Submitted:
12 January 2026
Posted:
20 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Data Source and Study Selection
2.2. Amplicon Processing and Taxonomic Assignment
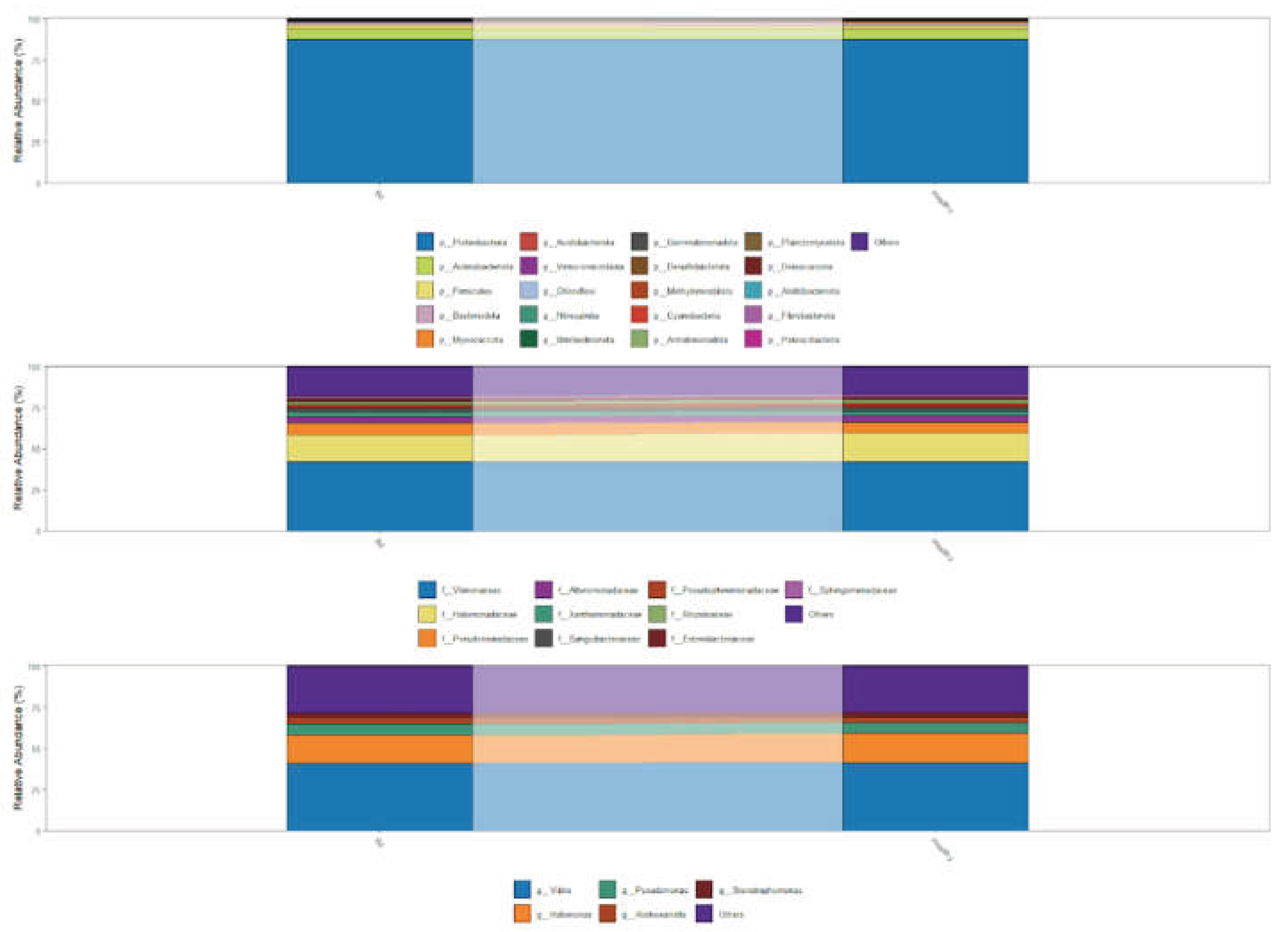
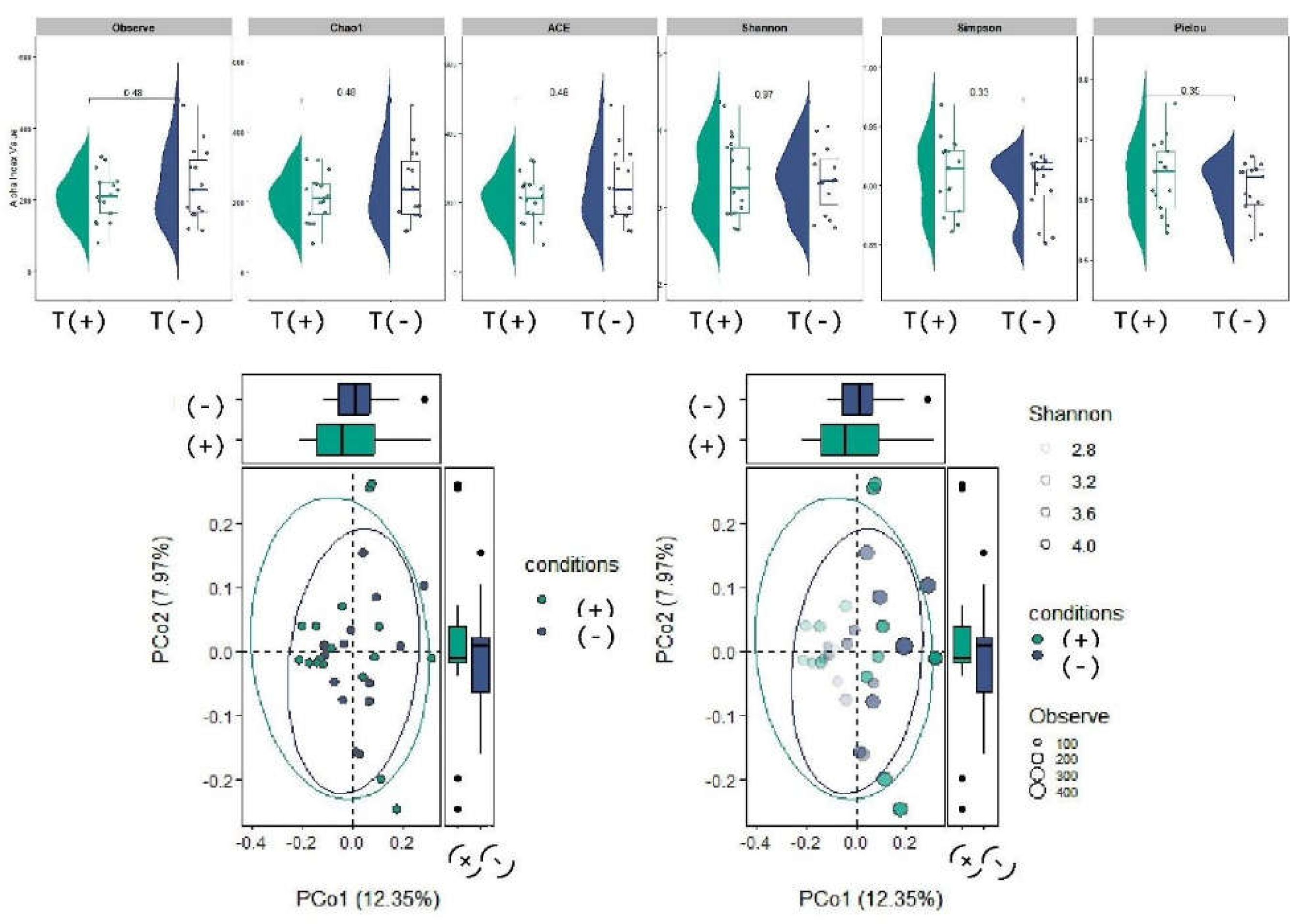
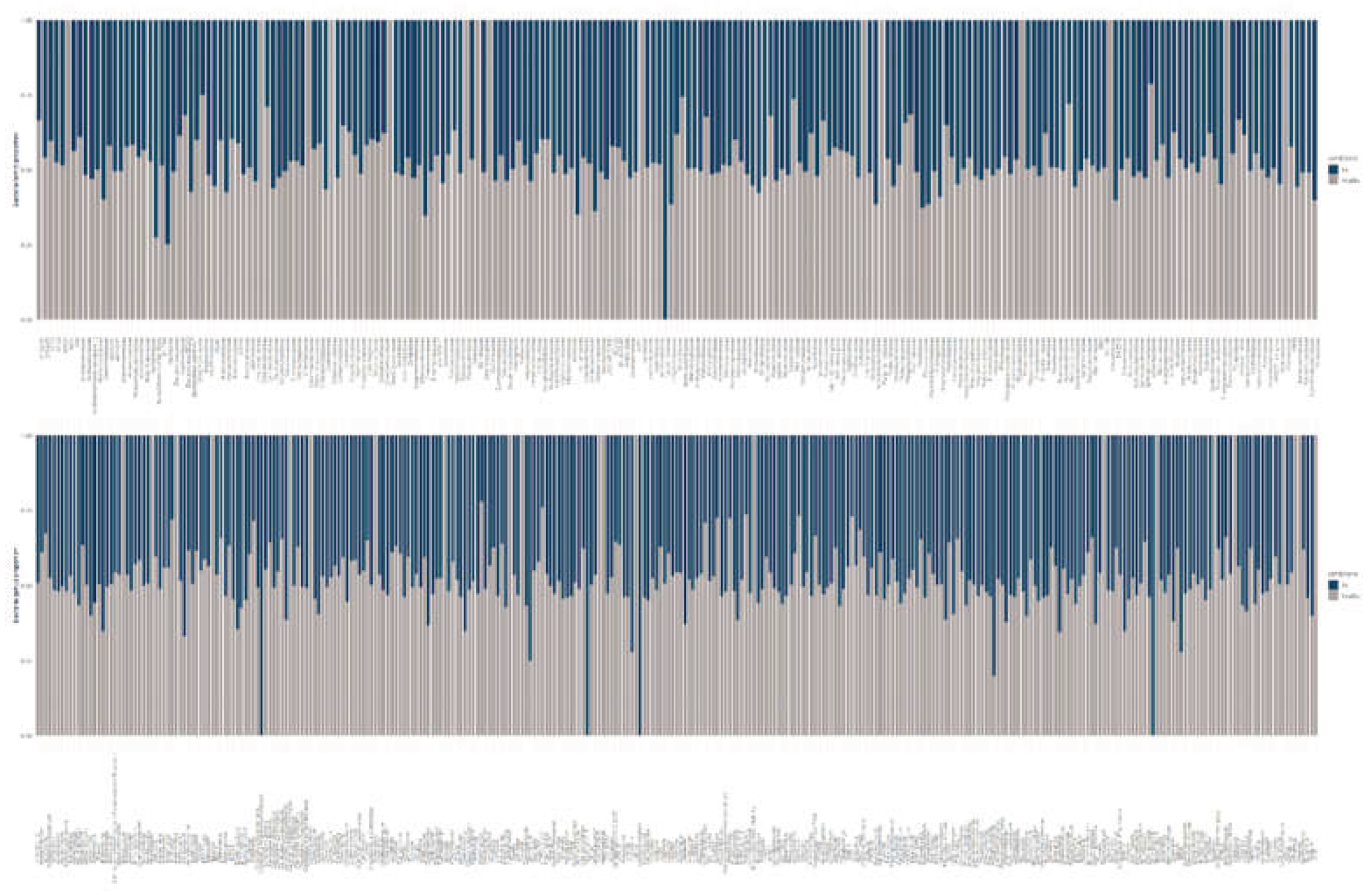
2.3. Community Composition and Diversity Analyses
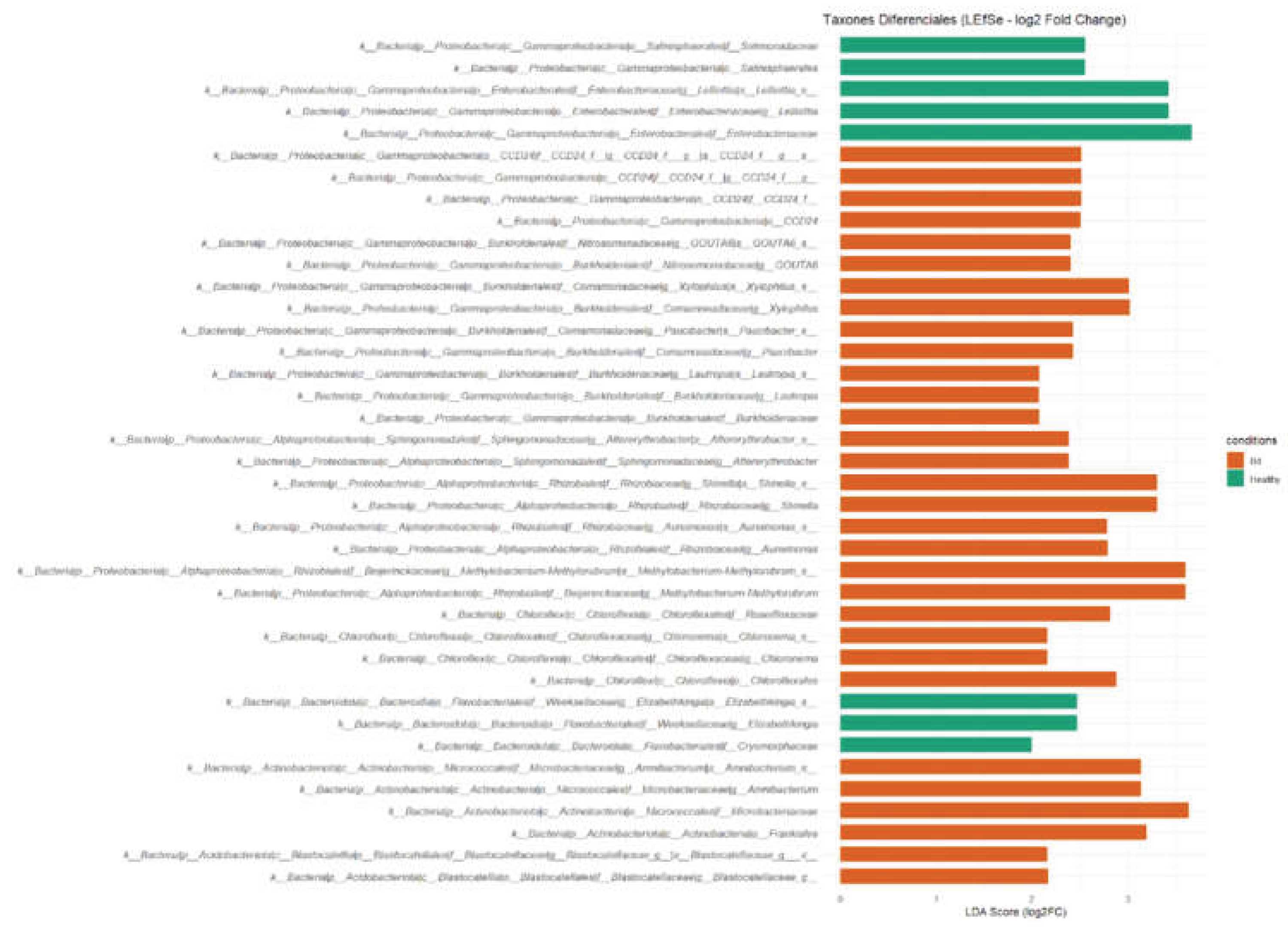
2.4. Differential Abundance and Biomarker Discovery
2.5. Microbial Association Network Inference
2.6. Network Topology and Robustness Analyses
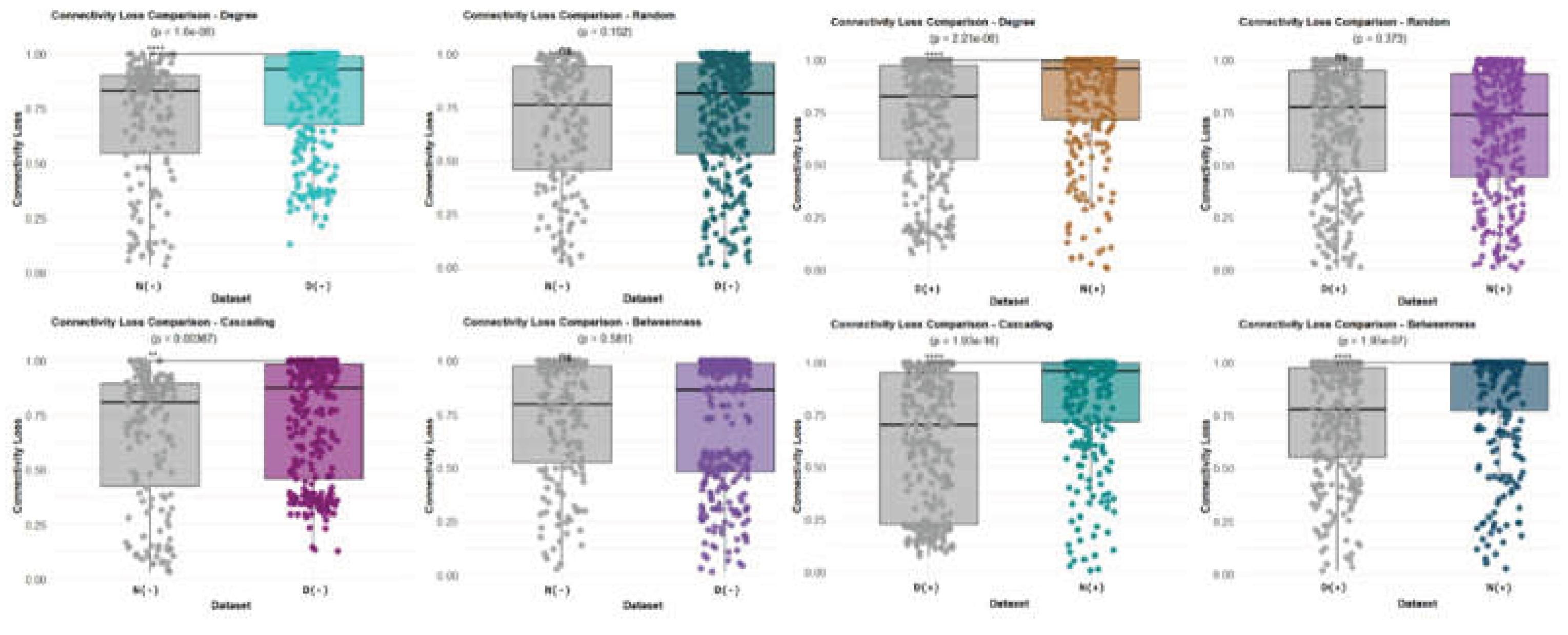
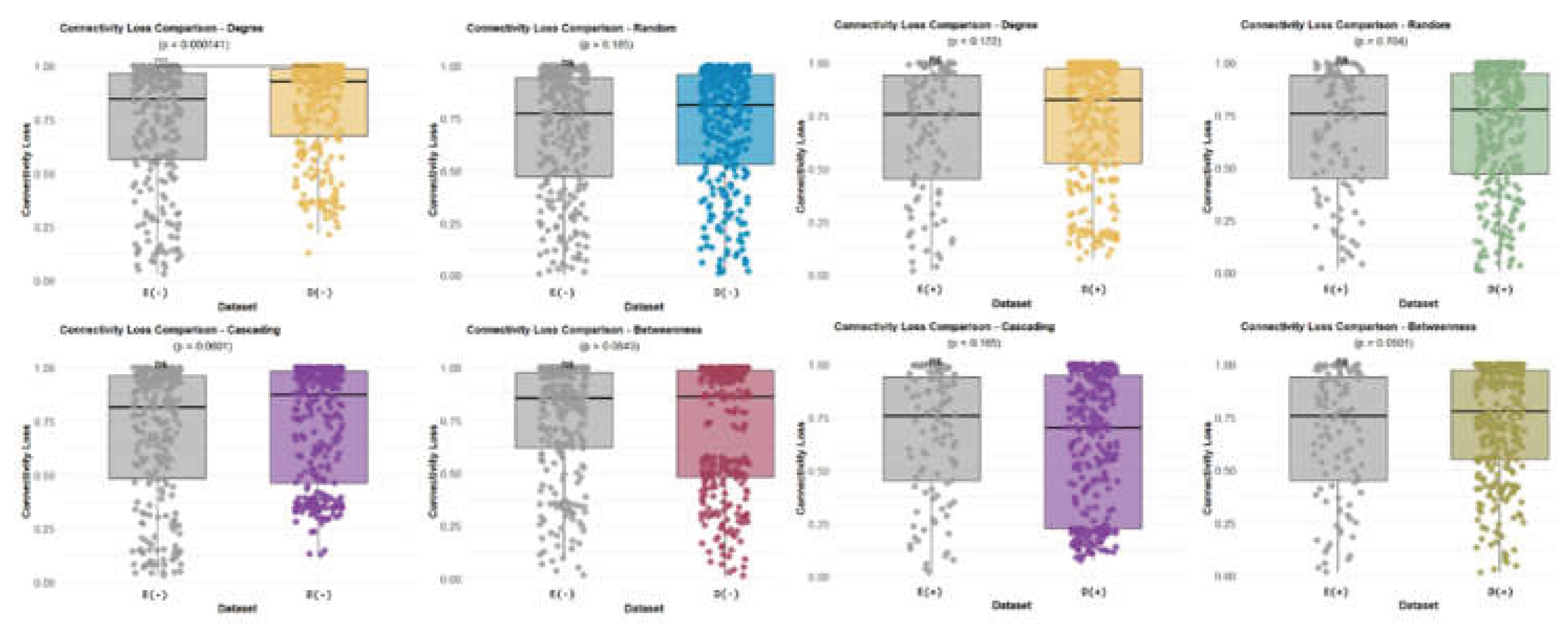
3. Results
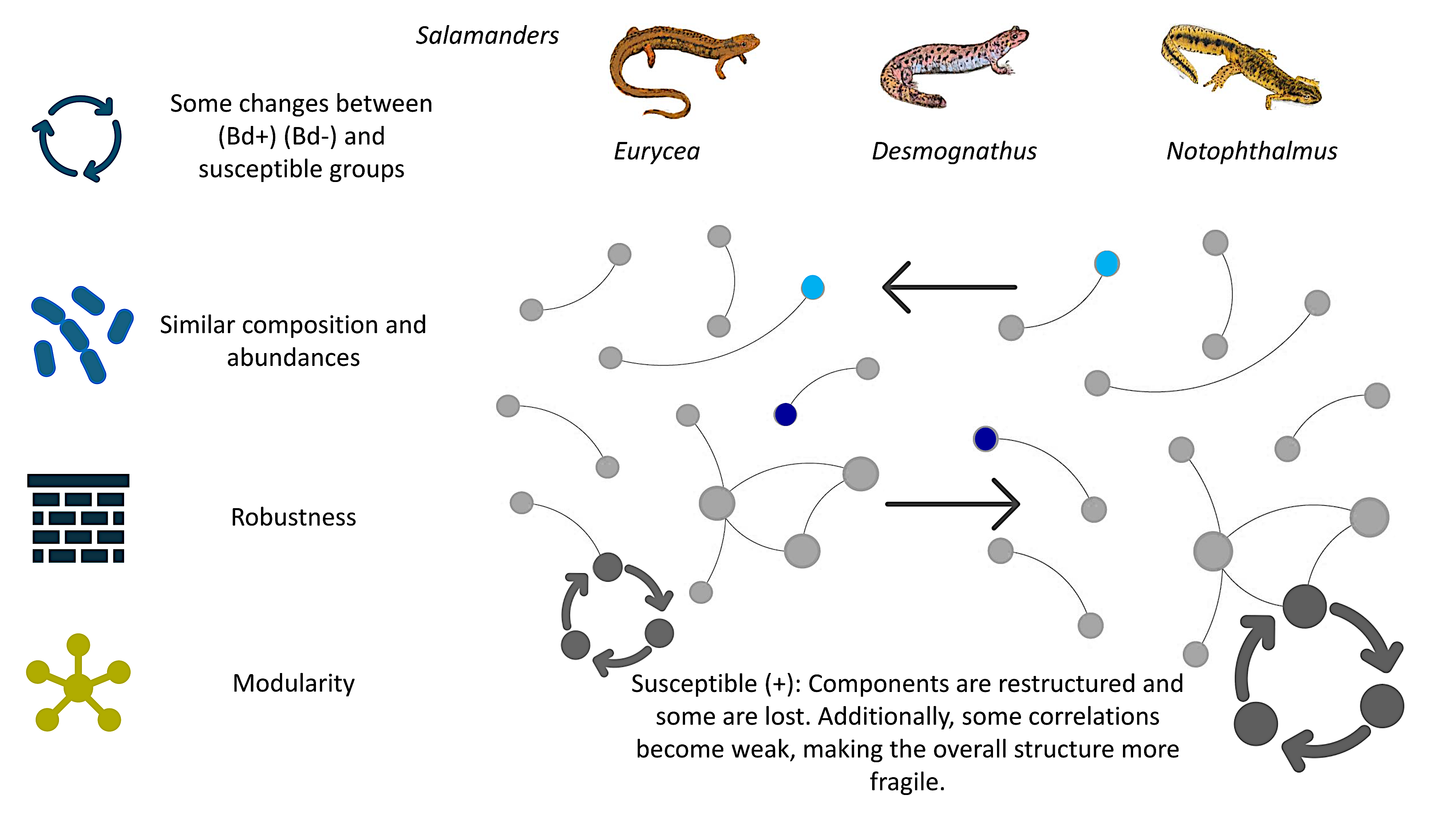
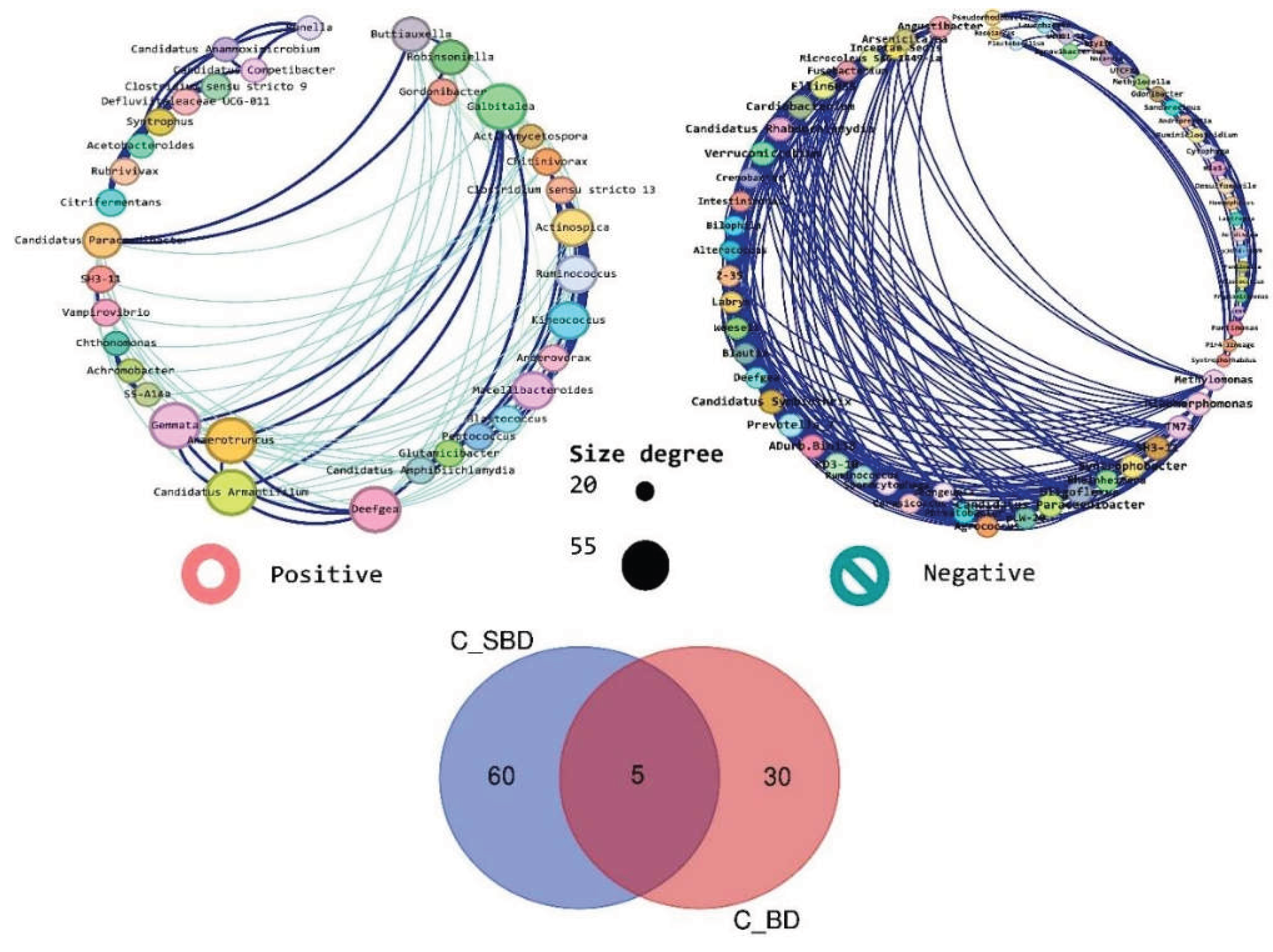
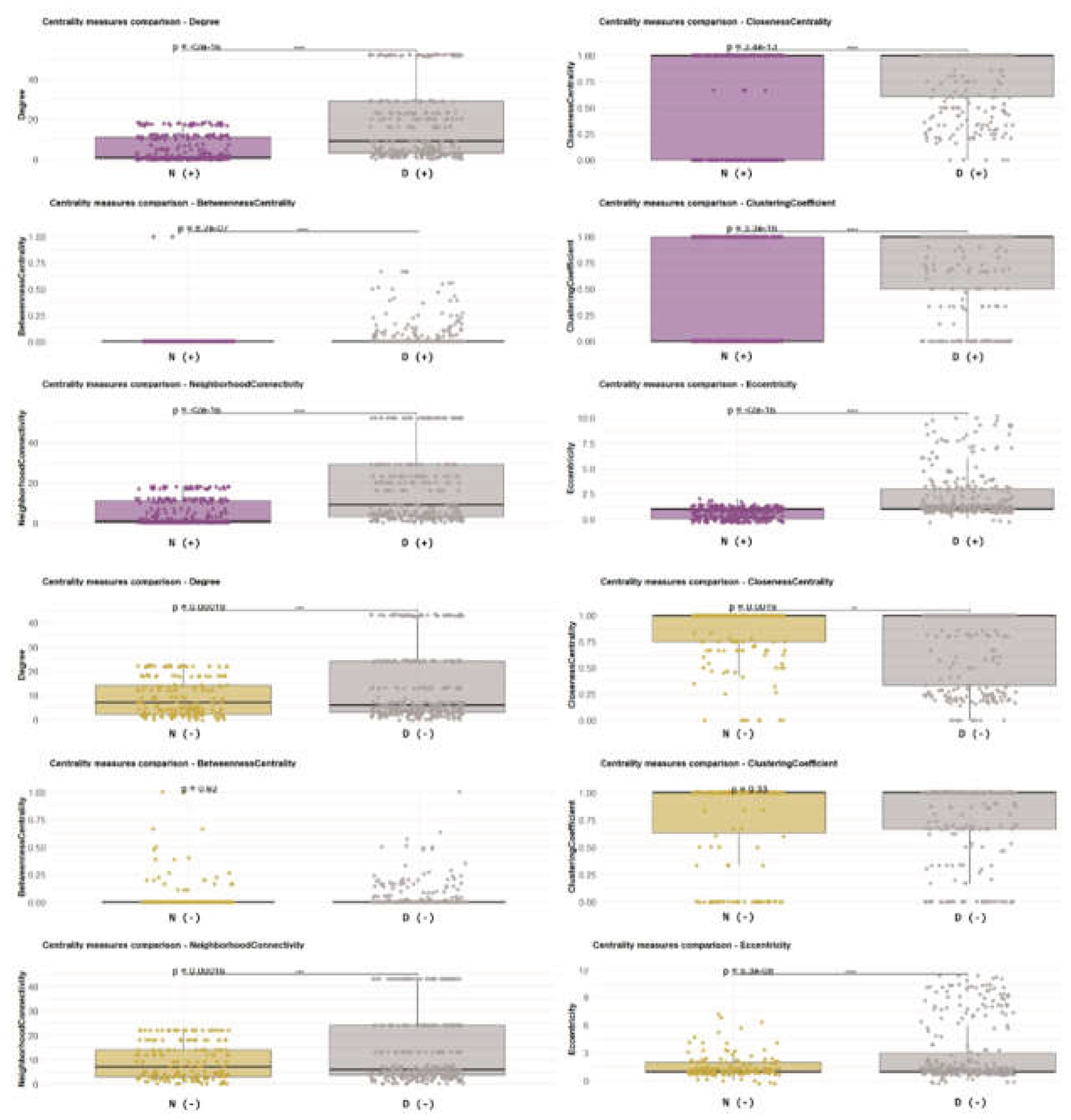
3.1. Network-Level Differences Between Bd− and Bd+ Salamanders
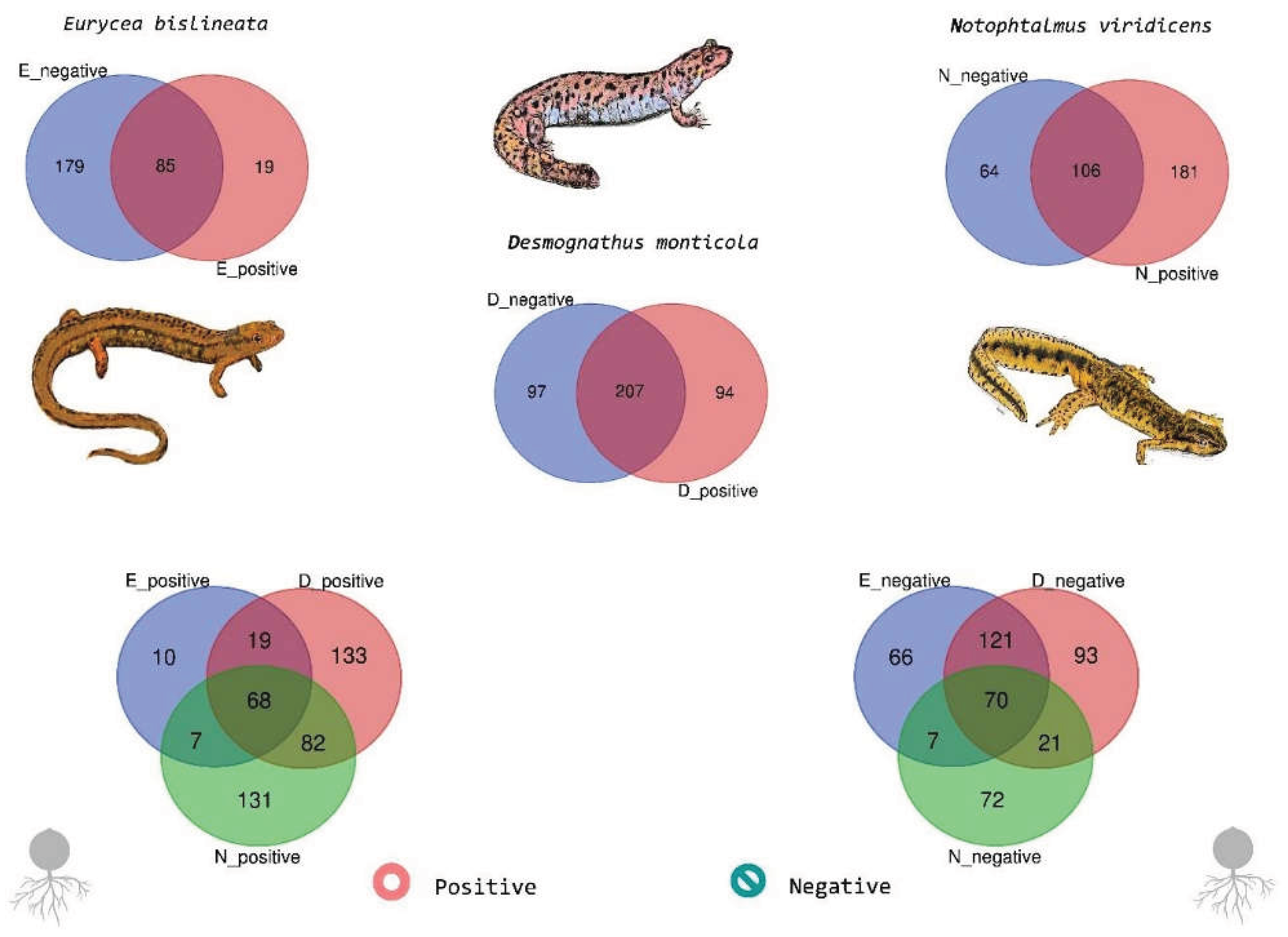
3.2. Positive vs. Negative Associations and Shared ASVs
3.3. Negative Associations and Taxa Linked to Vibrionaceae
3.4. Species-Level Composition and Shared Taxa Across Bd Status
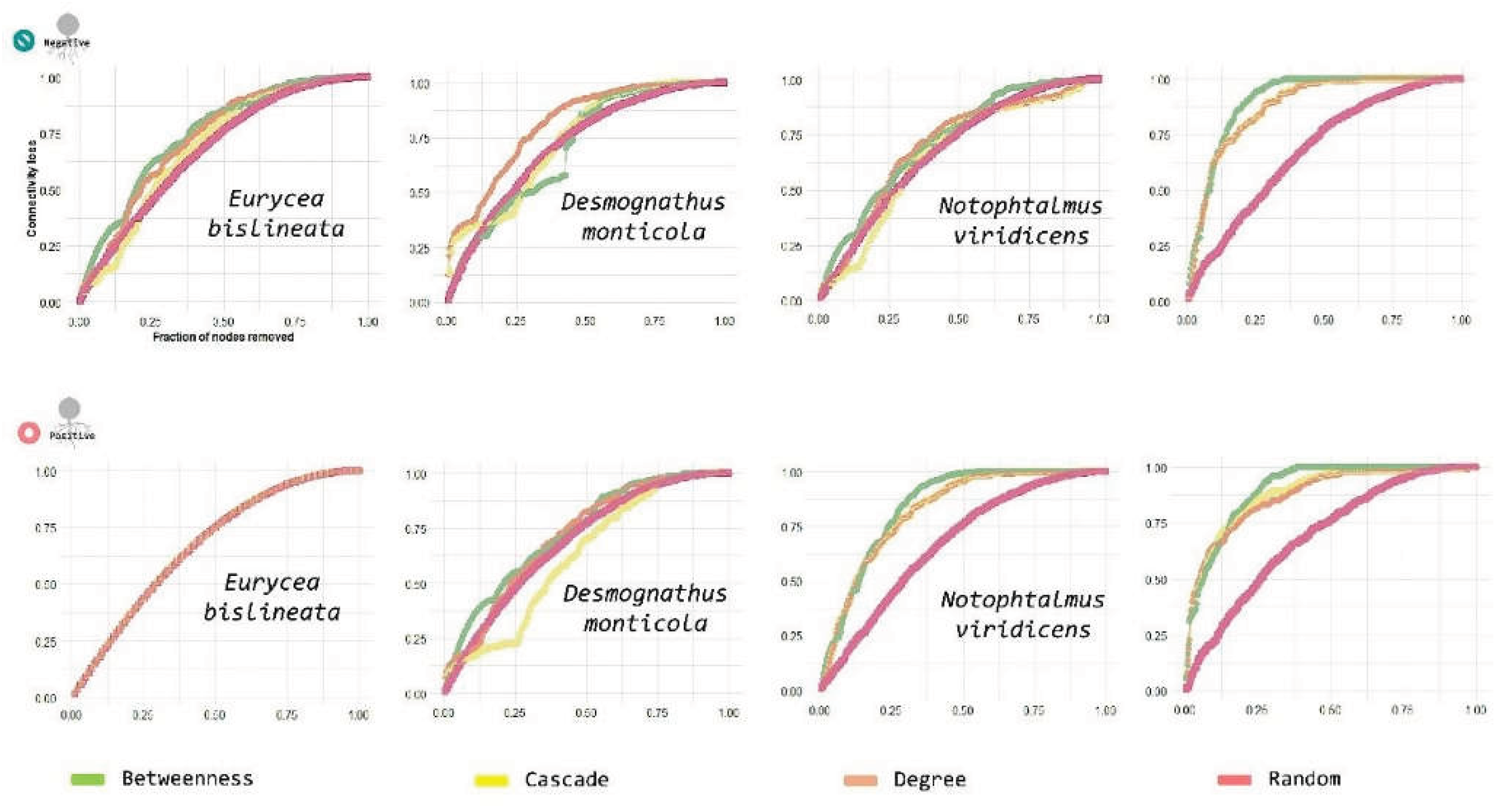
3.5. Eurycea
3.6. Desmognathus
3.7. Notophthalmus
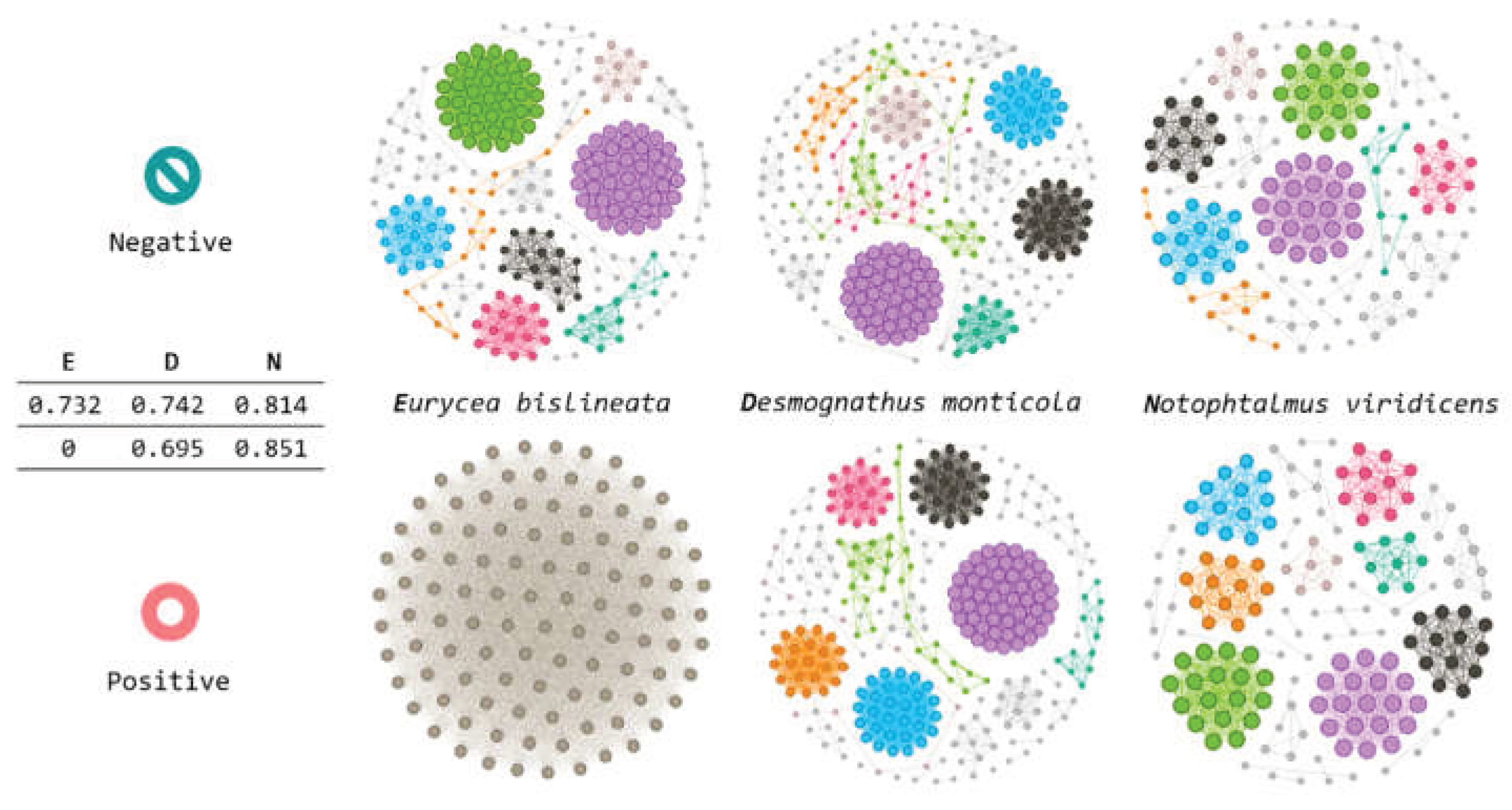
3.8. Module Membership Differences Across Conditions
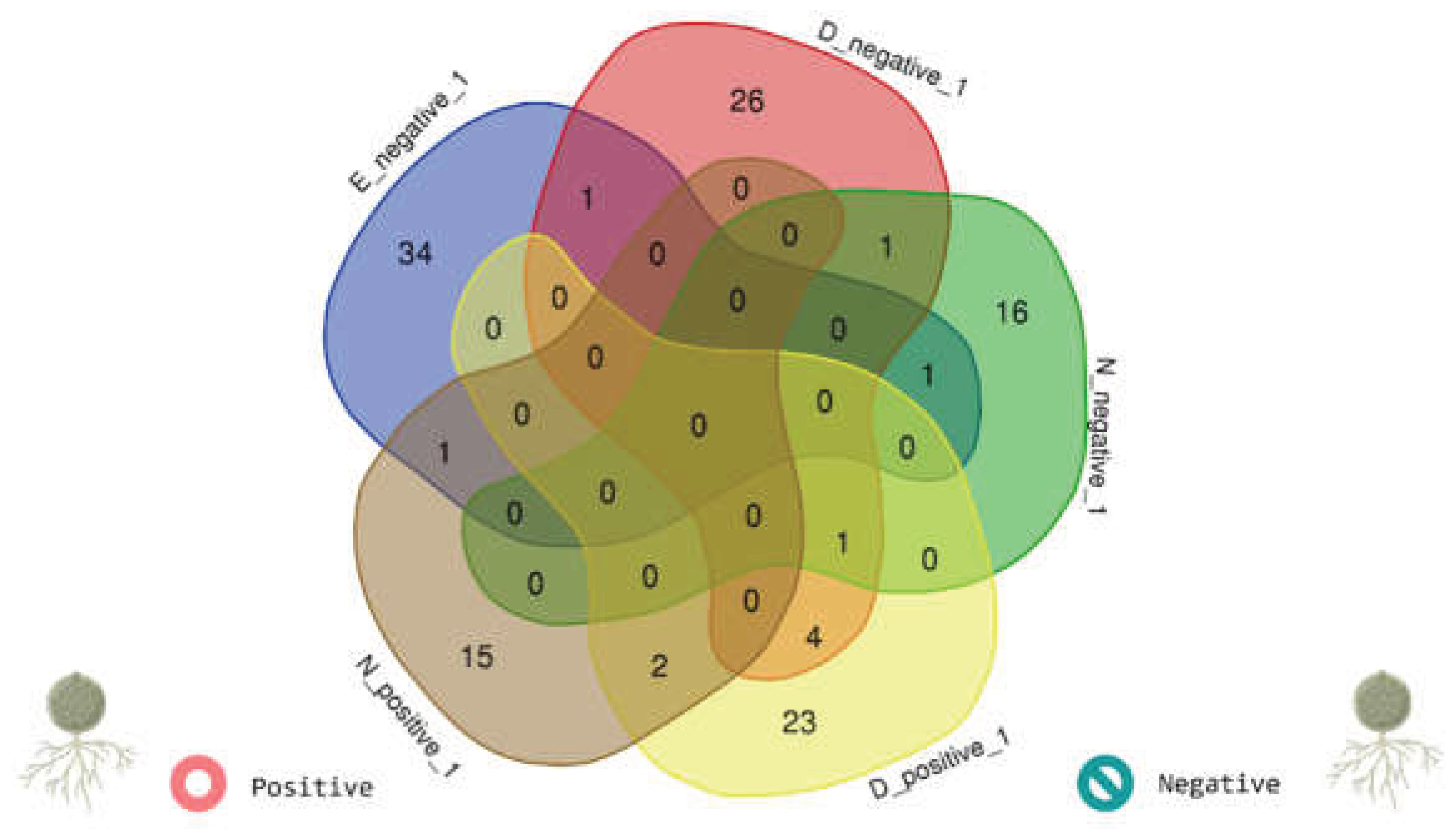
3.9. Core Network Analysis Under Stringent Association Thresholds
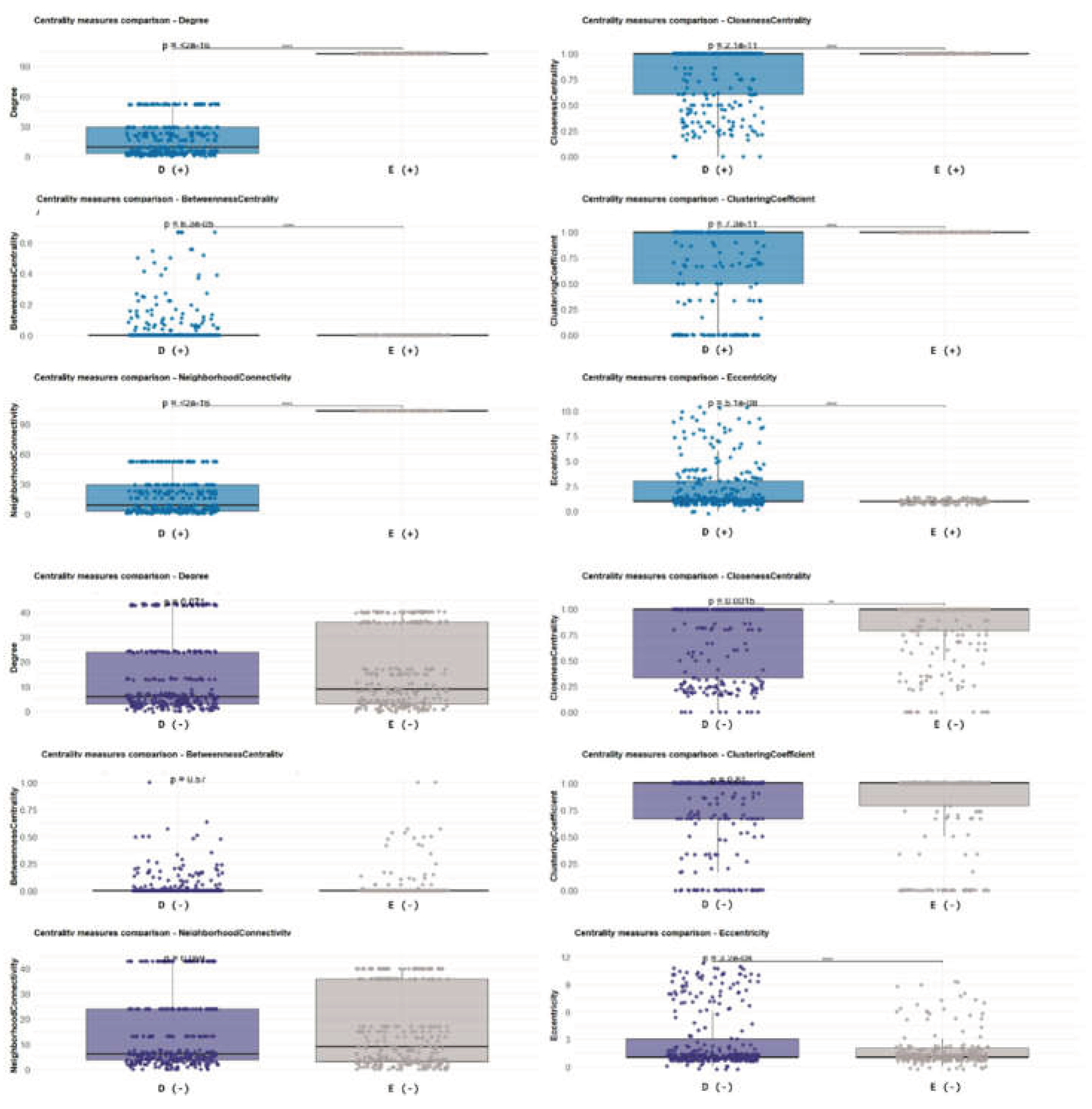
3.10. Susceptible vs Tolerant Hosts (Notophthalmus vs Desmognathus)
3.11. Tolerant vs Tolerant Hosts (Eurycea vs Desmognathus)
4. Discussion
4.1. Community Composition and Diversity: Stability at High Taxonomic Ranks
4.2. Differential Taxa and the Ecological Meaning of “Biomarkers”
4.3. Network Analyses: Reorganization of Associations Despite Similar Composition
- Module membership turnover: Even when focusing on the same nominal module (e.g., the first/pink module across networks), overlap in node membership across Bd− and Bd+ networks was minimal, implying that Bd detection is associated with a reshuffling of taxa among modules. This is consistent with a community in which different taxa can occupy similar ecological “roles” or niches under varying conditions.
- Core network differences: Under stringent thresholds defining “core” associations, Bd− communities exhibited a larger and denser core than Bd+ communities, with limited overlap in core membership (only a small set of shared taxa). This pattern is consistent with Bd detection being associated with the loss (or reduced detectability) of strongly coupled taxa and/or the weakening of high-strength associations among community members.
- Modularity changes were host-dependent: Bd− networks showed high modularity across host taxa, whereas Bd+ networks showed modularity responses that differed by species. The modularity value of zero observed for Bd+ Eurycea warrants cautious interpretation: it may reflect the collapse of the community structure, but it can also arise from methodological factors (e.g., sparse networks after filtering, small sample size, disconnected components, or algorithmic resolution limits). Nonetheless, considering the broader evidence of module membership turnover and core network shrinkage, our results are consistent with the hypothesis that Bd detection can disrupt modular organization in a host-specific manner.
