Submitted:
26 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
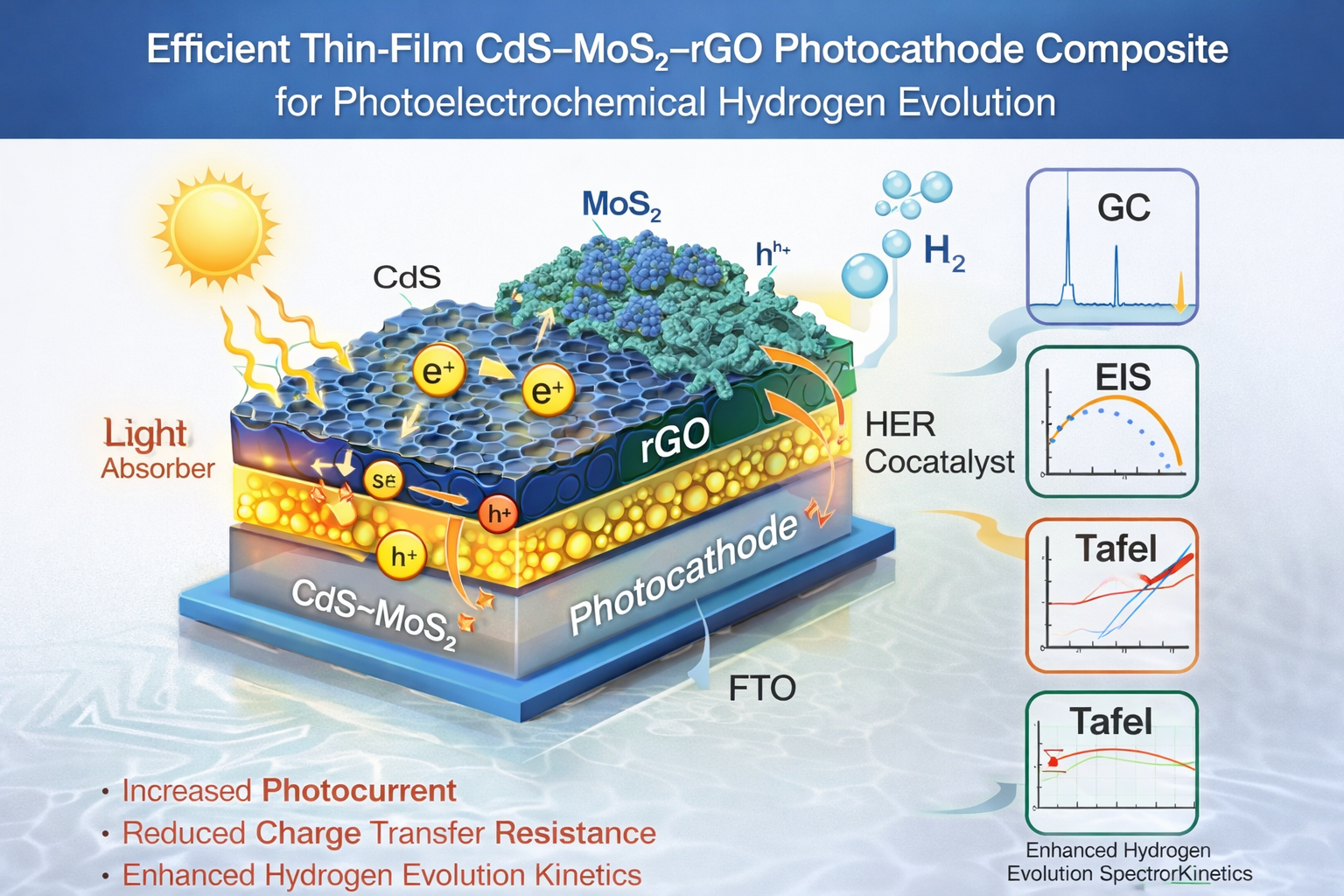
Keywords:
1. Introduction
2. Materials and Methods
2.1. Preparation of Graphene Oxide (GO)
2.2. Preparation of FTO Substrates
2.3. Preparation of CdS–MoS₂–rGO Composite Ink
2.4. Film Deposition and Thermal Treatment
2.5. Characterization Techniques
2.6. Photoelectrochemical Measurements
2.4. CdS-MoS2-rGO Film and Other Control Films Deposition (Three Thin Coats) and Thermal Conversion
2.5. Measurement Techniques; X-Ray Powered Diffraction (XRD)
2.6. Photoelectrochemical Measurement
3. Results and Discussion
3.1. Linear Sweep Voltammetry (LSV) and Photocurrent Density
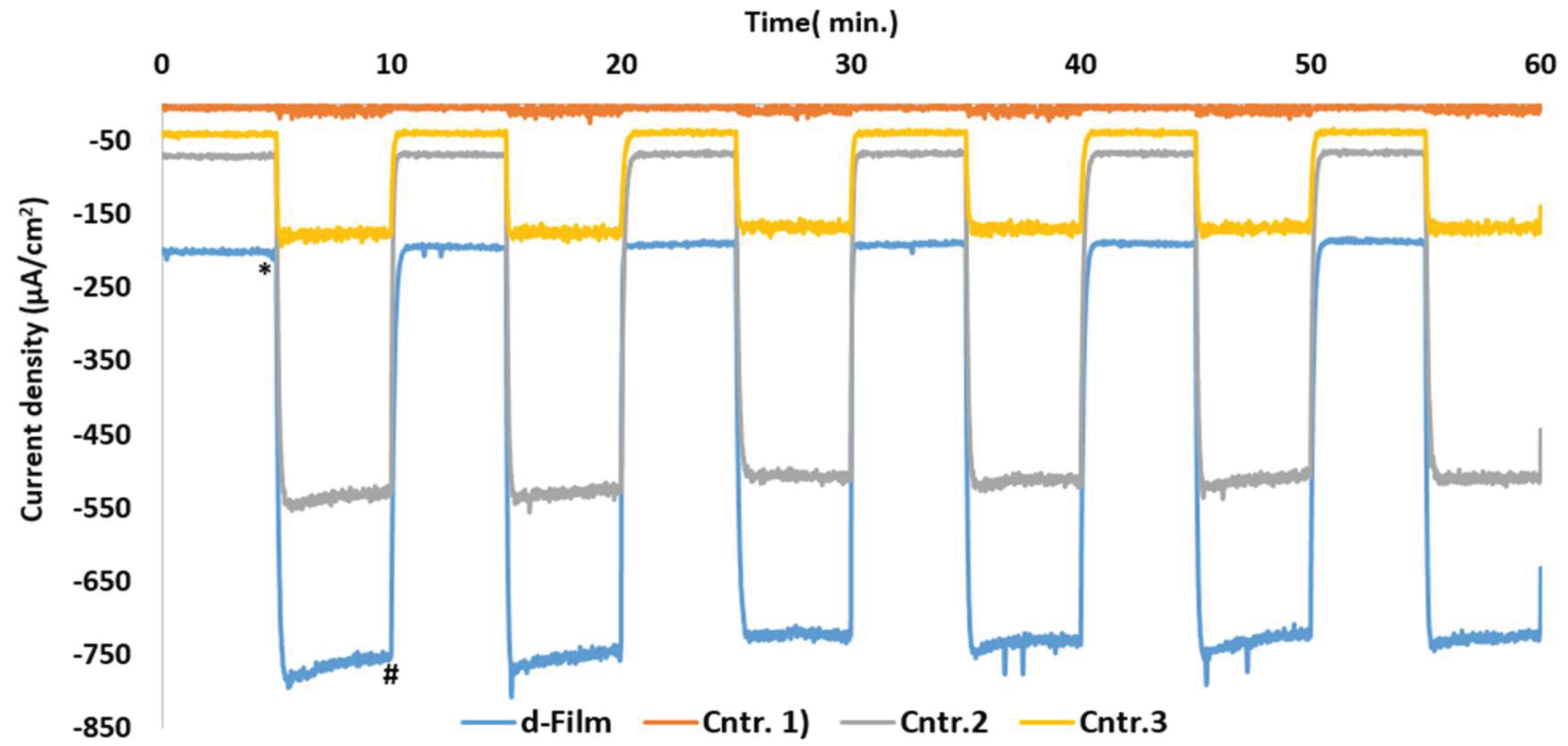
3.2. Chronoamperometric Investigations
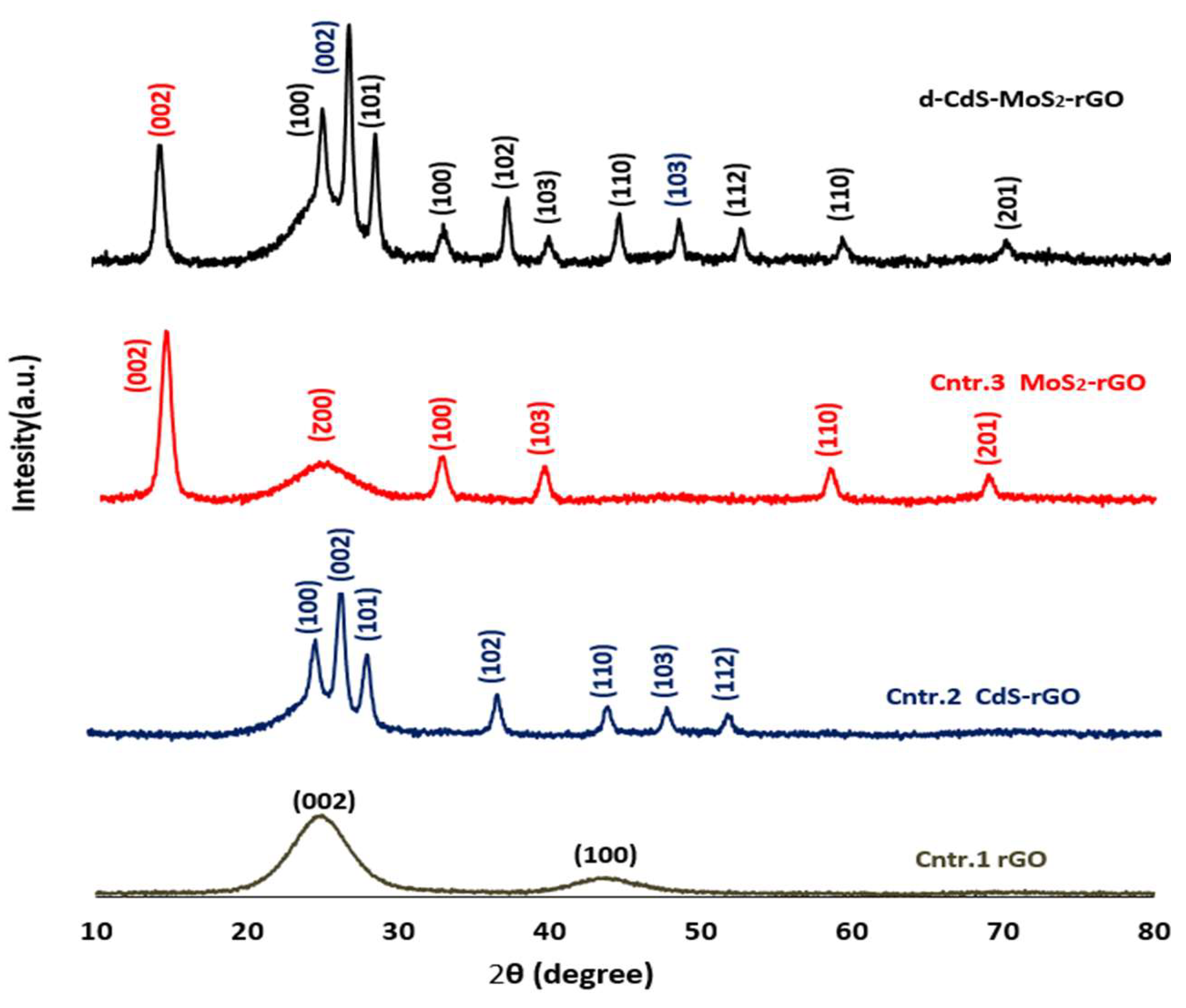
3.3. UV-Visible and X-Ray Diffraction (XRD) Studies
