Submitted:
30 January 2026
Posted:
03 February 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Clinical Paradox: Lipedematous Dynapenia
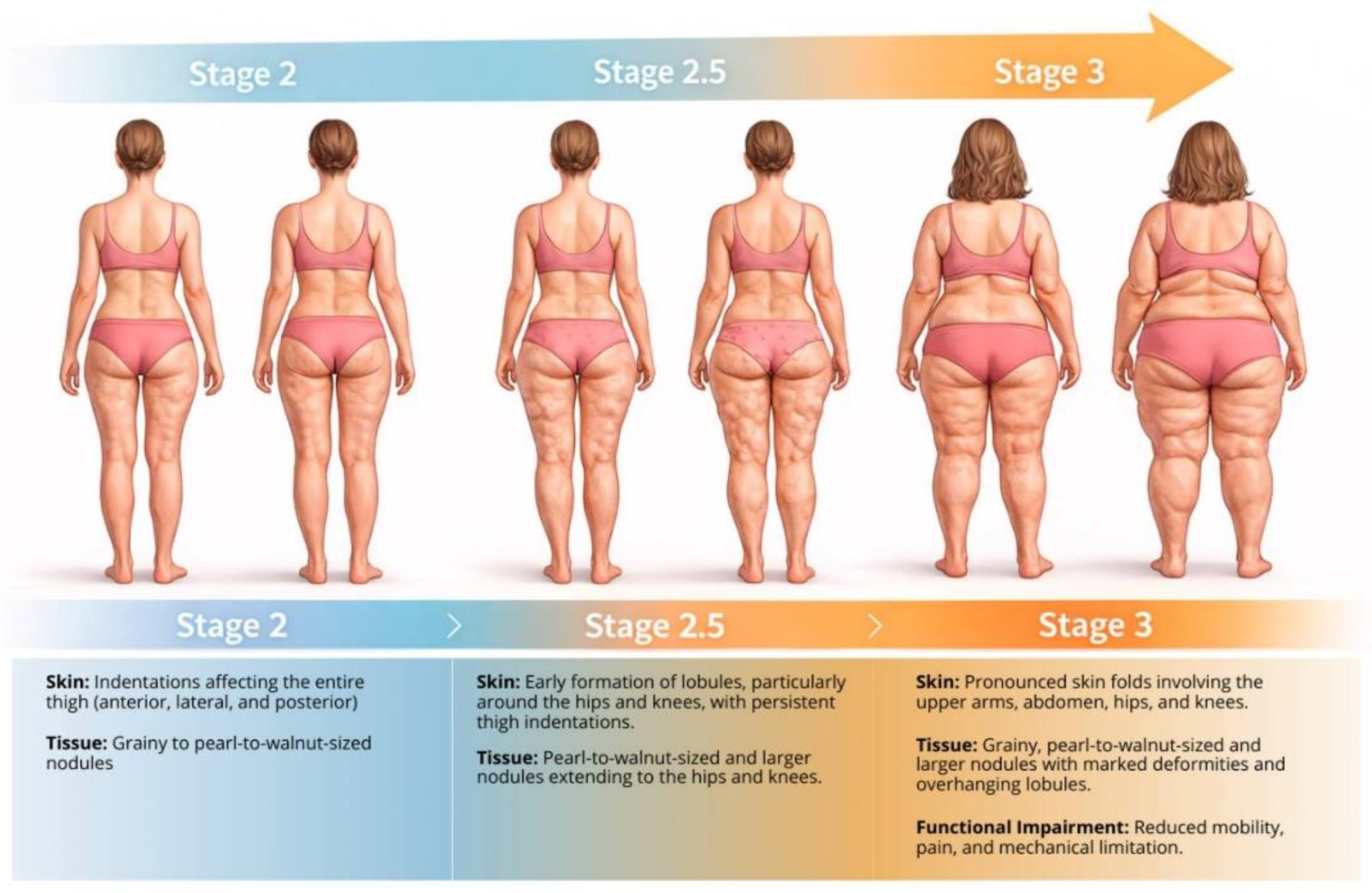
2.1. The Illusion of Mass: Pseudo-Hypertrophy versus Functional Reality
2.2. The Failure of Standard Metrics: Why BMI Is Blind
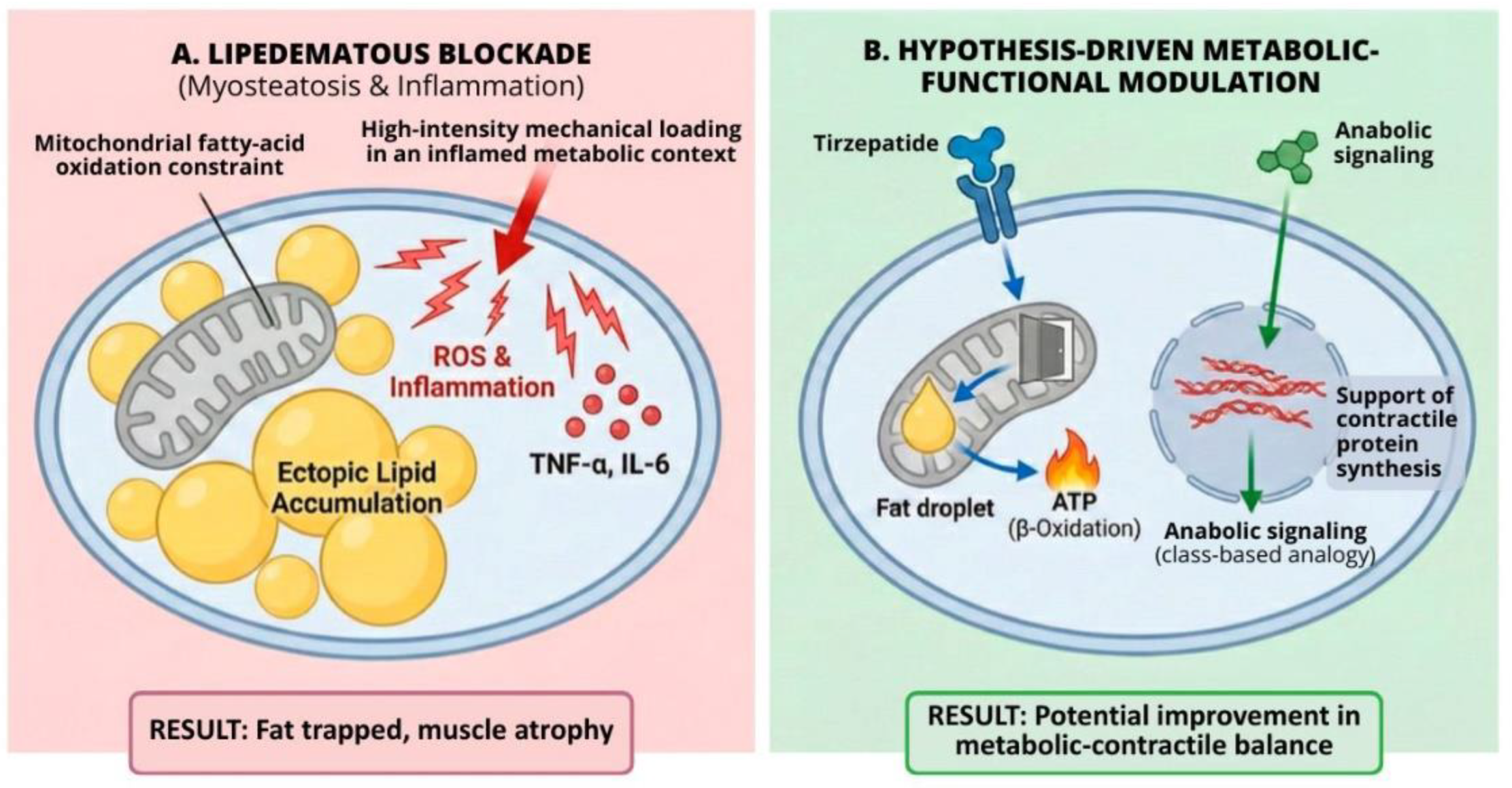
2.3. Inflammatory Myosteatosis as a Metabolic Bottleneck
2.4. The Vicious Cycle of Pain, Inactivity, and Functional Decline
3. The Therapeutic Deadlock and a Translational, Hypothesis-Driven Pharmacological Bypass
3.1. The Exercise Paradox: Why Mechanical Loading May Fail
3.2. The Dual-Target Hypothesis: Breaking the Pathophysiological Cycle
3.2.1. Metabolic Modulation: Tirzepatide and Mitochondrial Flexibility
3.2.2. Anabolic Signaling as a Functional Rescue Analogy
4. Discussion: Translational Implications and Safety Considerations
4.1. Identifying the Functional Inflection Point
4.2. Contextualizing the Anabolic Rationale Within Evidence-Based Medicine
4.3. Safety Considerations in a Hypothesis-Driven Framework
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAS | Anabolic Androgenic Steroids |
| BIA | Bioelectrical Impedance Analysis |
| BMI | Body Mass Index |
| CPT-1 | Carnitine Palmitoyltransferase 1 |
| ECM | Extracellular Matrix |
| ERα | Estrogen Receptor Alpha |
| ERβ | Estrogen Receptor Beta |
| FFM | Fat-Free Mass |
| GIP | Glucose-Dependent Insulinotropic Polypeptide |
| GLP-1 | Glucagon-Like Peptide-1 |
| HDL | High-Density Lipoprotein |
| HFpEF | Heart Failure with Preserved Ejection Fraction |
| IL-6 | Interleukin-6 |
| MRI | Magnetic Resonance Imaging |
| ROS | Reactive Oxygen Species |
| RQ | Respiratory Quotient |
| SAT | Subcutaneous Adipose Tissue |
| SIF | Italian Society of Phlebology |
| SISMES | Italian Society of Motor and Sports Sciences |
| SMM | Skeletal Muscle Mass |
| STS | Sit-to-Stand Test |
| TNF-α | Tumor Necrosis Factor Alpha |
| TUG | Timed Up and Go Test |
| UCP1 | Uncoupling Protein 1 |
References
- Allen, E.; Hines, E.; Hines, E. Lipedema of the Legs: A Syndrome Characterized by Fat Legs and Orthostatic Edema. Proc. Staff Meet. Mayo Clin 1940, 15, 184–187. [Google Scholar] [CrossRef]
