Submitted:
14 January 2026
Posted:
16 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
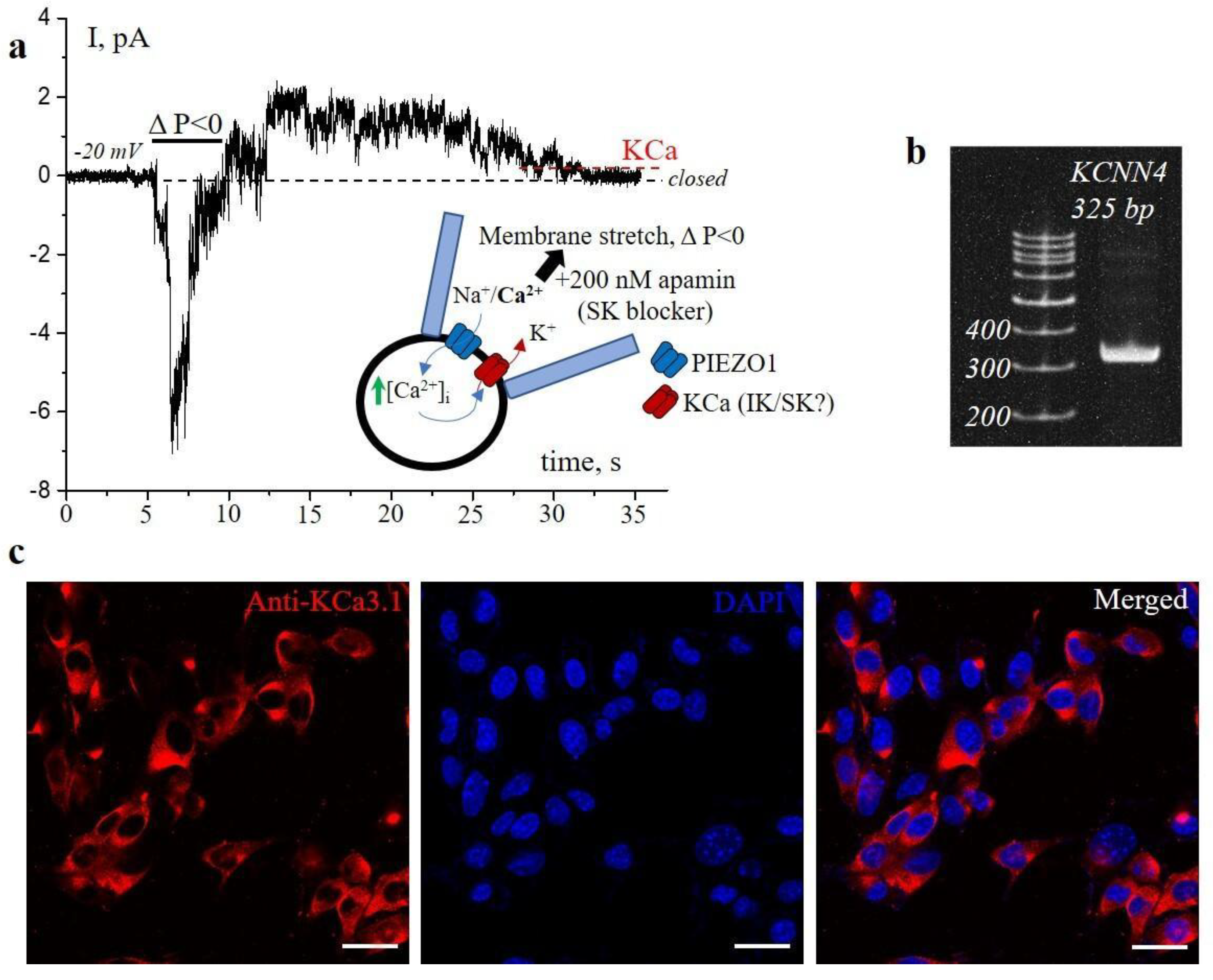
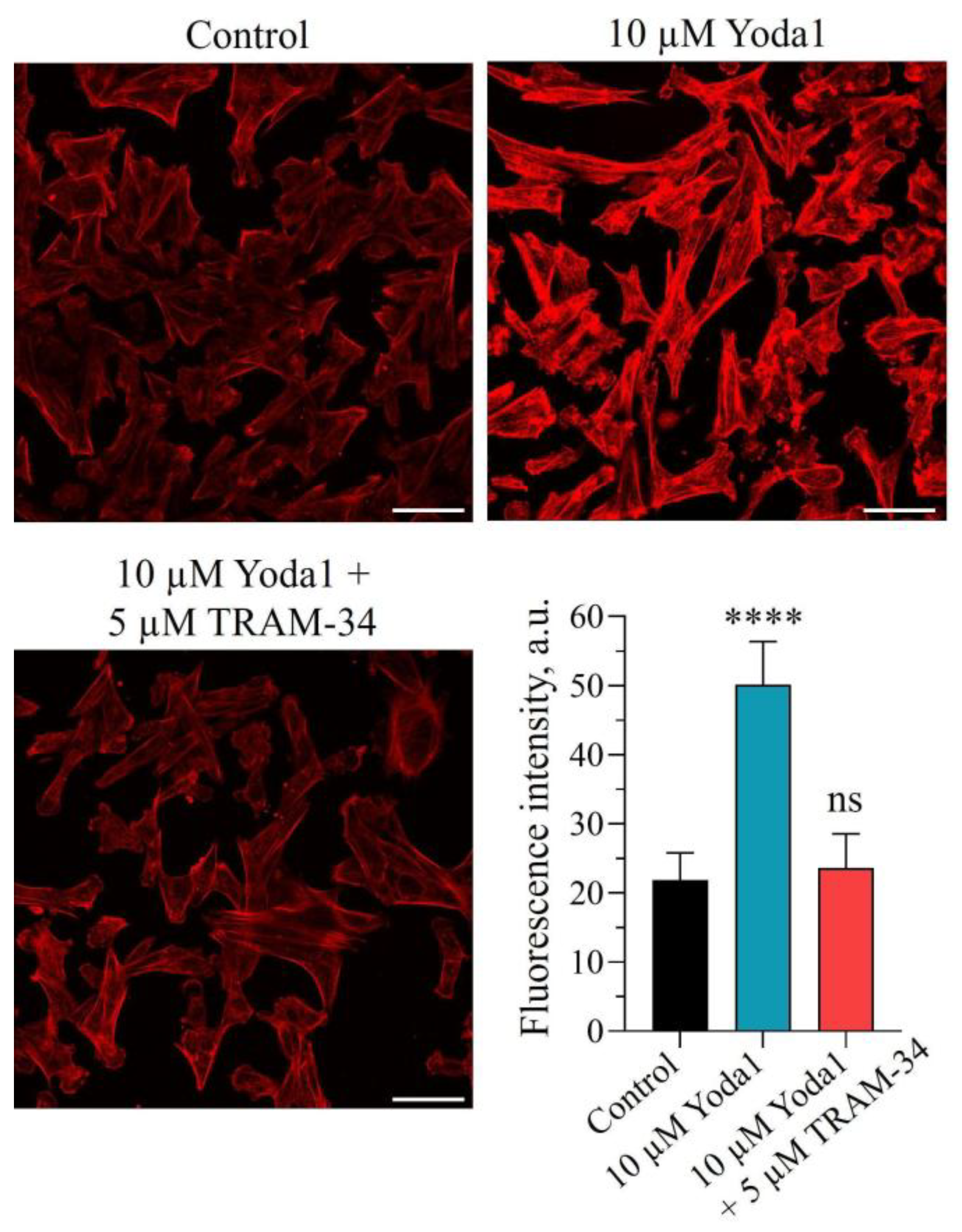
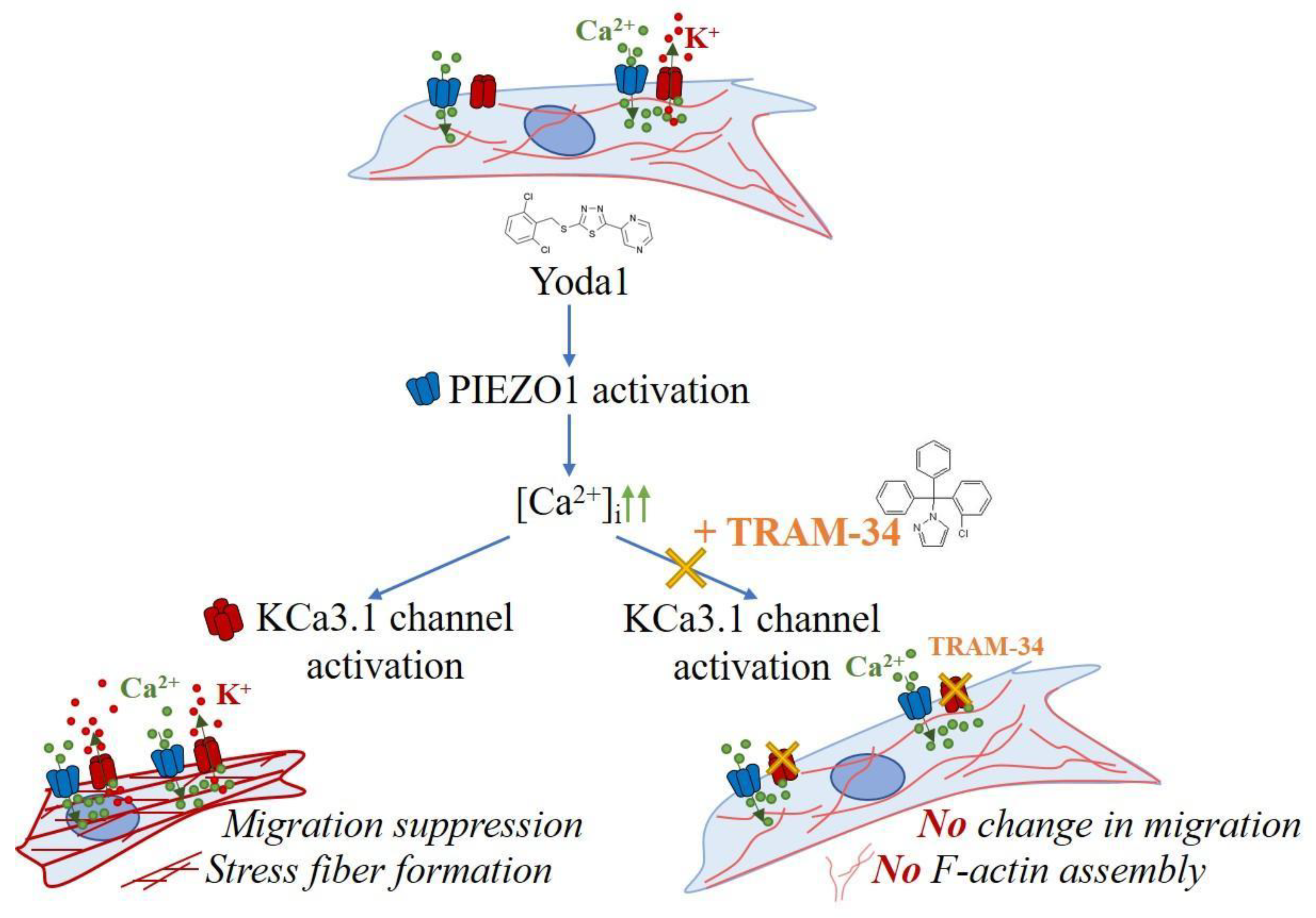
2. Results and Discussion
3. Materials and Methods
3.1. Cell Culture and Reagents
3.2. Patch-Clamp Experiments
3.3. RT-PCR
3.4. Wound Healing Migration Assay
3.5. Immunofluorescence
3.6. F-actin Labeling
3.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Xu, Y.; Wang, Y.; Yang, Y.; Fang, X.; Wu, L.; Hu, J.; Li, J.; Mei, S. Piezo1: the key regulators in central nervous system diseases. Frontiers in cellular neuroscience. 2024, 18, 1441806. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Wen, Z.; Cao, M.; Zhang, H.; Ling, SKK.; Fu, BSC.; Qin, L.; Xu, J.; Yung, PSH. The emerging role of Piezo1 in the musculoskeletal system and disease. Theranostics 2024, 14, 3963–3983. Available online: https://www.thno.org/v14p3963.htm. [CrossRef]
- Xiong, H.; Yang, J.; Guo, J.; Ma, A.; Wang, B.; Kang, Y. Mechanosensitive Piezo channels mediate the physiological and pathophysiological changes in the respiratory system. Respiratory research 2022, 23, 196. [Google Scholar] [CrossRef]
- Xiao, B. Mechanisms of mechanotransduction and physiological roles of PIEZO channels. Nature reviews. Molecular cell biology 2024, 25, 886–903. [Google Scholar] [CrossRef]
- Kuriyama, M.; Hirose, H.; Masuda, T.; Shudou, M.; Arafiles, J.V.V.; Imanishi, M.; Maekawa, M.; Hara, Y.; Futaki, S. Piezo1 activation using Yoda1 inhibits macropinocytosis in A431 human epidermoid carcinoma cells. Scientific reports 2022, 12, 6322. [Google Scholar] [CrossRef]
