Submitted:
26 January 2026
Posted:
26 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Industrial Production of Cumene
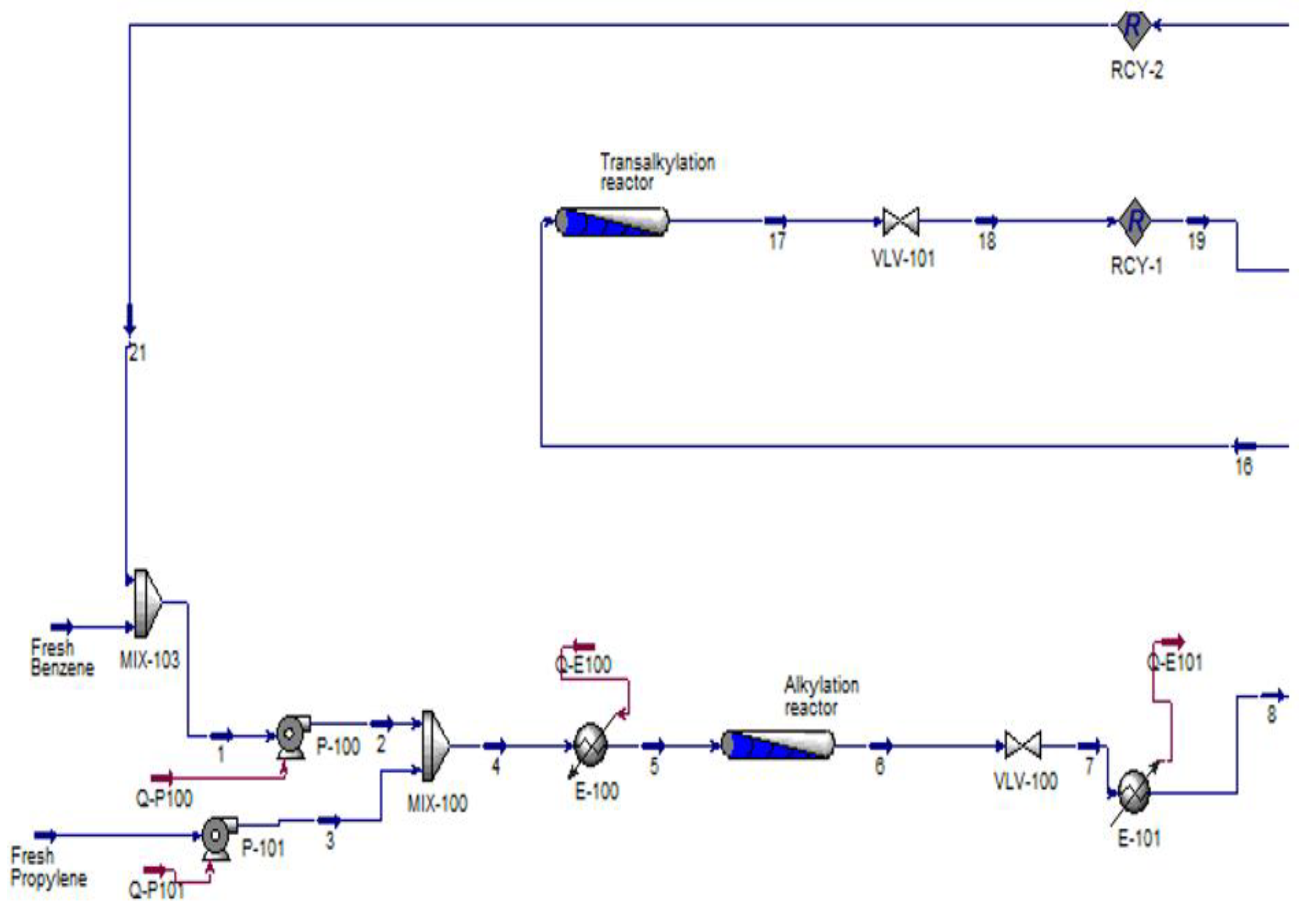
2.1. Alkylation Reaction