4.4. Central Taxa, Clustering, and Implications for Tolerance vs Susceptibility
4.5. Robustness: Vulnerability Under Targeted Attacks and Ecological Interpretation
4.6. Limitations and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Ethics statement
Data availability statement
Acknowledgments
Conflicts of Interest
References
- Crudele, L.; Gadaleta, R. M.; Cariello, M.; Moschetta, A. Gut microbiota in the pathogenesis and therapeutic approaches of diabetes. EBioMedicine 2023, 97, 104821. [Google Scholar] [CrossRef]
- Ting, N. L.-N.; Lau, H. C.-H.; Yu, J. Cancer pharmacomicrobiomics: Targeting microbiota to optimize cancer therapy outcomes. Gut 2022, 71(7), 1412–1425. [Google Scholar] [CrossRef]
- Kuznetzova, O. Ju.; Gorshenina, A. P.; Nesterov, O. V.; Maximovskaya, L. N. Complex treatment of recurrent herpetic stomatitis with dysbiosis. Stomatologiya 2017, 96(5), 31. [Google Scholar] [CrossRef] [PubMed]
- Vanhoefer, U.; Harstrick, A.; Achterrath, A.; Cao, W.; Seeber, S.; Rustum, S.Y.M. Irinotecan in the treatment of colorectal cancer: Clinical overview. Journal of Clinical Oncology: Official Journal of the American Society of Clinical Oncology 2001, 19(5), 1501–1518. [Google Scholar] [CrossRef] [PubMed]
- Torres-Carrillo, N.; Martínez-López, E.; Torres-Carrillo, N. M.; López-Quintero, A.; Moreno-Ortiz, J. M.; González-Mercado, A.; Gutiérrez-Hurtado, I. A. Pharmacomicrobiomics and Drug–Infection Interactions: The Impact of Commensal, Symbiotic and Pathogenic Microorganisms on a Host Response to Drug Therapy. International Journal of Molecular Sciences 2023, 24(23), 17100. [Google Scholar] [CrossRef]
- Brown, L. C.; Bobo, W. V.; Gall, C. A.; Müller, D. J.; Bousman, C. A. Pharmacomicrobiomics of Antidepressants in Depression: A Systematic Review. Journal of Personalized Medicine 2023, 13(7), 1086. [Google Scholar] [CrossRef] [PubMed]
- Menozzi, E.; Schapira, A. H. V. The Gut Microbiota in Parkinson Disease: Interactions with Drugs and Potential for Therapeutic Applications. CNS Drugs 2024, 38(5), 315–331. [Google Scholar] [CrossRef]
- Kueneman, J. G.; Parfrey, L. W.; Woodhams, D. C.; Archer, H. M.; Knight, R.; McKenzie, V. J. The amphibian skin-associated microbiome across species, space and life history stages. Molecular Ecology 2014, 23(6), 1238–1250. [Google Scholar] [CrossRef]
- Martínez-Ugalde, E.; Ávila-Akerberg, V.; González Martínez, T. M.; Vázquez Trejo, M.; Zavala Hernández, D.; Anaya-Morales, S. L.; Rebollar, E. A. The skin microbiota of the axolotl Ambystoma altamirani is highly influenced by metamorphosis and seasonality but not by pathogen infection. Animal Microbiome 2022, 4(1), 63. [Google Scholar] [CrossRef]
