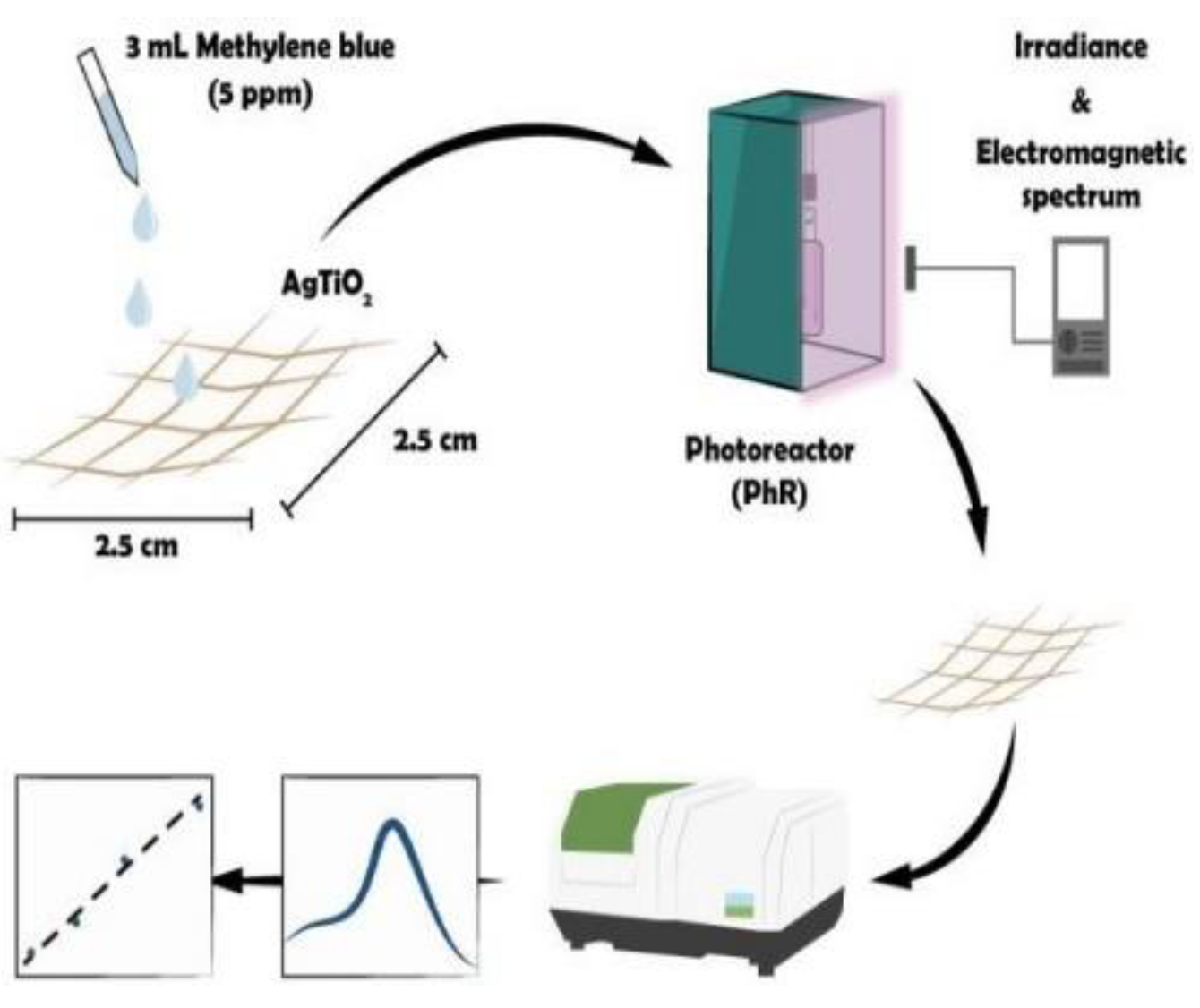
This section presents the characterization results, the electro-optical properties of the AgTiC and the emitted wavelength and irradiance of the radiation sources of the different PhRs used. Kinetic photocatalytic model was proposed considering the reactant equilibrium coverage (Langmuir isotherm), the pseudo photocatalytic steady state and the transfer processes of the photoinduced charge carrier species. Finally, the photocatalytic activity results for the degradation of MB are discussed and linked to the parameters above mentioned.
3.2. UV-vis Diffuse Reflectance Spectroscopy (UV-vis-DRS), Photoluminescence Spectroscopy (PL) and UV-vis Spectroscopy (UV-vis) of AgTiC
The bandgap energy (E
g) of the AgTiC was determined using the Kubelka-Munk function and the Tauc plot, assuming an indirect electronic transition from the valence band to the conduction band. The Eg value determined was 3.55 eV, therefore the minimum required wavelength for the photoactivation of AgTiC is ~ 360 nm. Whereas E
g values of reference materials were 3.2 eV and 3.0 eV for TiO
2-anatase and Degussa P25, respectively. The increase of the Eg value for AgTiC is attributed to the smaller size of TiO
2 NPs (9.6 nm) into the nanocomposite compared to the size of the TiO
2 particles in bulk TiO
2-anatase (> 50 nm) or Degussa P25 (~ 25 nm) [
27]. It is important to note that although the E
g value indicates that AgTiC should be photoactivated by high-energy photons, from the UV spectrum, the presence of the nanoplasmomic Ag NPs could promote the absorption of lower-energy photons from vis radiation (> 400 nm) due to the localized surface plasmon resonance phenomenon (LSRP). According to [
28], the Ag NPs promote the electron flow from the nanoplasmonic NPs towards the conduction band of the TiO
2. Which should enhance photocatalytic activity due to charge carrier species remaining on the surface of the photocatalyst, in a Z-type scheme as has been reported for several semiconductors in photocatalytic processes.