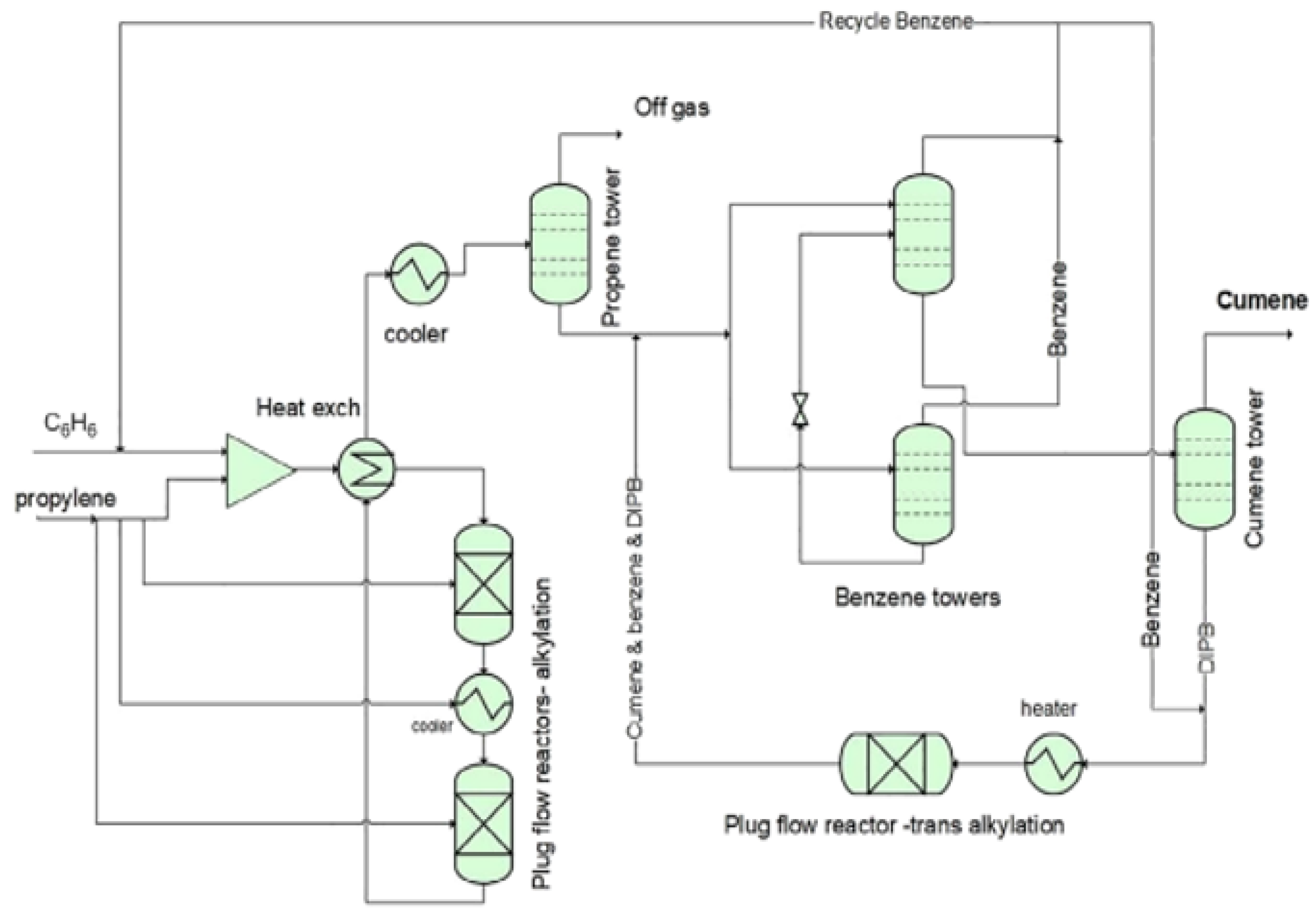
2.2. Transalkylation Reaction
2.3. Reaction Kinetics
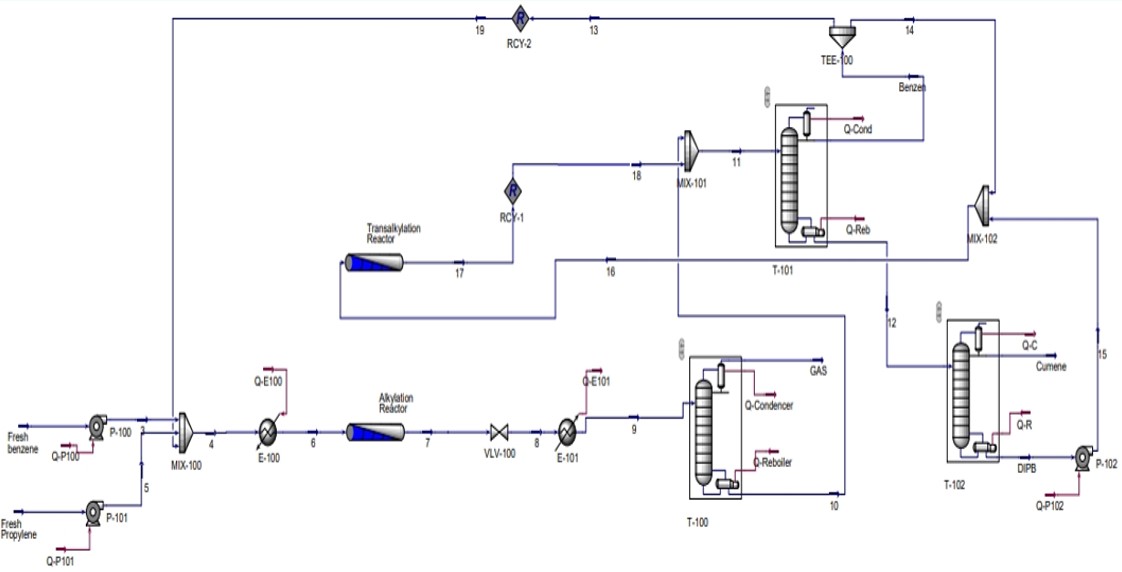
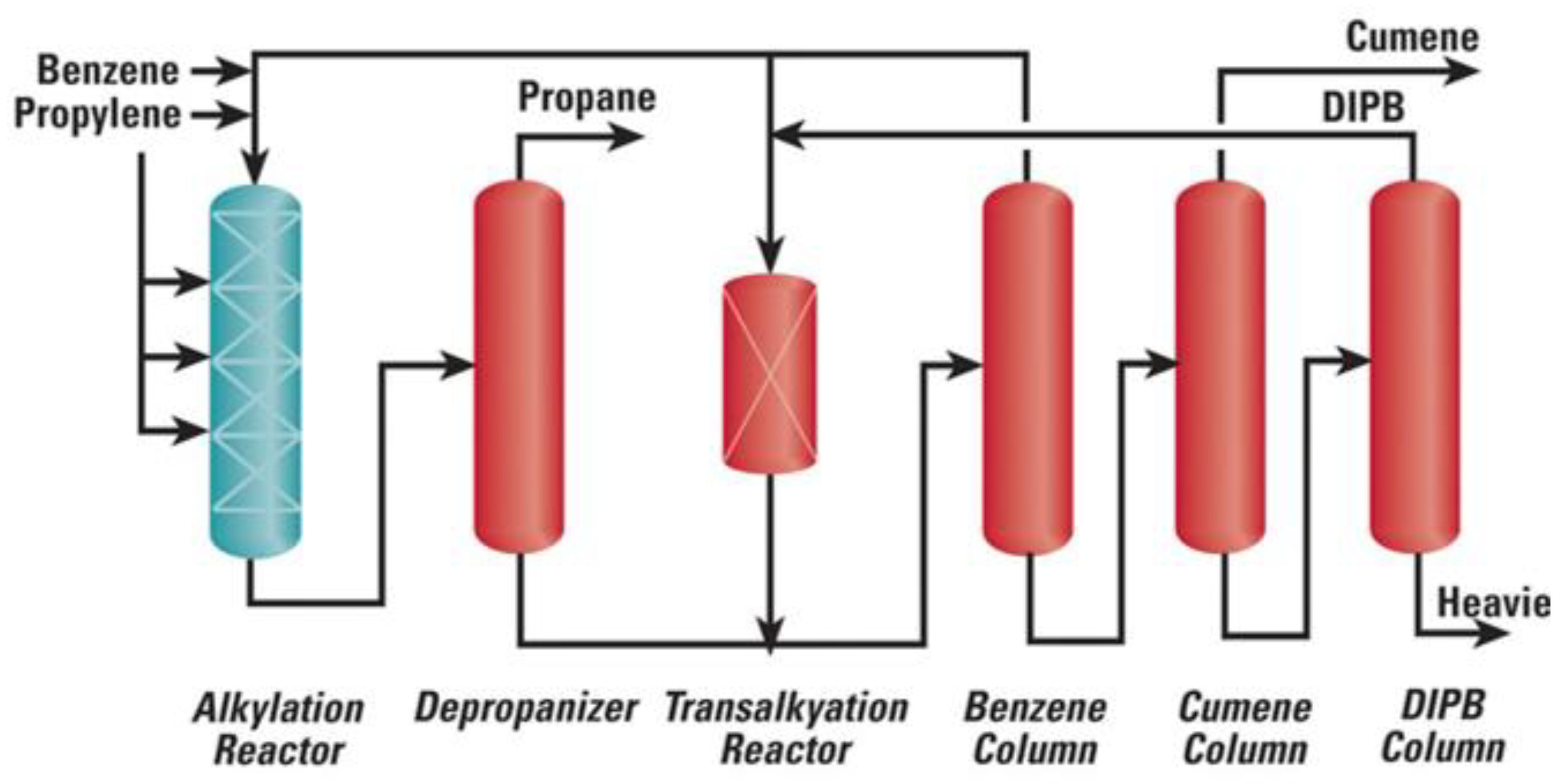
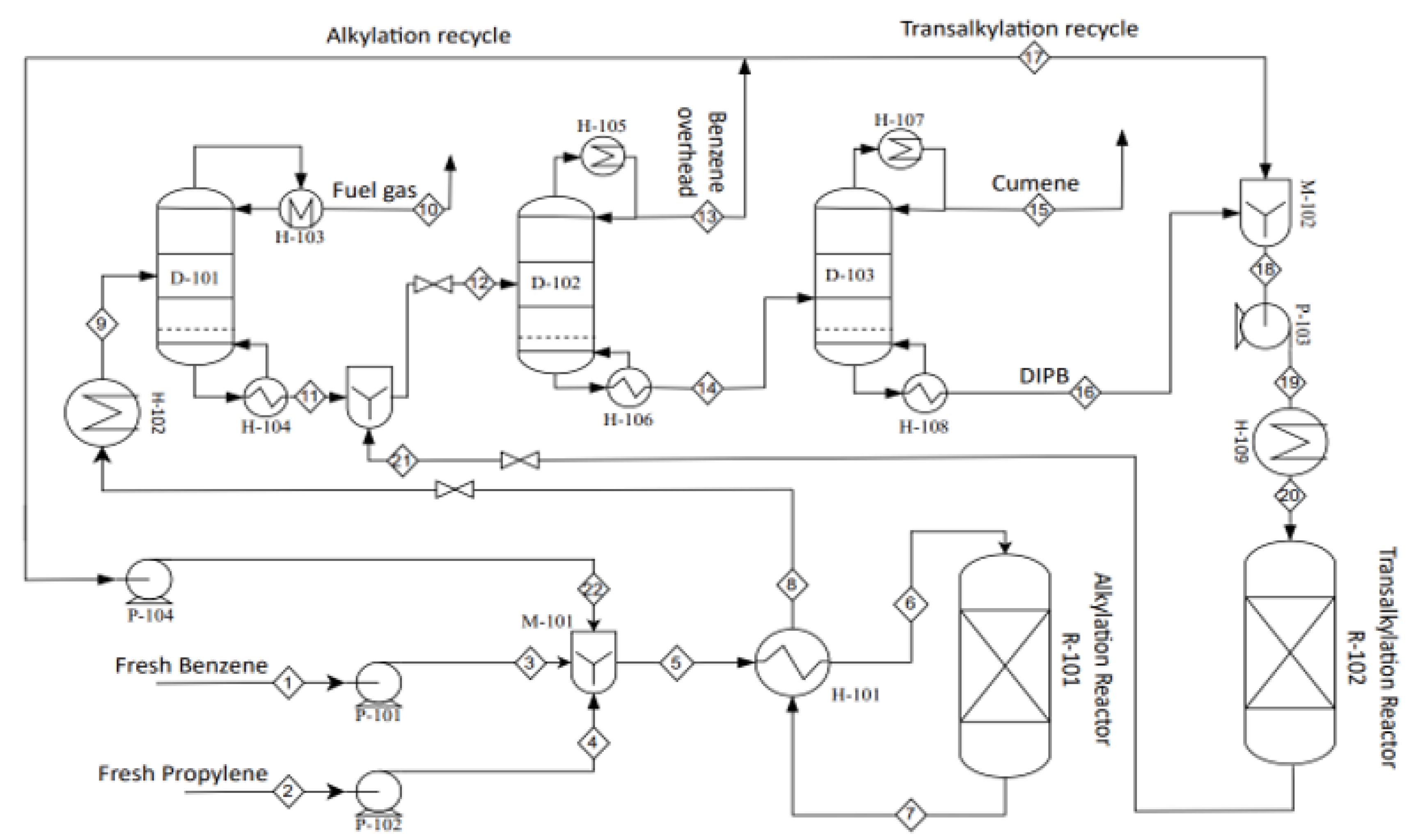
