Submitted:
12 January 2026
Posted:
14 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
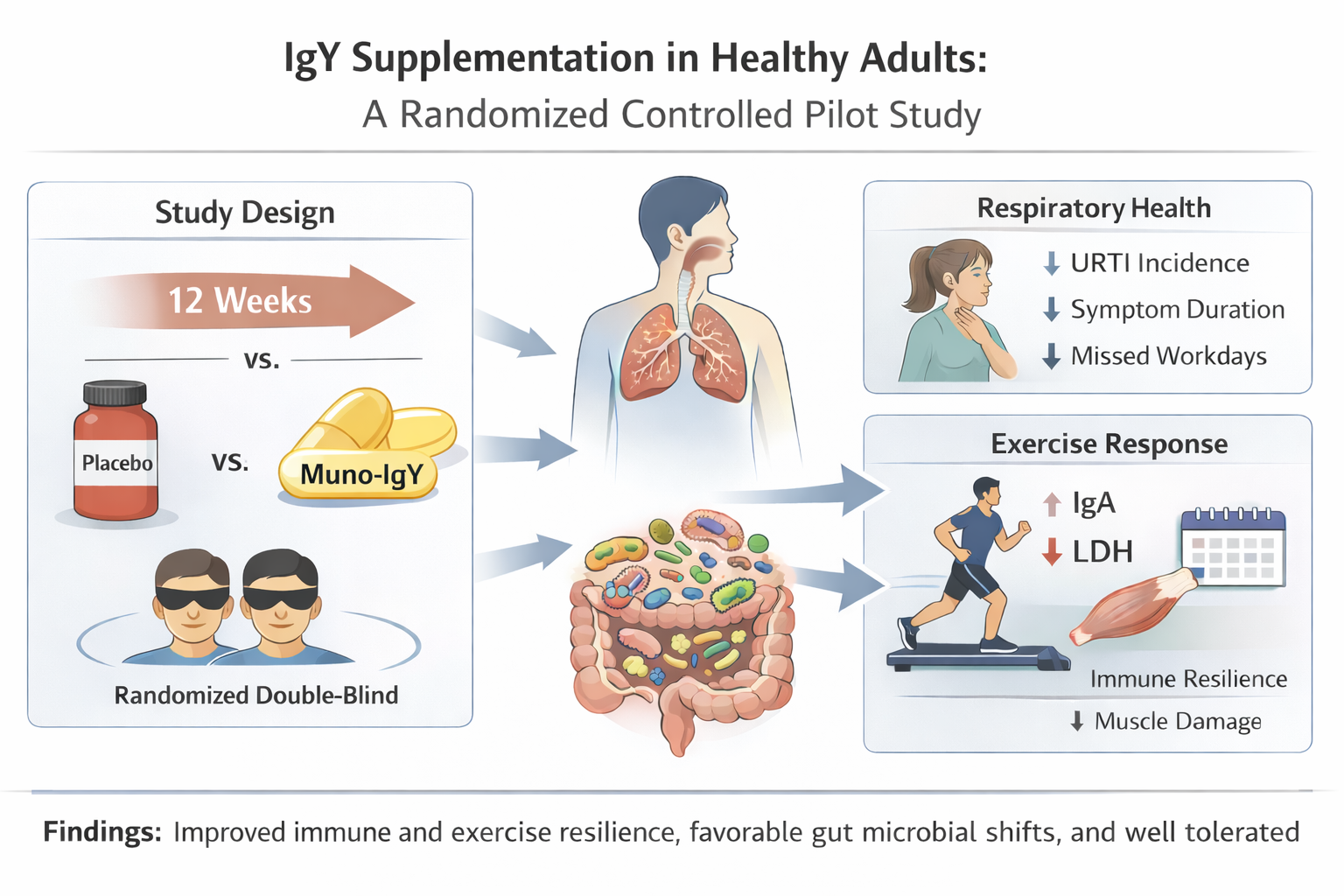
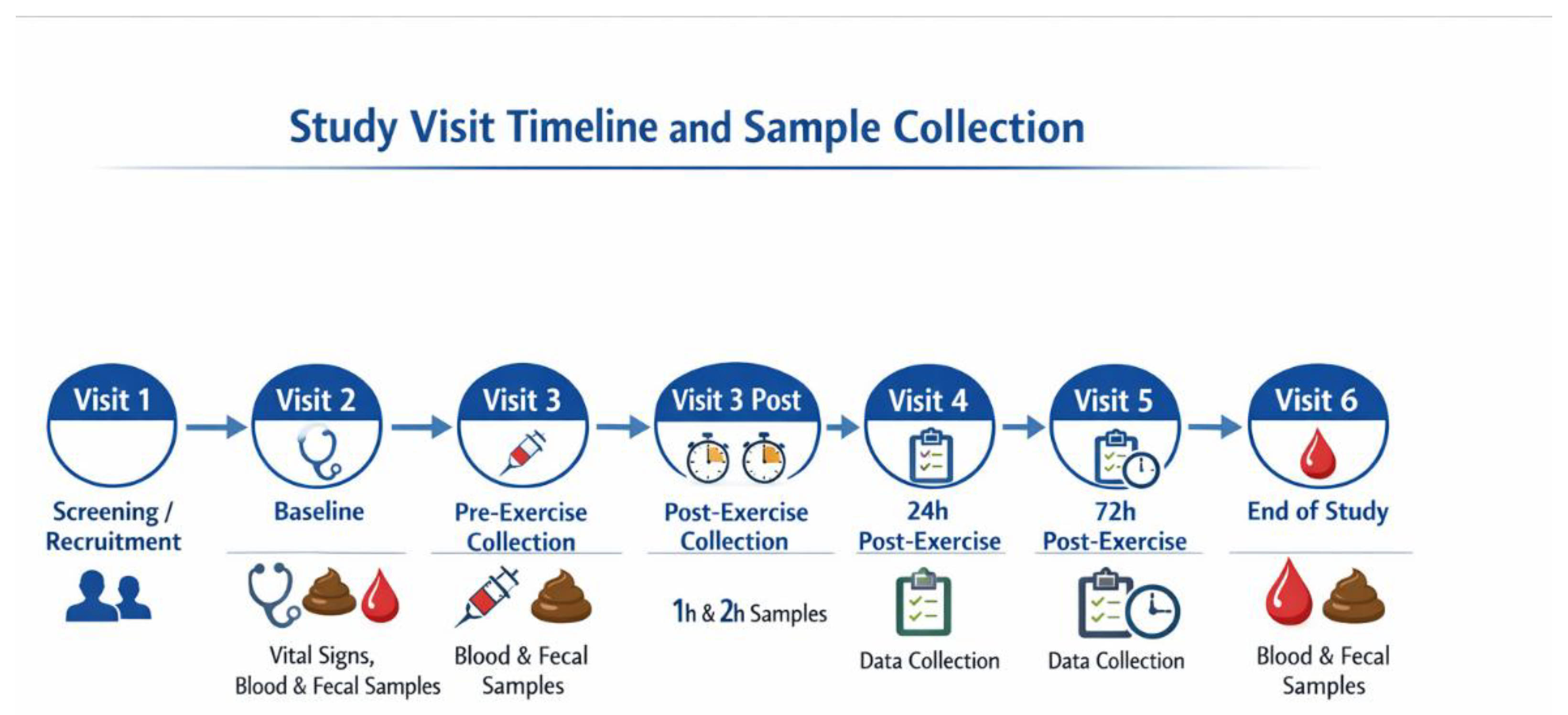
2.1. Study Design, Participants and Data Collection
2.2. Safety Assessments on Muno-IgY supplementation
2.2.1. Height, Weight, and Body Mass Index (BMI)
2.3. Effect of Muno-IgY on Immune Responses
2.3.1. Incidence, Severity, and Duration of Upper Respiratory Tract Infections
2.3.2. Serum IgA concentration
2.3.3. Inflammatory Biomarkers
2.4 Effect of Muno-IgY on Post-Exercise Inflammatory, Immunity and Muscle Damage Response
2.5 Effect of Muno-IgY supplementation on Gut Microbiome
3. Statistical Analysis
4. Results
4.1. Study population and Baseline characteristics
4.2. Effect of Muno-IgY on Anthropometrics, Vital Signs, and Clinical Laboratory Safety
4.3. Adverse Events and Safety Monitoring of Muno-IgY
4.4. Effect of Muno-IgY on Immune Responses
4.4.1. Incidence, Severity, and Duration of Upper Respiratory Tract Infections
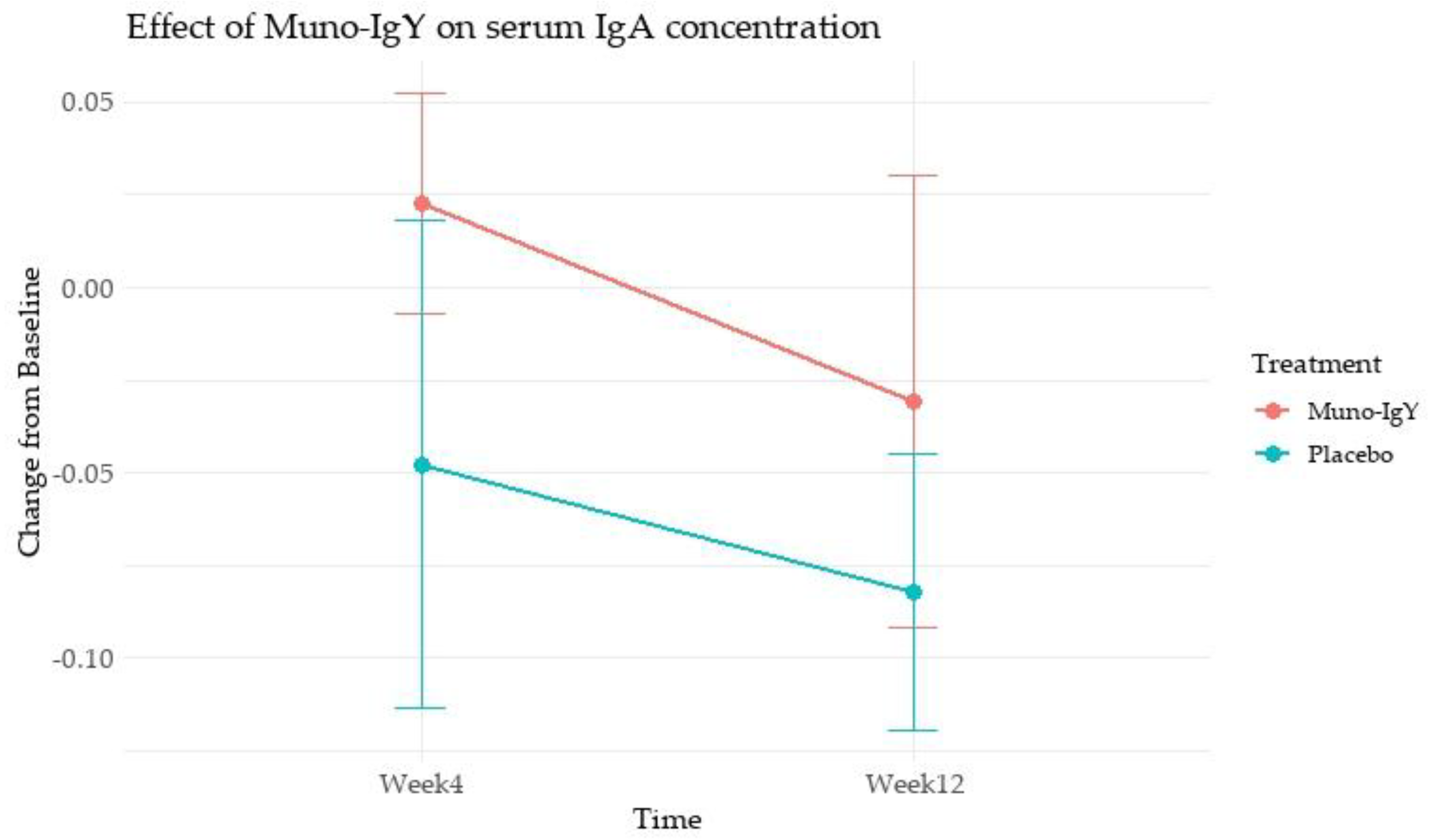
4.4.2 Serum IgA concentration
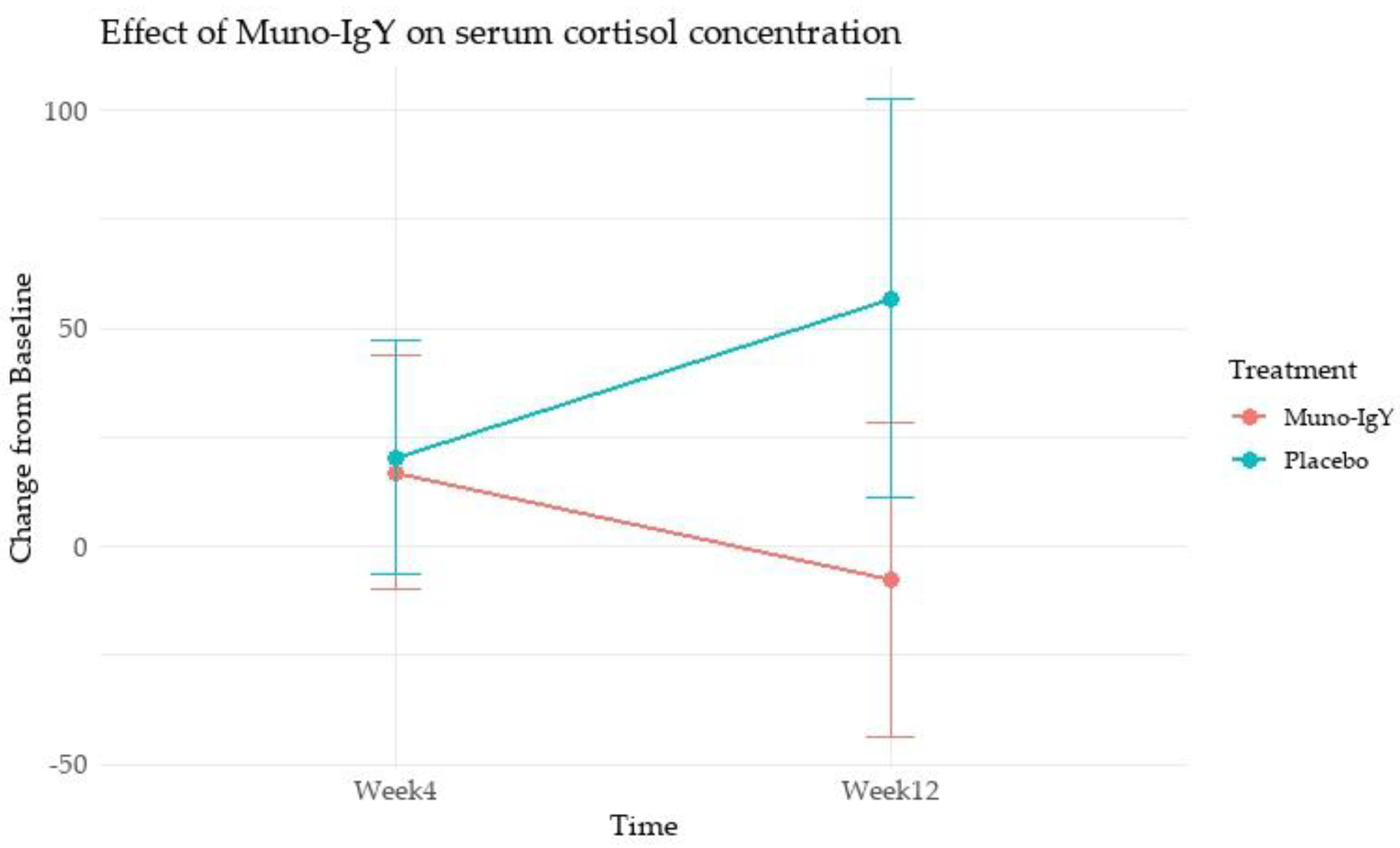
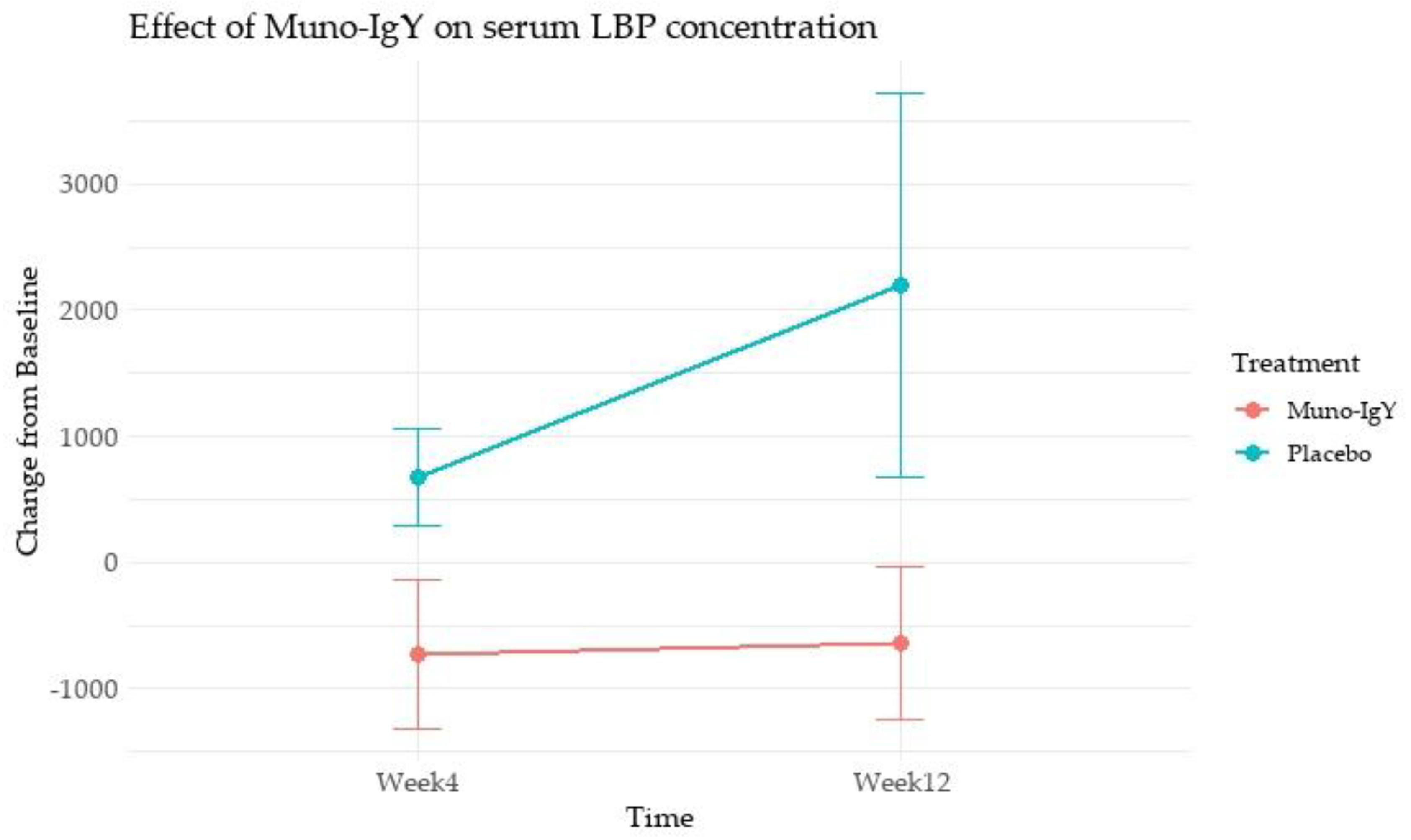
4.4.3 Inflammatory Biomarkers
4.5. Effect of Muno-IgY on Post-Exercise Inflammatory, Immunity and Muscle Damage Response
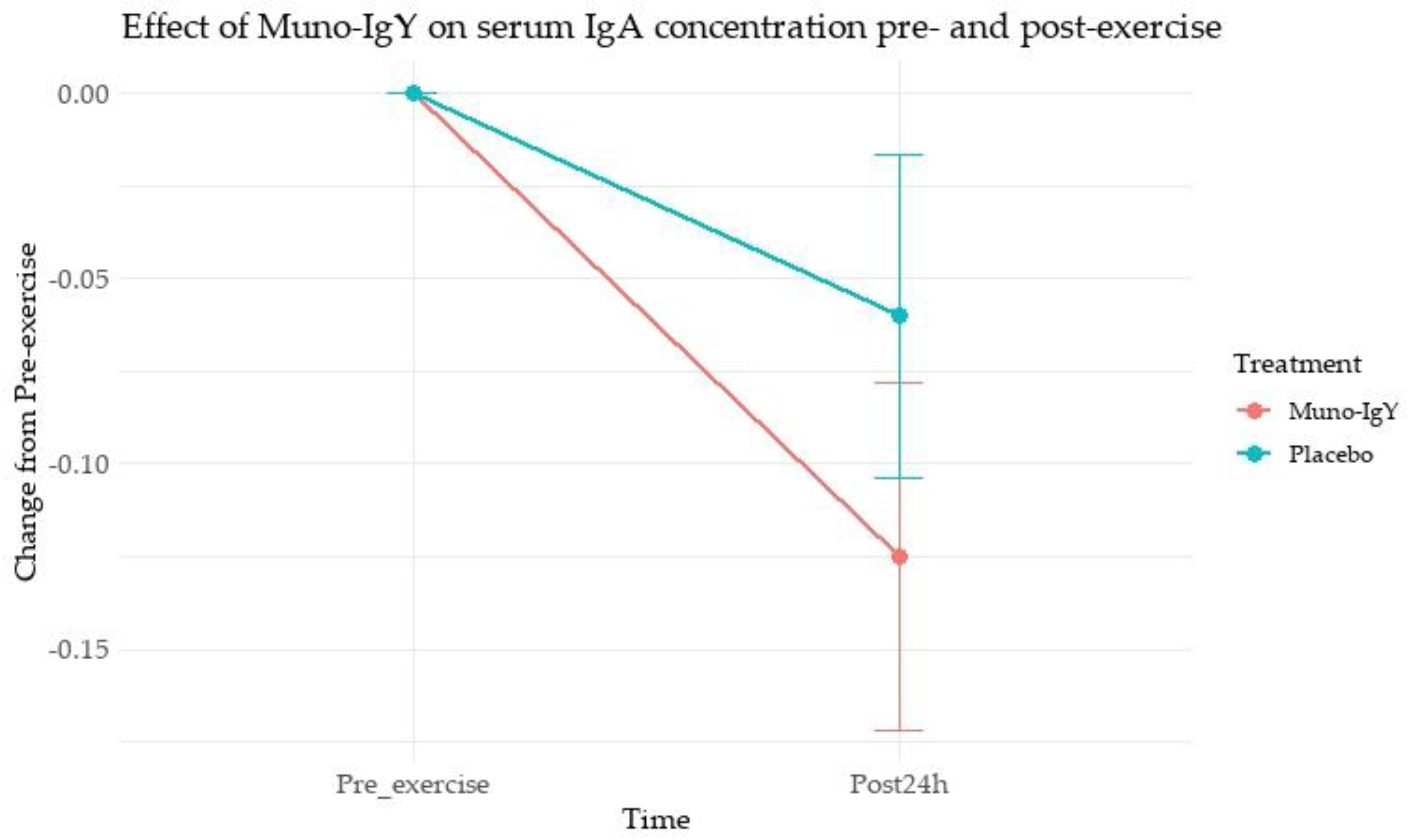
4.5.1 Serum IgA concentration
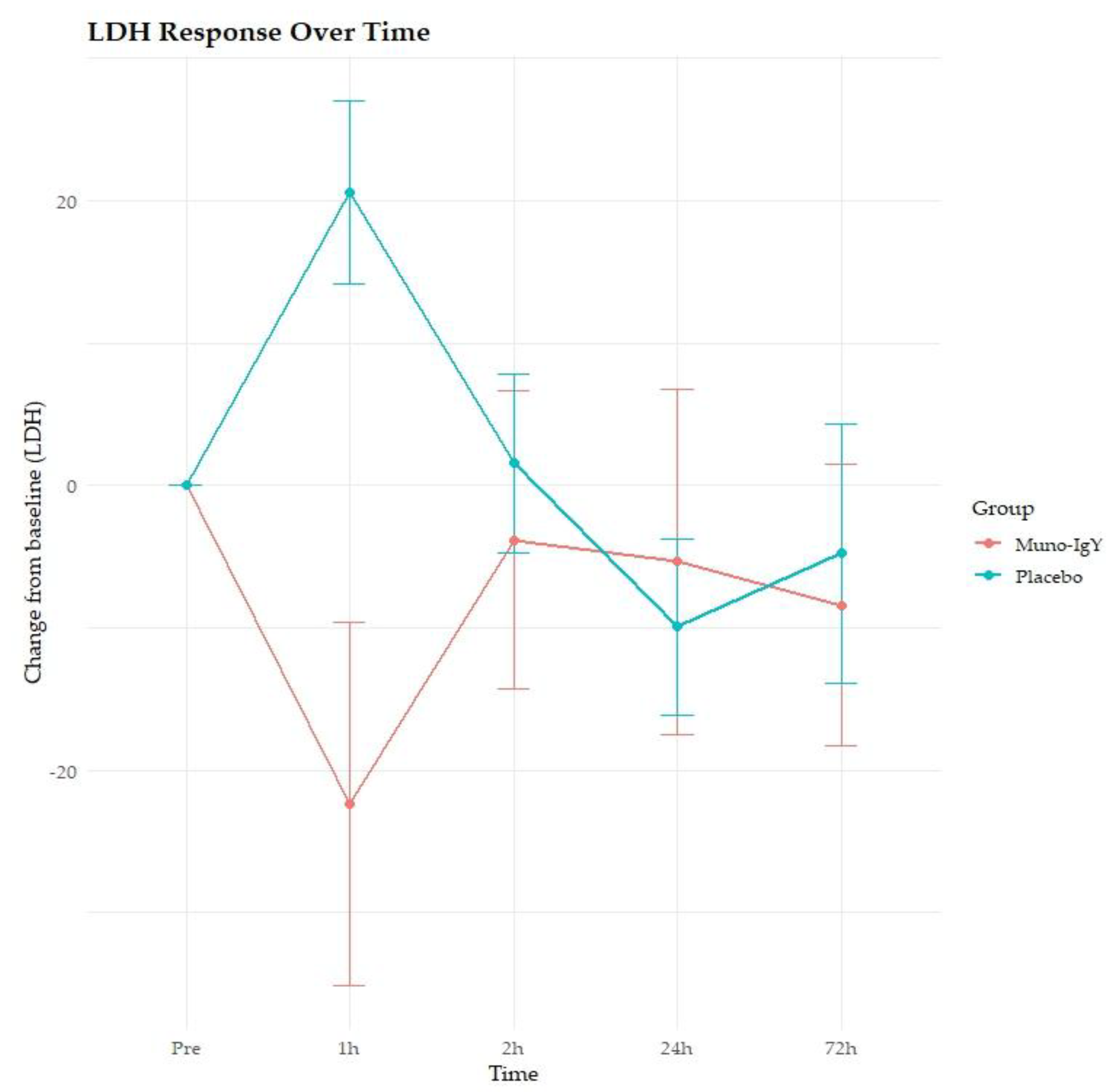
4.5.2 Serum Markers of Muscle Damage
4.5.3 Serum Markers of Inflammation
4.6. Effect of Muno-IgY supplementation on Gut Microbiome
4.6.1. Alpha Diversity
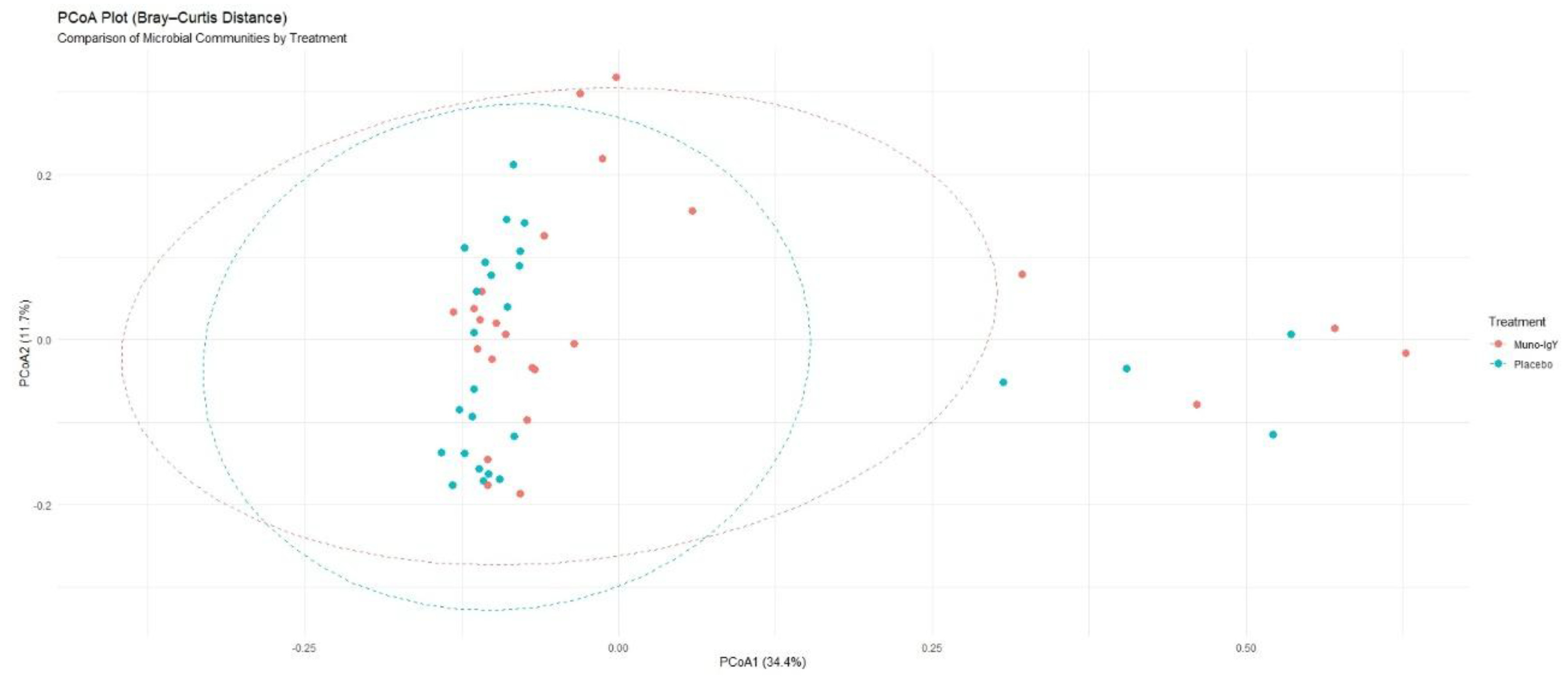
4.6.2 Beta Diversity
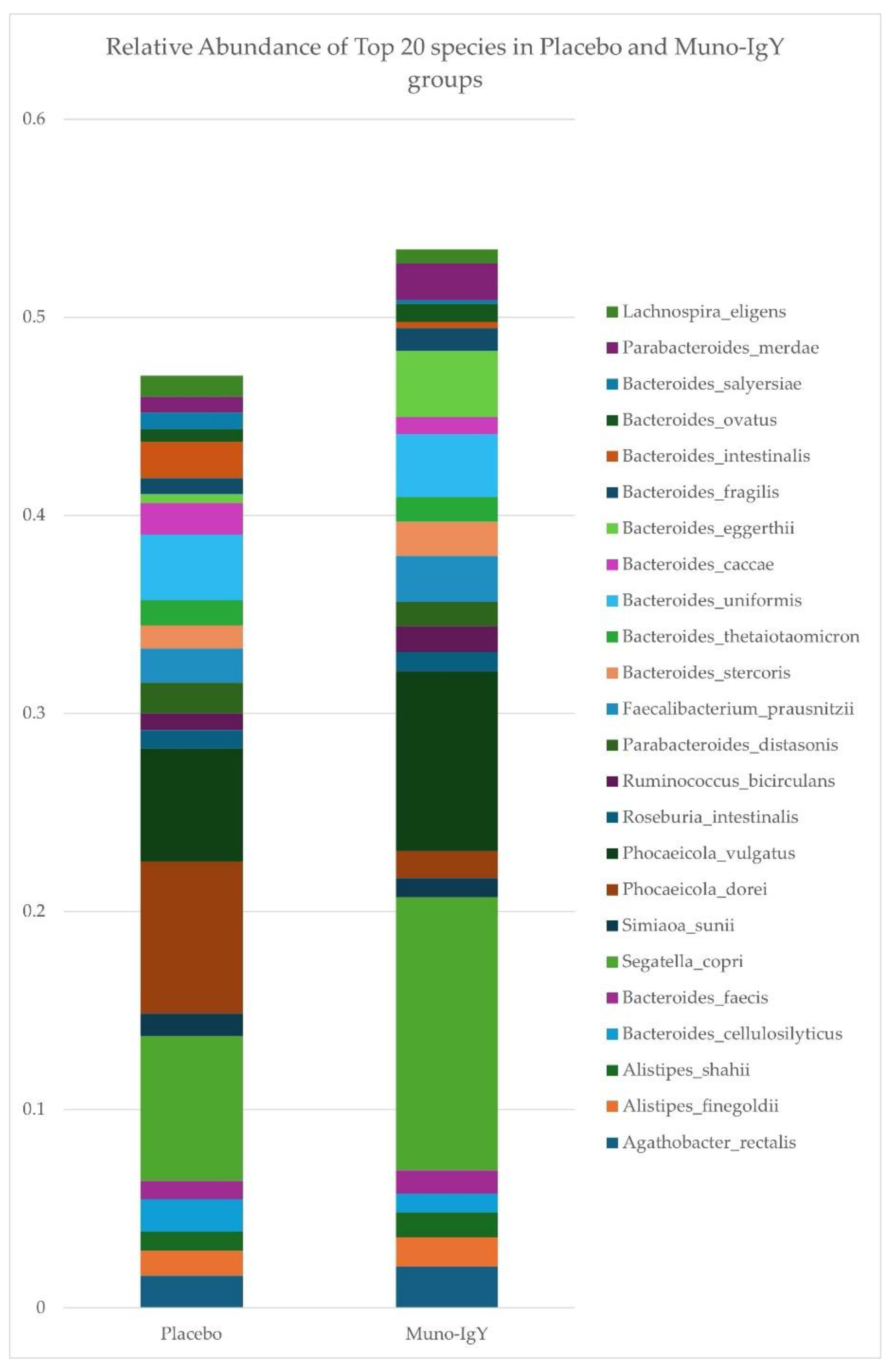
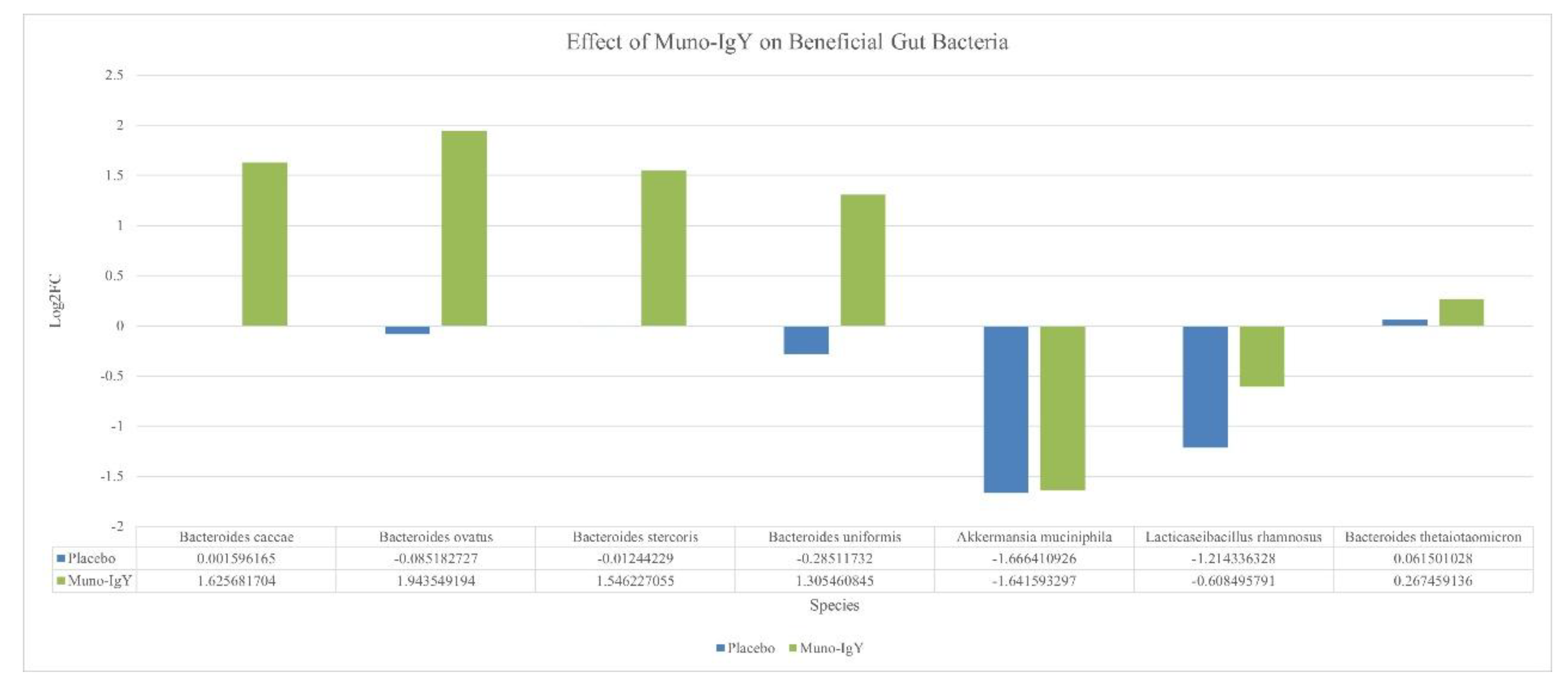
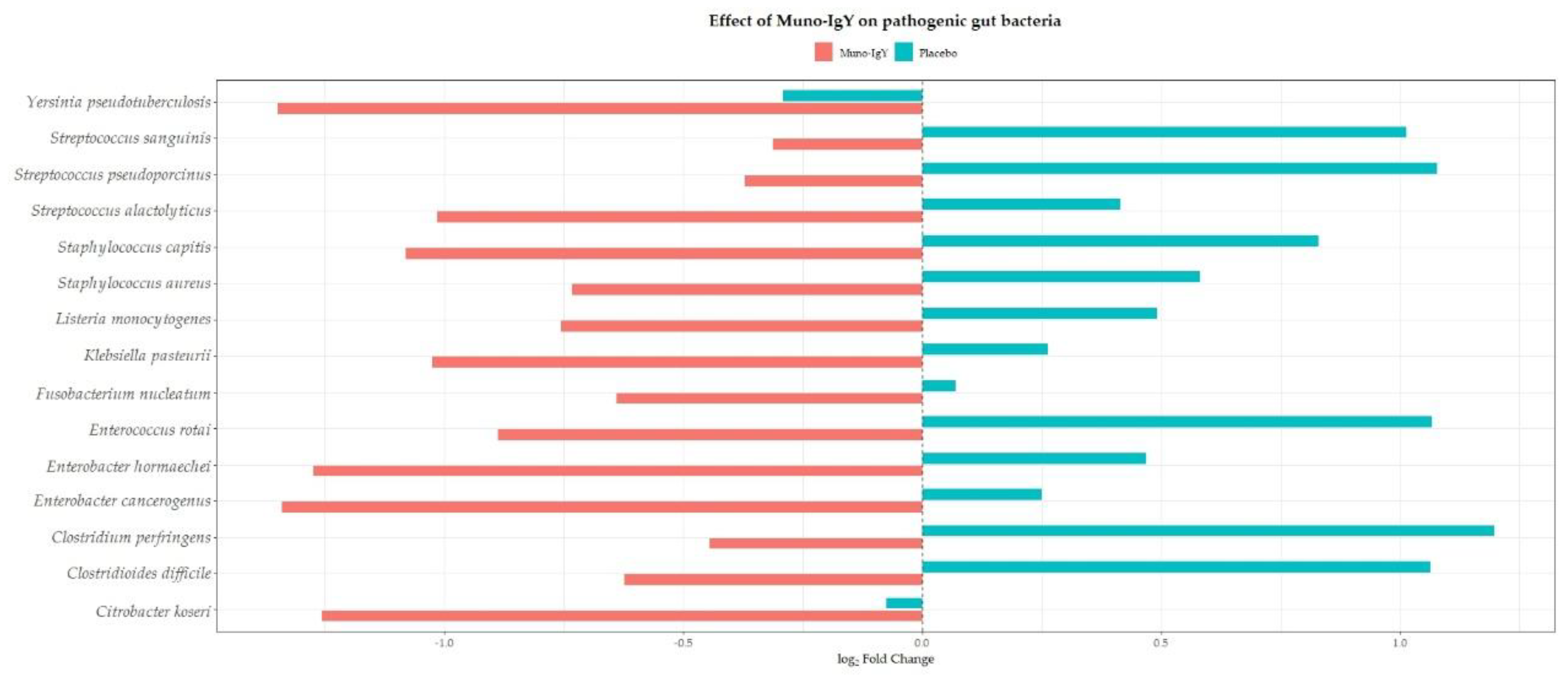
4.6.3 Species-Level Gut Microbiome Changes
5. Discussion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Jin, X.; Ren, J.; Li, R.; Gao, Y.; Zhang, H.; Li, J.; Zhang, J.; Wang, X.; Wang, G. Global burden of upper respiratory infections in 204 countries and territories, from 1990 to 2019. EClinicalMedicine 2021, 37. [Google Scholar] [CrossRef] [PubMed]
