, R.D.; Krishnakumar, B. Biomethanation of water hyacinth biomass. Biores. Techn 2018, 255, 288–292. [Google Scholar] [CrossRef]
- Missotten, J.A.M.; Michiels, J.M.; Degroote, J.; De Smet, S. Fermented liquid feed for pigs: An ancient technique for the future. J. Anim. Sci. Biotech. 2015, 6, 1–9. [Google Scholar] [CrossRef]
- Mako, A.A.; Ikusika, O.O. Utilization of Pleurotus sajor-caju biodegraded water hyacinth (Eichhornia crassipes) in a solid state fermentation by West African Dwarf goats in the humid tropics. Trop. Agric. 2020, 97. [Google Scholar]
- Du, Z.; Yang, F.; Fang, J.; Yamasaki, S.; Oya, T.; Nguluve, D.; Cai, Y. Silage preparation and sustainable livestock production of natural woody plant. Front. Plant Sci. 2023, 14, 1253178. [Google Scholar] [CrossRef] [PubMed]
- Muck, R.E.; Nadeau, E.M.G.; McAllister, T.A.; Contreras-Govea, F.E.; Santos, M.C.; Kung, L. Silage review: Recent advances and future uses of silage additives. J. Dairy Sci. 2018, 100, 3980–4000. [Google Scholar] [CrossRef] [PubMed]
- Tao, H.; Si, B.; Xu, W.; Tu, Y.; Diao, Q. Effect of Broussonetia papyrifera L. silage on blood biochemical parameters, growth performance, meat amino acids and fatty acids compositions in beef cattle. Asian-Australas. J. Anim. Sci. 2020, 33, 732–741. [Google Scholar] [CrossRef]
- Isnawati, I., Ni’matuzahroh N., Surtiningsih, T., Khaleyla, F., 2021. “Effect of Fermented Water Hyacinth (Eichhornia Crassipes) on Palatability, Performance and Spermatozoa Quality on Male Sheep (Ovis Aries).” Atlantis Press 209(Ijcse):350–56.
- Tumambing, C.R.; Degoma, M.K.P.; Bacorro, T.J.; Abes, E.E.C.; Manulat, G.L.; Villar, T.D.; Angeles, A.A. Ruminal volatile fatty acids, total sugars, milk yield and quality of dairy cows fed water hyacinth [Eichhornia crassipes (Mart.) Solms] and banana (Musa sp.) pseudostem. Philipp. J. Vet. Anim. Sci. 2019, 45, 140–145. [Google Scholar]
- Chen, C. Z.; Li, P.; Wang, W.B.; Li, Z.H. Response of growth performance, serum biochemical parameters, antioxidant capacity, and digestive enzyme activity to different feeding strategies in common carp (Cyprinus carpio) under high-temperature stress. Aquac. 2022, 548, 737636. [Google Scholar] [CrossRef]
- Fitrihidajati, H.; Isnawati, E.R. Effectiveness of ruminant feed formula from the fermented water hyacinth (Eichhornia Crassipes) to produce the high level protein of goat meat. Adv. Sci. Lett. 2017, 23, 11972–75. [Google Scholar] [CrossRef]
- Amit, N.C. Effect of feeding different inclusion rates of water hyacinth [Eichhornia crassipes (Mart.) Solms] on the body weight gain of growing dairy bull calves. Int. J. Res- granthaalayah. 2019, 7, 293–298. [Google Scholar] [CrossRef]
- Yaman, M.A., Koesmara, H., Raskita, R., 2024. Production efficiency of young Acehnese cattle by giving fermented of water hyacinth (Eichhornia crassipes), palm fronds, and sago stems with addition of digestive enzymes. In IOP Conference Series: Earth. Environ. Sci. 1341, 012105.
