Submitted:
29 January 2026
Posted:
30 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Conventional Transthoracic Echocardiography
2.3. Speckle-Tracking Echocardiography
2.4. Carotid Ultrasonography
2.5. Endpoint Definition
2.6. Statistical Analysis
3. Results
3.1. Clinical Findings
3.2. Conventional Echocardiographic and Carotid Ultrasound Parameters
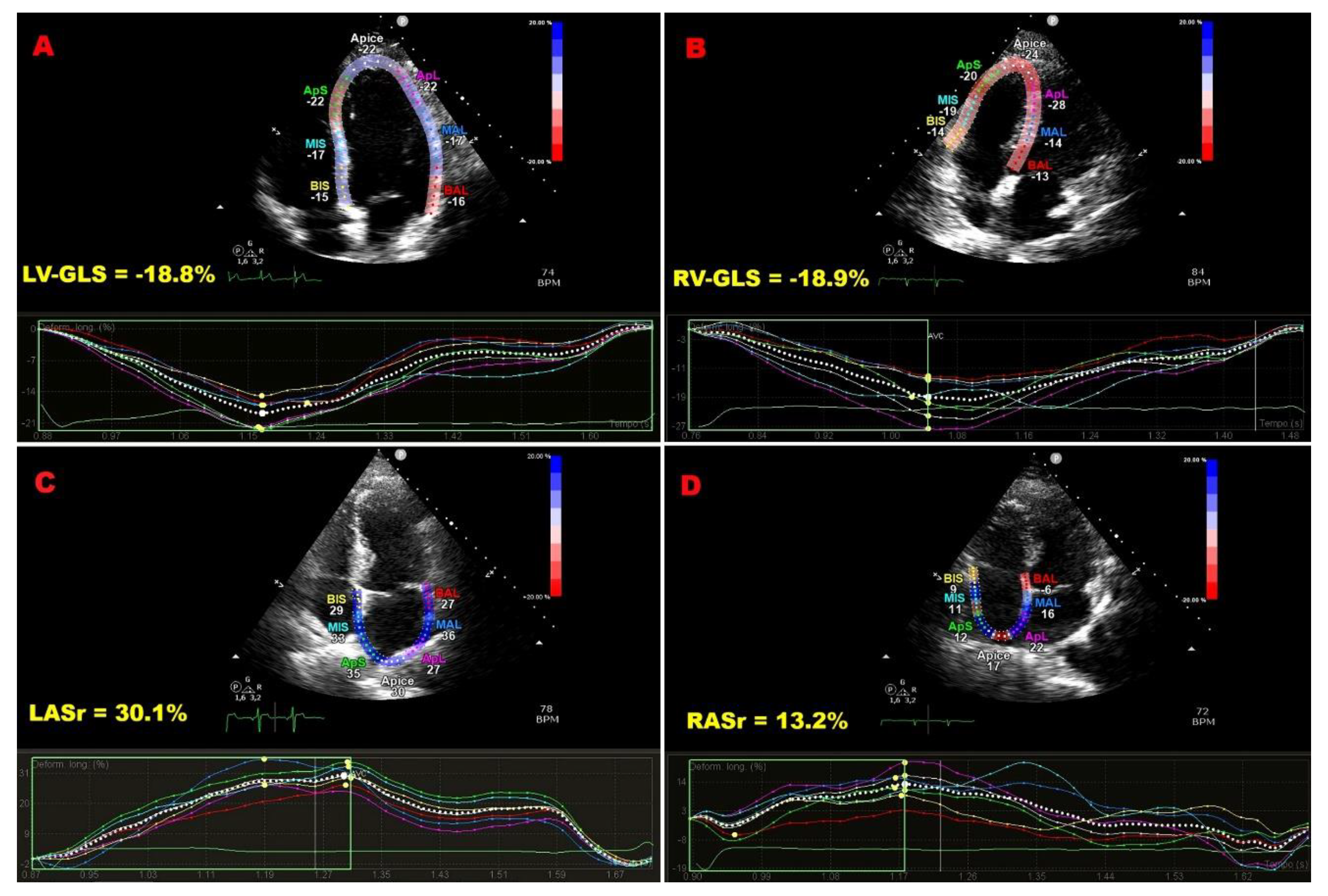
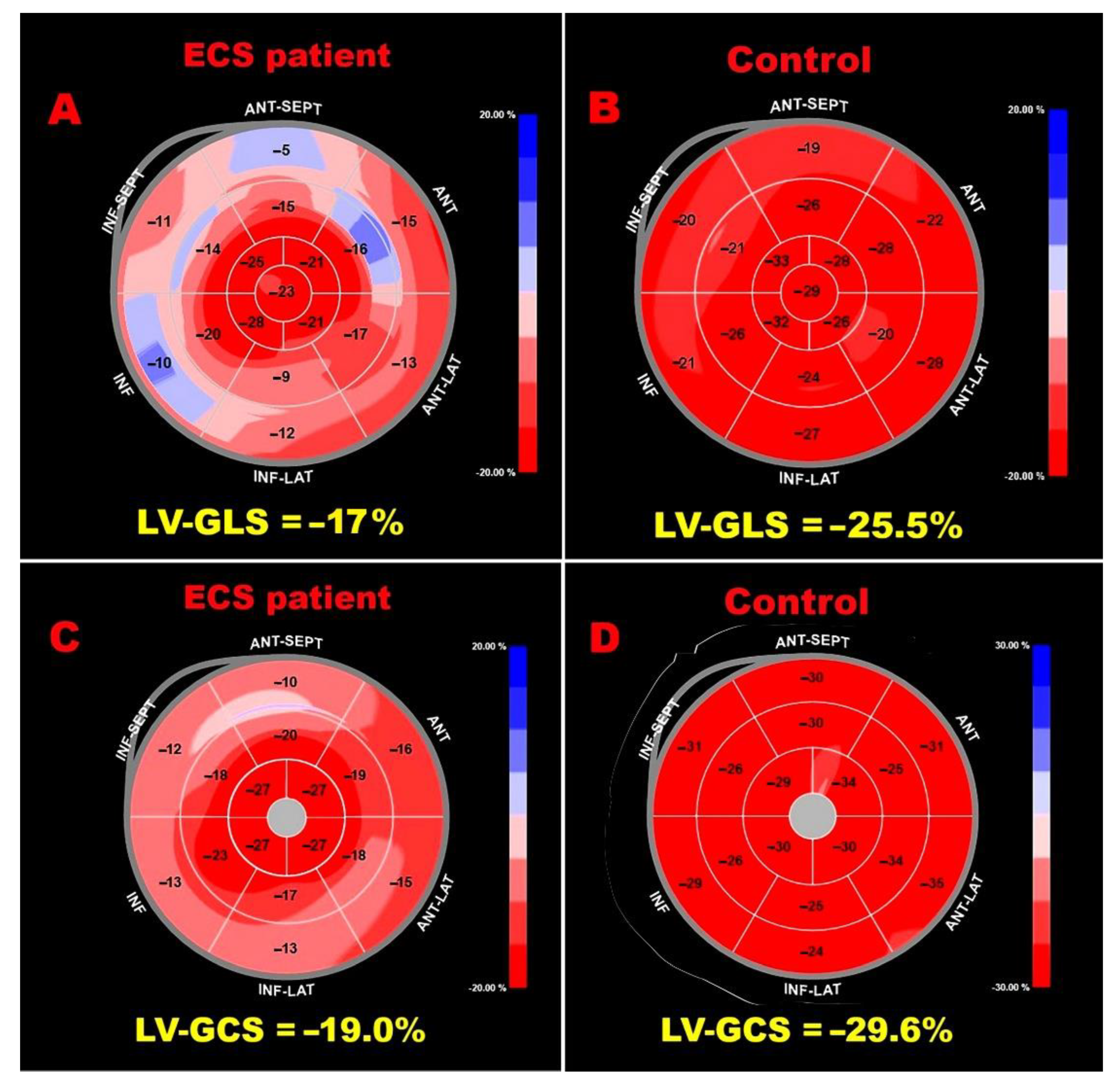
3.3. Speckle-Tracking Echocardiography–Derived Myocardial Strain Parameters
3.4. Variability Analysis
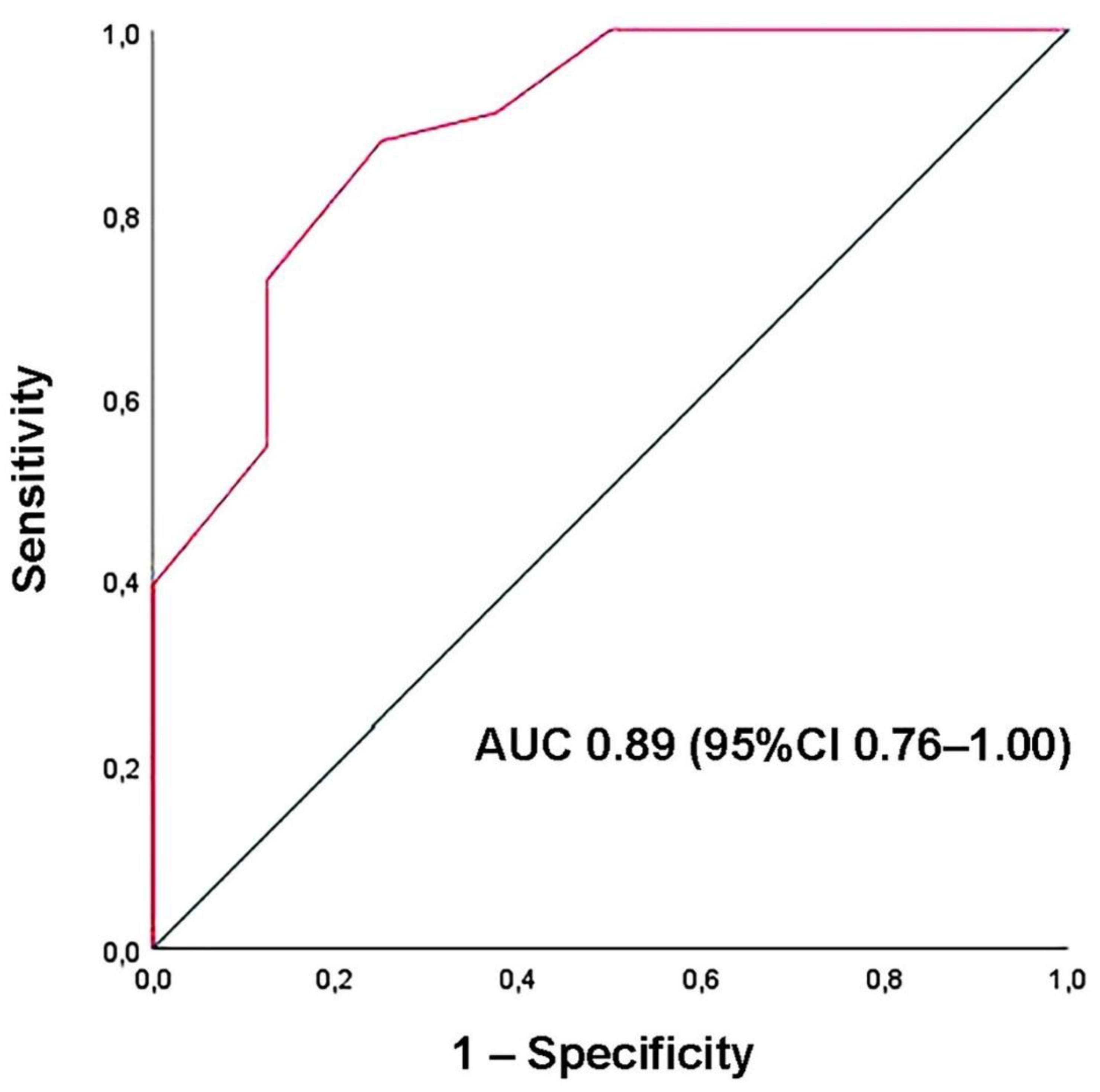
3.5. Univariate and Multivariate Logistic Regression
4. Discussion
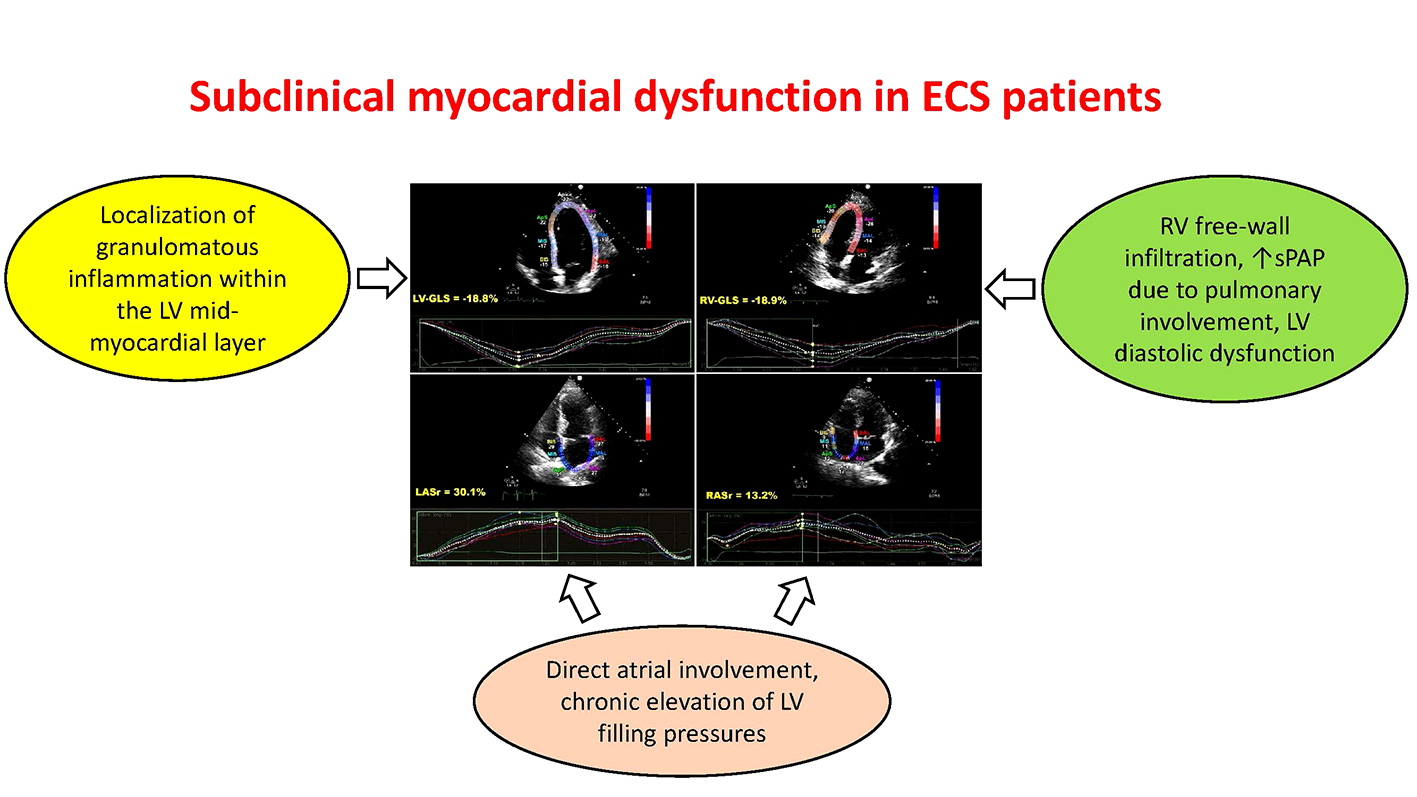
4.1. Main Findings of the Present Study
4.2. Comparison with Previous Studies and Interpretation of Results
4.3. Pathophysiological Mechanisms Underpinning Subclinical Myocardial Dysfunction in Sarcoidosis Patients
4.4. Implications for Clinical Practice
4.5. Study Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jain, R.; Yadav, D.; Puranik, N.; Guleria, R.; Jin, J.O. Sarcoidosis: Causes, Diagnosis, Clinical Features, and Treatments. J. Clin. Med. 2020, 9, 1081. [Google Scholar] [CrossRef] [PubMed]
- Iannuzzi, M.C.; Fontana, J.R. Sarcoidosis: Clinical presentation, immunopathogenesis, and therapeutics. JAMA 2011, 305, 391–399. [Google Scholar] [CrossRef] [PubMed]
- Silverman, K.J.; Hutchins, G.M.; Bulkley, B.H. Cardiac sarcoid: A clinicopathologic study of 84 unselected patients with systemic sarcoidosis. Circulation 1978, 58, 1204–1211. [Google Scholar] [CrossRef] [PubMed]
- Hulten, E.; Aslam, S.; Osborne, M.; Abbasi, S.; Bittencourt, M.S.; Blankstein, R. Cardiac sarcoidosis—State of the art review. Cardiovasc. Diagn. Ther. 2016, 6, 50–63. [Google Scholar] [CrossRef]
- Mankad, P.; Mitchell, B.; Birnie, D.; Kron, J. Cardiac sarcoidosis. Curr. Cardiol. Rep. 2019, 21, 152. [Google Scholar] [CrossRef]
- Jaiswal, R.; Vaisyambath, L.; Khayyat, A.; Unachukwu, N.; Nasyrlaeva, B.; Asad, M.; Fabara, S.P.; Balan, I.; Kolla, S.; Rabbani, R.; et al. Cardiac sarcoidosis diagnostic challenges and management: A case report and literature review. Cureus 2022, 14, e24850. [Google Scholar] [CrossRef]
- Perry, A.; Vuitch, F. Causes of death in patients with sarcoidosis: A morphologic study of 38 autopsies with clinicopathologic correlations. Arch. Pathol. Lab. Med. 1995, 119, 167–172. [Google Scholar]
- Trivieri, M.G.; Spagnolo, P.; Birnie, D.; Liu, P.; Drake, W.; Kovacic, J.C.; Baughman, R.; Fayad, Z.A.; Judson, M.A. Challenges in cardiac and pulmonary sarcoidosis: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2020, 76, 1878–1901. [Google Scholar] [CrossRef]
- Mohsen, A.; Jimenez, A.; Hood, R.E.; Dickfeld, T.; Saliaris, A.; Shorofsky, S.; Saba, M.M. Cardiac sarcoidosis: Electrophysiological outcomes on long-term follow-up and the role of the implantable cardioverter-defibrillator. J. Cardiovasc. Electrophysiol. 2014, 25, 171–176. [Google Scholar] [CrossRef]
- Patel, M.R.; Cawley, P.J.; Heitner, J.F.; Klem, I.; Parker, M.A.; Jaroudi, W.A.; Meine, T.J.; White, J.B.; Elliott, M.D.; Kim, H.W.; et al. Detection of myocardial damage in patients with sarcoidosis. Circulation 2009, 120, 1969–1977. [Google Scholar] [CrossRef]
- Ohira, H.; Birnie, D.H.; Pena, E.; Bernick, J.; McArdle, B.; Leung, E.; Wells, G.A.; Yoshinaga, K.; Tsujino, I.; Sato, T.; et al. Comparison of 18F-fluorodeoxyglucose positron emission tomography and cardiac magnetic resonance in corticosteroid-naive patients with conduction system disease due to cardiac sarcoidosis. Eur. J. Nucl. Med. Mol. Imaging 2016, 43, 259–269. [Google Scholar] [CrossRef]