- Wold, L.E.; Hines, E.A.; Allen, E.V. Lipedema of the Legs: A Syndrome Characterized by Fat Legs and Edema. Ann Intern Med 1951, 34, 1243–1250. [Google Scholar] [CrossRef]
- Földi, E.; Földi; Lipedema, M. Földi’s Textbook of Lymphology: For Physicians and Lymphedema Therapists; Elsevier, 2006. [Google Scholar]
- Herbst, K.L. Rare Adipose Disorders (RADs) Masquerading as Obesity. Acta Pharmacol Sin 2012, 33, 155–172. [Google Scholar] [CrossRef]
- Al-Ghadban, S.; Evancio, J.V.; Alfiscar, P.E.F.; Herbst, K.L. New Characterization of Lipedema Stages: Focus on Pain, Water, Fat and Skeletal Muscle. Life 2025, 15, 1397. [Google Scholar] [CrossRef]
- Christoffersen, V.; Tennfjord, M.K. Younger Women with Lipedema, Their Experiences with Healthcare Providers, and the Importance of Social Support and Belonging: A Qualitative Study. Int J Environ Res Public Health 2023, 20, 1925. [Google Scholar] [CrossRef]
- Falck, J.; Rolander, B.; Nygårdh, A.; Jonasson, L.-L.; Mårtensson, J. Women with Lipoedema: A National Survey on Their Health, Health-Related Quality of Life, and Sense of Coherence. BMC Women’s Health 2022, 22, 457. [Google Scholar] [CrossRef]
- Romeijn, J.R.M.; De Rooij, M.J.M.; Janssen, L.; Martens, H. Exploration of Patient Characteristics and Quality of Life in Patients with Lipoedema Using a Survey. Dermatol Ther (Heidelb) 2018, 8, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Dudek, J.E.; Białaszek, W.; Gabriel, M. Quality of Life, Its Factors, and Sociodemographic Characteristics of Polish Women with Lipedema. BMC Women’s Health 2021, 21, 27. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, J. Lipedema, Body-Related Emotional and Disordered Eating to Cope. Surg Obes Relat Dis 2023, 19, 667–668. [Google Scholar] [CrossRef]
- Clarke, C.; Kirby, J.N.; Best, T. Beyond the Physical: The Interplay of Experienced Weight Stigma, Internalised Weight Bias and Depression in Lipoedema. Clin Obes 2025, 15, e12727. [Google Scholar] [CrossRef] [PubMed]
- Torre, Y.S.-D.L.; Wadeea, R.; Rosas, V.; Herbst, K.L. Lipedema: Friend and Foe. Horm Mol Biol Clin Investig 2018, 33, 20170076. [Google Scholar] [CrossRef] [PubMed]