- Patil, P.V.; Patil, M.K. Factors affecting nutrient digestibility in animals. J. Agri. 2022, 2, 1–6. [Google Scholar]
- Dong, N.T.K.; Van Thu, N. The use of water hyacinth (Eichhornia crassipes) for improving metabolizable energy intake, nutrient digestibility and economic return of local yellow cattle. Tạp chí Khoa học Công nghệ Chăn nuôi 2020, 116, 42–47. [Google Scholar]
- Fanta, Y.; Kechero, Y.; Yemane, N. Nutritional response to water hyacinth (Eichhornia crassipes) challenges via blood biochemical profiles in goats and sheep. Heliyon 2024, 10. [Google Scholar] [CrossRef]
- Mekuriaw, S.; Tegegne, F.; Tsunekawa, A.; Ichinohe, T. Effects of substituting concentrate mix with water hyacinth (Eichhornia crassipes) leaves on feed intake, digestibility and growth performance of Washera sheep fed rice straw-based diet. Trop. Anim. heal. prod. 2018, 50, 965–972. [Google Scholar] [CrossRef]
- Nguyen, V.T. Effects of water hyacinth silage in diets on feed intake, digestibility and rumen parameters of sheep (Ovis aries) in the Mekong Delta of Vietnam. J. Innov. Sustain. Dev. 2016, 02, 8–12. [Google Scholar]
- Zurak, D.; Kljak, K.; Aladrović, J. Metabolism and utilisation of non-protein nitrogen compounds in ruminants: A review. J. Cent. Eur. Agri. 2023, 24, 1–14. [Google Scholar] [CrossRef]
- Abegunde, T.O.; Akinropo, T.F. Growth performance, nutrient digestibility and haematological parameters of West African Dwarf Goats fed water hyacinth ensiled with breadfruit. J. Anim. Vet. Adv. 2018, 18, 260–269. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, G.; Li, Y.; Zhang, Y. Effects of high forage/concentrate diet on volatile fatty acid production and the microorganisms involved in VFA production in cow rumen. Anim. 2020, 10, 223. [Google Scholar] [CrossRef] [PubMed]
- Castillo-GonzálezA, R.; Burrola-Barraza, M.E.; Domínguez-Viveros, J.; Chávez-Martínez, A. Rumen microorganisms and fermentation. Arch. Med. Vet. 2014, 46, 349–361. [Google Scholar] [CrossRef]
- Beckett, L.; Gleason, C.B.; Bedford, A.; Liebe, D.; Yohe, T.T.; Hall, M.B.; White, R.R. Rumen volatile fatty acid molar proportions, rumen epithelial gene expression, and blood metabolite concentration responses to ruminally degradable starch and fiber supplies. J. Dairy Sci. 2021, 104, 8857–8869. [Google Scholar] [CrossRef]
- Bharanidharan, R.; Arokiyaraj, S.; Kim, E.B.; Lee, C.H.; Woo, Y.W.; Na, Y.; Kim, K.H. Ruminal methane emissions, metabolic, and microbial profile of Holstein steers fed forage and concentrate, separately or as a total mixed ration. PloS ONE 2018, 13, e0202446. [Google Scholar] [CrossRef]
- Ungerfeld, E.M. Metabolic hydrogen flows in rumen fermentation: principles and possibilities of interventions. Front. Microbiol. 2020, 15, 589. [Google Scholar] [CrossRef] [PubMed]
- Trung, T.T.; Nhan, P. Using silaged water hyacinth to replace elephant grass in the diet of Boer crossbred goats. Adv. Anim. Vet. Sci. 2025, 13, 1191–1199. [Google Scholar] [CrossRef]
- Mamun, M.A.; Hassan, M.M.; Shaikat, A.H.; Islam, S.A.; Hoque, M.A.; Uddin, M.; Hossain, M.B. Biochemical analysis of blood of native cattle in the hilly area of Bangladesh. Bangladesh J. Vet. Med. 2013, 11, 51–56. [Google Scholar] [CrossRef]
- Xuan, N.H.; Loc, H.T.; Ngu, N.T. Blood biochemical profiles of Brahman crossbred cattle supplemented with different protein and energy sources. Vet. World 2018, 11, 1021. [Google Scholar] [CrossRef]
- Tawose, O.M.; Oluwadele, J.F.; Ekeocha, A.H. Haematological and biochemical responses of West African Dwarf goats fed water hyacinth and Cassava peel silage. Niger. J. Anim. Prod. 2023, 50, 109–128. [Google Scholar] [CrossRef]
- Ibtisham, F.; Nawab, A.A.; Li, G.; Xiao, M.; An, L.; Naseer, G. Effect of nutrition on reproductive efficiency of dairy animals. Med. weter. 2018, 74, 356–361. [Google Scholar] [CrossRef]
- Ratnasari, E.; Fitrihidajati, H.; Isnawati, I. Improving the quality of goat sperm through the implementation of fermented feed based on water hyacinth: fermege formula 3. In Int. Conf. Sci. Technol.; Atlantis Press, 2018; pp. 101–104. [Google Scholar]
- Gorelik, O.V., Galushina, P.S., Knysh, I.V., Bobkova, E.Y., Grigoryants, I.A., 2021. March. Relationship between cow milk yield and milk quality indicators. In IOP Conference Series: Earth Environ. Sci. 677, 032013.
