Submitted:
27 January 2026
Posted:
28 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
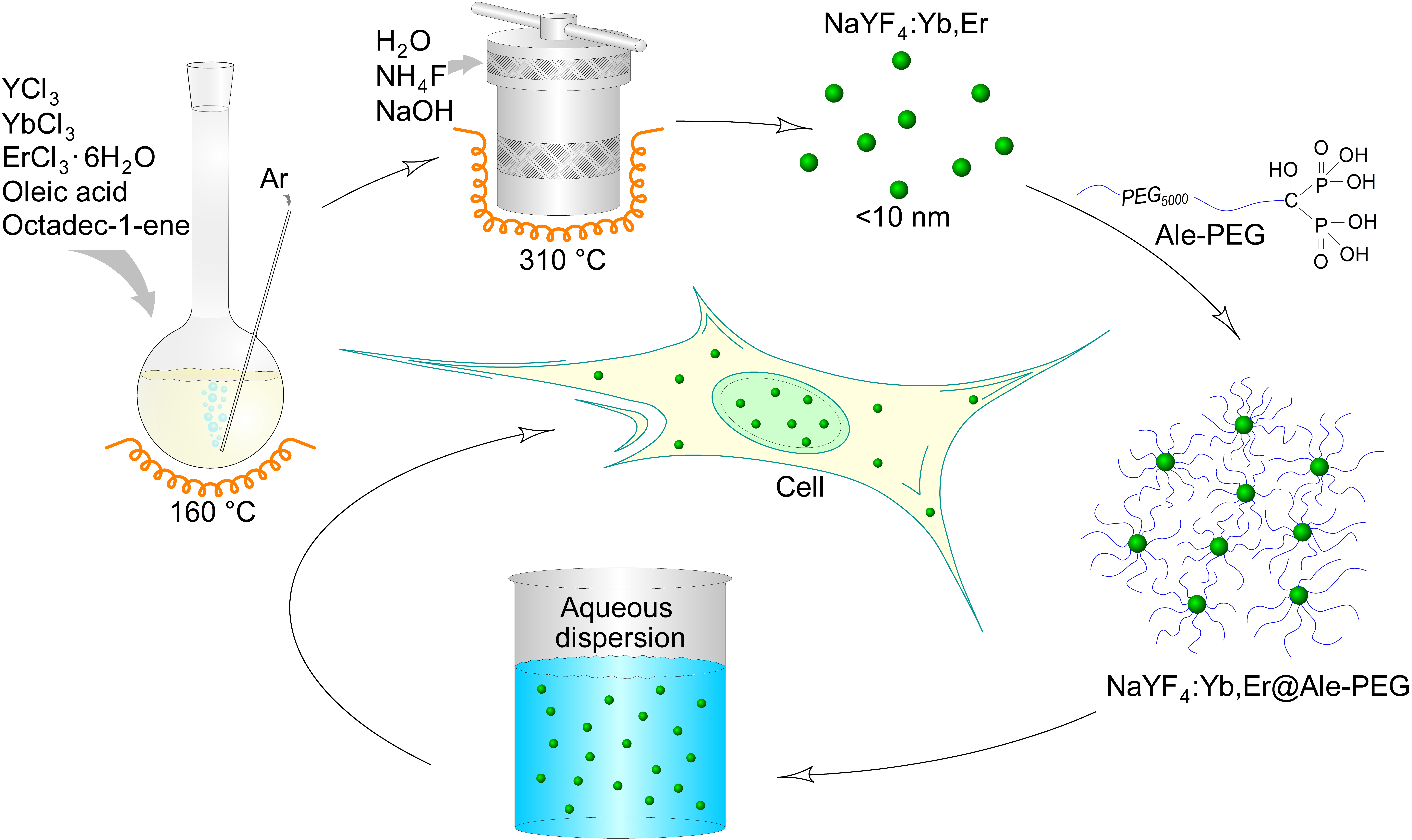
2.1. Materials
2.2. Synthesis of PEG-Alendronate (PEG-Ale)
2.3. Synthesis of UCC
2.4. PEGylation of UCC
2.5. Characterization Methods
2.6. Cells Experiments
2.6.1. Cell Culture and Labeling.
2.6.2. Microscopy.
3. Results and Discussion
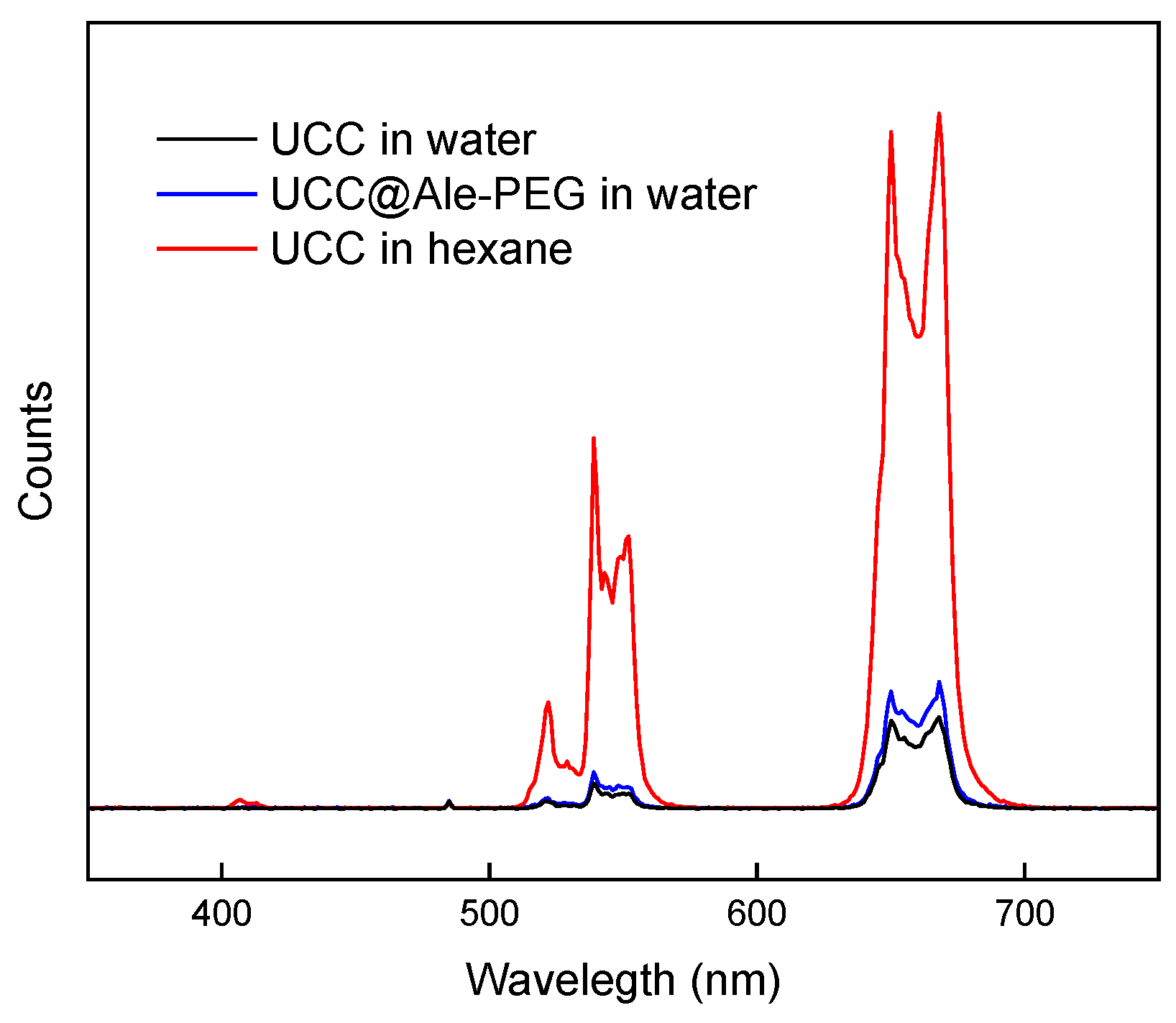
3.1. UCC
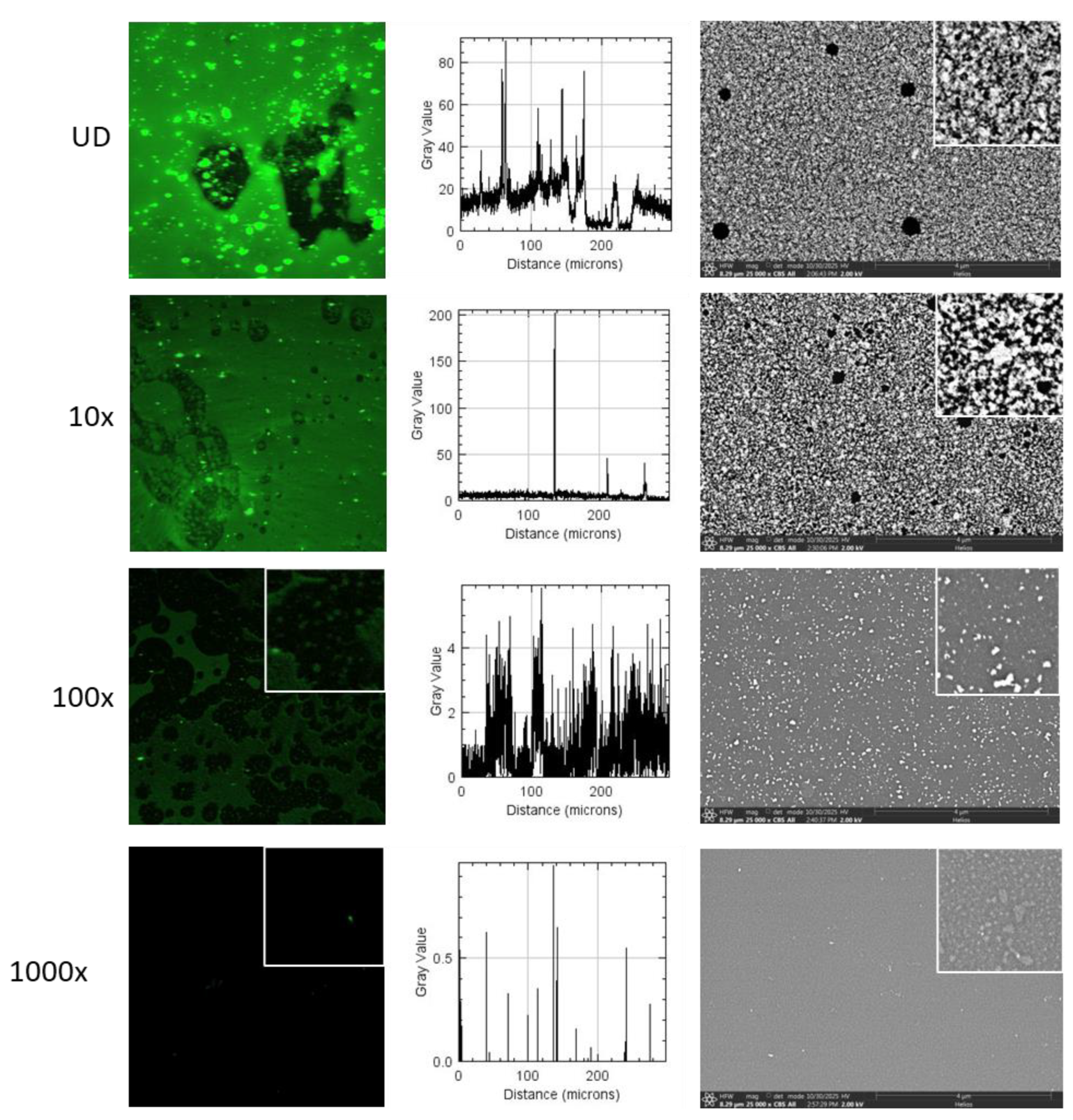
3.2. PEGylation of UCC
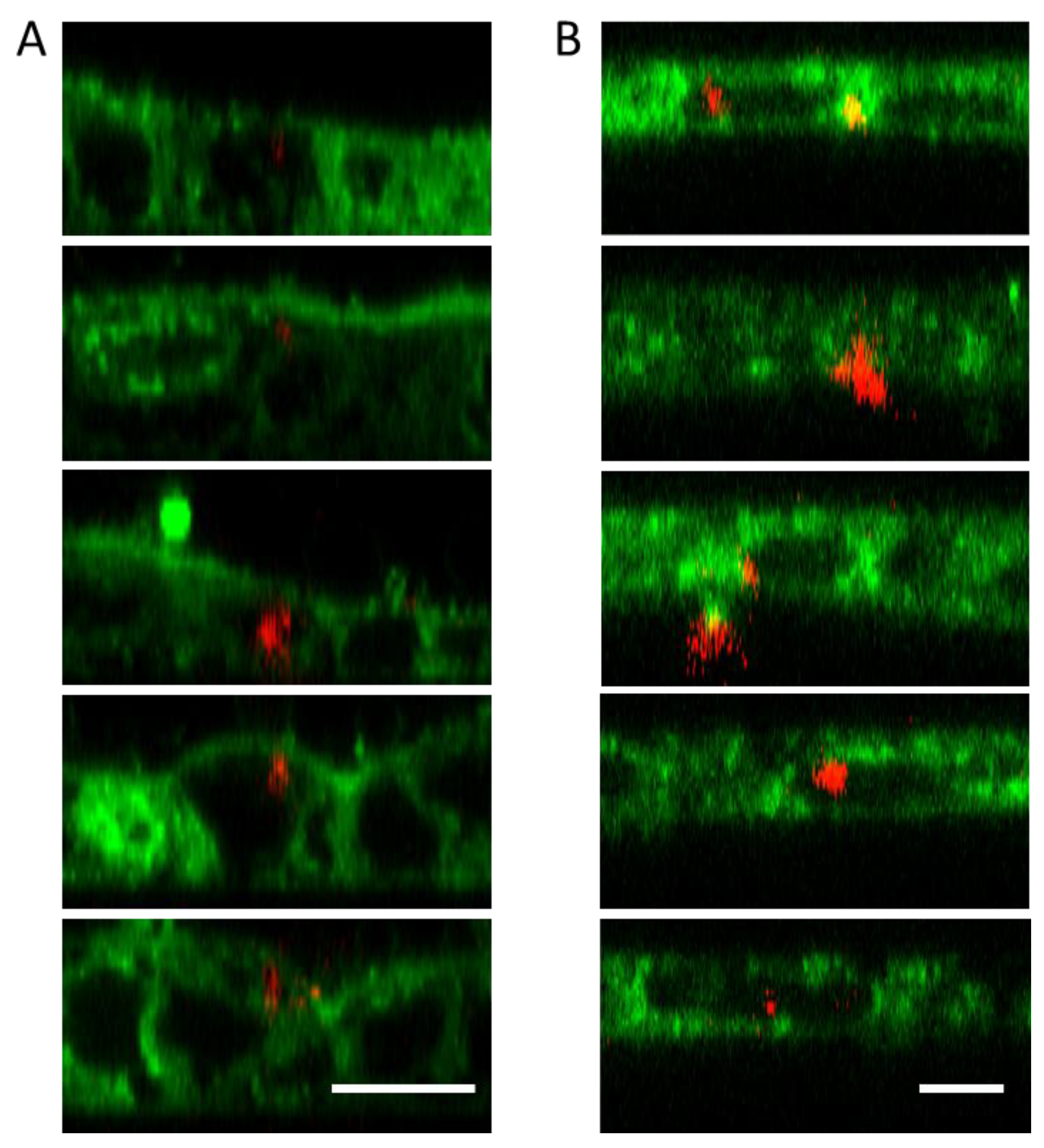
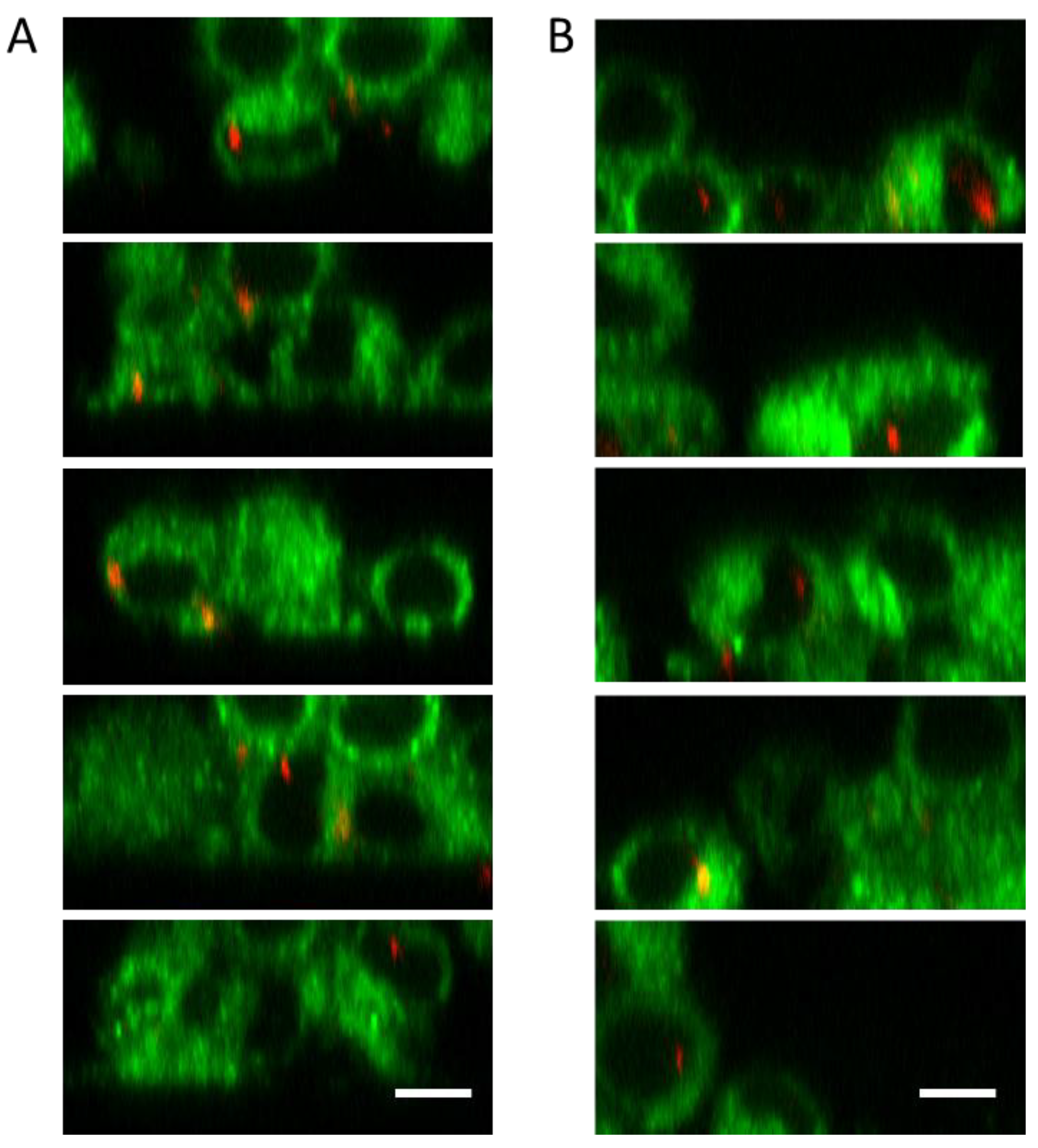
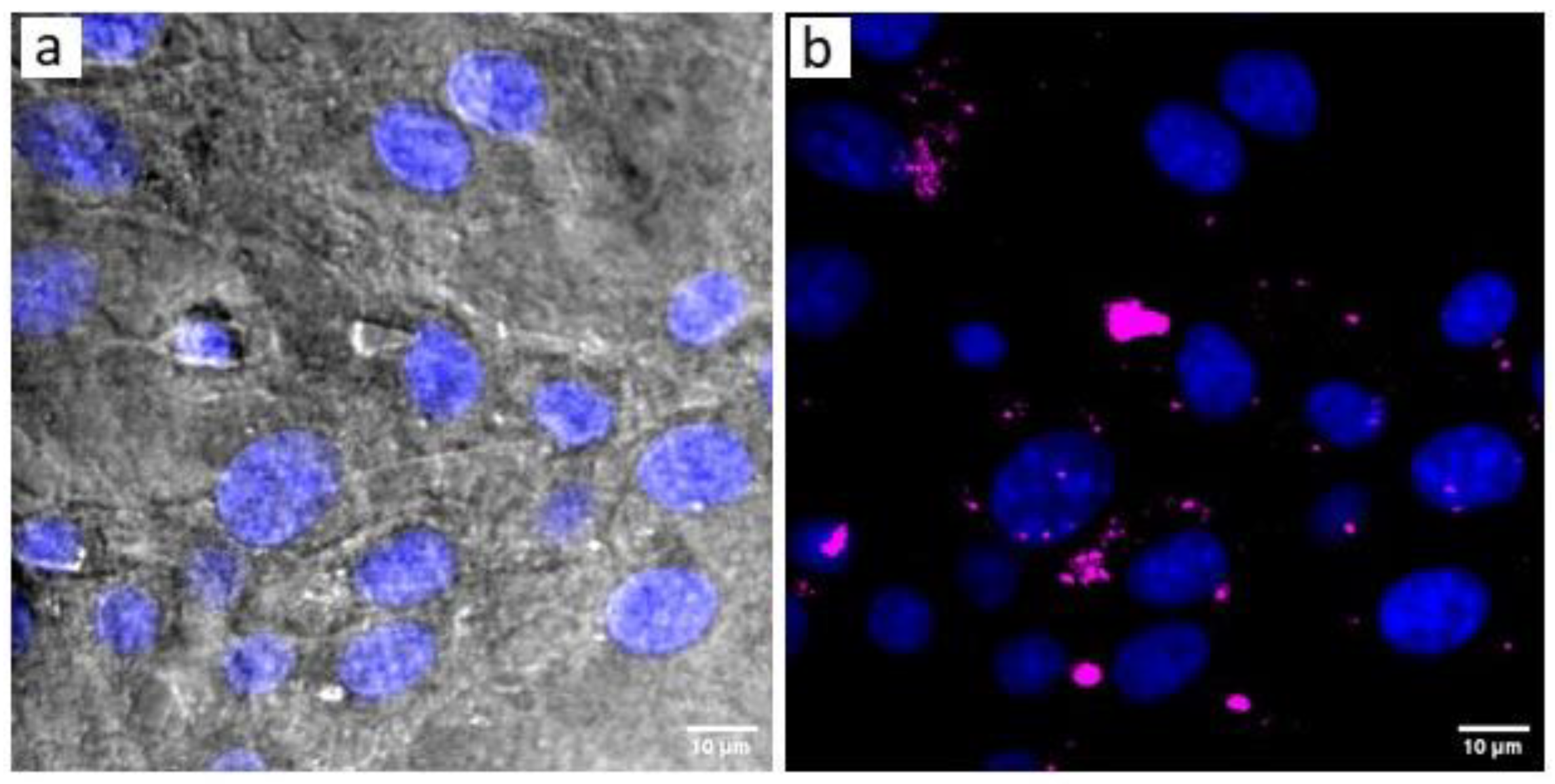
3.3. Cell Uptake Assay of the UCC@Ale-PEG
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
Abbreviations
| Ð | Dispersity |
| Dh | Hydrodynamic diameter |
| Dn | Number-average diameter |
| Dw | Weight-average diameter |
| DAPI | 4′,6-Diamidino-2-phenylindole |
| DLS | Dynamic light scattering |
| DMEM | Dulbecco’s modified Eagle’s medium |