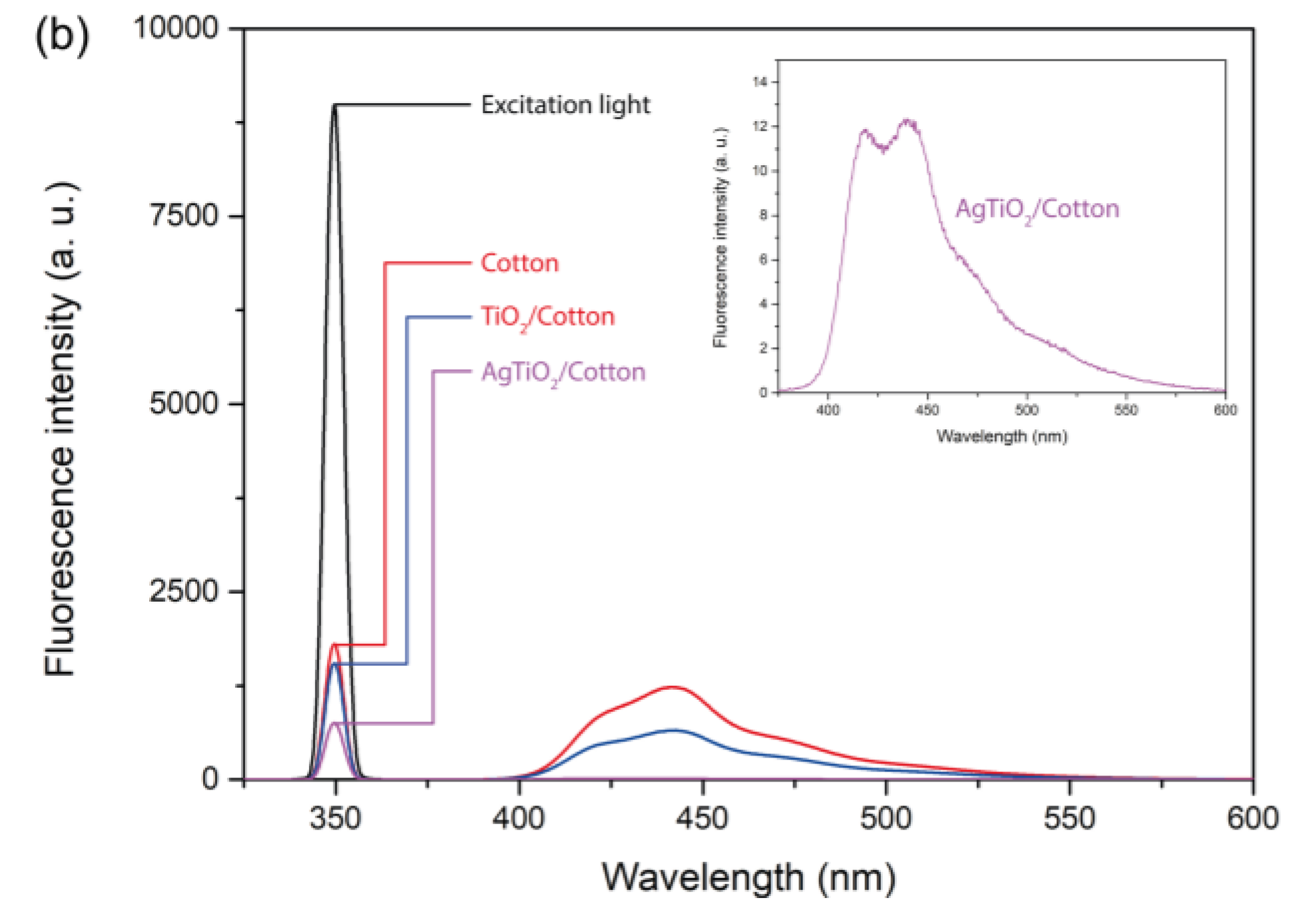
The fluorescence 3D excitation-emission matrix for AgTiC indicated that at excitation wavelength (λ
ex) of 350 nm, the intensity of the emitted fluorescence (λ
em) at 442 nm was maximal [
25]. The emitted band at 442 nm is attributed to the excitonic emission from TiO
2 due to vacancies on the surface [
29]. Therefore, it is to be assumed that the optimal wavelength for the photoactivation of the AgTiC nanocomposite should be at 350 nm, which corresponds to high-energy photons from UV spectrum, as has also been suggested by the determined E
g value.
Since fluorescence radiation is due to the fall of electrons from the conduction band to the valence band, the intensity of the emitted fluorescence band at 442 nm can be used to determinate the [e
-/h
+] recombination rate. Therefore, a higher intensity of the band at 442 nm indicates greater electron recombination, and conversely, a lower band intensity indicates a slower recombination rate. It can be observed in
Figure 2 the fluorescence band of AgTiC at 442 nm. For comparison purposes fluorescence band at 442 nm of reference compounds such as bare cotton and TiO
2/cotton are also shown. The intensity of the band at 442 nm of AgTiC was lower than reference compounds, which suggests that recombination rate of [e
-/h
+] of AgTiC was very low.
Based on the results, AgTiC promoted better separation of the charge carrier species [e-/h+], boosting the charge carrier migration towards the material surface for the interfacial electron transfer processes to the generation of the reactive oxygen species (ROS). Therefore, it is presumed that simultaneous incorporation of NPs (Ag and TiO2) on the cotton fabrics promotes the greater predominance of charge carrier species on the surface of the nanocomposite.
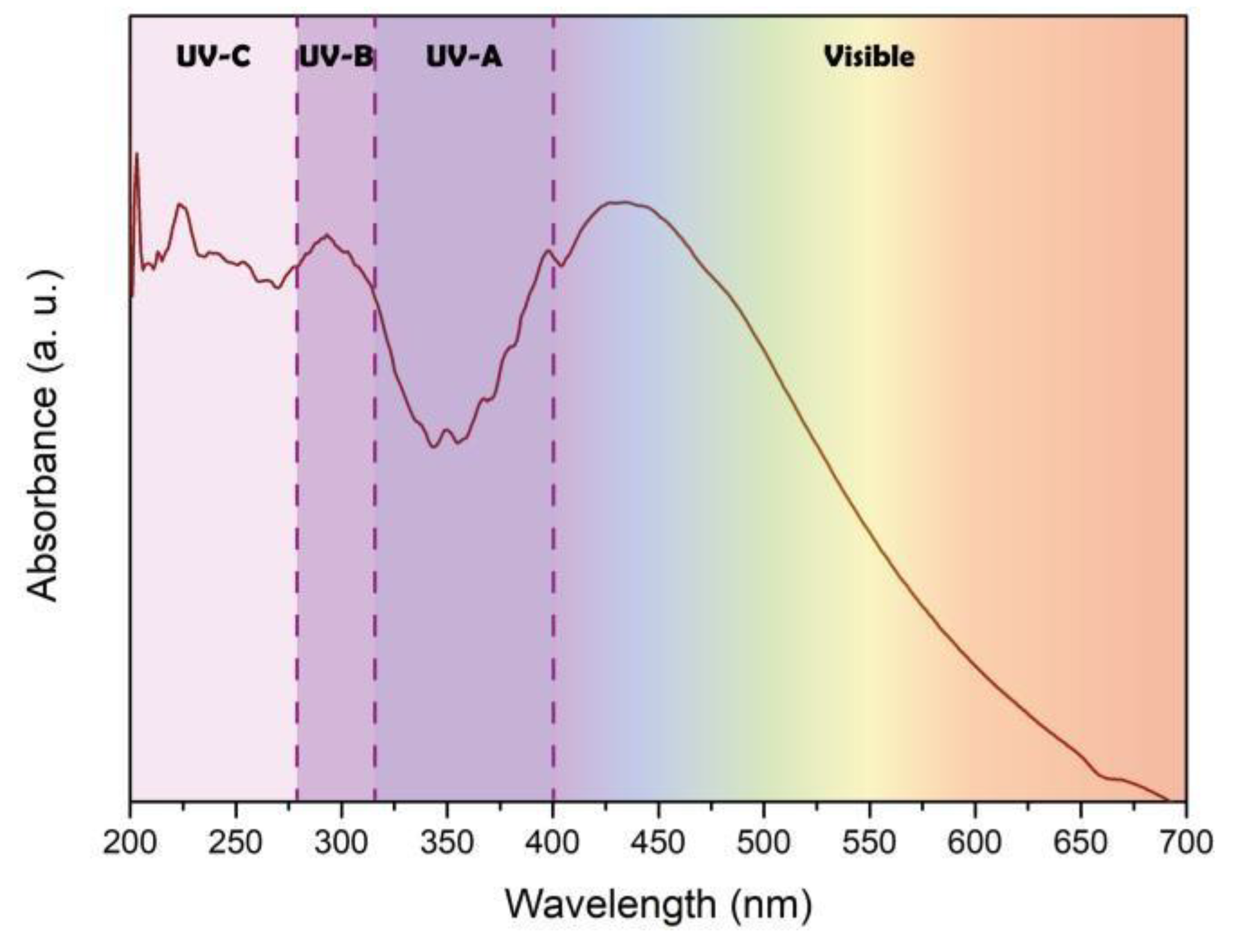
Figure 3 shows the UV-vis spectrum of AgTiC, which presents absorption of photons from 200 to 650 nm. The absorption at 442 nm corresponds to the wavelength of the maximal phosphorescence emission (see
Figure 2). The absorption at wavelengths corresponding to visible spectrum is attributed to the incorporation of Ag NPs, which extended the absorption of lower-energy photons. Therefore, AgTiC can be used as visible light-activated photocatalyst. Four intervals can be observed in
Figure 3 to identify the type of irradiation required to the activation of AgTiC: UVC (200-280 nm), UVB (280-320 nm), UVA (320-400 nm) and visible (> 400 nm).