3.4. SEM Measurement and Surface Morphology
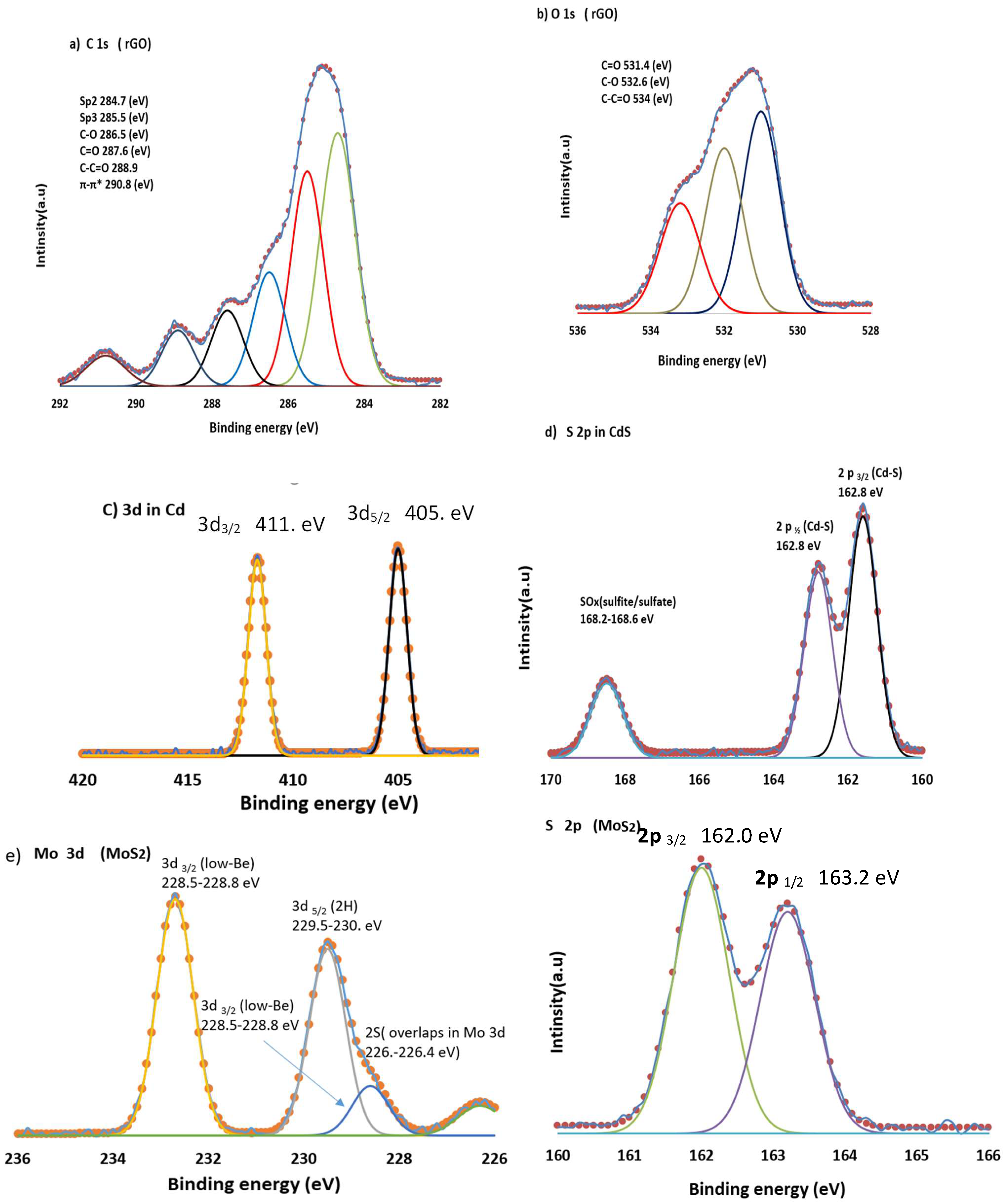
3.5. X-Ray Photoelectron Spectroscopy
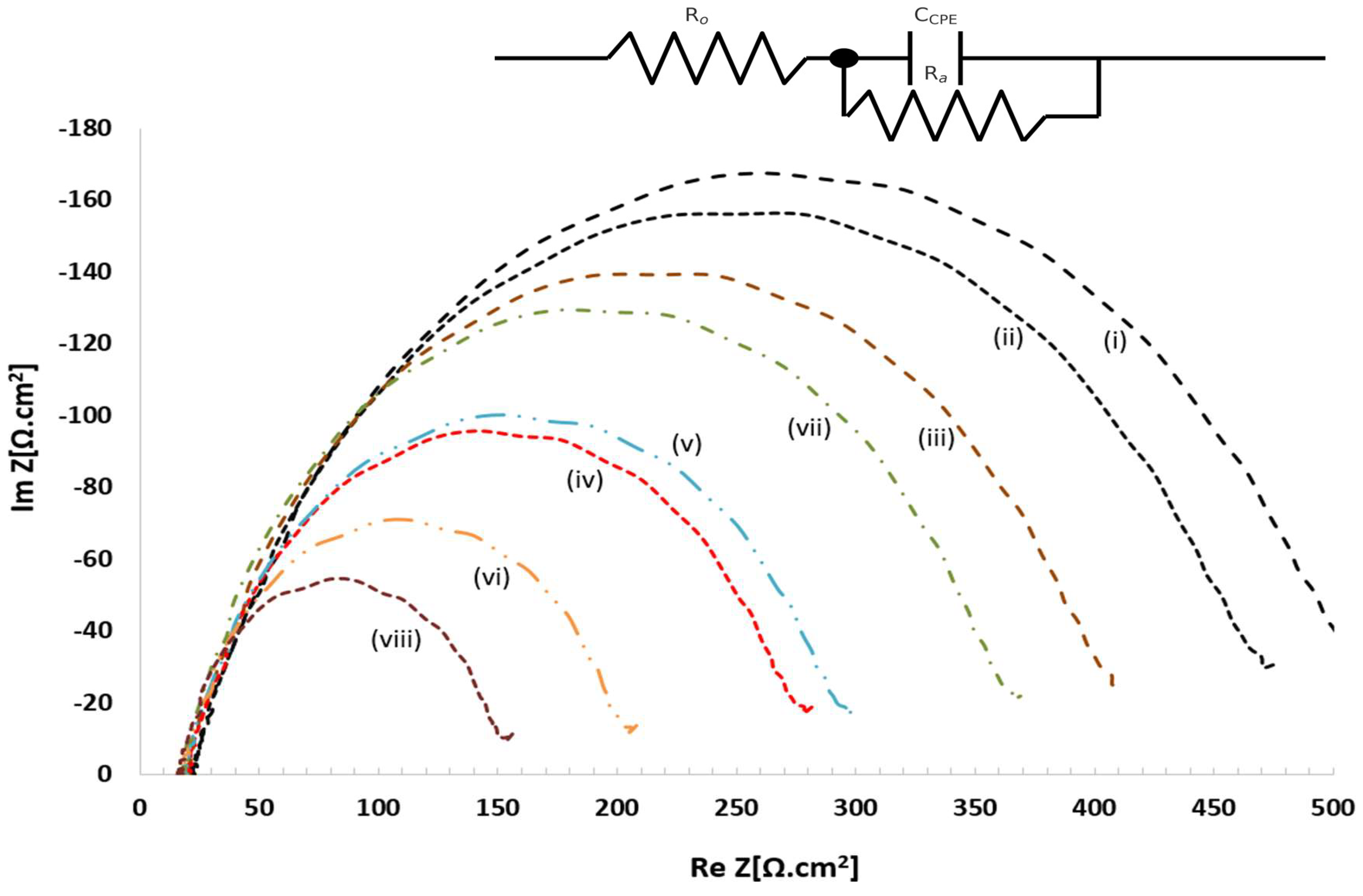
3.6. Electrochemical Impedance Spectroscopy (EIS) & Tafel Plots Studies
| Sample | Rct (Ω·cm²) | i₀ (mA·cm⁻²) | Tafel slope (mV·dec⁻¹) |
| (i) rGO — dark | 445 | 0.028 | 280 |
| (ii) rGO — light | 355 | 0.036 | 255 |
| (iii) MoS₂–rGO — dark | 332 | 0.041 | 211 |
| (iv) MoS₂–rGO — light | 217 | 0.058 | 176 |
| (v) CdS–rGO — dark | 242 | 0.053 | 146 |
| (vi) CdS–rGO — light | 157 | 0.083 | 126 |
| (vii) CdS–MoS₂–rGO — dark | 256 | 0.049 | 111 |
| (viii) CdS–MoS₂–rGO — light | 118 | 0.055 | 86 |
3.6. Gas Chromatography (GC) and Hydrogen Evolution Rates
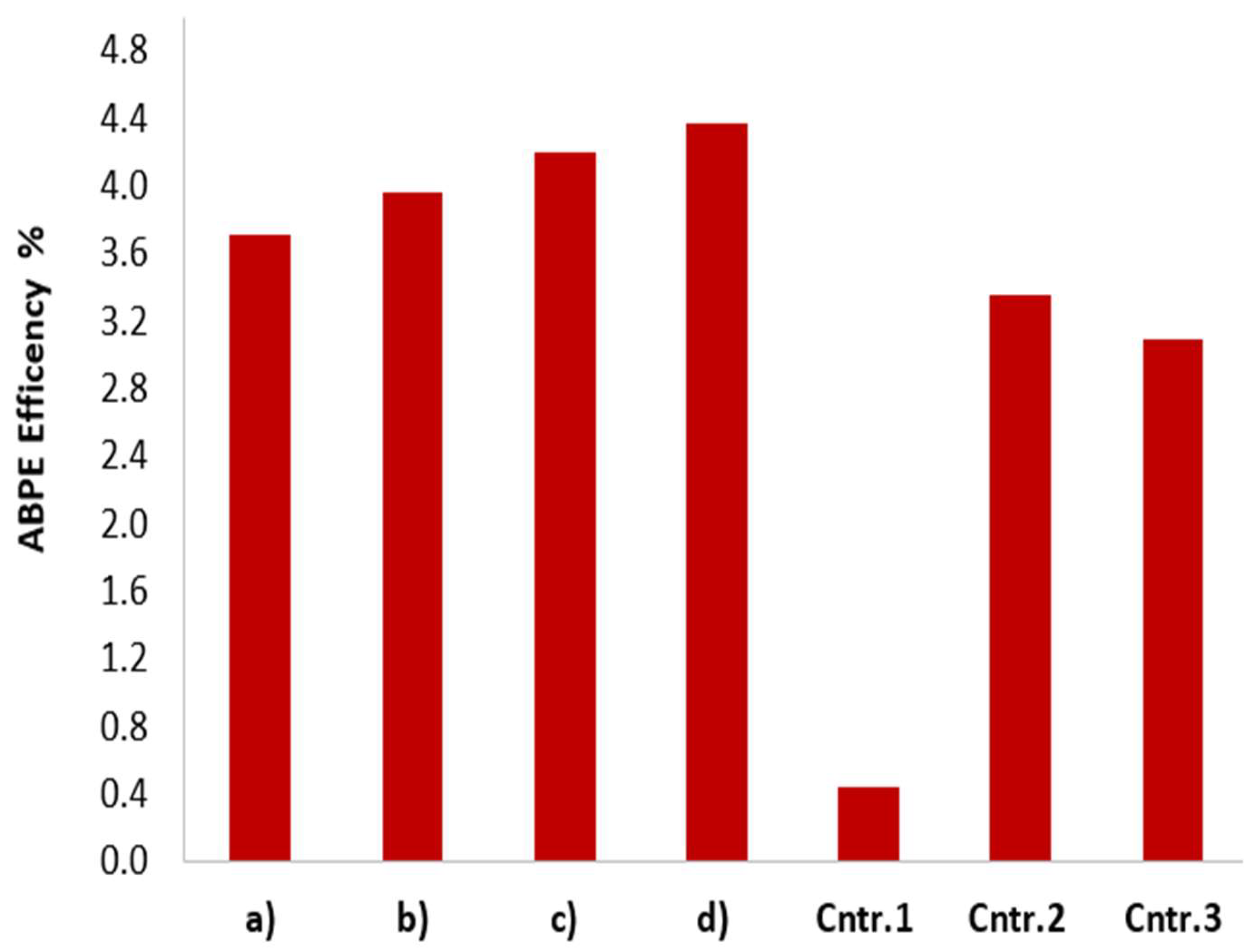