- Wuerthner, V. P.; Hua, J.; Hernández-Gómez, O. Life stage and proximity to roads shape the skin microbiota of eastern newts (Notophthalmus viridescens). Environmental Microbiology 2022, 24(9), 3954–3965. [Google Scholar] [CrossRef]
- Wan, B.; Chen, G.; Poon, E. S. K.; Fung, H. S.; Lau, A.; Sin, S. Y. W. Environmental factors and host sex influence the skin microbiota structure of Hong Kong newt (Paramesotriton hongkongensis) in a coldspot of chytridiomycosis in subtropical East Asia. Integrative Zoology 2024. [Google Scholar] [CrossRef]
- Dwaraka, V. B.; Voss, S. R. Towards comparative analyses of salamander limb regeneration. Journal of Experimental Zoology Part B: Molecular and Developmental Evolution 2021, 336(2), 129–144. [Google Scholar] [CrossRef]
- Demircan, T.; Gül, S.; Taşçı, E. A. Can Microbiome Modulate Regenerative Capacity? A Comparative Microbiome Study Reveals a Dominant Presence of Flavobacteriaceae in Blastema Tissue During Axolotl Limb Regeneration. OMICS: A Journal of Integrative Biology 2024, 28(6), 291–302. [Google Scholar] [CrossRef]
- Hunt, D. E.; Ward, C. S. A network-based approach to disturbance transmission through microbial interactions. Frontiers in Microbiology 2015, 6. [Google Scholar] [CrossRef]
- Analysis of biological networks; Junker, B. H., Schreiber, F., Eds.; Wiley-Interscience, 2008. [Google Scholar]
- Oldham, S.; Fulcher, B.; Parkes, L.; Arnatkevic Iūtė, A.; Suo, C.; Fornito, A. Consistency and differences between centrality measures across distinct classes of networks. PloS One 2019, 14(7), e0220061. [Google Scholar] [CrossRef]
- Lou, Y.; Wu, R.; Li, J.; Wang, L.; Tang, C.-B.; Chen, G. Classification-based prediction of network connectivity robustness. Neural Networks: The Official Journal of the International Neural Network Society 2023, 157, 136–146. [Google Scholar] [CrossRef]
- Saint-Antoine, M. M.; Singh, A. Network inference in systems biology: Recent developments, challenges, and applications. Current Opinion in Biotechnology 2020, 63, 89–98. [Google Scholar] [CrossRef] [PubMed]
- Fisher, M. C.; Garner, T. W. J.; Walker, S. F. Global Emergence of Batrachochytrium dendrobatidis and Amphibian Chytridiomycosis in Space, Time, and Host. Annual Review of Microbiology 2009, 63(1), 291–310. [Google Scholar] [CrossRef]
- Martel, A.; Spitzen-van Der Sluijs, A.; Blooi, M.; Bert, W.; Ducatelle, R.; Fisher, M. C.; Woeltjes, A.; Bosman, W.; Chiers, K.; Bossuyt, F.; Pasmans, F. Batrachochytrium salamandrivorans sp. Nov. Causes lethal chytridiomycosis in amphibians. Proceedings of the National Academy of Sciences 2013, 110(38), 15325–15329. [Google Scholar] [CrossRef] [PubMed]
- Olson, D. H.; Ronnenberg, K. L.; Glidden, C. K.; Christiansen, K. R.; Blaustein, A. R. Global Patterns of the Fungal Pathogen Batrachochytrium dendrobatidis Support Conservation Urgency. Frontiers in Veterinary Science 2021, 8, 685877. [Google Scholar] [CrossRef] [PubMed]
- Tornabene, B. J.; Hossack, B. R.; Crespi, E. J.; Breuner, C. W. Evaluating corticosterone as a biomarker for amphibians exposed to increased salinity and ambient corticosterone. Conservation Physiology 2021, 9(1), coab049. [Google Scholar] [CrossRef] [PubMed]