3.3. Determination of the Photoluminescence Quantum Yield (ϕpl) of AgTiC
It is important to note that generated charge carrier species could get three possible phenomena: 1) radiative recombination, 2) non-radiative recombination or 3) separation and charge species transfer. Radiative recombination phenomena are due to the relaxation processes of the [e-/h+], where [e-] from the conduction band fall to the valence band and occupy the [h+] sites. Non-radiative recombination phenomena refer to the [e-] in the conduction band that fall from a higher-energy electronic state to a lower-energy electronic state within the conduction band, the energy is lost as heat. Separation and charge species transfer refers to [e-/h+] species that are maintained on the surface of the semiconductor material and therefore, are involved in photocatalytic processes.
In this sense, the efficiency of the photocatalytic processes can be estimated by the photocatalytic quantum yield (Φ
pc), which can be interpreted as a parameter that indicates the number of charge carrier species directly involved in the photocatalytic process with respect to the total number of irradiated photons. Complementarily, the Φ
pc can be estimated by means of the photoluminescence quantum yield (ϕ
pl), which can be interpreted as the counterpart parameter. Photoluminescence quantum yield (ϕ
pl) is a parameter of semiconductor materials that quantify the capacity of the material to emit photons after this one is irradiated with photons that exceed the corresponding wavelength to its E
g. ϕ
pl is defined as the ratio of emitted photons respect to the absorbed photons [
30,
31]. The Φ
pc can be estimated by the consideration of the ϕ
pl and the non-radiative recombination processes [
6]. Therefore, the intensity of the emitted fluorescence band at 350 nm (see
Figure 2) can be used to estimate the ϕ
pl. Which can be determined by the comparison of the emitted fluorescence intensities at 350 nm of the AgTiC and the excitation light, where the last one corresponds to the total reflected light as phosphorescence (as reference).
A high value of ϕpl indicates that significant amount of photogenerated [e-/h+] recombine through radiative processes, releasing energy as light by fluorescence or phosphorescence phenomena. Conversely, low ϕpl could be interpreted as charge carrier species probably involved in photocatalytic processes. Therefore, desirable photocatalytic materials must have lower ϕpl values. Which suggests that radiative recombination phenomena are minimized and most likely separation and transport of charge carrier species to the photocatalyst surface happens. The ϕpl determined for AgTiC was 0.0175 meanwhile for TiO2/cotton was 0.21. Then, AgTiC absorbs more photons than TiO2/cotton, decreasing radiative recombination processes. Which suggests that photocatalytic activity should be better due to the greater amount of photogenerated charge carrier species on the surface on the photocatalytic material.
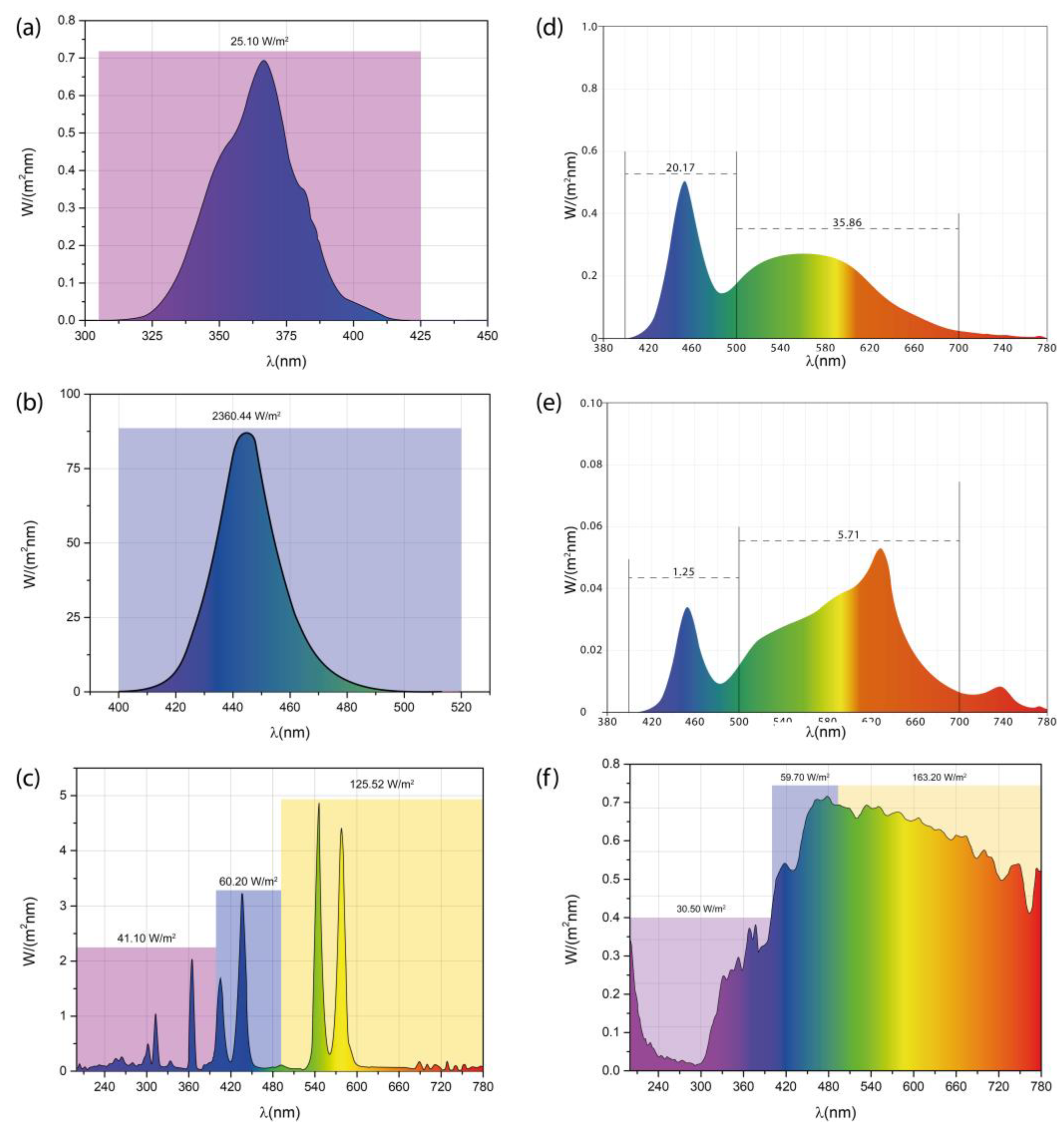
3.4. Irradiance and Emission Spectra of the Photoreactors (PhRs)
Figure 4 shows the irradiance (W/m
2) as a function of the radiation wavelength for the tested PhRs.
