Submitted:
12 January 2026
Posted:
21 January 2026
You are already at the latest version
Abstract
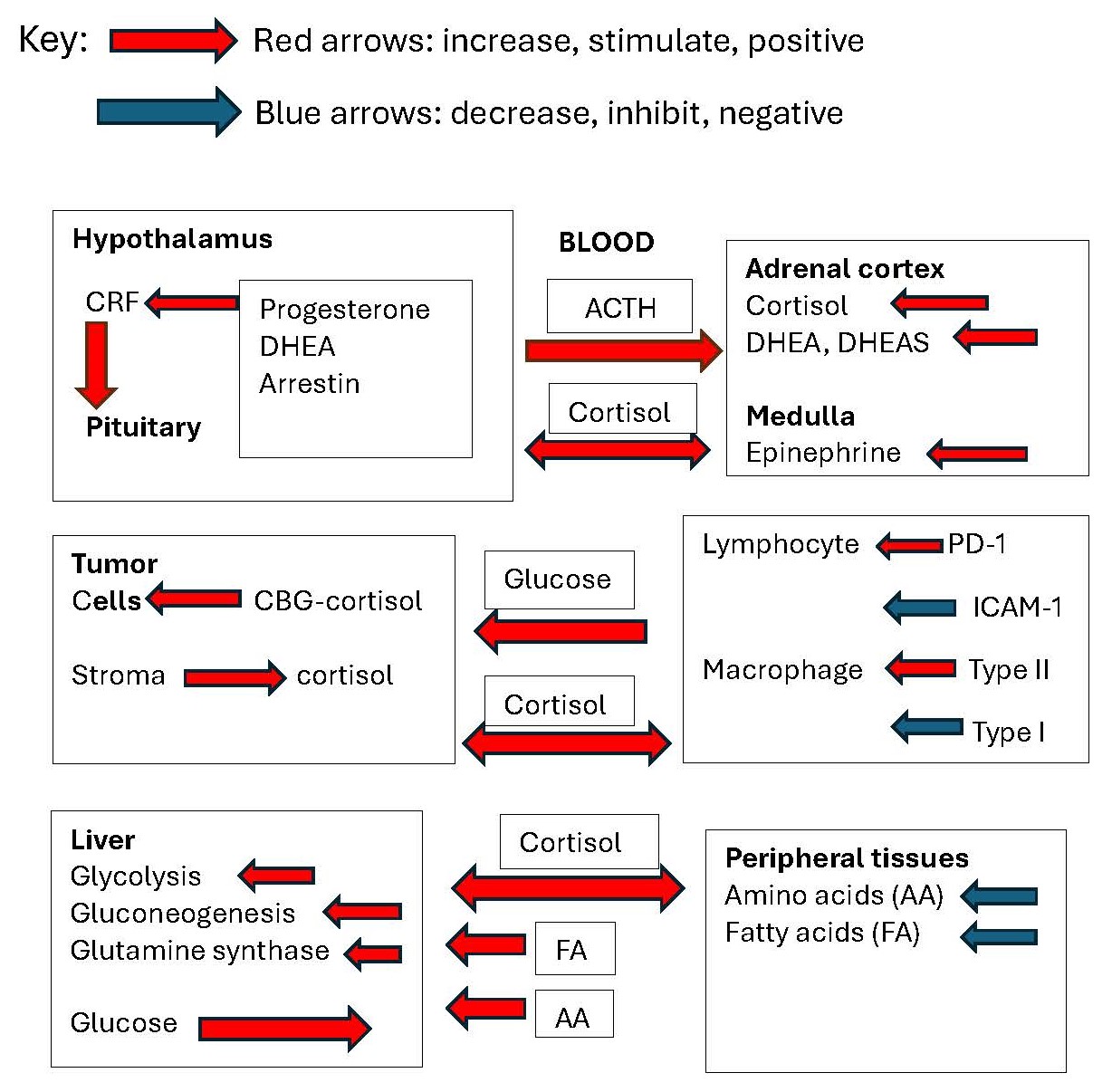
Keywords:
