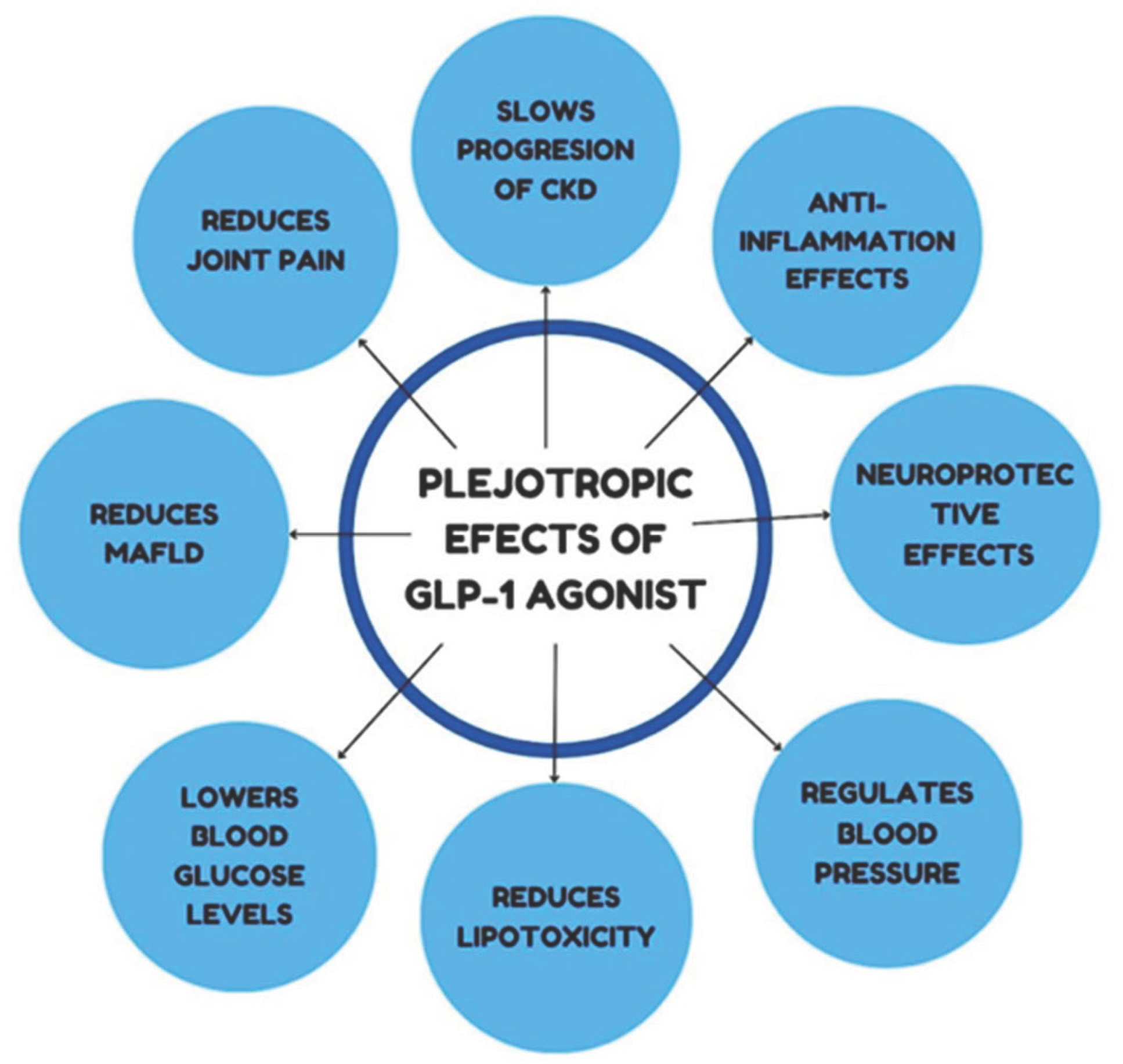
1. Introduction
GLP-1 receptor agonists (GLP-1 RAs) constitute a class of medications originally developed for the treatment of type 2 diabetes mellitus (T2DM). The first drug in this group, exenatide, was approved by the U.S. Food and Drug Administration (FDA) in April 2005. It was derived from a naturally occurring peptide found in the saliva of the Gila monster (Heloderma suspectum), which exhibits properties similar to those of endogenous human GLP-1 [
1,
2], and required twice-daily subcutaneous administration. Subsequently, longer-acting and more effective formulations were developed, including liraglutide (approved in 2009), followed by once-weekly agents such as dulaglutide (2014), semaglutide (2017), and tirzepatide (2023) [
3]. Most GLP-1 RAs are administered subcutaneously, with the notable exception of semaglutide, which is also available in an oral formulation. Currently, GLP-1 RAs are approved for the treatment of T2DM and obesity. However, during their clinical use, numerous non-metabolic effects have been observed, including delayed progression of chronic kidney disease (CKD), reduction in proteinuria [
4], lowered blood pressure, improved lipid profiles, and decreased risk of major adverse cardiovascular events (MACE). These pleiotropic properties have sparked considerable interest in their potential therapeutic applications beyond diabetes and obesity. One notable investigation in this area is the SELECT trial, a large international clinical study evaluating the effect of semaglutide on MACE incidence in individuals with overweight or obesity and established cardiovascular disease, but without diabetes. The trial demonstrated a reduction in MACE risk from 8.0% to 6.5%. It enrolled 17,604 patients with a median follow-up of approximately 40 months. Importantly, the reduction in cardiovascular risk was evident shortly after treatment initiation, suggesting that semaglutide’s benefits may extend beyond weight loss alone. The favorable cardiovascular effects of GLP-1 RAs were also confirmed in the randomized SUMMIT trial, which assessed tirzepatide in patients with obesity and heart failure with preserved ejection fraction (HFpEF). The results showed a 38% reduction in the risk of cardiovascular death or worsening heart failure, and a 56% reduction in hospitalizations due to heart failure compared to placebo [
3,
5]. GLP-1 analogs have also been shown to exert broader pharmacological effects, including anti-inflammatory, neuroprotective, antihypertensive, and lipotoxicity-reducing properties. These effects are relevant to both physiological and pathological processes implicated in Alzheimer’s disease (AD), hypertension, and metabolic dysfunction-associated steatohepatitis (MASH). Although the underlying mechanisms remain incompletely understood, further elucidation may facilitate the identification of novel therapeutic targets for AD, hypertension, and MASH [
6].
The aim of this paper is to present the current state of knowledge regarding the pleiotropic effects of GLP-1 receptor agonists and to explore their potential applications beyond glycemic control and weight management. Expanding the therapeutic use of these agents may represent a significant advancement in the treatment and prevention of various diseases, particularly among patients with metabolic syndrome.
2. Materials and Methods
The study presents an analysis of current literature data on the use of GLP-1 RAs, with particular emphasis on their pleiotropic effects. It also discusses newly identified mechanisms of action and potential future therapeutic targets for this drug class.
3. Results and Discussion
3.1. Mechanism of Action and Therapeutic Effects of GLP-1 RAs
Nowadays we know perfectly well that GLP-1 is an incretin hormone secreted by L cells of the small intestine. GLP-1 is an incretin hormone secreted by L cells of the small intestine. It exerts its effects by binding to the GLP-1 receptor (GLP-1R) and is subsequently inactivated by the enzyme dipeptidyl peptidase-4 (DPP-4) [
6]. Following food intake, GLP-1 activates receptors on pancreatic β-cells, leading to an increase in intracellular cAMP levels, which enhances insulin secretion and inhibits glucagon release from α-cells. GLP-1 receptor agonists (GLP-1 RAs) also delay gastric emptying, thereby attenuating postprandial glucose excursions. Moreover, they act on hypothalamic centers regulating appetite and satiety, resulting in reduced food intake and promoting weight loss. However, GLP-RAs have much more to offer as we will outline below.