- Dicpinigaitis, P. V.; Eccles, R.; Blaiss, M. S.; Wingertzahn, M. A. Impact of cough and common cold on productivity, absenteeism, and daily life in the United States: ACHOO Survey. Curr.Med.Res.Opin 2015, 31(8), 1519–1525. [Google Scholar]
- Engebretsen, L.; Steffen, K.; Alonso, J. M.; Aubry, M.; Dvorak, J.; Junge, A.; Meeuwisse, W.; Mountjoy, M.; Renström, P.; Wilkinson, M. Sports injuries and illnesses during the Winter Olympic Games 2010. Br.J.Sports Med. 2010, 44(11), 772–780. [Google Scholar] [CrossRef]
- Cicchella, A.; Stefanelli, C.; Massaro, M. Upper respiratory tract infections in sport and the immune system response. A review. Biology 2021, 10(5), 362. [Google Scholar] [CrossRef]
- Colbey, C.; Cox, A. J.; Pyne, D. B.; Zhang, P.; Cripps, A. W.; West, N. P. Upper respiratory symptoms, gut health and mucosal immunity in athletes. Sports Medicine 2018, 48 Suppl 1, 65–77. [Google Scholar] [CrossRef]
- Kang, W.; Kudsk, K. A. Is there evidence that the gut contributes to mucosal immunity in humans? J.Parenter.Enteral Nutr. 2007, 31(3), 246–258. [Google Scholar] [CrossRef] [PubMed]
- Schuijt, T. J.; Lankelma, J. M.; Scicluna, B. P.; e Melo, F. d. S.; Roelofs, J. J.; de Boer, J. D.; Hoogendijk, A. J.; de Beer, R.; de Vos, A.; Belzer, C. The gut microbiota plays a protective role in the host defence against pneumococcal pneumonia. Gut 2016, 65(4), 575–583. [Google Scholar] [CrossRef] [PubMed]
- Cox, A. J.; Pyne, D. B.; Saunders, P. U.; Fricker, P. A. Oral administration of the probiotic Lactobacillus fermentum VRI-003 and mucosal immunity in endurance athletes. Br.J.Sports Med. 2010, 44(4), 222–226. [Google Scholar]
- Clancy, R. L.; Gleeson, M.; Cox, A.; Callister, R.; Dorrington, M.; D’este, C.; Pang, G.; Pyne, D.; Fricker, P.; Henriksson, A. Reversal in fatigued athletes of a defect in interferon γ secretion after administration of Lactobacillus acidophilus. Br.J.Sports Med. 2006, 40(4), 351–354. [Google Scholar]
