Submitted:
20 January 2026
Posted:
21 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Taxonomy and Phylogenetic Framework: Species Delimitation, Lineage Structure, and Geographic Patterns
2.1. Traditional Morphological Classification and Its Controversies
2.2. Molecular Phylogenetics and DNA Barcoding
2.3. Multi-Locus Data, Coalescent Models, and Gene Flow
2.4. Natural Hybridization and Species Boundaries
3. Geologic History and Drainage Evolution as Drivers of Radiation: From “Plateau Uplift” to “Lineage Divergence”
3.1. How Episodic Uplift, Glaciations, and Canyon Formation Shape Schizothoracine Divergence
3.2. A Representative Case: Geomorphic Development of the Yarlung Tsangpo–Parlung Tsangpo System and Patterns of Gene Exchange
3.3. A Representative Case: The Tianshan–Pamir Corridor, Westward Dispersal, and Rapid Diversification
4. Polyploidy and Genome Evolution: From Ploidy Patterns to Early Rediploidization
4.1. Ploidy Landscape and Core Questions
4.2. “Young Tetraploids” and Early Rediploidization
4.3. Evidence Framework for Polyploid Origins and Post-WGD Processes
4.4. Comparison and Synthesis
5. Omics Resources and Methodological Advances: From Data Accumulation to Testable Evidence Chains
5.1. Inventory of Available Resources
5.2. Key Analytical Pipelines
7. Phenotypic, Morphological, and Life-History Adaptation: Linking “Molecular Modules” to Ecological Function
7.1. Morphological Differentiation and Niche Association
7.2. High-Throughput Phenotyping
7.3. Otolith Microchemistry and Life-History Reconstruction
7.4. Age and Growth
7.5. Reproductive Ecology and Trophic Ecology
8. Population Genetic Structure and Conservation Units: From Genetic Diversity to Management Units (MU/ESU)
8.1. Operationalizing MU/ESU in Schizothoracine Research
8.2. Upgrading Data Types and Elevating Evidence Strength
8.3. Multi-Scale Evidence Chains and Management Translation
9. Anthropogenic Pressures and Management Responses: Translating Evidence into an Actionable Checklist
9.1. Minimum Actionable Response Framework for Three Major Pressure Types
9.2. One-Page Action Checklist and Evaluation Metrics
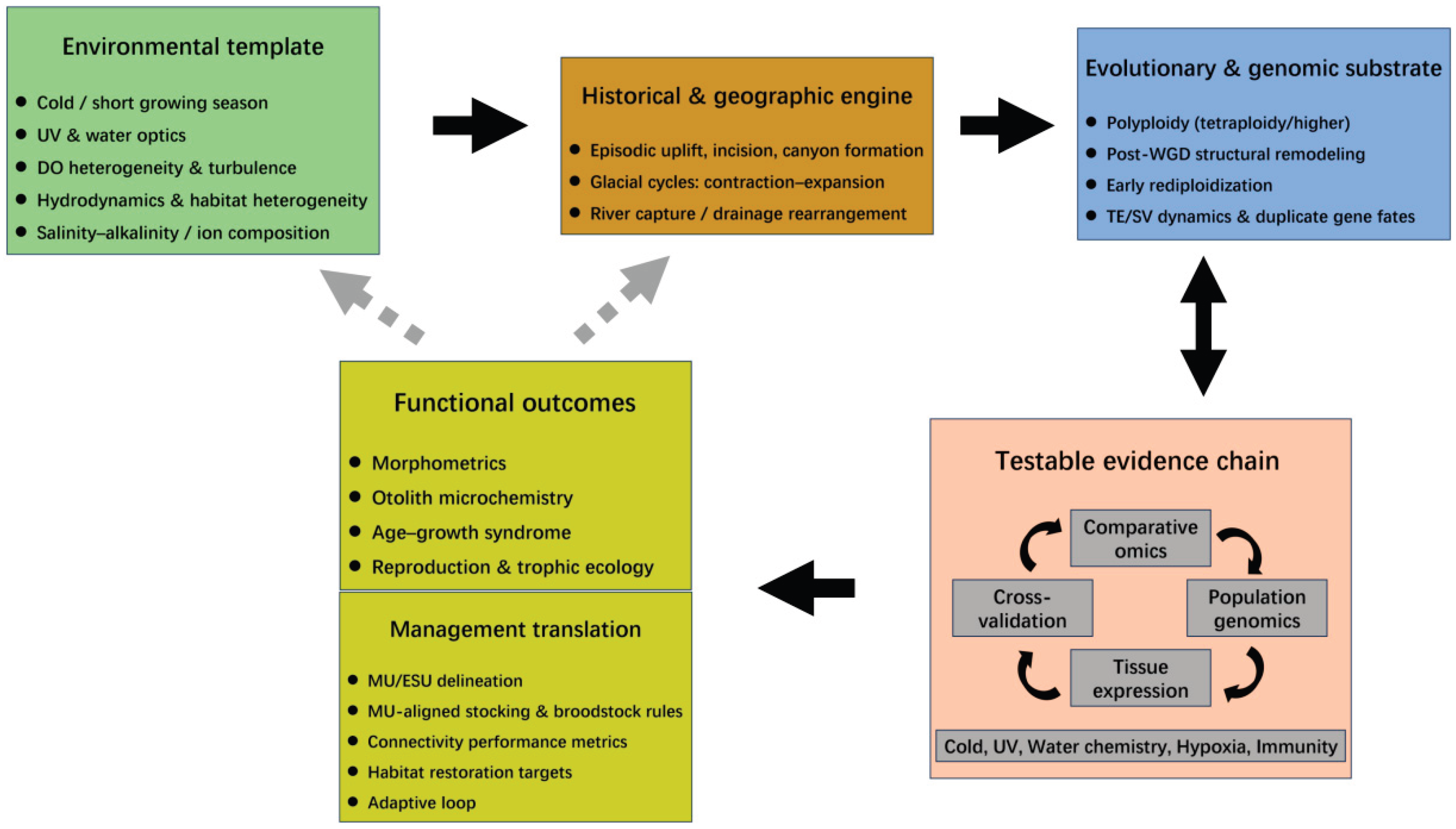
10. Integrated Framework and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Tian, F.; Liu, S.; Zhou, B.; Tang, Y.; Zhang, Y.; Zhang, C.; Zhao, K. Chromosome-level genome of Tibetan naked carp (Gymnocypris przewalskii) provides insights into Tibetan highland adaptation. DNA Res. 2022, 29, dsac025. [CrossRef]
- Xie, Y. G.; Qi, Y. L.; Luo, Z. H.; Qu, Y. N.; Yang, J.; Liu, S. Q.; Yang, H. L.; Xie, D. W.; Wang, Z.; Jiang, H. C.; et al. Lake salinization on the Qinghai-Tibetan Plateau alters viral community composition and lifestyles. Commun. Earth Environ. 2025, 6, 49. [CrossRef]
- Ren, W.; Gao, Y.; Qian, H.; Qu, W.; Shi, X.; Ma, Y.; Su, Z.; Ma, W. The Evolution and Drivers of Hydrochemistry in Nam Co Lake, the Third Largest Lake on the Tibetan Plateau, over the Last 20 Years. Sustainability 2025, 17, 2180. [CrossRef]
- Grabner, D.; Rothe, L. E.; Sures, B. Parasites and Pollutants: Effects of Multiple Stressors on Aquatic Organisms. Environ. Toxicol. Chem. 2023, 42, 1946-1959. [CrossRef]
- Hughes, S. J.; Santos, J.; Ferreira, T.; Mendes, A. Evaluating the response of biological assemblages as potential indicators for restoration measures in an intermittent Mediterranean river. Environ. Manage. 2010, 46, 285-301. [CrossRef]
- Vieira, T. B.; Tejerina-Garro, F. L. Relationships Between Environmental Conditions And Fish Assemblages InTropical Savanna Headwater Streams. Sci. Rep. 2020, 10, 2174. [CrossRef]
- Du, T.; Ding, C.; Yang, K.; Chen, J.; Liu, X.; Lv, W.; Ding, L.; He, D.; Tao, J. A global dataset on species occurrences and functional traits of Schizothoracinae fish. Sci. Data 2024, 11, 272. [CrossRef]
- He, Z.; Gao, K.; Chen, H.; Yang, D.; Pu, Y.; Zheng, L.; Jiao, Y.; Xiong, J.; Chen, Q.; Lai, B.; et al. Comparative Population Dynamics of Schizothorax wangchiachii (Cyprinidae: Schizothoracinae) in the Middle Reaches of the Yalong River and the Upper Reaches of the Jinsha River, China. Animals 2023, 13, 2209. [CrossRef]