- Birnie, D.H.; Sauer, W.H.; Bogun, F.; Cooper, J.M.; Culver, D.A.; Duvernoy, C.S.; Judson, M.A.; Kron, J.; Mehta, D.; Cosedis Nielsen, J.; et al. HRS expert consensus statement on the diagnosis and management of arrhythmias associated with cardiac sarcoidosis. Heart Rhythm 2014, 11, 1305–1323. [Google Scholar] [CrossRef]
- Vignaux, O.; Dhote, R.; Duboc, D.; Blanche, P.; Devaux, J.Y.; Weber, S.; Legmann, P. Detection of myocardial involvement in patients with sarcoidosis applying T2-weighted, contrast-enhanced, and cine magnetic resonance imaging: Initial results of a prospective study. J. Comput. Assist. Tomogr. 2002, 26, 762–767. [Google Scholar] [CrossRef]
- Potter, E.; Marwick, T.H. Assessment of left ventricular function by echocardiography: The case for routinely adding global longitudinal strain to ejection fraction. JACC Cardiovasc. Imaging 2018, 11, 260–274. [Google Scholar] [CrossRef]
- Shah, B.N.; De Villa, M.; Khattar, R.S.; Senior, R. Imaging cardiac sarcoidosis: The incremental benefit of speckle tracking echocardiography. Echocardiography 2013, 30, E213–E214. [Google Scholar] [CrossRef]
- Galderisi, M.; Cosyns, B.; Edvardsen, T.; Cardim, N.; Delgado, V.; Di Salvo, G.; Donal, E.; Sade, L.E.; Ernande, L.; Garbi, M.; et al. Standardization of adult transthoracic echocardiography reporting in agreement with recent chamber quantification, diastolic function, and heart valve disease recommendations: An expert consensus document of the European Association of Cardiovascular Imaging. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 1301–1310. [Google Scholar] [CrossRef]
- Kul, S.; Ozcelik, H.K.; Uyarel, H.; Karakus, G.; Guvenc, T.S.; Yalcınsoy, M.; Asoglu, E.; Kemik, A.S.; Tasal, A.; Gungor, S.; et al. Diagnostic value of strain echocardiography, galectin-3, and tenascin-C levels for the identification of patients with pulmonary and cardiac sarcoidosis. Lung 2014, 192, 533–542. [Google Scholar] [CrossRef] [PubMed]
- Orii, M.; Hirata, K.; Tanimoto, T.; Shiono, Y.; Shimamura, K.; Yamano, T.; Ino, Y.; Yamaguchi, T.; Kubo, T.; Tanaka, A.; et al. Myocardial damage detected by two-dimensional speckle-tracking echocardiography in patients with extracardiac sarcoidosis: Comparison with magnetic resonance imaging. J. Am. Soc. Echocardiogr. 2015, 28, 683–691. [Google Scholar] [CrossRef] [PubMed]
- Tigen, K.; Sunbul, M.; Karaahmet, T.; Tasar, O.; Dundar, C.; Yalcinsoy, M.; Takir, M.; Akkaya, E. Early detection of biventricular and atrial mechanical dysfunction using two-dimensional speckle tracking echocardiography in patients with sarcoidosis. Lung 2015, 193, 669–675. [Google Scholar] [CrossRef]
- Joyce, E.; Kamperidis, V.; Ninaber, M.K.; Katsanos, S.; Debonnaire, P.; Schalij, M.J.; Taube, C.; Bax, J.J.; Delgado, V.; Ajmone Marsan, N. Prevalence and correlates of early right ventricular dysfunction in sarcoidosis and its association with outcome. J. Am. Soc. Echocardiogr. 2016, 29, 871–878. [Google Scholar] [CrossRef] [PubMed]
- Murtagh, G.; Laffin, L.J.; Patel, K.V.; Patel, A.V.; Bonham, C.A.; Yu, Z.; Addetia, K.; El-Hangouche, N.; Maffesanti, F.; Mor-Avi, V.; et al. Improved detection of myocardial damage in sarcoidosis using longitudinal strain in patients with preserved left ventricular ejection fraction. Echocardiography 2016, 33, 1344–1352. [Google Scholar] [CrossRef]
- Değirmenci, H.; Demirelli, S.; Arısoy, A.; Ermiş, E.; Araz, Ö.; Bakırcı, E.M.; Hamur, H.; Büyüklü, M.; Topal, E. Myocardial deformation and total atrial conduction time in the prediction of cardiac involvement in patients with pulmonary sarcoidosis. Clin. Respir. J. 2017, 11, 68–77. [Google Scholar] [CrossRef]