3.2. Pleiotropic Effects of GLP-1 Receptor Agonists
Figure 1.
Pleiotropic Effects of GLP-1 Receptor Agonists.
Figure 1.
Pleiotropic Effects of GLP-1 Receptor Agonists.
3.2.1. Alcohol
According to the 2022 data from the
National Survey on Drug Use and Health, alcohol remains the most commonly used psychoactive substance in the United States. Alcohol Use Disorder (AUD) is a chronic condition associated with significant medical, social, and psychological consequences [
7]. Therefore, the search for new therapeutic strategies remains ongoing. GLP-1 receptor agonists (GLP-1 RAs) have attracted increasing attention from the scientific community, particularly due to their effects on the central nervous system and their potential application in addiction treatment.
As described by Klausen et al. (2022), GLP-1 plays an important role in modulating the activity of the brain’s reward system, which may represent a novel therapeutic target for substance use disorders.
The reward system comprises a network of neural structures responsible for motivation and behavior regulation, activated in response to satisfying needs and receiving pleasurable stimuli. Psychoactive substances such as alcohol, opioids, and amphetamines strongly stimulate this system, contributing to the development of addiction [
8].
Tirzepatide, a dual agonist of the GLP-1 and GIP receptors, influences central nervous system pathways involved in mood regulation, reward processing, and addictive behavior. The GLP-1 receptor plays a key role in the functioning of the reward system, and its activation by tirzepatide may modulate dopaminergic signaling, which is critical in the rewarding effects of food and alcohol [
9].
Preclinical studies conducted by Professor Elisabet Jerlhag’s team at the University of Gothenburg demonstrated that GLP-1 RAs reduce alcohol intake in rats. This effect is achieved, in part, through inhibition of dopamine release in the nucleus accumbens—a structure central to the brain’s reward circuitry. Experiments with semaglutide confirmed these findings, showing both reduced alcohol consumption and prevention of relapse [
10,
11].
A randomized clinical trial led by Christian S. Hendershot evaluated the effect of once-weekly subcutaneous semaglutide administration on alcohol intake in individuals with AUD. The study enrolled 48 participants—34 women and 14 men—with a mean age of 39.9 years. Most participants had a body mass index (BMI) exceeding 30. The results demonstrated that semaglutide treatment led to a reduction in alcohol use, both in terms of total alcohol consumption and peak breath alcohol concentration [
7].
Despite these promising findings from both preclinical and early clinical studies, the efficacy of GLP-1 RAs in treating AUD requires further investigation. Large-scale, randomized, multicenter clinical trials involving more diverse patient populations are necessary to validate these results.
3.2.2. Nicotine
Cigarette smoking remains the leading preventable cause of premature death. Despite numerous support programs, smoking cessation rates remain low. One of the key barriers is the occurrence of nicotine withdrawal symptoms and the associated weight gain following smoking cessation. This unfavorable metabolic consequence is a significant deterrent for many smokers attempting to quit or maintain abstinence. Weight gain is particularly prominent during the first months of cessation, with an average increase of approximately 2.3 kg within the first two months [
12].
GLP-1 receptor agonists (GLP-1 RAs) reduce caloric intake and body weight and have demonstrated potential in modulating addictive behaviors through their impact on the central reward system. They regulate synaptic dopamine availability and expression of the dopamine transporter (DAT), which plays a crucial role in motivation and reward-driven behaviors. Their influence on nicotine dependence also appears to involve the interpeduncular nucleus, a brain region implicated in nicotine withdrawal and craving [
13].
Preclinical studies have shown that GLP-1 RAs reduce nicotine self-administration and nicotine-seeking behavior in animal models. Notably, in some cases, these effects persisted even after discontinuation of substance exposure [
12].
In an observational study involving 222,942 patients with type 2 diabetes treated with various antidiabetic medications (including insulin, metformin, DPP-4 inhibitors, SGLT-2 inhibitors, sulfonylureas, thiazolidinediones, and GLP-1 RAs—among them 5,967 semaglutide users), semaglutide use was associated with a 32% lower risk of tobacco use disorder compared to insulin. Patients receiving semaglutide also showed reduced need for pharmacological smoking cessation aids and less frequent use of smoking cessation counseling [
14].
GLP-1 RAs may represent a novel therapeutic avenue to complement standard nicotine replacement therapies in individuals with nicotine dependence. These agents may not only assist in addressing the addiction itself but also mitigate cessation-related complications such as nicotine craving, withdrawal symptoms, weight gain, hyperphagia, and other metabolic disturbances.
3.2.3. Substance Use Disorders – Cannabinoids and Cocaine
Substance use disorders (SUDs) represent an escalating public health challenge both nationally and globally. In the United States, there is a steady increase in the prevalence of drug dependence, as reflected in epidemiological data, as well as a rise in drug-related hospitalizations and overdose deaths [
15,
16]. In recent years, an increased share of juvenile offenses has been attributed to drug-related criminal activity.
According to the 2019 European School Survey Project on Alcohol and Other Drugs (ESPAD), Polish adolescents ranked among the top four in Europe in the use of new psychoactive substances (NPS, commonly referred to as "legal highs") and were the leading group in amphetamine use [
17]. Drug addiction is a debilitating and chronic condition characterized by a recurrent cycle of intoxication, craving, withdrawal, and relapse. Regardless of the substance involved, addiction leads to the development of neuroanatomical abnormalities accompanied by functional brain changes and behavioral disturbances [
18].
Cocaine exerts its psychoactive effects primarily through activation of the mesolimbic dopaminergic pathway. Dopaminergic projections from the ventral tegmental area (VTA) to forebrain structures, including the nucleus accumbens (NAc), play a central role in cocaine reinforcement and the development of dependence [
19]. Cocaine increases dopamine levels and prevents its reuptake in the brain, which over time disrupts neural receptor function [
20].
GLP-1 receptor agonists (GLP-1 RAs) may reduce cocaine dependence by acting on several brain regions involved in addiction processes, such as the lateral septum, dorsolateral and ventral tegmental areas, nucleus of the solitary tract, and nucleus accumbens [
16].
In a mouse study conducted by Emil Egecioglu et al., systemic administration of exenatide inhibited cocaine uptake and expression in the central nervous system, leading to a reduction in cocaine-induced locomotor stimulation. A similar effect was observed in response to amphetamine administration [
21].
Further preclinical studies by Chaves Filho et al. demonstrated potential antimanic or mood-stabilizing effects of liraglutide in D-amphetamine-induced mania models in mice. Liraglutide mitigated amphetamine-related cognitive deficits in certain behavioral tasks, effectively counteracting hyperlocomotion and impairments in spatial and executive memory [
22].
In an observational study involving approximately 700,000 individuals, patients who initiated semaglutide therapy were found to have a 40% lower risk of being diagnosed with cannabis use disorder.
There is also growing speculation that GLP-1 RAs may reduce various forms of compulsive consumption—such as compulsive shopping or gambling—through their influence on the reward system. However, further research is required to clarify these potential effects [
23].