| DNA | Deoxyribonucleic acid |
| FBS | Fetal bovine serum |
| MeO-PEG-NHS | N-Hydroxylsuccinimide-functionalized methoxy poly(ethylene glycol) |
| MWCO | Molecular weight cut-off |
| NIR | Near-infrared radiation |
| PBS | Phosphate-buffered saline |
| PD | Polydispersity |
| PEG-Ale | Poly(ethylene glycol)-alendronate |
| ROI | Region of interest |
| SEM | Scanning electron microscopy |
| TEM | Transmission electron microscopy |
| UCC | Upconversion colloid |
References
- Zhang, Y.; Du, W.; Liu, X. Photophysics and its application in photon upconversion. Nanoscale 2024, 16, 2747. [Google Scholar] [CrossRef]
- Geng, S.; Li, H.; Lv, Z.; Zhai, Y.; Tian, B.; Luo, Y.; Zhou, Y.; Han, S.-T. Challenges and opportunities of upconversion nanoparticles for emerging NIR optoelectronic devices. Adv. Mater 2025, 2419678. [Google Scholar] [CrossRef]
- Ruggiero, E.; Alonso-de-Castro, S.; Habtemariam, A.; Salassa, L. Upconverting nanoparticles for the near infrared photoactivation of transition metal complexes: New opportunities and challenges in medicinal inorganic photochemistry. Dalton Trans. 2016, 45, 13012–13020. [Google Scholar] [CrossRef]
- Chen, G.; Qiu, H.; Prasad, P.; Chen, X. Upconversion nanoparticles: Design, nanochemistry, and applications. Chem. Rev. 2014, 114, 5161–5214. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Y. Orthogonal emissive upconversion nanoparticles: Material design and applications. Small 2021, 17, 2004552. [Google Scholar] [CrossRef]
- Liu, J.; Yu, M.; Zhou, C.; Zheng, J. Renal clearable inorganic nanoparticles: A new frontier of bionanotechnology. Mater. Today 2013, 38, 676–681. [Google Scholar] [CrossRef]
- Gargas, D.; Chan, E.; Ostrowski, A.; Aloni, S.; Altoe, M.; Barnard, E.; Sanii, B.; Urban, J.; Milliron, D.; Cohen, B.; Schuck, P. Engineering bright sub-10-nm upconverting nanocrystals for single-molecule imaging. Nat. Nanotechnol. 2014, 9, 300–305. [Google Scholar] [CrossRef] [PubMed]