Figure 4a shows the irradiance for the PhR-T5 and it can be observed emission of UV-A radiation and low contribution of visible radiation (400-420 nm). The total irradiance recorded from 306-425 nm was 25.10 W/m
2. The visible radiation (corresponding to violet color) represents less than 10% of the total emission spectrum, therefore PhR-T5 was classified as a source of UV-A radiation. Then, shorter wavelengths (more energetic) of the emitted UV-A radiation from PhR-T5 surpasses the wavelength required by the E
g of TiO
2 but larger wavelengths could not activate the LSPR of Ag NPs, although it is important to note that irradiance value determined was quite low. PhR-B (
Figure 4b) shows emissions in 400-500 nm, and the maximal emission was at ~450 nm. The total irradiance was 2360.44 W/m
2. Despite the greater irradiance value, the minimum wavelength required for the photoactivation of AgTiC is not achieved with PhR-B. However, the irradiated photons in this interval could activate the LSPR of Ag NPs.
The PhR-Hg (
Figure 4c) emitted radiation from UV (200-400 nm) and visible (> 400 nm) spectrum, the photons emitted from the visible spectrum correspond to a significant part of the blue spectrum. The total irradiance corresponding to the UV radiation (200-400 nm) was 41.10 W/m
2 meanwhile the visible radiation at 400-500 nm generates a total irradiance of 60.20 W/m
2. The total irradiance at wavelengths greater than 500 nm was 125.52 W/m
2. According to previous sections, the photons with wavelengths below 360 nm could photoactivate the AgTiC. Although it is likely that photons with greater wavelengths than 360 nm could photoactivate the LSPR of Ag NPs, which should trigger the photocatalytic activity of the AgTiC.
The spectrum of PhR-I (see
Figure 4d) shown emission from 420 to 780 nm, corresponding to visible spectrum. The total irradiance in 400-500 nm was 25.17 W/m
2. Meanwhile, the irradiance increased to 35.86 W/m
2 at wavelengths from 500 to 700 nm. Low irradiances were obtained by PhR-G (see
Figure 4e). Since, total irradiance of 1.25 W/m
2 at 420-500 nm was recorded, although a slight increase of the total irradiance (5.71 W/m
2) was observed at 500-700 nm. The emission spectra of PhR-I and PhR-G (
Figure 4d and
Figure 4e) confirm that these radiation sources do not emit UV-radiation and provide only visible radiation. For these last irradiation sources, it is likely that the photons emitted between 420 and 500 nm could provide the necessary energy to activate the LSPR of Ag NPs in AgTiC. For comparison purposes, the irradiance for the solar radiation (winter in Mexico City) is shown in
Figure 4f. The irradiances determined were significantly greater than other radiation sources used in this study. The total irradiance was 30.50 W/m
2 in 200-400 nm, and a higher value was recorded (59.70 W/m
2) in 400 to 500 nm. However, a significantly higher total irradiance of 163.20 W/m
2 was observed at wavelengths greater than 500 nm. It is important to note that photoactivation of AgTiC (or LSPR) cannot be achieved if the specific wavelength value that establishes the E
g is not fulfilled, despite high irradiance values.
All PhRs tested emitted UV and visible radiation, however the irradiances recorded were different. The UV irradiance trend among the tested PhRs was solar radiation > PhR-Hg > PhR-T5 > PhR-I > PhR-G. Meanwhile, the visible irradiance trend of the PhRs was PhR-B > solar radiation > PhR-Hg > PhR-I > PhR-G > PhR-T5. The UV irradiance of PhR-Hg was 2-fold, 33-fold and 1.6-fold greater than UV irradiance for PhR-I, PhR-G and PhR-T5, respectively. In similar sense, visible irradiance of PhR-Hg was 3.5-fold and 22-fold greater than visible irradiance for PhR-I and PhR-G, respectively. It is important to note that visible irradiation was not significant using PhR-T5. Also, the visible irradiance for solar radiation was higher than others radiation sources.
Based on the above results, the selected PhRs for the photocatalytic degradation of MB were those with high irradiance, mainly in the UV spectrum. Thus, the PhR-Hg and PhR-T5 were used as radiation sources for the assessment of photocatalytic activity using AgTiC. In
Table 2 the irradiances are reported as total values for the corresponding UV and visible intervals as established in
Figure 4. The irradiance of the PhR-Hg was adjusted by switching the supplied voltage (75 or 90 V), while the PhR-T5 was placed at different heights with respect to the reaction system. The recorded irradiance from the PhRs is shown for UV and visible irradiation at the corresponding photoreactor setups. Irradiance of PhR-Hg was significatively greater than those for the PhR-T5, for both types of radiation.
3.5. Kinetic Photocatalytic Model (KPM)
A model that encompasses all aspects involved in the overall photocatalytic process is required for the kinetic description of the photocatalytic reactions. According to [
32,
33,
34] the photocatalytic reaction rate must consider two fundamental aspects. The first one involves considering the heterogeneous reaction on the photocatalyst surface. The second one involves considering the optical and electronic processes that take place throughout the irradiation process. The photocatalytic reaction rate could be defined according to the photoinduced charge carrier transfer processes [
35,
36] as:
Where θA corresponds to the occupied surface fraction of the adsorbed reactant A, Φpc is the photocatalytic quantum efficiency and I is the irradiance. It is important to note that θA parameter involves the reactant equilibrium coverage in the same sense as Langmuir isotherm. However, for photocatalytic reactions the θA parameter is not the same as Langmuir stated.