- Ahmedsham, M.; Amza, N.; Tamiru, M. Review on milk and milk product safety, quality assurance and control. Int. J. Livest. Prod. 2018, 9, 67–78. [Google Scholar] [CrossRef]
- Ojha, K.S.; Kerry, J.P.; Tiwari, B.K. Emerging technologies for the meat processing industry. Adv. Technol. Meat Process. 2017, 297–318. [Google Scholar]
- Hocquette, J.F.; Ellies-Oury, M.P.; Lherm, M.; Pineau, C.; Deblitz, C.; Farmer, L. Current situation and future prospects for beef production in Europe—A review. Asian-Austral. J. Anim. Sci. 2018, 31, 1017. [Google Scholar] [CrossRef]
- Warner, A., 2020. Utilization of cotton byproducts in cattle finishing diets: impacts on performance, carcass traits, and ruminal degradability of diet components. MS thesis. Oklahoma state University.
- Mani, A.M. Utilization leaf meal of water hyacinth (Eichhornia crassipes) as a replacement protein source for growing Awassi lambs. Inter. J. Vet. Sci. 2019, 8, 54–60. [Google Scholar]
- Yosef, O.M. Estimation of nutritive value and apparent digestibility factor for water hyacinth silage in Awassi Sheep rations. Jordan. J. Agri. Sci. 2021, 17, 399–410. [Google Scholar] [CrossRef]
- De Vasconcelos, G.A.; Véras, R.M.L.; de Lima Silva, J.; Cardoso, D.B.; de Castro Soares, P.; de Morais, N.N.G.; Souza, A.C. Effect of water hyacinth (Eichhornia crassipes) hay inclusion in the diets of sheep. Trop. Anim. health prod. 2016, 48, 539–544. [Google Scholar] [CrossRef] [PubMed]
- Wimalarathne, H.D.A.; Perera, P.C.D. Potential of Water Hyacinth as livestock feed in Sri Lanka. Indian. J. Weed. Sci. 2019, 51, 101–105. [Google Scholar] [CrossRef]
- Guo, Yan. Water Hyacinth: Environmental Challenges, Management and Utilization; CRC Press, 2017. [Google Scholar]
- Pandiya, R.; Jona, S.E.; Dhinakaran, S.; Dinesh, J.; Deepika, R.; Aruna, S.; Manikandavelu, D. Water hyacinth a sustainable source of feed, manure and industrial products: A review. Agric. Rev. 2023, 44, 389–392. [Google Scholar]

| Amino acid (mg/100g) | Leaf | Stem | Leaf | Leaf |
|---|---|---|---|---|
| Glycine | 4.67 | 6.07 | 3.00 | 6.51 |
| Alanine | 6.98 | 6.77 | 3.20 | 6.45 |
| Serine | 4.20 | 4.25 | 2.25 | 10.21 |
| Proline | 2.68 | 2.40 | 2.72 | 5.62 |
| Valine | 3.36 | 3.09 | 2.81 | 7.46 |
| Threonine | 4.38 | 3.22 | 2.60 | 5.27 |
| Isoleucine | 3.06 | 4.58 | 2.29 | 5.47 |
| Leucine | 7.02 | 8.08 | 5.01 | 9.56 |
| Asparagine | 8.40 | 7.59 | 4.96 | 10.21 |
| Lysine | 7.73 | 5.21 | 3.72 | 5.06 |
| Methionine | 2.09 | 1.33 | 1.34 | 1.31 |
| Glutamine | 15.13 | 11.95 | 6.04 | 7.31 |
| Phenylalanine | 4.29 | 4.53 | 3.67 | 6.01 |
| Histidine | 2.93 | 2.01 | 1.10 | 2.22 |
| Arginine | 5.25 | 8.06 | 3.80 | 6.58 |
| Tyrosine | 2.20 | 2.33 | 2.20 | 2.92 |
| Cysteine | 1.78 | 2.31 | 0.72 | 0.38 |
| References | [35] | [35] | [45] | [46] |
| ANF | Effect | Presence in WH | References |
|---|---|---|---|
| Oxalates | Bind calcium, may cause kidney stones and reduce mineral bio-availability | High | [47,48] |
| Tannins | Reduce protein digestibility by binding proteins and inhibiting digestive enzymes | Moderate to high | [51] |
| Phytates (Phytic Acid) | Bind essential minerals such as iron, zinc, calcium, to reduce their absorption | Moderate | [52,53] |
| Saponins | Interfere with nutrient absorption and may have toxic effects at high levels | Present in small amounts | [54] |
| Alkaloids | Some may be toxic or interfere with metabolism | Trace | [47] |
| Cyanide | Can be toxic if consumed in high concentrations | Present in small amounts | [48,49] |
| Species | CP (%) | OM (%) | DM (%) | NDF (%) | References |
|---|---|---|---|---|---|
| Sheep | 78.4 | 68.8 | 66.4 | 66.1 | [72] |