- Bertsch, T.; Erbacher, G.; Elwell, R.; Partsch, H. Lipoedema: A Paradigm Shift and Consensus. J Wound Care 2020, 29, 1–51. [Google Scholar] [CrossRef]
- Viana, D.P.C.; Invitti, A.L.; Schor, E. Tirzepatide as a Potential Disease-Modifying Therapy in Lipedema: A Narrative Review on Bridging Metabolism, Inflammation, and Fibrosis. Int J Mol Sci 2025, 26, 10741. [Google Scholar] [CrossRef]
- van Esch-Smeenge, J.; Damstra, R.J.; Hendrickx, A.A. Muscle Strength and Functional Exercise Capacity in Patients with Lipoedema and Obesity: A Comparative Study. J Lymphoedema 2017, 12. [Google Scholar]
- Crescenzi, R.; Marton, A.; Donahue, P.M.C.; Mahany, H.B.; Lants, S.K.; Wang, P.; Beckman, J.A.; Donahue, M.J.; Titze, J. Tissue Sodium Content Is Elevated in the Skin and Subcutaneous Adipose Tissue in Women with Lipedema. Obesity 2018, 26, 310–317. [Google Scholar] [CrossRef]
- Gumucio, J.P.; Qasawa, A.H.; Ferrara, P.J.; Malik, A.N.; Funai, K.; Mcdonagh, B.; Mendias, C.L. Reduced Mitochondrial Lipid Oxidation Leads to Fat Accumulation in Myosteatosis. FASEB J 2019, 33, 7863–7881. [Google Scholar] [CrossRef]
- Goodpaster, B.H.; Wolf, D. Skeletal Muscle Lipid Accumulation in Obesity, Insulin Resistance, and Type 2 Diabetes. Pediatr Diabetes 2004, 5, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Aitzetmüller-Klietz, M.-L.; Busch, L.; Hamatschek, M.; Paul, M.; Schriek, C.; Wiebringhaus, P.; Aitzetmüller-Klietz, M.; Kückelhaus, M.; Hirsch, T. Understanding the Vicious Circle of Pain, Physical Activity, and Mental Health in Lipedema Patients—A Response Surface Analysis. J Clin Med. 2023, 12, 5319. [Google Scholar] [CrossRef] [PubMed]
- Viana, D. C. P.; Caseri Câmara, L.; Borges Palau, R. Menopause as a Critical Turning Point in Lipedema: The Estrogen Receptor Imbalance, Intracrine Estrogen, and Adipose Tissue Dysfunction Model. Int J Mol Sci 2025, 26, 7074. [Google Scholar] [CrossRef]
- Shiose, K.; Tanabe, Y.; Ohnishi, T.; Takahashi, H. Effect of Regional Muscle Damage and Inflammation Following Eccentric Exercise on Electrical Resistance and the Body Composition Assessment Using Bioimpedance Spectroscopy. J Physiol Sci 2019, 69, 895–901. [Google Scholar] [CrossRef]