- Schouver, E.D.; Moceri, P.; Doyen, D.; Tieulie, N.; Queyrel, V.; Baudouy, D.; Cerboni, P.; Gibelin, P.; Leroy, S.; Fuzibet, J.G.; et al. Early detection of cardiac involvement in sarcoidosis with two-dimensional speckle-tracking echocardiography. Int. J. Cardiol. 2017, 227, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Lei, J.; Scalzetti, E.; McGrath, M.; Feiglin, D.; Voelker, R.; Wang, J.; Iannuzzi, M.C.; Liu, K. Myocardial contractile patterns predict future cardiac events in sarcoidosis. Int. J. Cardiovasc. Imaging 2018, 34, 251–262. [Google Scholar] [CrossRef] [PubMed]
- Felekos, I.; Aggeli, C.; Gialafos, E.; Kouranos, V.; Rapti, A.; Sfikakis, P.; Koulouris, N.; Tousoulis, D. Global longitudinal strain and long-term outcomes in asymptomatic extracardiac sarcoid patients with no apparent cardiovascular disease. Echocardiography 2018, 35, 804–808. [Google Scholar] [CrossRef]
- Di Stefano, C.; Bruno, G.; Arciniegas Calle, M.C.; Acharya, G.A.; Fussner, L.M.; Ungprasert, P.; Cooper, L.T., Jr.; Blauwet, L.A.; Ryu, J.H.; Pellikka, P.A.; et al. Diagnostic and predictive value of speckle tracking echocardiography in cardiac sarcoidosis. BMC Cardiovasc. Disord. 2020, 20, 21. [Google Scholar] [CrossRef]
- Bayat, F.; Fahimi, A.; Tavana, S.; Tabary, M.; Khaheshi, I. Subclinical involvement of the heart and its associated factors in patients with sarcoidosis with normal systolic function using two-dimensional speckle tracking. Echocardiography 2020, 37, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Kaptan Ozen, D.; Mutlu, B.; Kocakaya, D.; Turan, B.; Sert Sekerci, S.; Ceyhan, B.; Kepez, A.; Erdogan, O. The effect of global longitudinal strain on impaired six-minute walk test performance in patients with sarcoidosis. Sarcoidosis Vasc. Diffuse Lung Dis. 2020, 37, 66–73. [Google Scholar] [CrossRef]
- Kusunose, K.; Fujiwara, M.; Yamada, H.; Nishio, S.; Saijo, Y.; Yamada, N.; Hirata, Y.; Torii, Y.; Ise, T.; Yamaguchi, K.; et al. Deterioration of biventricular strain is an early marker of cardiac involvement in confirmed sarcoidosis. Eur. Heart J. Cardiovasc. Imaging 2020, 21, 796–804. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. 2016 European guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef]
- Crouser, E.D.; Maier, L.A.; Wilson, K.C.; Bonham, C.A.; Morgenthau, A.S.; Patterson, K.C.; Abston, E.; Bernstein, R.C.; Blankstein, R.; Chen, E.S.; et al. Diagnosis and detection of sarcoidosis: An official American Thoracic Society clinical practice guideline. Am. J. Respir. Crit. Care Med. 2020, 201, e26–e51. [Google Scholar] [CrossRef] [PubMed]
- Baughman, R.P.; Valeyre, D.; Korsten, P.; Mathioudakis, A.G.; Wuyts, W.A.; Wells, A.; Rottoli, P.; Nunes, H.; Lower, E.E.; Judson, M.A.; et al. ERS clinical practice guidelines on treatment of sarcoidosis. Eur. Respir. J. 2021, 58, 2004079. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Bosch, J.P.; Lewis, J.B.; Greene, T.; Rogers, N.; Roth, D. A more accurate method to estimate glomerular filtration rate from serum creatinine: A new prediction equation. Ann. Intern. Med. 1999, 130, 461–470. [Google Scholar] [CrossRef] [PubMed]
- Lang, R.M.; Badano, L.P.; Mor-Avi, V.; Afilalo, J.; Armstrong, A.; Ernande, L.; Flachskampf, F.A.; Foster, E.; Goldstein, S.A.; Kuznetsova, T.; et al. Recommendations for cardiac chamber quantification by echocardiography in adults: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2015, 28, 1–39.e14. [Google Scholar] [CrossRef]
- Nagueh, S.F.; Smiseth, O.A.; Appleton, C.P.; Byrd, B.F., III; Dokainish, H.; Edvardsen, T.; Flachskampf, F.A.; Gillebert, T.C.; Klein, A.L.; Lancellotti, P.; et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: An update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J. Am. Soc. Echocardiogr. 2016, 29, 277–314. [Google Scholar] [CrossRef]