3.7. Applied-Bias Photon-to-Current Efficiency (ABPE)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xu, Y.; Schoonen, M.A. The absolute energy positions of conduction and valence bands of selected semiconducting minerals. Am. Miner. 2000, 85, 543–556. [CrossRef]
- Nasir, J.A.; Rehman, Z.-U.; Shah, S.N.A.; Khan, A.; Butler, I.S.; Catlow, C.R.A. Recent developments and perspectives in CdS-based photocatalysts for water splitting. J. Mater. Chem. A 2020, 8, 20752–20780. [CrossRef]
- Hinnemann, B.; Moses, P.G.; Bonde, J.; Jørgensen, K.P.; Nielsen, J.H.; Horch, S.; Chorkendorff, I.; Nørskov, J.K. Biomimetic Hydrogen Evolution: MoS2Nanoparticles as Catalyst for Hydrogen Evolution. J. Am. Chem. Soc. 2005, 127, 5308–5309. [CrossRef]
- Voiry, D.; Salehi, M.; Silva, R.; Fujita, T.; Chen, M.; Asefa, T.; Shenoy, V.B.; Eda, G.; Chhowalla, M. Conducting MoS2 Nanosheets as Catalysts for Hydrogen Evolution Reaction. Nano Lett. 2013, 13, 6222–6227. [CrossRef]