- Chen, W.; Yue, X.; He, S. Genetic differentiation of the Schizothorax species complex (Cyprinidae) in the Nujiang River (upper Salween). Sci. Rep. 2017, 7, 5944. [CrossRef]
- Zhou, C.; Xiao, S.; Liu, Y.; Mou, Z.; Zhou, J.; Pan, Y.; Zhang, C.; Wang, J.; Deng, X.; Zou, M.; et al. Comprehensive transcriptome data for endemic Schizothoracinae fish in the Tibetan Plateau. Sci. Data 2020, 7, 28. [CrossRef]
- Tang, Y.; Li, C.; Wanghe, K.; Feng, C.; Tong, C.; Tian, F.; Zhao, K. Convergent evolution misled taxonomy in schizothoracine fishes (Cypriniformes: Cyprinidae). Mol. Phylogenet. Evol. 2019, 134, 323-337. [CrossRef]
- Xiao, S.; Mou, Z.; Fan, D.; Zhou, H.; Zou, M.; Zou, Y.; Zhou, C.; Yang, R.; Liu, J.; Zhu, S.; et al. Genome of Tetraploid Fish Schizothorax o'connori Provides Insights into Early Re-diploidization and High-Altitude Adaptation. iScience 2020, 23, 101497. [CrossRef]
- Guo, X. Z.; Zhang, G. R.; Wei, K. J.; Ji, W.; Yan, R. J.; Wei, Q. W.; Gardner, J. P. A. Phylogeography of the threatened tetraploid fish, Schizothorax waltoni, in the YarlungTsangpo River on the southern Qinghai-Tibet Plateau: implications for conservation. Sci. Rep. 2019, 9, 2704. [CrossRef]
- Regmi, B.; Douglas, M. R.; Wangchuk, K.; Zbinden, Z. D.; Edds, D. R.; Tshering, S.; Douglas, M. E. The Himalayan uplift and evolution of aquatic biodiversity across Asia: Snowtrout (Cyprininae: Schizothorax) as a test case. PLoS One 2023, 18, e0289736. [CrossRef]
- Xu, M. R.; Liao, Z. Y.; Brock, J. R.; Du, K.; Li, G. Y.; Chen, Z. Q.; Wang, Y. H.; Gao, Z. N.; Agarwal, G.; Wei, K. H.; et al. Maternal dominance contributes to subgenome differentiation in allopolyploid fishes. Nat. Commun. 2023, 14, 8357. [CrossRef]
- Gao, K.; He, Z.; Xiong, J.; Chen, Q.; Lai, B.; Liu, F.; Chen, P.; Chen, M.; Luo, W.; Huang, J.; et al. Population structure and adaptability analysis of Schizothorax o'connori based on whole-genome resequencing. BMC Genomics 2024, 25, 145. [CrossRef]
- Zhou, B.; Sui, R.; Yu, L.; Qi, D.; Fu, S.; Luo, Y.; Qi, H.; Li, X.; Zhao, K.; Liu, S.; et al. Transcriptomics and proteomics provide insights into the adaptative strategies of Tibetan naked carps (Gymnocypris przewalskii) to saline-alkaline variations. BMC Genomics 2025, 26, 162. [CrossRef]
- Li, B.; Hu, F.; Li, W.; Su, W.; Zhu, J.; Jiang, W. Spawning habitat selection in Schizothorax wangchiachii using acoustic tagging and tracking. Front. Ecol. Evol. 2025, 13, 1615081. [CrossRef]
- Lu, Y.; Zhu, W. Y.; Liu, Q. Y.; Li, Y.; Tian, H. W.; Cheng, B. X.; Zhang, Z. Y.; Wu, Z. H.; Qing, J.; Sun, G.; et al. Impact of Low-Head Dam Removal on River Morphology and Habitat Suitability in Mountainous Rivers. Int. J. Environ. Res. Public Health 2022, 19, 11743. [CrossRef]
- Niu, J.; Zhang, R.; Hu, J.; Zhang, T.; Liu, H.; Minavar, M.; Zhang, H.; Xian, W. Chromosomal-scale genome assembly of the near-extinction big-head schizothorcin (Aspiorhynchus laticeps). Sci. Data 2022, 9, 556. [CrossRef]
- Yuan, Q.; Li, K.; Wang, Y.; Ji, Q.; Liang, R. Tolerance and Growth of Adult Schizothorax prenanti Exposed and Re-Exposed to Supersaturated Gas Downstream of a Dam. Water 2022, 14, 2501. [CrossRef]
- Cheng, L.; Song, D.; Yu, X.; Du, X.; Huo, T. Endangered Schizothoracin Fish in the Tarim River Basin Are Threatened by Introgressive Hybridization. Biology 2022, 11, 981. [CrossRef]
- He, L.; Bi, Y.; Weese, D.; Wu, J.; Xu, S.; Ren, H.; Zhang, F.; Liu, X.; Chen, L.; Zhang, J. Genetic Signature of River Capture Imprinted in Schizopygopsis Fish from the Eastern Tibetan Plateau. Genes 2024, 15, 1148. [CrossRef]
- Li, M.; An, R.; Chen, M.; Li, J. Evaluation of Volitional Swimming Behavior of Schizothorax prenanti Using an Open-Channel Flume with Spatially Heterogeneous Turbulent Flow. Animals 2022, 12, 752. [CrossRef]
- Hoelzel, A. R. Where to now with the evolutionarily significant unit? Trends Ecol. Evol. 2023, 38, 1134-1142. [CrossRef]