- Yang, X.; Zeng, H.; Wang, L.; Luo, S.; Zhou, Y. Activation of Piezo1 downregulates renin in juxtaglomerular cells and contributes to blood pressure homeostasis. Cell Biosci. 2022, 12, 197. [Google Scholar] [CrossRef]
- Lei, M.; Wang, W.; Zhang, H.; Gong, J.; Wang, Z.; Cai, H.; Yang, X.; Wang, S.; Ma, C. Cell-cell and cell-matrix adhesion regulated by Piezo1 is critical for stiffness-dependent DRG neuron aggregation. Cell reports 2023, 42, 113522. [Google Scholar] [CrossRef]
- Tsuchiya, M.; Hara, Y.; Okuda, M.; Itoh, K.; Nishioka, R.; Shiomi, A.; Nagao, K.; Mori, M.; Mori, Y.; Ikenouchi, J.; Suzuki, R.; Tanaka, M.; Ohwada, T.; et al. Cell surface flip-flop of phosphatidylserine is critical for PIEZO1-mediated myotube formation. Nature communications 2018, 9, 2049. [Google Scholar] [CrossRef]
- Vasileva, V.Y.; Sudarikova, A.V.; Chubinskiy-Nadezhdin, V.I. Functional coupling of Piezo1 channels and Ca2+-activated ion channels in the plasma membrane: fine-tunable interplay with wide-range signaling effects. American journal of physiology. Cell physiology 2025, 328, C1338–C1345. [Google Scholar] [CrossRef]
- Chubinskiy-Nadezhdin, V.I.; Negulyaev, Y.A.; Morachevskaya, E.A. Functional coupling of ion channels in cellular mechanotransduction. Biochemical and biophysical research communications 2014, 451, 421–424. [Google Scholar] [CrossRef]
- Chubinskiy-Nadezhdin, V.I.; Vasileva, V.Y.; Vassilieva, I.O.; Sudarikova, A.V.; Morachevskaya, E.A.; Negulyaev, Y.A. Agonist-induced Piezo1 activation suppresses migration of transformed fibroblasts. Biochemical and biophysical research communications 2019, 514, 173–179. [Google Scholar] [CrossRef]
- Grissmer, S.; Nguyen, A.N.; Cahalan, M.D. Calcium-activated potassium channels in resting and activated human T lymphocytes. Expression levels, calcium dependence, ion selectivity, and pharmacology. J Gen Physiol. 1993, 102, 601–630. [Google Scholar] [CrossRef]
- Wulff, H.; Kolski-Andreaco, A.; Sankaranarayanan, A.; Sabatier, J.M.; Shakkottai, V. Modulators of small- and intermediate-conductance calcium-activated potassium channels and their therapeutic indications. Current medicinal chemistry 2007, 14, 1437–1457. [Google Scholar] [CrossRef]
- Urrego, D.; Tomczak, A.P.; Zahed, F.; Stühmer, W.; Pardo, L.A. Potassium channels in cell cycle and cell proliferation. Philos Trans R Soc Lond B Biol Sci. 2014, 369, 20130094. [Google Scholar] [CrossRef]
- Ouadid-Ahidouch, H.; Ahidouch, A. K(+) channels and cell cycle progression in tumor cells. Front Physiol. 2013, 4, 220. [Google Scholar] [CrossRef]
- Catacuzzeno, L.; Fioretti, B.; Franciolini, F. Expression and Role of the Intermediate-Conductance Calcium-Activated Potassium Channel KCa3.1 in Glioblastoma. Journal of signal transduction 2012, 421564. [Google Scholar] [CrossRef]
- Carrisoza-Gaytan, R.; Mutchler, S.M.; Carattino, F.; Soong, J.; Dalghi, M.G.; Wu, P.; Wang, W.; Apodaca, G.; Satlin, L.M.; Kleyman, T.R. PIEZO1 is a distal nephron mechanosensor and is required for flow-induced K+ secretion. The Journal of clinical investigation 2024, 134, e174806. [Google Scholar] [CrossRef]
- Catacuzzeno, L.; Michelucci, A. Emerging connections between Piezo1 and BK channels in vascular smooth muscle cells. In European journal of physiology; Pflugers Archiv, 2024; Volume 476, pp. 1475–1477. [Google Scholar] [CrossRef]
- Jakob, D.; Klesen, A.; Allegrini, B.; Darkow, E.; Aria, D.; Emig, R.; Chica, A.S.; Rog-Zielinska, E.A.; Guth, T.; Beyersdorf, F.; Kari, F.A.; Proksch, S.; Hatem, S.N.; Karck, M.; Künzel, S.R.; Guizouarn, H.; Schmidt, C.; Kohl, P.; Ravens, U.; Peyronnet, R. Piezo1 and BKCa channels in human atrial fibroblasts: Interplay and remodelling in atrial fibrillation. Journal of molecular and cellular cardiology 2021, 158, 49–62. [Google Scholar] [CrossRef]
- Chubinskiy-Nadezhdin, V.I.; Vasileva, V.Y.; Pugovkina, N.A.; Vassilieva, I.O.; Morachevskaya, E.A.; Nikolsky, N.N.; Negulyaev, Y.A. Local calcium signalling is mediated by mechanosensitive ion channels in mesenchymal stem cells. Biochemical and biophysical research communications 2017, 482, 563–568. [Google Scholar] [CrossRef] [PubMed]
- Rossi, A.M.; Hirschhorn, R.R. Expression of growth-regulated genes in normal and SV40 transformed hamster fibroblasts. J Cell Biochem. 1991, 47, 165–173. [Google Scholar] [CrossRef]
- Chubinskiy-Nadezhdin, V.I.; Efremova, T.N.; Negulyaev, Y.A.; Morachevskaya, E.A. Coupled Activation of Mechanosensitive and Calcium-Dependent Potassium Channels in 3T3 and 3T3-SV40 Cells. Cell Tiss. Biol. 2018, 12, 231–237. [Google Scholar] [CrossRef]