- Lu, K.-Q.; Li, Y.-H.; Tang, Z.-R.; Xu, Y.-J. Roles of Graphene Oxide in Heterogeneous Photocatalysis. ACS Mater. Au 2021, 1, 37–54. [CrossRef]
- Hummers, W.S., Jr.; Offeman, R.E. Preparation of Graphitic Oxide. J. Am. Chem. Soc. 1958, 80, 1339. [CrossRef]
- Marcano, D.C., et al., Improved Synthesis of Graphene Oxide. ACS Nano, 2010. 4(8): p. 4806-4814.
- Ortega-Borges, R.; Lincot, D. Mechanism of Chemical Bath Deposition of Cadmium Sulfide Thin Films in the Ammonia-Thiourea System: In Situ Kinetic Study and Modelization. J. Electrochem. Soc. 1993, 140, 3464–3473. [CrossRef]
- Najm, A.S.; Naeem, H.S.; Alwarid, D.A.R.M.; Aljuhani, A.; Hasbullah, S.A.; Hasan, H.A.; Sopian, K.; Bais, B.; Al-Iessa, H.J.; Majdi, H.S.; et al. Mechanism of Chemical Bath Deposition of CdS Thin Films: Influence of Sulphur Precursor Concentration on Microstructural and Optoelectronic Characterizations. Coatings 2022, 12, 1400. [CrossRef]