- Alves, F.; Banks, S. C.; Edworthy, M.; Stojanovic, D.; Langmore, N. E.; Heinsohn, R. Using conservation genetics to prioritise management options for an endangered songbird. Heredity 2023, 130, 289-301. [CrossRef]
- Cao, W. X.; Chen, Y. Y.; Wu, Y. F.; Zhu, S. Q. Origin and evolution of schizothoracine fishes in relation to the upheaval of the Xizang Plateau. In Studies on the Period, Amplitude and Type of the Uplift of the Qinghai–Xizang Plateau; Tibetan Expedition Team of the Chinese Academy of Sciences, Ed.; Science Press: Beijing, China, 1981; pp. 118–130. https://www.cabidigitallibrary.org/doi/full/10.5555/19812608810.
- Qi, D.; Chao, Y.; Guo, S.; Zhao, L.; Li, T.; Wei, F.; Zhao, X. Convergent, parallel and correlated evolution of trophic morphologies in the subfamily schizothoracinae from the Qinghai-Tibetan plateau. PLoS One 2012, 7, e34070. [CrossRef]
- Zhang, C.; Tong, C.; Ludwig, A.; Tang, Y.; Liu, S.; Zhang, R.; Feng, C.; Li, G.; Peng, Z.; Zhao, K. Adaptive Evolution of the Eda Gene and Scales Loss in Schizothoracine Fishes in Response to Uplift of the Tibetan Plateau. Int. J. Mol. Sci. 2018, 19, 2953. [CrossRef]
- Li, Y.; Luo, Y.; Lv, Y.; Ou, Y.; Zhang, R.; Zhang, R. A Novel Mitochondrial Genome Resource for the Endemic Fish Gymnodiptychus integrigymnatus and Insights into the Phylogenetic Relationships of Schizothoracinae. Biology 2025, 14, 1760. [CrossRef]
- Ma, B.; Zhao, T.; Xu, B.; Zhong, L.; Wu, X.; Wei, K.; Zhang, Z.; Li, Y. Morphological variation in Schizothorax oconnori, Schizothorax waltoni (Teleostei: Cyprinidae: Schizothoracinae), and their natural hybrids from the middle Yarlung Zangbo River, Tibet. Ecol. Evol. 2024, 14, e11342. [CrossRef]
- Wang, T.; Qi, D.; Sun, S.; Liu, Z.; Du, Y.; Guo, S.; Ma, J. DNA barcodes and their characteristic diagnostic sites analysis of Schizothoracinae fishes in Qinghai province. Mitochondrial DNA A DNA Mapp. Seq. Anal. 2019, 30, 592-601. [CrossRef]
- Antil, S.; Abraham, J. S.; Sripoorna, S.; Maurya, S.; Dagar, J.; Makhija, S.; Bhagat, P.; Gupta, R.; Sood, U.; Lal, R.; et al. DNA barcoding, an effective tool for species identification: a review. Mol. Biol. Rep. 2023, 50, 761-775. [CrossRef]
- Rozimov, A.; Wang, Y.; Wang, M.; Zou, M.; Sobirov, J.; Karimov, E.; Kholmatov, B.; Freyhof, J.; Namozov, S.; Wang, C.; et al. Mitochondrial genome insights into the phylogenetics and biogeographic evolution of snow trout (Cyprinidae, Schizothorax) in the Tien Shan Mountains. Zoosystematics and Evolution 2025, 101, 91-102. [CrossRef]
- Yue, X.; Shi, J.; Zou, Y.; Wang, B. The complete mitochondrial genome sequence of Schizothorax lissolabiatus (Cypriniformes: Cyprinidae). Mitochondrial DNA A DNA Mapp. Seq. Anal. 2016, 27, 2450-2. [CrossRef]
- Qin, Q.; Chen, L.; Zhang, F.; Xu, J.; Zeng, Y. Characterization of the Complete Mitochondrial Genome of Schizothorax kozlovi (Cypriniformes, Cyprinidae, Schizothorax) and Insights into the Phylogenetic Relationships of Schizothorax. Animals 2024, 14, 721. [CrossRef]
- Andersen, M. M.; Balding, D. J. How many individuals share a mitochondrial genome? PLoS Genet. 2018, 14, e1007774. [CrossRef]
- DeRaad, D. A.; McCullough, J. M.; DeCicco, L. H.; Hime, P. M.; Joseph, L.; Andersen, M. J.; Moyle, R. G. Mitonuclear discordance results from incomplete lineage sorting, with no detectable evidence for gene flow, in a rapid radiation of Todiramphus kingfishers. Mol. Ecol. 2023, 32, 4844-4862. [CrossRef]
- Guo, X. Z.; Zhang, G. R.; Wei, K. J.; Yan, R. J.; Ji, W.; Yang, R. B.; Wei, Q. W.; Gardner, J. P. Phylogeography and population genetics of Schizothorax o'connori: strong subdivision in the Yarlung Tsangpo River inferred from mtDNA and microsatellite markers. Sci. Rep. 2016, 6, 29821. [CrossRef]
- Heled, J.; Drummond, A. J. Bayesian inference of population size history from multiple loci. BMC Evol. Biol. 2008, 8, 289. [CrossRef]
- Payseur, B. A.; Rieseberg, L. H. A genomic perspective on hybridization and speciation. Mol. Ecol. 2016, 25, 2337-60. [CrossRef]