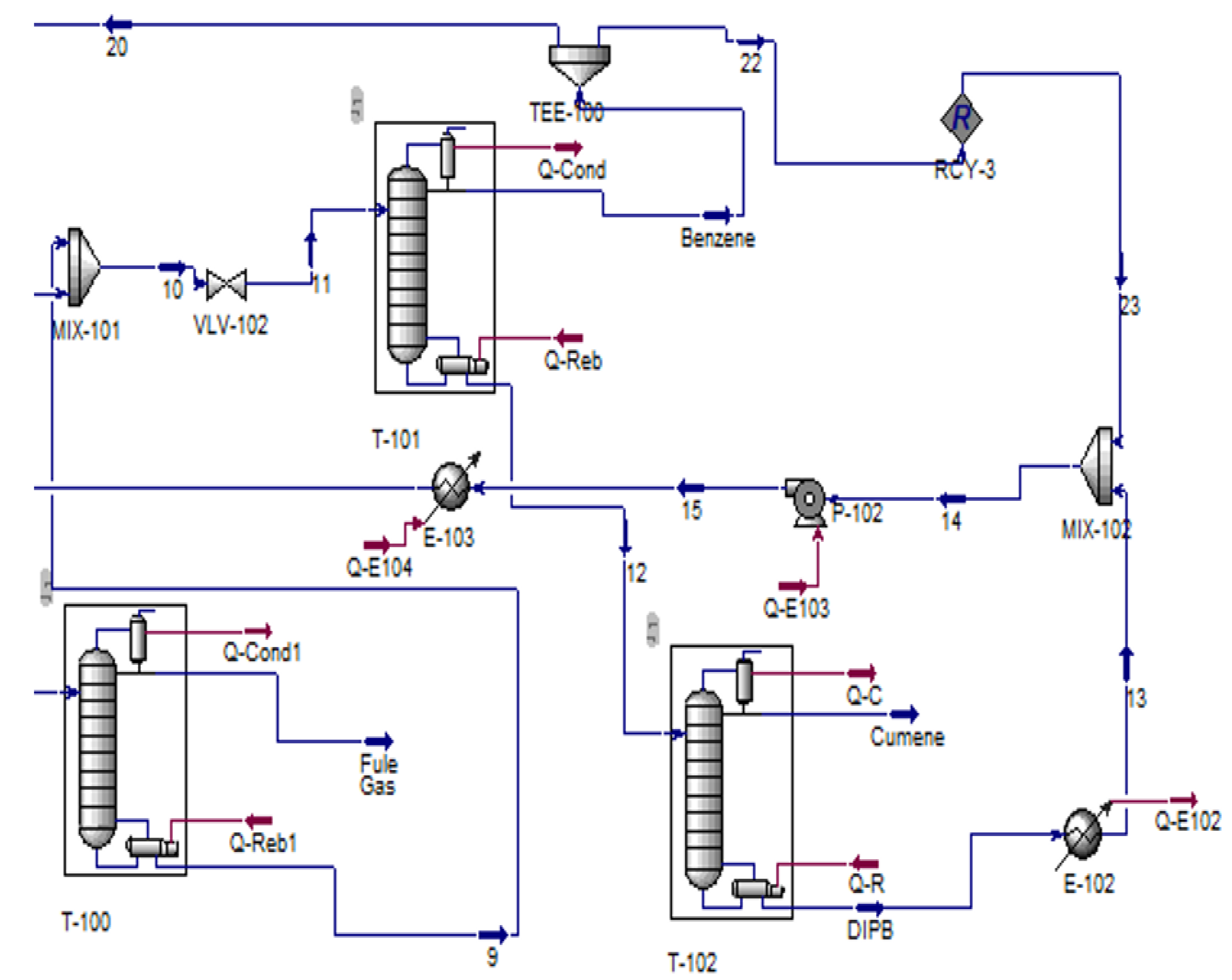
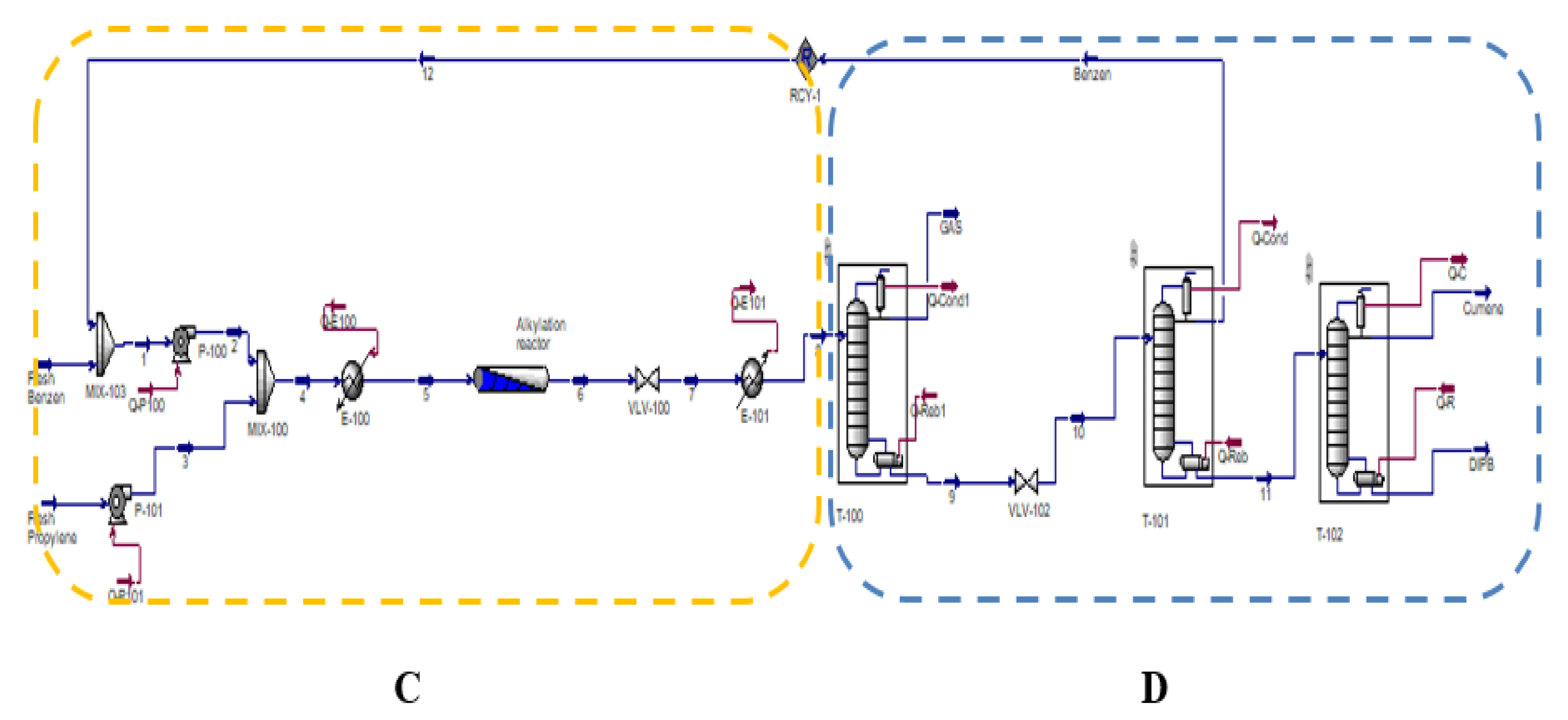
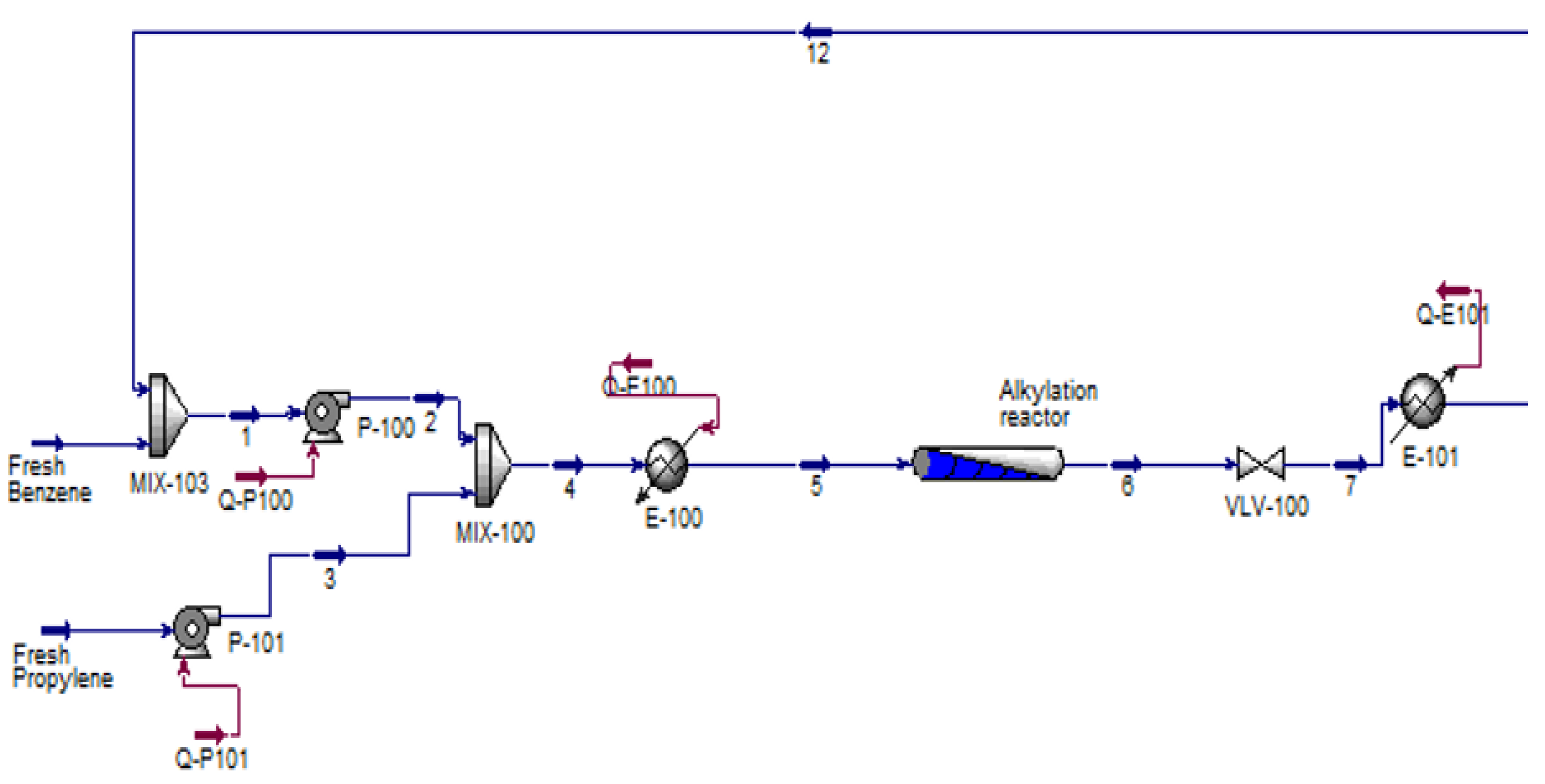
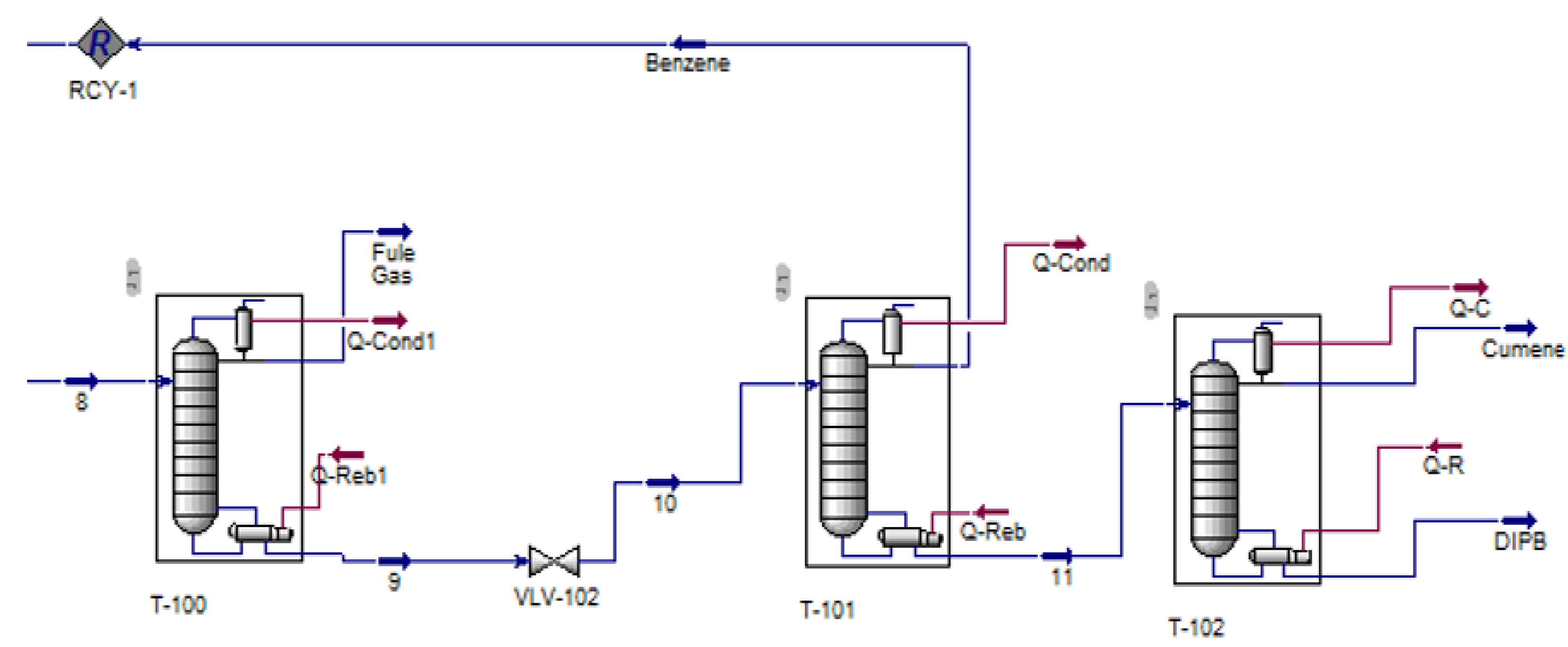
2.4. Cumene Production with Transalkylation Reactor