- Rollins-Smith, L. A.; Le Sage, E. H. Heat stress and amphibian immunity in a time of climate change. Philosophical Transactions of the Royal Society B: Biological Sciences 2023, 378(1882), 20220132. [Google Scholar] [CrossRef] [PubMed]
- Belasen, A. M.; Riolo, M. A.; Bletz, M. C.; Lyra, M. L.; Toledo, L. F.; James, T. Y. Geography, Host Genetics, and Cross-Domain Microbial Networks Structure the Skin Microbiota of Fragmented Brazilian Atlantic Forest Frog Populations. Ecology and Evolution 2021, 11(14), 9293–9307. [Google Scholar] [CrossRef]
- Neely, W. J.; Martins, R. A.; Mendonça Da Silva, C. M.; Ferreira Da Silva, T.; Fleck, L. E.; Whetstone, R. D.; Woodhams, D. C.; Cook, W. H.; Prist, P. R.; Valiati, V. H.; Greenspan, S. E.; Tozetti, A. M.; Earley, R. L.; Becker, C. G. Linking microbiome and stress hormone responses in wild tropical treefrogs across continuous and fragmented forests. Communications Biology 2023, 6(1), 1261. [Google Scholar] [CrossRef]
- Schilliger, L.; Paillusseau, C.; François, C.; Bonwitt, J. Major Emerging Fungal Diseases of Reptiles and Amphibians. Pathogens 2023, 12(3), 429. [Google Scholar] [CrossRef] [PubMed]
- Xie, G. Y.; Olson, D. H.; Blaustein, A. R. Projecting the Global Distribution of the Emerging Amphibian Fungal Pathogen, Batrachochytrium dendrobatidis, Based on IPCC Climate Futures. PLOS ONE 2016, 11(8), e0160746. [Google Scholar] [CrossRef]
- Nnadi, N. E.; Carter, D. A. Climate change and the emergence of fungal pathogens. PLOS Pathogens 2021, 17(4), e1009503. [Google Scholar] [CrossRef]
- Muletz-Wolz, C. R.; Fleischer, R. C.; Lips, K. R. Fungal disease and temperature alter skin microbiome structure in an experimental salamander system. Molecular Ecology 2019, 28(11), 2917–2931. [Google Scholar] [CrossRef]
- Woodhams, D. C.; Alford, R. A.; Antwis, R. E.; Archer, H.; Becker, M. H.; Belden, L. K.; Bell, S. C.; Bletz, M.; Daskin, J. H.; Davis, L. R.; Flechas, S. V.; Lauer, A.; Gonzalez, A.; Harris, R. N.; Holden, W. M.; Hughey, M. C.; Ibáñez, R.; Knight, R.; Kueneman, J.; McKenzie, V. Antifungal isolates database of amphibian skin-associated bacteria and functions against emerging fungal pathogens: Ecological Archives E096-059. Ecology 2015, 96(2), 595–595. [Google Scholar] [CrossRef]
- Piovia-Scott, J.; Rejmanek, D.; Woodhams, D. C.; Worth, S. J.; Kenny, H.; McKenzie, V.; Lawler, S. P.; Foley, J. E. Greater Species Richness of Bacterial Skin Symbionts Better Suppresses the Amphibian Fungal Pathogen Batrachochytrium Dendrobatidis. Microbial Ecology 2017, 74(1), 217–226. [Google Scholar] [CrossRef]
- Woodhams, D. C.; Rollins-Smith, L. A.; Reinert, L. K.; Lam, B. A.; Harris, R. N.; Briggs, C. J.; Vredenburg, V. T.; Patel, B. T.; Caprioli, R. M.; Chaurand, P.; Hunziker, P.; Bigler, L. Probiotics Modulate a Novel Amphibian Skin Defense Peptide That Is Antifungal and Facilitates Growth of Antifungal Bacteria. Microbial Ecology 2020, 79(1), 192–202. [Google Scholar] [CrossRef]