1. Formation and Secretion of Adrenocorticosteroids
1.1. Extra-Adrenal Sources of Cortisol
1.2. Stimuli for Activation of the Hypothalamic-Pituitary-Adrenal (HPA) Axis
1.3. Role of the 11-Hydroxysteroid Dehydrogenases (HSD11βs)
2. Dis-Regulation of Salivary and Serum Cortisol in Breast Cancer
2.1. Role of Ultradian Rhythms of Cortisol Secretion
2.2. Role of Obesity in Breast Cancer Progression
2.3. Associated Findings in Other Systems
3. Biochemical Activities of Cortisol
Cortisol Association with Illness
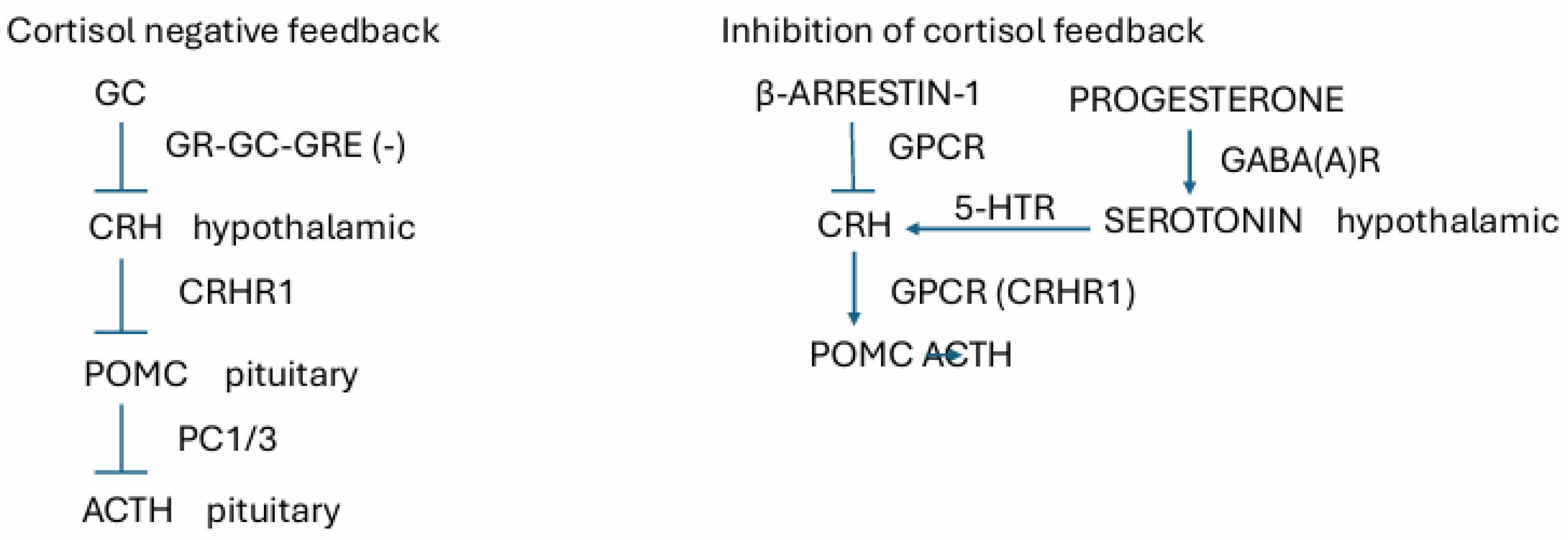
4. Suppression of Negative Feedback of Cortisol in the Hypothalamus
Reverse Effects of Stress on Cortisol Levels
5. Other Interacting Factors
5.1. Progesterone
5.2. Dehydroepiandrosterone (DHEA)
5.3. β-Arrestin
6. Mechanisms by Which Cortisol Modifies Cancer Risk
6.1. Cortisol Effects on Metabolism
6.2. Interaction of Cortisol with IGF-I
6.3. Potentiation of Stress Responses by Catecholamines
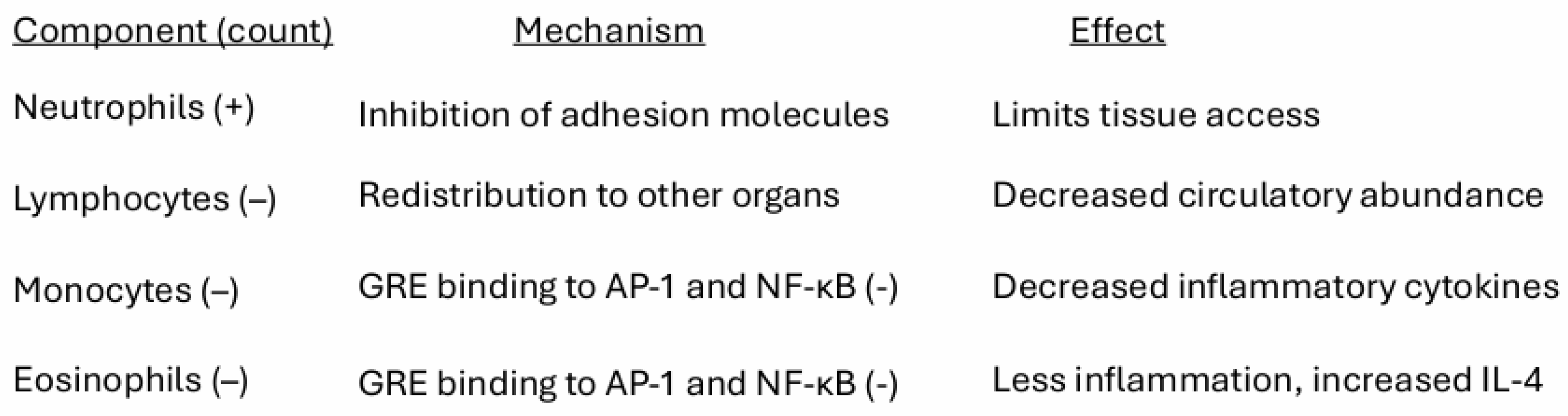
6.4. Effects of Cortisol on the Immune System
6.4.1. Effect of GC on Lymphocytes.
6.4.2. Effect of Cortisol on Tumor-Infiltrating Lymphocytes
6.5. The Tumor Microenvironment
Effects of Cortisol on Macrophages
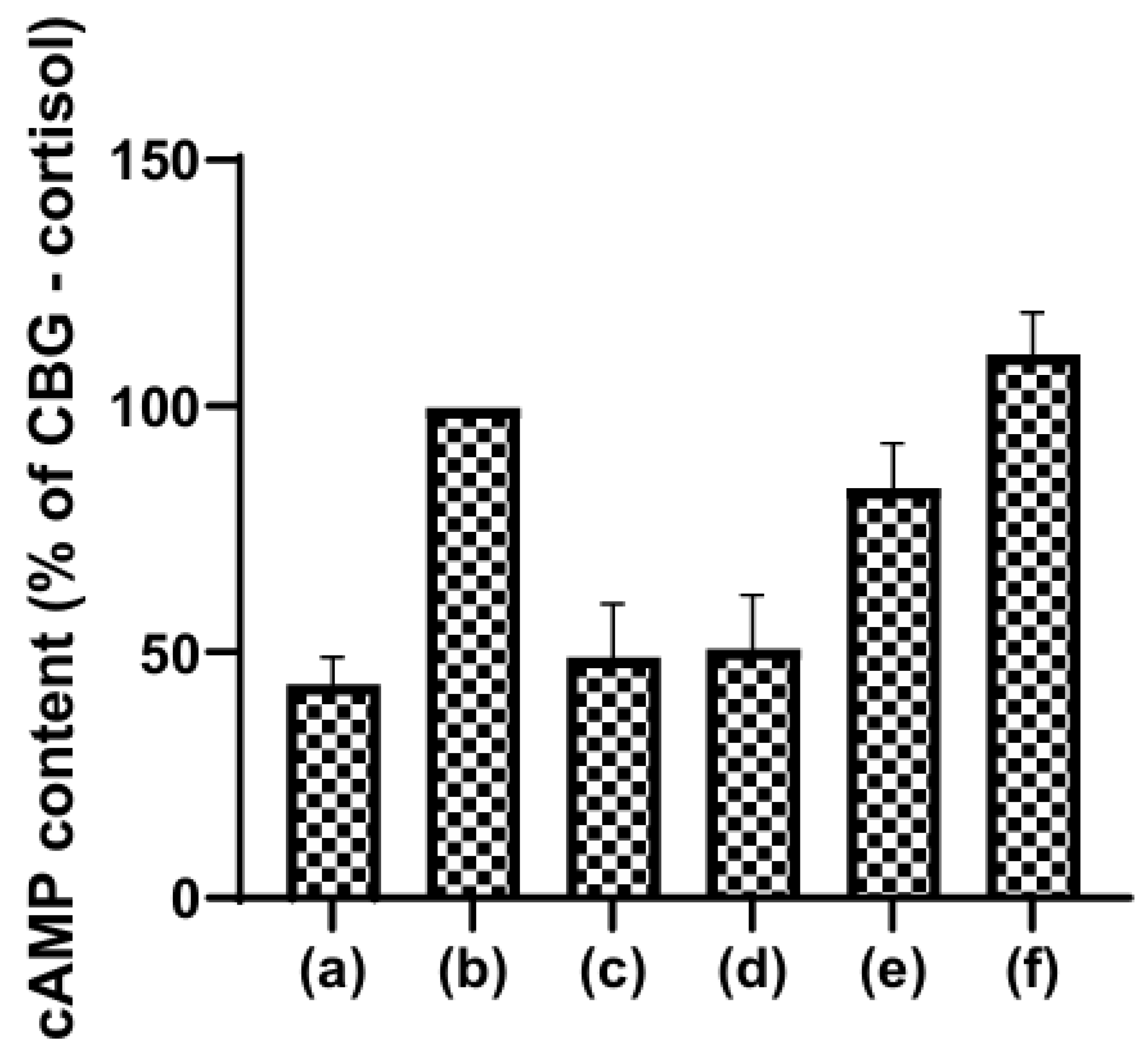
6.6. Cortisol Acting Through Corticosteroid Binding Globulin (CBG)
7. Cortisol vs DHEA as Risk Factors for Breast Cancer
8. Effects of Glucocorticoids in Cancer Therapy
8.1. Effect of Glucocorticoids on DNA Damage Response Therapy
8.2. Effect of Glucocorticoids on Immune Checkpoint Inhibitor Therapy
8.3. Effect of Glucocorticoids on Retinoid Drug Therapy
8.4. Effect of Glucocorticoids in PARP1-Related Drug Therapy
Summary
- Cortisol is regulated by input form the nervous system, and cortisol negative feedback is suppressed by feedback from a number of sources including DHEA and β-arrestin, and progesterone may stimulate ACTH secretion via stimulation of hypothalamic serotonin.
- Cortisol is produced in a number of organ sites in addition to the adrenal cortex in the body including the immune system, and peripheral activity is further regulated by 11β-oxidoreductases.
- Cortisol is often dis-regulated in metastatic breast cancer with increased variability and increased peripheral concentrations, and this is common to a number of other cancers and chronic disease states.
- Cortisol modifies metabolism in a manner that supports cancer by increasing glucose and glutamine availability, but is opposed by actions of IGF-I and DHEA.
- Cortisol bound to its intracellular receptor blocks transcription of Nf-κB and AP-1, reducing inflammatory cytokines and the antitumor immune activity. It also binds to membrane-bound CBG, activating adenylyl cyclase in T cells, suppressing proliferation.
- Cortisol is immunosuppressive, with specific effects on lymphocytes and macrophages. It also modifies the availability of immune cells to the tumor in part by suppressing ICAM-1 and by increasing the expression of immune checkpoint receptors such as PD-1 and Tim3 and promoting Treg cell proliferation.
- Cortisol is opposed by effects of DHEA and DHEA sulfate on metabolism and the immune system.
- Glucocorticoid agonists may provide benefit in retinoid therapy, PARP1 therapy, and therapy in ER+ cancers but not with therapy involving DNA damage in ER negative cancer or immune checkpoint blockade therapy.
References
- Kageyama, K.; Iwasaki, Y.; Daimon, M. Hypothalamic Regulation of Corticotropin-Releasing Factor under Stress and Stress Resilience. Int. J. Mol. Sci. 2021, 22, 12242. [Google Scholar] [CrossRef]
- Zhu, Y.; Wu, X.; Zhou, R.; Sie, O.; Niu, Z.; Wang, F.; Fang, Y. Hypothalamic-Pituitary-End-Organ Axes: Hormone Function in Female Patients with Major Depressive Disorder. Neurosci. Bull. 2021, 37, 1176–1187. [Google Scholar] [CrossRef]
- Engeland, W.C. Functional innervation of the adrenal cortex by the splanchnic nerve. Horm. Metab. Res. 1998, 30, 311–314. [Google Scholar] [CrossRef]
- Taves, M.D.; Gomez-Sanchez, C.E.; Soma, K.K. Extra-adrenal glucocorticoids and mineralocorticoids: Evidence for local synthesis, regulation, and function. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E11–E24. [Google Scholar] [CrossRef]
- Gomez-Sanchez, C.E.; Gomez-Sanchez, E.P. Extra-adrenal Glucocorticoid and Mineralocorticoid Biosynthesis. Endocrinology 2022, 163, bqac016. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Zbytek, B.; Pisarchik, A.; Slominski, R.M.; Zmijewski, M.A.; Wortsman, J. CRH functions as a growth factor/cytokine in the skin. J. Cell. Physiol. 2006, 206, 780–791. [Google Scholar] [CrossRef] [PubMed]
- Ottaviani, E.; Franchini, A.; Franceschi, C. Evolution of neuroendocrine thymus: Studies on POMC-derived peptides, cytokines and apoptosis in lower and higher vertebrates. J. Neuroimmunol. 1997, 72, 67–74. [Google Scholar] [CrossRef]
- Kishimoto, W.; Hiroi, T.; Shiraishi, M.; Osada, M.; Imaoka, S.; Kominami, S.; Igarashi, T.; Funae, Y. Cytochrome P450 2D catalyze steroid 21-hydroxylation in the brain. Endocrinology 2004, 145, 699–705. [Google Scholar] [CrossRef]
- Taylor, C.; Crosby, I.; Yip, V.; Maguire, P.; Pirmohamed, M.; Turner, R.M. A Review of the Important Role of CYP2D6 in Pharmacogenomics. Genes 2020, 11, 1295. [Google Scholar] [CrossRef] [PubMed]
- Briones, A.M.; Nguyen Dinh Cat, A.; Callera, G.E.; Yogi, A.; Burger, D.; He, Y.; Corrêa, J.W.; Gagnon, A.M.; Gomez-Sanchez, C.E.; Gomez-Sanchez, E.P.; et al. Adipocytes produce aldosterone through calcineurin-dependent signaling pathways: Implications in diabetes mellitus-associated obesity and vascular dysfunction. Hypertension 2012, 59, 1069–1078. [Google Scholar] [CrossRef]
- Phan, T.S.; Schink, L.; Mann, J.; Merk, V.M.; Zwicky, P.; Mundt, S.; Simon, D.; Kulms, D.; Abraham, S.; Legler, D.F.; et al. Keratinocytes control skin immune homeostasis through de novo-synthesized glucocorticoids. Sci. Adv. 2021, 7, eabe0337. [Google Scholar] [PubMed]
- Acharya, N.; Madi, A.; Zhang, H.; Klapholz, M.; Escobar, G.; Dulberg, S.; Christian, E.; Ferreira, M.; Dixon, K.O.; Fell, G.; et al. Endogenous Glucocorticoid Signaling Regulates CD8(+) T Cell Differentiation and Development of Dysfunction in the Tumor Microenvironment. Immunity 2020, 53, 658–671.e6. [Google Scholar]