- Borga, M.; West, J.; Bell, J.D.; Harvey, N.C.; Romu, T.; Heymsfield, S.B.; Dahlqvist Leinhard, O. Advanced Body Composition Assessment: From Body Mass Index to Body Composition Profiling. J Investig Med 2018, 66, 1–9. [Google Scholar] [CrossRef]
- Taylor, S.L.; Donahue, P.M.C.; Pridmore, M.D.; Garza, M.E.; Patel, N.J.; Custer, C.A.; Luo, Y.; Aday, A.W.; Beckman, J.A.; Donahue, M.J.; et al. Semiautomated Segmentation of Lower Extremity MRI Reveals Distinctive Subcutaneous Adipose Tissue in Lipedema: A Pilot Study. J. Med. Imag. 2023, 10. [Google Scholar] [CrossRef]
- Erbacher, G.; Bertsch, T. Lipoedema and Pain: What is the role of the psyche? – Results of a pilot study with 150 patients with Lipoedema. Phlebologie 2020, 49, 305–316. [Google Scholar] [CrossRef]
- Kunzová, M.; Lagová, E.; Keith, L. Mental and Physical Health Burden and Quality of Life in Czech Women with Lipedema. Front Glob Womens Health 2025, 6, 1629077. [Google Scholar] [CrossRef] [PubMed]
- Janota, B.; Michalska, P.; Janota, K. Lipedema: The Intersection of Physical and Mental Health. Arch Psych Psych 2025, 27, 59–66. [Google Scholar] [CrossRef]
- Annunziata, G.; Paoli, A.; Manzi, V.; Camajani, E.; Laterza, F.; Verde, L.; Capó, X.; Padua, E.; Bianco, A.; Carraro, A.; et al. The Role of Physical Exercise as a Therapeutic Tool to Improve Lipedema: A Consensus Statement from the Italian Society of Motor and Sports Sciences (Società Italiana Di Scienze Motorie e Sportive, SISMeS) and the Italian Society of Phlebology (Società Italiana Di Flebologia, SIF). Curr Obes Rep 2024, 13, 667–679. [Google Scholar] [CrossRef] [PubMed]
- Orr, R.; Fiatarone Singh, M. The Anabolic Androgenic Steroid Oxandrolone in the Treatment of Wasting and Catabolic Disorders: Review of Efficacy and Safety. Drugs 2004, 64, 725–750. [Google Scholar] [CrossRef]
- Ring, J.; Heinelt, M.; Sharma, S.; Letourneau, S.; Jeschke, M.G. Oxandrolone in the Treatment of Burn Injuries: A Systematic Review and Meta-Analysis. J Burn Care Res 2020, 41, 190–199. [Google Scholar] [CrossRef]