- Hohenlohe, P. A.; Funk, W. C.; Rajora, O. P. Population genomics for wildlife conservation and management. Mol. Ecol. 2021, 30, 62-82. [CrossRef]
- Cai, D.; Wang, X.; Li, G.; Jiao, R.; Kohn, B.; Zhu, W.; De Grave, J.; Lu, H. Fault systems impede incision of the Yarlung river into the Tibetan plateau. Commun. Earth Environ. 2023, 4, 200. [CrossRef]
- Stokes, M. F.; Kim, D.; Gallen, S. F.; Benavides, E.; Keck, B. P.; Wood, J.; Goldberg, S. L.; Larsen, I. J.; Mollish, J. M.; Simmons, J. W.; et al. Erosion of heterogeneous rock drives diversification of Appalachian fishes. Science 2023, 380, 855-859. [CrossRef]
- Swartz, E. R.; Chakona, A.; Skelton, P. H.; Bloomer, P. The genetic legacy of lower sea levels: does the confluence of rivers during the last glacial maximum explain the contemporary distribution of a primary freshwater fish (Pseudobarbus burchelli, Cyprinidae) across isolated river systems? Hydrobiologia 2014, 726, 109-121. [CrossRef]
- Robinson, R. A. J.; Brezina, C. A.; Parrish, R. R.; Horstwood, M. S. A.; Oo, N. W.; Bird, M. I.; Thein, M.; Walters, A. S.; Oliver, G. J. H.; Zaw, K. Large rivers and orogens: The evolution of the Yarlung Tsangpo–Irrawaddy system and the eastern Himalayan syntaxis. Gondwana Res. 2014, 26, 112-121. [CrossRef]
- Schmidt, J. L.; Zeitler, P. K.; Pazzaglia, F. J.; Tremblay, M. M.; Shuster, D. L.; Fox, M. Knickpoint evolution on the Yarlung river: Evidence for late Cenozoic uplift of the southeastern Tibetan plateau margin. Earth Planet. Sci. Lett. 2015, 430, 448-457. [CrossRef]
- Ledger, K. J.; Su, Y.; Jeon, J. Y.; Fullerton, A. H.; Kuligowski, D.; Bennett, T.; Denton, K.; McHenry, M.; McMillan, J. H.; et al. Using riverscape genetics to investigate the genetic response of two species and their life-history forms to dam removal. Front. Ecol. Evol. 2023, 11, 1225229. [CrossRef]
- Li, K.; Wang, J.; Wang, X.; Wang, M.; He, R.; Wang, M. Distribution pattern of fish richness in the Yarlung Zangbo River Basin. Diversity 2022, 14, 1142. [CrossRef]
- Barreto, S. B.; Knowles, L. L.; Affonso, P. R. A. M.; Batalha-Filho, H. Riverscape properties contribute to the origin and structure of a hybrid zone in a Neotropical freshwater fish. J. Evol. Biol. 2020, 33, 1530-1542. [CrossRef]
- Payseur, B. A.; Rieseberg, L. H. A genomic perspective on hybridization and speciation. Mol. Ecol. 2016, 25, 2337-60. [CrossRef]
- Liang, Y.; He, D.; Jia, Y.; Sun, H.; Chen, Y. Phylogeographic studies of schizothoracine fishes on the central Qinghai-Tibet Plateau reveal the highest known glacial microrefugia. Sci. Rep. 2017, 7, 10983. [CrossRef]
- Caves, J. K.; Bayshashov, B. U.; Zhamangara, A.; Ritch, A. J.; Ibarra, D. E.; Sjostrom, D. J.; Mix, H. T.; Winnick, M. J.; Chamberlain, C. P. Late Miocene uplift of the Tian Shan and Altai and reorganization of Central Asia climate. GSA Today 2017, 27, 20-26. [CrossRef]
- Dai, Y.; Han, H. Karyological analysis of two species in the subfamily Schizothoracinae (Cypriniformes: Cyprinidae) from China, with notes on karyotype evolution in Schizothoracinae. Turk. J. Fish. Aquat. Sci. 2018, 18, 175-186. [CrossRef]
- Chen, C. N.; Huang, Y. Y.; Li, H.; Long, Z. H.; Lai, J. S.; Liu, G. X.; Zhao, G. The complete mitochondrial genome of Schizothorax prenanti (Tchang) (Teleostei, Cyprinidae, Schizothoracinae). Mitochondrial DNA A DNA Mapp. Seq. Anal. 2016, 27, 253-254. [CrossRef]
- Gundappa, M. K.; To, T. H.; Grønvold, L.; Martin, S. A. M.; Lien, S.; Geist, J.; Hazlerigg, D.; Sandve, S. R.; Macqueen, D. J. Genome-Wide Reconstruction of Rediploidization Following Autopolyploidization across One Hundred Million Years of Salmonid Evolution. Mol. Biol. Evol. 2022, 39, msab310. [CrossRef]
- Krabbenhoft, T. J.; MacGuigan, D. J.; Backenstose, N. J. C.; Hannah Waterman H.; Lan T. Y.; Pelosi J. A.; Tan M.; Sandve S. R. Chromosome-level genome assembly of Chinese sucker (Myxocyprinus asiaticus) reveals strongly conserved synteny following a catostomid-specific whole-genome duplication. Genome Biol. Evol. 2021, 13, evab190. [CrossRef]
