Submitted:
16 January 2026
Posted:
19 January 2026
You are already at the latest version
Abstract
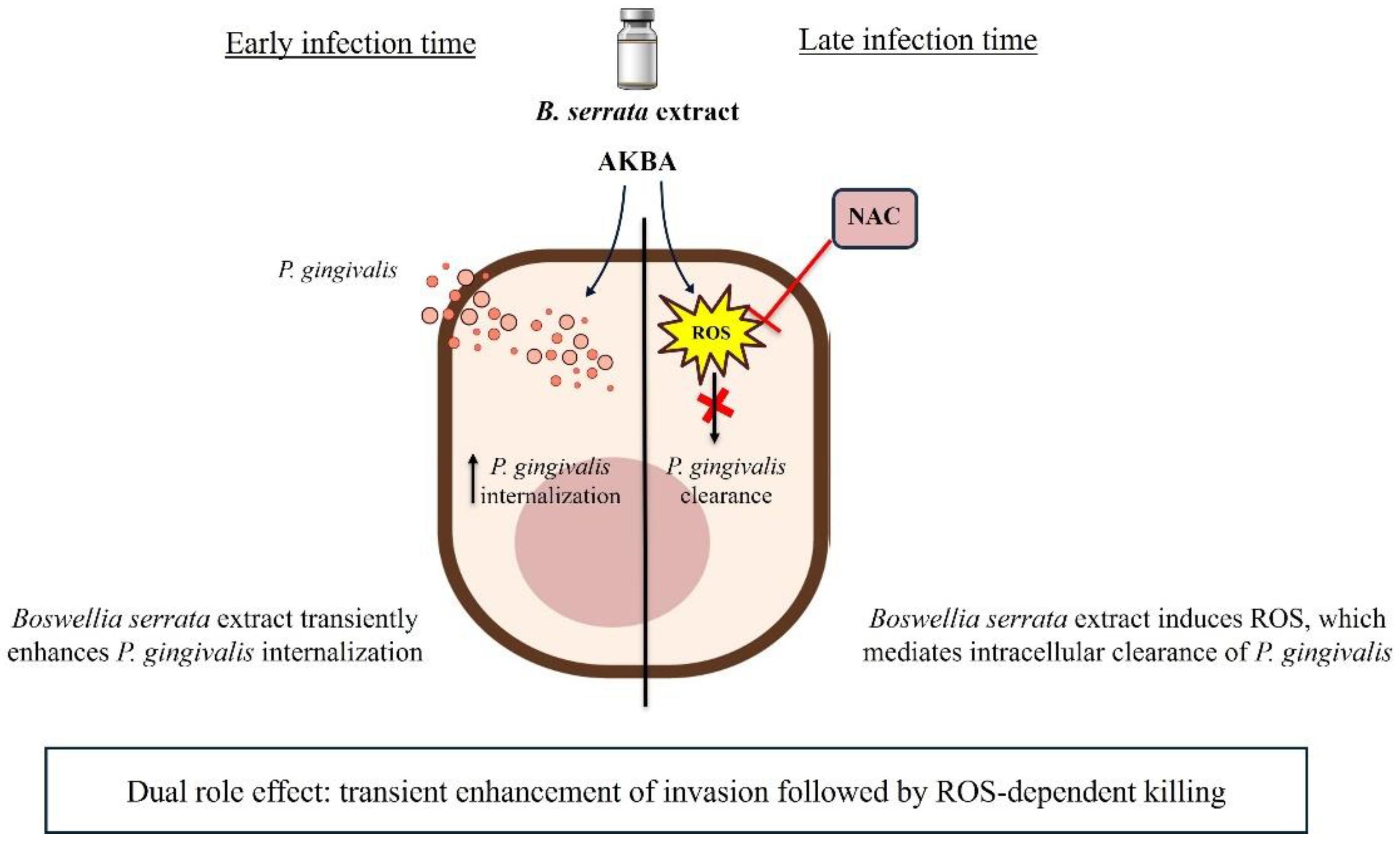
Porphyromonas gingivalis is a keystone pathogen in periodontitis, known for its ability to invade gingival epithelial cells and persist intracellularly. Conventional antimicrobials are often ineffective against intracellular pathogens, and natural products remain poorly explored in this context. Here, we investigated the antimicrobial effects of Boswellia serrata extract and its bioactive compounds on the dynamics of P. gingivalis infection in human gingival epithelial cells. During early times of infection, B. serrata extracts stimulate endocytic mechanisms and increased bacterial internalization, suggesting a modulation of epithelial uptake mechanisms. At later times of infection, B. serrata increased production of reactive oxygen species (ROS) in host cells and markedly reduced intracellular bacterial load. The antimicrobial effect was abolished by the ROS scavenger N-acetylcysteine, confirming a role for oxidative mechanisms in the clearance of P. gingivalis. Similar results were obtained with 3-O-acetyl-11-keto-β-boswellic acid (AKBA), one of the major boswellic acid derivatives found in B. serrata extract. These findings reveal a dual role of B. serrata compounds in response to P. gingivalis infection, in which B. serrata initially facilitates bacterial entry and subsequently promotes ROS-dependent intracellular killing. These findings provide new mechanistic insights into the regulation of host-pathogen interactions by the natural products found in B. serrata. Our results support the therapeutic potential of B. serrata–derived compounds for managing periodontal infections.