- Heinz, S. A.; Henson, D. A.; Austin, M. D.; Jin, F.; Nieman, D. C. Quercetin supplementation and upper respiratory tract infection: A randomized community clinical trial. Pharmacological research 2010, 62(3), 237–242. [Google Scholar] [CrossRef]
- Abbas, A. T.; El-Kafrawy, S. A.; Sohrab, S. S.; Azhar, E. I. A. IgY antibodies for the immunoprophylaxis and therapy of respiratory infections. Human vaccines & immunotherapeutics 2019, 15(1), 264–275. [Google Scholar]
- Wang, L.; Li, X.; Jin, L.; You, J.; Zhou, Y.; Li, S.; Xu, Y. Characterization of chicken egg yolk immunoglobulins (IgYs) specific for the most prevalent capsular serotypes of mastitis-causing Staphylococcus aureus. Vet.Microbiol 2011, 149(3-4), 415–421. [Google Scholar] [CrossRef] [PubMed]
- Lee, E. N.; Sunwoo, H. H.; Menninen, K.; Sim, J. S. In vitro studies of chicken egg yolk antibody (IgY) against Salmonella enteritidis and Salmonella typhimurium. Poult.Sci 2002, 81(5), 632–641. [Google Scholar]
- Zhen, Y.; Jin, L.; Guo, J.; Li, X.; Lu, Y.; Chen, J.; Xu, Y. Characterization of specific egg yolk immunoglobulin (IgY) against mastitis-causing Escherichia coli. Vet.Microbiol 2008, 130(1-2), 126–133. [Google Scholar] [CrossRef]
- Wang, Z.; Li, J.; Li, J.; Li, Y.; Wang, L.; Wang, Q.; Fang, L.; Ding, X.; Huang, P.; Yin, J. Protective effect of chicken egg yolk immunoglobulins (IgY) against enterotoxigenic Escherichia coli K88 adhesion in weaned piglets. BMC veterinary research 2019, 15(1), 234. [Google Scholar]
- Yang, Y.; Wen, J.; Zhao, S.; Zhang, K.; Zhou, Y. Prophylaxis and therapy of pandemic H1N1 virus infection using egg yolk antibody. J.Virol.Methods. 2014, 206, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H. H.; Tumpey, T. M.; Park, H.; Byun, Y.; Tran, L. D.; Nguyen, V. D.; Kilgore, P. E.; Czerkinsky, C.; Katz, J. M.; Seong, B. L. Prophylactic and therapeutic efficacy of avian antibodies against influenza virus H5N1 and H1N1 in mice. PloS one 2010, 5(4), e10152. [Google Scholar] [CrossRef]
