Submitted:
19 January 2026
Posted:
27 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Transpapillary Biliary Drainage
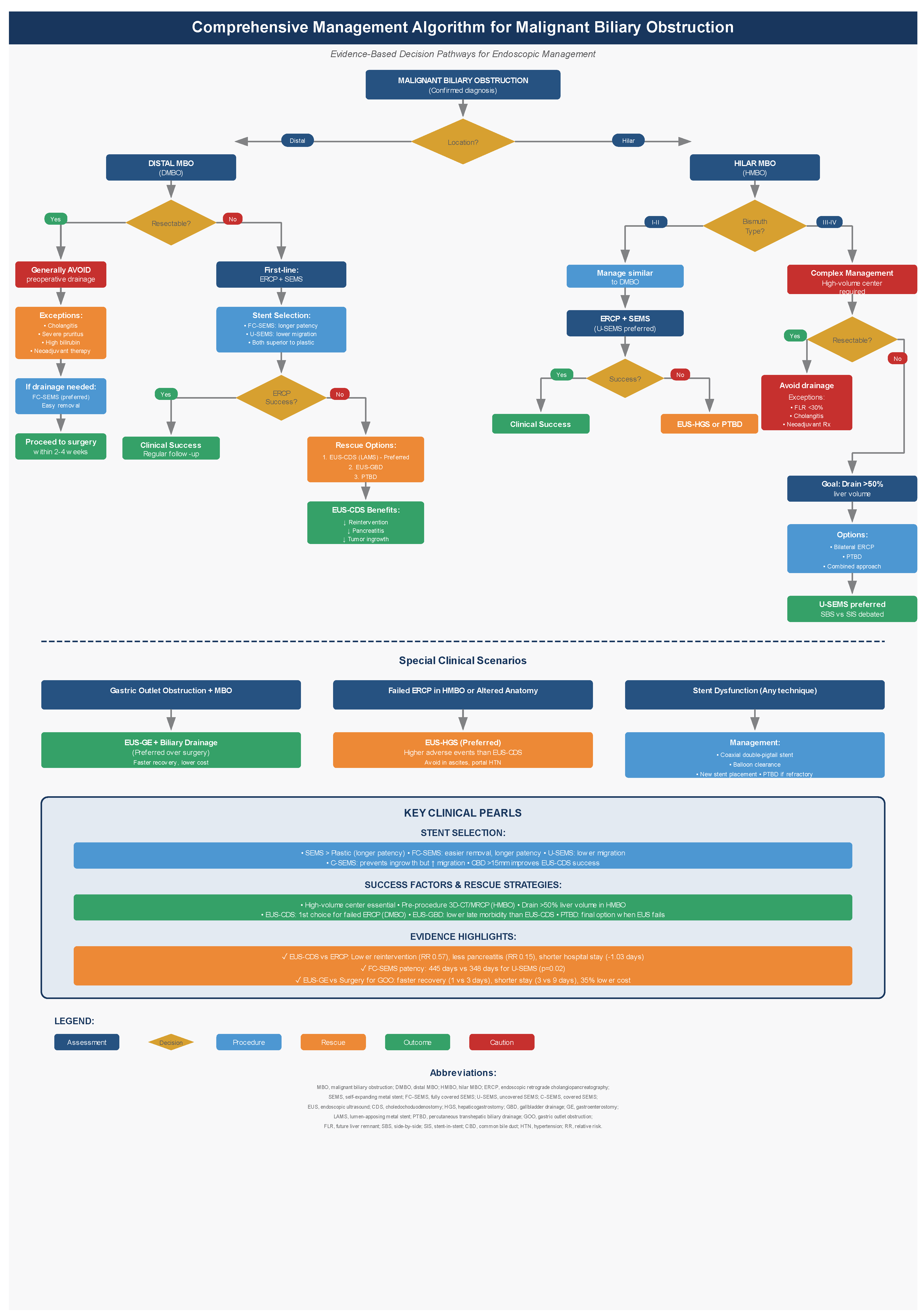
2.1. TBD in Unresectable Distal Malignant Biliary Obstruction
2.2. TBD in Resectable Distal Malignant Biliary Obstruction
2.3. TBD in Unresectable Hilar Malignant Biliary Obstruction
2.4. TBD in Resectable Hilar Malignant Biliary Obstruction
2.5. Endoscopic Ultrasound-Guided Biliary Drainage
2.6. Endoscopic Ultrasound-Guided Choledochoduodenostomy
2.7. Endoscopic Ultrasound-Guided Hepaticogastrostomy
2.8. EUS-Guided Rendezvous
2.9. EUS-Guided Antegrade Drainage
2.10. EUS-Guided Gallbladder Drainage
3. Emerging Developments and Future Directions
3.1. EUS-Guided Gastroenterostomy Versus Surgical Gastrojejunostomy
3.2. Artificial Intelligence in Predicting Stent Failure and Complications
4. Conclusion
| Clinical Scenario | First-Line Approach | Second-Line/Rescue | Key Considerations |
|---|---|---|---|
| Unresectable DMBO [12,14,23,24,25,32,33,39,40,41] | ERCP with FC-SEMS or U-SEMS | EUS-CDS with LAMS or EUS-GBD | SEMS preferred; C-SEMS for longer patency |
| Resectable/Borderline Resectable DMBO [12,14,42,43,44,45,46,47] | SEMS (FC-SEMS preferred) | Avoid preoperative drainage except: cholangitis, high bilirubin, neoadjuvant therapy | FC-SEMS allows easy removal; Place ≥1.5 cm below confluence |
| Unresectable HMBO (Bismuth I-II) [12,14] | ERCP with SEMS (U-SEMS preferred) | EUS-HGS or PTBD | Similar to DMBO management |
| Unresectable HMBO (Bismuth III-IV) [12,14,48,49,50,51,52,53,54,55,56,57,58,59,60] | Bilateral ERCP stenting (>50% liver volume) OR PTBD | EUS-HGS or Combined approach | High-volume center; 3D-CT/MRCP; U-SEMS preferred |
| Resectable HMBO [48,61,62,63,64,65,66,67] | Generally avoid drainage; If needed: ERCP or PTBD | Surgery within 2 weeks after target bilirubin | Indications: FLR <30%, cholangitis, high bilirubin, neoadjuvant therapy |
| Failed ERCP in DMBO [68,69,70,71,72,73,80,81,82,83] | EUS-CDS with LAMS OR EUS-GBD | PTBD | Lower reintervention, pancreatitis; CBD >15mm better; EUS-GBD lower late morbidity |
| Failed ERCP in HMBO [80,84,85,86,87,88] | EUS-HGS | PTBD | Higher adverse events; Avoid in ascites, portal hypertension; PC-SEMS reduces RBO |
| Gastric Outlet Obstruction + MBO [96] | EUS-GE with biliary drainage | Surgical gastrojejunostomy | Faster recovery (1 vs 3 days), shorter stay (3 vs 9 days), lower cost |
| Authors, year | Title | Type of study | Number of studies/ patients | Key findings |
|---|---|---|---|---|
| Lopimpisuth et al., 2025 [41] | Postprocedural cholecystitis following covered self-expandable metal stent placement in patients with distal malignant biliary obstruction |
Systematic Review and Meta-Analysis | 21 studies 5753 patients |
CSEMS showed higher post-ERCP acute cholecystitis rates, lower tumor in growth rates, and higher rates of stent migration |
| Chung et al., 2025 [58] | Efficacy and safety of covered self-expandable metal stents for malignant hilar biliary obstruction |
Systematic Review and Meta-Analysis | 7 studies 194 patients |
High technical and clinical success rates of CSEMS placement in MHBO. Adverse events: cholangitis, cholecystitis, and pancreatitis, were <10%. |
| Zafar et al., 2025 [70] | Efficacy of endoscopic ultrasound-guided biliary drainage of malignant biliary obstruction | Systematic Review and Meta-Analysis | 8 studies 670 patients |
EUS-BD performed better than ERCP-BD and PTBD in reducing stent dysfunction, postprocedural pancreatitis, and tumor ingrowth or overgrowth. |