Keywords:
1. Introduction
2. Results
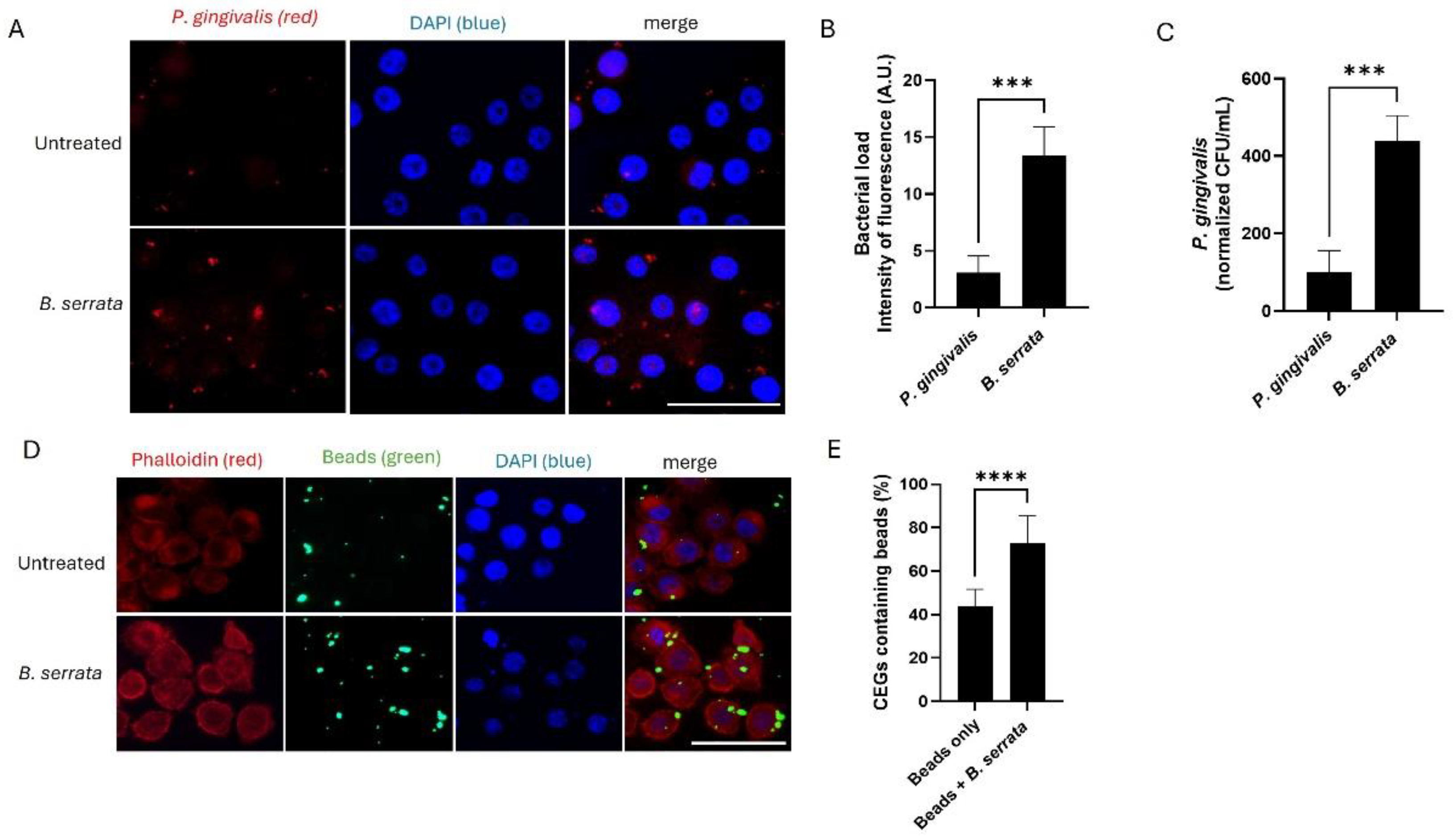
2.1. B. serrata Enhances P. gingivalis Internalization and Epithelial Endocytic Activity During Early Times of Infection
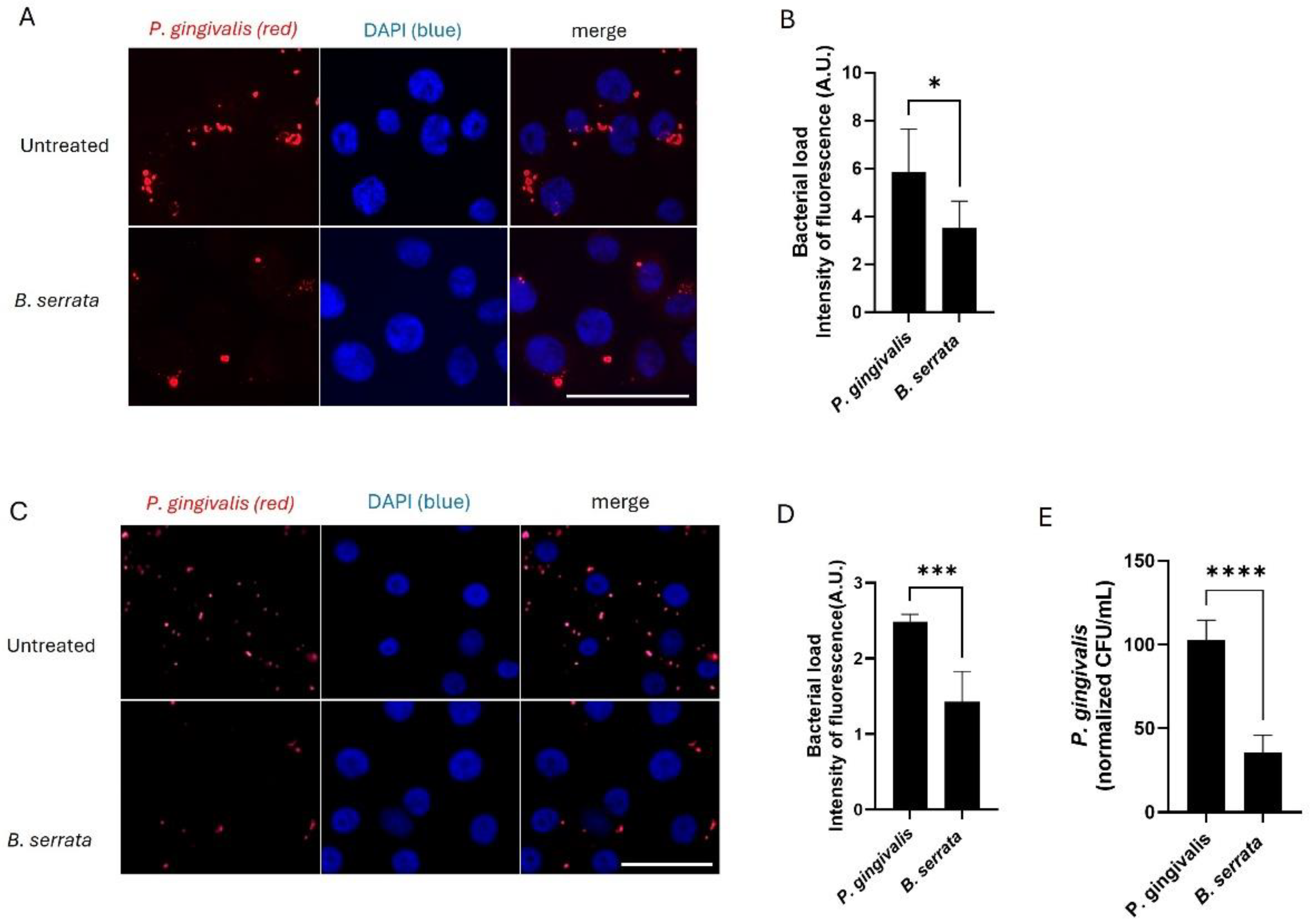
2.2. B. serrata Limits P. gingivalis Persistence at Later Times of Infection
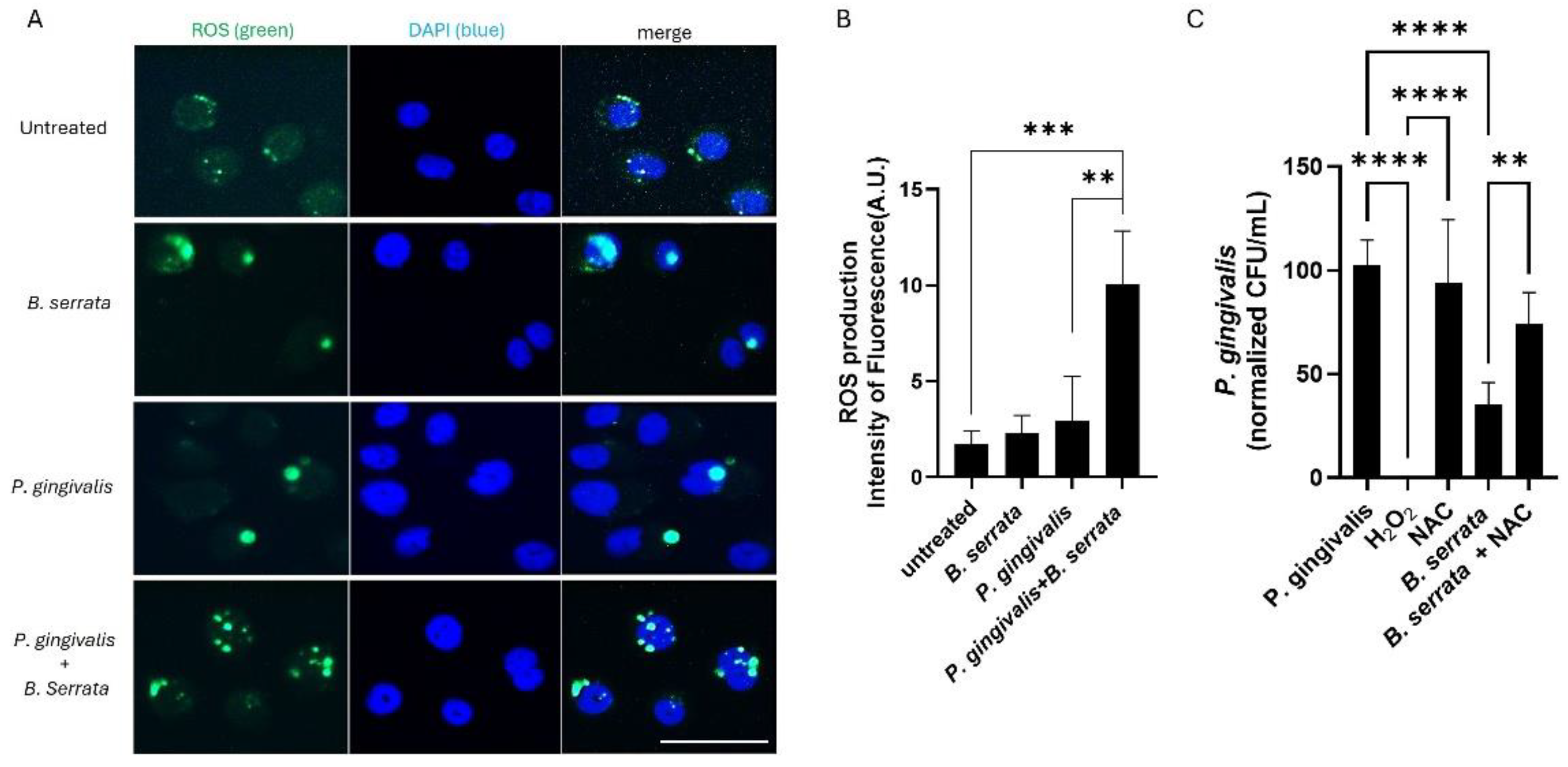
2.3. B. serrata Induces ROS Production and Restricts Intracellular P. gingivalis Survival in Human Gingival Epithelial Cells
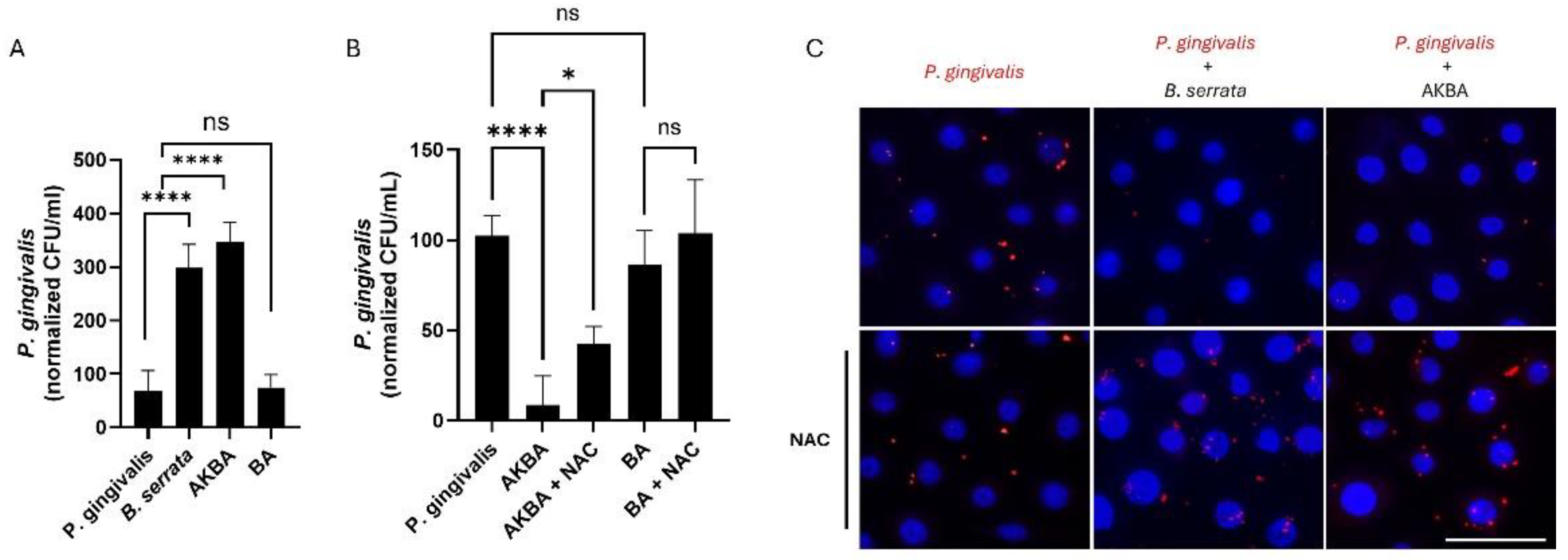
2.4. 3-O-acetyl-11-keto-β-boswellic Acids (AKBA), but Not β-boswellic Acids (BA), Promote ROS-Mediated Antimicrobial Activities
3. Discussion
4. Materials and Methods
4.1. Boswellia Serrata Extract and Compounds
4.2. Bacterial Strain and Cell Culture
4.5. Quantification of P. gingivalis Infection
4.6. Latex Bead Uptake Assay
4.7. Measurement of ROS Production
4.8. Statistics
5. Conclusions
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, K; Roberts, JS; Choi, CH; Atanasova, KR; Yilmaz, O. Porphyromonas gingivalis traffics into endoplasmic reticulum-rich-autophagosomes for successful survival in human gingival epithelial cells. Virulence 2018, 9, 845–859. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, O; Jungas, T; Verbeke, P; Ojcius, DM. Activation of the phosphatidylinositol 3-kinase/Akt pathway contributes to survival of primary epithelial cells infected with the periodontal pathogen Porphyromonas gingivalis. Infec Immun. 2004, 72, 3743–3751. [Google Scholar] [CrossRef]