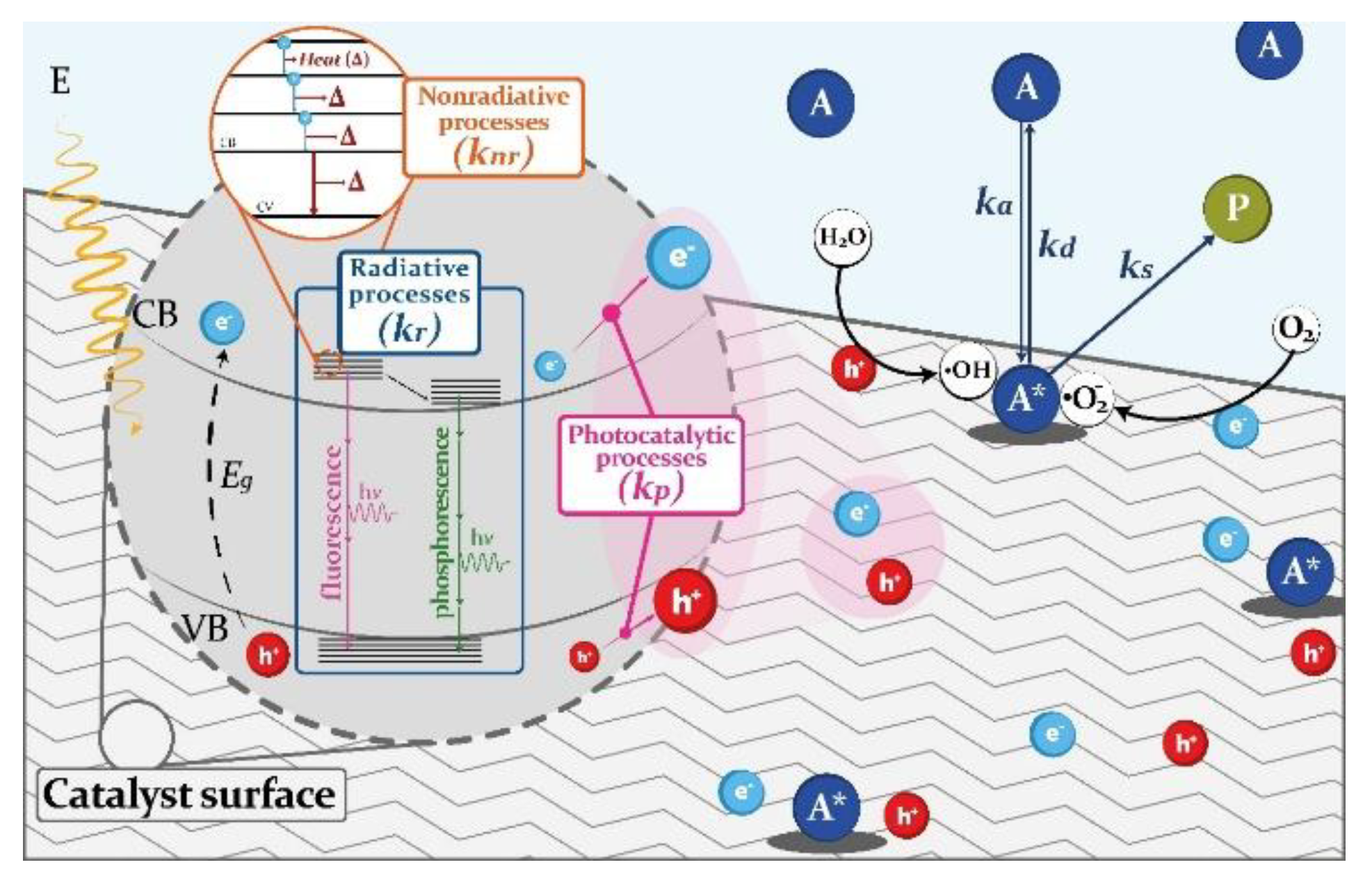
Diagram 2 shows the schematic representation of the photocatalytic process, also showing the recombination pathways of the charge carrier species.
Diagram 2.
Schematic representation of the kinetic photocatalytic model.
Diagram 2.
Schematic representation of the kinetic photocatalytic model.
According to Diagram 2, reactant A must be absorbed on the photocatalytic surface, according to step 1:
Where * is the adsorption site and A* corresponds to the adsorbed reactant A. According to [
33] photocatalytic reactions involve the generation of reactive intermediates such as free electrons [e
-], holes [h
+] and hydroxyl radicals [•OH
-]. The adsorption-desorption equilibrium of reactant A could not be reached due to the reactivity of the A* with [•OH
-] adsorbed on the surface. Thus, the slow step (single reactant adsorption) stated in the Langmuir-Hinshelwood model (LHM) is not fulfilled in photocatalysis, and a pseudo photocatalytic steady state approach must be utilized in the kinetic equation for reactant and surface radicals. This model assumes that reaction of adsorbed reactant with surface [•OH
-] occurs, according to step 2:
Where k
s refers to the surface reaction constant between adsorbed reactant A (A*) and the surface [•OH
-].
Therefore, the surface reaction rate must be as follows:
In consequence, for a pseudo photocatalytic steady state reactant coverage is:
Where, K0 corresponds to the adsorption equilibrium constant of reactant A, which is a function on the irradiance intensity and the absorbed concentration of [•OH -]. This pseudo photocatalytic steady state model is mathematically analogous to the LHM, but with adsorption constants conceptually different.
It can be observed in Diagram 2 a zoomed-in view of a volume-element of photocatalytic surface, after the irradiation process. The optical and electronic processes, due to the irradiation process on the photocatalytic material, can be described as charge transfer processes [
34]. The diagram shows the classification of the charge carrier transfer processes as radiative, non-radiative and photocatalytic processes. To clarify, it is important to note that in an irradiation process, the emitted photons with a wavelength greater than or equal to that the required by the E
g of the photocatalyst promote the generation of the pair [e
-/h
+]. Radiative processes refer to the recombination of the charge carrier species, where the [e
-] into the conduction band (CB) fall to the valence band (VB) and therefore, the [h
+] are occupied. The radiative processes are characterized by the radiative optical constant (k
r). In radiative processes the phenomena of fluorescence and phosphorescence could happen. Fluorescence implies that [e
-] falls from the singlet state in the CB to the singlet state in the VB, meanwhile in the phosphorescence the [e
-] falls from the triplet state in the CB to the singlet state in the VB.
In the non-radiative processes, the charge carrier species do not recombine, that is the [e-] in the CB do not fall to the VB. The [e-] held in the CB could transfer energy as heat in an energy decay processes within the CB. These non-radiative processes are characterized by the non-radiative optical constant (knr). If the [e-] does not transfer energy as heat, the charge carriers are kept on the surface of the photocatalytic material, and the generation of reactive oxygen species (ROS) occurs. Thus, [e-/h+] pairs that do not recombine are the charge carrier species involved in the photocatalytic degradation of the adsorbed reactants. These charge carrier transfer processes are characterized by the photocatalytic optical constant (kp). The intrinsic electro-optic processes can be quantified by the photocatalytic quantum efficiency (Φpc). This characteristic parameter of the semiconductor materials allows the evaluation of the effectiveness of a photocatalyst for the generation of charge carrier species [e-/h+] that are maintained on the surface of the material, through non-recombination processes. Therefore, Φpc indirectly assesses the effective generation of charge carrier species, which could very likely be involved in the improvement of the photocatalytic activity.