- Li, J. T.; Wang, Q.; Huang Yang, M. D.; Li, Q. S.; Cui, M. S.; Dong, Z. J.; Wang, H. W.; Yu, J. H.; Zhao, Y. J.; Yang, C. R.; et al. Parallel subgenome structure and divergent expression evolution of allo-tetraploid common carp and goldfish. Nat. Genet. 2021, 53, 1493-1503. [CrossRef]
- Redmond, A. K.; Casey, D.; Gundappa, M. K.; Macqueen, D. J.; McLysaght, A. Independent rediploidization masks shared whole genome duplication in the sturgeon-paddlefish ancestor. Nat. Commun. 2023, 14, 2879. [CrossRef]
- Lien, S.; Koop, B. F.; Sandve, S. R.; Miller, J. R.; Kent, M. P.; Nome, T.; Hvidsten, T. R.; Leong, J. S.; Minkley, D. R.; Zimin, A.; et al. The Atlantic salmon genome provides insights into rediploidization. Nature 2016, 533, 200-205. [CrossRef]
- Yang, L.; Mayden, R. L.; Naylor, G. J. P. Origin of Polyploidy, Phylogenetic Relationships, and Biogeography of Botiid Fishes (Teleostei: Cypriniformes). Biology 2025, 14, 531. [CrossRef]
- Glover, N. M.; Redestig, H.; Dessimoz, C. Homoeologs: What Are They and How Do We Infer Them? Trends Plant Sci. 2016, 21, 609-621. [CrossRef]
- Sancho, R.; Inda, L. A.; Díaz-Pérez, A.; Des Marais, D. L.; Gordon, S.; Vogel, J. P.; Lusinska, J.; Hasterok, R.; Contreras-Moreira, B.; Catalán, P. Tracking the ancestry of known and 'ghost' homeologous subgenomes in model grass Brachypodium polyploids. Plant J. 2022, 109, 1535-1558. [CrossRef]
- Soares, N. R.; Mollinari, M.; Oliveira, G. K.; Pereira, G. S.; Vieira, M. L. C. Meiosis in Polyploids and Implications for Genetic Mapping: A Review. Genes 2021, 12, 1517. [CrossRef]
- Xu, P.; Xu, J.; Liu, G.; Chen, L.; Zhou, Z.; Peng, W.; Jiang, Y.; Zhao, Z.; Jia, Z.; Sun, Y.; et al. The allotetraploid origin and asymmetrical genome evolution of the common carp Cyprinus carpio. Nat. Commun. 2019, 10, 4625. [CrossRef]
- Wang, L.; Zhu, P.; Mo, Q.; Luo, W.; Du, Z.; Jiang, J.; Yang, S.; Zhao, L.; Gong, Q.; Wang, Y. Comprehensive analysis of full-length transcriptomes of Schizothorax prenanti by single-molecule long-read sequencing. Genomics 2022, 114, 456-464. [CrossRef]
- Liang, X.; Wang, W.; Huang, J.; Luo, M.; Wangdui, N.; Sun, C.; Lu, J. A chromosome-level genome assembly of big-barbel schizothorcin, Schizothorax macropogon. Sci. Data 2024, 11, 1402. [CrossRef]
- Zhang, J.; Chen, Z.; Zhou, C.; Kong, X. Molecular phylogeny of the subfamily Schizothoracinae (Teleostei: Cypriniformes: Cyprinidae) inferred from complete mitochondrial genomes. Biochem. Syst. Ecol. 2016, 64, 6-13. [CrossRef]
- Tong, C.; Fei, T.; Zhang, C.; Zhao, K. Comprehensive transcriptomic analysis of Tibetan Schizothoracinae fish Gymnocypris przewalskii reveals how it adapts to a high altitude aquatic life. BMC Evol. Biol. 2017, 17, 74. [CrossRef]
- Zhang, D.; Yu, M.; Hu, P.; Peng, S.; Liu, Y.; Li, W.; Wang, C.; He, S.; Zhai, W.; Xu, Q.; et al. Genetic Adaptation of Schizothoracine Fish to the Phased Uplifting of the Qinghai-Tibetan Plateau. G3 2017, 7, 1267-1276. [CrossRef]
- Lei, L.; Deng, X.; Liu, F.; Gao, H.; Duan, Y.; Li, J.; Fu, S.; Li, H.; Zhou, Y.; Liao, R.; et al. Exploitation of Key Regulatory Modules and Genes for High-Salt Adaptation in Schizothoracine by Weighted Gene Co-Expression Network Analysis. Animals 2024, 15, 56. [CrossRef]
- Kirillin, G. B.; Shatwell, T.; Wen, L. Ice-covered lakes of Tibetan Plateau as solar heat collectors. Geophys. Res. Lett. 2021, 48, e2021GL093429. [CrossRef]
- Ban, X.; Dang, Y.; Shu, P.; Qi, H.; Luo, Y.; Xiao, F.; Feng, Q.; Zhou, Y. Estimation of phytoplankton primary productivity in Qinghai Lake using ocean color satellite data: Seasonal and interannual variations. Water 2024, 16, 1433. [CrossRef]
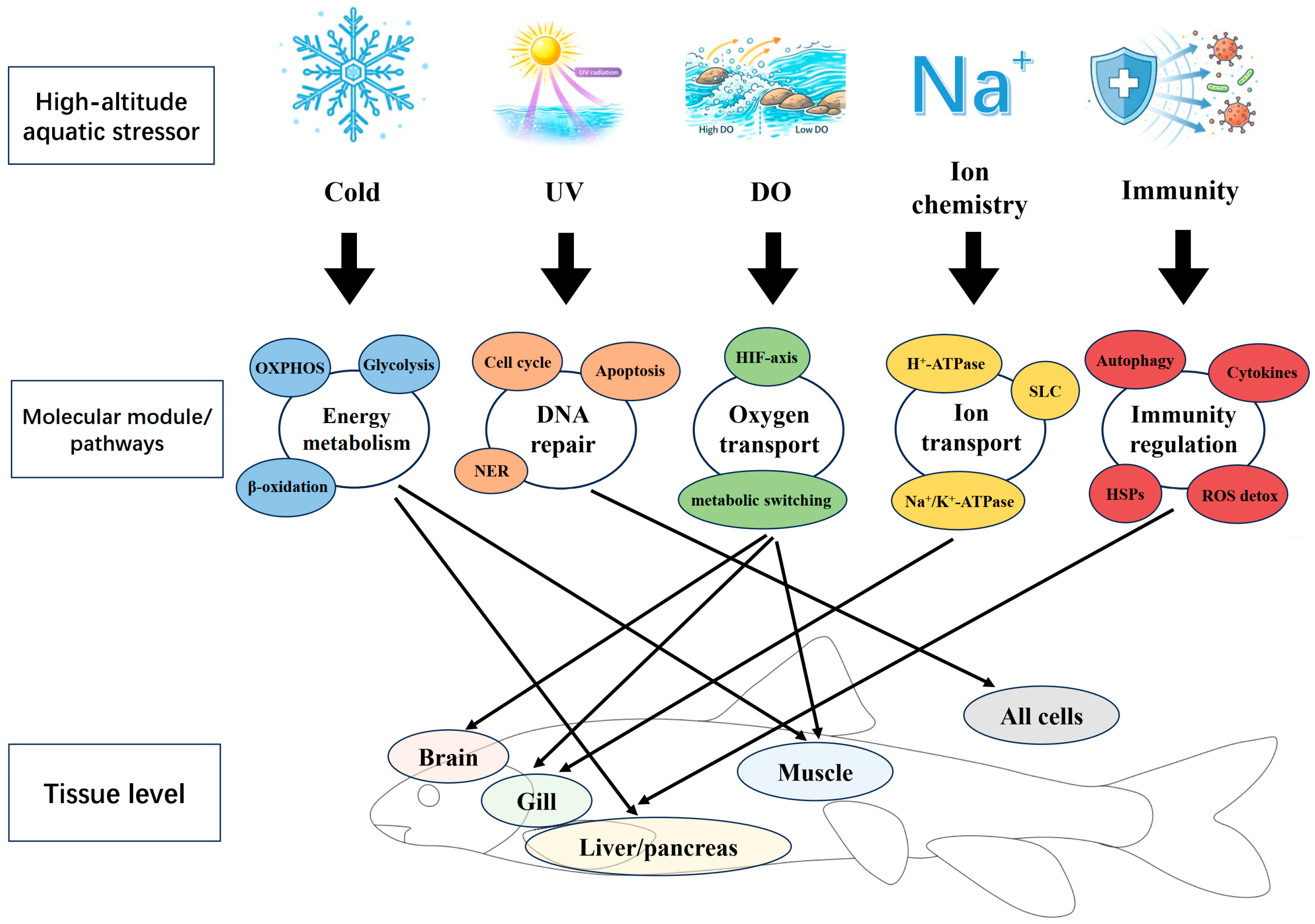
- Tong, C.; Tian, F.; Zhao, K. Genomic signature of highland adaptation in fish: a case study in Tibetan Schizothoracinae species. BMC Genomics 2017, 18, 948. [CrossRef]
- Xu, W.; Zhu, F.; Wang, D.; Chen, D.; Duan, X.; Liu, M.; Li, D. Comparative analysis of metabolites between different altitude Schizothorax nukiangensis (Cyprinidae, Schizothoracine) on the Qinghai-Tibet Plateau in Nujiang River. Water 2023, 15, 284. [CrossRef]