- Yilmaz, O; Verbeke, P; Lamont, RJ; Ojcius, DM. Intercellular spreading of Porphyromonas gingivalis infection in primary gingival epithelial cells. Infect Immun. 2006, 74, 703–710. [Google Scholar] [CrossRef]
- Bui, FQ; Almeida-da-Silva, CLC; Huynh, B; Trinh, A; Liu, J; Woodward, J; et al. Association between periodontal pathogens and systemic disease. Biomedical journal. 2019, 42, 27–35. [Google Scholar] [CrossRef]
- Hajishengallis, G. Periodontitis: from microbial immune subversion to systemic inflammation. Nat Rev Immunol. 2015, 15, 30–44. [Google Scholar] [CrossRef] [PubMed]
- Makkawi, H; Hoch, S; Burns, E; Hosur, K; Hajishengallis, G; Kirschning, CJ; et al. Porphyromonas gingivalis Stimulates TLR2-PI3K Signaling to Escape Immune Clearance and Induce Bone Resorption Independently of MyD88. Front Cell Infect Microbiol 2017, 7, 359. [Google Scholar] [CrossRef]
- Almeida-da-Silva, CLC; Ramos-Junior, ES; Morandini, AC; Rocha, GDC; Marinho, Y; Tamura, AS; et al. P2X7 receptor-mediated leukocyte recruitment and Porphyromonas gingivalis clearance requires IL-1beta production and autocrine IL-1 receptor activation. Immunobiology 2019, 224, 50–59. [Google Scholar] [CrossRef]
- Ramos-Junior, ES; Morandini, AC; Almeida-da-Silva, CLC; Franco, EJ; Potempa, J; Nguyen, KA; et al. A Dual Role for P2X7 Receptor during Porphyromonas gingivalis Infection. Journal of dental research 2015, 94, 1233–1242. [Google Scholar] [CrossRef]
- Morandini, AC; Ramos-Junior, ES; Potempa, J; Nguyen, KA; Oliveira, AC; Bellio, M; et al. Porphyromonas gingivalis fimbriae dampen P2X7-dependent IL-1beta secretion. J Innate Immunity, 2014; in press. [Google Scholar] [CrossRef] [PubMed]