- Duncan, M. J.; Rosenkranz, R. R.; Vandelanotte, C.; Caperchione, C. M.; Rebar, A. L.; Maeder, A. J.; Tague, R.; Savage, T. N.; Van Itallie, A.; Mummery, W. K. What is the impact of obtaining medical clearance to participate in a randomised controlled trial examining a physical activity intervention on the socio-demographic and risk factor profiles of included participants? Trials 2016, 17(1), 580. [Google Scholar] [CrossRef]
- Hansen, A. W.; Grønbæk, M.; Helge, J. W.; Severin, M.; Curtis, T.; Tolstrup, J. S. Effect of a web-based intervention to promote physical activity and improve health among physically inactive adults: a population-based randomized controlled trial. Journal of medical Internet research 2012, 14(5), e2109. [Google Scholar] [CrossRef]
- Giuliano, F.; Jackson, G.; Montorsi, F.; Martin-Morales, A.; Raillard, P. Safety of sildenafil citrate: Review of 67 double-blind placebo-controlled trials and the postmarketing safety database. Int.J.Clin.Pract. 2010, 64(2), 240–255. [Google Scholar] [CrossRef]
- Moro, P. L.; Leung, J.; Marquez, P.; Kim, Y.; Wei, S.; Su, J. R.; Marin, M. Safety surveillance of varicella vaccines in the vaccine adverse event reporting system, United States, 2006–2020. J.Infect.Dis. 2022, 226 Supplement_4, S431–S440. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Sun, G.; Zhu, H.; Ye, B.; Zheng, Z.; He, X.; Pan, H. Effects of different peripheral fatigue protocol on lower limb biomechanical changes during landing and its impact on the risk of anterior cruciate ligament injury: a systematic review. Frontiers in Bioengineering and Biotechnology 2025, 13, 1587573. [Google Scholar] [CrossRef] [PubMed]
- Miraples, A.; McFadyen, B. J.; Khalaf, E. M.; Raizada, M. N. Pollen DNA Isolation Methods: Literature Review and Benefit of Adding Chloroform: Isoamyl Alcohol to Commercial DNA Kits. 2025. [Google Scholar] [CrossRef]
- Han, S.; Wen, Y.; Yang, F.; He, P. Chicken egg yolk antibody (IgY) protects mice against enterotoxigenic Escherichia coli infection through improving intestinal health and immune response. Frontiers in cellular and infection microbiology 2021, 11, 662710. [Google Scholar] [CrossRef]