| Lauri et al., 2024 [73] | Primary drainage of distal malignant biliary obstruction |
A comparative network meta-analysis | 6 RCTs 583 patients |
EUS-CDS with LAMS had the highest technical and clinical success rates and was significantly superior to ERCP as the upfront technique for dMBO treatment. |
| van de Pavert et al., 2025 [96] | Endoscopic versus surgical gastroenterostomy for palliation of malignant gastric outlet obstruction (ENDURO) |
Multicenter RCT | 98 patients (48 endoscopic, 50 surgical) |
Endoscopic group demonstrated shorter time to solid oral intake (1 vs 3 days). Comparable reintervention rates (5 vs 6 patients). Overall adverse events 58% endoscopic vs 64% surgical (RR 0.91, 95% CI 0.66-1.25). Three fatal events in surgical group vs one in endoscopic group. |
References
- Okamoto, T. Malignant biliary obstruction due to metastatic non-hepato-pancreato-biliary cancer. World J Gastroenterol. 2022, 28, 985–1008. [Google Scholar] [CrossRef]
- Qiu, Feng MM; Yang, Tianchi MM; Han, Wei, PhD. Comparison of Biliary Drainage Techniques for MBO: A Meta-Analysis. Pancreas 2025, 54(9), p e796–e805. [Google Scholar] [CrossRef]
- Mikalsen, I. M.; Breder, S.; Medhus, A. W.; Folseraas, T.; Aabakken, L.; Ånonsen, K. V. ERCP for the initial management of malignant biliary obstruction – real world data on 596 procedures. Scandinavian Journal of Gastroenterology 2024, 59(3), 369–377. [Google Scholar] [CrossRef]
- Tsetis, D; Krokidis, Μ; Negru, D; et al. Malignant biliary obstruction: the current role of interventional radiology. Ann Gastroenterol. 2016, 29(1), 33–36. [Google Scholar]
- Pachisia, AV; Govil, D. Ultrasound-guided bedside percutaneous transhepatic biliary drainage in critically ill: a friend indeed. Indian J Crit Care Med. 2023, 27, 4–5. [Google Scholar]
- Binda, C.; Trebbi, M.; Coluccio, C.; Giuffrida, P.; Perini, B.; Gibiino, G.; Fabbri, S.; Liverani, E.; Fabbri, C. Endoscopic management of malignant biliary obstructions. Annals of gastroenterology 2024, 37(3), 291–302. [Google Scholar] [CrossRef] [PubMed]
- Canakis, A; Kahaleh, M. Endoscopic palliation of malignant biliary obstruction. World J Gastrointest Endosc 2022, 14, 581–596. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, Y; Viesca, M; Arvanitakis, M. Early diagnosis and management of malignant distal biliary obstruction: a review on current recommendations and guidelines. Clin Exp Gastroenterol 2019, 12, 415–432. [Google Scholar] [CrossRef]
- Fugazza, A; Fabbri, C; Di Mitri, R; i-EUS Group. EUS-guided choledochoduodenostomy for malignant distal biliary obstruction after failed ERCP: a retrospective nationwide analysis. Gastrointest Endosc 2022, 95, 896–904. [Google Scholar] [CrossRef]
- Pietrzak, J.; Przybyłkowski, A. Endoscopic Treatment of Malignant Hilar Biliary Obstruction. Cancers 2023, 15(24), 5819. [Google Scholar] [CrossRef] [PubMed]
- Mosconi, C.; Renzulli, M.; Giampalma, E.; Galuppi, A.; Balacchi, C.; Brandi, G.; Ercolani, G.; Bianchi, G.; Golfieri, R. Unresectable perihilar cholangiocarcinoma: Multimodal palliative treatment. Anticancer. Res. 2013, 33, 2747–2753. [Google Scholar]
- Elmunzer, BJ; Maranki, JL; Gómez, V; et al. ACG Clinical Guideline: Diagnosis and management of biliary strictures. Am J Gastroenterol 2023, 118, 405–426. [Google Scholar] [CrossRef]
- van der Merwe, SW; van Wanrooij, RLJ; Bronswijk, M; et al. Therapeutic endoscopic ultrasound: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy 2022, 54, 185–205. [Google Scholar] [CrossRef]
- Dumonceau, JM; Tringali, A; Papanikolaou, IS; et al. Endoscopic biliary stenting: indications, choice of stents, and results: European Society of Gastrointestinal Endoscopy (ESGE) Clinical Guideline – Updated October 2017. Endoscopy 2018, 50, 910–930. [Google Scholar] [CrossRef]
- Ekkelenkamp, VE; de Man, RA; Ter Borg, F; et al. Prospective evaluation of ERCP performance: results of a nationwide quality registry. Endoscopy 2015, 47, 503–507. [Google Scholar] [CrossRef] [PubMed]
- de Jong, M. J. P.; van Delft, F.; van Geenen; et al. Endoscopic ultrasound-guided choledochoduodenostomy results in fewer complications than percutaneous drainage following failed ERCP in malignant distal biliary obstruction. Endoscopy 2025, 57(9), 1004–1015. [Google Scholar] [CrossRef] [PubMed]
- Alves de Lima, SL; Bustamante, FAC; Hourneaux de Moura, EG; et al. Endoscopic palliative treatment vs surgical bypass in malignant low bile duct obstruction:a systematic review and meta-analysis. Int J Hepatobiliary Pancreat Dis. 2015, 5, 35–45. [Google Scholar] [CrossRef]
- Inamdar, S; Slattery, E; Bhalla, R; Sejpal, DV; Trindade, AJ. Comparison of adverse events for endoscopic vs percutaneous biliary drainage in the treatment of malignant biliary tract obstruction in an inpatient national cohort. JAMA Oncol. 2016, 2, 112–117. [Google Scholar] [CrossRef]
- Deniz, S; Öcal, O; Wildgruber, M; et al. Percutaneous transhepatic biliary drainage (PTBD) in patients with biliary leakage: Technical and clinical outcomes. Medicine (Baltimore) 2023, 102, e35213. [Google Scholar] [CrossRef]
- Nennstiel, S; Weber, A; Frick, G; et al. Drainage-related complications in percutaneous transhepatic biliary drainage:an analysis over 10 years. J Clin Gastroenterol. 2015, 49, 764–770. [Google Scholar] [CrossRef] [PubMed]
- Park, C. H.; Park, S. W.; Jung, J. H.; Jung, E. S.; Kim, J. H.; Park, D. H. Comparative Efficacy of Various Stents for Palliation in Patients with Malignant Extrahepatic Biliary Obstruction: A Systematic Review and Network Meta-Analysis. Journal of personalized medicine 2021, 11(2), 86. [Google Scholar] [CrossRef]
- Walta, D.C.; Fausel, C.S.; Brant, B. Endoscopic biliary stents and obstructive jaundice. Am. J. Surg. 1987, 153, 444–447. [Google Scholar] [CrossRef]
- A Almadi, M.; Barkun, A.; Martel, M. Plastic vs. self-expandable metal stents for palliation in malignant biliary obstruction: A series of meta-analyses. Am. J. Gastroenterol. 2017, 112, 260–273. [Google Scholar] [CrossRef] [PubMed]
- Isayama, H.; Hamada, T.; Yasuda, I.; et al. TOKYO criteria 2014 for transpapillary biliary stenting. Dig. Endosc. 2015, 27, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Sawas, T.; Al Halabi, S.; Parsi, M.A.; Vargo, J. Self-expandable metal stents versus plastic stents for malignant biliary obstruction: A meta-analysis. Gastrointest. Endosc. 2015, 82, 256–267. [Google Scholar] [CrossRef] [PubMed]
- Walter, D.; Van Boeckel, P.G.A.; Groenen, M.J.; et al. Cost efficacy of metal stents for palliation of extrahepatic bile duct obstruction in a randomized controlled trial. Gastroenterology 2015, 149, 130–138. [Google Scholar] [CrossRef]
- Bor, R.; Fábián, A.; Szűcs, M.; et al. Comparison of therapeutic efficacy and treatment costs of self-expandable metal stents and plastic stents for management of malignant biliary obstruction. BMC Gastroenterol 2023, 23, 41. [Google Scholar] [CrossRef]
- Tamura, T; Ashida, R; Kawaji, Y; et al. Self-expandable metal vs. plastic stents for preoperative biliary drainage in patients receiving neoadjuvant chemotherapy. Clin Endosc. 2025, 58(6), 817–825. [Google Scholar] [CrossRef]
- Endo, Y; Tanaka, M; Kitago, M; et al. Comparison between plastic and metallic biliary stent placement for preoperative patients with pancreatic head cancer: a systematic review and meta-analysis. Ann Surg Oncol 2024, 31, 1319–1327. [Google Scholar] [CrossRef]
- Coté, GA; Kumar, N; Ansstas, M; et al. Risk of post-ERCP pancreatitis with placement of self-expandable metallic stents. Gastrointest Endosc 2010. [Google Scholar] [CrossRef]
- Tamura, T; Yamai, T; Uza, N; et al. Adverse events of self-expandable metal stent placement for malignant distal biliary obstruction: a large multicenter study. Gastrointest Endosc 2024, 99, 61–72. [Google Scholar] [CrossRef]
- Tringali, A; Hassan, C; Rota, M; et al. Covered vs. uncovered self-expandable metal stents for malignant distal biliary strictures: a systematic review and meta-analysis. Endoscopy 2018, 50(06), 631–41. [Google Scholar]
- Vanella, G; Coluccio, C; Cucchetti, A; et al. Fully covered versus partially covered self-expandable metal stents for palliation of distal malignant biliary obstruction: a systematic review and meta-analysis. Gastrointest Endosc. 2024, 99(3), 314–322. [Google Scholar] [CrossRef] [PubMed]
- Park, C. H.; Park, S. W.; Jung, J. H.; Jung, E. S.; Kim, J. H.; Park, D. H. Comparative Efficacy of Various Stents for Palliation in Patients with Malignant Extrahepatic Biliary Obstruction: A Systematic Review and Network Meta-Analysis. Journal of personalized medicine 2021, 11(2), 86. [Google Scholar] [CrossRef] [PubMed]
- Lee, JH; Krishna, SG; Singh, A; et al. Comparison of the utility of covered metal stents versus uncovered metal stents in the management of malignant biliary strictures in 749 patients. Gastrointest Endosc. 2013, 78, 312–324. [Google Scholar] [CrossRef] [PubMed]
- Isayama, H; Komatsu, Y; Tsujino, T; et al. A prospective randomised study of “covered”versus “uncovered”diamond stents for the management of distal malignant biliary obstruction. Gut 2004, 53, 729–734. [Google Scholar] [CrossRef]
- Kitano, M; Yamashita, Y; Tanaka, K; et al. Covered self-expandable metal stents with an anti-migration system improve patency duration without increased complications compared with uncovered stents for distal biliary obstruction caused by pancreatic carcinoma: a randomized multicenter trial. Am J Gastroenterol. 2013, 108, 1713–1722. [Google Scholar] [CrossRef]
- Ghazi, R; AbiMansour, JP; Mahmoud, T; et al. Uncovered versus fully covered self-expandable metal stents for the management of distal malignant biliary obstruction. Gastrointest Endosc. 2023, 98, 577–584. [Google Scholar] [CrossRef]
- Zhang, D.; Luo, T.; Gao, F.; et al. Comparison of fully covered versus uncovered self-expandable metallic stents in treating inoperable malignant distal biliary obstruction: a retrospective study. Therapeutic advances in gastrointestinal endoscopy 2025, 18. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, Y; Tachikawa, A; Shimokawa, T; et al. Covered versus uncovered metal stent for endoscopic drainage of a malignant distal biliary obstruction: Meta-analysis. Dig Endosc. 2022, 34, 938–951. [Google Scholar] [CrossRef]
- Lopimpisuth, C; Vedantam, S; Danpanichkul, P; et al. Postprocedural cholecystitis following covered self-expandable metal stent placement in patients with distal malignant biliary obstruction: a systematic review and meta-analysis. Gastrointest Endosc 2025. [Google Scholar] [CrossRef]