- Liu, S.; Tian, F.; Qi, D.; Qi, H.; Wang, Y.; Xu, S.; Zhao, K. Physiological, metabolomic, and transcriptomic reveal metabolic pathway alterations in Gymnocypris przewalskii due to cold exposure. BMC Genomics 2023, 24, 545. [CrossRef]
- Watanabe, S.; Overholt, E. P.; Schladow, S. G.; Vincent, W. F.; Williamson, C. E. Climate change and underwater light: Large-scale changes in ultraviolet radiation transparency associated with intensifying wet–dry cycles. Limnol. Oceanogr. Lett. 2025, 10, 485-494. [CrossRef]
- Qi, D.; Chao, Y.; Wu, R.; Xia, M.; Chen, Q.; Zheng, Z. Transcriptome Analysis Provides Insights Into the Adaptive Responses to Hypoxia of a Schizothoracine Fish (Gymnocypris eckloni). Front. Physiol. 2018, 9, 1326. [CrossRef]
- Guan, L.; Chi, W.; Xiao, W.; Chen, L.; He, S. Analysis of hypoxia-inducible factor alpha polyploidization reveals adaptation to Tibetan Plateau in the evolution of schizothoracine fish. BMC Evol. Biol. 2014, 14, 192. [CrossRef]
- Benson, A.; Zane, M.; Becker, T. E.; et al. Quantifying reaeration rates in alpine streams using deliberate gas tracer experiments. Water 2014, 6, 1013-1027. [CrossRef]
- Liang, Y.; Hou, Y.; Hu, W.; et al. Flow velocity preference of Schizothorax oconnori Lloyd swimming upstream. Glob. Ecol. Conserv. 2021, 32, e01902. [CrossRef]
- Mucha, S.; Chapman, L. J.; Krahe, R. Normoxia exposure reduces hemoglobin concentration and gill size in a hypoxia-tolerant tropical freshwater fish. Environ. Biol. Fishes 2023, 106, 1405-1423. [CrossRef]
- Jin, Y.; Zhu, B.; Wang, F.; et al. Analysis of water chemistry characteristics and main ion controlling factors of lakes in the Nagqu area of the Qinghai–Tibet plateau in summer. Water 2023, 15, 2900. [CrossRef]
- Tong, C.; Li, M. Genomic signature of accelerated evolution in a saline-alkaline lake-dwelling Schizothoracine fish. Int. J. Biol. Macromol. 2020, 149, 341-347. [CrossRef]
- Kousar, Y.; Singh, M.; Singh, D. Stock assessment of Schizothorax richardsonii (Gray, 1832) using geometric morphometrics and mitochondrial marker COX1 from tributaries of the Chenab River, India. J. Appl. Nat. Sci. 2025, 17, 600-613. [CrossRef]
- Gul, S.; Rashid, I. Morphological variations among Schizothorax species from Kashmir Himalayas. Zool. Anz. 2025, 316, 130-139. [CrossRef]
- Rajput, V.; Johnson, J. A.; Sivakumar, K. Environmental effects on the morphology of the snow trout Schizothorax richardsonii (Gray, 1832). TAPROBANICA: J. Asian Biodivers. 2013, 5, 102-110. [CrossRef]
- Martinez-Leiva, L.; Landeira, J. M.; Fatira, E.; Díaz-Pérez, J.; Hernández-León, S.; Roo, J.; Tuset, V. M. Energetic Implications of Morphological Changes between Fish Larval and Juvenile Stages Using Geometric Morphometrics of Body Shape. Animals 2023, 13, 370. [CrossRef]
- Reyes Corral, W. D.; Aguirre, W. E. Effects of temperature and water turbulence on vertebral number and body shape in Astyanax mexicanus (Teleostei: Characidae). PLoS One 2019, 14, e0219677. [CrossRef]
- Hetzel, C.; Forsythe, P. Phenotypic plasticity of a generalist fish species resident to lotic environments: Insights from the Great Lakes region. Ecol. Evol. 2023, 13, e10715. [CrossRef]
- Zhou, Y.; He, Z.; Cui, W.; Lu, Q.; Qin, J.; Han, Z.; Liu, J.; He, T. Strontium and Magnesium in Otoliths Can Trace Schizothorax grahami (Regan, 1904) Life History. Animals 2025, 15, 3170. [CrossRef]
- Gao, H.; Fu, S.; Xing, M.; et al. A high-throughput phenome-based analysis of morphological variation and environmental adaptation in extremely high-altitude schizothoracine fishes. Water Biol. Secur. 2025, 4, 100381. [CrossRef]
- Zhou, L.; Jin, Z. D.; Li, F. C. Mineralogy of the otoliths of naked carp Gymnocypris przewalskii (Kessler) from Lake Qinghai and its Sr/Ca potential implications for migratory pattern. Sci. China Earth Sci. 2012, 55, 983–990. [CrossRef]
- Belay, T. H.; Mengist, A. B. The application of otolith chemistry in fish life history assessment. Fish. Aquat. Sci. 2025, 28, 583-595. [CrossRef]