The intrinsic optical and electronic processes are triggered by the irradiance (I) intensity on the photocatalyst. That is, high irradiance values promote photocatalytic activity more than low irradiance values, as stated [
33]. Where the irradiance must be raised to an exponent that refers to the magnitude of the irradiance on the catalyst. The value of this exponent being less than one when irradiance is low. Demonstrating a significant dependence of the photocatalytic activity with the irradiance. In this sense, in the present work the effective irradiance (I
e) can be defined as the flux of photons with energy equal or higher than the E
g of the semiconductor material, which is necessary to generate the [e
-/h
+] pairs. That is, I
e must correspond to the minimum wavelength of the incident photons, which photoactivates the semiconductor material. Thus, in this work is proposed the kinetic photocatalytic model (KPM), which encompasses the surface reaction contributions (equations 1 to 6), the concurrent electronic and optical processes and the effective irradiance dependence, as follows:
3.6. Photocatalytic Activity Results for MB Degradation
Photocatalytic activities were determined using PhR-Hg, PhR-T5 and PhR-B since these photoreactor configurations reached an effective irradiance to activate the photocatalyst. It is important to note that photoactivation of the LSPR phenomenon can be achieved under these PhRs, contributing to the photocatalytic activity.
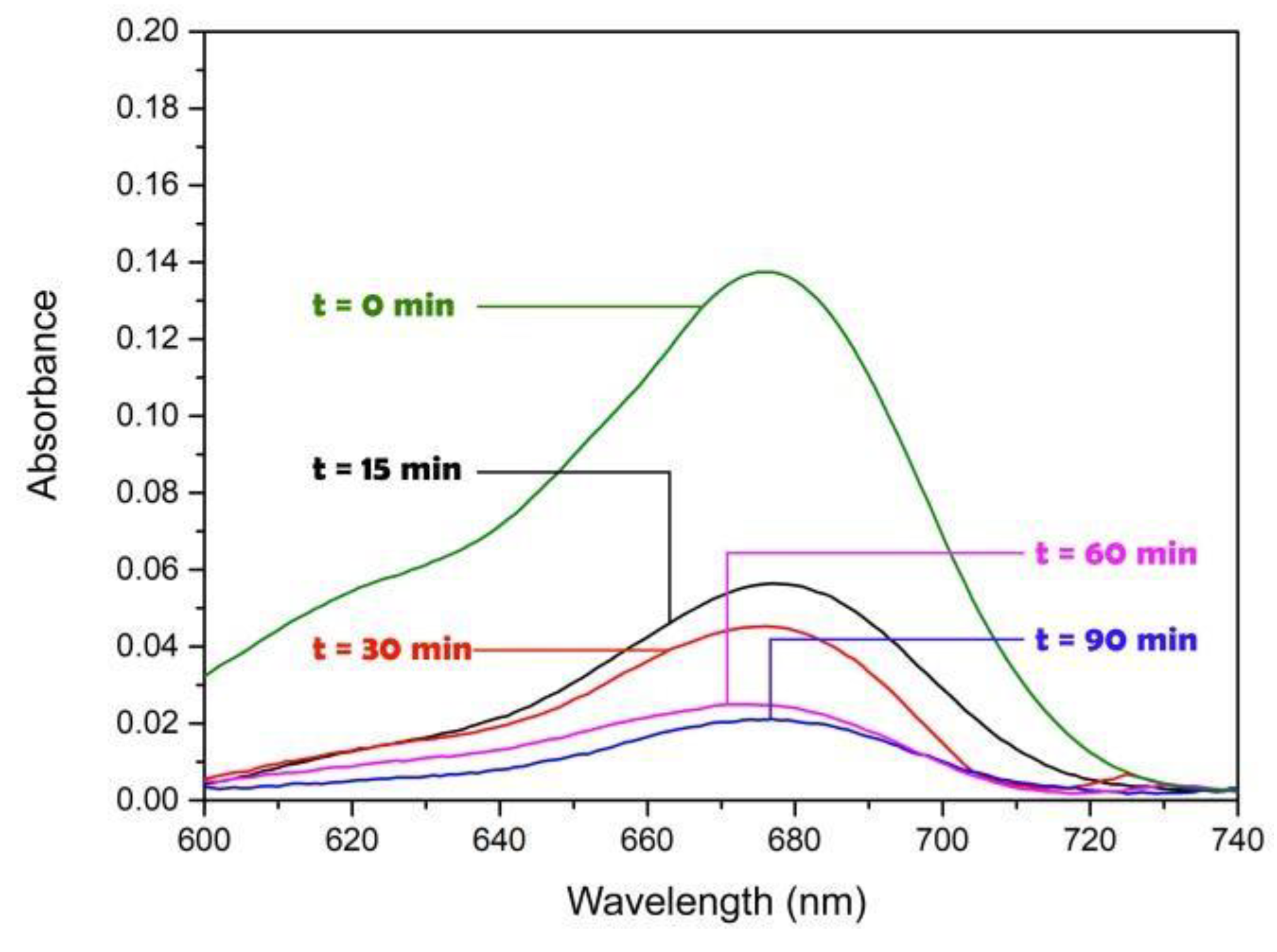
Figure 5 shows the UV-vis DRS spectra of MB as a function of the wavelength. It can be observed the photodegradation of MB after 90 min using the PhR-Hg at 75 V, where an irradiance of 41.10 W/m
2 was used.
It can be observed in
Figure 5 that under PhR-Hg at 75 V (41.10 W/m
2), the photoactivation of the AgTiC was achieved since the adsorption band of MB (λ
max = 672 nm) decreases with the reaction time. MB was degraded almost 88 % after 90 min under PhR-Hg. Similar results were obtained by PhR-T5 and solar radiation for MB degradation. With PhR-B the MB degradation reached was near to 33%. For comparison purposes, the MB degradation was carried out on bare cotton fabrics in PhRs tested. The degradation of MB by photolysis was less than 5% at 90 min, as has also been reported by [
37]. The photocatalytic efficiency under the different PhRs can be directly correlated with the irradiance profiles shown in
Figure 4. For PhR-T5 the emitted radiation was mainly UV-A, therefore the wavelength was enough for the photoactivation of AgTiC. Which allows the generation of the [e
-/h
+] pair from TiO
2, although the LSPR effect will not occur under this radiation source because its emitted wavelength was not enough for the LSPR photoactivation. According to
Figure 4 PhR-B cannot photoactivate AgTiC due to the low-energy emitted wavelength at 450 nm. In this sense, PhR-Hg and solar radiation can achieve photoactivation of AgTiC and LSPR phenomena, more than other PhRs used in this study.