- Miethke, M; Pieroni, M; Weber, T; Bronstrup, M; Hammann, P; Halby, L; et al. Towards the sustainable discovery and development of new antibiotics. Nat Rev Chem. 2021, 5, 726–749. [Google Scholar] [CrossRef] [PubMed]
- Almeida-da-Silva, CLC; Sivakumar, N; Asadi, H; Chang-Chien, A; Qoronfleh, MW; Ojcius, DM; et al. Effects of Frankincense Compounds on Infection, Inflammation, and Oral Health. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Raja, AF; Ali, F; Khan, IA; Shawl, AS; Arora, DS. Acetyl-11-keto-beta-boswellic acid (AKBA); targeting oral cavity pathogens. BMC Res Notes 2011, 4, 406. [Google Scholar] [CrossRef]
- Vang, D; Moreira-Souza, ACA; Zusman, N; Moncada, G; Matshik Dakafay, H; Asadi, H; et al. Frankincense (Boswellia serrata) Extract Effects on Growth and Biofilm Formation of Porphyromonas gingivalis, and Its Intracellular Infection in Human Gingival Epithelial Cells. Current issues in molecular biology 2024, 46, 2991–3004. [Google Scholar] [CrossRef]
- Jaros, P; Timkina, E; Michailidu, J; Marsik, D; Kulisova, M; Kolouchova, I; et al. Boswellic Acids as Effective Antibacterial Antibiofilm Agents. Molecules 2022, 27. [Google Scholar] [CrossRef]
- Choi, CH; Spooner, R; DeGuzman, J; Koutouzis, T; Ojcius, DM; Yilmaz, O. Porphyromonas gingivalis-nucleoside-diphosphate-kinase inhibits ATP-induced reactive-oxygen-species via P2X7 receptor/NADPH-oxidase signalling and contributes to persistence. Cell Microbiol. 2013, 15, 961–976. [Google Scholar] [CrossRef]