- Bates, K. A.; Sommer, U.; Hopkins, K. P.; Shelton, J. M. G.; Wierzbicki, C.; Sergeant, C.; Tapley, B.; Michaels, C. J.; Schmeller, D. S.; Loyau, A.; Bosch, J.; Viant, M. R.; Harrison, X. A.; Garner, T. W. J.; Fisher, M. C. Microbiome function predicts amphibian chytridiomycosis disease dynamics. Microbiome 2022, 10(1), 44. [Google Scholar] [CrossRef]
- Chen, M. Y.; Alexiev, A.; McKenzie, V. J. Bacterial Biofilm Thickness and Fungal Inhibitory Bacterial Richness Both Prevent Establishment of the Amphibian Fungal Pathogen Batrachochytrium dendrobatidis. Applied and Environmental Microbiology 2022, 88(5), e01604-21. [Google Scholar] [CrossRef]
- MacAlpine, J.; Robbins, N.; Cowen, L. E. Bacterial-fungal interactions and their impact on microbial pathogenesis. Molecular Ecology 2023, 32(10), 2565–2581. [Google Scholar] [CrossRef]
- Kinnula, H.; Mappes, J.; Sundberg, L.-R. Coinfection outcome in an opportunistic pathogen depends on the inter-strain interactions. BMC Evolutionary Biology 2017, 17(1), 77. [Google Scholar] [CrossRef] [PubMed]
- Fast, D.; Kostiuk, B.; Foley, E.; Pukatzki, S. Commensal-Pathogen Competition Impacts Host Viability. 2018. [Google Scholar] [CrossRef] [PubMed]
- Santus, W.; Devlin, J. R.; Behnsen, J. Crossing Kingdoms: How the Mycobiota and Fungal-Bacterial Interactions Impact Host Health and Disease. Infection and Immunity 2021, 89(4), e00648-20. [Google Scholar] [CrossRef]
- Holmes, M. L.; Shine, R.; Waddle, A. W. Spontaneous reoccurrence of Batrachochytrium dendrobatidis infections in Australian green tree frogs (Litoria caerulea) following successful heat therapy: Case report. Veterinary Research Communications 2024, 48(5), 3229–3237. [Google Scholar] [CrossRef] [PubMed]
- Gibson, J. F.; Pidwill, G. R.; Carnell, O. T.; Surewaard, B. G. J.; Shamarina, D.; Sutton, J. A. F.; Jeffery, C.; Derré-Bobillot, A.; Archambaud, C.; Siggins, M. K.; Pollitt, E. J. G.; Johnston, S. A.; Serror, P.; Sriskandan, S.; Renshaw, S. A.; Foster, S. J. Commensal bacteria augment Staphylococcus aureus infection by inactivation of phagocyte-derived reactive oxygen species. PLoS Pathogens 2021, 17(9), e1009880. [Google Scholar] [CrossRef]
- Hardman, R. H.; Reinert, L. K.; Irwin, K. J.; Oziminski, K.; Rollins-Smith, L.; Miller, D. L. Disease state associated with chronic toe lesions in hellbenders may alter anti-chytrid skin defenses. Scientific Reports 2023, 13(1). [Google Scholar] [CrossRef]
- Hu, H.-L.; Chen, J.-M.; Chen, J.-Y.; Seah, R. W. X.; Ding, G.-H. Microbial Diversity of the Chinese Tiger Frog (Hoplobatrachus rugulosus) on Healthy versus Ulcerated Skin. Animals 2022, 12(10), 1241. [Google Scholar] [CrossRef]
- Li, Z.; Wang, G.; Zhang, K.; Gong, W.; Yu, E.; Tian, J.; Xie, J.; Yu, D. Epizootic ulcerative syndrome causes cutaneous dysbacteriosis in hybrid snakehead (Channa maculata♀ × Channa argus♂). PeerJ 2019, 7, e6674. [Google Scholar] [CrossRef]