- Kratina, G. J.; DeVries, D. R.; Wright, R. A.; Peatman, E.; Rider, S. J.; Zhao, H. Using fish hard-part microchemistry and genetics to quantify population impacts of low-use lock-and-dam structures on the Alabama River. Trans. Am. Fish. Soc. 2023, 152, 490-512. [CrossRef]
- Wang, G.; Tang, Q.; Chen, Z.; Guo, D.; Zhou, L.; Lai, H.; Li, G. Otolith microchemistry and demographic history provide new insight into the migratory behavior and heterogeneous genetic divergence of coilia grayii in the pearl river. Fishes 2022, 7, 23. [CrossRef]
- Sturrock, A. M.; Hunter, E.; Milton, J. A.; Eimf; Johnson, R. C.; Waring, C. P.; Trueman, C. N. Quantifying physiological influences on otolith microchemistry. Methods Ecol. Evol. 2015, 6, 806-816. [CrossRef]
- Ma, B.; Xie, C.; Huo, B.; Yang, X.; Huang, H. Age and growth of a long-lived fish Schizothorax o'connori in the Yarlung Tsangpo River, Tibet. Zool. Stud. 2010, 49, 749-759. [CrossRef]
- Ma, B.; Xie, C.; Huo, B.; Yang, X.; Li, P. Age validation, and comparison of otolith, vertebra and opercular bone for estimating age of Schizothorax o’connori in the Yarlung Tsangpo River, Tibet. Environ. Biol. Fishes 2011, 90, 159-169. [CrossRef]
- Huo, B.; Ma, B. S.; Xie, C. X.; Duan, Y. J.; Yang, X. F.; Huang, H. P. Stock assessment and management implications of an endemic fish, Oxygymnocypris stewartii, in the Yarlung Zangbo River in Tibet, China. Zool. Stud. 2015, 54, 53. [CrossRef]
- Pfennig, M. B.; Crane, D. P.; Smith, N. G.; Buckmeier, D. L. Age estimation and validation in otoliths, spines, and fin rays from four central Texas fishes. N. Am. J. Fish. Manag. 2024, 44, 590-606. [CrossRef]
- Xu, L.; Zhou, Y.; Cui, W.; Lu, Q.; Liu, J.; Duan, C.; He, T. Characteristics of surface flow field and substrate in the spawning ground of Schizothorax grahami. Isr. J. Aquac. 2024, 76, 1-12. [CrossRef]
- Pan, H.; Liu, H.; Liu, F.; Xie, J.; Zhou, Y.; Zheng, Q.; Guo, M. Gut microbiota: a new frontier in understanding and protecting endangered plateau schizothorax fish. Front Microbiol. 2025, 16, 1592312. https://pubmed.ncbi.nlm.nih.gov/40584040/. [CrossRef]
- Zhong, C.; Chen, L.; Huang, Z.; Hu, Y.; Jiang, Y.; Zhou, J.; Long, X. Comparison of metabolism, gut histology, and microbiota between Schizothorax lissolabiatus and Schizothorax griseus under identical farming conditions. Front. Mar. Sci. 2024, 11, 1510780. [CrossRef]
- Mamoozadeh, N. R.; Wade, M. J.; Reid, B. N.; Bardwell, E.; Collins, E. E.; Hugentobler, S. A.; Jackson, S. A.; Kline, B. C.; Rothkopf, H. E.; Zhang, A.; Meek, M. H. A practical introduction to effective population size for fisheries management. Trans. Am. Fish. Soc. 2025, 154, 352-371. [CrossRef]
- Funk, W. C.; McKay, J. K.; Hohenlohe, P. A.; Allendorf, F. W. Harnessing genomics for delineating conservation units. Trends Ecol. Evol. 2012, 27, 489-496. [CrossRef]
- Semlitsch, R. D.; Hotz, H.; Guex, G. D. Competition among tadpoles of coexisting hemiclones of hybridogenetic Rana esculenta: support for the frozen niche variation model. Evolution 1997, 51, 1249-1261. [CrossRef]
- Liao, J.; Chen, S.; Liu, P.; Fontaneto, D.; Han, B. P. Environmental selection and gene flow jointly determine the population genetic diversity and structure of Diaphanosoma dubium along a watershed elevation. Glob. Ecol. Conserv. 2024, 49, e02773. [CrossRef]
- Yan, T.; Gao, K.; He, L.; Pu, Y.; Tang, Z.; Xiong, J.; Chen, Q.; Lai, B.; Liu, F.; Chen, P.; Chen, M.; Luo, W.; Huang, J.; Ding, W.; Yang, D.; He, Z. Genomic-based revelation of genetic structure and adaptive characterization of Schizopygopsis malacanthus in the Jinsha River and Yalong River. BMC Genomics 2025, 26, 870. [CrossRef]
- He, J.; He, Z.; Yang, D.; Ma, Z.; Chen, H.; Zhang, Q.; Deng, F.; Ye, L.; Pu, Y.; Zhang, M.; Yang, S.; Yang, S.; Yan, T. Genetic Variation in Schizothorax kozlovi Nikolsky in the Upper Reaches of the Chinese Yangtze River Based on Genotyping for Simplified Genome Sequencing. Animals 2022, 12, 2181. [CrossRef]