- Parker, K.J.; Schatzberg, A.F.; Lyons, D.M. Neuroendocrine aspects of hypercortisolism in major depression. Horm. Behav. 2003, 43, 60–66. [Google Scholar] [CrossRef]
- Steckler, T.; Holsboer, F.; Reul, J.M. Glucocorticoids and depression. Baillieres Best Pract. Res. Clin. Endocrinol. Metab. 1999, 13, 597–614. [Google Scholar] [CrossRef]
- Tirabassi, G.; Boscaro, M.; Arnaldi, G. Harmful effects of functional hypercortisolism: A working hypothesis. Endocrine 2014, 46, 370–386. [Google Scholar] [PubMed]
- Ceccato, F.; Terzolo, M.; Scaroni, C. Who and how to screen for endogenous hypercortisolism in a high-risk population: A special issue of the journal of endocrinological investigations. J. Endocrinol. Investig. 2024, 48, 1–2. [Google Scholar] [CrossRef]
- Abraham, S.B.; Rubino, D.; Sinaii, N.; Ramsey, S.; Nieman, L.K. Cortisol, obesity, and the metabolic syndrome: A cross-sectional study of obese subjects and review of the literature. Obesity 2013, 21, E105–E117. [Google Scholar] [CrossRef] [PubMed]
- Zoli, A.; Lizzio, M.M.; Ferlisi, E.M.; Massafra, V.; Mirone, L.; Barini, A.; Scuderi, F.; Bartolozzi, F.; Magaró, M. ACTH, cortisol and prolactin in active rheumatoid arthritis. Clin. Rheumatol. 2002, 21, 289–293. [Google Scholar] [CrossRef]
- Masi, A.T.; Elmore, K.B.; Rehman, A.A.; Chatterton, R.T.; Goertzen, N.J.; Aldag, J.C. Lower Serum Androstenedione Levels in Pre-Rheumatoid Arthritis versus Normal Control Women: Correlations with Lower Serum Cortisol Levels. Autoimmune Dis. 2013, 2013, 593493. [Google Scholar] [CrossRef]
- Peeters, F.; Nicolson, N.A.; Berkhof, J. Levels and variability of daily life cortisol secretion in major depression. Psychiatry Res. 2004, 126, 1–13. [Google Scholar] [CrossRef]
- Cohen, L.; Cole, S.W.; Sood, A.K.; Prinsloo, S.; Kirschbaum, C.; Arevalo, J.M.; Jennings, N.B.; Scott, S.; Vence, L.; Wei, Q.; et al. Depressive symptoms and cortisol rhythmicity predict survival in patients with renal cell carcinoma: Role of inflammatory signaling. PLoS ONE 2012, 7, e42324. [Google Scholar] [CrossRef]
- Sapolsky, R.M.; Romero, L.M.; Munck, A.U. How do glucocorticoids influence stress responses? Integrating permissive, suppressive, stimulatory, and preparative actions. Endocr. Rev. 2000, 21, 55–89. [Google Scholar]
- Morgan, C.A., 3rd; Southwick, S.; Hazlett, G.; Rasmusson, A.; Hoyt, G.; Zimolo, Z.; Charney, D. Relationships among plasma dehydroepiandrosterone sulfate and cortisol levels, symptoms of dissociation, and objective performance in humans exposed to acute stress. Arch. Gen. Psychiatry 2004, 61, 819–825. [Google Scholar] [CrossRef]
- Balsalobre-Fernandez, C.; Tejero-Gonzalez, C.M.; del Campo-Vecino, J. Relationships between training load, salivary cortisol responses and performance during season training in middle and long distance runners. PLoS ONE 2014, 9, e106066. [Google Scholar] [CrossRef]
- Chatterton, R.T., Jr.; Vogelsong, K.M.; Lu, Y.C.; Hudgens, G.A. Hormonal responses to psychological stress in men preparing for skydiving. J. Clin. Endocrinol. Metab. 1997, 82, 2503–2509. [Google Scholar] [CrossRef] [PubMed]
- Topor, L.S.; Asai, M.; Dunn, J.; Majzoub, J.A. Cortisol stimulates secretion of dehydroepiandrosterone in human adrenocortical cells through inhibition of 3betaHSD2. J. Clin. Endocrinol. Metab. 2011, 96, E31–E39. [Google Scholar] [CrossRef] [PubMed]
- Charalampopoulos, I.; Dermitzaki, E.; Vardouli, L.; Tsatsanis, C.; Stournaras, C.; Margioris, A.N.; Gravanis, A. Dehydroepiandrosterone sulfate and allopregnanolone directly stimulate catecholamine production via induction of tyrosine hydroxylase and secretion by affecting actin polymerization. Endocrinology 2005, 146, 3309–3318. [Google Scholar] [CrossRef]
- Cadegiani, F.A.; Kater, C.E. Adrenal fatigue does not exist: A systematic review. BMC Endocr. Disord. 2016, 16, 48. [Google Scholar]
- Garbrecht, M.R.; Klein, J.M.; McCarthy, T.A.; Schmidt, T.J.; Krozowski, Z.S.; Snyder, J.M. 11-Beta hydroxysteroid dehydrogenase type 2 in human adult and fetal lung and its regulation by sex steroids. Pediatr. Res. 2007, 62, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Edwards, C.R.; Benediktsson, R.; Lindsay, R.S.; Seckl, J.R. 11 beta-Hydroxysteroid dehydrogenases: Key enzymes in determining tissue-specific glucocorticoid effects. Steroids 1996, 61, 263–269. [Google Scholar] [CrossRef]
- Kaur, K.; Hardy, R.; Ahasan, M.M.; Eijken, M.; van Leeuwen, J.P.; Filer, A.; Thomas, A.M.; Raza, K.; Buckley, C.D.; Stewart, P.M.; et al. Synergistic induction of local glucocorticoid generation by inflammatory cytokines and glucocorticoids: Implications for inflammation associated bone loss. Ann. Rheum. Dis. 2010, 69, 1185–1190. [Google Scholar] [CrossRef]
- Thieringer, R.; Le Grand, C.B.; Carbin, L.; Cai, T.Q.; Wong, B.; Wright, S.D.; Hermanowski-Vosatka, A. 11 Beta-hydroxysteroid dehydrogenase type 1 is induced in human monocytes upon differentiation to macrophages. J. Immunol. 2001, 167, 30–35. [Google Scholar]
- Gilmour, J.S.; Coutinho, A.E.; Cailhier, J.F.; Man, T.Y.; Clay, M.; Thomas, G.; Harris, H.J.; Mullins, J.J.; Seckl, J.R.; Savill, J.S.; et al. Local amplification of glucocorticoids by 11 beta-hydroxysteroid dehydrogenase type 1 promotes macrophage phagocytosis of apoptotic leukocytes. J. Immunol. 2006, 176, 7605–7611. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.Y.; Ding, X.; Daynes, R.A. The expression of 11 beta-hydroxysteroid dehydrogenase type I by lymphocytes provides a novel means for intracrine regulation of glucocorticoid activities. J. Immunol. 2005, 174, 879–889. [Google Scholar] [CrossRef]
- Freeman, L.; Hewison, M.; Hughes, S.V.; Evans, K.N.; Hardie, D.; Means, T.K.; Chakraverty, R. Expression of 11beta-hydroxysteroid dehydrogenase type 1 permits regulation of glucocorticoid bioavailability by human dendritic cells. Blood 2005, 106, 2042–2049. [Google Scholar] [CrossRef]
- Sephton, S.E.; Sapolsky, R.M.; Kraemer, H.C.; Spiegel, D. Diurnal cortisol rhythm as a predictor of breast cancer survival. J. Natl. Cancer Inst. 2000, 92, 994–1000. [Google Scholar] [CrossRef] [PubMed]
- Abercrombie, H.C.; Giese-Davis, J.; Sephton, S.; Epel, E.S.; Turner-Cobb, J.M.; Spiegel, D. Flattened cortisol rhythms in metastatic breast cancer patients. Psychoneuroendocrinology 2004, 29, 1082–1092. [Google Scholar] [CrossRef]