- Bletz, M. C.; Kelly, M.; Sabino-Pinto, J.; Bales, E.; Van Praet, S.; Bert, W.; Boyen, F.; Vences, M.; Steinfartz, S.; Pasmans, F.; Martel, A. Disruption of skin microbiota contributes to salamander disease. Proceedings of the Royal Society B: Biological Sciences 2018, 285(1885), 20180758. [Google Scholar] [CrossRef]
- Harris, R.; Lauer, A.; Simon, M.; Banning, J.; Alford, R. Addition of antifungal skin bacteria to salamanders ameliorates the effects of chytridiomycosis. Diseases of Aquatic Organisms 2009, 83, 11–16. [Google Scholar] [CrossRef]
- Becker, M. H.; Brucker, R. M.; Schwantes, C. R.; Harris, R. N.; Minbiole, K. P. C. The Bacterially Produced Metabolite Violacein Is Associated with Survival of Amphibians Infected with a Lethal Fungus. Applied and Environmental Microbiology 2009, 75(21), 6635–6638. [Google Scholar] [CrossRef]
- Agate, L.; Beam, D.; Bucci, C.; Dukashin, Y.; Jo’Beh, R.; O’Brien, K.; Jude, B. A. The Search for Violacein-Producing Microbes to Combat Batrachochytrium dendrobatidis: A Collaborative Research Project between Secondary School and College Research Students. Journal of Microbiology & Biology Education 2016, 17(1), 70–73. [Google Scholar] [CrossRef]
- Gass, J.; Voyles, J. When Defenses Fail: Atelopus zeteki Skin Secretions Increase Growth of the Pathogen Batrachochytrium dendrobatidis. Integrative and Comparative Biology 2022, 62(6), 1595–1605. [Google Scholar] [CrossRef] [PubMed]
- Jiménez, R. R.; Carfagno, A.; Linhoff, L.; Gratwicke, B.; Woodhams, D. C.; Chafran, L. S.; Bletz, M. C.; Bishop, B.; Muletz-Wolz, C. R. Inhibitory Bacterial Diversity and Mucosome Function Differentiate Susceptibility of Appalachian Salamanders to Chytrid Fungal Infection. Applied and Environmental Microbiology 2022, 88(8), e01818-21. [Google Scholar] [CrossRef]
- Van Rooij, P.; Martel, A.; Haesebrouck, F.; Pasmans, F. Amphibian chytridiomycosis: A review with focus on fungus-host interactions. Veterinary Research 2015, 46(1), 137. [Google Scholar] [CrossRef] [PubMed]
- Corduneanu, A.; Wu-Chuang, A.; Maitre, A.; Obregon, D.; Sándor, A. D.; Cabezas-Cruz, A. Structural differences in the gut microbiome of bats using terrestrial vs. Aquatic feeding resources. BMC Microbiology 2023, 23(1), 93. [Google Scholar] [CrossRef] [PubMed]
- Woodhams, D. C.; LaBumbard, B. C.; Barnhart, K. L.; Becker, M. H.; Bletz, M. C.; Escobar, L. A.; Flechas, S. V.; Forman, M. E.; Iannetta, A. A.; Joyce, M. D.; Rabemananjara, F.; Gratwicke, B.; Vences, M.; Minbiole, K. P. C. Prodigiosin, Violacein, and Volatile Organic Compounds Produced by Widespread Cutaneous Bacteria of Amphibians Can Inhibit Two Batrachochytrium Fungal Pathogens. Microbial Ecology 2018, 75(4), 1049–1062. [Google Scholar] [CrossRef] [PubMed]
- Myers, J. M.; Ramsey, J. P.; Blackman, A. L.; Nichols, A. E.; Minbiole, K. P. C.; Harris, R. N. Synergistic inhibition of the lethal fungal pathogen Batrachochytrium dendrobatidis: The combined effect of symbiotic bacterial metabolites and antimicrobial peptides of the frog Rana muscosa. Journal of Chemical Ecology 2012, 38(8), 958–965. [Google Scholar] [CrossRef]