- Humbert, M.; Kovacs, G.; Hoeper, M.M.; Badagliacca, R.; Berger, R.M.F.; Brida, M.; Carlsen, J.; Coats, A.J.S.; Escribano-Subias, P.; Ferrari, P.; et al. 2022 ESC/ERS guidelines for the diagnosis and treatment of pulmonary hypertension. Eur. Respir. J. 2023, 61, 2200879. [Google Scholar] [CrossRef]
- Nishimura, R.A.; Otto, C.M.; Bonow, R.O.; Carabello, B.A.; Erwin, J.P., III; Fleisher, L.A.; Jneid, H.; Mack, M.J.; McLeod, C.J.; O’Gara, P.T.; et al. 2017 AHA/ACC focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease. Circulation 2017, 135, e1159–e1195. [Google Scholar] [CrossRef]
- Voigt, J.U.; Pedrizzetti, G.; Lysyansky, P.; Marwick, T.H.; Houle, H.; Baumann, R.; Pedri, S.; Ito, Y.; Abe, Y.; Metz, S.; et al. Definitions for a common standard for two-dimensional speckle tracking echocardiography: Consensus document of the EACVI/ASE/Industry Task Force. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 1–11. [Google Scholar] [CrossRef]
- Badano, L.P.; Muraru, D.; Parati, G.; Haugaa, K.; Voigt, J.U. How to do right ventricular strain. Eur. Heart J. Cardiovasc. Imaging 2020, 21, 825–827. [Google Scholar] [CrossRef]
- Voigt, J.U.; Mălăescu, G.G.; Haugaa, K.; Badano, L. How to do left atrial strain. Eur. Heart J. Cardiovasc. Imaging 2020, 21, 715–717. [Google Scholar] [CrossRef]
- Sonaglioni, A.; Vincenti, A.; Baravelli, M.; Rigamonti, E.; Tagliabue, E.; Bassi, P.; Nicolosi, G.L.; Anzà, C.; Lombardo, M. Prognostic value of global left atrial peak strain in patients with acute ischemic stroke and no evidence of atrial fibrillation. Int. J. Cardiovasc. Imaging 2019, 35, 603–613. [Google Scholar] [CrossRef]
- Yingchoncharoen, T.; Agarwal, S.; Popović, Z.B.; Marwick, T.H. Normal ranges of left ventricular strain: A meta-analysis. J. Am. Soc. Echocardiogr. 2013, 26, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Muraru, D.; Onciul, S.; Peluso, D.; Soriani, N.; Cucchini, U.; Aruta, P.; Romeo, G.; Cavalli, G.; Iliceto, S.; Badano, L.P. Sex- and method-specific reference values for right ventricular strain by two-dimensional speckle-tracking echocardiography. Circ. Cardiovasc. Imaging 2016, 9, e003866. [Google Scholar] [CrossRef] [PubMed]
- Pathan, F.; D’Elia, N.; Nolan, M.T.; Marwick, T.H.; Negishi, K. Normal ranges of left atrial strain by speckle-tracking echocardiography: A systematic review and meta-analysis. J. Am. Soc. Echocardiogr. 2017, 30, 59–70.e8. [Google Scholar] [CrossRef] [PubMed]
- Krittanawong, C.; Maitra, N.S.; Hassan Virk, H.U.; Farrell, A.; Hamzeh, I.; Arya, B.; Pressman, G.S.; Wang, Z.; Marwick, T.H. Normal ranges of right atrial strain: A systematic review and meta-analysis. JACC Cardiovasc. Imaging 2023, 16, 282–294. [Google Scholar] [CrossRef]
- Stein, J.H.; Korcarz, C.E.; Hurst, R.T.; Lonn, E.; Kendall, C.B.; Mohler, E.R.; Najjar, S.S.; Rembold, C.M.; Post, W.S.; et al. Use of carotid ultrasound to identify subclinical vascular disease and evaluate cardiovascular disease risk: A consensus statement from the American Society of Echocardiography Carotid Intima-Media Thickness Task Force. J. Am. Soc. Echocardiogr. 2008, 21, 93–111. [Google Scholar] [CrossRef]
- Sonaglioni, A.; Fagiani, V.; Rigoni, M.; Nicolosi, G.L.; Lucidi, A.; Caminati, A.; Lombardo, M.; Harari, S. Echocardiographic evaluation of left ventricular mechanics in sarcoidosis patients without overt heart disease: A systematic review and meta-analysis. Sarcoidosis Vasc. Diffuse Lung Dis. 2024, 41, e2024042. [Google Scholar] [CrossRef]
- Phelan, D.; Collier, P.; Thavendiranathan, P.; Popović, Z.B.; Hanna, M.; Plana, J.C.; Marwick, T.H.; Thomas, J.D. Relative apical sparing of longitudinal strain using two-dimensional speckle-tracking echocardiography is both sensitive and specific for the diagnosis of cardiac amyloidosis. Heart 2012, 98, 1442–1448. [Google Scholar] [CrossRef]