- Bao, N.; Shen, L.; Takata, T.; Domen, K.; Gupta, A.; Yanagisawa, A.K.; Grimes‖, C.A. Facile Cd−Thiourea Complex Thermolysis Synthesis of Phase-Controlled CdS Nanocrystals for Photocatalytic Hydrogen Production under Visible Light. J. Phys. Chem. C 2007, 111, 17527–17534. [CrossRef]
- Yin, Y.; Han, J.; Zhang, Y.; Zhang, X.; Xu, P.; Yuan, Q.; Samad, L.; Wang, X.; Wang, Y.; Zhang, Z.; et al. Contributions of Phase, Sulfur Vacancies, and Edges to the Hydrogen Evolution Reaction Catalytic Activity of Porous Molybdenum Disulfide Nanosheets. J. Am. Chem. Soc. 2016, 138, 7965–7972. [CrossRef]
- Mondal, A.; Prabhakaran, A.; Gupta, S.; Subramanian, V.R. Boosting Photocatalytic Activity Using Reduced Graphene Oxide (RGO)/Semiconductor Nanocomposites: Issues and Future Scope. ACS Omega 2021, 6, 8734–8743. [CrossRef]
- Ren, H.; Zou, X.; Cheng, J.; Ling, T.; Bai, X.; Chen, D. Facile Solution Spin-Coating SnO2 Thin Film Covering Cracks of TiO2 Hole Blocking Layer for Perovskite Solar Cells. Coatings 2018, 8, 314. [CrossRef]
- Weidling, A.M.; Turkani, V.S.; Akhavan, V.; Schroder, K.A.; Swisher, S.L. Large-area photonic lift-off process for flexible thin-film transistors. npj Flex. Electron. 2022, 6, 1–10. [CrossRef]
- Kolhe, S.; Kuljarni, S.; Nigavekar, A.; Sharma, S. Effects of air annealing on chemincally deposited CdS films exmained by XPS and XRD. Sol. Energy Mater. 1984, 10, 47–54. [CrossRef]
- Allen J. Bard and Larry R. Faulkner, Electrochemical Methods: Fundamentals and Applications, New York: Wiley, 2001, 2nd ed. Russian Journal of Electrochemistry, 2002. 38(12): p. 1364-1365.