- Touitou, Y.; Bogdan, A.; Levi, F.; Benavides, M.; Auzeby, A. Disruption of the circadian patterns of serum cortisol in breast and ovarian cancer patients: Relationships with tumour marker antigens. Br. J. Cancer 1996, 74, 1248–1252. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Giskeodegard, G.F.; Skarra, S.; Engstrom, M.J.; Hagen, L.; Geisler, J.; Mikkola, T.S.; Tikkanen, M.J.; Debik, J.; Reidunsdatter, R.J.; et al. Association of serum cortisol and cortisone levels and risk of recurrence after endocrine treatment in breast cancer. Clin. Exp. Med. 2023, 23, 3883–3893. [Google Scholar] [CrossRef]
- Lightman, S.L.; Conway-Campbell, B.L. The crucial role of pulsatile activity of the HPA axis for continuous dynamic equilibration. Nat. Rev. Neurosci. 2010, 11, 710–718. [Google Scholar] [CrossRef]
- Zhao, H.; Wang, J.; Fang, D.; Lee, O.; Chatterton, R.T.; Stearns, V.; Khan, S.A.; Bulun, S.E. Adiposity Results in Metabolic and Inflammation Differences in Premenopausal and Postmenopausal Women Consistent with the Difference in Breast Cancer Risk. Horm. Cancer 2018, 9, 229–239. [Google Scholar] [CrossRef]
- Morris, P.G.; Hudis, C.A.; Giri, D.; Morrow, M.; Falcone, D.J.; Zhou, X.K.; Du, B.; Brogi, E.; Crawford, C.B.; Kopelovich, L.; et al. Inflammation and increased aromatase expression occur in the breast tissue of obese women with breast cancer. Cancer Prev. Res. 2011, 4, 1021–1029. [Google Scholar] [CrossRef]
- Scheinman, R.I.; Gualberto, A.; Jewell, C.M.; Cidlowski, J.A.; Baldwin, A.S., Jr. Characterization of mechanisms involved in transrepression of NF-kappa B by activated glucocorticoid receptors. Mol. Cell Biol. 1995, 15, 943–953. [Google Scholar] [CrossRef]
- Saini, J.; Singh, S.; Ebbehoj, A.; Zhang, C.D.; Nathani, R.; Fell, V.; Atkinson, E.; Achenbach, S.; Rivard, A.; Singh, R.; et al. Steroid Profiling and Circadian Cortisol Secretion in Patients With Mild Autonomous Cortisol Secretion: A Cross-sectional Study. J. Clin. Endocrinol. Metab. 2025, 110, 542–553. [Google Scholar] [CrossRef] [PubMed]
- Chatterton, R.T., Jr. Dehydroepiandrosterone (DHEA) in relation to breast cancer. Vitam. Horm. 2024, 129, 61–97. [Google Scholar] [PubMed]
- Mormont, M.C.; Levi, F. Circadian-system alterations during cancer processes: A review. Int. J. Cancer 1997, 70, 241–247. [Google Scholar] [CrossRef]
- Rosmond, R.; Dallman, M.F.; Bjorntorp, P. Stress-related cortisol secretion in men: Relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. J. Clin. Endocrinol. Metab. 1998, 83, 1853–1859. [Google Scholar] [CrossRef]
- Kronfol, Z.; Nair, M.; Zhang, Q.; Hill, E.E.; Brown, M.B. Circadian immune measures in healthy volunteers: Relationship to hypothalamic-pituitary-adrenal axis hormones and sympathetic neurotransmitters. Psychosom. Med. 1997, 59, 42–50. [Google Scholar] [CrossRef]
- Arendt, J. Shift work: Coping with the biological clock. Occup. Med. 2010, 60, 10–20. [Google Scholar] [CrossRef]
- Chandola, T.; Brunner, E.; Marmot, M. Chronic stress at work and the metabolic syndrome: Prospective study. BMJ 2006, 332, 521–525. [Google Scholar] [CrossRef]
- Buxton, O.M.; Marcelli, E. Short and long sleep are positively associated with obesity, diabetes, hypertension, and cardiovascular disease among adults in the United States. Soc. Sci. Med. 2010, 71, 1027–1036. [Google Scholar] [CrossRef]
- McEwen, B.S. Protective and damaging effects of stress mediators. N. Engl. J. Med. 1998, 338, 171–179. [Google Scholar] [CrossRef]
- Lee, J.H.; Meyer, E.J.; Nenke, M.A.; Falhammar, H.; Torpy, D.J. Corticosteroid-binding globulin (CBG): Spatiotemporal distribution of cortisol in sepsis. Trends Endocrinol. Metab. TEM 2023, 34, 181–190. [Google Scholar] [CrossRef]
- Salvador, E.; Shityakov, S.; Forster, C. Glucocorticoids and endothelial cell barrier function. Cell Tissue Res. 2014, 355, 597–605. [Google Scholar] [CrossRef]
- De Bosscher, K.; Desmet, S.J.; Clarisse, D.; Estebanez-Perpina, E.; Brunsveld, L. Nuclear receptor crosstalk—Defining the mechanisms for therapeutic innovation. Nat. Rev. Endocrinol. 2020, 16, 363–377. [Google Scholar] [CrossRef]
- Subramaniam, N.; Cairns, W.; Okret, S. Glucocorticoids repress transcription from a negative glucocorticoid response element recognized by two homeodomain-containing proteins, Pbx and Oct-1. J. Biol. Chem. 1998, 273, 23567–23574. [Google Scholar] [CrossRef]
- Verstrepen, L.; Bekaert, T.; Chau, T.L.; Tavernier, J.; Chariot, A.; Beyaert, R. TLR-4, IL-1R and TNF-R signaling to NF-kappaB: Variations on a common theme. Cell Mol. Life Sci. 2008, 65, 2964–2978. [Google Scholar] [CrossRef] [PubMed]
- Yang-Yen, H.F.; Chambard, J.C.; Sun, Y.L.; Smeal, T.; Schmidt, T.J.; Drouin, J.; Karin, M. Transcriptional interference between c-Jun and the glucocorticoid receptor: Mutual inhibition of DNA binding due to direct protein-protein interaction. Cell 1990, 62, 1205–1215. [Google Scholar] [CrossRef] [PubMed]
- Kuo, T.; McQueen, A.; Chen, T.C.; Wang, J.C. Regulation of Glucose Homeostasis by Glucocorticoids. Adv. Exp. Med. Biol. 2015, 872, 99–126. [Google Scholar] [PubMed]
- Vegiopoulos, A.; Herzig, S. Glucocorticoids, metabolism and metabolic diseases. Mol. Cell. Endocrinol. 2007, 275, 43–61. [Google Scholar] [CrossRef]
- Joels, M. Impact of glucocorticoids on brain function: Relevance for mood disorders. Psychoneuroendocrinology 2011, 36, 406–414. [Google Scholar] [CrossRef]
- Liston, C.; Cichon, J.M.; Jeanneteau, F.; Jia, Z.; Chao, M.V.; Gan, W.B. Circadian glucocorticoid oscillations promote learning-dependent synapse formation and maintenance. Nat. Neurosci. 2013, 16, 698–705. [Google Scholar] [CrossRef]
- Olejniczak, I.; Oster, H.; Ray, D.W. Glucocorticoid circadian rhythms in immune function. Semin. Immunopathol. 2022, 44, 153–163. [Google Scholar] [PubMed]
- Forster, C. Tight junctions and the modulation of barrier function in disease. Histochem. Cell Biol. 2008, 130, 55–70. [Google Scholar] [CrossRef]
- Shang, X.; Lin, X.; Alvarez, E.; Manorek, G.; Howell, S.B. Tight junction proteins claudin-3 and claudin-4 control tumor growth and metastases. Neoplasia 2012, 14, 974–985. [Google Scholar] [CrossRef] [PubMed]