- Scheufele, F; Schorn, S; Demir, IE; et al. Preoperative biliary stenting versus operation first in jaundiced patients due to malignant lesions in the pancreatic head: A meta-analysis of current literature. Surgery 2017, 161, 939–950. [Google Scholar] [CrossRef]
- van der Gaag, NA; Rauws, EA; van Eijck, CH; Bruno, MJ; van der Harst, E; Kubben, FJ; Gerritsen, JJ; Greve, JW; Gerhards, MF; de Hingh, IH; Klinkenbijl, JH; Nio, CY; de Castro, SM; Busch, OR; van Gulik, TM; Bossuyt, PM; Gouma, DJ. Preoperative biliary drainage for cancer of the head of the pancreas. N Engl J Med 2010, 362(2), 129–37. [Google Scholar] [CrossRef]
- Jang, S; Stevens, T; Parsi, MA; et al. Superiority of self-expandable metallic stents over plastic stents in treatment of malignant distal biliary strictures. Clinical Gastroenterology and Hepatology 2022, 20(2), e182-95. [Google Scholar] [CrossRef]
- Aadam, AA; Evans, DB; Khan, A; et al. Efficacy and safety of self-expandable metal stents for biliary decompression in patients receiving neoadjuvant therapy for pancreatic cancer: a prospective study. Gastrointestinal endoscopy 2012, 76(1), 67–75. [Google Scholar] [CrossRef]
- Siddiqui, AA; Mehendiratta, V; Loren, D; et al. Self-expanding metal stents (SEMS) for preoperative biliary decompression in patients with resectable and borderline-resectable pancreatic cancer: outcomes in 241 patients. Digestive diseases and sciences 2013, 58(6), 1744–50. [Google Scholar] [CrossRef] [PubMed]
- Ballard, DD; Rahman, S; Ginnebaugh, B; et al. Safety and efficacy of self-expanding metal stents for biliary drainage in patients receiving neoadjuvant therapy for pancreatic cancer. Endoscopy International Open 2018, 6(06), E714-21. [Google Scholar] [CrossRef] [PubMed]
- Mansour, JC; Aloia, TA; Crane, CH; et al. Hilar cholangiocarcinoma: expert consensus statement. HPB (Oxford) 2015, 17, 691–9. [Google Scholar] [CrossRef] [PubMed]
- Lee, TH. Proper management of inoperable malignant hilar biliary obstruction. International Journal of Gastrointestinal Intervention 2021, 10(3), 120–7. [Google Scholar] [CrossRef]
- Rerknimitr, R; Angsuwatcharakon, P; Ratanachu-ek, T; et al. Asia-Pacific consensus recommendations for endoscopic and interventional management of hilar cholangiocarcinoma. J Gastroenterol Hepatol. 2013, 28, 593–607. [Google Scholar] [CrossRef]
- Dhar, J.; Gupta, P.; Samanta, J. The role of endoscopy in malignant hilar obstruction. Annals of gastroenterology 2023, 36(4), 347–359. [Google Scholar] [CrossRef]
- Paik, WH; Park, YS; Hwang, JH; et al. Palliative treatment with self-expandable metallic stents in patients with advanced type III or IV hilar cholangiocarcinoma: a percutaneous versus endoscopic approach. Gastrointest Endosc. 2009, 69, 55–62. [Google Scholar] [CrossRef]
- Qumseya, BJ; Jamil, LH; Elmunzer, BJ; et al. ASGE guideline on the role of endoscopy in the management of malignant hilar obstruction. Gastrointest Endosc. 2021, 94, 222–234. [Google Scholar] [CrossRef]
- Vienne, A; Hobeika, E; Gouya, H; et al. Prediction of drainage effectiveness during endoscopic stenting of malignant hilar strictures: the role of liver volume assessment. Gastrointest Endosc. 2010, 72, 728–735. [Google Scholar] [CrossRef]
- Lee, TH; Moon, JH; Choi, JH; et al. Prospective comparison of endoscopic bilateral stent-in-stent versus stent-by-stent deployment for inoperable advanced malignant hilar biliary stricture. Gastrointest Endosc. 2019, 90, 222–230. [Google Scholar] [CrossRef] [PubMed]