- Stegen, G.; Pasmans, F.; Schmidt, B. R.; Rouffaer, L. O.; Van Praet, S.; Schaub, M.; Canessa, S.; Laudelout, A.; Kinet, T.; Adriaensen, C.; Haesebrouck, F.; Bert, W.; Bossuyt, F.; Martel, A. Drivers of salamander extirpation mediated by Batrachochytrium salamandrivorans. Nature 2017, 544(7650), 353–356. [Google Scholar] [CrossRef]
- Mulla, L.; Hernández-Gómez, O. Wildfires disturb the natural skin microbiota of terrestrial salamanders. Environmental Microbiology 2023, 25(11), 2203–2215. [Google Scholar] [CrossRef] [PubMed]
- Cordeiro, I. F.; Lemes, C. G. de C.; Sanchez, A. B.; da Silva, A. K.; de Paula, C. H.; de Matos, R. C.; Ribeiro, D. F.; de Matos, J. P.; Garcia, C. C. M.; Beirão, M.; Becker, C. G.; Pires, M. R. S.; Moreira, L. M. Amphibian tolerance to arsenic: Microbiome-mediated insights. Scientific Reports 2024, 14(1), 10193. [Google Scholar] [CrossRef]
- Li, S.; Wang, X.; Lu, Y.; Wang, J.; Yu, D.; Zhou, Z.; Wei, J.; Liu, L.; Liu, J.; Liu, F.; Su, Y.; Xu, H. Co-infections of Klebsiella pneumoniae and Elizabethkingia miricola in black-spotted frogs (Pelophylax nigromaculatus). Microbial Pathogenesis 2023, 180, 106150. [Google Scholar] [CrossRef]
- Chapman, P. A.; Hudson, D.; Morgan, X. C.; Beck, C. W. The role of family and environment in determining the skin bacterial communities of captive aquatic frogs, Xenopus laevis. FEMS Microbiology Ecology 2024, 100(11), fiae131. [Google Scholar] [CrossRef]
- Zhu, W.; Zhao, C.; Feng, J.; Chang, J.; Zhu, W.; Chang, L.; Liu, J.; Xie, F.; Li, C.; Jiang, J.; Zhao, T. Effects of Habitat River Microbiome on the Symbiotic Microbiota and Multi-Organ Gene Expression of Captive-Bred Chinese Giant Salamander. Frontiers in Microbiology 2022, 13, 884880. [Google Scholar] [CrossRef]
- Zhang, H.; Ma, H.; Deng, J.; Zhao, H.; Fang, C.; Zhang, J.; Wang, Q.; Zhang, H.; Jiang, W.; Kong, F. Seasonality influences skin bacterial community structure and anti-Bd function in two anuran species. Frontiers in Microbiology 2024, 15, 1463563. [Google Scholar] [CrossRef] [PubMed]
- Chew, A.; West, M.; Berger, L.; Brannelly, L. A. The impacts of water quality on the amphibian chytrid fungal pathogen: A systematic review. Environmental Microbiology Reports 2024, 16(3), e13274. [Google Scholar] [CrossRef]
- Mutnale, M. C.; Reddy, G. S.; Vasudevan, K. Bacterial Community in the Skin Microbiome of Frogs in a Coldspot of Chytridiomycosis Infection. Microbial Ecology 2021, 82(2), 554–558. [Google Scholar] [CrossRef]
- Hu, H.-L.; Chen, J.-M.; Chen, J.-Y.; Seah, R. W. X.; Ding, G.-H. Microbial Diversity of the Chinese Tiger Frog (Hoplobatrachus rugulosus) on Healthy versus Ulcerated Skin. Animals: An Open Access Journal from MDPI 2022, 12(10), 1241. [Google Scholar] [CrossRef]
- Portillo, A.; Santibáñez, P.; Palomar, A. M.; Santibáñez, S.; Oteo, J. A. ‘Candidatus Neoehrlichia mikurensis’ in Europe. New Microbes and New Infections 2018, 22, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Yu, H.-J.; Han, X.-Y.; Li, C.; Ye, R.-Z.; Du, L.-F.; Liu, Y.-T.; Zhang, M.-Z.; Shi, X.-Y.; Zhu, D.-Y.; Shi, W.; Jia, N.; Jiang, J.-F.; Sun, Y.; Zhao, L.; Cui, X.-M.; Cao, W.-C. Genomic characterization of an emerging Rickettsia barbariae isolated from tick eggs in northwestern China. Emerging Microbes & Infections 2024, 13(1), 2396870. [Google Scholar] [CrossRef] [PubMed]