- Leitman, M.; Tyomkin, V. Apical sparing in routine echocardiography: Occurrence and clinical significance. J. Cardiovasc. Dev. Dis. 2024, 11, 262. [Google Scholar] [CrossRef]
- Ferkh, A.; Tjahjadi, C.; Stefani, L.; Geenty, P.; Byth, K.; De Silva, K.; Boyd, A.C.; Richards, D.; Mollee, P.; Korczyk, D.; et al. Cardiac “hypertrophy” phenotyping: Differentiating aetiologies with increased left ventricular wall thickness on echocardiography. Front. Cardiovasc. Med. 2023, 10, 1183485. [Google Scholar] [CrossRef]
- Sonaglioni, A.; Fagiani, V.; Nicolosi, G.L.; Lombardo, M. Echocardiographic assessment of left ventricular mechanics in individuals with mitral valve prolapse: A systematic review and meta-analysis. Int. J. Cardiovasc. Imaging 2024, 40, 1617–1629. [Google Scholar] [CrossRef] [PubMed]
- Stokke, T.M.; Hasselberg, N.E.; Smedsrud, M.K.; Sarvari, S.I.; Haugaa, K.H.; Smiseth, O.A.; Edvardsen, T.; Remme, E.W. Geometry as a confounder when assessing ventricular systolic function: Comparison between ejection fraction and strain. J. Am. Coll. Cardiol. 2017, 70, 942–954. [Google Scholar] [CrossRef]
- Yilmaz, Y.; Kul, S.; Kavas, M.; Erman, H.; Aciksari, G.; Ozcan, F.B.; Yalcinkaya, E.; Kanbay, A.; Caliskan, M. Is there an association between sarcoidosis and atherosclerosis? Int. J. Cardiovasc. Imaging 2021, 37, 559–567. [Google Scholar] [CrossRef]
- Satny, M.; Hubacek, J.A.; Vrablik, M. Statins and inflammation. Curr. Atheroscler. Rep. 2021, 23, 80. [Google Scholar] [CrossRef]
- Liberale, L.; Carbone, F.; Montecucco, F.; Sahebkar, A. Statins reduce vascular inflammation in atherogenesis: A review of underlying molecular mechanisms. Int. J. Biochem. Cell Biol. 2020, 122, 105735. [Google Scholar] [CrossRef]
- Lehtonen, J.; Uusitalo, V.; Pöyhönen, P.; Mäyränpää, M.I.; Kupari, M. Cardiac sarcoidosis: Phenotypes, diagnosis, treatment, and prognosis. Eur. Heart J. 2023, 44, 1495–1510. [Google Scholar] [CrossRef]
- Kitai, T.; Nabeta, T.; Naruse, Y.; Taniguchi, T.; Yoshioka, K.; Miyakoshi, C.; Kurashima, S.; Miyoshi, Y.; Tanaka, H.; Okumura, T.; et al. Comparisons between biopsy-proven versus clinically diagnosed cardiac sarcoidosis. Heart 2022, 108, 1887–1894. [Google Scholar] [CrossRef]
- Lynch, J.P., III; Hwang, J.; Bradfield, J.; Fishbein, M.; Shivkumar, K.; Tung, R. Cardiac involvement in sarcoidosis: Evolving concepts in diagnosis and treatment. Semin. Respir. Crit. Care Med. 2014, 35, 372–390. [Google Scholar] [CrossRef]
- Aggeli, C.; Felekos, I.; Tousoulis, D.; Gialafos, E.; Rapti, A.; Stefanadis, C. Myocardial mechanics for the early detection of cardiac sarcoidosis. Int. J. Cardiol. 2013, 168, 4820–4821. [Google Scholar] [CrossRef] [PubMed]
- Fahy, G.J.; Marwick, T.; McCreery, C.J.; Quigley, P.J.; Maurer, B.J. Doppler echocardiographic detection of left ventricular diastolic dysfunction in patients with pulmonary sarcoidosis. Chest 1996, 109, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Aydin Kaderli, A.; Gullulu, S.; Coskun, F.; Yilmaz, D.; Uzaslan, E. Impaired left ventricular systolic and diastolic functions in patients with early grade pulmonary sarcoidosis. Eur. J. Echocardiogr. 2010, 11, 809–813. [Google Scholar] [CrossRef]
- Kato, Y.; Morimoto, S.; Uemura, A.; Hiramitsu, S.; Ito, T.; Hishida, H. Efficacy of corticosteroids in sarcoidosis presenting with atrioventricular block. Sarcoidosis Vasc. Diffuse Lung Dis. 2003, 20, 133–137. [Google Scholar] [PubMed]
- Yodogawa, K.; Seino, Y.; Ohara, T.; Takayama, H.; Katoh, T.; Mizuno, K. Effect of corticosteroid therapy on ventricular arrhythmias in patients with cardiac sarcoidosis. Ann. Noninvasive Electrocardiol. 2011, 16, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Nakano, S.; Kimura, F.; Osman, N.; Sugi, K.; Tanno, J.; Uchida, Y.; Shiono, A.; Senbonmatsu, T.; Nishimura, S. Improved myocardial strain measured by strain-encoded magnetic resonance imaging in a patient with cardiac sarcoidosis. Can. J. Cardiol. 2013, 29, 1531.e9–1531.e11. [Google Scholar] [CrossRef]