- Runkle, E.A.; Mu, D. Tight junction proteins: From barrier to tumorigenesis. Cancer Lett. 2013, 337, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, A.E.; Chapman, K.E. The anti-inflammatory and immunosuppressive effects of glucocorticoids, recent developments and mechanistic insights. Mol. Cell Endocrinol. 2011, 335, 2–13. [Google Scholar] [CrossRef]
- Munck, A.; Naray-Fejes-Toth, A. Glucocorticoids and stress: Permissive and suppressive actions. Ann. N. Y Acad. Sci. 1994, 746, 115–130; discussion 31–33. [Google Scholar]
- De La Balze, F.A.; Reifenstein, E.C., Jr.; Albright, F. Differential blood counts in certain adrenal cortical disorders (Cushing’s syndrome, Addison’s disease and panhypopituitarism). J. Clin. Endocrinol. Metab. 1946, 6, 312–319. [Google Scholar] [CrossRef]
- Gjerstad, J.K.; Lightman, S.L.; Spiga, F. Role of glucocorticoid negative feedback in the regulation of HPA axis pulsatility. Stress 2018, 21, 403–416. [Google Scholar] [CrossRef]
- Dougherty, T.F.; White, A. Pituitary-adrenal cortical control of lymphocyte structure and function as revealed by experimental X-radiation. Endocrinology 1946, 39, 370–385. [Google Scholar] [CrossRef] [PubMed]
- Hechter, O.; Grossman, A.; Chatterton, R.T., Jr. Relationship of dehydroepiandrosterone and cortisol in disease. Med. Hypotheses 1997, 49, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Roelfsema, F.; van Heemst, D.; Iranmanesh, A.; Takahashi, P.; Yang, R.; Veldhuis, J.D. Impact of age, sex and body mass index on cortisol secretion in 143 healthy adults. Endocr. Connect. 2017, 6, 500–509. [Google Scholar] [CrossRef]
- Orentreich, N.; Brind, J.L.; Rizer, R.L.; Vogelman, J.H. Age changes and sex differences in serum dehydroepiandrosterone sulfate concentrations throughout adulthood. J. Clin. Endocrinol. Metab. 1984, 59, 551–555. [Google Scholar] [CrossRef]
- Parker, L.N.; Levin, E.R.; Lifrak, E.T. Evidence for adrenocortical adaptation to severe illness. J. Clin. Endocrinol. Metab. 1985, 60, 947–952. [Google Scholar] [CrossRef]
- Wynn, P.C.; Harwood, J.P.; Catt, K.J.; Aguilera, G. Corticotropin-releasing factor (CRF) induces desensitization of the rat pituitary CRF receptor-adenylate cyclase complex. Endocrinology 1988, 122, 351–358. [Google Scholar] [CrossRef]
- Beyer, H.S.; Matta, S.G.; Sharp, B.M. Regulation of the messenger ribonucleic acid for corticotropin-releasing factor in the paraventricular nucleus and other brain sites of the rat. Endocrinology 1988, 123, 2117–2123. [Google Scholar] [CrossRef]
- Oki, Y.; Peatman, T.W.; Qu, Z.C.; Orth, D.N. Effects of intracellular Ca2+ depletion and glucocorticoid on stimulated adrenocorticotropin release by rat anterior pituitary cells in a microperifusion system. Endocrinology 1991, 128, 1589–1596. [Google Scholar] [CrossRef]
- Spiegel, D.; Giese-Davis, J.; Taylor, C.B.; Kraemer, H. Stress sensitivity in metastatic breast cancer: Analysis of hypothalamic-pituitary-adrenal axis function. Psychoneuroendocrinology 2006, 31, 1231–1244. [Google Scholar] [CrossRef]
- Ceulemans, D.L.; Westenberg, H.G.; van Praag, H.M. The effect of stress on the dexamethasone suppression test. Psychiatry Res. 1985, 14, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Carroll, B.J.; Greden, J.F.; Feinberg, M.; Lohr, N.; James, N.M.; Steiner, M.; Haskett, R.F.; Albala, A.A.; DeVigne, J.-P.; Tarika, J. Neuroendocrine evaluation of depression in borderline patients. Psychiatr. Clin. North. Am. 1981, 4, 89–99. [Google Scholar]
- Meltzer, H.Y.; Fang, V.S.; Tricou, B.J.; Robertson, A.; Piyaka, S.K. Effect of dexamethasone on plasma prolactin and cortisol levels in psychiatric patients. Am. J. Psychiatry 1982, 139, 763–768. [Google Scholar] [CrossRef]
- Kalin, N.H.; Cohen, R.M.; Kraemer, G.W.; Risch, S.C.; Shelton, S.; Cohen, M.; McKinney, W.T.; Murphy, D.L. The dexamethasone suppression test as a measure of hypothalamic-pituitary feedback sensitivity and its relationship to behavioral arousal. Neuroendocrinology 1981, 32, 92–95. [Google Scholar] [CrossRef]
- van der Pompe, G.; Antoni, M.H.; Heijnen, C.J. Elevated basal cortisol levels and attenuated ACTH and cortisol responses to a behavioral challenge in women with metastatic breast cancer. Psychoneuroendocrinology 1996, 21, 361–374. [Google Scholar] [CrossRef]
- Miller, R.G.; Rubin, R.T.; Clark, B.R.; Crawford, W.R.; Arthur, R.J. The stress of aircraft carrier landings. Psychosom. Med. 1970, 32, 581–588. [Google Scholar] [CrossRef]
- Bourne, P.G.; Rose, R.M.; Mason, J.W. 17-OHCS levels in combat. Arch. Gen. Psychiatry 1968, 19, 135–140. [Google Scholar] [CrossRef] [PubMed]
- De Souza, E.B.; Van Loon, G.R. Stress-induced inhibition of the corticosterone response to a subsequent stress in rats: A nonadrenocorticotropin-mediated mechanism. Endocrinology 1982, 110, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Kontula, K.; Paavonen, T.; Luukkainen, T.; Andersson, L.C. Binding of progestins to the glucocorticoid receptor. Correlation to their glucocorticoid-like effects on in vitro functions of human mononuclear leukocytes. Biochem. Pharmacol. 1983, 32, 1511–1518. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, G.G.; Baxter, J.D.; Tomkins, G.M. Glucocorticoid receptors: Relations between steroid binding and biological effects. J. Mol. Biol. 1972, 67, 99–115. [Google Scholar] [CrossRef]
- Brunton, P.J. Resetting the dynamic range of hypothalamic-pituitary-adrenal axis stress responses through pregnancy. J. Neuroendocrinol. 2010, 22, 1198–1213. [Google Scholar] [CrossRef]
- Nierop, A.; Wirtz, P.H.; Bratsikas, A.; Zimmermann, R.; Ehlert, U. Stress-buffering effects of psychosocial resources on physiological and psychological stress response in pregnant women. Biol. Psychol. 2008, 78, 261–268. [Google Scholar] [CrossRef]
- Keller-Wood, M. ACTH responses to hypotension and feedback inhibition of ACTH increased by chronic progesterone treatment. Am. J. Physiol. 1998, 274, R81–R87. [Google Scholar] [CrossRef] [PubMed]
- Guennoun, R. Progesterone in the Brain: Hormone, Neurosteroid and Neuroprotectant. Int. J. Mol. Sci. 2020, 21, 5271. [Google Scholar] [CrossRef] [PubMed]
- Ladisich, W. Influence of progesterone on serotonin metabolism: A possible causal factor for mood changes. Psychoneuroendocrinology 1977, 2, 257–266. [Google Scholar] [CrossRef]
- Jorgensen, H.; Knigge, U.; Kjaer, A.; Moller, M.; Warberg, J. Serotonergic stimulation of corticotropin-releasing hormone and pro-opiomelanocortin gene expression. J. Neuroendocrinol. 2002, 14, 788–795. [Google Scholar] [CrossRef]
- Brown, G.P.; Courtney, G.A.; Marotta, S.F. A comparative study of adrenal progesterone secretion during the estrous cycles of hamsters and rats. Steroids 1976, 28, 283–294. [Google Scholar] [CrossRef]
- Herrera, A.Y.; Nielsen, S.E.; Mather, M. Stress-induced increases in progesterone and cortisol in naturally cycling women. Neurobiol. Stress 2016, 3, 96–104. [Google Scholar] [CrossRef]
- Childs, E.; Dlugos, A.; De Wit, H. Cardiovascular, hormonal, and emotional responses to the TSST in relation to sex and menstrual cycle phase. Psychophysiology 2010, 47, 550–559. [Google Scholar] [CrossRef] [PubMed]
- Baulieu, E.E. Androgens and aging men. Mol. Cell. Endocrinol. 2002, 198, 41–49. [Google Scholar] [CrossRef]
- Baulieu, E.E.; Thomas, G.; Legrain, S.; Lahlou, N.; Roger, M.; Debuire, B.; Faucounau, V.; Girard, L.; Hervy, M.-P.; Latour, F.; et al. Dehydroepiandrosterone (DHEA), DHEA sulfate, and aging: Contribution of the DHEAge Study to a sociobiomedical issue. Proc. Natl. Acad. Sci. USA 2000, 97, 4279–4284. [Google Scholar] [CrossRef]
- Yiallouris, A.; Tsioutis, C.; Agapidaki, E.; Zafeiri, M.; Agouridis, A.P.; Ntourakis, D.; Johnson, E.O. Adrenal Aging and Its Implications on Stress Responsiveness in Humans. Front. Endocrinol. 2019, 10, 54. [Google Scholar] [CrossRef]
- Greaves, R.F.; Wudy, S.A.; Badoer, E.; Zacharin, M.; Hirst, J.J.; Quinn, T.; Walker, D.W. A tale of two steroids: The importance of the androgens DHEA and DHEAS for early neurodevelopment. J. Steroid Biochem. Mol. Biol. 2019, 188, 77–85. [Google Scholar] [CrossRef]
- Solerte, S.B.; Fioravanti, M.; Vignati, G.; Giustina, A.; Cravello, L.; Ferrari, E. Dehydroepiandrosterone sulfate enhances natural killer cell cytotoxicity in humans via locally generated immunoreactive insulin-like growth factor I. J. Clin. Endocrinol. Metab. 1999, 84, 3260–3267. [Google Scholar] [CrossRef]
- Mirlekar, B.; Pylayeva-Gupta, Y. IL-12 Family Cytokines in Cancer and Immunotherapy. Cancers 2021, 13, 167. [Google Scholar] [CrossRef]
- Oakley, R.H.; Olivares-Reyes, J.A.; Hudson, C.C.; Flores-Vega, F.; Dautzenberg, F.M.; Hauger, R.L. Carboxyl-terminal and intracellular loop sites for CRF1 receptor phosphorylation and beta-arrestin-2 recruitment: A mechanism regulating stress and anxiety responses. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R209–R222. [Google Scholar] [CrossRef]
- Kohout, T.A.; Lefkowitz, R.J. Regulation of G protein-coupled receptor kinases and arrestins during receptor desensitization. Mol. Pharmacol. 2003, 63, 9–18. [Google Scholar] [CrossRef]
- Reiter, E.; Lefkowitz, R.J. GRKs and beta-arrestins: Roles in receptor silencing, trafficking and signaling. Trends Endocrinol. Metab. TEM 2006, 17, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Andryszkiewicz, W.; Gasiorowska, J.; Kubler, M.; Kublinska, K.; Palkiewicz, A.; Wiatkowski, A.; Szwedowicz, U.; Choromańska, A. Glucose Metabolism and Tumor Microenvironment: Mechanistic Insights and Therapeutic Implications. Int. J. Mol. Sci. 2025, 26, 1879. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.L.; Centrella, M.; Canalis, E. Cortisol inhibits the synthesis of insulin-like growth factor-I in skeletal cells. Endocrinology 1990, 126, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Chatterton, R.T., Jr.; Murray, C.L.; Hellman, L. Endocrine effects on leukocytopoiesis in the rat. I. Evidence for growth hormone secretion as the leukocytopoietic stimulus following acute cortisol-induced lymphopenia. Endocrinology 1973, 92, 775–787. [Google Scholar] [CrossRef]
- Gelato, M.C. Growth hormone-insulinlike growth factor I and immune function. Trends Endocrinol. Metab. TEM 1993, 4, 106–110. [Google Scholar] [CrossRef]
- Bozzola, M.; Valtorta, A.; Moretta, A.; Cisternino, M.; Biscaldi, I.; Schimpff, R.M. In vitro and in vivo effect of growth hormone on cytotoxic activity. J. Pediatr. 1990, 117, 596–599. [Google Scholar] [CrossRef]
- Geffner, M. Effects of growth hormone and insulin-like growth factor I on T- and B-lymphocytes and immune function. Acta Paediatr. Suppl. 1997, 423, 76–79. [Google Scholar] [CrossRef]
- Klug, S.T.; Ellestad, L.E.; Porter, T.E. Pituitary-Targeted Knockout of Glucocorticoid Receptors Disrupts Growth Hormone Expression During Embryonic Development. Endocrinology 2025, 166, bqaf119. [Google Scholar] [CrossRef]
- Basu, R.; Boguszewski, C.L.; Kopchick, J.J. Growth Hormone Action as a Target in Cancer: Significance, Mechanisms, and Possible Therapies. Endocr. Rev. 2025, 46, 224–280. [Google Scholar] [CrossRef]
- Betito, K.; Diorio, J.; Meaney, M.J.; Boksa, P. Adrenal phenylethanolamine N -methyltransferase induction in relation to glucocorticoid receptor dynamics: Evidence that acute exposure to high cortisol levels is sufficient to induce the enzyme. J. Neurochem. 1992, 58, 1853–1862. [Google Scholar] [CrossRef] [PubMed]
- Adameova, A.; Abdellatif, Y.; Dhalla, N.S. Role of the excessive amounts of circulating catecholamines and glucocorticoids in stress-induced heart disease. Can. J. Physiol. Pharmacol. 2009, 87, 493–514. [Google Scholar] [CrossRef]
- Hadcock, J.R.; Malbon, C.C. Regulation of beta-adrenergic receptors by “permissive” hormones: Glucocorticoids increase steady-state levels of receptor mRNA. Proc. Natl. Acad. Sci. USA 1988, 85, 8415–8419. [Google Scholar] [CrossRef]
- Fauci, A.S.; Dale, D.C.; Balow, J.E. Glucocorticosteroid therapy: Mechanisms of action and clinical considerations. Ann. Intern. Med. 1976, 84, 304–315. [Google Scholar] [CrossRef] [PubMed]
- Rhen, T.; Cidlowski, J.A. Antiinflammatory action of glucocorticoids--new mechanisms for old drugs. N. Engl. J. Med. 2005, 353, 1711–1723. [Google Scholar] [CrossRef] [PubMed]
- Ugor, E.; Prenek, L.; Pap, R.; Berta, G.; Ernszt, D.; Najbauer, J.; Németh, P.; Boldizsár, F.; Berki, T. Glucocorticoid hormone treatment enhances the cytokine production of regulatory T cells by upregulation of Foxp3 expression. Immunobiology 2018, 223, 422–431. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S.; Yamaguchi, T.; Nomura, T.; Ono, M. Regulatory T cells and immune tolerance. Cell 2008, 133, 775–787. [Google Scholar] [CrossRef]