- Nastasa, V.; Minea, B.; Pasca, A.; Bostanaru-Iliescu, A.; Stefan, A.; Gologan, D.; Capota, R.; Foia, L.; Mares, M. Long-Term Oral Administration of Hyperimmune Egg-Based IgY-Rich Formulations Induces Mucosal Immune Response and Systemic Increases of Cytokines Involved in Th2-and Th17-Type Immune Responses in C57BL/6 Mice. International Journal of Molecular Sciences 2024, 25(16), 8701. [Google Scholar] [CrossRef]
- Rehan, I. F.; Youssef, M.; Abdel-Rahman, M. A.; Fahmy, S. G.; Ahmed, E.; Ahmed, A. S.; Maky, M. A.; Diab, H. M.; Shanab, O.; Alkahtani, S. The impact of probiotics and egg yolk IgY on behavior and blood parameters in a broiler immune stress model. Frontiers in Veterinary Science 2020, 7, 145. [Google Scholar] [CrossRef]
- Munteanu, C.; Schwartz, B. The relationship between nutrition and the immune system. Frontiers in nutrition 2022, 9, 1082500. [Google Scholar] [CrossRef]
- Lim, P. S.; Chang, Y.; Wu, T. Serum lipopolysaccharide-binding protein is associated with chronic inflammation and metabolic syndrome in hemodialysis patients. Blood Purif. 2019, 47(1-3), 28–36. [Google Scholar] [CrossRef]
- Kong, Y.; Li, Z.; Tang, T.; Wu, H.; Liu, J.; Gu, L.; Zhao, T.; Huang, Q. The level of lipopolysaccharide-binding protein is elevated in adult patients with obstructive sleep apnea. BMC pulmonary medicine 2018, 18(1), 90. [Google Scholar] [CrossRef]
- Karalus, M.; Grodzicka, J. The Impact of Intense Physical Exercise on Humoral and Autoimmune Markers: Changes in IgG, IgA, and Anti-TPO Antibody Levels in Healthy Adults. Progress in Health Sciences 2025, 15(2), 1–6. [Google Scholar] [CrossRef]
- Abdukalykovna, I. Z.; Jamil, R. S.; Naji, H. A.; Jamal, Z. A.; Saleh, S.; Nurlanovna, O. A. Immune Response to Probiotics and Physical Activity: A Clinical Study. Trends in Immunotherapy 2025, 120–129. [Google Scholar] [CrossRef]
- Boukhris, O.; Trabelsi, K.; Abdessalem, R.; Hsouna, H.; Ammar, A.; Glenn, J. M.; Bott, N.; Irandoust, K.; Taheri, M.; Turki, M. Effects of the 5-m shuttle run test on markers of muscle damage, inflammation, and fatigue in healthy male athletes. International Journal of Environmental Research and Public Health 2020, 17(12), 4375. [Google Scholar] [CrossRef]