3.7. Pseudo First Order Kinetic Photocatalytic Model (PFO-KPM)
Since the photocatalytic degradation of MB using AgTiC was carried out in solid phase, the adsorption-desorption equilibrium of reactant A is not carried out. Then, the KPM can be simplified as pseudo first order kinetic photocatalytic model (PFO-KPM) as follows:
Where k=K
0(Φ
pc)(I
eβ). If K
0C
A ≪ 1, then:
Thus k, the pseudo first order kinetic constant, can be determined as the scope of the of the following equation:
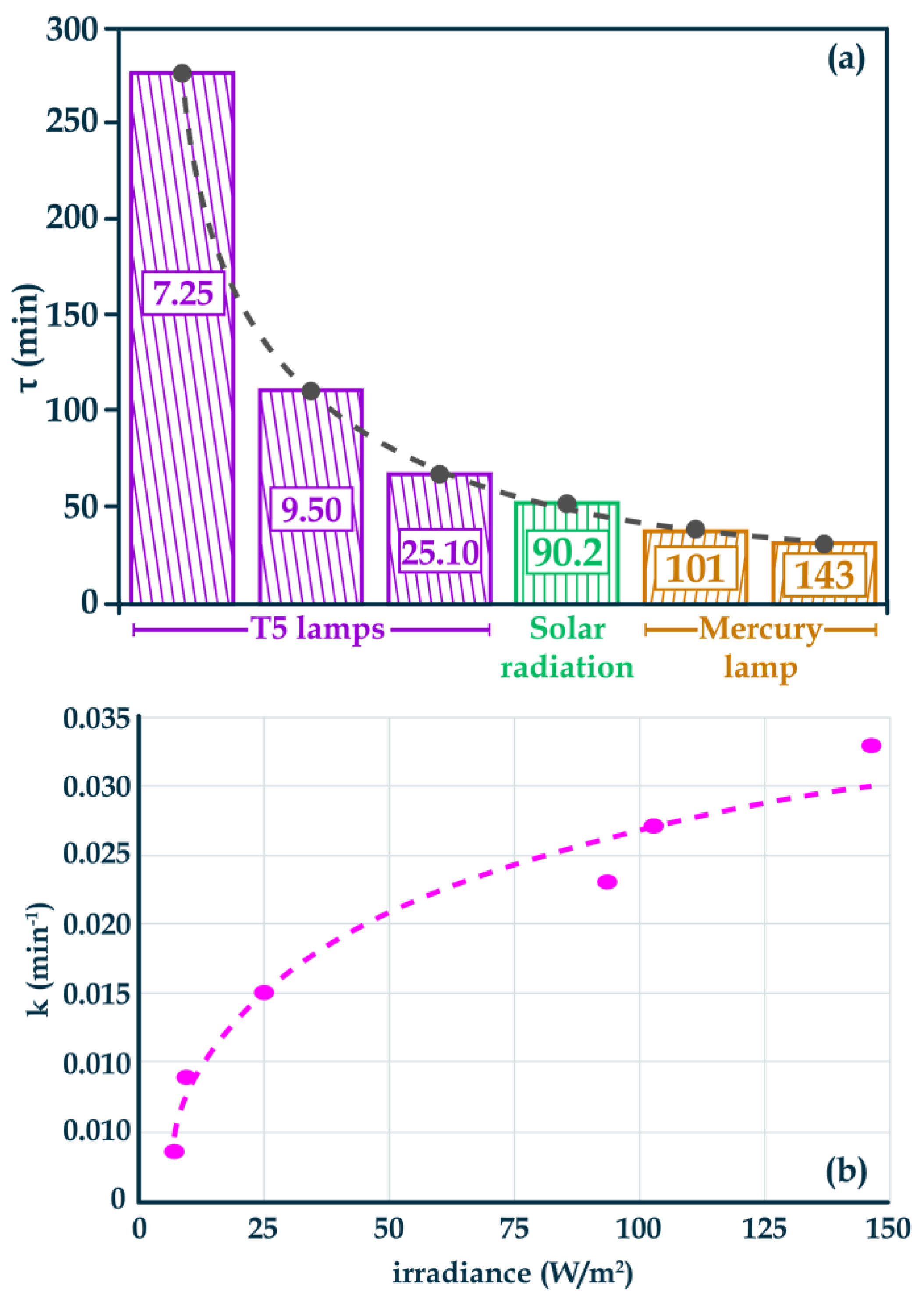
Figure 6 shows the photocatalytic activity of the AgTiC represented as pseudo-first order kinetic constant and the characteristic degradation time (τ), which is the reciprocal of k. Therefore, lower τ implies higher MB degradation and therefore, higher photocatalytic activity.
Figure 6a shows the τ0 as a function of the total irradiance for PhR-Hg and PhR-T5, solar radiation was included for comparison. The total irradiance used is shown inside the bars. Where, for these photocatalytic tests, total irradiance refers to the sum of the UV and visible irradiance reported in
Table 2, for each PhR configuration.
The photocatalytic results obtained with PhR-B (k = 0.0047 min
-1, τ = 212.77 min) were excluded from the comparison due to their low relative photocatalytic activity. Therefore, despite high irradiance of PhR-B in the blue region of the visible spectrum (2360.44 W/m
2) the LSPR phenomenon from Ag NPs could not be activated by the low-energy wavelength. In this sense, the photocatalytic activity with PhR-Hg and solar radiation was greater than with PhR-B. In
Figure 6a two irradiances are shown for PhR-Hg, 101 W/m
2 with 75 V and 143 W/m2 for 90 V. For PhR-T5 three irradiances are shown, 7.25 W/m2 at 11.5 cm, 9.50 W/m
2 at 4.5 cm and 25.10 W/m
2 at 0 cm. For all PhRs, the irradiated wavelengths were from 200 to 500 nm, being able to photoactivate AgTiC with the highest energy wavelengths
According to the results shown in
Figure 6a, it is observed that higher irradiance values promoted better photocatalytic activity. That is, for PhR-Hg the irradiance of 143 W/m
2 enhanced the photocatalytic activity regarding to the photocatalytic activity obtained under the irradiance of 101 W/m
2. The same trend was observed for the photocatalytic activities obtained with different PhR-T5 configurations. Based on the τ indicated in
Figure 6a, MB was degraded more effectively with PhR-Hg at 143 W/m
2, meanwhile the less effective PhR configuration was PhR-T5 at 7.25 W/m
2. That is, higher irradiance from PhR-Hg enhanced the photocatalytic MB degradation significatively regarding to lowest irradiance from PhR-T5. It is important to note that solar radiation inefficiently degraded the MB, due to the low-energy photons contained in the UV spectrum of this radiation source.
Figure 6b shows k values with PhR-Hg, PhR-T5 and for solar irradiation as a function of the total irradiance. The comparison of the highest tested irradiance for PhR-Hg (143 W/m2, at 90 V) with the lowest tested irradiance for PhR-T5 (7.25 W/m
2 at 11.5 cm), indicated that irradiance was near to 20-times higher for PhR-Hg. The corresponding k values for PhR-Hg (143 W/m
2, at 90 V) and PhR-T5 (7.25 W/m
2 at 11.5 cm) were 0.033 and 0.0036 min
-1, respectively. Which involves an improvement of the photocatalytic activity by almost 10-times for the PhR providing highest irradiance. That is, the photocatalytic activity was enhanced 10-times when the irradiance increased 20-times. It is important to note that tested PhRs emitted photons with different wavelengths, with the photons corresponding to the effective irradiance being those that enhanced the photocatalytic activity, as observed for PhR-Hg. These results highlight that photocatalytic activity is a function of the magnitude (intensity) of the irradiance as well as the wavelength emitted by the PhR.