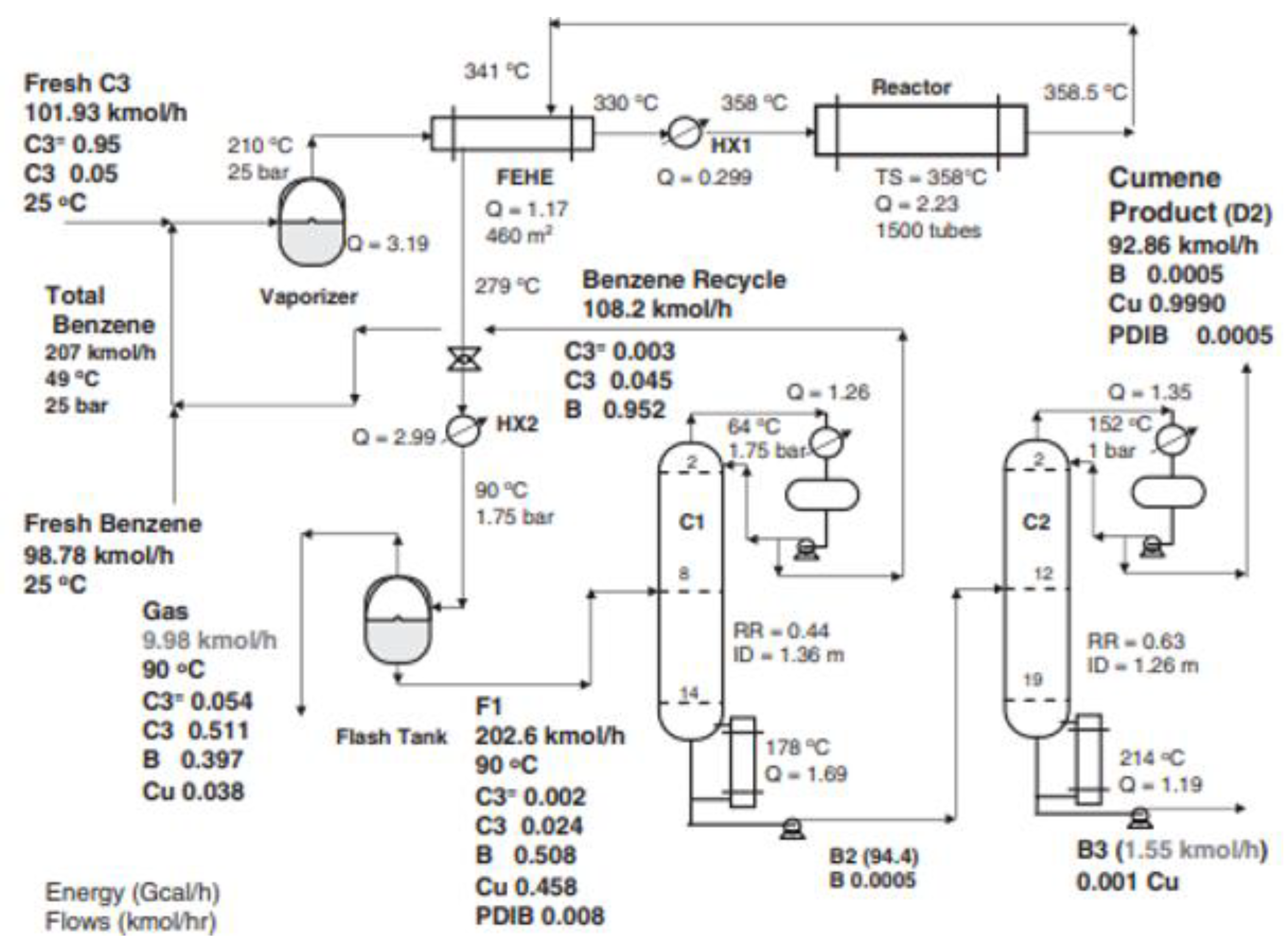
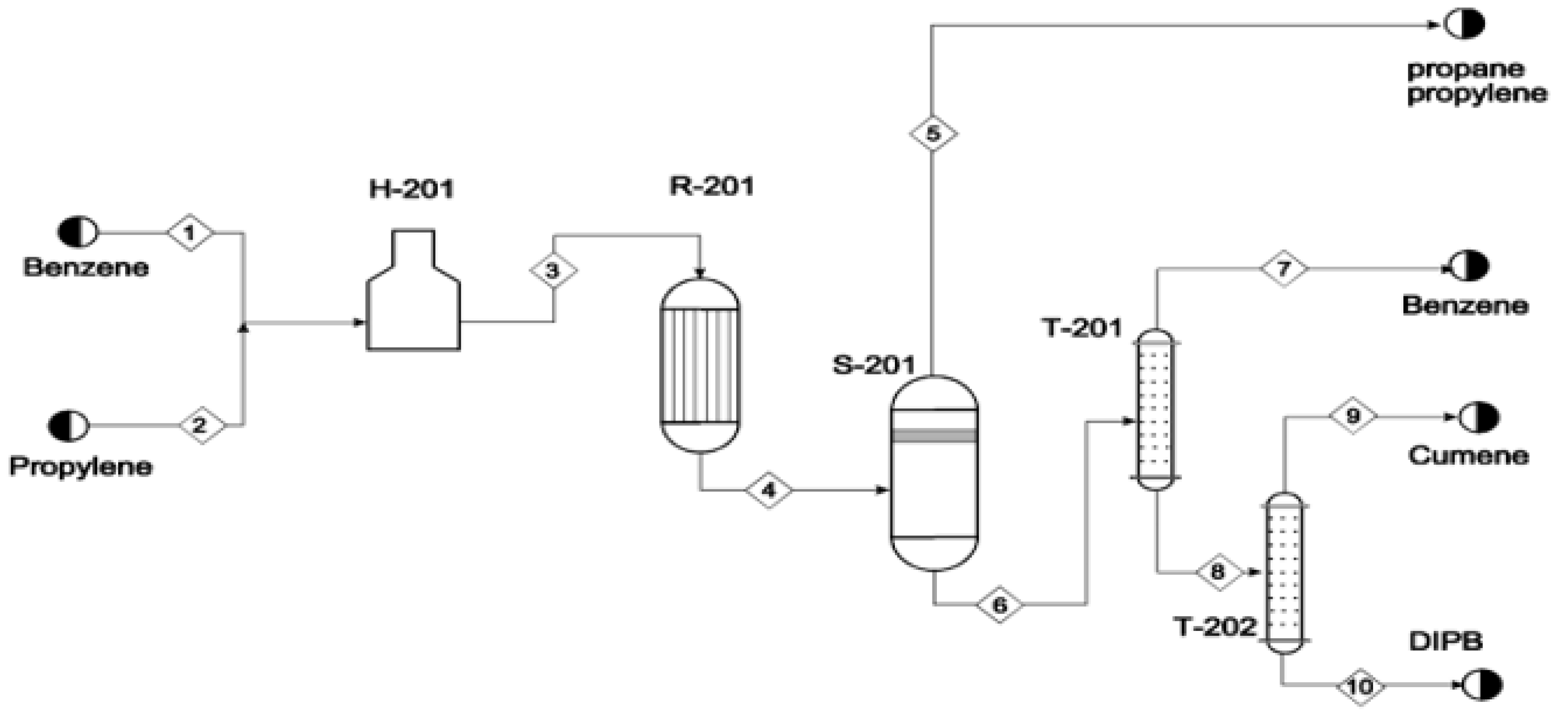
2.5. Cumene Production without Transalkylation Reactor
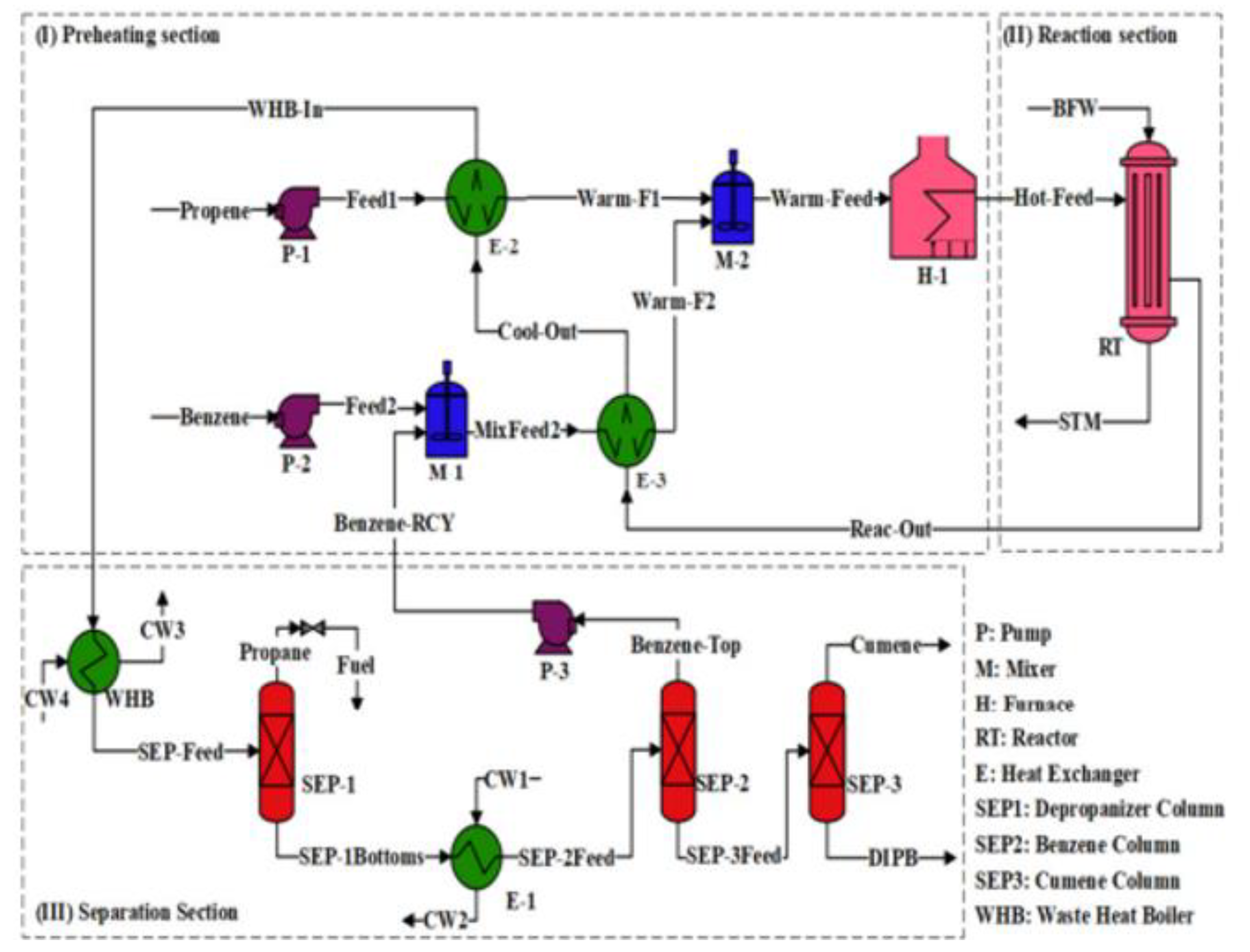
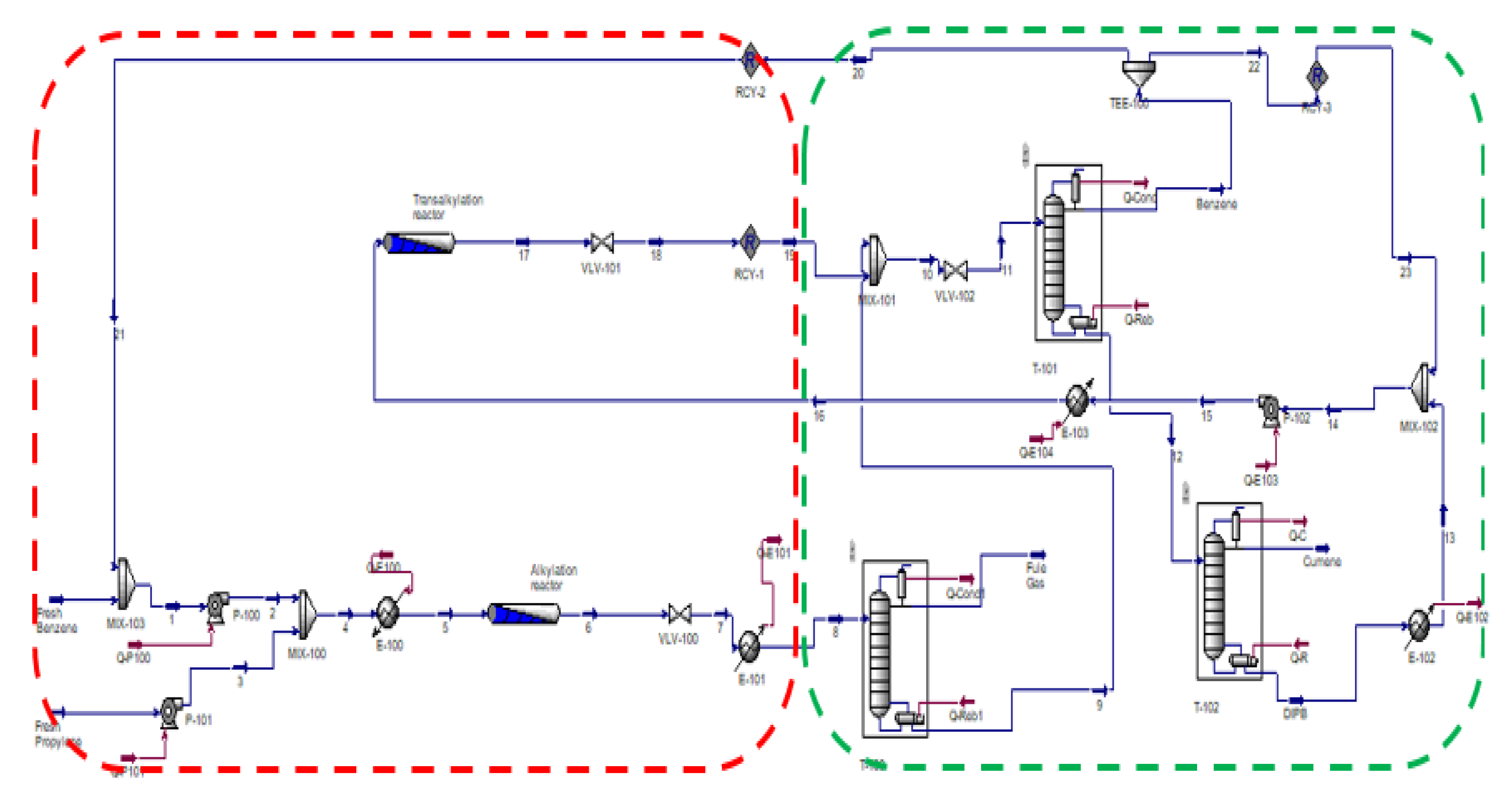
3. Methodology
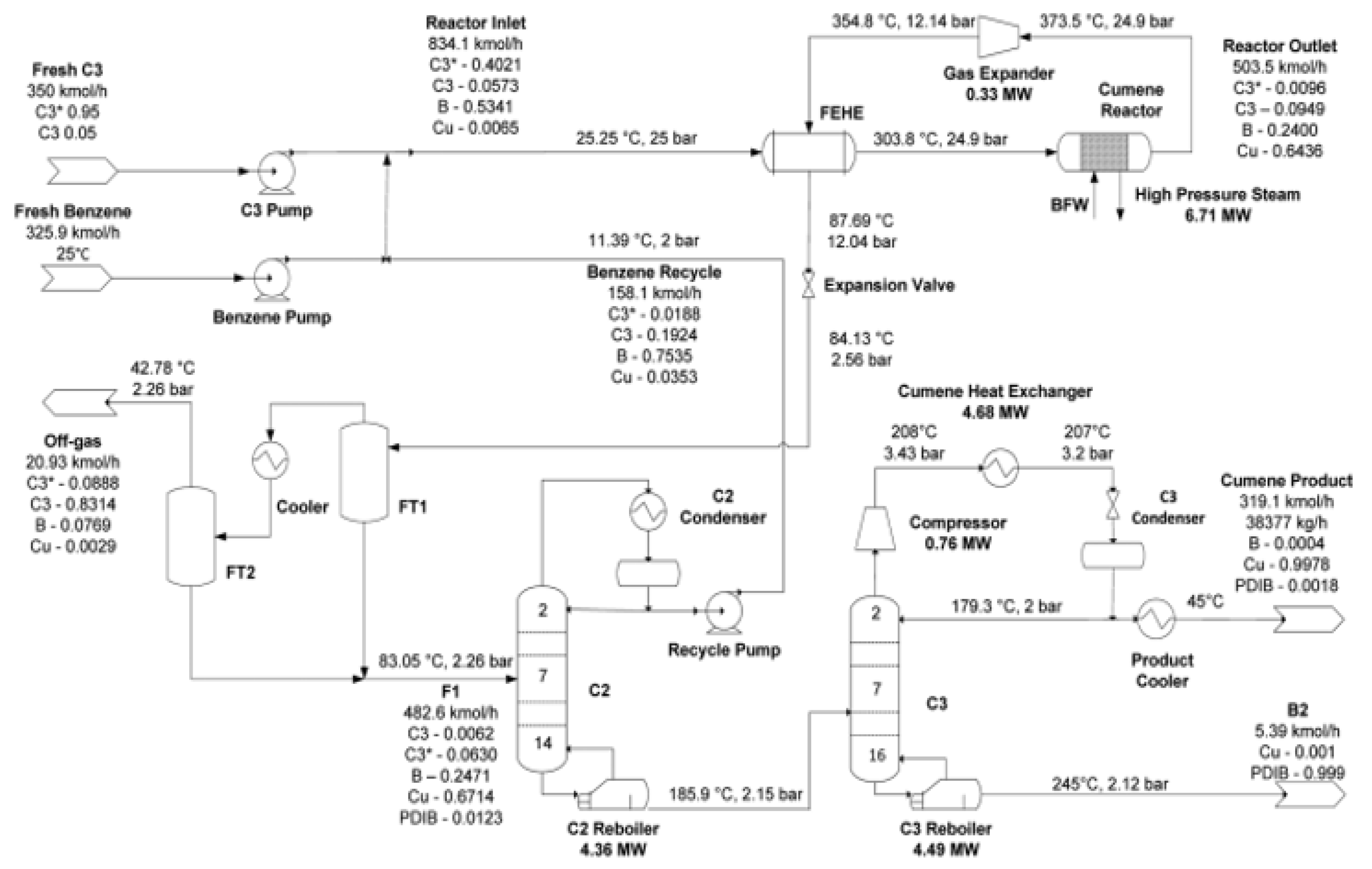
4. Different Simulation Approaches for Cumene Production
5. Results and Discussions
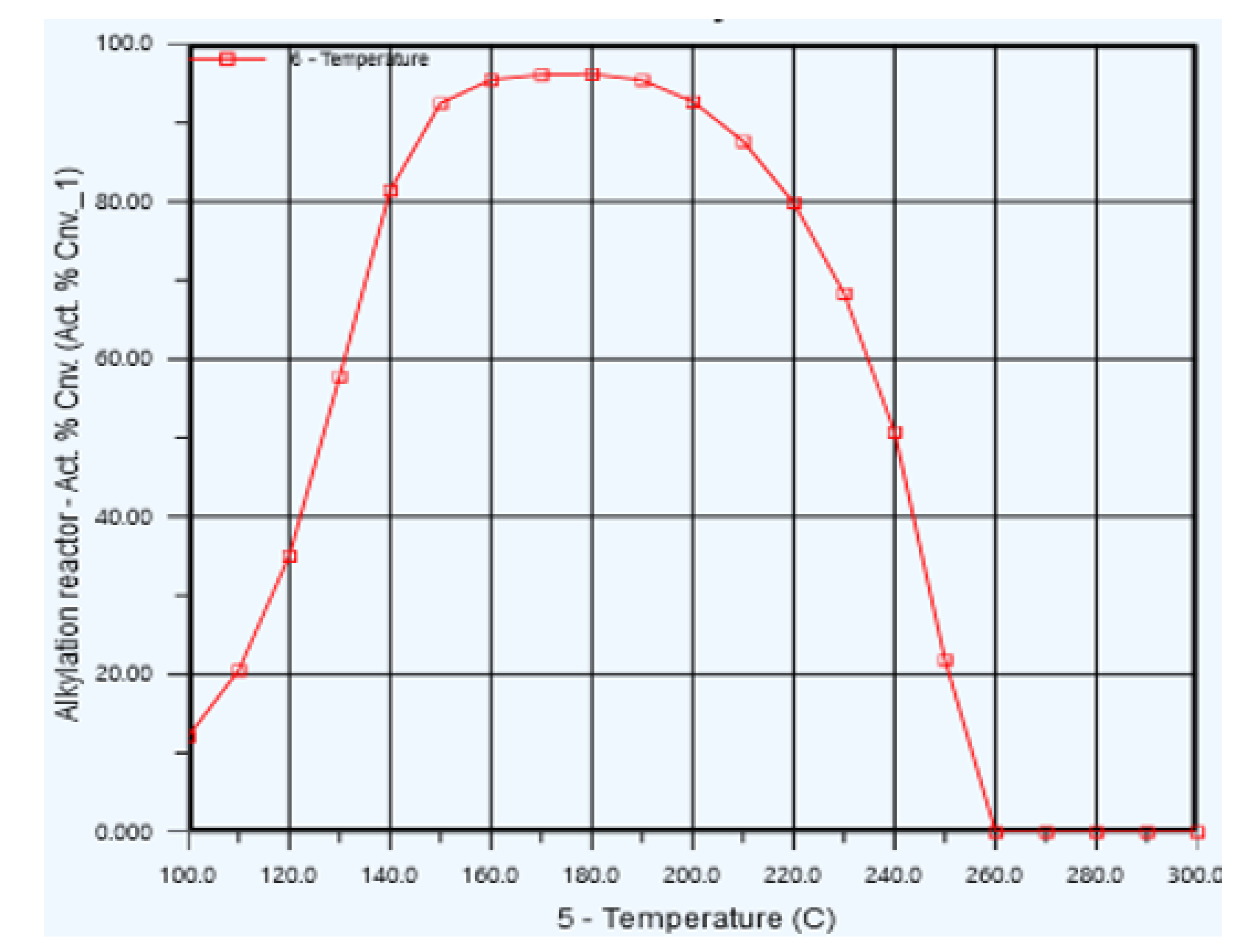
5.1. Reactor Temperature Optimization
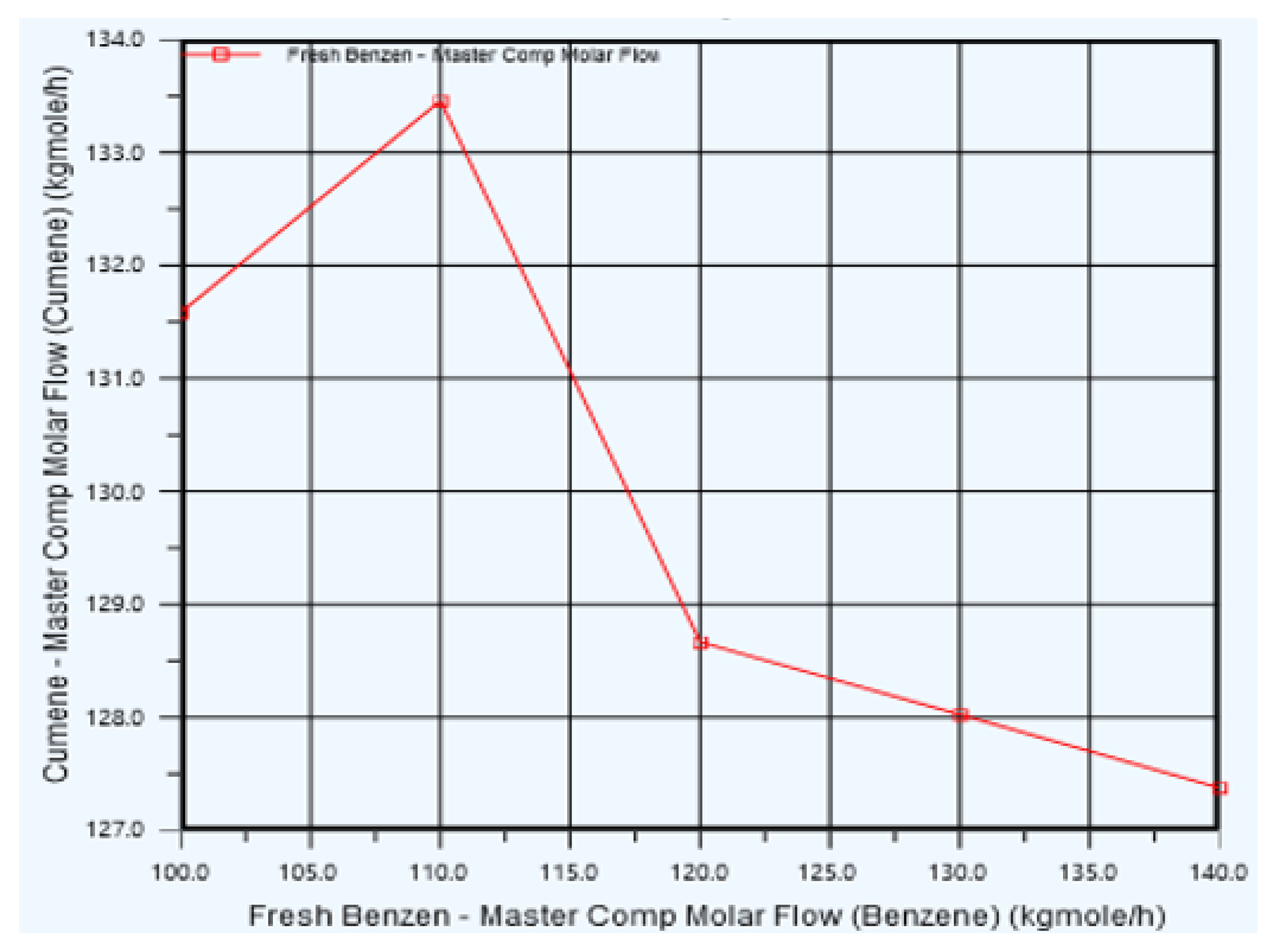
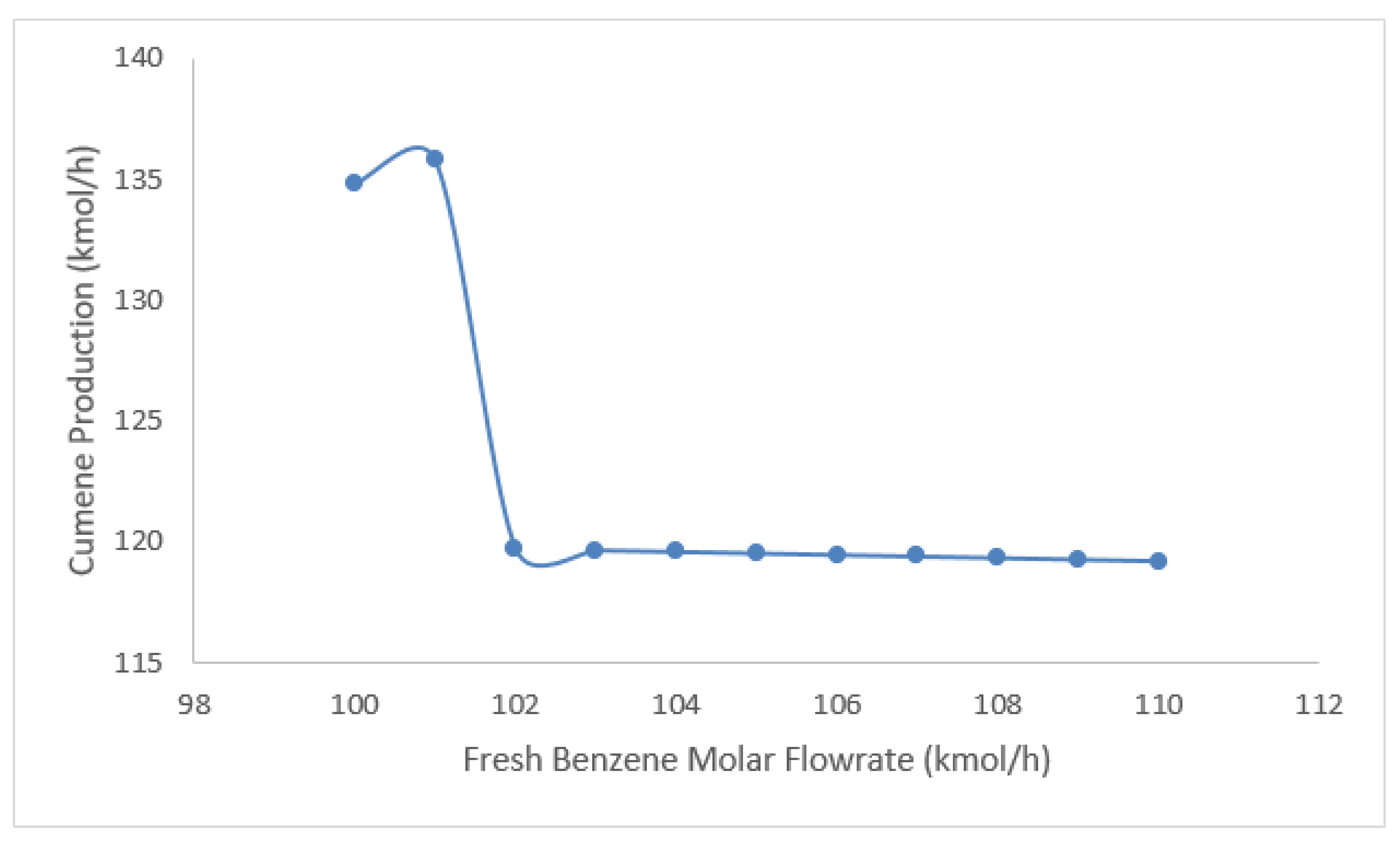
5.2. Fresh Benzene feed Optimization
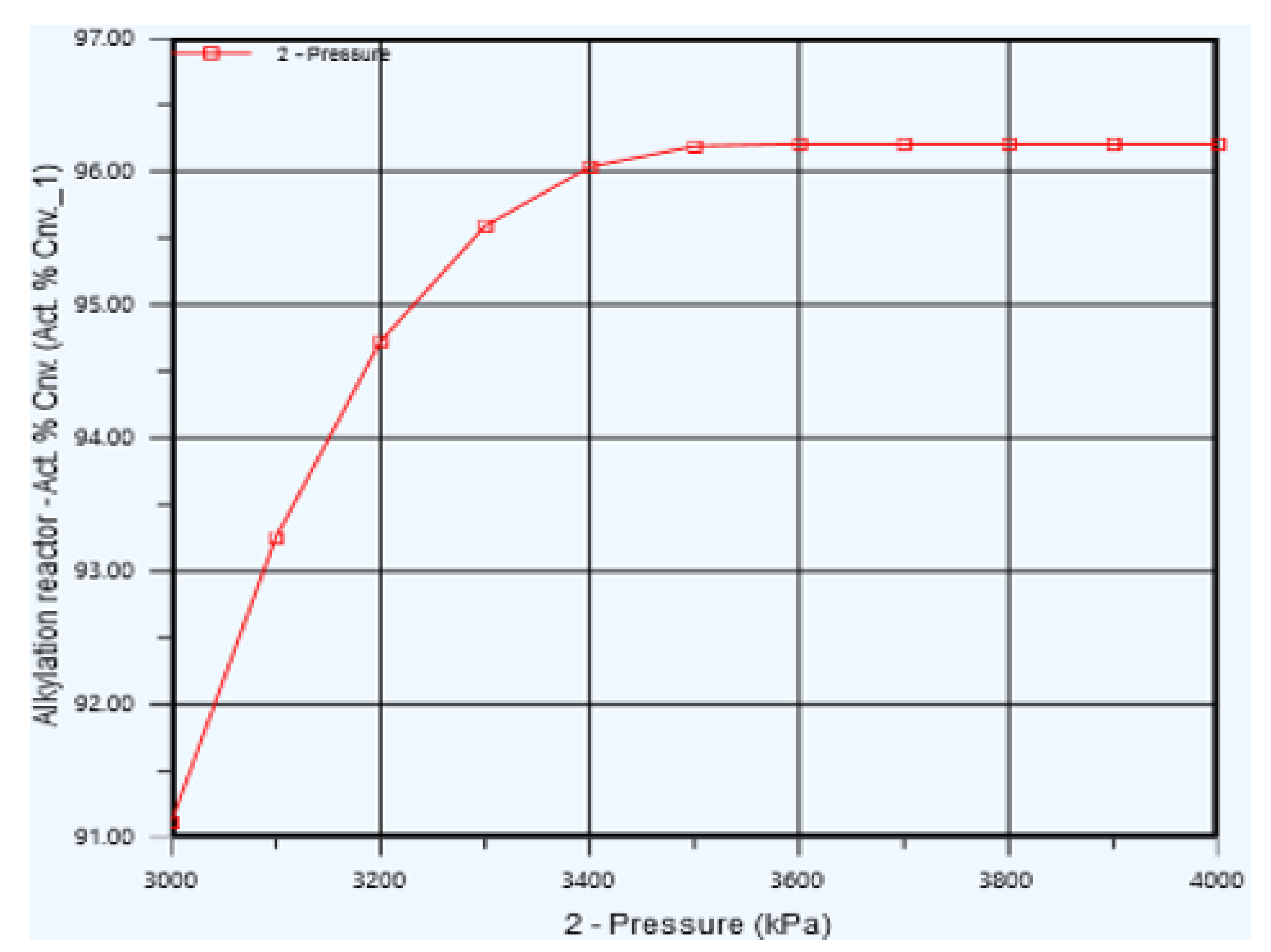
5.3. Reactor Pressure Optimization
6. Conclusion
- Achieving a maximum propylene conversion of 96.24% at an optimized temperature of 178°C, pressure of 3600 kPa, and fresh benzene flowrate of 101 kmol/h.
- Reducing the reaction temperature from 180°C to 178°C while maintaining high conversion, leading to energy savings and improved efficiency.
- Increasing cumene production from 127.7 kmol/h (base case) to 135.8 kmol/h after optimization, reflecting enhanced productivity.
- Lowering raw material consumption and waste generation, contributing to better sustainability and reduced operational cost.
- Demonstrating the capability of Aspen HYSYS to predict process behavior and optimize key parameters prior to industrial implementation.
Author Contributions
Funding
Data Availability Statement
References
- Ullmann, F.; Gerhartz, W. Ullmann’s Encyclopedia of Industrial Chemistry; VCH: Weineim, 1986. [Google Scholar]
- Lei, Z.; Dai, C.; Wang, Y.; Chen, B. Process optimization on alkylation of benzene with pro-pylene. Energy & Fuels 2009, 23(6), 3159–3166. [Google Scholar]
- Sengupta, S. L.; Pal, S.; Lahiri, S. K. Design and simulation of cumene plant. International Journal of Chemical Engineering and Applications 2011, 2(3), 192–196. [Google Scholar]
- Flegiel, F.; Sharma, S.; Rangaiah, G. P. Development and Multiobjective Optimization of Improved Cumene Production Processes. Materials and Manufacturing Processes 2015, 30(4), 444–457. [Google Scholar] [CrossRef]
- Mahmoudian, F.; Moghaddam, A. H.; Davachi, S. M. Genetic-based multi-objective optimization of alkylation process by a hybrid model of statistical and artificial intelligence approaches. Can. J. Chem. Eng. 2022, 100(1), 90–102. [Google Scholar] [CrossRef]
- Ahmad, F.; Ahmad, N.; Al-Khazaal, A. A. Z. Machine Learning-assisted Prediction and Optimization of Exergy Efficiency and Destruction of Cumene Plant under Uncertainty. Eng. Technol. Appl. Sci. Res. 2024, 14(1), 12892–12899. [Google Scholar] [CrossRef]
- Luyben, W. L. Principles and Case Studies of Simultaneous Design, 1st ed.; Wiley, 2011. [Google Scholar] [CrossRef]
- Ben Hamida, M.B.; Ali, A.B.M.; Sawaran Singh, N.S.; Mostafa, L. Optimization of MXene-Based Aqueous Ionic Liquids for Solar Systems Using Conventional and AI-Based Techniques. Sci Rep 2025, 15, 20565. [Google Scholar] [CrossRef] [PubMed]
- Ayadi, B.; Adnan; Khan, S.U.; Bani-Fwaz, M.Z.; Khidhir, D.M.; Ali, M.M.; Bashir, M.N.; Ben Hamida, M.B. Machine Learning Investigation of Wavy Fin Using Penta Type Nanomaterial with Multiple Parameters: Computational Design. Case Studies in Thermal Engineering 2025, 74, 106816. [Google Scholar] [CrossRef]
- Ben Hamida, M.B.; Basem, A.; Rajab, H.; Othayq, M.M. Dynamic Optimization of a Solar–sCO2–CAES Hybrid Energy System: Integration of Green Hydrogen Production and AI-Based Energy Management. International Journal of Hydrogen Energy 2025, 146, 149888. [Google Scholar] [CrossRef]
- Ayub, A.; Zulkefal, M.; Sethi, H. Artificial-Intelligence-Assisted Investigation of Quality and Yield of Cumene Production. In Proceedings of the CEMP 2023; MDPI, April 10 2024; p. 10. [Google Scholar]
- Amooey, A.A.; Mousapour, M.; Nabavi, S.R. Multiobjective Particles Swarm Optimization and Multicriteria Decision Making of Improved Cumene Production Process Including Economic, Environmental, and Safety Criteria. Can J Chem Eng 2024, 102, 1203–1224. [Google Scholar] [CrossRef]
- Al-Ghamdi, B.; Al-Ghazal, M.; Al-Abdulkareem, Q.; Al-Sharif, A.; Al-Saad, I. Production of Cumene via Alkylation of Benzene and Propylene By Honeywell UOP QMax Technology. ResearchGate, Dec. 2019. [Google Scholar]
- Hazmi, H.A. Process Simulation & Sensitvity Analysis of Cumene Production from an Integrated Alkylation and Transalkylation Reaction. JCPE 2022, 7, 64–78. [Google Scholar] [CrossRef]
- Zhai, J.; Liu, Y.; Li, L.; Zhu, Y.; Zhong, W.; Sun, L. Applications of dividing wall column technology to industrial-scale cumene production. Chem. Eng. Res. Des. 2015, 102, 138–149. [Google Scholar] [CrossRef]
- Norouzi, H. R.; Fatemi, S. Economic optimization of the cumene production process using response surface methodology. Chem. Eng. Commun. 2012, 199(11), 1375–1393. [Google Scholar] [CrossRef]
- Pathak, A. S.; Agarwal, S.; Gera, V.; Kaistha, N. Design and control of a vapor-phase conventional process and reactive distillation process for cumene production. Ind. Eng. Chem. Res. 2011, 50(6), 3312–3326. [Google Scholar] [CrossRef]
| Reactor | Reaction | Rate expression |
Rate constants |
| Alkylation | Cumene reaction | ||
| DIPB reaction |
|||
| Transalkylation | Forward | ||
| Backward |
| Cases | Temperature (°C) | Propylene conversion |
| Case 1 | 170 | 95.71 |
| Case 2 | 171 | 94.28 |
| Case 3 | 172 | 83.85 |
| Case 4 | 173 | 93.14 |
| Case 5 | 174 | 92.77 |
| Case 6 | 175 | 87.01 |
| Case 7 | 176 | 70.11 |
| Case 8 | 177 | 68.03 |
| Case 9 | 178 | 96.20 |
| Case 10 | 179 | 96.20 |
| Case 11 | 180 | 96.20 |
| Cases | Fresh benzene Flow (Kgmole/h) | Cumene Flow (Kgmole/h) |
| Case 1 | 100 | 134.844 |
| Case 2 | 101 | 135.792 |
| Case 3 | 102 | 119.725 |
| Case 4 | 103 | 119.665 |
| Case 5 | 104 | 119.605 |
| Case 6 | 105 | 119.542 |
| Case 7 | 106 | 119.477 |
| Case 8 | 107 | 119.413 |
| Case 9 | 108 | 119.349 |
| Case 10 | 109 | 119.284 |
| Case 11 | 110 | 119.219 |
| Parameter | Before Optimization | After optimization |
| Alkylation Reactor Temperature () | 170 | 178 |
| Propylene Conversion (%) | 96.13 | 96.20 |
| Fresh Benzene Feed (kmol/h) | 127.8 | 101 |
| Cumene Production (kmol/h) | 127.7 | 135.792 |
| Pressure (kpa) | 3540 | 3600 |
| Propylene Conversion (%) | 96.20 | 96.24 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




