Submitted:
14 January 2026
Posted:
15 January 2026
You are already at the latest version
Abstract
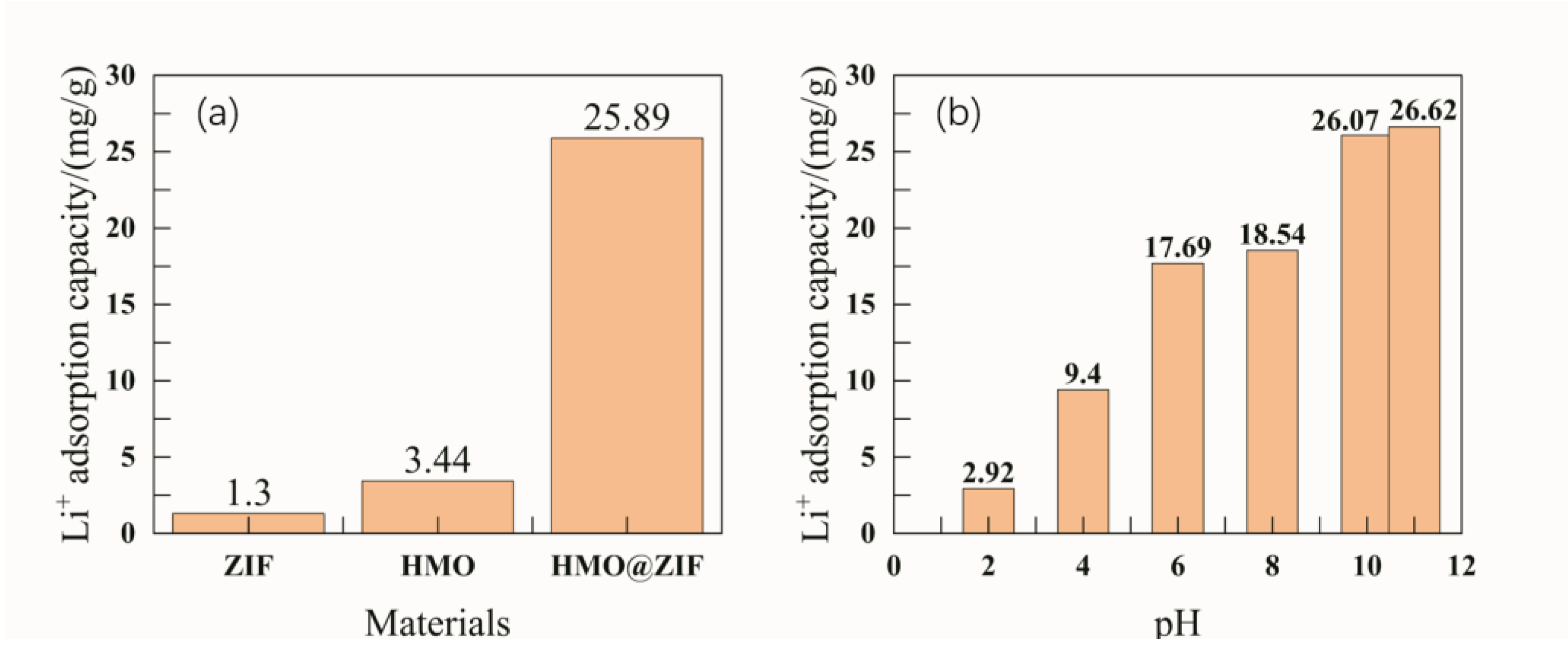
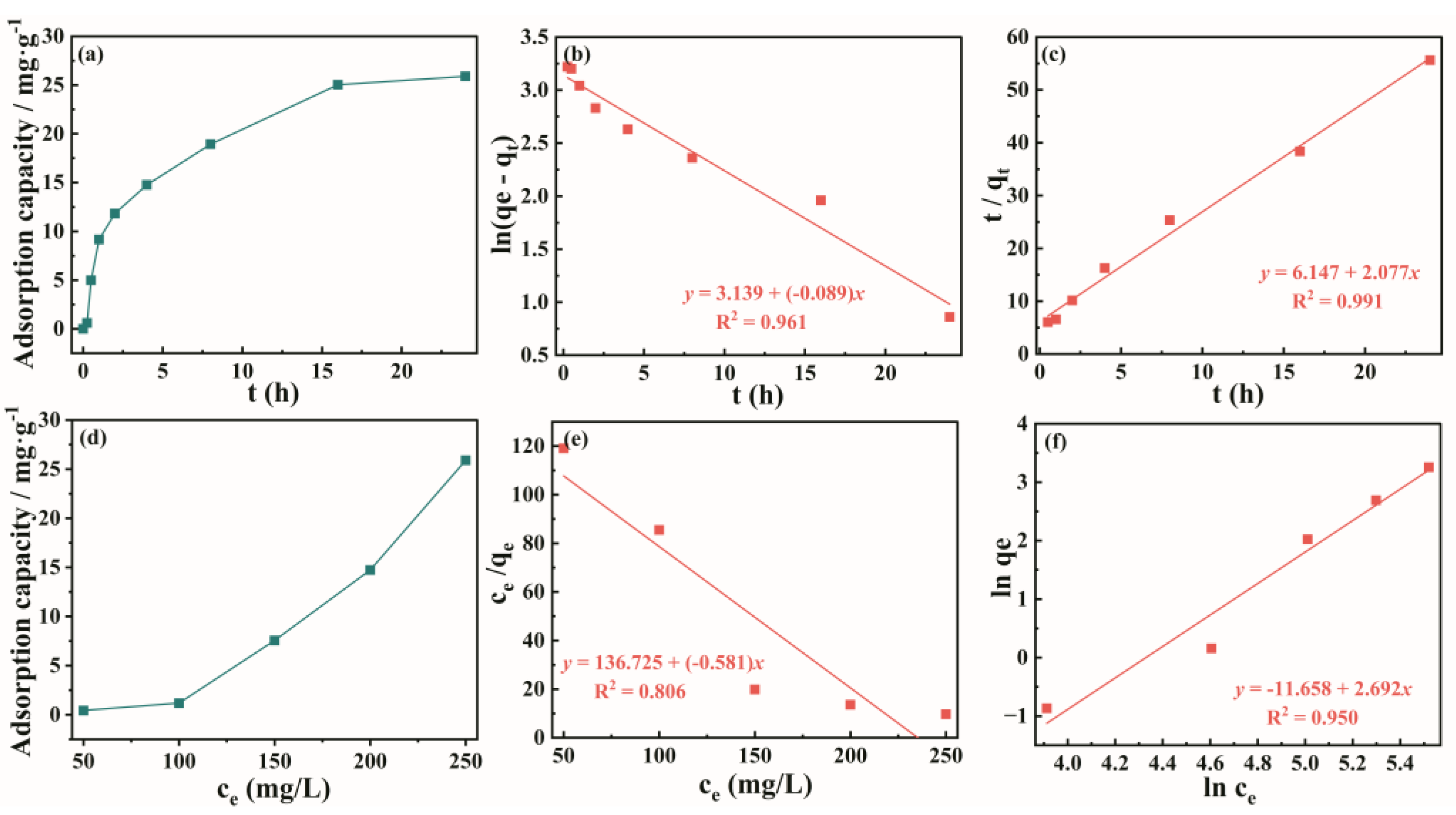
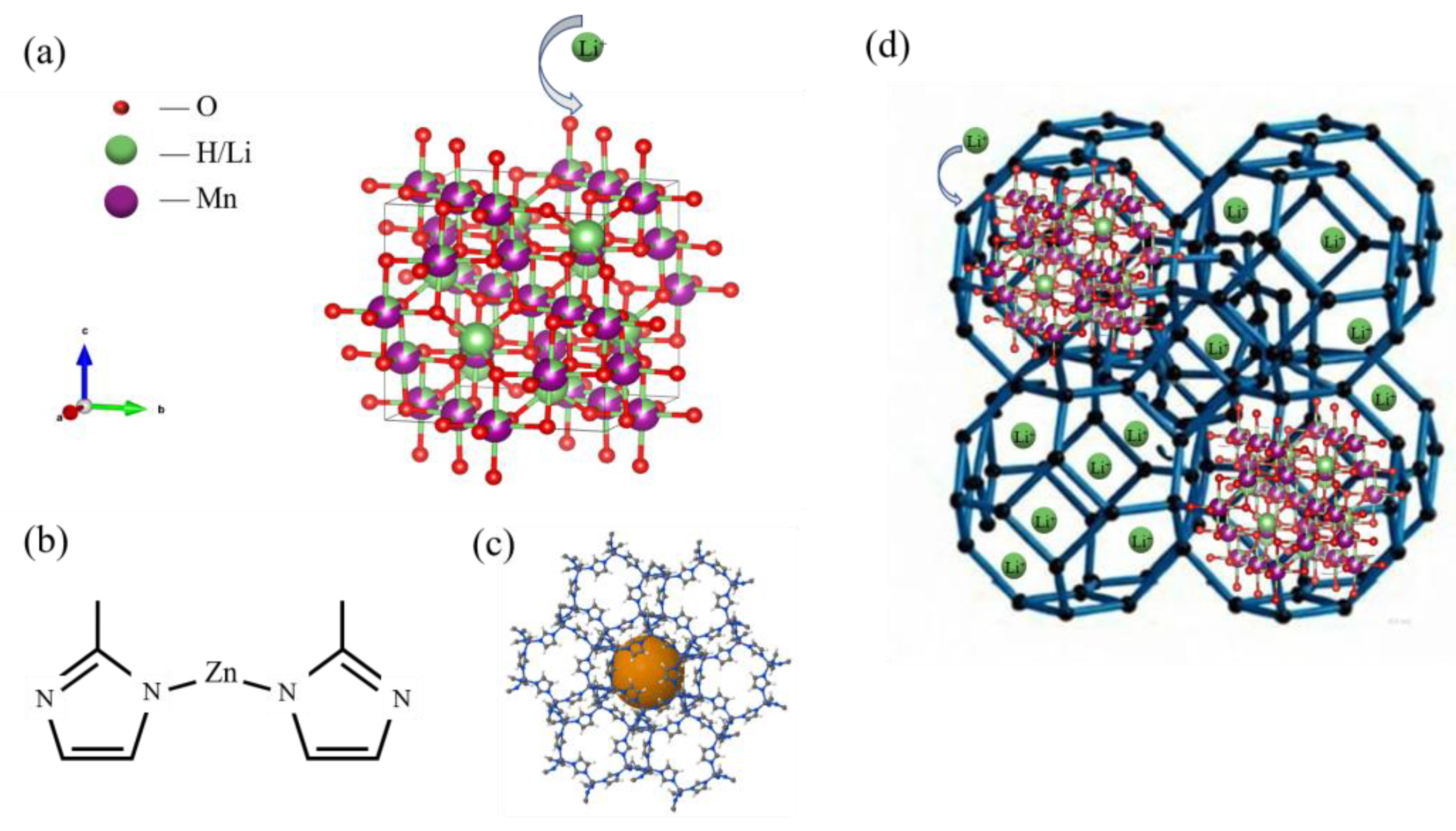
The critical role of lithium in powering the new energy economy necessitates prioritizing efficient extraction methods. This study investigates a novel zeolitic imidazolate framework (ZIF-8)-coated manganese-based lithium ion sieve (LIS) for enhanced lithium recovery. The precursor of LIS, Li1.6Mn1.6O4, was synthesized via the hydrothermal method, followed by acid pickling to obtain the spinel lithium ion sieve H1.6Mn1.6O4. The material was then immersed in a 2-methylimidazole/Zn(NO3)2 solution, undergoing ultrasonic-assisted hydrothermal growth to form ZIF@H1.6Mn1.6O4 composites. Under optimized conditions (30 °C, pH=11, 24 h), the composite demonstrated superior lithium extraction performance compared to single-phase adsorbents, reaching 26.44 mg/g at the solution with 250 mg/L Li+. The adsorption capacity of the composite increased with Li+ concentration and reaction time. The adsorption kinetics followed a pseudo-second-order kinetic model and is dominated by chemisorption.
Keywords:
1. Introduction
2. Materials and Methods
2.1. The Preparation of H1.6Mn1.6O4
2.2. The Preparation of ZIF-8@H1.6Mn1.6O4
2.3. Lithium Ion Adsorption Process
2.4. Characterization
3. Results
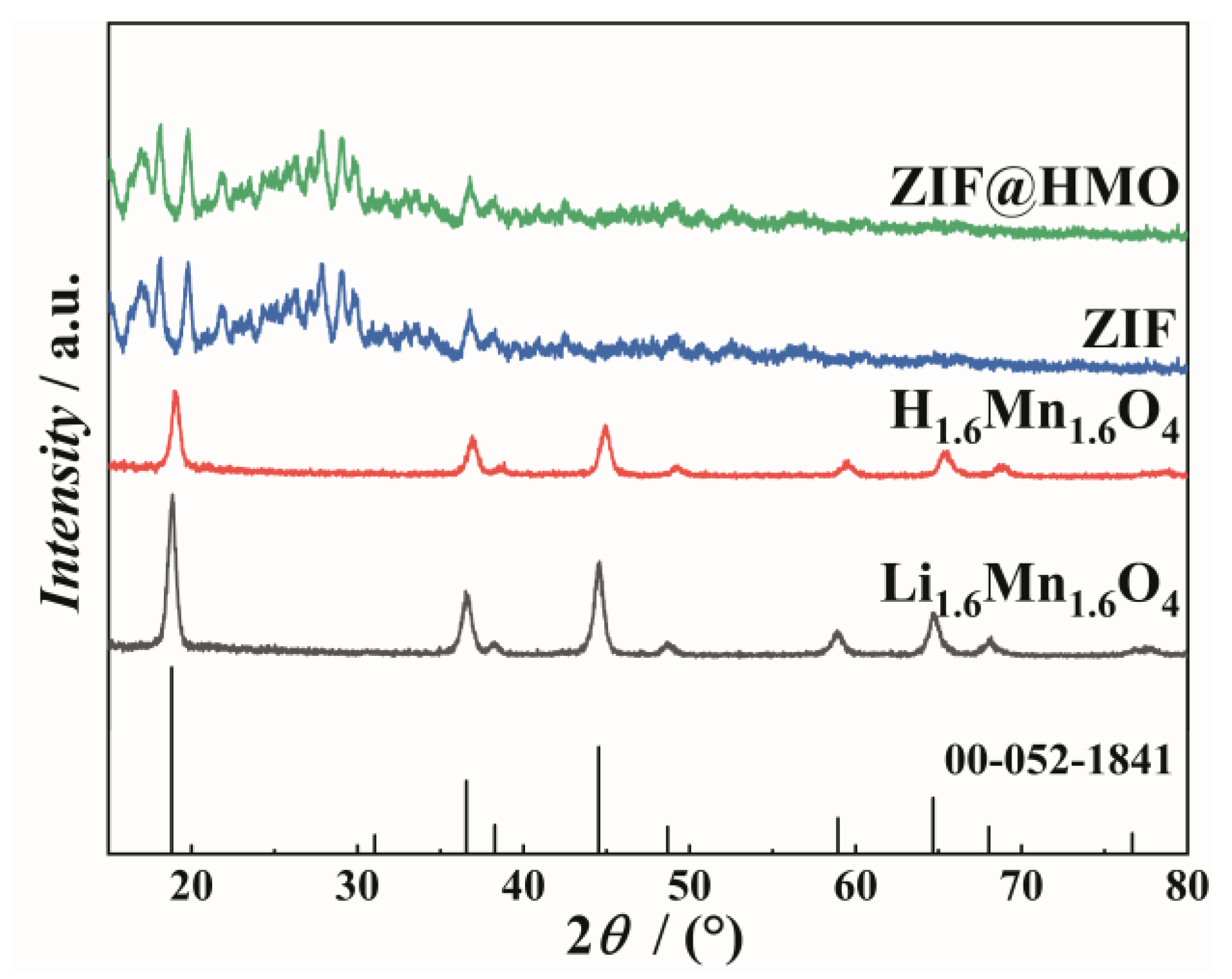
3.1. XRD Patterns of Materials
3.2. The Morphology of Ion Sieves
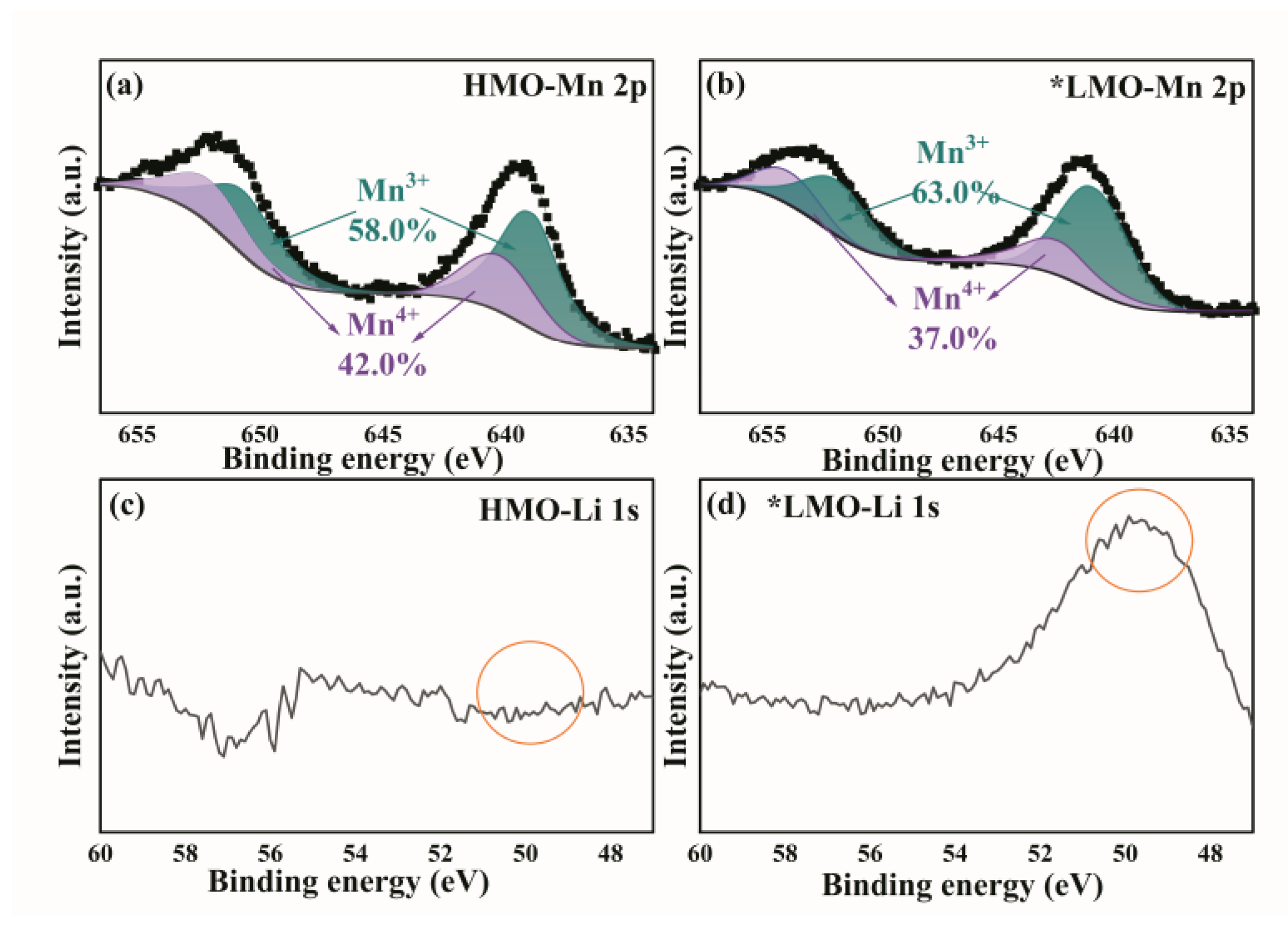
3.3. XPS Results Before and After Delithiation of Adsorbent Materials
3.4. Adsorption Performance of ZIF-8@H1.6Mn1.6O4
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HMO | Protoned Manganese Oxide |
| LIS | Lithium Ion Sieve |
| LMO | Lithium Manganese Oxide |
| MOFs | Metal-organic framework materials |
| ZIFs | Zeolitic Imidazolate Frameworks |
References
- Chitrakar, R.; Kanoh, H.; Miyai, Y.; Ooi, K. A New Type of Manganese Oxide (MnO2·0.5H2O) Derived from Li1.6Mn1.6O4 and Its Lithium Ion-Sieve Properties. Chem. Mater. 2000, 12, 3151–3157. [Google Scholar] [CrossRef]
- Chitrakar, R.; Kanoh, H.; Miyai, Y.; Ooi, K. Recovery of Lithium from Seawater Using Manganese Oxide Adsorbent (H1.6Mn1.6O4) Derived from Li1.6Mn1.6O4. Ind. Eng. Chem. Res. 2001, 40, 2054–2058. [Google Scholar] [CrossRef]
- Gao, A.; Hou, X.; Sun, Z.; Li, S.; Li, H.; Zhang, J. Lithium-Desorption Mechanism in LiMn2O4, Li1.33Mn1.67O4, and Li1.6Mn1.6O4 According to Precisely Controlled Acid Treatment and Density Functional Theory Calculations. J. Mater. Chem. A 2019, 7, 20878–20890. [Google Scholar] [CrossRef]