3.8. Photocatalytic Activity as a Function of the Effective Irradiance
The relationship of the photocatalytic activity regarding the irradiance could be determined based on the photocatalytic activity results (section 3.5 and 3.5.1) and the KPM discussed in section 3.4. According to
Figure 6 photocatalytic activity was enhanced at higher irradiances and when the wavelength irradiated is equal or lower than that required by the E
g of AgTiC. According to the Tauc plot analysis the Eg of the AgTiC was 3.55 eV. Therefore, the required wavelength for the photoactivation of AgTiC should be equal to or lower than 360 nm. Thus, it is imperative to identify the specific wavelengths to activate the photocatalyst and therefore assess in a proper way photocatalytic activity as a function of the irradiance. In this sense, the PhR-Hg (
Figure 4c) emitted radiation from 200 nm to 780 nm. Thus, only the UV irradiance, equal to or lower than 360 nm, is suitable to photoactivate AgTiC. As mentioned previously, this specific irradiance at adequate wavelength is named the effective irradiance (I
e). Also, for PhR-Hg, it is very likely that photons from 400–500 nm could activate the LSPR from Ag NPs. Conversely, the photons from visible spectrum between 500-780 nm are not significantly absorbed by AgTiC and, therefore, do not contribute to the photocatalytic process.
The photocatalytic activity as a function of the irradiance should be determined and this relationship must be considered in the KPM. As well as it is important consider the I_e instead of total irradiance. To this, experimental photocatalytic data (for PhR-Hg and PhR-T5) were fitted considering the effective irradiance to determine the dependence of the photocatalytic activity with the irradiance, as follows:
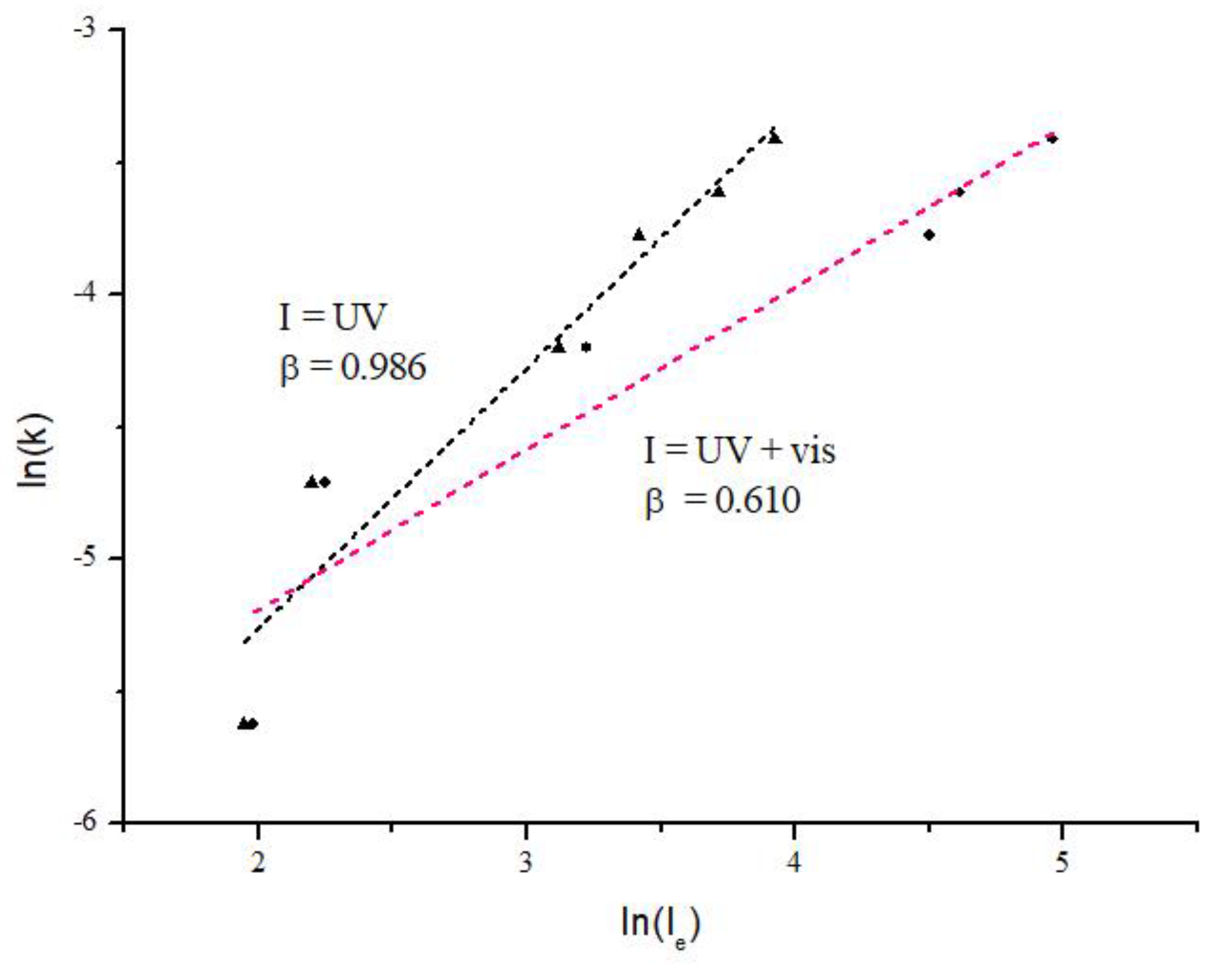
Figure 7 shows the plot of ln(k) vs ln(I
e) for the photocatalytic tests carried out. It can be observed photocatalytic results under UV and UV+visible irradiation. For the UV irradiation tests the β coefficient, determined as the slope of the linear equation, was 0.99 (R
2 = 0.93). This suggests that irradiance intensity with wavelength equal or lower than the required according to the E
g of AgTiC has a direct function with photocatalytic activity. For comparison purposes the photocatalytic activity under UV+visible irradiation was carried out considering the region equal or lower than 500 nm. It is important to note that visible radiation could only activate the LSPR of small fraction of the Ag NPs. The analysis of the data ln(k) vs ln(I
e) under UV+visible irradiance indicated a β of 0.61 (R
2 = 0.90). This value of β suggests the presence of intrinsic inefficiencies for the absorption of incident photons, both for the photoactivation of TiO
2 and Ag NPs, as had been previously assumed. The above results indicate that the type of radiation was preponderant to the photoactivation of the AgTiC, and therefore for the enhancement of the photocatalytic activity for the MB degradation.
Therefore, a representative kinetic correlation for the description of the photoactivity of AgTiC must consider only the fraction of UV irradiance (I
e), which is effective for the photoactivation. Then, it can be stated that high effective irradiance values are directly proportional to photocatalytic activity. It is important to note that experimental photocatalytic data obtained were fitted well with the equation (9). Where pseudo first order photocatalytic kinetic model and β=1 was considered, as follows: