Submitted:
20 January 2026
Posted:
21 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animal Model
2.2. mRNA Quantification using RT PCR
2.3. Protein Quantification and Analysis
2.4. Quantifying Leukocytes Adherent to Retinal Vasculature
2.5. Measuring Retinal Vascular Permeability
2.6. Statistical Analysis
3. Results
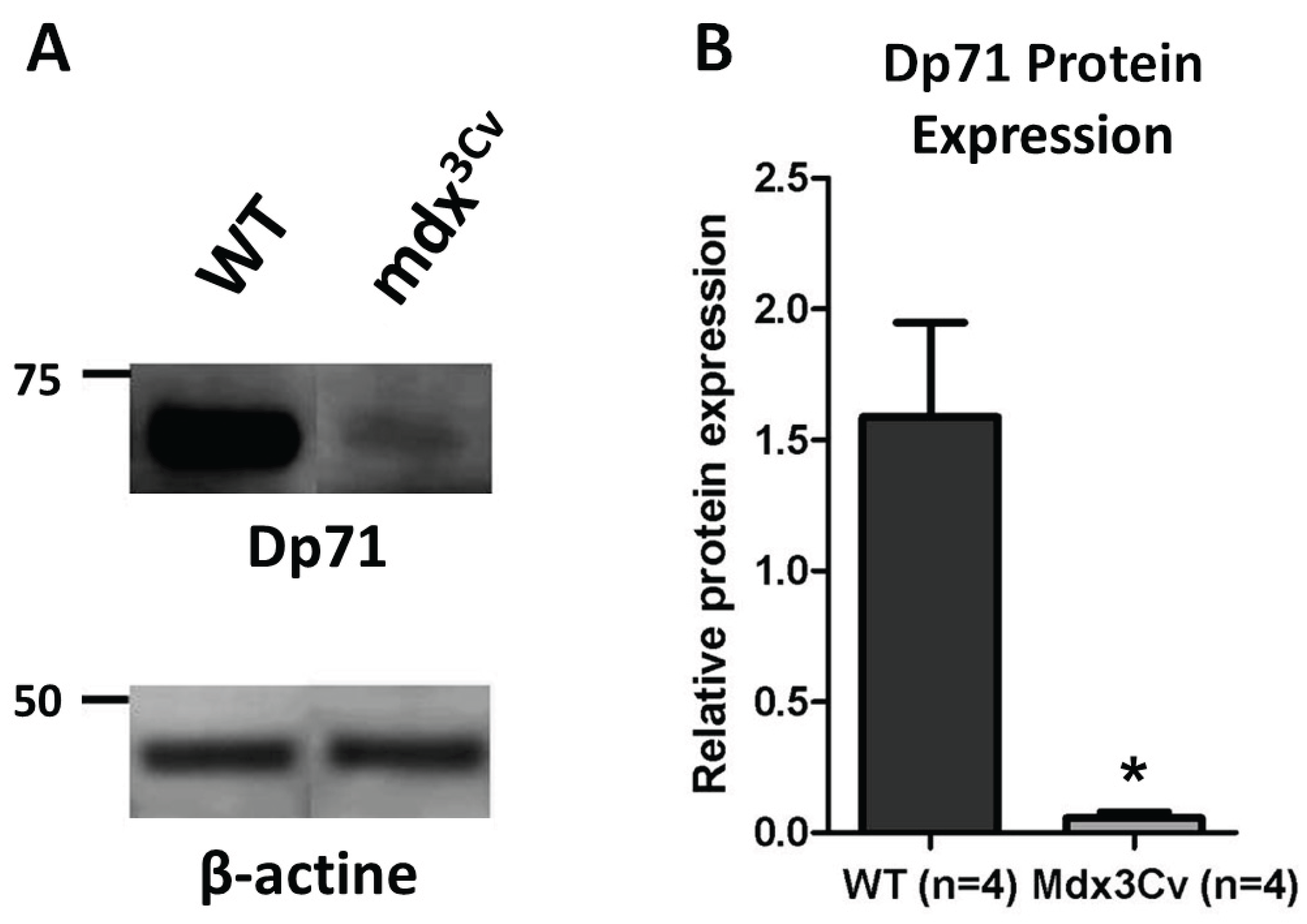
3.1. Dp71 expression in mdx3Cv mouse model
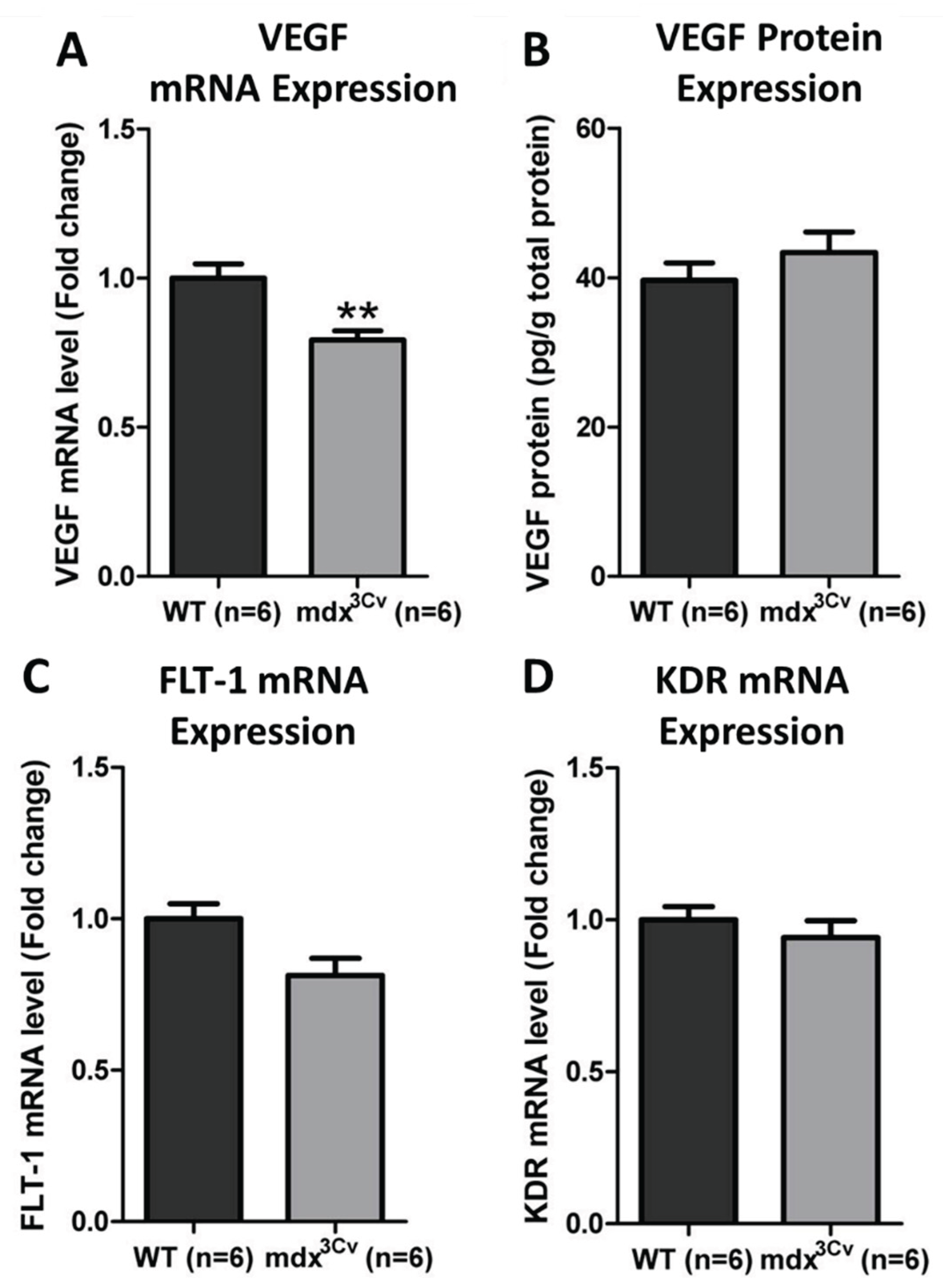
3.2. VEGF Pathway Gene Expression Remains Intact
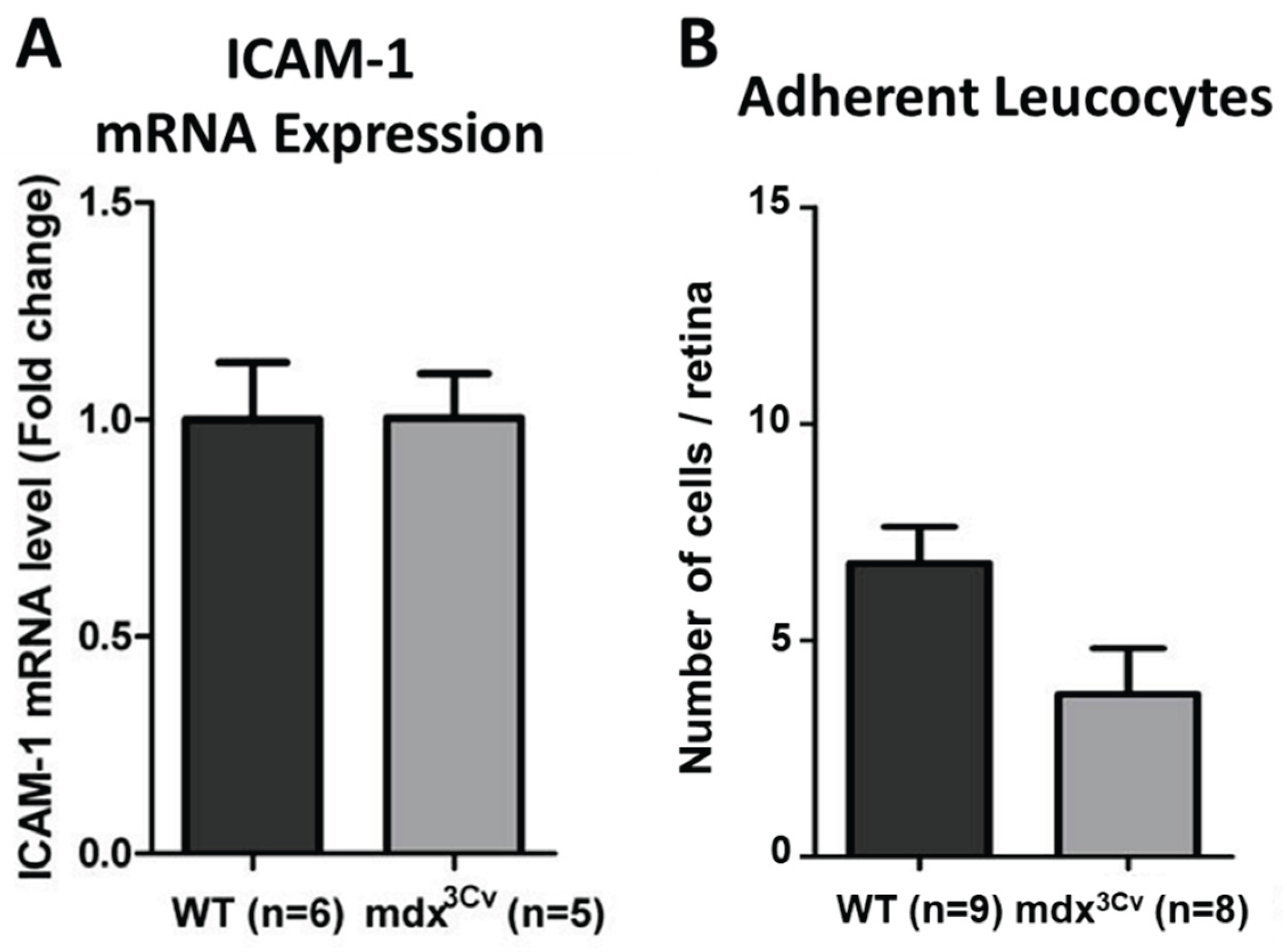
3.3. Cell-mediated inflammation is unchanged from WT in mdx3Cv
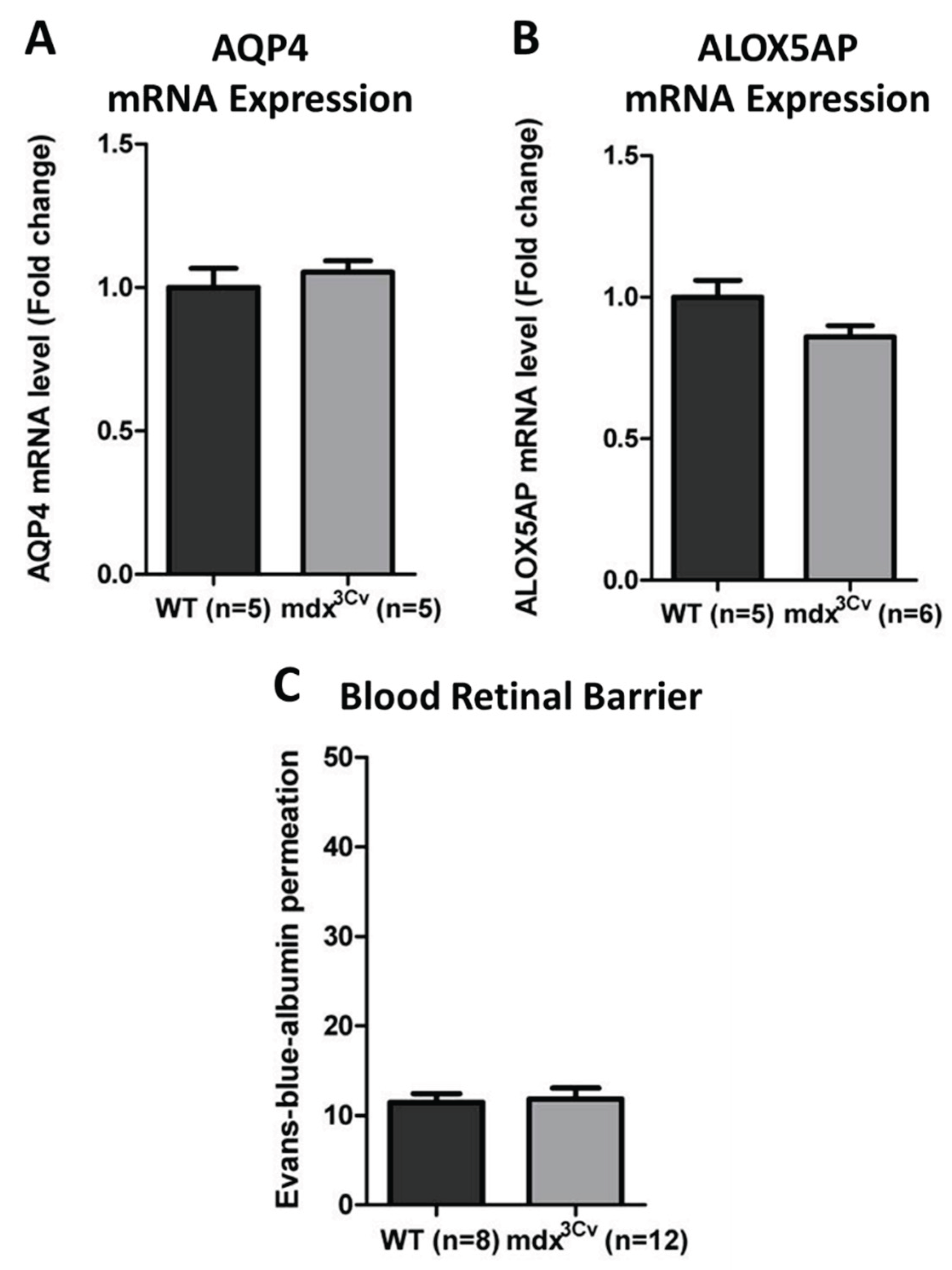
3.4. Preserved Permeability of the Blood-Retinal Barrier to Inflammatory Markers
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ALOX5AP | Arachidonate 5-Lipoxygenase Activating Protein |
| AQP4 | Aquaporin-4 |
| AMD | Age-related Macular Degeneration |
| BRB | Blood–Retinal Barrier |
| DAPC | Dystrophin-Associated Protein Complex |
| DMD | Duchenne Muscular Dystrophy |
| Dp | Dystrophin Protein Isoform |
| DR | Diabetic Retinopathy |
| ERG | Electroretinography |
| FLT-1 | Fms-Like Tyrosine kinase 1 (VEGFR-1) |
| ICAM-1 | Intracellular Adhesion Molecule 1 |
| ILM | Inner Limiting Membrane |
| KDR | Kinase Insert Domain Receptor (VEGFR-2) |
| RT-qPCR | Reverse Transcription quantitative Polymerase Chain Reaction |
| SEM | Standard Error of the Mean |
| VEGF | Vascular Endothelial Growth Factor |
| VEGFR | Vascular Endothelial Growth Factor Receptor |
| WT | Wild Type |
References
- Ehmsen, J.; Poon, E.; Davies, K. The Dystrophin-Associated Protein Complex. J Cell Sci 2002, 115, 2801–2803. [Google Scholar] [CrossRef]
- Tennyson, C.N.; Klamut, H.J.; Worton, R.G. The Human Dystrophin Gene Requires 16 Hours to Be Transcribed and Is Cotranscriptionally Spliced. Nat Genet 1995, 9, 184–190. [Google Scholar] [CrossRef]
- Tokarz, S.A.; Duncan, N.M.; Rash, S.M.; Sadeghi, A.; Dewan, A.K.; Pillers, D.A. Redefinition of Dystrophin Isoform Distribution in Mouse Tissue by RT-PCR Implies Role in Nonmuscle Manifestations of Duchenne Muscular Dystrophy. Mol Genet Metab 1998, 65, 272–281. [Google Scholar] [CrossRef]
- Vaillend, C.; Aoki, Y.; Mercuri, E.; Hendriksen, J.; Tetorou, K.; Goyenvalle, A.; Muntoni, F. Duchenne Muscular Dystrophy: Recent Insights in Brain Related Comorbidities. Nat Commun 2025, 16, 1298. [Google Scholar] [CrossRef] [PubMed]
- Ervasti, J.M. Dystrophin, Its Interactions with Other Proteins, and Implications for Muscular Dystrophy. Biochim Biophys Acta 2007, 1772, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, V.; Pavlakis, S. Duchenne Muscular Dystrophy. In StatPearls; StatPearls Publishing: Treasure Island (FL), 2025. [Google Scholar]
- Crisafulli, S.; Sultana, J.; Fontana, A.; Salvo, F.; Messina, S.; Trifirò, G. Global Epidemiology of Duchenne Muscular Dystrophy: An Updated Systematic Review and Meta-Analysis. Orphanet J Rare Dis 2020, 15, 141. [Google Scholar] [CrossRef] [PubMed]
- GB, D.D.B.; Duchenne De Boulogne GB De La Paralysie Musculaire Pseudo-Hypertrophique Ou Paralysie Myo-Sclérosique : Extrait Des Archives Générales de Médecine; Editeur des archives générales de médecine, 1868.
- Markati, T.; Oskoui, M.; Farrar, M.A.; Duong, T.; Goemans, N.; Servais, L. Emerging Therapies for Duchenne Muscular Dystrophy. Lancet Neurol 2022, 21, 814–829. [Google Scholar] [CrossRef]
- Pascual-Morena, C.; Cavero-Redondo, I.; Álvarez-Bueno, C.; Jiménez-López, E.; Saz-Lara, A.; Martínez-García, I.; Martínez-Vizcaíno, V. Global Prevalence of Intellectual Developmental Disorder in Dystrophinopathies: A Systematic Review and Meta-Analysis. Dev Med Child Neurol 2023, 65, 734–744. [Google Scholar] [CrossRef]
- Perumal, A.R.; Rajeswaran, J.; Nalini, A. Neuropsychological Profile of Duchenne Muscular Dystrophy. Appl Neuropsychol Child 2015, 4, 49–57. [Google Scholar] [CrossRef]
- Ramani, P.K.; Fawcett, K.; Guntrum, D.; Samuel, H.; Ciafaloni, E.; Veerapandiyan, A. Epilepsy Characteristics in Duchenne and Becker Muscular Dystrophies. Child Neurol Open 2023, 10, 2329048X231159484. [Google Scholar] [CrossRef]
- Parravicini, S.; Quaranta, C.A.; Dainesi, M.I.; Berardinelli, A. The Hidden Face of Duchenne (Neuro)Muscular Dystrophy. Preliminary Evidence of Social Cognition Impairment as a Feature of the Neuropsychological Phenotype of DMD. Front Psychol 2024, 15, 1504174. [Google Scholar] [CrossRef]
- Fagan, X.J.; Levy, J.; Al-Qureshi, S.; Harper, C.A. Proliferative Retinopathy in Duchenne Muscular Dystrophy and Its Response to Bevacizumab. Clin Exp Ophthalmol 2012, 40, 906–907. [Google Scholar] [CrossRef]
- Park, S.H.; Jo, Y.J.; Lee, J.J.; Park, S.W.; Lee, J.E. Proliferative Retinopathy Developed in a Duchenne Muscular Dystrophy Patient with Normal Cardiac Function. J Retin 2019, 4, 36–39. [Google Scholar] [CrossRef]
- Louie, K.; Apte, R.S.; Mori, K.; Gehlbach, P. Severe Proliferative Retinopathy in a Patient with Advanced Muscular Dystrophy. Br J Ophthalmol 2004, 88, 1604–1605. [Google Scholar] [CrossRef]
- Hahn, P.; Lin, P.; Fekrat, S. Ultra-Widefield Imaging of Duchenne Muscular Dystrophy-Associated Proliferative Retinal Vasculopathy Improved with Panretinal Laser Photocoagulation Alone. Ophthalmic Surg Lasers Imaging Retina 2013, 44, 293–295. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.F.; Oliveira, A.G.F.; Feitosa-Santana, C.; Zatz, M.; Ventura, D.F. Red-Green Color Vision Impairment in Duchenne Muscular Dystrophy. Am J Hum Genet 2007, 80, 1064–1075. [Google Scholar] [CrossRef] [PubMed]
- Blake, D.J.; Kröger, S. The Neurobiology of Duchenne Muscular Dystrophy: Learning Lessons from Muscle? Trends Neurosci 2000, 23, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Ricotti, V.; Jägle, H.; Theodorou, M.; Moore, A.T.; Muntoni, F.; Thompson, D.A. Ocular and Neurodevelopmental Features of Duchenne Muscular Dystrophy: A Signature of Dystrophin Function in the Central Nervous System. Eur J Hum Genet 2016, 24, 562–568. [Google Scholar] [CrossRef]
- Pillers, D.A.; Bulman, D.E.; Weleber, R.G.; Sigesmund, D.A.; Musarella, M.A.; Powell, B.R.; Murphey, W.H.; Westall, C.; Panton, C.; Becker, L.E. Dystrophin Expression in the Human Retina Is Required for Normal Function as Defined by Electroretinography. Nat Genet 1993, 4, 82–86. [Google Scholar] [CrossRef]
- Jiang, X.; Mahroo, O.A. Negative Electroretinograms: Genetic and Acquired Causes, Diagnostic Approaches and Physiological Insights. Eye (Lond) 2021, 35, 2419–2437. [Google Scholar] [CrossRef]
- Barboni, M.T.S.; Joachimsthaler, A.; Roux, M.J.; Nagy, Z.Z.; Ventura, D.F.; Rendon, A.; Kremers, J.; Vaillend, C. Retinal Dystrophins and the Retinopathy of Duchenne Muscular Dystrophy. Prog Retin Eye Res 2023, 95, 101137. [Google Scholar] [CrossRef]
- Howard, P.L.; Dally, G.Y.; Wong, M.H.; Ho, A.; Weleber, R.G.; Pillers, D.A.; Ray, P.N. Localization of Dystrophin Isoform Dp71 to the Inner Limiting Membrane of the Retina Suggests a Unique Functional Contribution of Dp71 in the Retina. Hum Mol Genet 1998, 7, 1385–1391. [Google Scholar] [CrossRef]
- Satz, J.S.; Philp, A.R.; Nguyen, H.; Kusano, H.; Lee, J.; Turk, R.; Riker, M.J.; Hernández, J.; Weiss, R.M.; Anderson, M.G.; et al. Visual Impairment in the Absence of Dystroglycan. J Neurosci 2009, 29, 13136–13146. [Google Scholar] [CrossRef]
- Shen, W.; Fruttiger, M.; Zhu, L.; Chung, S.H.; Barnett, N.L.; Kirk, J.K.; Lee, S.; Coorey, N.J.; Killingsworth, M.; Sherman, L.S.; et al. Conditional Müllercell Ablation Causes Independent Neuronal and Vascular Pathologies in a Novel Transgenic Model. J Neurosci 2012, 32, 15715–15727. [Google Scholar] [CrossRef]
- Tout, S.; Chan-Ling, T.; Holländer, H.; Stone, J. The Role of Müller Cells in the Formation of the Blood-Retinal Barrier. Neuroscience 1993, 55, 291–301. [Google Scholar] [CrossRef]
- Mizutani, M.; Gerhardinger, C.; Lorenzi, M. Müller Cell Changes in Human Diabetic Retinopathy. Diabetes 1998, 47, 445–449. [Google Scholar] [CrossRef]
- Paulson, O.B.; Newman, E.A. Does the Release of Potassium from Astrocyte Endfeet Regulate Cerebral Blood Flow? Science 1987, 237, 896–898. [Google Scholar] [CrossRef] [PubMed]
- Watkins, S.; Robel, S.; Kimbrough, I.F.; Robert, S.M.; Ellis-Davies, G.; Sontheimer, H. Disruption of Astrocyte-Vascular Coupling and the Blood-Brain Barrier by Invading Glioma Cells. Nat Commun 2014, 5, 4196. [Google Scholar] [CrossRef] [PubMed]
- Shelton, M.D.; Distler, A.M.; Kern, T.S.; Mieyal, J.J. Glutaredoxin Regulates Autocrine and Paracrine Proinflammatory Responses in Retinal Glial (Muller) Cells. J Biol Chem 2009, 284, 4760–4766. [Google Scholar] [CrossRef]
- Dalloz, C.; Claudepierre, T.; Rodius, F.; Mornet, D.; Sahel, J.; Rendon, A. Differential Distribution of the Members of the Dystrophin Glycoprotein Complex in Mouse Retina: Effect of the Mdx(3Cv) Mutation. Mol Cell Neurosci 2001, 17, 908–920. [Google Scholar] [CrossRef] [PubMed]
- Blank, M.; Koulen, P.; Blake, D.J.; Kröger, S. Dystrophin and Beta-Dystroglycan in Photoreceptor Terminals from Normal and mdx3Cv Mouse Retinae. Eur J Neurosci 1999, 11, 2121–2133. [Google Scholar] [CrossRef]
- Cox, G.A.; Phelps, S.F.; Chapman, V.M.; Chamberlain, J.S. New Mdx Mutation Disrupts Expression of Muscle and Nonmuscle Isoforms of Dystrophin. Nat Genet 1993, 4, 87–93. [Google Scholar] [CrossRef]
- El Mathari, B.; Sene, A.; Charles-Messance, H.; Vacca, O.; Guillonneau, X.; Grepin, C.; Sennlaub, F.; Sahel, J.-A.; Rendon, A.; Tadayoni, R. Dystrophin Dp71 Gene Deletion Induces Retinal Vascular Inflammation and Capillary Degeneration. Hum Mol Genet 2015, 24, 3939–3947. [Google Scholar] [CrossRef]
- Wei, L.; Sun, X.; Fan, C.; Li, R.; Zhou, S.; Yu, H. The Pathophysiological Mechanisms Underlying Diabetic Retinopathy. Front Cell Dev Biol 2022, 10, 963615. [Google Scholar] [CrossRef] [PubMed]
- Callan, A.; Heckman, J.; Tah, G.; Lopez, S.; Valdez, L.; Tsin, A. VEGF in Diabetic Retinopathy and Age-Related Macular Degeneration. International Journal of Molecular Sciences 2025, 26, 4992. [Google Scholar] [CrossRef] [PubMed]
- Behl, T.; Kotwani, A. Exploring the Various Aspects of the Pathological Role of Vascular Endothelial Growth Factor (VEGF) in Diabetic Retinopathy. Pharmacol Res 2015, 99, 137–148. [Google Scholar] [CrossRef]
- Khalfaoui, T.; Lizard, G.; Beltaief, O.; Colin, D.; Ben Hamida, J.; Errais, K.; Ammous, I.; Zbiba, W.; Tounsi, L.; Zhioua, R.; et al. Immunohistochemical Analysis of Cellular Adhesion Molecules (ICAM-1, VCAM-1) and VEGF in Fibrovascular Membranes of Patients with Proliferative Diabetic Retinopathy: Preliminary Study. Pathol Biol (Paris) 2009, 57, 513–517. [Google Scholar] [CrossRef]
- Bui, T.M.; Wiesolek, H.L.; Sumagin, R. ICAM-1: A Master Regulator of Cellular Responses in Inflammation, Injury Resolution, and Tumorigenesis. J Leukoc Biol 2020, 108, 787–799. [Google Scholar] [CrossRef]
- Shimizu, J.; Kawai, M.; Kanazawa, I. [논문]Sarcolemmal Coexpression of Intercellular Adhesion Molecule-1 (ICAM-1) and HLA-DR in Inflammatory Myopathy. Neuropathology : official journal of the Japanese Society of Neuropathology 1994, 14, 149–157. [Google Scholar] [CrossRef]
- Frank, P.G.; Lisanti, M.P. ICAM-1: Role in Inflammation and in the Regulation of Vascular Permeability. Am J Physiol Heart Circ Physiol 2008, 295, H926–H927. [Google Scholar] [CrossRef]
- Yao, Y.; Du, J.; Li, R.; Zhao, L.; Luo, N.; Zhai, J.Y.; Long, L. Association between ICAM-1 Level and Diabetic Retinopathy: A Review and Meta-Analysis. Postgrad Med J 2019, 95, 162–168. [Google Scholar] [CrossRef]
- Barile, G.R.; Chang, S.S.; Park, L.S.; Reppucci, V.S.; Schiff, W.M.; Schmidt, A.M. Soluble Cellular Adhesion Molecules in Proliferative Vitreoretinopathy and Proliferative Diabetic Retinopathy. Curr Eye Res 1999, 19, 219–227. [Google Scholar] [CrossRef]
- Limb, G.; Chignell, A. Vitreous Levels of Intercellular Adhesion Molecule 1 (ICAM-1) as a Risk Indicator of Proliferative Vitreoretinopathy. Br J Ophthalmol 1999, 83, 953–956. [Google Scholar] [CrossRef] [PubMed]
- Limb, G.A.; Webster, L.; Soomro, H.; Janikoun, S.; Shilling, J. Platelet Expression of Tumour Necrosis Factor-Alpha (TNF-α), TNF Receptors and Intercellular Adhesion Molecule-1 (ICAM-1) in Patients with Proliferative Diabetic Retinopathy. Clin Exp Immunol 1999, 118, 213–218. [Google Scholar] [CrossRef]
- Ma, N.; Hunt, N.H.; Madigan, M.C.; Chan-Ling, T. Correlation between Enhanced Vascular Permeability, up-Regulation of Cellular Adhesion Molecules and Monocyte Adhesion to the Endothelium in the Retina during the Development of Fatal Murine Cerebral Malaria. Am J Pathol 1996, 149, 1745–1762. [Google Scholar]
- Nagelhus, E.A.; Ottersen, O.P. Physiological Roles of Aquaporin-4 in Brain. Physiol Rev 2013, 93, 1543–1562. [Google Scholar] [CrossRef]
- Li, J.; Patil, R.V.; Verkman, A.S. Mildly Abnormal Retinal Function in Transgenic Mice without Müller Cell Aquaporin-4 Water Channels. Invest Ophthalmol Vis Sci 2002, 43, 573–579. [Google Scholar]