- Hung, SC; Choi, CH; Said-Sadier, N; Johnson, L; Atanasova, KR; Sellami, H; et al. P2X4 assembles with P2X7 and pannexin-1 in gingival epithelial cells and modulates ATP-induced reactive oxygen species production and inflammasome activation. PLoS ONE, 2013; in press. [Google Scholar] [CrossRef]
- Nathan, C; Cunningham-Bussel, A. Beyond oxidative stress: an immunologist’s guide to reactive oxygen species. Nat Rev Immunol. 2013, 13, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Alfei, S; Schito, GC; Schito, AM; Zuccari, G. Reactive Oxygen Species (ROS)-Mediated Antibacterial Oxidative Therapies: Available Methods to Generate ROS and a Novel Option Proposal. Int J Mol Sci. 2024, 25. [Google Scholar] [CrossRef] [PubMed]
- Moreira-Souza, ACA; Almeida-da-Silva, CLC; Rangel, TP; Rocha, GDC; Bellio, M; Zamboni, DS; et al. The P2X7 Receptor Mediates Toxoplasma gondii Control in Macrophages through Canonical NLRP3 Inflammasome Activation and Reactive Oxygen Species Production. Frontiers in immunology 2017, 8, 1257. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, SA; Al-Shanon, AF; Al-Saffar, AZ; Tawang, A; Al-Obaidi, JR. Antiproliferative and cell cycle arrest potentials of 3-O-acetyl-11-keto-beta-boswellic acid against MCF-7 cells in vitro. J Genet Eng Biotechnol. 2023, 21, 75. [Google Scholar] [CrossRef]
- Li, C; He, Q; Xu, Y; Lou, H; Fan, P. Synthesis of 3-O-Acetyl-11-keto-beta-boswellic Acid (AKBA)-Derived Amides and Their Mitochondria-Targeted Antitumor Activities. ACS Omega 2022, 7, 9853–9866. [Google Scholar] [CrossRef]