- Arabolla Rodríguez, R.; Della Santina Mohallem, N.; Avila Santos, M.; Sena Costa, D. A.; Andrey Montoro, L.; Mosqueda Laffita, Y.; Tavera Carrasco, L. A.; Perez-Cappe, E. L. Unveiling the Role of Mn-Interstitial Defect and Particle Size on the Jahn-Teller Distortion of the LiMn2O4 Cathode Material. J. Power Sources 2021, 490, 229519. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, P.; Cao, Y.-L.; Xie, Y.-L. Co Ion-Modified Li1.6Mn1.6O4 with Enhanced Li+ Adsorption Performance and Cyclic Stability. J. Mater. Sci. 2025. [Google Scholar] [CrossRef]
- Cho, J.; Kim, T.-J.; Kim, Y. J.; Park, B. Complete Blocking of Mn3+ Ion Dissolution from a LiMn2O4 Spinel Intercalation Compound by Co3O4 Coating. Chem. Commun. 2001, No. 12, 1074–1075. [Google Scholar] [CrossRef]
- Phan, A.; Doonan, C. J.; Uribe-Romo, F. J.; Knobler, C. B.; O’Keeffe, M.; Yaghi, O. M. Synthesis, Structure, and Carbon Dioxide Capture Properties of Zeolitic Imidazolate Frameworks. Acc. Chem. Res. 2010, 43, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Fathinia, S.; Mehdi Salarirad, M. Superior Selective Adsorption of Lithium Using Hierarchical Granular Nanostructures (PAN@LIS-MnO2@ZIF-8) from Its Low-Grade Aqueous Sources. Sep. Purif. Technol. 2025, 359, 130553. [Google Scholar] [CrossRef]
- Zhang, C.; Mu, Y.; Zhang, W.; Zhao, S.; Wang, Y. PVC-Based Hybrid Membranes Containing Metal-Organic Frameworks for Li+/Mg2+ Separation. J. Membr. Sci. 2020, 596, 117724. [Google Scholar] [CrossRef]
- Guo, Y.; Ying, Y.; Mao, Y.; Peng, X.; Chen, B. Polystyrene Sulfonate Threaded through a Metal-Organic Framework Membrane for Fast and Selective Lithium-Ion Separation. Angew. Chem. Int. Ed Engl. 2016, 55, 15120–15124. [Google Scholar] [CrossRef] [PubMed]