- Petkova-Kirova, P.; Murciano, N.; Iacono, G.; Jansen, J.; Simionato, G.; Qiao, M.; Van der Zwaan, C.; Rotordam, M.G.; John, T.; Hertz, L.; Hoogendijk, A.J.; Becker, N.; Wagner, C.; Von Lindern, M.; Egee, S.; Van den Akker, E.; Kaestner, L. The Gárdos Channel and Piezo1 Revisited: Comparison between Reticulocytes and Mature Red Blood Cells. International journal of molecular sciences 2024, 25, 1416. [Google Scholar] [CrossRef]
- Michelucci, A.; Sforna, L.; Di Battista, A.; Franciolini, F.; Catacuzzeno, L. Ca2+ -activated K+ channels regulate cell volume in human glioblastoma cells. Journal of cellular physiology 2023, 238, 2120–2134. [Google Scholar] [CrossRef] [PubMed]
- Mulhall, E.M.; Gharpure, A.; Lee, R.M.; Dubin, A.E.; Aaron, J.S.; Marshall, K.L.; Spencer, K.R.; Reiche, M.A.; Henderson, S.C.; Chew, T.L.; Patapoutian, A. Direct observation of the conformational states of PIEZO1. Nature 2023, 620, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Jetta, D.; Shireen, T.; Hua, S.Z. Epithelial cells sense local stiffness via Piezo1 mediated cytoskeletal reorganization. Frontiers in cell and developmental biology 2023, 11, 1198109. [Google Scholar] [CrossRef]
- Atcha, H.; Jairaman, A.; Holt, J.R.; Meli, V.S.; Nagalla, R.R.; Veerasubramanian, P.K.; Brumm, K.T.; Lim, H.E.; Othy, S.; Cahalan, M.D.; Pathak, M.M.; Liu, W.F. Mechanically activated ion channel Piezo1 modulates macrophage polarization and stiffness sensing. Nat Commun. 2021, 12, 3256. [Google Scholar] [CrossRef]