- Vivier, V.; Orazem, M.E. Impedance Analysis of Electrochemical Systems. Chem. Rev. 2022, 122, 11131–11168. [CrossRef]
- Yan, Y.; Xia, B.; Xu, Z.; Wang, X. Recent Development of Molybdenum Sulfides as Advanced Electrocatalysts for Hydrogen Evolution Reaction. ACS Catal. 2014, 4, 1693–1705. [CrossRef]
- Kamat, P.V. Graphene-Based Nanoarchitectures. Anchoring Semiconductor and Metal Nanoparticles on a Two-Dimensional Carbon Support. J. Phys. Chem. Lett. 2009, 1, 520–527. [CrossRef]
- Xiang, Q.; Cheng, B.; Yu, J. Graphene-Based Photocatalysts for Solar-Fuel Generation. Angew. Chem. Int. Ed. Engl. 2015, 54, 11350–11366. [CrossRef]
- Zong, X.; Wu, G.; Yan, H.; Ma, G.; Shi, J.; Wen, F.; Wang, L.; Li, C. Photocatalytic H2 Evolution on MoS2/CdS Catalysts under Visible Light Irradiation. J. Phys. Chem. C 2010, 114, 1963–1968. [CrossRef]
- Seo, D.-B.; Trung, T.N.; Bae, S.-S.; Kim, E.-T. Improved Photoelectrochemical Performance of MoS2 through Morphology-Controlled Chemical Vapor Deposition Growth on Graphene. Nanomaterials 2021, 11, 1585. [CrossRef]
- Krishna Kumar, A.S.; Jiang, S.-J.; Warchoł, J.K. Synthesis and Characterization of Two-Dimensional Transition Metal Dichalcogenide Magnetic MoS2@Fe3O4 Nanoparticles for Adsorption of Cr(VI)/Cr(III). ACS Omega 2017, 2, 6187–6200. [CrossRef]
- Sumair Ahmed, S., et al., Improved Performance of CuFe2O4/rGO Nanohybrid as an Anode Material for Lithium-ion Batteries Prepared Via Facile One-step Method. Current Nanoscience, 2019. 15(4): p. 420-429.
- Maticiuc, N.; Hiie, J.; Mikli, V.; Potlog, T.; Valdna, V. Structural and optical properties of cadmium sulfide thin films modified by hydrogen annealing. Mater. Sci. Semicond. Process. 2014, 26, 169–174. [CrossRef]
- Kar, S.; Ghosh, S.; Pal, T. MoS2-CdS composite for photocatalytic reduction of hexavalent chromium and thin film optoelectronic device applications. Sci. Rep. 2024, 14, 1–12. [CrossRef]
- Jones, L.A.H.; Xing, Z.; Swallow, J.E.N.; Shiel, H.; Featherstone, T.J.; Smiles, M.J.; Fleck, N.; Thakur, P.K.; Lee, T.-L.; Hardwick, L.J.; et al. Band Alignments, Electronic Structure, and Core-Level Spectra of Bulk Molybdenum Dichalcogenides (MoS2, MoSe2, and MoTe2). J. Phys. Chem. C 2022, 126, 21022–21033. [CrossRef]
- Hamid, K.; Bin Mukhlish, M.Z.; Uddin, T. Sunlight-activated heterostructure MoS2/CdS nanocomposite photocatalyst with enhanced photocatalytic activity: band alignment and mechanism study. RSC Adv. 2024, 14, 38908–38923. [CrossRef]
- Johra, F.T.; Jung, W.-G. Hydrothermally reduced graphene oxide as a supercapacitor. Appl. Surf. Sci. 2015, 357, 1911–1914. [CrossRef]
- Ziabari, A.A.; Ghodsi, F. Growth, characterization and studying of sol–gel derived CdS nanoscrystalline thin films incorporated in polyethyleneglycol: Effects of post-heat treatment. Sol. Energy Mater. Sol. Cells 2012, 105, 249–262. [CrossRef]
- Yan, Z.; Du, L.; Phillips, D.L. Multilayer core–shell MoS2/CdS nanorods with very high photocatalytic activity for hydrogen production under visible-light excitation and investigation of the photocatalytic mechanism by femtosecond transient absorption spectroscopy. RSC Adv. 2017, 7, 55993–55999. [CrossRef]
- Yao, Y.; Ao, K.; Lv, P.; Wei, Q. MoS2 Coexisting in 1T and 2H Phases Synthesized by Common Hydrothermal Method for Hydrogen Evolution Reaction. Nanomaterials 2019, 9, 844. [CrossRef]
- Macdonald, J.R., Impedance spectroscopy: emphasizing solid materials and systems. Applied Optics, 1989. 28(6): p. 1083.
- Macdonald, J.R., et al., Impedance spectroscopy: theory, experiment, and applications. 2018: John Wiley & Sons.
- Lasia, A., Electrochemical impedance spectroscopy and its applications, in Modern aspects of electrochemistry. 2002, Springer. p. 143-248.
- Kékedy-Nagy, L., et al., An Electrochemical Study of Ammonium Dihydrogen Phosphate on Mg and Mg Alloy Electrodes. Electrocatalysis, 2021. 12(3): p. 251-263.