- Bessa, A. L.; Oliveira, V. N.; Agostini, G. G.; Oliveira, R. J.; Oliveira, A. C.; White, G. E.; Wells, G. D.; Teixeira, D. N.; Espindola, F. S. Exercise intensity and recovery: biomarkers of injury, inflammation, and oxidative stress. The Journal of Strength & Conditioning Research 2016, 30(2), 311–319. [Google Scholar]
- Cullen, J. M.; Shahzad, S.; Dhillon, J. A systematic review on the effects of exercise on gut microbial diversity, taxonomic composition, and microbial metabolites: Identifying research gaps and future directions. Frontiers in Physiology 2023, 14, 1292673. [Google Scholar] [CrossRef]
- Donaldson, G. P.; Ladinsky, M. S.; Yu, K. B.; Sanders, J. G.; Yoo, B. B.; Chou, W.; Conner, M. E.; Earl, A. M.; Knight, R.; Bjorkman, P. J. Gut microbiota utilize immunoglobulin A for mucosal colonization. Science 2018, 360(6390), 795–800. [Google Scholar] [CrossRef]
- Sey, E. A.; Warris, A. The gut-lung axis: the impact of the gut mycobiome on pulmonary diseases and infections. Oxford Open Immunology 2024, 5(1), iqae008. [Google Scholar] [CrossRef]
- Yin, Q.; da Silva, A. C.; Zorrilla, F.; Almeida, A. S.; Patil, K. R.; Almeida, A. Ecological dynamics of Enterobacteriaceae in the human gut microbiome across global populations. Nature Microbiology 2025, 10(2), 541–553. [Google Scholar] [CrossRef] [PubMed]
- Pabst, O.; Slack, E. IgA and the intestinal microbiota: the importance of being specific. Mucosal immunology 2020, 13(1), 12–21. [Google Scholar] [PubMed]
| Summary of Adverse Event Data Elements Collected During the Study | |
| Parameter | Description |
| Event description | Brief description of symptoms or diagnosis |
| Onset/End | Date and time of event start and resolution |
| Severity | Mild: no limitation; Moderate: some limitation; Severe: unable to perform usual activities |
| Action taken | Dose unchanged, reduced, interrupted, withdrawn, or other actions |