- Mohamed, HRH; Ibrahim, EGS; El-Sherif, AA. Frankincense oil nanoemulsion induces selective cytotoxicity and over ROS-mediated oxidative stress and apoptotic DNA damage in Hep-G2 hepatic cancer cells. Sci Rep. 2025, 15, 32457. [Google Scholar] [CrossRef]
- Pan, D; Wang, Q; Tang, S; Wu, X; Cai, L; Wang, Z; et al. Acetyl-11-keto-beta-boswellic acid inhibits cell proliferation and growth of oral squamous cell carcinoma via RAB7B-mediated autophagy. Toxicol Appl Pharmacol. 2024, 485, 116906. [Google Scholar] [CrossRef] [PubMed]
- Altmann, A; Poeckel, D; Fischer, L; Schubert-Zsilavecz, M; Steinhilber, D; Werz, O. Coupling of boswellic acid-induced Ca2+ mobilisation and MAPK activation to lipid metabolism and peroxide formation in human leucocytes. British journal of pharmacology 2004, 141, 223–232. [Google Scholar] [CrossRef]
- Siddiqui, MZ. Boswellia serrata, a potential antiinflammatory agent: an overview. Indian J Pharm Sci. 2011, 73, 255–261. [Google Scholar]
- Krieglstein, CF; Anthoni, C; Rijcken, EJ; Laukotter, M; Spiegel, HU; Boden, SE; et al. Acetyl-11-keto-beta-boswellic acid, a constituent of a herbal medicine from Boswellia serrata resin, attenuates experimental ileitis. Int J Colorectal Dis. 2001, 16, 88–95. [Google Scholar] [CrossRef]
- Gerhardt, H; Seifert, F; Buvari, P; Vogelsang, H; Repges, R. Therapy of active Crohn disease with Boswellia serrata extract H 15. Z Gastroenterol. 2001, 39, 11–17. [Google Scholar] [CrossRef]
- Majeed, M; Majeed, S; Narayanan, NK; Nagabhushanam, K. A pilot, randomized, double-blind, placebo-controlled trial to assess the safety and efficacy of a novel Boswellia serrata extract in the management of osteoarthritis of the knee. Phytother Res. 2019, 33, 1457–1468. [Google Scholar] [CrossRef]