- Hartnell, R. E.; Stockley, L.; Keay, W.; Rosec, J.-P.; Hervio-Heath, D.; Van Den Berg, H.; Leoni, F.; Ottaviani, D.; Henigman, U.; Denayer, S.; Serbruyns, B.; Georgsson, F.; Krumova-Valcheva, G.; Gyurova, E.; Blanco, C.; Copin, S.; Strauch, E.; Wieczorek, K.; Lopatek, M.; Baker-Austin, C. A pan-European ring trial to validate an International Standard for detection of Vibrio cholerae, Vibrio parahaemolyticus and Vibrio vulnificus in seafoods. International Journal of Food Microbiology 2019, 288, 58–65. [Google Scholar] [CrossRef] [PubMed]
- Davoodi, S.; Foley, E. Host-Microbe-Pathogen Interactions: A Review of Vibrio cholerae Pathogenesis in Drosophila. Frontiers in Immunology 2020, 10, 3128. [Google Scholar] [CrossRef]
- Baker-Austin, C.; Oliver, J. D. Vibrio vulnificus: New insights into a deadly opportunistic pathogen. Environmental Microbiology 2018, 20(2), 423–430. [Google Scholar] [CrossRef]
- Aghnatios, R.; Drancourt, M. Gemmata Species: Planctomycetes of Medical Interest. Future Microbiology 2016, 11(5), 659–667. [Google Scholar] [CrossRef]
- Tett, A.; Pasolli, E.; Masetti, G.; Ercolini, D.; Segata, N. diversity, niches and interactions with the human host. Nature Reviews Microbiology 2021, 19(9), 585–599. [Google Scholar] [CrossRef]
- Carniel, E. The high-pathogenicity island: An iron-uptake island. Microbes and Infection 2001, 3(7), 561–569. [Google Scholar] [CrossRef]
- Zhang, H.; Ma, H.; Deng, J.; Zhao, H.; Fang, C.; Zhang, J.; Wang, Q.; Zhang, H.; Jiang, W.; Kong, F. Seasonality influences skin bacterial community structure and anti-Bd function in two anuran species. Frontiers in Microbiology 2024, 15, 1463563. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Cui, S.; Mao, B.; Zhang, Q.; Zhao, J.; Zhang, H.; Tang, X.; Chen, W. Ellagic acid and intestinal microflora metabolite urolithin A: A review on its sources, metabolic distribution, health benefits, and biotransformation. Critical Reviews in Food Science and Nutrition 2023, 63(24), 6900–6922. [Google Scholar] [CrossRef] [PubMed]
- Venturelli, O. S.; Carr, A. C.; Fisher, G.; Hsu, R. H.; Lau, R.; Bowen, B. P.; Hromada, S.; Northen, T.; Arkin, A. P. Deciphering microbial interactions in synthetic human gut microbiome communities. Molecular Systems Biology 2018, 14(6), e8157. [Google Scholar] [CrossRef] [PubMed]
- Faust, K.; Raes, J. Microbial interactions: From networks to models. Nature Reviews Microbiology 2012, 10(8), 538–550. [Google Scholar] [CrossRef]
- Louca, S.; Polz, M. F.; Mazel, F.; Albright, M. B. N.; Huber, J. A.; O’Connor, M. I.; Ackermann, M.; Hahn, A. S.; Srivastava, D. S.; Crowe, S. A.; Doebeli, M.; Parfrey, L. W. Function and functional redundancy in microbial systems. Nature Ecology & Evolution 2018, 2(6), 936–943. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