- Ou, R.; Zhang, H.; Wei, J.; Kim, S.; Wan, L.; Nguyen, N. S.; Hu, Y.; Zhang, X.; Simon, G. P.; Wang, H. Thermoresponsive Amphoteric Metal–Organic Frameworks for Efficient and Reversible Adsorption of Multiple Salts from Water. Adv. Mater. 2018, 30, 1802767. [Google Scholar] [CrossRef] [PubMed]
- Thackeray, M. M. Manganese Oxides for Lithium Batteries. Prog. Solid State Chem. 1997, 25, 1–71. [Google Scholar] [CrossRef]
- Gummow, R. J.; de Kock, A.; Thackeray, M. M. Improved Capacity Retention in Rechargeable 4 V Lithium/Lithium-Manganese Oxide (Spinel) Cells. Solid State Ion. 1994, 69, 59–67. [Google Scholar] [CrossRef]
- Chitrakar, R.; Kanoh, H.; Miyai, Y.; Ooi, K. Recovery of Lithium from Seawater Using Manganese Oxide Adsorbent (H1.6Mn1.6O4) Derived from Li1.6 Mn1.6 O4. Ind. Eng. Chem. Res. 2001, 40, 2054–2058. [Google Scholar] [CrossRef]
- Wang, C.; Liu, X.; Keser Demir, N.; Chen, J. P.; Li, K. Applications of Water Stable Metal–Organic Frameworks. Chem. Soc. Rev. 2016, 45, 5107–5134. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Wu, M.; Guo, Y.; Mamrol, N.; Yang, X.; Gao, C.; Van Der Bruggen, B. Metal-Organic Framework Based Membranes for Selective Separation of Target Ions. J. Membr. Sci. 2021, 634, 119407. [Google Scholar] [CrossRef]
- Razmjou, A.; Asadnia, M.; Hosseini, E.; Habibnejad Korayem, A.; Chen, V. Design Principles of Ion Selective Nanostructured Membranes for the Extraction of Lithium Ions. Nat. Commun. 2019, 10, 5793. [Google Scholar] [CrossRef] [PubMed]
- Daiguji, H. Ion Transport in Nanofluidic Channels. Chem. Soc. Rev. 2010, 39, 901–911. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