3.2.4. Opioids
The misuse of opioid analgesics has become a pressing contemporary social and medical issue. This problem is fueled by inappropriate use of opioids—either outside the framework of the WHO analgesic ladder or for recreational purposes—due to their euphoric properties. With the aging population steadily increasing, there is a growing susceptibility to chronic pain syndromes, often associated with multiple comorbidities. Chronic pain not only reduces quality of life but also poses a therapeutic challenge for healthcare providers [
24].
In many cases, opioid therapy remains the only viable option for improving patients' daily functioning and overall well-being. However, frequent opioid use in older adults—often in the treatment of nonspecific pain—has contributed to the rise in opioid overuse within this population. The use of GLP-1 receptor agonists (GLP-1 RAs), due to their activity in the nucleus accumbens and related brain regions, has been associated with a reduction in opioid dependence, including oxycodone [
25].
In a randomized controlled trial evaluating GLP-1 RAs for opioid use disorder (OUD), participants receiving low-dose liraglutide reported a 40% reduction in opioid craving over a 3-week period. However, over half of the study participants discontinued participation before completion, significantly limiting the study’s overall validity.
An observational study involving 33,000 patients with type 2 diabetes showed that treatment with semaglutide was associated with a nearly 50% lower risk of opioid overdose compared to other antidiabetic medications over a one-year follow-up period [
26].
A recent literature review of both preclinical and clinical studies further supports that GLP-1 RAs may reduce the risk of opioid overdose, opioid-seeking behaviors, and opioid self-administration. Multiple clinical trials aimed at evaluating their use in the treatment of opioid use disorder are currently underway, with results expected by 2027 [
25].
3.2.5. Mental Health – Depression and Suicide
Depressive disorders are among the most prevalent psychiatric conditions, characterized by diverse clinical presentations and a profound impact on psychosocial functioning. According to the World Health Organization (WHO), depression is one of the leading causes of global disability, both mental and physical.
Psychological pain is an integral component of depressive disorders and is strongly linked to an increased risk of suicidal ideation and suicidal behavior [
27]. Individuals with depression exhibit increased sensitivity to both psychological and physical pain [
28]. In recent years, there has been growing interest in the potential psychotropic effects of GLP-1 RAs on mental health.
Initially, pharmacovigilance reports involving liraglutide and semaglutide raised concerns about a possible association with increased suicidal ideation and self-injurious behavior. However, recently published clinical data indicate no causal relationship between GLP-1 RA use and suicidal behavior in individuals with psychiatric conditions [
29].
A large-scale retrospective study conducted by Jianxing Zhou et al. (2018–2022) on 93,431 clinical cases, including 204 involving suicidal and self-injurious behavior (SSIB), found no significant association between GLP-1 RA treatment and the occurrence of SSIB [
30].
Furthermore, preclinical studies in animal models have demonstrated antidepressant-like effects of GLP-1 RAs, including the reversal of depressive-like behaviors induced by chronic stress and inflammation [
31].
A meta-analysis by Xinda Chen et al., including over 2,000 participants across five randomized controlled trials and one prospective cohort study, showed that adults treated with GLP-1 RAs experienced significantly fewer depressive symptoms compared to those treated with other antidiabetic medications. These findings suggest that GLP-1 RAs may offer therapeutic benefit in alleviating depressive symptoms, particularly among patients treated for obesity [
32,
33].
Moreover, both preclinical and early human studies suggest an anxiolytic potential of GLP-1 RAs. The hypothesized mechanisms include modulation of serotonin and GABAergic neurotransmission, enhancement of neuroplasticity, and anti-inflammatory properties [
34].
3.2.6. Kidneys
Type 2 diabetes (T2DM) significantly increases the risk of developing cardiovascular diseases and diabetic nephropathy, which can progress to chronic kidney disease (CKD). A study by Vlado Perkovic's team demonstrated that semaglutide significantly reduces the risk of major kidney function decline and cardiovascular death in patients with T2DM and CKD [
35].
Authors of studies analyzing the renal effects of GLP-1 receptor agonists (GLP-1 RAs) in patients with diabetes point to genetic evidence supporting the nephroprotective role of these agents in both diabetic nephropathy and IgA nephropathy. Inflammation plays a central role in the pathogenesis of IgA nephropathy, and the anti-inflammatory properties of GLP-1 RAs may form the basis of their therapeutic potential [
36]. However, clinical trials are needed to confirm their efficacy and safety in this setting [
37].
GLP-1 RAs also activate AMP-activated protein kinase (AMPK), a key enzyme regulating cellular energy metabolism, including in cardiac muscle. This mechanism may contribute to their cardiovascular protective effects.
Meta-analyses of randomized controlled trials (RCTs) have shown that GLP-1 RAs reduce the risk and progression of CKD. While the exact mechanism remains unclear, these effects are likely mediated by AMPK activation and testosterone suppression, the latter of which has also been implicated in prostate cancer development.
3.2.7. Prostate
Prostate cancer (PCa) is the second most commonly diagnosed malignancy and a leading cause of cancer-related mortality among men in the United States. Insulin metabolism may be a key link between PCa, obesity, and metabolic disorders. One of the molecular factors identified in this signaling cascade is the FOXC2 (forkhead box C2) transcription factor, which may act as a connector between PCa, metabolic syndrome, and obesity. FOXC2 is known to suppress genes involved in insulin resistance while promoting proliferation of prostate cancer cells.
The interaction between insulin resistance and prostate cancer remains insufficiently understood. Insulin plays a critical role in the crosstalk between adipocytes and tumor cells. Insulin resistance has been shown to stimulate PCa proliferation and contribute to adipocyte remodeling [
38]. The tumor microenvironment, including surrounding adipose tissue, is essential in this communication, involving secreted molecules and overexpressed genes that promote cancer progression [
39].
Adiponectin levels are reduced in the presence of surrounding adipocytes, increasing the risk of PCa progression [
40]. Various adipokines secreted from adipose tissue modulate tumor growth, and hypoxic conditions in the tumor microenvironment have been linked to increased metastatic potential of PCa cells.
FOXC2, an effector of the EGFR signaling pathway, plays a key role in energy metabolism, cell growth, and differentiation in obesity and prostate cancer. In obesity, elevated leptin and reduced adiponectin levels promote chronic low-grade inflammation via pro-inflammatory cytokines. When adipose tissue becomes insulin-resistant, EGFR and IGF-1 receptors are activated, triggering overexpression of pro-tumorigenic factors. Insulin and TNF-α are known to induce FOXC2 in 3T3-L1 adipocytes. Meanwhile, free IGF-1 and its binding proteins (IGFBP-1 and IGFBP-3) exert opposing antitumor effects [
41].
Although the direct or indirect role of IGF signaling in FOXC2 regulation and PCa development is not yet fully established, existing evidence supports the need for further studies to better understand these interactions. Lifestyle, diet, and genetic predispositions also play significant roles in the pathophysiology of metabolic disorders and PCa.
There is growing evidence that GLP-1 RAs reduce prostate cancer risk. In the LEADER trial, patients treated with liraglutide had a lower incidence of prostate cancer compared to placebo [
42]. Similarly, a large-scale cohort study involving 1.1 million individuals with obesity demonstrated a reduction in prostate cancer risk among users of GLP-1 Ras [
43]. This association has also been noted for gastrointestinal, skin, breast, and hematopoietic malignancies, most of which are known to be obesity-related.