- Michel, M.C.; Knapp, J.; Ratjen, H. Sensitization by dexamethasone of lymphocyte cyclic AMP formation: Evidence for increased function of the adenylyl cyclase catalyst. Br. J. Pharmacol. 1994, 113, 240–246. [Google Scholar] [CrossRef]
- Stork, P.J.; Schmitt, J.M. Crosstalk between cAMP and MAP kinase signaling in the regulation of cell proliferation. Trends Cell Biol. 2002, 12, 258–266. [Google Scholar] [CrossRef] [PubMed]
- van de Stolpe, A.; Caldenhoven, E.; Stade, B.G.; Koenderman, L.; Raaijmakers, J.A.; Johnson, J.P.; van der Saag, P. 12-O-tetradecanoylphorbol-13-acetate- and tumor necrosis factor alpha-mediated induction of intercellular adhesion molecule-1 is inhibited by dexamethasone. Functional analysis of the human intercellular adhesion molecular-1 promoter. J. Biol. Chem. 1994, 269, 6185–6192. [Google Scholar] [CrossRef]
- Liden, J.; Rafter, I.; Truss, M.; Gustafsson, J.A.; Okret, S. Glucocorticoid effects on NF-kappaB binding in the transcription of the ICAM-1 gene. Biochem. Biophys. Res. Commun. 2000, 273, 1008–1014. [Google Scholar] [CrossRef]
- Boxman, I.L.; Ruwhof, C.; Boerman, O.C.; Lowik, C.W.; Ponec, M. Role of fibroblasts in the regulation of proinflammatory interleukin IL-1, IL-6 and IL-8 levels induced by keratinocyte-derived IL-1. Arch. Dermatol. Res. 1996, 288, 391–398. [Google Scholar] [CrossRef]
- Postlethwaite, A.E.; Lachman, L.B.; Kang, A.H. Induction of fibroblast proliferation by interleukin-1 derived from human monocytic leukemia cells. Arthritis Rheum. 1984, 27, 995–1001. [Google Scholar] [CrossRef]
- Schmidt, J.A.; Mizel, S.B.; Cohen, D.; Green, I. Interleukin 1, a potential regulator of fibroblast proliferation. J. Immunol. 1982, 128, 2177–2182. [Google Scholar] [CrossRef] [PubMed]
- Taher, M.Y.; Davies, D.M.; Maher, J. The role of the interleukin (IL)-6/IL-6 receptor axis in cancer. Biochem. Soc. Trans. 2018, 46, 1449–1462. [Google Scholar] [CrossRef]
- Waugh, D.J.; Wilson, C. The interleukin-8 pathway in cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2008, 14, 6735–6741. [Google Scholar] [CrossRef]
- Finak, G.; Bertos, N.; Pepin, F.; Sadekova, S.; Souleimanova, M.; Zhao, H.; Chen, H.; Omeroglu, G.; Meterissian, S.; Omeroglu, A.; et al. Stromal gene expression predicts clinical outcome in breast cancer. Nat. Med. 2008, 14, 518–527. [Google Scholar] [CrossRef]
- Smith, R.A.; Lea, R.A.; Weinstein, S.R.; Griffiths, L.R. Progesterone, glucocorticoid, but not estrogen receptor mRNA is altered in breast cancer stroma. Cancer Lett. 2007, 255, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Ray, A.; Prefontaine, K.E. Physical association and functional antagonism between the p65 subunit of transcription factor NF-kappa B and the glucocorticoid receptor. Proc. Natl. Acad. Sci. USA 1994, 91, 752–756. [Google Scholar] [CrossRef] [PubMed]
- Ayroldi, E.; Macchiarulo, A.; Riccardi, C. Targeting glucocorticoid side effects: Selective glucocorticoid receptor modulator or glucocorticoid-induced leucine zipper? A perspective. FASEB J. 2014, 28, 5055–5070. [Google Scholar] [CrossRef]
- Singer, M.; Wang, C.; Cong, L.; Marjanovic, N.D.; Kowalczyk, M.S.; Zhang, H.; Nyman, J.; Sakuishi, K.; Kurtulus, S.; Gennert, D.; et al. A Distinct Gene Module for Dysfunction Uncoupled from Activation in Tumor-Infiltrating T Cells. Cell 2016, 166, 1500–1511.e9. [Google Scholar] [CrossRef]
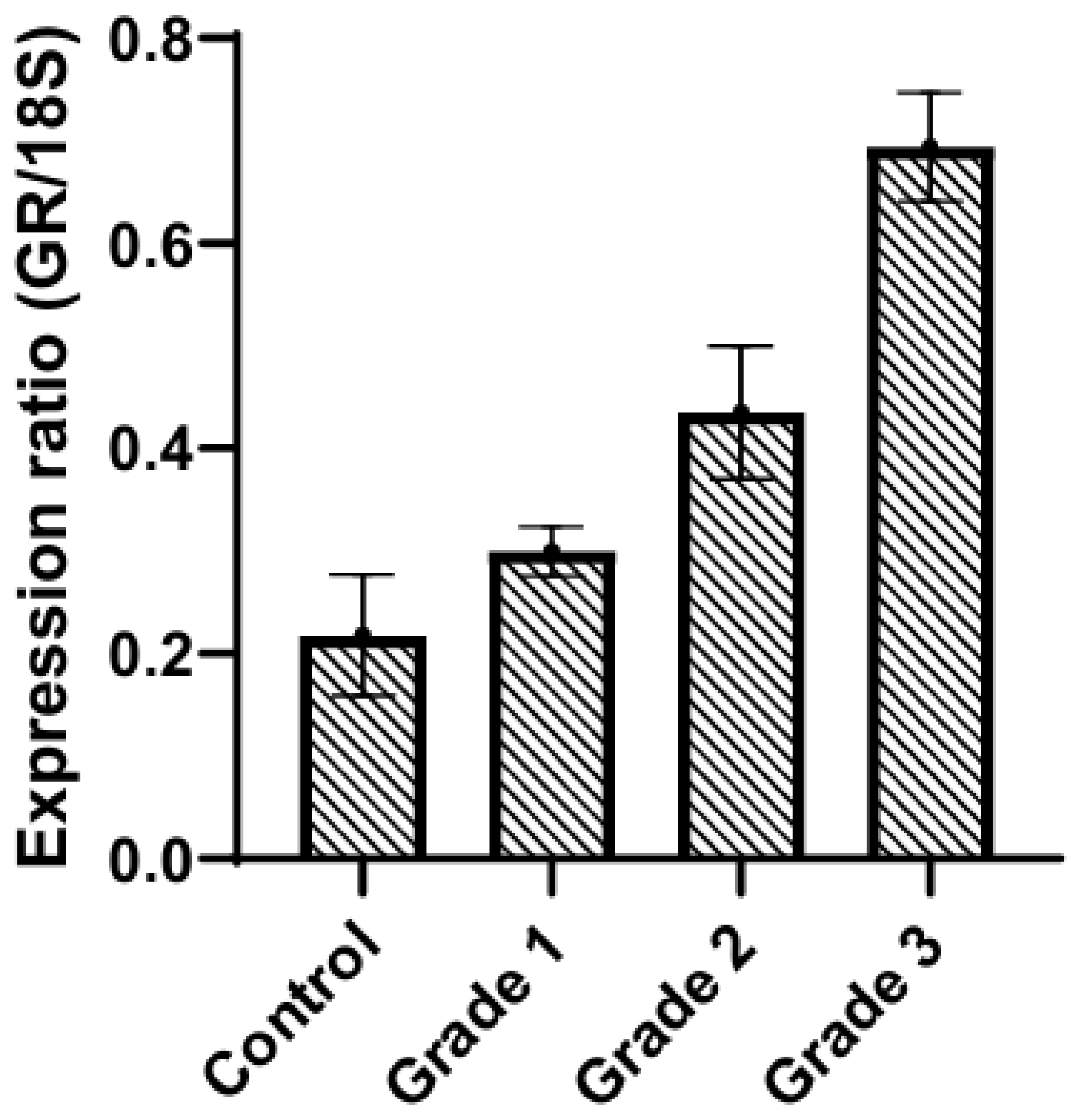
- Matossian, M.D.; Shiang, C.; Dolcen, D.N.; Dreyer, M.; Hatogai, K.; Hall, K.; Saha, P.; Biernacka, A.; Sweis, R.F.; Karrison, T.; et al. High tumor glucocorticoid receptor expression in early-stage, triple-negative breast cancer is associated with increased T-regulatory cell infiltration. Breast Cancer Res. Treat. 2025, 209, 563–572. [Google Scholar] [CrossRef]
- Tonsing-Carter, E.; Hernandez, K.M.; Kim, C.R.; Harkless, R.V.; Oh, A.; Bowie, K.R.; West-Szymanski, D.C.; Betancourt-Ponce, M.A.; Green, B.D.; Lastra, R.R.; et al. Glucocorticoid receptor modulation decreases ER-positive breast cancer cell proliferation and suppresses wild-type and mutant ER chromatin association. Breast Cancer Res. 2019, 21, 82. [Google Scholar] [CrossRef] [PubMed]
- Kelly, W.J.; Gilbert, M.R. Glucocorticoids and immune checkpoint inhibitors in glioblastoma. J. Neurooncol. 2021, 151, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Rico, S.; Rosenberg, D.; Elkayam, O.; Gertel, S. Implications of glucocorticoid-induced co-inhibitory molecule expression on combined glucocorticoid and immune checkpoint inhibitor therapy. Immunopharmacol Immunotoxicol. 2025, 47, 599–610. [Google Scholar] [CrossRef]
- Oishi, S.; Takano, R.; Tamura, S.; Tani, S.; Iwaizumi, M.; Hamaya, Y.; Takagaki, K.; Nagata, T.; Seto, S.; Horii, T.; et al. M2 polarization of murine peritoneal macrophages induces regulatory cytokine production and suppresses T-cell proliferation. Immunology 2016, 149, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Snyder, D.S.; Unanue, E.R. Corticosteroids inhibit murine macrophage Ia expression and interleukin 1 production. J. Immunol. 1982, 129, 1803–1805. [Google Scholar] [CrossRef] [PubMed]
- Musa, B.U.; Seal, U.S.; Doe, R.P.; Lamusga, R.F.; Lewis, M.D.; Glaser, R. Elevation of certain plasma proteins in man following estrogen administration: A dose-response relationship. J. Clin. Endocrinol. Metab. 1965, 25, 1163–1166. [Google Scholar] [CrossRef]
- Levin, N.; Akana, S.F.; Jacobson, L.; Kuhn, R.W.; Siiteri, P.K.; Dallman, M.F. Plasma adrenocorticotropin is more sensitive than transcortin production or thymus weight to inhibition by corticosterone in rats. Endocrinology 1987, 121, 1104–1110. [Google Scholar] [CrossRef]
- Rosner, W.; Aden, D.P.; Khan, M.S. Hormonal influences on the secretion of steroid-binding proteins by a human hepatoma-derived cell line. J. Clin. Endocrinol. Metab. 1984, 59, 806–808. [Google Scholar] [CrossRef]
- Mihrshahi, R.; Lewis, J.G.; Ali, S.O. Hormonal effects on the secretion and glycoform profile of corticosteroid-binding globulin. J. Steroid Biochem. Mol. Biol. 2006, 101, 275–285. [Google Scholar] [CrossRef]