- Wan, X.; Lu, D.; Wang, X.; Liu, G.; Fu, Y.; Hu, C.; Rong, N.; Wang, H.; Cheng, Z. Enhanced Photoelectrochemical Water Oxidation on BiVO4 Photoanodes Functionalized by Bimetallic Dicyanamide Molecular Catalysts. Sustainability 2023, 15, 3129. [CrossRef]

| Sample | CdS (wt%) | MoS₂ (wt%) | rGO (wt%) | m(CdS) (mg) | m(MoS₂) (mg) | m(rGO) (mg) | Cd(NO₃)₂ (mL) | Na₂MoO₄ (mL) | GO (4mg mL⁻¹) (µL) | water (mL) | EtOH (mL) | Thiourea† (mg) |
| a (93:5:2) | 93.0 | 5.0 | 2.0 | 11.16 | 0.60 | 0.24 | 0.772 | 0.037 | 60 | 0.690 | 8.435 | 61.6 |
| b (96:3:1) | 96.0 | 3.0 | 1.0 | 11.52 | 0.36 | 0.12 | 0.797 | 0.022 | 30 | 0.680 | 8.465 | 62.4 |
| c (97:2:1) | 97.0 | 2.0 | 1.0 | 11.64 | 0.24 | 0.12 | 0.806 | 0.015 | 30 | 0.679 | 8.465 | 62.5 |
| d (86:9:5) | 86.0 | 9.0 | 5.0 | 10.32 | 1.08 | 0.60 | 0.714 | 0.067 | 150 | 0.718 | 8.345 | 61.5 |
| Ctrl 1 (0/0/100) | 0.0 | 0.0 | 100 | 0.0 | 0.0 | 12.0 | 0.0 | 0.0 | 3000 | 1.500 | 5.495 | 0.0 |
| Ctrl 2 (95/0/5) | 95.0 | 0.0 | 5.0 | 11.40 | 0.0 | 0.60 | 0.789 | 0.0 | 150 | 0.711 | 8.345 | 60.1 |
| Ctrl 3 (0/95/5) | 0.0 | 95.0 | 5.0 | 0.0 | 11.40 | 0.60 | 0.0 | 0.712 | 150 | 0.788 | 8.345 | 54.2 |
| Samples & controls film | Dark current density (µA·cm⁻²) | Light current density (µA·cm⁻²) | Dark onset potential (V) | Light onset potential (V) |
| a) | 193 | 709 | −0.68 | −0.54 |
| b) | 196 | 757 | −0.66 | −0.52 |
| c) | 201 | 803 | −0.65 | −0.52 |
| d) | 209 | 836 | −0.64 | −0.49 |
| Cntr.1 (rGO) | 30 | 86 | −0.82 | −0.73 |
| Cntr.2 (CdS–rGO) | 160 | 643 | −0.72 | −0.55 |
| Cntr.3 (MoS₂–rGO) | 150 | 592 | −0.75 | −0.61 |
| Label | Film type | R₀ (Ω·cm²) | Rct (Ω·cm²) | QCPE (µΩ⁻¹·cm⁻²·sⁿ ×10⁻⁴) | nCPE | Cdl (µF·cm⁻²) | kct (s⁻¹) |
| (i) | rGO — dark | 40 | 450 | 1.5 | 0.82 | 6 | 370 |
| (ii) | rGO — light | 39 | 360 | 2.0 | 0.85 | 7 | 397 |
| (iii) | MoS₂–rGO — dark | 38 | 310 | 2.2 | 0.86 | 7.5 | 430 |
| (iv) | MoS₂–rGO — light | 38 | 220 | 2.8 | 0.88 | 9 | 505 |
| (v) | CdS–rGO — dark | 38 | 240 | 2.5 | 0.88 | 8.5 | 490 |
| (vi) | CdS–rGO — light | 37 | 155 | 3.3 | 0.90 | 10 | 645 |
| (vii) | CdS–MoS₂–rGO — dark | 37 | 260 | 3.0 | 0.90 | 9.5 | 405 |
| (viii) | CdS–MoS₂–rGO — light | 36 | 120 | 3.8 | 0.92 | 11 | 758 |
| Label | Film type | R₀ (Ω·cm²) | Rct (Ω·cm²) | QCPE (µΩ⁻¹·cm⁻²·sⁿ ×10⁻⁴) | nCPE | Cdl (µF·cm⁻²) | kct (s⁻¹) |
| (i) | rGO — dark | 40 | 450 | 1.5 | 0.82 | 6 | 370 |
| (ii) | rGO — light | 39 | 360 | 2.0 | 0.85 | 7 | 397 |
| (iii) | MoS₂–rGO — dark | 38 | 310 | 2.2 | 0.86 | 7.5 | 430 |
| (iv) | MoS₂–rGO — light | 38 | 220 | 2.8 | 0.88 | 9 | 505 |
| (v) | CdS–rGO — dark | 38 | 240 | 2.5 | 0.88 | 8.5 | 490 |
| (vi) | CdS–rGO — light | 37 | 155 | 3.3 | 0.90 | 10 | 645 |
| (vii) | CdS–MoS₂–rGO — dark | 37 | 260 | 3.0 | 0.90 | 9.5 | 405 |
| (viii) | CdS–MoS₂–rGO — light | 36 | 120 | 3.8 | 0.92 | 11 | 758 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