- De Palma, GD; Galloro, G; Siciliano, S; et al. Unilateral versus bilateral endoscopic hepatic duct drainage in patients with malignant hilar biliary obstruction: results of a prospective, randomized, and controlled study. Gastrointest Endosc. 2001, 53, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Ashat, M.; Arora, S.; Klair, J. S.; et al. Bilateral vs unilateral placement of metal stents for inoperable high-grade hilar biliary strictures: A systemic review and meta-analysis. World journal of gastroenterology 2019, 25(34), 5210–5219. [Google Scholar] [CrossRef]
- Chung, KH; Lee, KJ; Joseph, AA; et al. Efficacy and safety of covered self-expandable metal stents for malignant hilar biliary obstruction: systematic review and meta-analysis. Gastrointest Endosc. 2025, 101(2), 350–357. [Google Scholar] [CrossRef]
- Chen, L.; Gao, G. M.; Li, D. L.; Chen, Z. K. Side-by-side versus stent-in-stent bilateral stenting for malignant hilar biliary obstruction: a meta-analysis. Videosurgery and other miniinvasive techniques 2022, 17(2), 279–288. [Google Scholar] [CrossRef]
- Imamura, S.; Watanabe, K.; Inoue, K.; et al. Side-by-side versus stent-in-stent technique for stent deployment during systemic chemotherapy in biliary tract cancer patients. DEN open 2025, 5(1), e70075. [Google Scholar] [CrossRef] [PubMed]
- Wiggers, JK; Groot Koerkamp, B; Cieslak, KP; et al. Postoperative mortality after liver resection for perihilar cholangiocarcinoma. J Am Coll Surg. 2016, 223, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Celotti, A; Solaini, L; Montori, G; et al. Preoperative biliary drainage in hilar cholangiocarcinoma: Systematic review and meta-analysis. Eur J Surg Oncol. 2017, 43, 1628–1635. [Google Scholar] [CrossRef] [PubMed]
- Hameed, A; Pang, T; Chiou, J; et al. Percutaneous vs. endoscopic pre-operative biliary drainage in hilar cholangiocarcinoma. HPB (Oxford) 2016, 18, 400–410. [Google Scholar] [CrossRef] [PubMed]
- Al Mahjoub, A; Menahem, B; Fohlen, A; et al. Preoperative biliary drainage in patients with resectable perihilar cholangiocarcinoma. J Vasc Interv Radiol. 2017, 28, 576–582. [Google Scholar] [CrossRef]
- Coelen, RJS; Roos, E; Wiggers, JK; et al. Endoscopic versus percutaneous biliary drainage in patients with resectable perihilar cholangiocarcinoma. Lancet Gastroenterol Hepatol. 2018, 3, 681–690. [Google Scholar] [CrossRef]
- Kawakami, H; Kuwatani, M; Onodera, M; et al. Endoscopic nasobiliary drainage is the most suitable preoperative biliary drainage method. J Gastroenterol. 2011, 46, 242–248. [Google Scholar] [CrossRef]
- Paik, WH; Loganathan, N; Hwang, JH. Preoperative biliary drainage in hilar cholangiocarcinoma: when and how? World J Gastrointest Endosc. 2014, 6, 68–73. [Google Scholar] [CrossRef]
- Barbosa, EC; do Espírito Santo, PA; Baraldo, S; et al. EUS- versus ERCP-guided biliary drainage for malignant biliary obstruction. Gastrointest Endosc 2024, 100, 395–405. [Google Scholar] [CrossRef]
- Paik, WH; Lee, TH; Park, DH; et al. EUS-Guided Biliary Drainage Versus ERCP for the Primary Palliation of Malignant Biliary Obstruction: A Multicenter Randomized Clinical Trial. Am J Gastroenterol. 2018, 113, 987–997. [Google Scholar] [CrossRef]