- Mirea, O.; Pagourelias, E.D.; Duchenne, J.; Bogaert, J.; Thomas, J.D.; Badano, L.P.; Voigt, J.U.; et al. Intervendor differences in the accuracy of detecting regional functional abnormalities: A report from the EACVI-ASE strain standardization task force. JACC Cardiovasc. Imaging 2018, 11, 25–34. [Google Scholar] [CrossRef]
- Negishi, T.; Negishi, K.; Thavendiranathan, P.; Cho, G.Y.; Popescu, B.A.; Vinereanu, D.; Kurosawa, K.; Penicka, M.; Marwick, T.H.; et al. Effect of experience and training on the concordance and precision of strain measurements. JACC Cardiovasc. Imaging 2017, 10, 518–522. [Google Scholar] [CrossRef] [PubMed]
- Rösner, A.; Barbosa, D.; Aarsæther, E.; Kjønås, D.; Schirmer, H.; D’hooge, J. The influence of frame rate on two-dimensional speckle-tracking strain measurements: A study on in silico-simulated models and images recorded in patients. Eur. Heart J. Cardiovasc. Imaging 2015, 16, 1137–1147. [Google Scholar] [CrossRef]
- Sonaglioni, A.; Fagiani, V.; Nicolosi, G.L.; Lombardo, M. The influence of pectus excavatum on biventricular mechanics: A systematic review and meta-analysis. Minerva Cardiol. Angiol. 2024. [Google Scholar] [CrossRef]
| CLINICAL VARIABLES | Sarcoidosis patients (n = 41) | Controls (n = 30) |
P Value |
|---|---|---|---|
| Demographics and anthropometrics | |||
| Age (yrs) | 57.4 ± 10.2 | 58.5 ± 11.1 | 0.67 |
| Male sex (%) | 24 (58.5) | 16 (53.3) | 0.85 |
| BSA (m2) | 1.96 ± 0.23 | 1.92 ± 0.13 | 0.36 |
| BMI (Kg/m2) | 27.9 ± 4.9 | 26.3 ± 3.5 | 0.11 |
| Cardiovascular risk factors | |||
| Smoking (%) | 13 (31.7) | 10 (33.3) | 0.89 |
| Hypertension (%) | 18 (43.9) | 15 (50.0) | 0.61 |
| Type 2 diabetes (%) | 4 (9.7) | 7 (23.3) | 0.18 |
| Dyslipidemia (%) | 11 (26.8) | 8 (26.7) | 0.99 |
| Clinical staging | |||
| I (%) | 11 (26.8) | / | / |
| II (%) | 23 (56.1) | / | / |
| III (%) | 7 (17.1) | / | / |
| Yrs from diagnosis | 7.2 (1-20) | / | / |
| Spirometry parameters | |||
| FVC (l) | 3.7 ± 1.1 | / | / |
| FVC (%) | 91.3 ± 14.4 | / | / |
| FEV (l) | 2.79 (1.21-6.04) | / | / |
| FEV1 (%) | 88.4 ± 17.6 | / | / |
| FEV1/FVC ratio | 76.1 ± 8.9 | / | / |
| TLC (l) | 5.5 ± 1.4 | / | / |
| TLC (%) | 88 (50-116) | / | / |
| DLCO (ml/min/mmHg) | 6.7 ± 1.5 | / | / |
| DLCO (%) | 81.8 ± 14.1 | / | / |
| Normal pattern (%) | 22 (53.6) | / | / |
| Obstructive pattern (%) | 9 (21.9) | / | / |
| Restrictive pattern (%) | 5 (12.2) | / | / |
| Mixed pattern (%) | 1 (2.4) | / | / |
| Impaired diffusion capacity (%) | 10 (24.4) | / | / |
| 6MWT (m) | 480.2 ± 93.9 | / | / |
| ΔSaO2 (%) | 4.0 ± 2.9 | / | / |
| Blood tests | |||
| Serum hemoglobin (g/dl) | 13.6 ± 1.3 | 13.7 ± 1.2 | 0.74 |
| RDW (%) | 13.3 ± 0.9 | 13.1 ± 0.8 | 0.33 |
| NLR | 0.63 ± 0.14 | 0.59 ± 0.12 | 0.20 |
| Serum creatinine (mg/dl) | 0.85 ± 0.17 | 0.90 ± 0.14 | 0.18 |
| eGFR (ml/min/1.73 m2) | 87.9 ± 18.4 | 93.8 ± 15.5 | 0.15 |
| Serum glucose (mg/dl) | 95.5 ± 11.4 | 100.1 ± 18.2 | 0.23 |
| Serum total cholesterol (mg/dl) | 196.6 ± 52.7 | 204.1 ± 43.4 | 0.51 |
| Comorbidities | |||
| Cancers (%) | 1 (2.4) | 2 (6.7) | 0.57 |
| COPD (%) | 3 (7.3) | 1 (3.3) | 0.63 |
| OSAS (%) | 3 (7.3) | 1 (3.3) | 0.63 |
| GERD (%) | 6 (14.6) | 6 (20.0) | 0.74 |
| Rheumatoid arthritis (%) | 4 (9.7) | 1 (3.3) | 0.39 |
| Hypothyroidism (%) | 4 (9.7) | 4 (13.3) | 0.71 |
| Mixed anxiety–depressive disorder (%) | 1 (2.4) | 3 (10.0) | 0.32 |
| Hemodynamics | |||
| Heart rate (bpm) | 72.1 ± 12.2 | 69.0 ± 8.4 | 0.23 |
| SBP (mmHg) | 129.8 (104-190) | 132.5 (105-165) | 0.46 |
| DBP (mmHg) | 77.6 (63-100) | 79.3 (65-100) | 0.45 |
| Respiratory treatment | |||
| Oxygen therapy (%) | 1 (2.4) | / | / |
| Oral corticosteroids (%) | 14 (34.1) | / | / |
| Inhalation therapy (%) | 14 (34.1) | / | / |
| Immunosuppressants (%) | 3 (7.3) | / | / |
| Nonrespiratory treatment | |||
| Antiplatelets (%) | 4 (9.7) | 4 (13.3) | 0.72 |
| Anticoagulants (%) | 1 (2.4) | 2 (6.7) | 0.57 |
| ACEi-ARBs (%) | 13 (31.7) | 11 (36.7) | 0.66 |
| Calcium channel blockers (%) | 6 (14.6) | 6 (20.0) | 0.57 |
| Beta blockers (%) | 11 (26.8) | 12 (40.0) | 0.25 |
| Diuretics (%) | 6 (14.6) | 7 (23.3) | 0.37 |
| Statins (%) | 6 (14.6) | 12 (40.0) | 0.02 |
| Hydroxychloroquine (%) | 4 (9.7) | 1 (3.3) | 0.39 |
| Oral hypoglycaemic agents (%) | 4 (9.7) | 7 (23.3) | 0.11 |
| Proton pump inhibitors (%) | 6 (14.6) | 6 (20.0) | 0.74 |
| Conventional TTE variables | Sarcoidosis (n = 41) | Controls (n = 30) | P value |
|---|---|---|---|
| LV size and systolic function | |||
| IVS thickness (mm) | 11.5 ± 2.3 | 11.3 ± 2.6 | 0.74 |
| PW thickness (mm) | 8.6 ± 1.3 | 8.7 ± 1.5 | 0.77 |
| LVEDD (mm) | 47.2 ± 4.1 | 48.2 ± 3.6 | 0.28 |
| RWT | 0.37 ± 0.06 | 0.36 ± 0.07 | 0.53 |
| LVMi (g/m2) | 87.1 (40.2–165.8) | 89.3 (48–123) | 0.70 |
| LVEDVi (ml/m2) | 39.2 (24.2–71.9) | 42.5 (29.3–58.8) | 0.13 |
| LVESVi (ml/m2) | 13.6 (7.0–30.8) | 15.2 (12.6–17.7) | 0.13 |
| LVEF (%) | 65.8 (45–75) | 64.2 (57–70) | 0.21 |
| LV geometric pattern | |||
| Normal (%) | 29 (70.7) | 20 (66.7) | 0.80 |
| Concentric remodeling (%) | 7 (17.1) | 2 (6.7) | 0.29 |
| Eccentric hypertrophy (%) | 3 (7.3) | 5 (16.6) | 0.27 |
| Concentric hypertrophy (%) | 2 (4.9) | 3 (10.0) | 0.64 |
| LV diastolic function | |||
| E/A ratio | 0.87 (0.51–2.0) | 0.80 ± 0.20 | 0.26 |
| E/average e’ ratio | 10.1 (6.0–21.5) | 8.5 (4.5–12.4) | 0.03 |
| LA size | |||
| LA A-P diameter (mm) | 40.8 (27.5–62.0) | 40.4 (35–46) | 0.73 |
| LA longitudinal diamerer (mm) | 51.7 (40.0–89.0) | 50.3 (42–55) | 0.37 |
| LAVi (ml/m2) | 28.5 (14.8–90.6) | 32.1 (22.0–43.3) | 0.16 |
| Valvular regurgitation | |||
| More than mild MR (%) | 2 (4.9) | 3 (13.3) | 0.64 |
| More than mild AR (%) | 1 (2.4) | 3 (13.3) | 0.30 |
| More than mild TR (%) | 5 (12.2) | 5 (16.6) | 0.73 |
| RV size and systolic function | |||
| RVIT (mm) | 30.6 (24.5–52.0) | 31.1 (25–37) | 0.59 |
| TAPSE (mm) | 22.4 ± 3.7 | 22.5 ± 3.0 | 0.90 |
| Pulmonary hemodynamics | |||
| TRV (m/sec) | 2.45 (2.0–2.95) | 2.47 (2.18–2.74) | 0.71 |
| sPAP (mmHg) | 29.3 (21–39) | 30.1 (24–40) | 0.49 |
| Aortic dimensions | |||
| Aortic root (mm) | 34.8 (27–49) | 34.2 (29–44) | 0.56 |
| Ascending aorta (mm) | 33.4 (26.5–41.0) | 33.1 (28–42.5) | 0.76 |
| Aortic arch (mm) | 28.3 (22.5–35.0) | 27.5 (23.5–34) | 0.21 |
| Carotid ultrasound variables | |||
| CCA-EDD (mm) | 7.6 (6.2–9.5) | 7.5 (7.0–8.3) | 0.49 |
| CCA-IMT (mm) | 0.99 (0.55–1.50) | 0.92 (0.60–1.10) | 0.09 |
| Carotid RWT | 0.26 (0.18–0.32) | 0.24 (0.16–0.30) | 0.04 |
| Carotid CSA (mm2) | 26.9 (12.8–47.1) | 24.4 (15.3–29.2) | 0.08 |
| Left carotid artery stenosis (%) | 30 (15–45) | 22.5 (10–40) | 0.001 |
| Right carotid artery stenosis (%) | 28.6 (20–45) | 23.3 (10–45) | 0.02 |
| STE variables | Sarcoidosis (n = 41) | Controls (n = 30) | P value |
|---|---|---|---|
| LV regional and global longitudinal strain | |||
| LS 4C view (%) | –18.5 ± 2.5 | –20.7 ± 2.7 | <0.001 |
| LS 2C view (%) | –18.4 ± 3.2 | –20.5 ± 2.9 | 0.005 |
| LS 3C view (%) | –17.4 ± 3.6 | –20.1 ± 3.1 | 0.001 |
| GLS (%) | –18.1 ± 2.6 | –20.4 ± 2.9 | 0.001 |
| LV-GLS less negative than -20% (%) | 33 (80.5) | 6 (20.0) | <0.001 |
| LV regional and global longitudinal strain rate | |||
| LSR 4C view (s-1) | –1.01 (–0.7,–1.3) | –1.15 (–0,97,–1.33) | 0.001 |