Moreover, GLP-1 RAs were found to significantly reduce all-cause mortality in cancer survivors [
44]. Notably, in patients with prostate cancer, GLP-1 RAs may serve as adjuvants to standard treatments, potentially enhancing the effectiveness of radiotherapy, hormone therapy, and chemotherapy [
45].
3.2.8. Neuroprotection
Alzheimer’s disease (AD) is the most common neurodegenerative disorder and represents a significant global health challenge. Its progressive cognitive decline markedly reduces patients' quality of life, ultimately leading to disability and premature death [
46,
47].
From an epidemiological and pathophysiological perspective, AD and type 2 diabetes mellitus (T2DM) share numerous similarities and are sometimes referred to as “sister diseases” [
8,
9,
48]. T2DM increases the risk of developing AD, while neurodegenerative processes impair systemic glucose metabolism. Consequently, AD has been termed “type 3 diabetes” [
9].
Given the underlying mechanisms and etiology of AD, there is growing interest in the potential neuroprotective effects of GLP-1 receptor agonists (GLP-1 RAs) [
49]. This hypothesis is supported by studies from teams at Harbin Medical University and Huazhong University of Science and Technology, which demonstrated that GLP-1 RAs exert anti-inflammatory effects in the central nervous system and reduce microglial activation. They also decrease pathological deposition of β-amyloid and tau proteins, potentially by enhancing autophagy and clearance of damaged cellular structures. Additionally, GLP-1 RAs promote long-term synaptic potentiation and cognitive functions and reduce oxidative stress by lowering free radical production and increasing antioxidant enzyme activity [
46].
These findings are corroborated by a large cohort study conducted by researchers at Tulane University (Siddeeque et al., 2024), which found that semaglutide use was associated with a 37% reduction in the risk of developing AD and over 50% reduction in all-cause mortality. Furthermore, GLP-1 RAs reduced the risk of Lewy body dementia and vascular dementia by 41% and 56%, respectively [
50]. This study analyzed data from 5,307,845 obese adult patients across 73 healthcare organizations in 17 countries, forming two cohorts of 102,935 patients each.
Regarding Parkinson’s disease (PD), clinical studies have yielded inconclusive results. In preclinical models, tirzepatide significantly reduced neuroinflammation and oxidative stress in PD mice, increased striatal dopamine levels, and promoted mitochondrial homeostasis [
51]. GLP-1 RAs have been shown to promote survival of dopaminergic neurons by inhibiting apoptosis and inflammation, as well as resensitizing brain insulin signaling, thus targeting the metabolic impairments underlying PD.
Although Siddeeque et al. observed a trend toward risk reduction, it did not reach statistical significance [
50]. Moreover, a systematic review and meta-analysis by researchers from Universidade do Estado do Amazonas and collaborators (Nogueira et al., 2024) found no improvement in any part of the MDS-UPDRS scale (I–IV) after 6 and 12 months of GLP-1 RA treatment. No significant changes in quality of life (PDQ-39) were observed, and treatment was associated with increased gastrointestinal adverse events and weight loss [
52]. Similarly, the latest meta-analysis assessing GLP-1 RAs' impact on key clinical outcomes in PD showed no significant improvements in MDS-UPDRS Parts II and IV [
53].
Studies investigating GLP-1's role in amyotrophic lateral sclerosis (ALS) pathogenesis and progression showed elevated GLP-1 levels in ALS patients compared to healthy controls, although the correlation with the ALS functional rating scale (ALSFRS-R) was insignificant [
54].
Despite the undeniable neuroprotective role of GLP-1 RAs through restoration of neurite outgrowth, increased neurotrophic factors, and reinforcement of the blood-brain barrier, the divergent results across various neurodegenerative disorders highlight the need for further research to clarify their potential therapeutic role in these conditions.
3.2.9. Fatty Liver Disease
Metabolic dysfunction-associated steatotic liver disease (MASLD), characterized by excessive fat accumulation within hepatocytes, can contribute to the development of cirrhosis and liver failure [
55,
56]. GLP-1 receptor agonists (GLP-1 RAs) reduce liver fat content and liver enzyme levels, decrease inflammation, oxidative stress, and liver fibrosis, improve hepatic de novo lipogenesis, thereby enhancing liver function [
57,
58]. It is believed that weight loss may contribute to these effects, although the exact mechanisms remain unclear. Endogenous glucose-induced GLP-1 secretion is reduced in patients with MASLD and metabolic dysfunction-associated steatohepatitis (MASH), which may represent another starting point for research into MASLD pathophysiology [
58,
59].
One study compared exenatide at a dose of 10 μg twice daily to insulin boluses in 60 patients with obesity, type 2 diabetes, and MASLD with elevated liver enzymes. After 12 weeks, levels of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and γ-glutamyltransferase were lower in the exenatide group compared to insulin [
57]. In another randomized clinical trial, retatrutide administered in advanced MASLD eliminated 86% of liver fat in patients.
Clinical trials of GLP-1 RAs have demonstrated various potential benefits in patients with MASH and MASLD, including resolution of steatohepatitis, reduction in liver stiffness, and fibrosis progression. The impact of GLP-1 RAs on the spectrum of fibrosis severity in MASH represents a crucial area for future research. Currently, trials of retatrutide in individuals with diabetes and obesity or overweight (TRIUMPH-2, NCT05929079) are ongoing, with results expected in 2026 Studies on a long-acting triple agonist of GLP-1/glucagon/GIP, HM15211 (efocipegtrutide), are in phase 2 for MASH (NCT04505436), with outcomes anticipated in 2025 [
57,
58].
GLP-1 is also significantly related to metabolic dysfunction-associated steatohepatitis (MASH), characterized by liver inflammation and hepatocyte injury. MASH is currently recognized as a leading cause of the rising incidence of hepatocellular carcinoma (HCC) [
50], although its molecular mechanisms are complex and multifactorial [
52]. Furthermore, GLP-1 analogues have been shown to effectively reduce body weight and liver fat content in patients with MASH [
57].
3.2.10. Hepatocellular Carcinoma
Liver cancer ranks as the sixth most common cancer worldwide. Hepatocellular carcinoma (HCC) accounts for approximately 72% of primary liver cancers [
32]. HCC primarily develops in patients with post-inflammatory liver cirrhosis (80–90%), associated with HBV or HCV infections and their coexistence. Modifiable risk factors such as type 2 diabetes, obesity, alcohol and tobacco abuse, metabolic dysfunction-associated steatotic liver disease (MASLD), and metabolic dysfunction-associated steatohepatitis (MASH) also influence HCC development.
A cohort study of 1,890,020 patients with type 2 diabetes demonstrated that GLP-1 RAs were associated with a lower risk of HCC compared to other antidiabetic medications (insulin, metformin, DPP-4 inhibitors, SGLT2 inhibitors, sulfonylureas, and thiazolidinediones). GLP-1 RAs were also linked to reduced risk of liver function decompensation compared to other antidiabetics in diabetic patients. They were effective across different stages of MASLD, MASH, liver fibrosis, and cirrhosis, with the greatest efficacy observed in patients without prior liver disease. GLP-1 RAs indirectly reduce HCC risk by influencing the reward system and addiction to smoking and alcohol. Moreover, combination therapies including GLP-1 RAs offer additional benefits over monotherapies [
14].
Well-established metabolic effects of GLP-1 RAs appear to be accompanied by epigenetic modulatory properties, such as inhibiting lactate-mediated histone lactylation in fibrotic contexts, presenting a novel strategy to inhibit progression from chronic liver injury through fibrosis to HCC [
60].