- Wang, F.; Wang, L.; Liu, D.; Gao, Q.; Nie, M.; Zhu, S.; Chao, Y.; Yang, C.; Zhang, C.; Yi, R.; et al. Chromosome-level assembly of Gymnocypris eckloni genome. Sci. Data 2022, 9, 464. [CrossRef]
- Wang, Q.; Lan, T.; Li, H.; Sahu, S. K.; Shi, M.; Zhu, Y.; Han, L.; Yang, S.; Li, Q.; Zhang, L.; Deng, Z.; Liu, H.; Hua, Y. Whole-genome resequencing of Chinese pangolins reveals a population structure and provides insights into their conservation. Commun. Biol. 2022, 5, 821. [CrossRef]
- Wedekind, C. Sexual selection and life-history decisions: implications for supportive breeding and the management of captive populations. Conserv. Biol. 2002, 16, 1204-1211. http://www.jstor.org/stable/3095316.
- Han, H.; Wang, L.; Zhang, C.; Li, H.; Ma, B. Population Structure, Growth Characteristics, Resource Dynamics, and Management Strategies of Schizopygopsis younghusbandi in Four Tributaries of the Yarlung Zangbo River, Tibet. Biology 2025, 14, 707. [CrossRef]
- Hixon, M. A.; Johnson, D. W.; Sogard, S. M. BOFFFFs: on the importance of conserving old-growth age structure in fishery populations. ICES J. Mar. Sci. 2014, 71, 2171-2185. [CrossRef]
- Cooke, S. J.; Hinch, S. G. Improving the reliability of fishway attraction and passage efficiency estimates to inform fishway engineering, science, and practice. Ecol. Eng. 2013, 58, 123-132. [CrossRef]
- Bunt, C. M.; Castro-Santos, T.; Haro, A. Performance of fish passage structures at upstream barriers to migration. River Res. Appl. 2012, 28, 457-478. [CrossRef]
- Frankham, R.; Ballou, J. D.; Eldridge, M. D.; Lacy, R. C.; Ralls, K.; Dudash, M. R.; Fenster, C. B. Predicting the probability of outbreeding depression. Conserv. Biol. 2011, 25, 465-475. https://10.1111/j.1523-1739.2011.01662.x.
- Humphries, P.; King, A.; McCasker, N.; Kopf, R. K.; Stoffels, R.; Zampatti, B.; Price, A. Riverscape recruitment: a conceptual synthesis of drivers of fish recruitment in rivers. Can. J. Fish. Aquat. Sci. 2020, 77, 213-225. [CrossRef]
- King, A. J.; Gwinn, D. C.; Tonkin, Z.; Mahoney, J.; Raymond, S.; Beesley, L. Using abiotic drivers of fish spawning to inform environmental flow management. J. Appl. Ecol. 2016, 53, 34-43. [CrossRef]
- Krellwitz, E. M.; Gido, K. B.; Mehl, H. E.; Totten, L. A.; Jones, T. C. Abiotic Drivers of Spawning and Early Life Stage Assemblage Dynamics of Great Plains Fishes. River Res. Appl. 2026, 42, 3-18. [CrossRef]
- Liu, Q. Y.; Li, J.; An, R. D.; Li, Y. Ecohydraulogical characteristic index system of Schizopygopsis younghusbandi during spawning periods in the Yarlung Tsangpo River. Int. J. Environ. Res. Public. Health 2018, 15, 1949. [CrossRef]
- Laikre, L.; Schwartz, M. K.; Waples, R. S.; Ryman, N.; GeM Working Group. Compromising genetic diversity in the wild: unmonitored large-scale release of plants and animals. Trends Ecol. Evol. 2010, 25, 520-529. https://doi: 10.1016/j.tree.2010.06.013.
- Palmé, A. Assessing and monitoring genetic patterns for conservation purposes with special emphasis on Scandinavia. Department of Zoology, Stockholm University, 2010. https://www.diva-portal.org/smash/get/diva2:353156/FULLTEXT01.pdf.
- Nie, M. A.; Schultz, C. A. Decision-making triggers in adaptive management. Conserv. Biol. 2012, 26, 1137-1144. http://www.jstor.org/stable/23360129.
| Objective | Action (minimum) | Metric (quantified) | Focus | Data | Rule | Reference |
| Structure protection | Closed season + Min size | Size/age rebound; recruitment ↑ | Key reaches; spawning season | P | No improv. ×2 yrs → tighten | [13,113,114,124] |
| Connectivity | Set passage target | Arrival efficiency; passage efficiency; number of passable days |
Migration season; representative reaches | C | No improv. ×2 yrs → redesign/adjust | [99,115,116,122,123,124] |
| Stocking risk control | MU-aligned source & release | Source compliance; anomaly warning | Transition zone | G + R | Violation → stop/rewire | [104,105,117] |
| Habitat restoration | Microhabitat restoration | Depth/ velocity/substrate/cover area | Key reach | H | No improv. ×2 yrs → relocate/retune | [118,119,120,121,124] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