- Albanese, A.; Tang, P.; Chan, W. The effect of nanoparticle size, shape, and surface chemistry on biological systems. Annu. Rev. Biomed. Eng. 2012, 14, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Liu, M.; Chen, J.; Li, Z.; Yuan, Q. Rational design and biomedical applications of DNA-functionalized upconversion nanoparticles. Chin. Chem. Lett. 2018, 29, 1321–1332. [Google Scholar] [CrossRef]
- Boyer, J.C.; Vetrone, F.; Cuccia, L.A.; Capobianco, J.A. Synthesis of colloidal upconverting NaYF4 nanocrystals doped with Er3+, Yb3+ and Tm3+, Yb3+ via thermal decomposition of lanthanide trifluoroacetate precursors. J. Am. Chem. Soc. 2006, 128, 7444–7445. [Google Scholar] [CrossRef]
- Kang, N.; Ai, C.-C.; Zhou, Y.-M.; Wang, Z.; Ren, L. Nanotechnology 2018, 29, 075601. [CrossRef] [PubMed]
- Phuong, H.; Huong, T.; Vinh, L.; Thao, D.; Tu, L.; Cong, T.; Van, N.; Tien, T. SCN–IgG functionalized NaYF4:Yb3+/(Er3+,Tm3+) upconversion nanoparticles for targeted fluorescence imaging of liver cancer cells. RSC Adv. 2025, 15, 22682–2268. [Google Scholar] [CrossRef] [PubMed]
- Salomão, P.M.A.; Oliveira, F.A.; Rodrigues, P.D.; Al-Ahj, L.P.; Gasque, K.C.D.S.; Jeggle, P.; Buzalaf, M.A.R.; Oliveira, R.C.; Edwardson, J.M.; Magalhães, A.C. The cytotoxic effect of TiF4 and NaF on fibroblasts is influenced by the experimental model, fluoride concentration and exposure time. PLOS ONE 2017, 12, e0179471. [Google Scholar] [CrossRef]
- Chang, Y.C.; Chou, M.Y. Cytotoxicity of fluoride on human pulp cell cultures in vitro. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2001, 91, 230–234. [Google Scholar] [CrossRef]
- Ansari, A.A.; Parchur, A.K.; Chen, G. Surface modified lanthanide upconversion nanoparticles for drug delivery, cellular uptake mechanism, and current challenges in NIR-driven therapies. Coord. Chem. Rev. 2022, 457, 214423. [Google Scholar] [CrossRef]
- Chang, H.; Xie, J.; Zhao, B.; Liu, B.; Xu, S.; Ren, N.; Xie, X.; Huang, L.; Huang, W. Rare earth ion-doped upconversion nanocrystals: Synthesis and surface modification. Nanomaterials (Basel) 2014, 25, 1–25. [Google Scholar] [CrossRef]
- Malhotra, K.; Kumar, B.; Piunno, P.A.E.; Krull, U.J. Cellular uptake of upconversion nanoparticles based on surface polymer coatings and protein corona. ACS Appl. Mater. Interfaces 2024, 16, 35985–36001. [Google Scholar] [CrossRef]
- Muhr, V.; Wilhelm, S.; Hirsch, T.; Wolfbeis, O.S. Upconversion nanoparticles: From hydrophobic to hydrophilic surfaces. Acc. Chem. Res. 2014, 47, 3481–3493. [Google Scholar] [CrossRef] [PubMed]
- Vasylyshyn, T.; Patsula, V.; Větvička, D.; Shapoval, O.; Pankrác, J.; Kabešová, M.; Beneš, J.; Horák, D. Intraperitoneal versus intravenous administration of Flamma®-conjugated PEG-alendronate-coated upconversion nanoparticles in mice pancreatic cancer model. Nanoscale Adv. 2025, 7, 144–154. [Google Scholar] [CrossRef]
- Zhou, B.; Shi, B.; Jin, D.; Liu, X. Controlling upconversion nanocrystals for emerging applications. Nat. Nanotechnol. 2015, 10, 924–936. [Google Scholar] [CrossRef]
- Kostiv, U.; Lobaz, V.; Kučka, J.; Švec, P.; Sedláček, O.; Hrubý, M.; Janoušková, O.; Francová, P.; Kolářová, V.; Šefc, L.; Horák, D. A simple neridronate-based surface coating strategy for upconversion nanoparticles: Highly colloidally stable 125I-radiolabeled NaYF4:Yb3+/Er3+@PEG nanoparticles for multimodal in vivo tissue imaging. Nanoscale 2017, 9, 16680–16688. [Google Scholar] [CrossRef]
- Nahorniak, M.; Horák, D.; Šlouf, M.; Steinhart, M.; Shapoval, O.; Engstová, H.; Ježek, P. Lanthanide-based UCNPs: Toxicity evaluation and interaction of ultrasmall core vs. core–shell nanoparticles with cells. Mater. Adv. 2025, 6, 6907–6918. [Google Scholar] [CrossRef]
- Nahorniak, M.; Oleksa, V.; Vasylyshyn, T.; Pop-Georgievski, O.; Rydvalová, E.; Filipová, M.; Horák, D. Cytotoxicity evaluation of photosensitizer-conjugated hexagonal upconverting nanoparticles. Nanomaterials 2023, 13, 1535. [Google Scholar] [CrossRef] [PubMed]
- Giakoumettis, D.; Kritis, A.; Foroglou, N. C6 cell line: The gold standard in glioma research. Hippokratia 2018, 22, 105–112. [Google Scholar] [PubMed]
- Wang, M.; Abbineni, G.; Clevenger, A.; Mao, C.; Xu, S. Upconversion nanoparticles: Synthesis, surface modification and biological applications. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 710–729. [Google Scholar] [CrossRef] [PubMed]

| Colloid |
Dn (nm) |
Ð | Dh (nm) | PD | ζ-potential, (mV) |
| UCC | 9 | 1.09 | 105 (18*) | 0.19 (0.17*) | 25 |
| UCC@Ale-PEG | 11 | 1.13 | 90 | 0.14 | 14 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