- Cherepanova, MO; Subotyalov, MA. Component Composition and Biological Activity of Oleo-Gum Resin from Boswellia serrata (Burseraceae). Dokl Biol Sci. 2023, 512, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Raja, AF; Ali, F; Khan, IA; Shawl, AS; Arora, DS; Shah, BA; et al. Antistaphylococcal and biofilm inhibitory activities of acetyl-11-keto-beta-boswellic acid from Boswellia serrata. BMC Microbiol. 2011, 11, 54. [Google Scholar] [CrossRef] [PubMed]
- Cho, JA; Park, E. Curcumin utilizes the anti-inflammatory response pathway to protect the intestine against bacterial invasion. Nutr Res Pract. 2015, 9, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Daglia, M. Polyphenols as antimicrobial agents. Curr Opin Biotechnol. 2012, 23, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Yang, YH; Li, W; Ren, LW; Yang, H; Zhang, YZ; Zhang, S; et al. S670, an amide derivative of 3-O-acetyl-11-keto-beta-boswellic acid, induces ferroptosis in human glioblastoma cells by generating ROS and inhibiting STX17-mediated fusion of autophagosome and lysosome. Acta Pharmacol Sin. 2024, 45, 209–222. [Google Scholar] [CrossRef]
- Nakano, K; Sasaki, S; Kataoka, T. Bioactive Evaluation of Ursane-Type Pentacyclic Triterpenoids: beta-Boswellic Acid Interferes with the Glycosylation and Transport of Intercellular Adhesion Molecule-1 in Human Lung Adenocarcinoma A549 Cells. Molecules 2022, 27. [Google Scholar] [CrossRef] [PubMed]