3.2.11. Sleep Apnea
Obstructive sleep apnea (OSA) is a component of metabolic syndrome. The randomized SURMOUNT trial, including patients with obstructive sleep apnea, demonstrated that tirzepatide treatment for one year resulted in a 20% reduction in body weight and a 60% decrease in apnea episodes during sleep [
61]. In the SCALE study focusing on sleep apnea in obese individuals with moderate to severe OSA who were unwilling or unable to use continuous positive airway pressure (CPAP), liraglutide was associated with a significantly greater reduction in apnea-hypopnea index and hypoxemia compared to placebo, alongside weight loss and improvements in systolic blood pressure and HbA1c levels [
58].
3.2.12. Arthritis
Osteoarthritis (OA) is a leading cause of disability, affecting over 300 million people worldwide. OA commonly involves the knees, hands, hips, and spine. Risk factors for knee and hip OA include age, sex, obesity, cardiometabolic factors, injuries, physical activity, and anatomical abnormalities. OA is suspected to be a component of metabolic syndrome [
62]. Randomized controlled trials have shown that overweight/obese individuals who lost an average of 14% of their body weight during semaglutide therapy experienced significant improvement in knee osteoarthritis pain, likely related to both weight loss and the anti-inflammatory properties of the drug. These results encourage consideration of GLP-1 RAs as potential treatment in patients requiring joint replacement surgery; however, further research is needed [
63]. The discovery of anti-inflammatory, immunoregulatory, and differentiating effects of GLP-1 analogues at the joint tissue and cellular level raises the hypothesis of their potential use in OA treatment [
62].
3.2.13. PCOS, Libido, Fertility
Polycystic ovary syndrome (PCOS) is the most common cause of anovulatory infertility, affecting approximately 20% of women of reproductive age (according to Rotterdam criteria). Over half of women with PCOS struggle with overweight or obesity, which negatively impacts implantation rates, pregnancy numbers, and increases miscarriage risk [
64].
GLP-1 RAs demonstrate beneficial effects in women with PCOS, who often present with comorbidities such as obesity, insulin resistance, glucose metabolism disorders, dyslipidemia, hypertension, or metabolic syndrome, all increasing diabetes risk. This therapy helps regulate appetite, improve glycemic control, induce weight loss, and gradually reduce metabolic disturbances that indirectly affect hormonal balance [
18,
65,
66,
67]. In patients with the so-called metabolic phenotype, an additional benefit is the reduced cardiovascular risk.
In an open-label, prospective, randomized clinical trial conducted over 12 months (September 2014 – May 2015) with 28 obese women diagnosed with PCOS per Rotterdam criteria, preconception low-dose liraglutide combined with metformin was more effective than metformin alone in increasing pregnancy rates in IVF procedures, despite similar weight loss effects in both groups [
64].
Studies suggest that GLP-1 RAs may support fertility by enhancing the preovulatory luteinizing hormone surge, leading to increased estrogen levels and restoration of normal menstrual cycles. Especially in women with PCOS, many begin ovulating again after significant visceral fat loss during tirzepatide or semaglutide therapy [
58,
68,
69].
A literature review by Abdalla et al. showed that exenatide, both as monotherapy and combined with metformin, improves menstrual regularity and ovulation rates in overweight or obese women with PCOS, directly increasing fertility [
70]. In a study by Karen Elkind-Hirsch et al. involving 60 obese women with oligo-ovulation and PCOS (70% completed the study), combination therapy with exenatide and metformin resulted in greater improvement in menstrual cyclicity, endogenous ovulatory function, body weight, waist circumference, insulin resistance, and hyperandrogenism compared to monotherapy [
71].
Notably, women on GLP-1 analogues experience decreased testosterone levels—a key hormone for maintaining normal libido, whose excess can impair fertility. However, the impact of GLP-1 RAs on libido remains controversial; some patients report decreased libido during therapy, while overweight or obese individuals who lost weight report improved self-esteem, vitality, and sexual activity [
72].
In men, obesity significantly affects the hypothalamic-pituitary-gonadal axis, resulting in decreased testosterone levels and diminished libido. Although data are limited, a possible role for GLP-1 RAs in treating functional hypogonadism related to overweight and obesity has been suggested [
73]. In a retrospective observational study on 43 men, liraglutide proved superior to testosterone supplementation in obese men with functional hypogonadism [
74].
In diabetic mice, liraglutide was shown to improve erectile function by relaxing corpus cavernosum smooth muscle cells and alleviate hypogonadism symptoms post-orchidectomy, while exenatide improved sperm quality in obese mice [
75,
76]. GLP-1 receptors have also been found in human testes, and some studies confirm improvements in erectile dysfunction in obese diabetic men treated with GLP-1 RAs. Positive effects on semen quality were suggested in a study by La Vignera et al., but it remains unclear whether these were due to weight loss or direct drug effects [
74].
5. Conclusions
GLP-1 receptor agonists exhibit multifaceted therapeutic effects, providing significant clinical benefits not only in the treatment of type 2 diabetes but also obesity, cardiovascular diseases, neurodegenerative disorders, and certain cancers. Furthermore, they offer considerable advantages in conditions such as sleep apnea, musculoskeletal diseases, PCOS, prostate enlargement, liver dysfunction, and even substance use disorders and mental health improvement. They are characterized by a favorable safety profile, with adverse effects typically mild and not leading to treatment discontinuation. Despite promising results from current studies, further large-scale, well-designed clinical trials are necessary to fully evaluate and harness the potential of this drug class in clinical practice. Considering the entirety of scientific evidence presented, GLP-1 RAs represent a major breakthrough and the future of multiple medical fields, with their full potential yet to be completely uncovered.
Author Contributions
Conceptualization, W.M., P.W, P.W. ; methodology, W.M., P.W, P.W.; software, R.M.; validation, J.J.,W.M., MS, formal analysis, J.J.; investigation, P.W,P.W..; resources, KWK., R.M.; data curation, J.M.G. writing—original draft preparation, W.M., P.W, P.W.; writing—review and editing, W.M.; visualization, M.S.,K.W.K.; supervision, J.J; project administration, W.M.,J.J. All authors have read and agreed to the published version of the manuscript.
Funding
Please add: This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Publicly available datasets were analyzed in this study.
Acknowledgments
We thank the anonymous referees for their useful suggestions. This work utilized. ChatGPT, a large language model-based assistant, for language translation and text structuring. The authors retained full responsibility for all final content.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
Abbreviations are explained in the text.