- Jeschke, M.G.; Finnerty, C.C.; Suman, O.E.; Kulp, G.; Mlcak, R.P.; Herndon, D.N. The Effect of Oxandrolone on the Endocrinologic, Inflammatory, and Hypermetabolic Responses During the Acute Phase Postburn. Ann Surg 2007, 246, 351–362. [Google Scholar] [CrossRef]
- Grunfeld, C.; Kotler, D.P.; Dobs, A.; Glesby, M.; Bhasin, S. Oxandrolone in the Treatment of HIV-Associated Weight Loss in Men: A Randomized, Double-Blind, Placebo-Controlled Study. J Acquir Immune Defic Syndr 2006, 41, 304–314. [Google Scholar] [CrossRef]
- Pharo, A.; Salvato, P.; Vergel, N.; Carroll, E.; Sauer, L.; Mooney, M. Oxandrolone: Anabolic Steroid Use in HIV Positive Women. Nutrition 1997, 13, 268. [Google Scholar] [CrossRef]
- Fontanarosa, P.B.; Romeyn, M.; Gunn, N., Iii. Resistance Exercise and Oxandrolone for Men With HIV-Related Weight Loss. JAMA 2000, 284, 176. [Google Scholar] [CrossRef]
- Fox-Wheeler, S.; Heller, L.; Salata, C.M.; Kaufman, F.; Loro, M.L.; Gilsanz, V.; Haight, M.; Umman, G.C.; Barton, N.; Church, J.A. Evaluation of the Effects of Oxandrolone on Malnourished HIV-Positive Pediatric Patients. Pediatrics 1999, 104, e73–e73. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.T.; Herndon, D.N.; Tanksley, J.D.; Jennings, K.; Klein, G.L.; Mlcak, R.P.; Clayton, R.P.; Crites, N.N.; Hays, J.P.; Andersen, C.; et al. Five-Year Outcomes after Long-Term Oxandrolone Administration in Severely Burned Children: A Randomized Clinical Trial. Shock 2016, 45, 367–374. [Google Scholar] [CrossRef] [PubMed]
- de Mello Gindri, I.; Almeida, G.; Saraiva, C.; Ferrari, G.; Dallacosta, D.; Roesler, C.R. The Safety and Effectiveness of Oxandrolone on Different Clinical Conditions: A Systematic Review. Endocr. Metab. Sci. 2025, 18. [Google Scholar] [CrossRef]
- Li, H.; Guo, Y.; Yang, Z.; Roy, M.; Guo, Q. The Efficacy and Safety of Oxandrolone Treatment for Patients with Severe Burns: A Systematic Review and Meta-Analysis. Burns 2016, 42, 717–727. [Google Scholar] [CrossRef]
- Jastreboff, A.M.; Aronne, L.J.; Ahmad, N.N.; Wharton, S.; Connery, L.; Alves, B.; Kiyosue, A.; Zhang, S.; Liu, B.; Bunck, M.C.; et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med 2022, 387, 205–216. [Google Scholar] [CrossRef]