| LSR 2C view (s-1) | –0.98 ± 0.14 | –1.13 ± 0.16 | <0.001 |
| LSR 3C view (s-1) | –1.00 (–0.6,–1.3) | –1.11 (–0.94,–1.28) | 0.01 |
| GLSR (s-1) | –1.00 ± 0.11 | –1.13 ± 0.17 | <0.001 |
| LV regional and global circumferential strain | |||
| CS at base level (%) | –16.5 ± 4.5 | –21.3 ± 2.2 | <0.001 |
| CS at papillary muscles level (%) | –19.4 ± 4.5 | –24.5 ± 2.9 | <0.001 |
| CS at apex level (%) | –27.2 ± 6.2 | –26.5 ± 6.4 | 0.65 |
| GCS (%) | –20.2 (–13.6,–29.6) | –24.1 (–20,3,–32.9) | <0.001 |
| LV-GCS less negative than -23.3% (%) | 8 (19.5) | 3 (10.0) | 0.34 |
| LV regional and global circumferential strain rate | |||
| CSR 4C view (s-1) | –1.14 ± 0.20 | –1.37 ± 0.22 | <0.001 |
| CSR 2C view (s-1) | –1.29 ± 0.26 | –1.65 ± 0.29 | <0.001 |
| CSR 3C view (s-1) | –1.80 ± 0.36 | –1.78 ± 0.35 | 0.81 |
| GCSR (s-1) | –1.34 ± 0.23 | –1.60 ± 0.29 | <0.001 |
| LA phasic strain | |||
| LAScd (%) | 22.9 (5.7–36.8) | 28.4 (21–37.5) | <0.001 |
| LASct (%) | –7.4 (–1,–19) | –9.6 (–1,–16.4) | 0.07 |
| LASr (%) | 30.2 (17–55.8) | 38.1 (27.8–46) | <0.001 |
| LASr less positive than 39% (%) | 29 (70.7) | 6 (20.0) | <0.001 |
| LA phasic strain rate | |||
| SRs (s-1) | 1.61 (0.6–2.7) | 1.98 (1.2–3.8) | 0.008 |
| SRe (s-1) | –1.65 (–0.9,–3.7) | –2.47 (–1.7,–4.2) | <0.001 |
| SRl (s-1) | –2.44 ± 0.66 | 2.74 ± 0.56 | 0.04 |
| LASr/E/average e’ ratio (%) | 3.56 ± 1.34 | 4.58 ± 1.37 | 0.003 |
| RV regional and global strain | |||
| FWLS (%) | –17.5 (–12,–27.5) | –24.6 (–19.0,–29.6) | <0.001 |
| GLS (%) | –16.8 (–11.5,–26.5) | –23.5 (–18,–28.5) | <0.001 |
| RV-GLS less negative than -20% (%) | 35 (85.4) | 2 (6.7) | <0.001 |
| RV global strain rate | |||
| GLSR (s-1) | –1.06 ± 0.18 | 1.22 ± 0.19 | <0.001 |
| RA phasic strain | |||
| RAScd (%) | 21.6 ± 7.5 | 25.5 ± 5.8 | 0.02 |
| RASct (%) | –6.9 (–1,–20) | –9.7 (–4,–25) | 0.02 |
| RASr (%) | 28.5 (17–49.4) | 35.2 ± 7.7 | <0.001 |
| RASr less positive than 35% (%) | 28 (68.3) | 5 (16.7) | <0.001 |
| RA phasic strain rate | |||
| SRs (s-1) | 1.79 (1–3.3) | 2.01 (1.21–3.51) | 0.12 |
| SRe (s-1) | –1.46 ± 0.41 | –1.68 ± 0.44 | 0.04 |
| SRl (s-1) | –1.85 ± 0.56 | –2.15 ± 0.61 | 0.04 |
| Timing (minutes) | 9.1 ± 1.5 | 8.0 ± 1.8 | 0.009 |
| LV-GLS | LV-GCS | |||||
|---|---|---|---|---|---|---|
| PATIENT LIST | INITIAL MEASUREMENT | REMEASUREMENTS | INITIAL MEASUREMENT | REMEASUREMENTS | ||
| Rater 1 | Rater 2 | Rater 1 | Rater 2 | |||
| 1) L.B. | 15.3 | 16 | 16.5 | 20.4 | 20.8 | 22 |
| 2) A.D. | 16.8 | 17 | 17.1 | 19 | 19.5 | 21 |
| 3) A.C. | 19 | 18 | 17 | 22.7 | 20.3 | 19.5 |
| 4) F.N. | 15.1 | 13.5 | 12 | 13.6 | 15 | 15.6 |
| 5) M.L. | 19.6 | 20 | 20.2 | 13.8 | 13.1 | 12.6 |
| 6) M.B. | 16.6 | 16.9 | 17 | 14.7 | 13.5 | 13 |
| 7) C.M. | 18.4 | 18.5 | 18.8 | 15.4 | 15 | 14.5 |
| 8) M.F. | 18 | 17 | 16.5 | 16.5 | 16 | 15 |
| 9) F.B. | 18.9 | 18 | 16 | 17 | 16.1 | 15.8 |
| 10) L.S. | 16.4 | 15.5 | 15 | 17.1 | 17 | 17.1 |
| 11) S.R. | 19.8 | 19 | 19 | 17.3 | 16.7 | 16.5 |
| 12) G.S. | 24.4 | 23.5 | 23 | 17.5 | 17 | 16.8 |
| 13) M.P. | 16.2 | 17 | 17.1 | 17.5 | 18 | 18.1 |
| 14) P.B. | 19.2 | 20 | 20.2 | 18 | 18.3 | 18.9 |
| 15) D.S. | 17.9 | 18.8 | 18.9 | 18.2 | 17.5 | 17 |
| ICC (95%CI) | 0.93 (0.81–0.98) |
0.82 (0.54–0.93) |
0.92 (0.80–0.97) |
0.83 (0.56–0.94) |
||
| UNIVARIATE LOGISTIC REGRESSION ANALYSIS |
MULTIVARIATE LOGISTIC REGRESSION ANALYSIS |
|||||
|---|---|---|---|---|---|---|
| VARIABLES | OR | 95% CI | P value | OR | 95% CI | P value |
| Male sex | 6.00 | 1.04–34.7 | 0.046 | 3.04 | 0.26–35.5 | 0.37 |
| Years from diagnosis | 2.27 | 1.26–4.09 | 0.006 | 2.26 | 1.10–4.65 | 0.03 |
| NLR | 1.10 | 1.01–1.20 | 0.025 | 1.12 | 0.98–1.28 | 0.10 |
| SBP (mmHg) | 1.01 | 0.96–1.06 | 0.62 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