References
- Parkes, DG; Mace, KF; Trautmann, ME. Discovery and development of exenatide: the first antidiabetic agent to leverage the multiple benefits of the incretin hormone, GLP-1. Expert Opin Drug Discov. 2013, 8(2), 219–44. [Google Scholar] [CrossRef]
- Knop, FK; Brønden, A; Vilsbøll, T. Exenatide: pharmacokinetics, clinical use, and future directions. Expert Opin Pharmacother. 2017, 18(6), 555–71. [Google Scholar] [CrossRef]
- Nauck, MA; Quast, DR; Wefers, J; Meier, JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Mol Metab. 2021, 46, 101102. [Google Scholar] [CrossRef]
- Yao, YX; Tang, C; Si, FL; Lv, JC; Shi, SF; Zhou, XJ; et al. Glucagon-like peptide-1 receptor agonists, inflammation, and kidney diseases: evidence from Mendelian randomization. Ren Fail. 2025, 47(1), 2478488. [Google Scholar] [CrossRef]
- O’Keefe, JH; Franco, WG; O’Keefe, EL. Anti-consumption agents: Tirzepatide and semaglutide for treating obesity-related diseases and addictions, and improving life expectancy. Prog Cardiovasc Dis. 2025, 89, 102–12. [Google Scholar] [CrossRef] [PubMed]
- Li QX, Gao H, Guo YX, Wang BY, Hua R xuan, Gao L, et al. GLP-1 and Underlying Beneficial Actions in Alzheimer’s Disease, Hypertension, and NASH. Front Endocrinol. 2021, 12, 721198. [CrossRef] [PubMed]
- Hendershot, CS; Bremmer, MP; Paladino, MB; Kostantinis, G; Gilmore, TA; Sullivan, NR; et al. Once-Weekly Semaglutide in Adults With Alcohol Use Disorder: A Randomized Clinical Trial. JAMA Psychiatry 2025, 82(4), 395. [Google Scholar] [CrossRef]
- Klausen, MK; Thomsen, M; Wortwein, G; Fink-Jensen, A. The role of glucagon-like peptide 1 (GLP-1) in addictive disorders. Br J Pharmacol. 2022, 179(4), 625–41. [Google Scholar] [CrossRef] [PubMed]
- Schooling, CM; Yang, G; Soliman, GA; Leung, GM. A Hypothesis That Glucagon-like Peptide-1 Receptor Agonists Exert Immediate and Multifaceted Effects by Activating Adenosine Monophosphate-Activate Protein Kinase (AMPK). Life 2025, 15(2), 253. [Google Scholar] [CrossRef]
- Jerlhag, E. GLP-1 signaling and alcohol-mediated behaviors; preclinical and clinical evidence. Neuropharmacology 2018, 136, 343–9. [Google Scholar] [CrossRef]
- Jerlhag, E. GLP-1 Receptor Agonists: Promising Therapeutic Targets for Alcohol Use Disorder. Endocrinology 2025, 166(4), bqaf028. [Google Scholar] [CrossRef]
- Lee, S; Li, M; Le, GH; Teopiz, KM; Vinberg, M; Ho, R; et al. Glucagon-like peptide-1 receptor agonists (GLP-1RAs) as treatment for nicotine cessation in psychiatric populations: a systematic review. Ann Gen Psychiatry 2024, 23(1), 45. [Google Scholar] [CrossRef] [PubMed]
- Tuesta, LM; Chen, Z; Duncan, A; Fowler, CD; Ishikawa, M; Lee, BR; et al. GLP-1 acts on habenular avoidance circuits to control nicotine intake. Nat Neurosci. 2017, 20(5), 708–16. [Google Scholar] [CrossRef] [PubMed]
- Wang, L; Berger, NA; Kaelber, DC; Xu, R. Association of GLP-1 Receptor Agonists and Hepatocellular Carcinoma Incidence and Hepatic Decompensation in Patients With Type 2 Diabetes. Gastroenterology 2024, 167(4), 689–703. [Google Scholar] [CrossRef]
- Martinelli, S; Mazzotta, A; Longaroni, M; Petrucciani, N. Potential role of glucagon-like peptide-1 (GLP-1) receptor agonists in substance use disorder: A systematic review of randomized trials. Drug Alcohol Depend. 2024, 264, 112424. [Google Scholar] [CrossRef]
- Hernandez, NS; Ige, KY; Mietlicki-Baase, EG; Molina-Castro, GC; Turner, CA; Hayes, MR; et al. Glucagon-like peptide-1 receptor activation in the ventral tegmental area attenuates cocaine seeking in rats. Neuropsychopharmacology 2018, 43(10), 2000–8. [Google Scholar] [CrossRef] [PubMed]
- Stachelska, A. Aktualne tendencje przestępczość narkotykowej oraz zażywania narkotyków wśród nieletnich w Polsce. PRAWO WIĘŹ 2025, 55(2), 14. [Google Scholar] [CrossRef]
- Ceceli, AO; Bradberry, CW; Goldstein, RZ. The neurobiology of drug addiction: cross-species insights into the dysfunction and recovery of the prefrontal cortex. Neuropsychopharmacology 2022, 47(1), 276–91. [Google Scholar] [CrossRef]
- Schmidt, HD; Mietlicki-Baase, EG; Ige, KY; Maurer, JJ; Reiner, DJ; Zimmer, DJ; et al. Glucagon-Like Peptide-1 Receptor Activation in the Ventral Tegmental Area Decreases the Reinforcing Efficacy of Cocaine. Neuropsychopharmacology 2016, 41(7), 1917–28. [Google Scholar] [CrossRef]
- Beheshti, I. Cocaine Destroys Gray Matter Brain Cells and Accelerates Brain Aging. Biology 2023, 12(5), 752. [Google Scholar] [CrossRef]
- Egecioglu, E; Engel, JA; Jerlhag, E. The Glucagon-Like Peptide 1 Analogue, Exendin-4, Attenuates the
Rewarding Properties of Psychostimulant Drugs in Mice. Holscher C, redaktor. In PLoS ONE; Holscher, C, Ed.; 2013; Volume 8, 7. [Google Scholar]
- Chaves Filho, AJM; Cunha, NL; De Souza, AG; Soares, MVR; Jucá, PM; De Queiroz, T; et al. The GLP-1 receptor agonist liraglutide reverses mania-like alterations and memory deficits induced by D-amphetamine and augments lithium effects in mice: Relevance for bipolar disorder. Prog Neuropsychopharmacol Biol Psychiatry 2020, 99, 109872. [Google Scholar] [CrossRef]
- Arillotta, D; Floresta, G; Papanti Pelletier, GD; Guirguis, A; Corkery, JM; Martinotti, G; et al. Exploring the Potential Impact of GLP-1 Receptor Agonists on Substance Use, Compulsive Behavior, and Libido: Insights from Social Media Using a Mixed-Methods Approach. Brain Sci. 2024, 14(6), 617. [Google Scholar] [CrossRef]
- Au, HCT; Lam, PH; Kabir, F; Huang, CL; Dri, CE; Le, GH; et al. Glucagon-like peptide-1 receptor agonists for the treatment of opioid use disorders: a systematic review. Acta Neuropsychiatr. 2025, 37, e85. [Google Scholar] [CrossRef]
- Qeadan, F; McCunn, A; Tingey, B. The association between glucose-dependent insulinotropic polypeptide and/or glucagon-like peptide-1 receptor agonist prescriptions and substance-related outcomes in patients with opioid and alcohol use disorders: A real-world data analysis. Addiction 2025, 120(2), 236–50. [Google Scholar] [CrossRef] [PubMed]
- Wang, W; Volkow, ND; Wang, Q; Berger, NA; Davis, PB; Kaelber, DC; et al. Semaglutide and Opioid Overdose Risk in Patients With Type 2 Diabetes and Opioid Use Disorder. JAMA Netw Open 2024, 7(9), e2435247. [Google Scholar] [CrossRef] [PubMed]