- Roach, K.M.; Bradding, P. Ca2+ signalling in fibroblasts and the therapeutic potential of KCa3.1 channel blockers in fibrotic diseases. British journal of pharmacology 2020, 177, 1003–1024. [Google Scholar] [CrossRef] [PubMed]
- Bonito, B.; Sauter, D.R.P.; Schwab, A.; Djamgoz, M.B.; Novak, I. KCa3.1 (IK) modulates pancreatic cancer cell migration, invasion and proliferation: anomalous effects on TRAM-34. Pflugers Arch - Eur J Physiol. 2016, 468, 1865–1875. [Google Scholar] [CrossRef]
- Cruse, G.; Singh, S.R.; Duffy, S.M.; Doe, C.; Saunders, R.; Brightling, C.E.; Bradding, P. Functional KCa3.1 K+ channels are required for human fibrocyte migration. The Journal of allergy and clinical immunology 2011, 128, 1303–1309.e2. [Google Scholar] [CrossRef]
- Rapetti-Mauss, R.; Picard, V.; Guitton, C.; Ghazal, K.; Proulle, V.; Badens, C.; Soriani, O.; Garçon, L.; Guizouarn, H. Red blood cell Gardos channel (KCNN4): the essential determinant of erythrocyte dehydration in hereditary xerocytosis. Haematologica 2017, 102, e415–e418. [Google Scholar] [CrossRef]
- Guéguinou, M.; Chantôme, A.; Fromont, G.; Bougnoux, P.; Vandier, C.; Potier-Cartereau, M. KCa and Ca(2+) channels: the complex thought. Biochim Biophys Acta. 2014, 1843, 2322–2333. [Google Scholar] [CrossRef] [PubMed]
- Morishita, K.; Watanabe, K.; Ichijo, H. Cell volume regulation in cancer cell migration driven by osmotic water flow. Cancer Sci. 2019, 110, 2337–2347. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