- Zafar, Y; Azam, H; Azhar, MAB; Shaheen, F; Javaid, SS; Manzoor, L; Masood, M; Krishnamoorthi, R. Efficacy of endoscopic ultrasound-guided biliary drainage of malignant biliary obstruction: a systematic review and meta-analysis of randomized controlled trials. Clin Endosc. 2025, 58(4), 533–543. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lambin, T.; Leblanc, S.; Napoléon, B. Advances in EUS-Guided Biliary Drainage for the Management of Pancreatic Cancer. Cancers 2025, 17(21), 3428. [Google Scholar] [CrossRef]
- Amato, A.; Sinagra, E.; Celsa, C.; et al. Efficacy of Lumen-Apposing Metal Stents or Self-Expandable Metal Stents for Endoscopic Ultrasound-Guided Choledochoduodenostomy. Endoscopy 2021, 53, 1037–1047. [Google Scholar] [CrossRef] [PubMed]
- Lauri, G; Archibugi, L; Arcidiacono, PG; et al. Primary drainage of distal malignant biliary obstruction: A comparative network meta-analysis. Dig Liver Dis. 2024, 56(12), 2004–2010. [Google Scholar] [CrossRef] [PubMed]
- Uemura, R.S.; Khan, M.A.; Otoch, J.P.; et al. EUS-Guided Choledochoduodenostomy Versus Hepaticogastrostomy. J. Clin. Gastroenterol. 2018, 52, 123–130. [Google Scholar] [CrossRef]
- Krishnamoorthi, R.; Dasari, C.S.; Chandrasekar, V.T.; et al. Effectiveness and Safety of EUS-Guided Choledochoduodenostomy Using Lumen-Apposing Metal Stents. Surg. Endosc. 2020, 34, 2866–2877. [Google Scholar] [CrossRef]
- Li, J.-S.; Tang, J.; Fang, J.; et al. Adverse Events in Endoscopic Ultrasound-Guided Choledochoduodenostomy with Lumen-Apposing Metal Stents. J. Gastroenterol. Hepatol. 2024, 39, 1769–1779. [Google Scholar] [CrossRef]
- Fugazza, A.; Khalaf, K.; Spadaccini, M.; et al. Outcomes Predictors in Endoscopic Ultrasound-Guided Choledochoduodenostomy with Lumen-Apposing Metal Stent. Endosc. Int. Open. 2024, 12, E456–E462. [Google Scholar]
- Khoury, T.; Sbeit, W.; Fumex, F.; et al. Endoscopic Ultrasound- versus ERCP-Guided Primary Drainage of Inoperable Malignant Distal Biliary Obstruction. Endoscopy 2024, 56, 955–963. [Google Scholar] [CrossRef]
- Ogura, T; Itoi, T. Technical Tips and Recent Development of Endoscopic Ultrasound-Guided Choledochoduodenostomy. DEN Open 2021, 1, e8. [Google Scholar] [CrossRef]
- Mohan, BP; Shakhatreh, M; Garg, R; et al. Efficacy and safety of endoscopic ultrasound-guided choledochoduodenostomy. J Clin Gastroenterol. 2019, 53, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Beunon, C; Debourdeau, A; Schaefer, M; et al. Technical failure of endoscopic ultrasound-guided choledochoduodenostomy. Endoscopy 2025, 57(9), 990–1000. [Google Scholar] [CrossRef]
- Chen, YI; Long, C; Sahai, AV; et al. Stent misdeployment and factors associated with failure in endoscopic ultrasound-guided choledochoduodenostomy. Endoscopy 2025, 57(4), 330–338. [Google Scholar] [PubMed]
- Vanella, G; Bronswijk, M; Dell’Anna, G; et al. Classification, risk factors, and management of lumen apposing metal stent dysfunction. Dig Endosc. 2023, 35, 377–388. [Google Scholar] [CrossRef]
- Giovannini, M.; Moutardier, V.; Pesenti, C.; et al. Endoscopic ultrasound-guided bilioduodenal anastomosis. Endoscopy 2001, 33, 898–900. [Google Scholar] [CrossRef]
- Mazza, S.; Masciangelo, G.; Mauro, A.; et al. Endoscopic Ultrasound-Guided Hepaticogastrostomy in Malignant Biliary Obstruction. Diagnostics 2024, 14(23), 2644. [Google Scholar] [CrossRef]
- van Wanrooij, R.L.J.; Bronswijk, M.; Kunda, R.; et al. Therapeutic Endoscopic Ultrasound: ESGE Technical Review. Endoscopy 2022, 54, 310–332. [Google Scholar] [CrossRef]
- Rizqiansyah, CY; Awatara, PID; Amar, N; et al. Efficacy and safety of EUS hepaticogastrostomy versus choledochoduodenostomy. JGH Open 2024, 8(10), e70037. [Google Scholar] [CrossRef] [PubMed]