- Diacovich, L; Gorvel, JP. Bacterial manipulation of innate immunity to promote infection. Nat Rev Microbiol. 2010, 8, 117–128. [Google Scholar] [CrossRef]
- Schleimer, RP; Kato, A; Kern, R; Kuperman, D; Avila, PC. Epithelium: at the interface of innate and adaptive immune responses. J Allergy Clin Immunol. 2007, 120, 1279–1284. [Google Scholar] [CrossRef]
- Santana, PT; Martel, J; Lai, HC; Perfettini, JL; Kanellopoulos, JM; Young, JD; et al. Is the inflammasome relevant for epithelial cell function? Microbes Infect. 2016, 18, 93–101. [Google Scholar] [CrossRef]
- Ammon, HP. Boswellic Acids and Their Role in Chronic Inflammatory Diseases. Adv Exp Med Biol. 2016, 928, 291–327. [Google Scholar]
- Fakhri, KU; Sharma, D; Fatma, H; Yasin, D; Alam, M; Sami, N; et al. The Dual Role of Dietary Phytochemicals in Oxidative Stress: Implications for Oncogenesis, Cancer Chemoprevention, and ncRNA Regulation. Antioxidants (Basel) 2025, 14. [Google Scholar] [CrossRef]
- Zhou, C; Wang, Y; Zhang, Q; Zhou, G; Ma, X; Jiang, X; et al. Acetyl-11-Keto-Beta-Boswellic Acid Activates the Nrf2/HO-1 Signaling Pathway in Schwann Cells to Reduce Oxidative Stress and Promote Sciatic Nerve Injury Repair. Planta Med. 2023, 89, 1468–1482. [Google Scholar] [CrossRef]
- Ahmad, S; Khan, SA; Kindelin, A; Mohseni, T; Bhatia, K; Hoda, MN; et al. Acetyl-11-keto-beta-boswellic acid (AKBA) Attenuates Oxidative Stress, Inflammation, Complement Activation and Cell Death in Brain Endothelial Cells Following OGD/Reperfusion. Neuromolecular Med. 2019, 21, 505–516. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