- Bartalena, L.; Hammond, G.L.; Farsetti, A.; Flink, I.L.; Robbins, J. Interleukin-6 inhibits corticosteroid-binding globulin synthesis by human hepatoblastoma-derived (Hep G2) cells. Endocrinology 1993, 133, 291–296. [Google Scholar] [CrossRef]
- Maitra, U.S.; Khan, M.S.; Rosner, W. Corticosteroid-binding globulin receptor of the rat hepatic membrane: Solubilization, partial characterization, and the effect of steroids on binding. Endocrinology 1993, 133, 1817–1822. [Google Scholar] [CrossRef]
- Nakhla, A.M.; Khan, M.S.; Rosner, W. Induction of adenylate cyclase in a mammary carcinoma cell line by human corticosteroid-binding globulin. Biochem. Biophys. Res. Commun. 1988, 153, 1012–1018. [Google Scholar] [CrossRef]
- Adamson, R.H.; Liu, B.; Fry, G.N.; Rubin, L.L.; Curry, F.E. Microvascular permeability and number of tight junctions are modulated by cAMP. Am. J. Physiol. 1998, 274, H1885–H1894. [Google Scholar] [CrossRef] [PubMed]
- Ruddle, N.H.; Akirav, E.M. Secondary lymphoid organs: Responding to genetic and environmental cues in ontogeny and the immune response. J. Immunol. 2009, 183, 2205–2212. [Google Scholar] [CrossRef]
- Grader-Beck, T.; van Puijenbroek, A.A.; Nadler, L.M.; Boussiotis, V.A. cAMP inhibits both Ras and Rap1 activation in primary human T lymphocytes, but only Ras inhibition correlates with blockade of cell cycle progression. Blood 2003, 101, 998–1006. [Google Scholar] [CrossRef]
- Eigler, A.; Siegmund, B.; Emmerich, U.; Baumann, K.H.; Hartmann, G.; Endres, S. Anti-inflammatory activities of cAMP-elevating agents: Enhancement of IL-10 synthesis and concurrent suppression of TNF production. J. Leukoc. Biol. 1998, 63, 101–107. [Google Scholar] [CrossRef]
- Chatterton, R.T. Functions of dehydroepiandrosterone in relation to breast cancer. Steroids 2022, 179, 108970. [Google Scholar] [CrossRef] [PubMed]
- Sriramulu, S.; Thoidingjam, S.; Brown, S.L.; Siddiqui, F.; Movsas, B.; Nyati, S. Molecular targets that sensitize cancer to radiation killing: From the bench to the bedside. Biomed Pharmacother. 2023, 158, 114126. [Google Scholar] [CrossRef]
- Berry, M.R.; Fan, T.M. Target-Based Radiosensitization Strategies: Concepts and Companion Animal Model Outlook. Front. Oncol. 2021, 11, 768692. [Google Scholar] [CrossRef]
- Kvols, L.K. Radiation sensitizers: A selective review of molecules targeting DNA and non-DNA targets. J. Nucl. Med. 2005, 46, 187S–190S. [Google Scholar] [PubMed]
- Flint, M.S.; Baum, A.; Chambers, W.H.; Jenkins, F.J. Induction of DNA damage, alteration of DNA repair and transcriptional activation by stress hormones. Psychoneuroendocrinology 2007, 32, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Flaherty, R.L.; Owen, M.; Fagan-Murphy, A.; Intabli, H.; Healy, D.; Patel, A.; Allen, M.C.; Patel, B.A.; Flint, M.S. Glucocorticoids induce production of reactive oxygen species/reactive nitrogen species and DNA damage through an iNOS mediated pathway in breast cancer. Breast Cancer Res. 2017, 19, 35. [Google Scholar] [CrossRef]
- West, D.C.; Kocherginsky, M.; Tonsing-Carter, E.Y.; Dolcen, D.N.; Hosfield, D.J.; Lastra, R.R.; Sinnwell, J.P.; Thompson, K.J.; Bowie, K.R.; Harkless, R.V.; et al. Discovery of a Glucocorticoid Receptor (GR) Activity Signature Using Selective GR Antagonism in ER-Negative Breast Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2018, 24, 3433–3446. [Google Scholar] [CrossRef]
- Porter, B.A.; Frerich, C.; Laine, M.; Clark, A.B.; Durdana, I.; Lee, J.; Taya, M.; Sahoo, S.; Greene, G.L.; Bennett, L.; et al. Glucocorticoid Receptor Activation in Lobular Breast Cancer Is Associated with Reduced Cell Proliferation and Promotion of Metastases. Cancers 2023, 15, 4679. [Google Scholar] [CrossRef]
- Danilo, M.; Chennupati, V.; Silva, J.G.; Siegert, S.; Held, W. Suppression of Tcf1 by Inflammatory Cytokines Facilitates Effector CD8 T Cell Differentiation. Cell Rep. 2018, 22, 2107–2117. [Google Scholar] [CrossRef] [PubMed]
- Wherry, E.J.; Kurachi, M. Molecular and cellular insights into T cell exhaustion. Nat. Rev. Immunol. 2015, 15, 486–499. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.T.; Anderson, A.C.; Tan, W.G.; West, E.E.; Ha, S.J.; Araki, K.; Freeman, G.J.; Kuchroo, V.K.; Ahmed, R. Cooperation of Tim-3 and PD-1 in CD8 T-cell exhaustion during chronic viral infection. Proc. Natl. Acad. Sci. USA 2010, 107, 14733–14738. [Google Scholar] [CrossRef] [PubMed]
- den Hollander, P.; Savage, M.I.; Brown, P.H. Targeted therapy for breast cancer prevention. Front. Oncol. 2013, 3, 250. [Google Scholar] [CrossRef]
- Hidalgo, A.A.; Trump, D.L.; Johnson, C.S. Glucocorticoid regulation of the vitamin D receptor. J. Steroid Biochem. Mol. Biol. 2010, 121, 372–375. [Google Scholar] [CrossRef]
- Bagnoud, M.; Remlinger, J.; Massy, M.; Lodygin, D.; Salmen, A.; Chan, A.; Lühder, F.; Hoepner, R. In Vivo and In Vitro Evidence for an Interplay between the Glucocorticoid Receptor and the Vitamin D Receptor Signaling. Cells 2023, 12, 2291. [Google Scholar] [CrossRef]
- Sartorius, C.A.; Takimoto, G.S.; Richer, J.K.; Tung, L.; Horwitz, K.B. Association of the Ku autoantigen/DNA-dependent protein kinase holoenzyme and poly(ADP-ribose) polymerase with the DNA binding domain of progesterone receptors. J. Mol. Endocrinol. 2000, 24, 165–182. [Google Scholar] [CrossRef]
- Kraus, W.L.; Lis, J.T. PARP goes transcription. Cell 2003, 113, 677–683. [Google Scholar] [CrossRef]
- Kraus, W.L. Transcriptional control by PARP-1: Chromatin modulation, enhancer-binding, coregulation, and insulation. Curr. Opin. Cell Biol. 2008, 20, 294–302. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, J.; Li, H.; Chen, L.; Yao, D. Dual-target inhibitors of PARP1 in cancer therapy: A drug discovery perspective. Drug Discov. Today 2023, 28, 103607. [Google Scholar] [CrossRef] [PubMed]
- Mitre-Aguilar, I.B.; Barrios-Garcia, T.; Ruiz-Lopez, V.M.; Cabrera-Quintero, A.J.; Mejia-Dominguez, N.R.; Ventura-Gallegos, J.L.; Moreno-Mitre, D.; Aranda-Gutierrez, A.; Mejia-Rangel, J.; Escalona-Guzman, A.R.; et al. Glucocorticoid-dependent expression of IAP participates in the protection against TNF-mediated cytotoxicity in MCF7 cells. BMC Cancer 2019, 19, 356. [Google Scholar] [CrossRef]
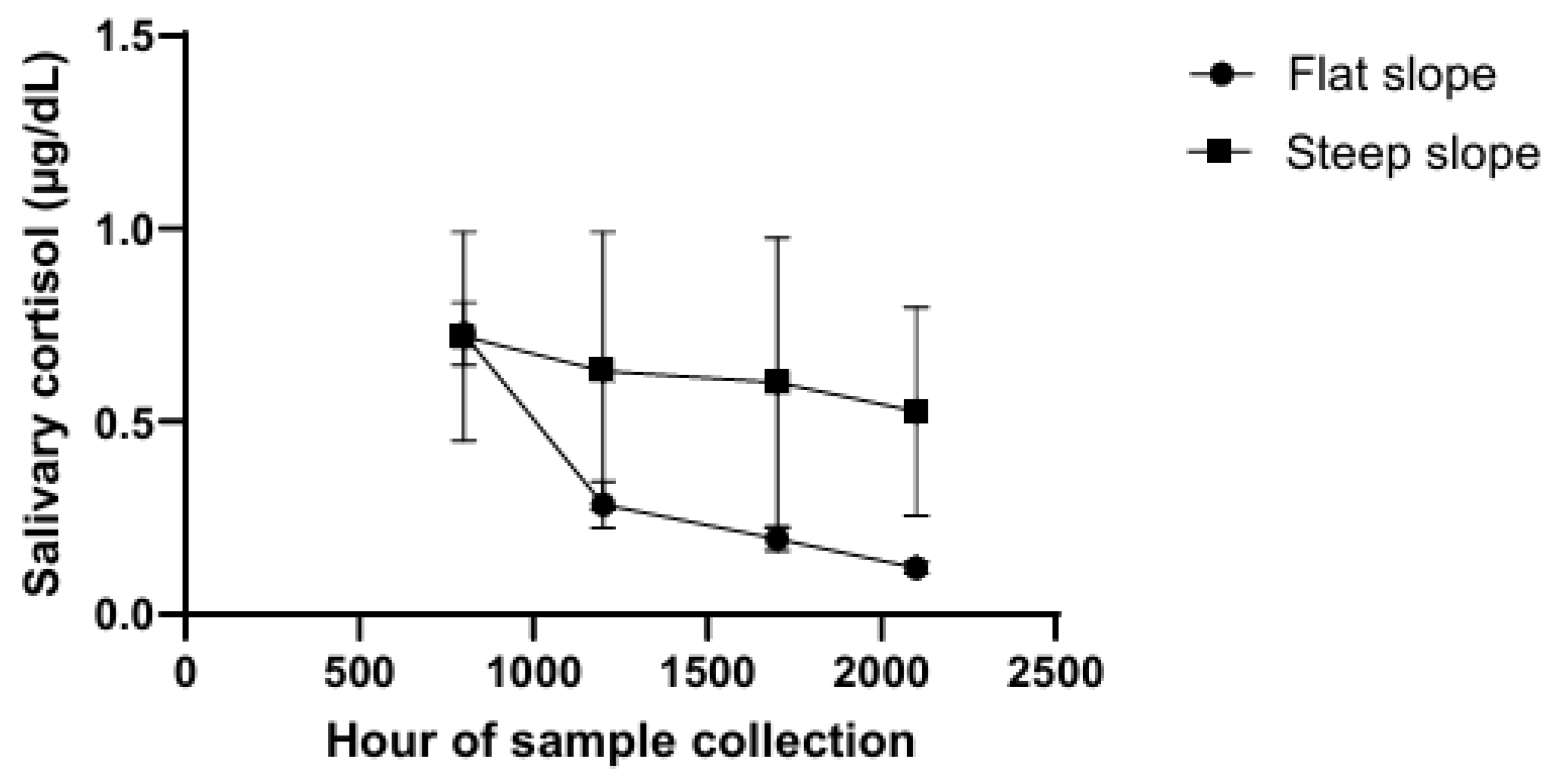
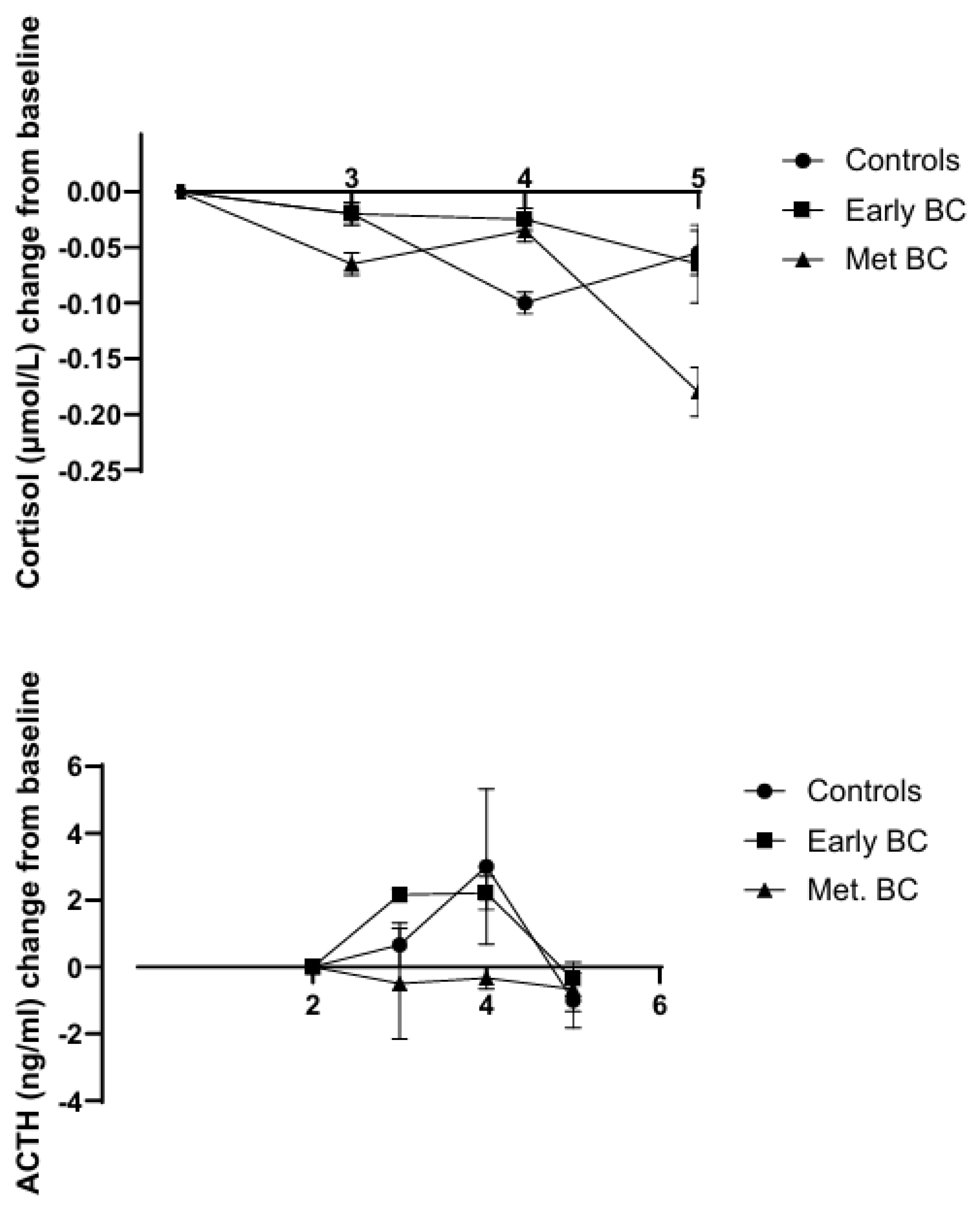
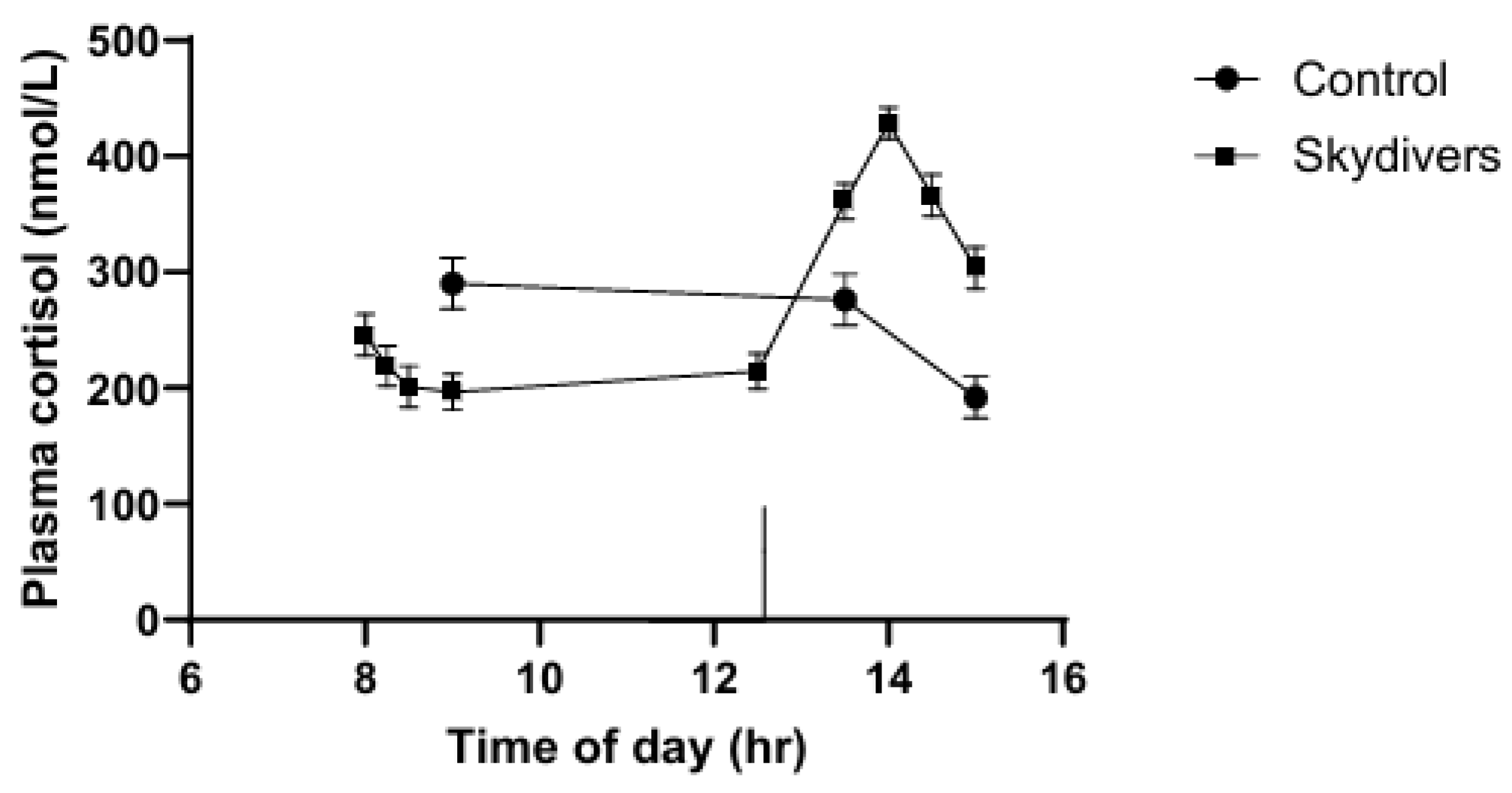
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