| Outcome | Fatal, recovered/resolved, recovered with sequelae, not resolved, unknown |
| Causality | Related, suspected, or not related to the study product |
| Pregnancy monitoring | Abnormal pregnancy outcomes were considered AEs/SAEs. Participants with pregnancy were withdrawn or followed to resolution. |
| Characteristic | Muno-IgY (n=14) | Placebo (n=14) | Total (N=28) |
| Age, years | |||
| Mean ± SD | 47.5 ± 7.1 | 48.1 ± 8.2 | 47.8 ± 7.6 |
| Range | 36-58 | 35-65 | 35-65 |
| Sex, n (%) | |||
| Female | 12 (85.7) | 12 (85.7) | 24 (85.7) |
| Male | 2 (14.3) | 2 (14.3) | 4 (14.3) |
| Race, n (%) | |||
| White | 10 (71.4) | 10 (71.4) | 20 (71.4) |
| Asian | 1 (7.1) | 1 (7.1) | 2 (7.1) |
| Black or African American | 1 (7.1) | 1 (7.1) | 2 (7.1) |
| Not reported | 2 (14.3) | 2 (14.3) | 4 (14.3) |
| Ethnicity, n (%) | |||
| Not Hispanic or Latino | 12 (85.7) | 12 (85.7) | 24 (85.7) |
| Hispanic or Latino | 2 (14.3) | 2 (14.3) | 4 (14.3) |
| BMI (kg/m²) | |||
| Mean ± SD | 25.4 ± 2.7 | 25.8 ± 2.3 | 25.6 ± 2.5 |
| Range | 20.2-29.5 | 21.3-29.8 | 20.2-29.8 |
| Vital signs | |||
| Systolic BP (mmHg), mean ± SD | 110.9 ± 10.4 | 113.3 ± 9.5 | 112.1 ± 9.9 |
| Diastolic BP (mmHg), mean ± SD | 74.9 ± 5.8 | 75.6 ± 6.6 | 75.3 ± 6.1 |
| Heart rate (bpm), mean ± SD | 65.4 ± 7.9 | 67.2 ± 7.6 | 66.3 ± 7.7 |
| Respiratory rate (breaths/min), mean ± SD | 14.4 ± 2.2 | 13.9 ± 1.9 | 14.1 ± 2.0 |
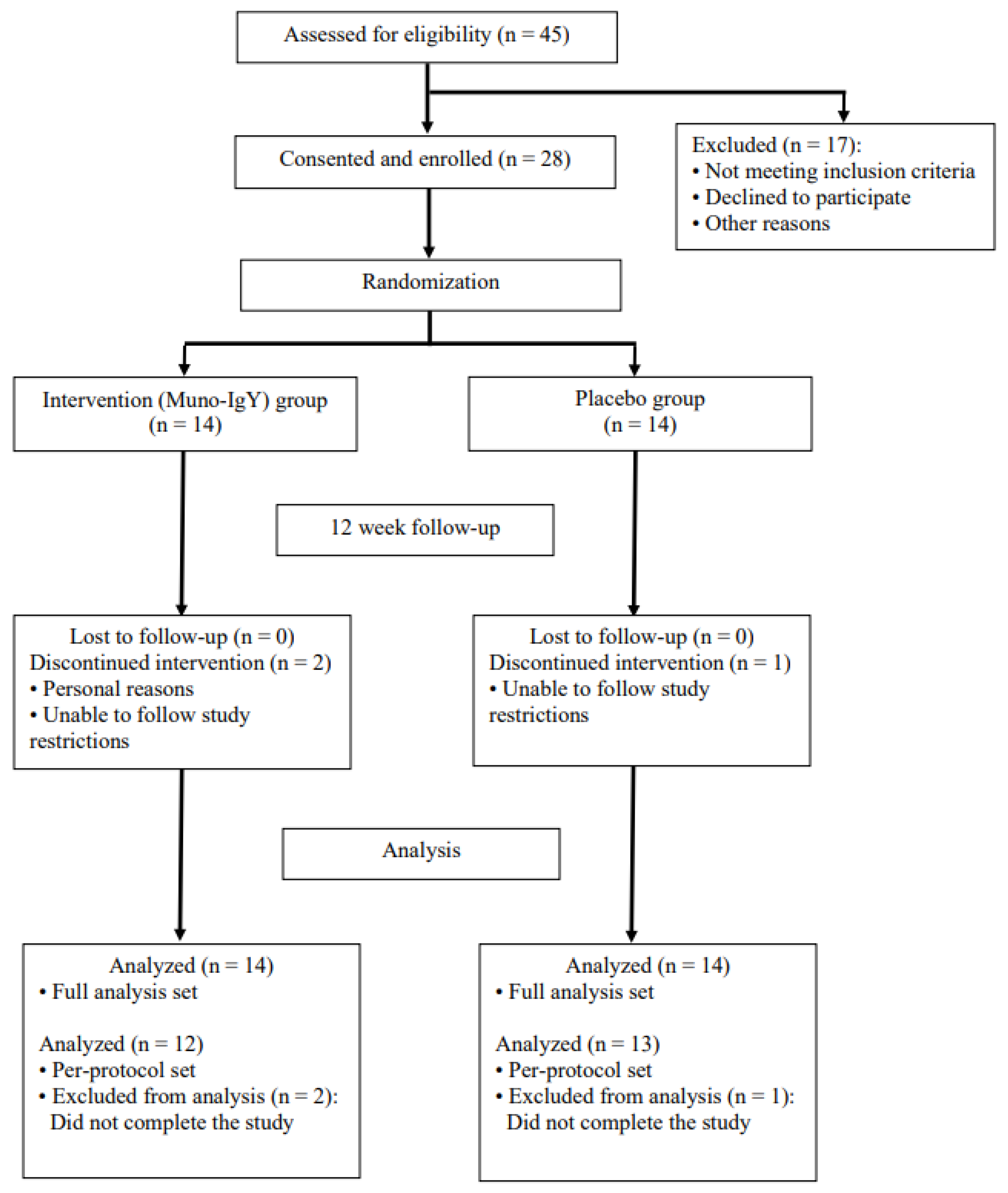
| Placebo | Muno-IgY | Total | |
| Screened | 45 | ||
| Met criteria but not randomized | 2 | ||
| Screening failure | 15 | ||
| Randomized | 14 (100%) | 14 (100%) | 28 (100%) |
| Included in the Safety population | 14 (100%) | 14 (100%) | 28 (100%) |
| Included in the FAS population | 14 (100%) | 14 (100%) | 28 (100%) |
| Included in the PPS population | 12 (85.7%) | 13 (92.9%) | 25 (89.3%) |
| Completed study | 12 (85.7%) | 13 (92.9%) | 25 (89.3%) |
| Excluded from PPS | 2 (14.3%) | 1 (7.1%) | 3 (10.7%) |
| Withdrawal by subject | 2 (14.3%) | 1 (7.1%) | 3 (10.7%) |
| Muno-IgY (N=14) | Placebo (N=14) |
Total (N=28) |
||