- Hedjoudje, A; Pokossy Epée, J; Perez-Cuadrado-Robles, E; et al. Long-term outcomes of endoscopic ultrasound-guided hepaticogastrostomy. United European Gastroenterol J 2024, 12(8), 1044–1055. [Google Scholar] [CrossRef] [PubMed]
- Gornals, J. B.; Sumalla-Garcia, A.; Quintana, S.; et al. Endoscopic ultrasound-guided biliary rendezvous after failed cannulation. Annals of Medicine 2024, 56(1). [Google Scholar] [CrossRef]
- Iwashita, T; Uemura, S; Tezuka, R; et al. Current status of endoscopic ultrasound-guided antegrade intervention. Dig Endosc. 2023, 35, 264–274. [Google Scholar] [CrossRef]
- Khoury, T.; Farraj, M.; Sbeit, W.; et al. EUS-Guided Gallbladder Drainage of Inoperable Malignant Distal Biliary Obstruction. Cancers 2025, 17, 1983. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, G.E.M.; Crinò, S.F.; Vanella, G.; et al. EUS-Guided Gallbladder Drainage as a Rescue in Distal Malignant Biliary Obstruction. Endosc. Ultrasound 2025, 14, 41–47. [Google Scholar] [CrossRef]
- Kamal, F.; Khan, M.A.; Lee-Smith, W.; et al. Efficacy and Safety of EUS-Guided Gallbladder Drainage for Rescue Treatment. Endosc. Ultrasound 2022, 12, 8–15. [Google Scholar] [CrossRef]
- Debourdeau, A.; Daniel, J.; Caillo, L.; et al. Effectiveness of endoscopic ultrasound-guided choledochoduodenostomy vs. EUS-guided gallbladder drainage. Digestive endoscopy 2025, 37(1), 103–114. [Google Scholar] [CrossRef]
- Fernandes, Matheus Vanzin; et al. Endoscopic Ultrasound-Guided Gastroenterostomy vs Enteral Stenting for Malignant Gastric Outlet Obstruction: A Systematic Review and Meta-analysis. Techniques and Innovations in Gastrointestinal Endoscopy 2025, Volume 27(Issue 3). [Google Scholar] [CrossRef]
- van de Pavert, YL; et al. Endoscopic versus surgical gastroenterostomy for palliation of malignant gastric outlet obstruction (ENDURO). The Lancet Gastroenterology & Hepatology Volume 10(Issue 12), 1065–1074. [CrossRef]
- Zhao, R.; Gu, L.; Ke, X.; et al. Risk prediction of cholangitis after stent implantation based on machine learning. Sci Rep 2024, 14, 13715. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Qi, J.; Zhang, X.; et al. Development and application of an early warning model for predicting early mortality. Oncology letters 2025, 29(5), 237. [Google Scholar]
- Jin, H.; Sun, X.; Fu, C.; et al. Machine learning-based prediction model for post-ERCP cholangitis. Surg Endosc 2025, 39, 5107–5126. [Google Scholar] [CrossRef] [PubMed]
| Stent Type | Advantages | Disadvantages | Clinical Recommendations |
|---|---|---|---|
| Plastic Stents |
|
|
Limited role; consider for temporary drainage or very short life expectancy |
| Uncovered SEMS |
|
|
Suitable for unresectable DMBO; preferred for hilar obstruction |
| Covered SEMS (FC/PC) |
|
|
FC-SEMS preferred for resectable/borderline resectable disease; consider for unresectable DMBO |
| Technique | Primary Indication | Technical Success | Clinical Success | Major Complications |
|---|---|---|---|---|
| EUS-CDS | Failed ERCP in DMBO; preferred EUS-BD approach for distal obstruction | 93.5-96% | 88-96% |
|
| EUS-HGS | Failed ERCP in HMBO; gastric outlet obstruction; altered anatomy | Similar to EUS-CDS | Similar to EUS-CDS |
|
| EUS-RV | Failed ERCP (second attempt); benign biliary disease with normal anatomy | 72-96% (expert hands) | 84-86% |
|
| EUS-AG | Failed ERCP with preservation of normal anatomy | 92% | Not reported |
|
| EUS-GBD | Failed ERCP in DMBO; alternative to EUS-CDS with potentially lower late morbidity | 99.2-100% | 85-89% |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