- Claudepierre, T.; Dalloz, C.; Mornet, D.; Matsumura, K.; Sahel, J.; Rendon, A. Characterization of the Intermolecular Associations of the Dystrophin-Associated Glycoprotein Complex in Retinal Müller Glial Cells. J Cell Sci 2000, 113 Pt 19, 3409–3417. [Google Scholar] [CrossRef] [PubMed]
- Peters-Golden, M.; Henderson, W.R. Leukotrienes. N Engl J Med 2007, 357, 1841–1854. [Google Scholar] [CrossRef]
- Bringmann, A.; Pannicke, T.; Grosche, J.; Francke, M.; Wiedemann, P.; Skatchkov, S.N.; Osborne, N.N.; Reichenbach, A. Müller Cells in the Healthy and Diseased Retina. Prog Retin Eye Res 2006, 25, 397–424. [Google Scholar] [CrossRef] [PubMed]
- Antonetti, D.A.; Klein, R.; Gardner, T.W. Diabetic Retinopathy. N Engl J Med 2012, 366, 1227–1239. [Google Scholar] [CrossRef] [PubMed]
- Klaassen, I.; Van Noorden, C.J.F.; Schlingemann, R.O. Molecular Basis of the Inner Blood-Retinal Barrier and Its Breakdown in Diabetic Macular Edema and Other Pathological Conditions. Prog Retin Eye Res 2013, 34, 19–48. [Google Scholar] [CrossRef]
- Ferrara, N.; Adamis, A.P. Ten Years of Anti-Vascular Endothelial Growth Factor Therapy. Nat Rev Drug Discov 2016, 15, 385–403. [Google Scholar] [CrossRef] [PubMed]
- Apte, R.S.; Chen, D.S.; Ferrara, N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell 2019, 176, 1248–1264. [Google Scholar] [CrossRef] [PubMed]
- Olsson, A.-K.; Dimberg, A.; Kreuger, J.; Claesson-Welsh, L. VEGF Receptor Signalling - in Control of Vascular Function. Nat Rev Mol Cell Biol 2006, 7, 359–371. [Google Scholar] [CrossRef]
- Campochiaro, P.A. Ocular Neovascularization. J Mol Med (Berl) 2013, 91, 311–321. [Google Scholar] [CrossRef]
- Farea, M.; Rani, A.Q.M.; Maeta, K.; Nishio, H.; Matsuo, M. Dystrophin Dp71ab Is Monoclonally Expressed in Human Satellite Cells and Enhances Proliferation of Myoblast Cells. Sci Rep 2020, 10, 17123. [Google Scholar] [CrossRef]
- Karuppasamy, M.; Alexander, M.S. Restoration of Brain Dystrophin Using Tricyclo-DNA ASOs Restores Neurobehavioral Deficits in DMD Mice. Mol Ther Nucleic Acids 2023, 32, 841–842. [Google Scholar] [CrossRef]
- Zarrouki, F.; Relizani, K.; Bizot, F.; Tensorer, T.; Garcia, L.; Vaillend, C.; Goyenvalle, A. Partial Restoration of Brain Dystrophin and Behavioral Deficits by Exon Skipping in the Muscular Dystrophy X-Linked (Mdx) Mouse. Ann Neurol 2022, 92, 213–229. [Google Scholar] [CrossRef]
- Vacca, O.; Zarrouki, F.; Izabelle, C.; Belmaati Cherkaoui, M.; Rendon, A.; Dalkara, D.; Vaillend, C. AAV-Mediated Restoration of Dystrophin-Dp71 in the Brain of Dp71-Null Mice: Molecular, Cellular and Behavioral Outcomes. Cells 2024, 13, 718. [Google Scholar] [CrossRef]
- Feng, L.; Liang, L.; Zhang, S.; Yang, J.; Yue, Y.; Zhang, X. HMGB1 Downregulation in Retinal Pigment Epithelial Cells Protects against Diabetic Retinopathy through the Autophagy-Lysosome Pathway. Autophagy 2022, 18, 320–339. [Google Scholar] [CrossRef] [PubMed]
- Wu, A.H.; He, L.; Long, W.; Zhou, Q.; Zhu, S.; Wang, P.; Fan, S.; Wang, H. Novel Mechanisms of Herbal Therapies for Inhibiting HMGB1 Secretion or Action. Evid Based Complement Alternat Med 2015, 2015, 456305. [Google Scholar] [CrossRef] [PubMed]
- Semeraro, F.; Morescalchi, F.; Cancarini, A.; Russo, A.; Rezzola, S.; Costagliola, C. Diabetic Retinopathy, a Vascular and Inflammatory Disease: Therapeutic Implications. Diabetes Metab 2019, 45, 517–527. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