| Sheep | 79.2 | 73.2 | 72.8 | 71.8 | [71] |
| Cattle | 67.3 | 65.4 | 63 | 64.3 | [69] |
| Buffaloes | 71.6 | 73.4 | 72.4 | 72.7 | [38] |
| Goat | 82.72 | 76.97 | 75.32 | 73.61 | [70] |
| Sheep | 75.32 | 70.40 | 68.80 | 71.42 | [70] |
| Ruminal parameters | ||||||
|---|---|---|---|---|---|---|
| Animal | WH | Inclusion (%) | pH | N-NH3 (mg/100g) | VFAs (mmol) | References |
| Goats | Silage | 25, 50 & 75 | 7.05 - 7.50 | 23.1 - 27.7 | 73.5 - 85.2 | [80] |
| Cattle | Fresh | 0, 25, 50 & 75 | 7.03 -7.10 | 20.8 - 22.8 | 84.6 - 91.0 | [69] |
| Sheep | Silage | 0, 15, 30 & 45 | 6.87 - 7.08 | 34.6 - 38.5 | 108 - 115 | [72] |
| Goats | Fungal-treated | 0, 30, 45, 60 & 90 | 6.15 - 6.31 | 20.1 - 25.6 | 55.2 - 76.1 | [58] |
| Buffaloes | Fresh | 25, 50, 75 & 100 | 6.95 -7.05 | 17.5 - 25.2 | 74.2 - 87.2 | [38] |
| Species | Inclusions levels (%) | Processing | Response | References |
|---|---|---|---|---|
| Sheep | 0, 50, 75 & 100 | Wilted | Wilted WH leaves substitute concentrate mix up to 75% results in the optimum growth of Washera sheep. | [71] |
| Sheep | 0, 10, 20 & 30 | Fermented | 20% fermented WM and corncob have the highest palatability and WG in male sheep. Sheep fed 30% fermented WH and corncob produced spermatozoa with the highest quality. |
[62] |
| Sheep | 0, 25, 50 & 75 | Silage | 25% replacement of WH silage had a positive effect on TDE, DE and ME. | [92] |
|
Sheep and goats |
0, 12.5, 25 & 37.5 |
Fodder |
Sheep had a high FI, and goats had a low FI. 37.5% replacement with WH enhances nutritive intake, digestibility, and body weight gain. |
70] |
|
Sheep |
0, 20, 40, 60 & 80 |
Hay |
WH hay decreased the amount of certain nutrients that the sheep could consume and digest. |
[93] |
|
Awassi Lamb |
0, 5, 10 & 20 |
Fodder |
WH leaf substitute concentrate mix up to 10% has optimum growth of Awassi sheep. |
[91] |
|
Dwarf Goats |
0, 15, 30, 45 & 60 |
Silage |
The optimal level of WH supplementation for West African Dwarf goats was determined to be 15%, as this level resulted in the best hematology and biochemical indices, with no negative impact on their blood indices. |
[83] |
|
West African dwarf goats |
0, 5, 10 & 15 |
Sun dried |
Feeding West African dwarf goats with WH up to 10% in their diets had favourable effects on growth performance, health, and possibly immune response as well as profitability. |
[2] |
|
Goats |
0, 20, 30 & 40 |
Forage |
Highest body weight gain observed at 20% inclusion levels. The study suggested that WH can be included in the goat diet only up to 40% without any adverse effects on growth performance. |
[35] |
| Goats | 0, 50, 60, 70, 80 & 90 | Chopped and Silage | 60% WH diets ensiled with 30% breadfruit improved DWG and FCR. | [74] |
|
Swamp buffaloes |
0, 25, 50, 75 & 100 |
Silage |
WH can replace para grass in the buffalo diet up to 100%. 50% of WH replacement improves utilization of nutrients and energy, lowers feed cost, and improves the environment. |
[38] |
|
Dairy bull calves |
0, 10, 20 & 40 |
40% WH inclusion increased intake in calves but did not have any effect on the BWG of the calves. A WH inclusion rate of 10 - 20% DM basis can substitute for Napier grass. |
[66] |
|
|
Young Acehnese cattle |
50 |
Fermented |
Fermented WH, palm fronds, and sago stems improve average consumption, feed cost, and daily body weight gain. |
[67] |
|
Local yellow cattle |
0, 25, 50 & 75 |
Fresh |
Fresh WH improves dietary nutrient digestibility, metabolizable energy, and positive live weight change. The optimum level of WH replacement to rice straw in cattle diet is 50%. |
[69] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