| Overall | 7 (50%) | 9 (64.3%) | 16 (57.1%) | |
| Relation | Related | 0 (0%) | 0 (0%) | 0 (0%) |
| Suspected | 1 (7.1%) | 0 (0%) | 1 (3.6%) | |
| Not related | 6 (42.9%) | 9 (64.3%) | 15 (53.6%) | |
| Severity | Severe | 0 (0%) | 0 (0%) | 0 (0%) |
| Moderate | 0 (0%) | 0 (0%) | 0 (0%) | |
| Mild | 7 (50%) | 9 (64.3%) | 16 (57.1%) | |
| Discontinuation | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Number of participants | Percentage of participants with URTI | |||||
| Muno-IgY | Placebo | Total | Muno-IgY | Placebo | Total | p-value |
| 2 | 5 | 7 | 14.3% | 35.7% | 25.0% | 0.38 |
| Diversity Index | Placebo | Muno-IgY | p-value |
| Shannon | 3.38 ± 0.57 | 3.43 ± 0.75 | 0.59 |
| Simpson | 0.86 ± 0.08 | 0.85 ± 0.12 | 0.53 |
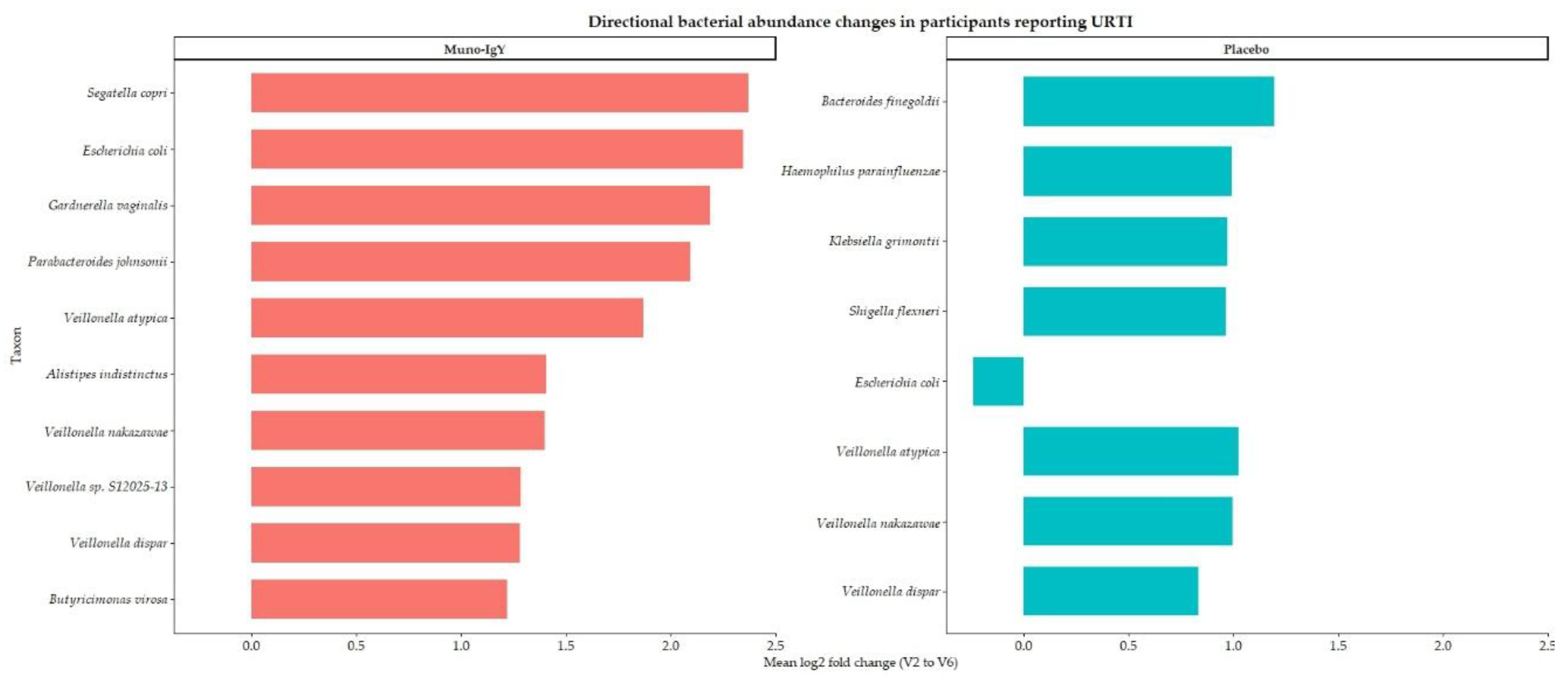
| Taxon | Treatment | Mean fold change | Median fold change | Mean log2FC | Median log2FC |
| Escherichia coli | Placebo | 14.36 | 0.33 | -0.24 | -1.62 |
| Klebsiella grimontii | Placebo | 12.87 | 0.92 | 0.97 | -0.12 |
| Shigella flexneri | Placebo | 12.27 | 0.96 | 0.96 | -0.06 |
| Bacteroides finegoldii | Placebo | 3.59 | 1.99 | 1.20 | 0.99 |
| Haemophilus parainfluenzae | Placebo | 4.01 | 2.35 | 0.99 | 1.23 |
| Veillonella atypica | Placebo | 3.99 | 1.60 | 1.02 | 0.68 |
| Veillonella dispar | Placebo | 3.89 | 1.19 | 0.83 | 0.25 |
| Veillonella nakazawae | Placebo | 6.62 | 1.30 | 1.00 | 0.38 |
| Segatella copri | Muno-IgY | 11.47 | 11.47 | 2.37 | 2.37 |
| Parabacteroides johnsonii | Muno-IgY | 6.57 | 6.57 | 2.09 | 2.09 |
| Escherichia coli | Muno-IgY | 5.92 | 5.92 | 2.35 | 2.35 |
| Gardnerella vaginalis | Muno-IgY | 8.49 | 8.49 | 2.19 | 2.19 |
| Veillonella atypica | Muno-IgY | 4.39 | 4.39 | 1.87 | 1.87 |
| Veillonella dispar | Muno-IgY | 3.28 | 3.28 | 1.28 | 1.28 |
| Veillonella nakazawae | Muno-IgY | 4.38 | 4.38 | 1.40 | 1.40 |
| Veillonella sp. S12025-13 | Muno-IgY | 4.46 | 4.46 | 1.28 | 1.28 |
| Butyricimonas virosa | Muno-IgY | 3.20 | 3.20 | 1.22 | 1.22 |
| Alistipes indistinctus | Muno-IgY | 4.87 | 4.87 | 1.41 | 1.41 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