- Monroe, SM; Harkness, KL. Major Depression and Its Recurrences: Life Course Matters. Annu Rev Clin Psychol. 2022, 18(1), 329–57. [Google Scholar] [CrossRef] [PubMed]
- Conejero, I; Olié, E; Calati, R; Ducasse, D; Courtet, P. Psychological Pain, Depression, and Suicide: Recent Evidences and Future Directions. Curr Psychiatry Rep. 2018, 20(5), 33. [Google Scholar] [CrossRef]
- McIntyre, RS. Glucagon-like peptide-1 receptor agonists (GLP-1 RAs) and suicidality: what do we know and future vistas. Expert Opin Drug Saf. 2024, 23(5), 539–42. [Google Scholar] [CrossRef]
- Zhou, J; Zheng, Y; Xu, B; Long, S; Zhu, L e; Liu, Y; et al. Exploration of the potential association between GLP-1 receptor agonists and suicidal or self-injurious behaviors: a pharmacovigilance study based on the FDA Adverse Event Reporting System database. BMC Med. 2024, 22(1), 65. [Google Scholar] [CrossRef]
- Darwish, AB; El Sayed, NS; Salama, AAA; Saad, MA. Dulaglutide impedes depressive-like behavior persuaded by chronic social defeat stress model in male C57BL/6 mice: Implications on GLP-1R and cAMP/PKA signaling pathway in the hippocampus. Life Sci. 2023, 320, 121546. [Google Scholar] [CrossRef]
- Chen, X; Zhao, P; Wang, W; Guo, L; Pan, Q. The Antidepressant Effects of GLP-1 Receptor Agonists: A Systematic Review and Meta-Analysis. Am J Geriatr Psychiatry 2024, 32(1), 117–27. [Google Scholar] [CrossRef]
- Himmerich, H. Glucagon-like-peptide-1-Rezeptoragonisten: eine neue pharmakologische Behandlungsoption für psychiatrische Erkrankungen? Nervenarzt 2025, 96(3), 247–54. [Google Scholar] [CrossRef]
- Komsuoglu Celikyurt, I; Mutlu, O; Ulak, G; Uyar, E; Bektaş, E; Yildiz Akar, F; et al. Exenatide Treatment Exerts Anxiolytic- and Antidepressant-Like Effects and Reverses Neuropathy in a Mouse Model of Type-2 Diabetes. Med Sci Monit Basic Res. 2014, 20, 112–7. [Google Scholar] [CrossRef] [PubMed]
- Perkovic, V; Tuttle, KR; Rossing, P; Mahaffey, KW; Mann, JFE; Bakris, G; et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. N Engl J Med. 2024, 391(2), 109–21. [Google Scholar] [CrossRef]
- Sourris, KC; Ding, Y; Maxwell, SS; Al-sharea, A; Kantharidis, P; Mohan, M; et al. Glucagon-like peptide-1 receptor signaling modifies the extent of diabetic kidney disease through dampening the receptor for advanced glycation end products–induced inflammation. Kidney Int. stycznia 2024, 105(1), 132–49. [Google Scholar] [CrossRef]
- Chen, J; Dong, X; Lin, Y; Lv, C. The critical role of GLP-1 signaling pathways in the pathology of Parkinson’s disease and diabetes. Pathol - Res Pract. 2025, 270, 155985. [Google Scholar] [CrossRef] [PubMed]
- Santoro, A; McGraw, TE; Kahn, BB. Insulin action in adipocytes, adipose remodeling, and systemic effects. Cell Metab. 2021, 33(4), 748–57. [Google Scholar] [CrossRef] [PubMed]
- Cancel, M; Pouillot, W; Mahéo, K; Fontaine, A; Crottès, D; Fromont, G. Interplay between Prostate Cancer and Adipose Microenvironment: A Complex and Flexible Scenario. Int J Mol Sci. 2022, 23(18), 10762. [Google Scholar] [CrossRef]
- Hu, X; Hu, C; Zhang, C; Zhang, M; Long, S; Cao, Z. Role of Adiponectin in prostate cancer. Int Braz J Urol. 2019, 45(2), 220–8. [Google Scholar] [CrossRef]
- Pallegar NK, Christian SL. Adipocytes in the Tumour Microenvironment. W: Birbrair A, redaktor. Tumor Microenvironment. Cham: Springer International Publishing; 2020. s. 1–13. (Advances in Experimental Medicine and Biology; t. 1234).
- Karzai, FH; Madan, RA; Dahut, WL. Metabolic Syndrome in Prostate Cancer: Impact on Risk and Outcomes. Future Oncol. 2016, 12(16), 1947–55. [Google Scholar] [CrossRef]
- Levy, S; Attia, A; Elshazli, RM; Abdelmaksoud, A; Tatum, D; Aiash, H; et al. Differential Effects of GLP-1 Receptor Agonists on Cancer Risk in Obesity: A Nationwide Analysis of 1.1 Million Patients. Cancers 2024, 17(1), 78. [Google Scholar] [CrossRef]
- Chen PH, Hibler EA. Abstract 735: The associations between the use of GLP-1 receptor agonists, cancer
recurrence and all-cause mortality among cancer survivors. Cancer Res. 4 kwietnia
2023;83(7_Supplement):735–735. [CrossRef]
- Eftekhari, S; Montazeri, H; Tarighi, P. Synergistic anti-tumor effects of Liraglutide, a glucagon-like peptide-1 receptor agonist, along with Docetaxel on LNCaP prostate cancer cell line. Eur J Pharmacol. 2020, 878, 173102. [Google Scholar] [CrossRef]
- Du H, Meng X, Yao Y, Xu J. The mechanism and efficacy of GLP-1 receptor agonists in the treatment of
Alzheimer’s disease. Front Endocrinol [Internet]. 2022;Volume 13-2022.
- Scheltens, P; De Strooper, B; Kivipelto, M; Holstege, H; Chételat, G; Teunissen, CE; et al. Alzheimer’s disease. The Lancet 2021, 397(10284), 1577–90. [Google Scholar] [CrossRef]
- Xie, Y; Choi, T; Al-Aly, Z. Author Correction: Mapping the effectiveness and risks of GLP-1 receptor agonists. Nat Med. 2025, 31(3), 1038–1038. [Google Scholar] [CrossRef] [PubMed]
- Colin, IM; Szczepanski, LW; Gérard, AC; et al. Emerging evidence for the use of antidiabetic drugs, glucagon-like peptide-1 receptor agonists, for the treatment of Alzheimer’s disease. Eur Endocrinol. 2023, 19(1), 16. [Google Scholar] [CrossRef] [PubMed]
- Siddeeque, N; Hussein, MH; Abdelmaksoud, A; Bishop, J; Attia, AS; Elshazli, RM; et al. Neuroprotective effects of GLP-1 receptor agonists in neurodegenerative Disorders: A Large-Scale Propensity-Matched cohort study. Int Immunopharmacol. 2024, 143, 113537. [Google Scholar] [CrossRef]
- Delvadia, P; Dhote, V; Mandloi, AS; Soni, R; Shah, J. Dual GLP-1 and GIP Agonist Tirzepatide Exerted Neuroprotective Action in a Parkinson’s Disease Rat Model. ACS Chem Neurosci. 2025, 16(5), 818–25. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, LOS; Mazetto, RASV; Defante, MLR; Antunes, VLJ; Gonçalves, OR; Corso, AMS; et al. Efficacy and safety of glucagon-like peptide 1 agonists for Parkinson’s disease: a systematic review and meta-analysis. Arq Neuropsiquiatr. 2025, 83(04), 001–10. [Google Scholar] [CrossRef]