| Domain | Operational definition & assessment (examples) | Pathophysiological significance | Suggested objective endpoints |
|---|---|---|---|
| 1. Objective dynapenia (function) | Timed Up and Go (TUG) >10 s; Sit-to-Stand (5×STS or 30 s STS) below age/sex norms; handgrip or quadriceps strength below normative values (specify dynamometer and units). | Dissociation between limb volume and contractile performance, indicating failure of muscle quality rather than simple mass loss. | TUG, STS, handgrip, isokinetic quadriceps torque, gait speed, mobility-related patient-reported outcomes. |
| 2. Mechanical intolerance and refractoriness to loading | Post-exertional pain persisting >24 h; measurable limb volume increase within 24–48 h after activity; persistence of symptoms despite ≥6 months of documented conservative therapy (compression, physiotherapy, diet). | Captures the “exercise paradox,” in which inflammatory and edematous responses limit tolerance to mechanical loading and promote disuse atrophy. | Pain NRS/VAS, post-exertional symptom diary, limb volume change, pressure pain thresholds, accelerometry-based activity metrics. |
| 3. Myosteatosis and impaired muscle quality (structure) | MRI Dixon fat fraction or muscle radiodensity; ultrasound echo-intensity where advanced imaging is unavailable; sodium-MRI markers when feasible. | Links dynapenia to ectopic lipid deposition, inflammatory muscle remodeling, and reduced contractile efficiency. | Muscle fat fraction, radiodensity/echo-intensity, correlation with strength and mobility, longitudinal structural change. |
| 4. Metabolic–inflammatory milieu consistent with oxidative mismatch | Optional enrichment markers: insulin resistance indices, inflammatory cytokines, adipokines, and fibrosis/ECM-related biomarkers. | Tests the proposed lipolysis–oxidation mismatch and immunometabolic drivers sustaining myosteatosis and functional decline. | Metabolic flexibility (RQ/indirect calorimetry), insulin sensitivity, inflammatory and fibrosis-related panels, association with imaging and function. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