- Helal, MM; AbouShawareb, H; Abbas, OH; Haddad, R; Zain, Y; Osman, ASA; et al. GLP-1 receptor agonists in Parkinson’s disease: an updated comprehensive systematic review with meta-analysis. Diabetol Metab Syndr. 2025, 17(1), 352. [Google Scholar] [CrossRef]
- Moțățăianu, A; Mănescu, IB; Șerban, G; Bărcuțean, L; Ion, V; Bălașa, R; et al. Exploring the Role of Metabolic Hormones in Amyotrophic Lateral Sclerosis. Int J Mol Sci. 2024, 25(10), 5059. [Google Scholar] [CrossRef]
- Arai, T; Atsukawa, M; Tsubota, A; Oikawa, T; Tada, T; Matsuura, K; et al. Beneficial effect of oral semaglutide for type 2 diabetes mellitus in patients with metabolic dysfunction-associated steatotic liver disease: A prospective, multicentre, observational study. Diabetes Obes Metab. 2024, 26(11), 4958–65. [Google Scholar] [CrossRef] [PubMed]
- Newsome, PN; Buchholtz, K; Cusi, K; Linder, M; Okanoue, T; Ratziu, V; et al. A Placebo-Controlled Trial of Subcutaneous Semaglutide in Nonalcoholic Steatohepatitis. N Engl J Med. 2021, 384(12), 1113–24. [Google Scholar] [CrossRef]
- Abdelmalek, MF; Harrison, SA; Sanyal, AJ. The role of glucagon-like peptide-1 receptor agonists in metabolic dysfunction-associated steatohepatitis. Diabetes Obes Metab. 2024, 26(6), 2001–16. [Google Scholar] [CrossRef] [PubMed]
- Alkhouri, N; Charlton, M; Gray, M; Noureddin, M. The pleiotropic effects of glucagon-like peptide-1 receptor agonists in patients with metabolic dysfunction-associated steatohepatitis: a review for gastroenterologists. Expert Opin Investig Drugs 2025, 34(3), 169–95. [Google Scholar] [CrossRef]
- Bernsmeier, C; Meyer-Gerspach, AC; Blaser, LS; Jeker, L; Steinert, RE; Heim, MH; et al. Glucose-Induced Glucagon-Like Peptide 1 Secretion Is Deficient in Patients with Non-Alcoholic Fatty Liver Disease. Petta S, PLoS ONE 2014, 9(1), e87488. [Google Scholar] [CrossRef]
- Liu, C; Zhang, Q; Zhou, H; Jin, L; Liu, C; Yang, M; et al. GLP-1R activation attenuates the progression of pulmonary fibrosis via disrupting NLRP3 inflammasome/PFKFB3-driven glycolysis interaction and histone lactylation. J Transl Med. 2024, 22(1), 954. [Google Scholar] [CrossRef] [PubMed]
- Malhotra A, Grunstein RR, Fietze I, Weaver TE, Redline S, Azarbarzin A, et al. Tirzepatide for the
Treatment of Obstructive Sleep Apnea and Obesity. N Engl J Med. 3 października 2024;391(13):1193–205. [CrossRef]
- Meurot, C; Jacques, C; Martin, C; Sudre, L; Breton, J; Rattenbach, R; et al. Targeting the GLP-1/GLP-1R axis to treat osteoarthritis: A new opportunity? J Orthop Transl. 2022, 32, 121–9. [Google Scholar] [CrossRef]
- Baser, O; Rodchenko, K; Vivier, E; Baser, I; Lu, Y; Mohamed, M. The impact of approved anti-obesity medications on osteoarthritis. Expert Opin Pharmacother. 2024, 25(11), 1565–73. [Google Scholar] [CrossRef]
- Salamun V, Jensterle M, Janez A, Vrtacnik Bokal E. Liraglutide increases IVF pregnancy rates in obese PCOS women with poor response to first-line reproductive treatments: a pilot randomized study. Eur J Endocrinol. 2018, 179(1), 1–11. [CrossRef]
- Bednarz, K; Kowalczyk, K; Cwynar, M; Czapla, D; Czarkowski, W; Kmita, D; et al. The Role of Glp-1 Receptor Agonists in Insulin Resistance with Concomitant Obesity Treatment in Polycystic Ovary Syndrome. Int J Mol Sci. 2022, 23(8), 4334. [Google Scholar] [CrossRef]
- Bader, S; Bhatti, R; Mussa, B; Abusanana, S. A systematic review of GLP-1 on anthropometrics, metabolic and endocrine parameters in patients with PCOS. Womens Health 2024, 20, 17455057241234530. [Google Scholar] [CrossRef] [PubMed]
- Austregésilo De Athayde De Hollanda Morais, B; Martins Prizão, V; De Moura De Souza, M; Ximenes Mendes, B; Rodrigues Defante, ML; Cosendey Martins, O; et al. The efficacy and safety of GLP-1 agonists in PCOS women living with obesity in promoting weight loss and hormonal regulation: A meta-analysis of randomized controlled trials. J Diabetes Complications 2024, 38(10), 108834. [Google Scholar] [CrossRef]
- Monney, M; Mavromati, M; Leboulleux, S; Gariani, K. Endocrine and metabolic effects of GLP-1 receptor agonists on women with PCOS, a narrative review. Endocr Connect. 2025, 14(5), e240529. [Google Scholar] [CrossRef] [PubMed]
- Siamashvili, M; Davis, SN. Update on the effects of GLP-1 receptor agonists for the treatment of polycystic ovary syndrome. Expert Rev Clin Pharmacol. 2021, 14(9), 1081–9. [Google Scholar] [CrossRef]
- Abdalla, MA; Deshmukh, H; Atkin, S; Sathyapalan, T. The potential role of incretin-based therapies for polycystic ovary syndrome: a narrative review of the current evidence. Ther Adv Endocrinol Metab. 2021, 12, 2042018821989238. [Google Scholar] [CrossRef]
- Elkind-Hirsch, K; Marrioneaux, O; Bhushan, M; Vernor, D; Bhushan, R. Comparison of Single and Combined Treatment with Exenatide and Metformin on Menstrual Cyclicity in Overweight Women with Polycystic Ovary Syndrome. J Clin Endocrinol Metab. 2008, 93(7), 2670–8. [Google Scholar] [CrossRef] [PubMed]
- Frangie Machado, M; Shunk, T; Hansen, G; Harvey, C; Fulford, B; Hauf, S; et al. Clinical Effects of Glucagon-Like Peptide-1 Agonist Use for Weight Loss in Women With Polycystic Ovary Syndrome: A Scoping Review. Cureus 2024. [Google Scholar] [CrossRef]
- Salvio, G; Ciarloni, A; Ambo, N; Bordoni, M; Perrone, M; Rossi, S; et al. Effects of glucagon-like peptide 1 receptor agonists on testicular dysfunction: A systematic review and meta-analysis. Andrology 2025, 13(8), 2022–34. [Google Scholar] [CrossRef]
- La Vignera, S; Condorelli, RA; Calogero, AE; Cannarella, R; Aversa, A. Sexual and Reproductive Outcomes in Obese Fertile Men with Functional Hypogonadism after Treatment with Liraglutide: Preliminary Results. J Clin Med. 2023, 12(2), 672. [Google Scholar] [CrossRef] [PubMed]
- Yuan, P; Ma, D; Gao, X; Wang, J; Li, R; Liu, Z; et al. Liraglutide Ameliorates Erectile Dysfunction via Regulating Oxidative Stress, the RhoA/ROCK Pathway and Autophagy in Diabetes Mellitus. Front Pharmacol. 2020, 11, 1257. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H; Meng, J; Li, X; Zhou, S; Qu, D; Wang, N; et al. Pro-GLP-1, a Pro-drug of GLP-1, is neuroprotective in cerebral ischemia. Eur J Pharm Sci. 2015, 70, 82–91. [Google Scholar] [CrossRef] [PubMed]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |