1. Introduction
B vitamins are a group of water-soluble micronutrients which encompassing thiamine (vitamin B1), riboflavin (vitamin B2), niacin (vitamin B3), pantothenic acid (vitamin B5), pyridoxine (vitamin B6), biotin (vitamin B8), folate (vitamin B9), and cobalamin (vitamin B12), indispensable for sustaining redox balance and mitigating oxidative stress, in addition to their canonical roles in energy metabolism, neurotransmitter synthesis, and DNA repair (Ding et al., 2022; Fitzpatrick & Chapman, 2020; Hellmann & Mooney, 2010; Kennedy, 2016; Lee et al., 2023; Yang et al., 2024). Many B vitamins serve as core cofactors or precursors for the body’s antioxidant system: for example, vitamin B2 supports the activity of glutathione reductase, an enzyme that regenerates the key antioxidant glutathione (GSH) (Asensi-Fabado & Munné-Bosch, 2010); vitamin B3 is converted to NAD(P)H, which fuels ROS-scavenging enzymes (Yang et al., 2024); and vitamin B6 (pyridoxine) regulates the metabolism of ROS-scavenging amino acids (Hellmann & Mooney, 2010). Deficiencies in these vitamins disrupt redox homeostasis, exacerbating oxidative stress and increasing risks of disorders such as pellagra, which due to vitamin B3 deficiency, characterized by oxidative damage to skin and neurons (Hrubša et al., 2022), and fetal neural tube defects, arising by vitamin B9 or vitamin B12 deficiency, linked to oxidative DNA damage (Degnan et al., 2014), as well as neurological impairments, reinforced by vitamin B1 deficiency, associated with mitochondrial ROS overproduction (Mkrtchyan et al., 2018). These issues are with global prevalence, particularly in populations relying on plant-based diets lacking adequate antioxidant-rich B vitamin sources (Fitzpatrick et al., 2024; Hrubša et al., 2022; Liu et al., 2022; Mkrtchyan et al., 2018).
While animal products have long been recognized as primary sources of B vitamins, plant-based foods are gaining prominence amid rising vegetarian and flexitarian trends, offering sustainable natural antioxidant reservoirs (Bonku et al., 2020; Kim et al., 2024). Plant-based B vitamin contents vary substantially by species, genotype and tissue type, with many oilseeds and leafy vegetables underexplored for their antioxidant-linked B vitamin potential (Garg et al., 2021; Lebiedzińska & Szefer, 2006). For instance, black soybean seeds show significant intraspecific variation in vitamin B6 content (Kim et al., 2024), and maize displays exhibited broad variability in B vitamins content such as, vitamin B1 (1.08-2.65 μg/g), vitamin B3 (0.2-2.14 μg/g), and vitamin B6 (1.4-10.65 μg/g) (Safiul Azam et al., 2022). Existing studies have predominantly focused on crops, such as rice and wheat, or individual vitamins, with limited attention to comparative analyses across multiple oilseed crops and leafy vegetables, or to the influence of tissue type (seeds, leaves, seedlings), and genotype factors on vitamin accumulation, limiting the development of natural antioxidant sources (Garg et al., 2021; Kim et al., 2014; Lebiedzińska & Szefer, 2006; Mangel et al., 2022; Watanabe et al., 2014).
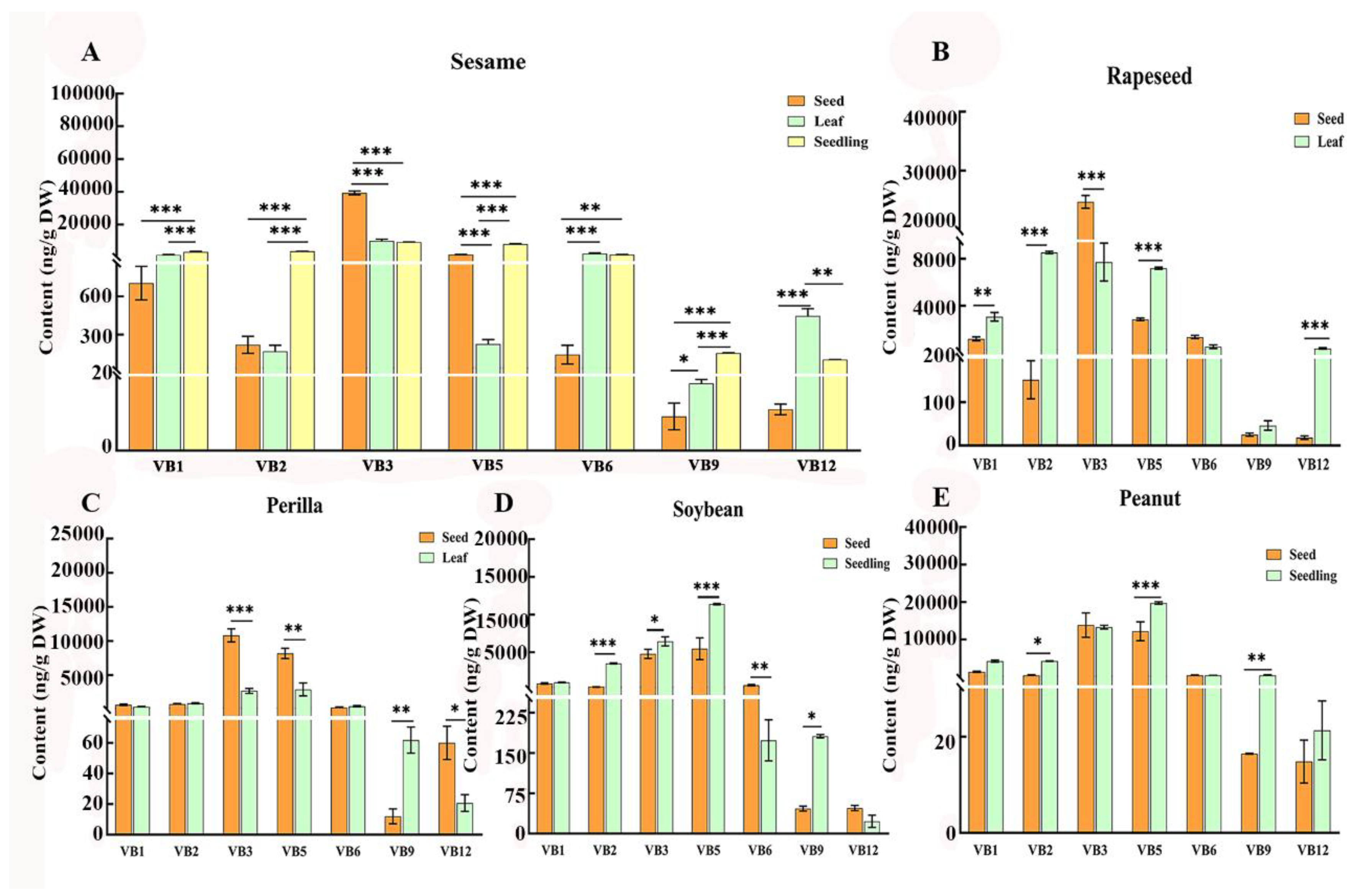
Notably, sesame, a crop valued for its oil content, has been overlooked for its micronutrient and antioxidant potential, despite preliminary evidence of its B vitamin richness (Abdiani et al. 2024; Kefale et al., 2023; Wang et al., 2018). This oversight is significant, as sesame is widely cultivated across diverse agro-climatic zones globally, making it a potential candidate for targeted biofortification for nutritional security. Given that oxidative stress underlies numerous chronic diseases and over two billion people face micronutrient deficiencies worldwide (Titcomb & Tanumihardjo, 2019), identifying plant-based sources with high, tissue-specific antioxidant B vitamin content is critical for advancing natural antioxidant research and dietary interventions. Hence, we systematically quantified seven B vitamins in seeds, leaves, and seedlings of five major oilseed crops including sesame, peanut, soybean, rapeseed, and perilla and two common leafy vegetables such as spinach and lettuce using LC-MS/MS. The aim of the study was to characterize the interspecific and tissue-specific variations in B vitamin profiles, and assess vitamin B3 natural variation in sesame germplasms, identify superior plant-based natural antioxidant sources. These findings will be useful to provide a scientific foundation for optimizing dietary antioxidant intake, developing functional foods and designing crop biofortification strategies targeting B vitamin enrichment.
2. Materials and Methods
2.1. Plant Materials, Growth Conditions and Sample Collection
Sesame, soybean, peanut, rapeseed, perilla, lettuce and spinach were used in the present study. The study materials were sourced from the National Medium-Term oilseed crops Gene Bank of China (Wuhan, China). Whereas, the leafy vegetables were purchased from local markets. Sesame, soybean, peanut, rapeseed and perilla were grown under natural conditions at the experimental station of the Oil Crops Research Institute of Chinese Academy of Agricultural Sciences (OCRI-CAAS, Wuhan, China) during 2024. Each material was planted with three cultivars. Field management of the trials were followed the standard agronomic practices for each crop. Fresh leaves of sesame, perilla and rapeseed were sampled from three-weeks-old plants. Seeds of sesame, soybean, peanut, rapeseed and perilla were harvested at physiological maturity stage. One-week-old seedlings of soybean, peanut, and sesame were sampled for further experiments. The study was conducted following with the local and national plant regulations.
2.2. Reagents and Instruments
Analytical standards for vitamins B1 (thiamine) (CAS 67-03-8, China), B2 (riboflavin) (CAS 83-88-5, China), B3 (niacin) (CAS 59-67-6, China), B5 (pantothenic acid) (CAS 137-08-6, China), B6 (pyridoxine) (CAS 58-56-0, China), B9 (folic acid) (CAS 59-30-3, China), and B12 (cobalamin) (CAS 68-19-9, China)were purchased from Shanghai Yuanye Biotechnology. HPLC-grade acetonitrile and formic acid were acquired from Merck, and analytical-grade hydrochloric acid and sodium hydroxide were supplied by Cologne Chemicals. Ultrapure water (18.2 MΩ·cm) was generated using a Milli-Q system. Instruments included a Shimadzu ATY224 electronic balance, KQ3200DA ultrasonic cleaner, H3-18KR high-speed centrifuge, Sartorius pH meter, and AB Sciex QTRAP 5500 LC-MS/MS system.
2.3. Sample Preparation and Extraction
For B vitamins extraction, 0.5 g of sample was accurately weighed into a centrifuge tube, mixed with 4.5 mL ultrapure water, vortexed for 30 seconds, and sonicated in an ice bath for 30 minutes. The extract pH was adjusted to 4.5 with 6 M hydrochloric acid, incubated for 2 minutes, and neutralized to pH 7.0 with 2 M sodium hydroxide. After centrifugation at 10,000 × g for 10 minutes, the supernatant was diluted to 10 mL, filtered through a 0.22-μm PTFE membrane, and stored at -20 °C prior to analysis.
2.4. LC-MS/MS Analysis
Chromatographic separation was performed on a Waters XSelect® HSS T3 column (4.6 mm × 250 mm, 3.5 μm) at 30 °C using a mobile phase of 0.2% formic acid in water and acetonitrile. The gradient elution program was 5% acetonitrile for 2 minutes, increasing to 20% over 8 minutes, 95% over 2 minutes, held for 1 minute, and returned to 5% over 1 minute, at a flow rate of 0.40 mL/min with a 2-μL injection volume.
Mass spectrometry was conducted in ESI+ mode with MRM detection. Source parameters included a curtain gas of 30 psi, +4500 V spray voltage, 500 °C temperature, and 50 psi for nebulizer and auxiliary gases. Quantitative analysis used the following parent/daughter ion pairs: VB1 (265.0→122.0), VB2 (377.0→243.2), VB3 (124.0→80.0), VB5 (220.0→202.0), VB6 (170.0→152.0), VB9 (442.0→295.2), VB12 (678.0→147.0), with optimized collision energies (REF). Calibration curves (1–1000 ng/mL) were prepared in 50% methanol-water, and quality control samples were analyzed every 10 injections to ensure method reproducibility.
2.5. Statistical Analysis
All the experiments were performed with three biological replicates, and the presented values are the mean ± SD. Data processing was performed using IBM SPSS Statistics 27. Statistical significance was determined using analysis of variance (ANOVA) with Duncan’s multiple range test (p < 0.05) and Student’s t-test (*, P < 0.05 is considered significantly different, and **, P < 0.01 is considered highly significantly different). Heat maps were generated by GraphPad Prism 8 (standardization via mean centering and standard deviation).
3. Results and Discussion
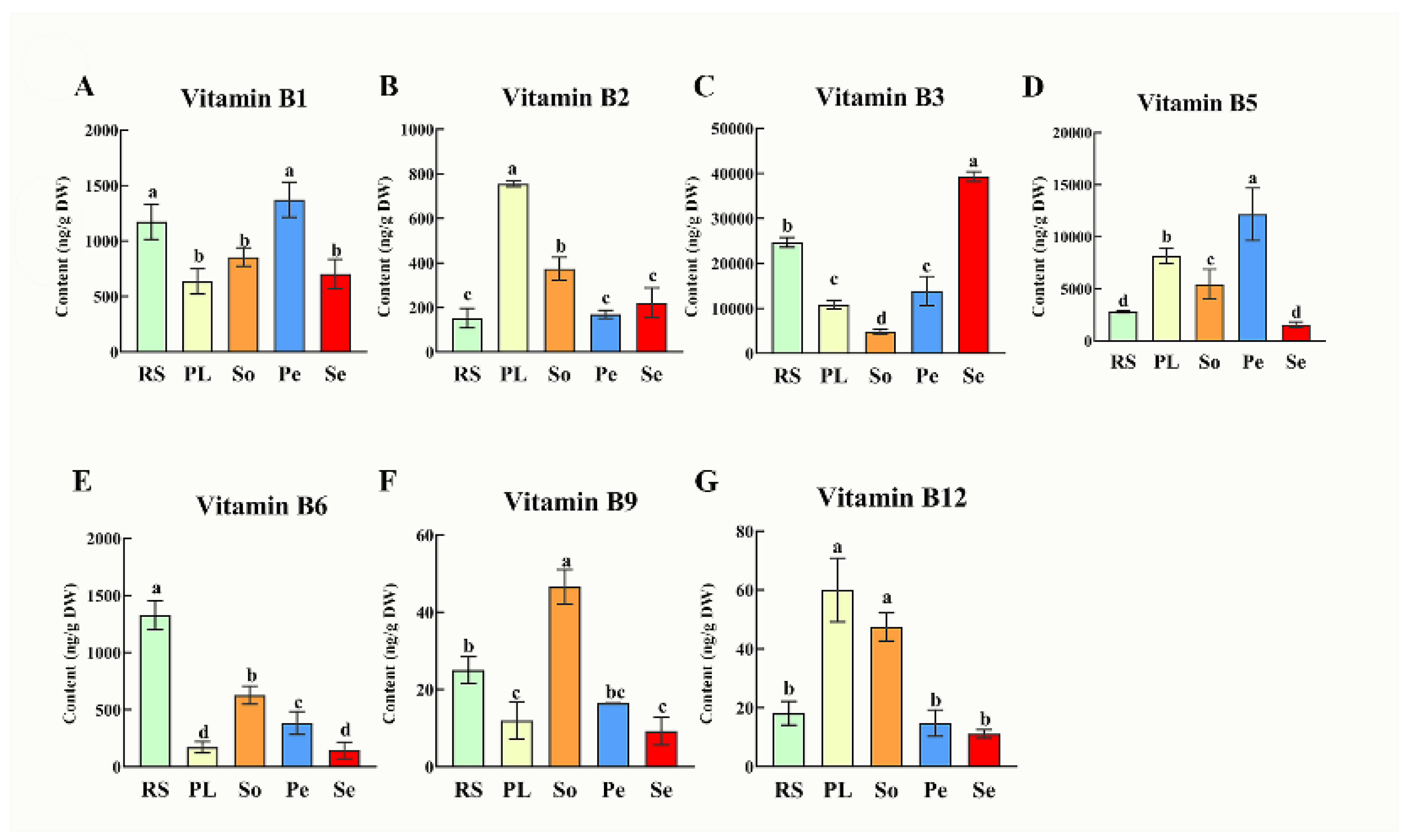
3.1. Sesame Seed Showed Notable High Vitamin B3 in Oil Crops
B vitamins unable synthesize and stored by humans and these micronutrients are essential on their daily diet (Liu et al., 2022). Plants or plant-based foods are good sources of B vitamins, with natural variations in their contents contributing to diverse nutritional applications (Lebiedzińska & Szefer, 2006). In the present study we detected the seven types of B-vitamin complex (vitamin B
1, B
2, B
3 B
5, B
6, B
9, and B
12) in seeds of five oilseed crops such as, sesame, soybean, peanut, rapeseed and perilla. Quantitative analysis revealed significant interspecific differences in B-vitamin profiles across the five oilseed crops seeds (
Figure 1). Sesame seeds exhibited the highest content of vitamin B
3 (39.3 μg/g), with 1.6-fold levels higher than rapeseed (24.7 μg/g), 2.8-fold higher than peanut (13.8 μg/g), 3.6-fold higher than perilla (10.8 μg/g) and 8.2-fold higher than soybean (4.78 μg/g). This exceptional vitamin B
3 content not only positions sesame seeds as a superior natural functional ingredient for niacin supplementation but also highlights their potential as a natural antioxidant resource. vitamin B
3 plays a critical role in maintaining cellular redox homeostasis by participating in the synthesis of nicotinamide adenine dinucleotide (NAD+) and nicotinamide adenine dinucleotide phosphate (NADP+), key cofactors involved in antioxidant defense and ROS scavenging (Lindschinger et al., 2019).The high content of B
3 available in sesame seed makes the crop ideal for incorporation into processed foods like bread, snacks, and plant-based meat alternatives to simultaneously enhance nutritional value and antioxidant capacity. Additionally, the higher sesame seeds’ B
3 concentration in the present study exceeds that of major cereal crops: for instance, it is 22.9-245-fold higher than the bioavailable B
3 in wheat (0.16-1.74 μg/g dw) (Shewry et al., 2011) and 18.4-200-fold higher than the B
3 range in maize (0.196-2.1375 μg/g) (Safiul Azam et al., 2022), highlighting sesame’s unique potential to enhance niacin intake, with particularly valuable for preventing pellagra, a B
3 deficiency disorder, in regions reliant on cereal-dominated diets (Hrubša et al., 2022).
Among other oilseed seeds, peanut seeds revealed the highest vitamin B1 and vitamin B5 contents, with a value of 1.37 μg/g and 12.2 μg/g, respectively. These findings could benefit regions with rice-dominant diets, where thiamine (vitamin B1) deficiency contributes to beriberi disease, an effect potentially reinforced by vitamin B1’s role in protecting mitochondrial function against oxidative damage (Nguyen et al., 2024). Moreover, rapeseed showed the highest vitamin B6, with a content of 1.33 μg/g. The vitamin B9 content in soybean (0.046 μg/g) was significantly higher compared to other crops. Perilla was recorded higher in vitamin B2 (0.75 μg/g) and vitamin B12 (0.06 μg/g) contents than other crops. Among the seven studied B-vitamin complex, vitamin B3 was highly available in content in the five oilseed crops ranged between 4.78 to 39.3 μg/g, emphasizing oilseeds as a collective valuable resource for both nutritional supplementation and antioxidant defense. These variations indicated that different oilseed seeds can serve as complementary sources to meet specific B-vitamin and antioxidant needs, diversifying seed-based nutritional strategies.
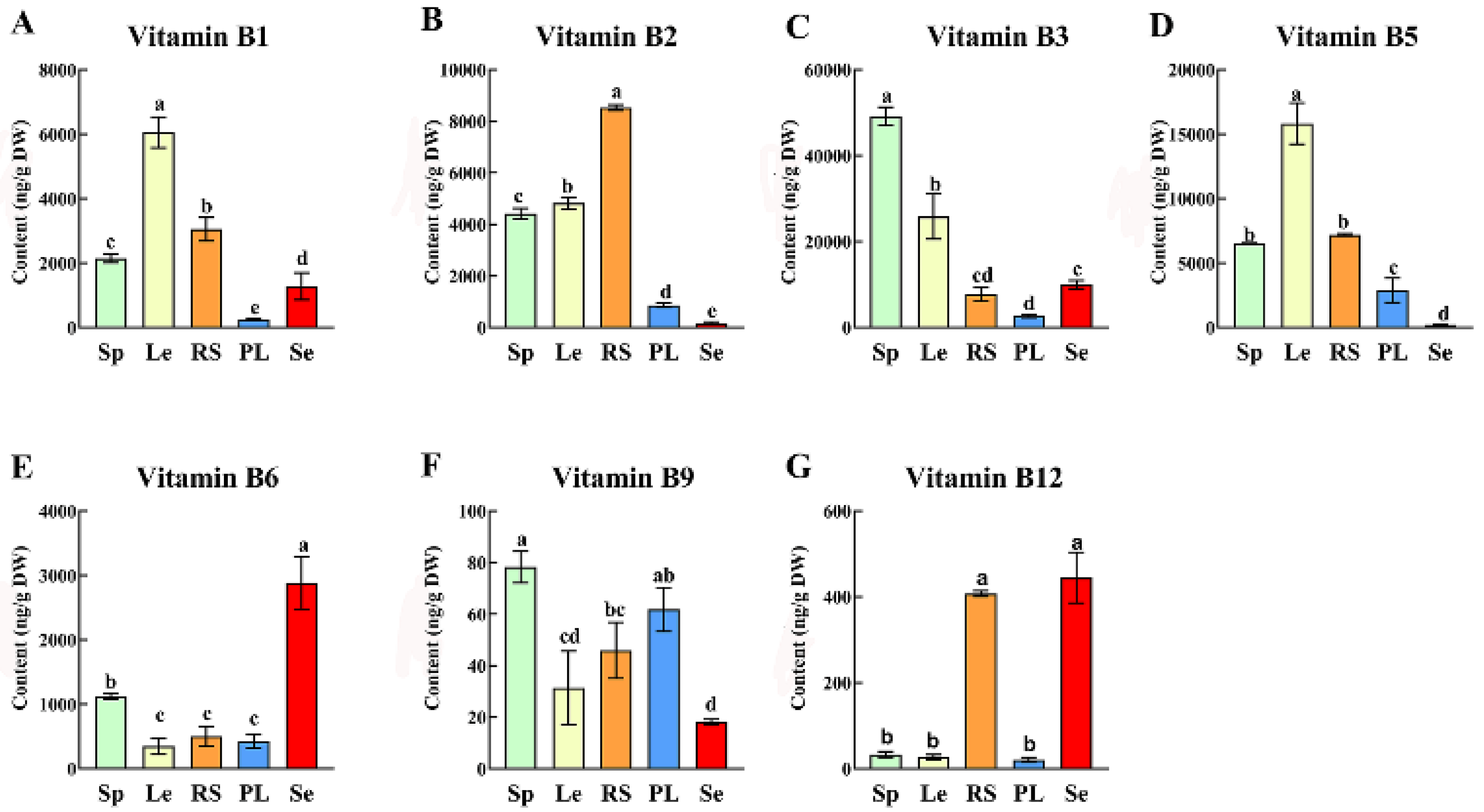
3.2. B Vitamin Profiles in Leaves: Sesame as a Superior Source of Vitamin B6 and Vitamin B12
Based on quality and high preference for cooking oil production, we selected three edible oilseed crops including sesame, rapeseed and perilla leaves, and two regularly consumed leafy vegetables (spinach and lettuce) to determine the B-vitamin variations in the study (
Figure 2). Of the leave samples, lettuce showed the highest vitamin B
1 (6.05 μg/g) and vitamin B
5 (15.83 μg/g). The highest vitamins B
2, B
3, and B
6, values of 8.53 μg/g, 49.17 μg/g and 2.88 μg/g were recorded in the leaves of rapeseed, spinach, and sesame, respectively. Similarly, higher values in vitamin B
9 (0.08 μg/g) and B
12 (0.44 μg/g) were recorded for spinach and sesame leaves, respectively.
Typically, the vitamin B6 content in sesame leaves was 2.57, 8.31, 5.76 and 6.8 times higher than in spinach, lettuce, rapeseed, and perilla, respectively. Vitamin B6 (pyridoxine) is a critical antioxidant cofactor that modulates the glutathione (GSH) system, an essential component of cellular antioxidant defense (Bdeir et al., 2024). This function is particularly important as major staples like rice, wheat, and cassava often fail to meet recommended dietary allowances for B6 when consumed as primary staples, leading to compromised antioxidant capacity and increased oxidative stress (Hellmann & Mooney, 2010). In contrast to the present finding, low B6 content (1.27-2.97 μg/g dw) reported by Shewry et al. (2011) in wheat grains, making sesame leaf a critical complementary source for B6, supporting neurotransmitter biosynthesis and ROS scavenging (Hellmann & Mooney, 2010; Watanabe et al., 2014).
Moreover, sesame (0.44 μg/g) and rapeseed (0.4 μg/g) leaves exhibited the highest vitamin B12, which were both accounts about 13, 16, and 20 times higher that of spinach, lettuce and perilla, respectively. Vitamin B12 is usually scarce in plant-based diets, but plays a pivotal role in mitigating oxidative stress by reducing homocysteine accumulation (Firat et al., 2022; Pahlavani et al., 2023). Notably, sesame leaves (0.44 μg/g) and rapeseed leaves (0.4 μg/g) outperform commonly cited plant sources like shiitake mushrooms, which was regarded as an important source of vitamin B12, whose B12 content varies widely from 0.013 to 0.127 μg/g (Watanabe et al., 2014). This stability and concentration in sesame leaves address a key gap in vegetarian diets, providing a reliable source of B12 that not only prevents megaloblastic anemia but also enhances antioxidant status by reducing oxidative stress and DNA damage (Firat et al., 2022; Belal et al., 2023).
In contrast, lettuce leaves contained twice the B1 and B5 levels of sesame, rapeseed, perilla, and spinach. Collectively, these findings highlight the tissue-specific distribution of B vitamins and their antioxidant functions, with sesame leaves standing out as a dual-source of B6 and B12 for both nutritional supplementation and oxidative stress mitigation, particularly valuable for vegetarians and populations at risk of B-vitamin deficiency-related oxidative damage.
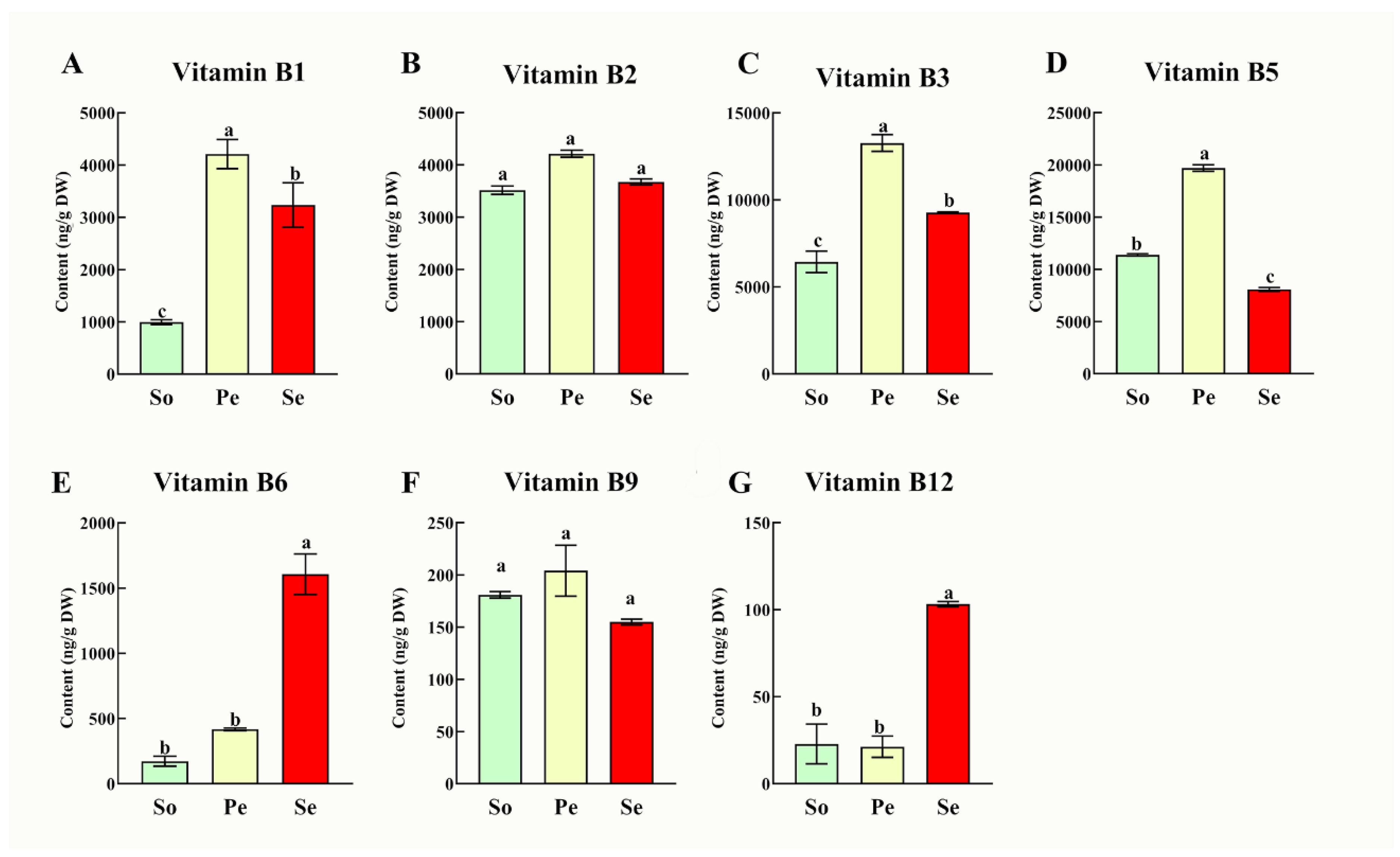
3.3. B-Vitamin Accumulation in Seedlings Indicating Complementary Nutritional Reservoirs
We also investigated the B vitamin variations in the seedlings of three edible oilseed crops including, sesame, peanut and soybean (
Figure 3). Results revealed that there were no significant differences in contents of vitamin B
2 and B
9, while, remarkable variation occurred among the other five B vitamins (B
1, B
3, B
5, B
6, B
12). Peanut seedlings evidenced rich in vitamin B
1 (4.21 μg/g), B
3 (13.27 μg/g), and B
5 (19.7 μg/g), while sesame followed by the contents of 3.23 μg/g and 9.28 μg/g and 8.08 μg/g, respectively. Sesame seedlings had stood out with the highest vitamin B
6 (1.6 μg/g) and B
12 (0.1 μg/g), reinforcing sesame’s multi-tissue potential as a source of antioxidant-related B vitamins.
Seedlings represent underutilized nutritional reservoirs, with their B-vitamin profiles reflecting adaptations to early growth under oxidative stress. Vitamin B6 in sesame seedlings contributes to ROS scavenging and GSH maintenance (Bdeir et al., 2024), while B12 supports DNA integrity by reducing oxidative damage (Firat et al., 2022). These properties make seedlings ideal for integration into urban farming and ready-to-eat products (e.g., salads, spreads), offering convenient sources of both nutrients and antioxidants for modern lifestyles. The complementary B-vitamin profiles of peanut, soybean, and sesame seedlings further enable diversified dietary strategies, where each crop’s seedlings can be leveraged to target specific antioxidant and nutritional needs.
3.4. Tissue-Specific Accumulation of B-Vitamin in Different Oilseed Crops
The vitamin B contents were compared between the seed and non-seed tissues (leaf, and seedling) in the five oilseed crops. Results revealed significant differences among the studied tissues of the oilseed crops in the present study, highlighting the need for comprehensive utilization of crop tissues for optimal B-vitamin and antioxidant intake. Overall, sesame seed had the highest vitamin B
3, which were 3.9 and 4.2 times higher than those in leaves and seedlings, respectively. Both sesame leaf and seedling were significantly higher in vitamin B
6 and vitamin B
12, which was about 15 to 33 times of that in seeds (
Figure 4A). This pattern might associate with the functional implications of these vitamins, with B
3 (niacin) in seeds likely mediating oxidative protection during storage and B
6 and B
12 in photosynthetic tissues (leaves) and actively growing seedlings supporting ROS scavenging during photosynthesis and early development stages (Mangel et al., 2019). Rapeseed leaves had 2.5 times higher vitamin B
1 and vitamin B
5, 56 times higher vitamin B
2, and 19 times higher vitamin B
12 than seeds, while seeds had 3 times higher vitamin B
3 than leaves (
Figure 4B). Perilla seeds had 4 times higher vitamin B
3 and 2.5 times higher vitamin B
5 than leaves, but vitamin B
9 content in seeds was 5 times lower than that in leaves (
Figure 4C). Soybean seedlings had 9 times higher vitamin B
2, 1.3 times higher vitamin B
3, and 3 times higher vitamin B
9 than seeds (
Figure 4D). In peanut, seedlings were significantly higher in vitamin B
5 and vitamin B
9 than seeds (
Figure 4E). The results suggested the seed of sesame and rapeseed are better resource for vitamin B
3 than its leaf and seedling; the leaf of sesame and rapeseed can supply more vitamin B
12 than its seed; Perilla seeds are good resource for vitamin B
3 and vitamin B
5 than its leaf; and rapeseed leaf and soybean seedling are good resource for vitamin B
2 than its seeds. The results revealed distinct tissue-specific patterns. These patterns challenge the traditional focus on seeds, emphasizing that non-seed tissues (leaves, seedlings) can be equally valuable for dietary B-vitamin intake and antioxidant defense. Collectively, these findings support the comprehensive utilization of oilseed crops, where each tissue can be targeted for specific B-vitamin and antioxidant applications.
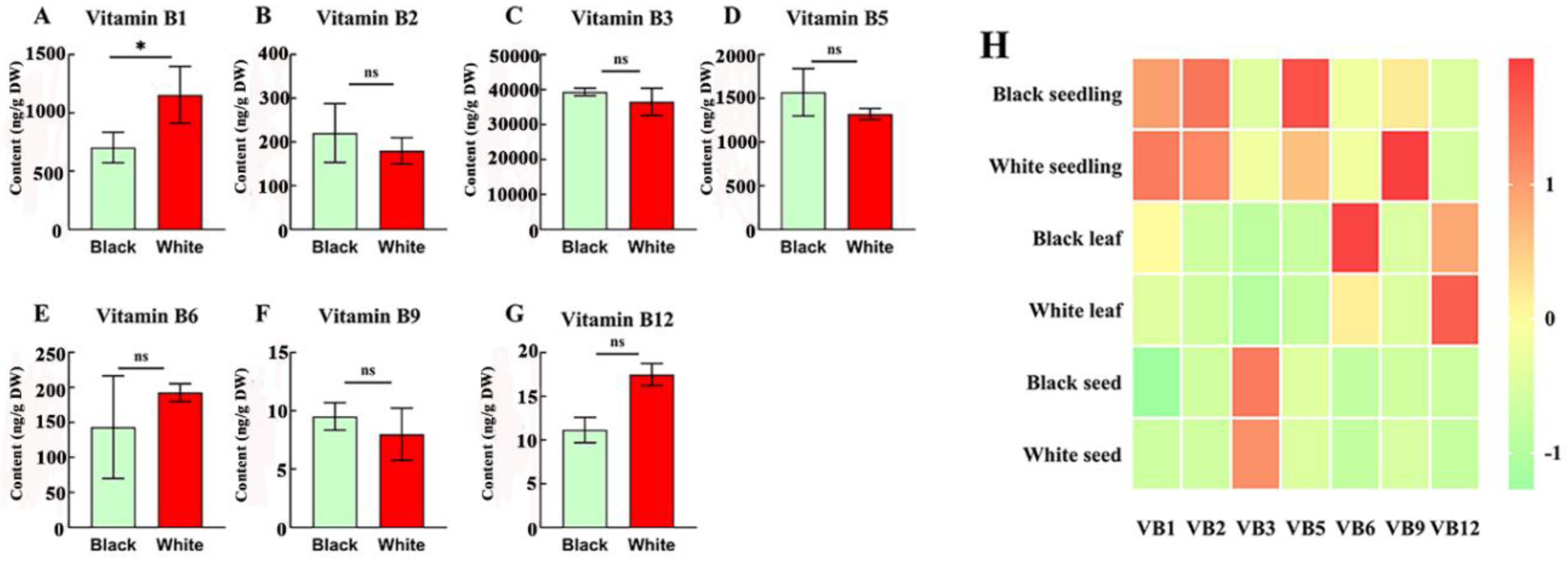
3.5. B-Vitamin Profiles in Black and White Sesame Seeds Across Tissues
Black sesame seeds usually considered as good value for antioxidative ability in the treatment of diverse diseases, while white seeded for oil extraction (Radchuk and Borisjuk, 2014). Comparative analysis of black and white sesame seeds revealed tissue-specific B-vitamin distributions, with seed coat color influencing only vitamin B
1 content in seeds. White sesame seeds showed significant difference and had 1.5 higher vitamin B
1 content of 1.09 μg/g than black seeded sesame (0.7 μg/g) (
Figure 5A), while there was no significant differences (ns) observed in the remain B vitamins in the current study (
Figure 5B-G). The heatmap results showed both white and black sesame seedlings exhibited the highest vitamins B
1 (3.23 and 2.36 μg/g, respectively)
, B
2, (3.67 and 3.65 μg/g, respectively), and B
5 (8.1 and 10.5 μg/g, respectively), whereas seeds had peak vitamin B
3 (35.5 and 39.3 μg/g, respectively). The leaves of both seed colors stood out with the highest vitamin B
12 (0.44 and 0.48 μg/g respectively), but black sesame leaves showed with 2.76-22-fold higher vitamin B
6 than other two tissues (
Figure 5H). These finding indicates that black sesame seeds may offer advantage for vitamin B
5 in seedling and vitamin B
6 in leaf, supporting targeted utilization based on tissue and seed coat color.
3.6. Natural Variations in Vitamin B3 Content Among Sesame Germplasm
Vitamin B
3 (niacin) is a key antioxidant nutrient that enhances cellular redox defense by promoting NAD(P)H-dependent ROS scavenging and inhibiting lipid peroxidation (Hrubša et al., 2022). To identify sesame germplasm with high vitamin B
3 content and thereby expand vitamin B
3 supply sources, we analyzed vitamin B
3 variability across different sesame germplasms (
Table 1). The results showed that vitamin B
3 content in sesame germplasm exhibited substantial variability across the tested germplasms. Among 92 total samples, the content of vitamin B
3 ranged from 23.57 to 82.25 μg/g, with a mean of 39.28 μg/g and a CV of 15.12%. White sesame showed a range of 23.57-55.87 μg/g, with a mean of 38.79 μg/g and a CV of 16.44%, while black sesame had a narrower distribution, ranged from 25.37-45.14 μg/g, with a mean of 38.7 μg/g and a CV of 13.93%. Notably, yellow sesame ranged from 26.24-82.25 μg/g, exhibited the highest mean (40.66 μg/g) and greatest variability (CV = 32.83%, matching the overall maximum), whereas brown sesame displayed intermediate variation (24.08–48.64 μg/g; mean = 37.90 μg/g; CV = 18.94%). These results underscore the broad natural variation in vitamin B
3 content across sesame germplasm, informing the screening of high- vitamin B
3 accessions to boost vitamin B
3 supply. This genetic diversity enables targeted breeding for high-B
3 germplasms. The identification of germplasms with B
3 contents enables the development of tailored fortification from daily staples to high-dose products and enhancing sesame’s role in addressing global niacin inadequacies and oxidative stress-related health issues.
3.7. Implications for Dietary Diversification, Biofortification and Antioxidant Defense
Globally, achieving a balanced and diverse diet remains a challenge for several people, attributable to micronutrient deficiencies, particularly in low-income settings (Bechoff et al., 2023). A diversified diet implies the adequate intake of essential nutrients and antioxidants, and plant-based sources are increasingly recognized for their dual role in nutrition and oxidative stress mitigation (Ray & Dutta, 2022). Understanding the plant-based sources and profiles are essential for dietary diversification and fortification to contribute nutrient deficiency globally. The distinct species- and tissue-specific B-vitamin patterns highlight the potential of oilseeds, particularly sesame, as multifunctional resources. Sesame’s seeds (high B3), leaves (high B6, B12), and seedlings (high B6, B12) allow comprehensive dietary integration, from seed-based products to leafy greens and sprouts. These tissues collectively provide a spectrum of B vitamins that support antioxidant pathways: B3 for NAD(P)H synthesis, B6 for GSH maintenance, and B12 for homocysteine reduction (Lindschinger et al., 2019; Firat et al., 2022). Furthermore, sesame’s adaptability to diverse agro-climatic zones and compatibility with local diets in regions like South Asia and East Africa further enhance its utility as a sustainable solution for global nutritional and antioxidant needs.
Leveraging the observed variations, future efforts can focus on breeding high-B-vitamin germplasms and promoting the inclusion of non-seed tissues in diets. These strategies hold promise for addressing global micronutrient deficiencies through sustainable. Additionally, integrating sesame into biofortification programs can enhance the antioxidant capacity of staple diets, reducing the risk of chronic diseases linked to oxidative stress (e.g., cardiovascular disease, neurological disorders) (Ding et al., 2022; Kennedy, 2016).
4. Conclusion
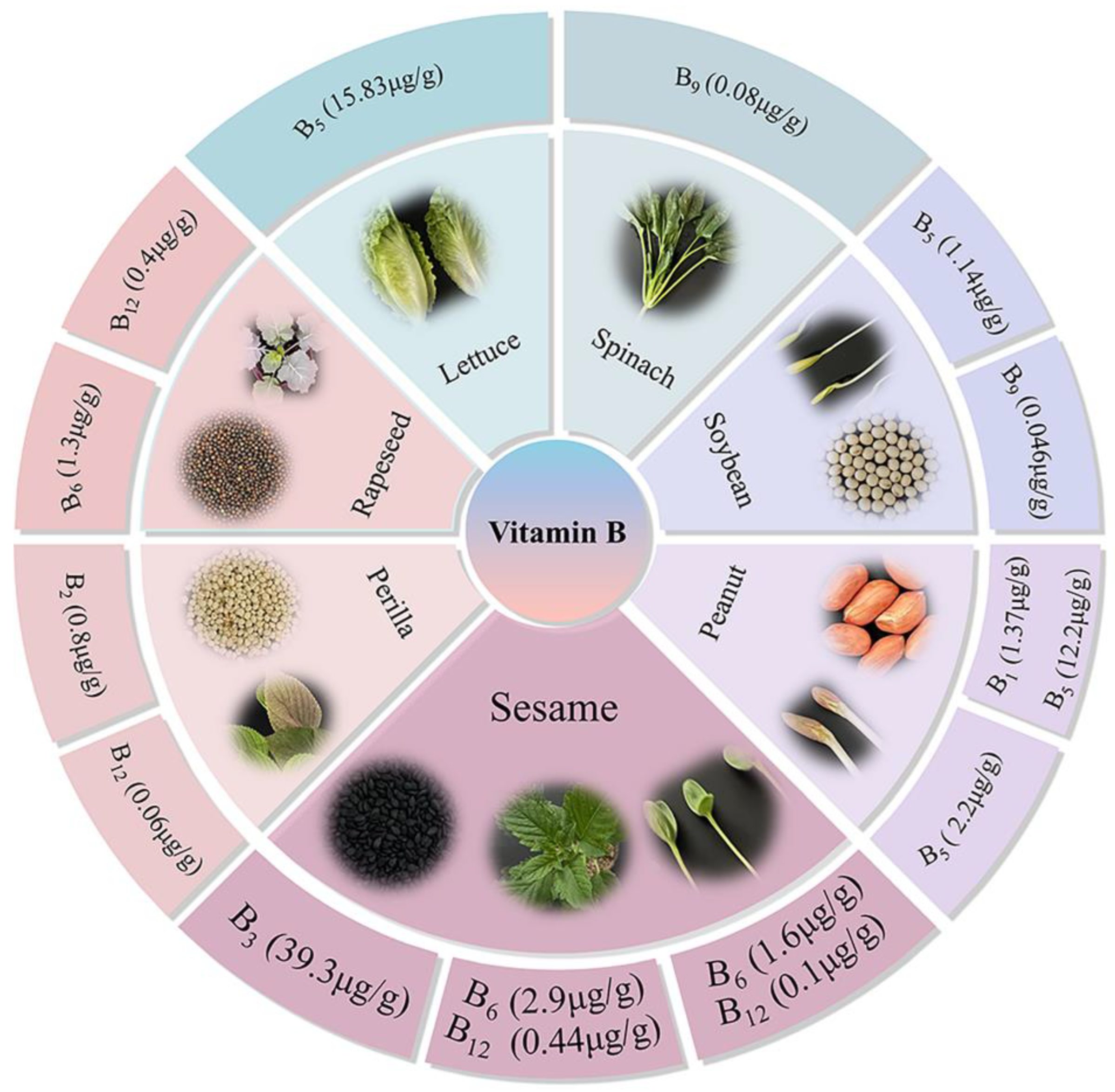
This study aims to characterize the interspecific and tissue-specific variations in B vitamin profiles, and assess vitamin B
3 variations in sesame germplasms. Results revealed that significant interspecific differences in B-vitamin variations across the five oilseed crops seeds and two leafy greens (
Figure 6). Notably, Sesame seeds exhibited exceptional vitamin B
3 contents compared to other oilseeds crops, and this makes the crop regarded as a natural functional ingredient for niacin supplementation. Furthermore, sesame’s tissues such as, leaves and seedlings are rich sources of vitamins B
6 and B
12. This multi tissue potential of the crop, allows for comprehensive dietary integration, from seed-based products to leafy greens and sprouted seedlings. Collectively, sesame’s unique B vitamin profile, coupled with its developmental and genetic variability, positions it as a cornerstone for biofortification programs, advancing efforts to address global B-vitamin inadequacies and enhance antioxidant nutrition through plant-based solutions. By leveraging the identified variations, future research should focus on vitamin bioavailability, processing effects, and genetic mechanisms underlying B-vitamin accumulation to fully realize sesame’s potential as a functional crop.
CRediT authorship contribution statement
Yijia Zhang, Ting Zhou and Linhai Wang conceived and designed the project and experiments. Yijia Zhang performed the experiments. Yijia Zhang analyzed the data, Ting Zhou and Desawi Hdru Teklu wrote the manuscript. Zishu Luo, Rong Zhou and Lei Wang assisted in preparing the materials. Jun You and Huan Li assisted in data analyzing. Linhai Wang revised the manuscript. All authors read and approved the manuscript.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Funding
This research was funded by the China Agriculture Research System (CARS-14), National Key R & D Program of China (2024YFD1600100), the Agricultural Science and Technology Innovation Program of the Chinese Academy of Agricultural Sciences (CAAS-ASTIP-2025-OCRI), Hubei International Science and Technology Cooperation Project (2024EHA055), Science and Technology Innovation Project of Hubei province (2025-620-000-001-031), Fundamental Research Funds for Central Non-profit Scientific Institution (1610172023003), and the National Center for Crops Germplasm Resources (NCCGR-2024-016).
Acknowledgments
We thank all of the laboratory members for their collaboration and constructive suggestions and Wuhan ProNets Testing Technology Co, Ltd. for B vitamins detection.
References
- Abdiani, N.; Kolahi, M.; Javaheriyan, M.; Sabaeian, M. Effect of storage conditions on nutritional value, oil content, and oil composition of sesame seeds. Journal of Agriculture and Food Research 2024, 16, 101117. [Google Scholar] [CrossRef]
- Asensi-Fabado, M. A.; Munné-Bosch, S. Vitamins in plants: Occurrence, biosynthesis and antioxidant function. Trends in Plant Science 2010, 15(10), 582–592. [Google Scholar] [CrossRef]
- Bechoff, A.; Bruyn, J.; Alpha, A.; Wieringa, F.; Greffeuille, V. Exploring the Complementarity of Fortification and Dietary Diversification to Combat Micronutrient Deficiencies: A Scoping Review. Current Developments in Nutrition 2023, 7(2), 100033. [Google Scholar] [CrossRef]
- Bdeir, R.; Aljabali, S. M.; Banihani, S. A. Role of pyridoxine and oxidative stress in asthenozoospermia. Heliyon 2024, 10, e34799. [Google Scholar] [CrossRef]
- Belal, A.; Mahmoud, R.; Mohamed, E. E.; Farghali, A.; Abo El-Ela, F. I.; Gamal, A.; Halfaya, F. M.; Khaled, E.; Farahat, A. A.; Hassan, A. H. E.; Ghoneim, M. M.; Taha, M.; Zaky, M. Y. A Novel Hydroxyapatite/Vitamin B12 Nanoformula for Treatment of Bone Damage: Preparation, Characterization, and Anti-Arthritic, Anti-Inflammatory, and Antioxidant Activities in Chemically Induced Arthritic Rats. Pharmaceuticals 2023, 16, 551. [Google Scholar] [CrossRef] [PubMed]
- Bonku, R.; Mikiashvili, N.; Yu, J. Impacts of Protease Treatment on the Contents of Tocopherols and B Vitamins in Peanuts. Journal of Food Research 2020, 9(6), 6. [Google Scholar] [CrossRef]
- Degnan, P. H.; Taga, M. E.; Goodman, A. L. Vitamin B12 as a Modulator of Gut Microbial Ecology. Cell Metabolism 2014, 20(5), 769–778. [Google Scholar] [CrossRef]
- Ding, H.; Reiss, A. B.; Pinkhasov, A.; Kasselman, L. J. Plants, Plants, and More Plants: Plant-Derived Nutrients and Their Protective Roles in Cognitive Function, Alzheimer’s Disease, and Other Dementias. Medicina 2022, 58(8), 1025. [Google Scholar] [CrossRef]
- Firat, C. K.; Ozkan, B. N.; Guler, E. M. Beneficial effects of vitamin B12 treatment in pediatric patients diagnosed with vitamin B12 deficiency regarding total-native thiol, oxidative stress, and mononuclear leukocyte DNA damage. Free Radical Research 2022, 56, 631–639. [Google Scholar] [CrossRef]
- Fitzpatrick, T. B.; Chapman, L. M. The importance of thiamine (vitamin B1) in plant health: From crop yield to biofortification. Journal of Biological Chemistry 2020, 295(34), 12002–12013. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, T. B.; Dalvit, I.; Chang, F.; Wang, K.; Fudge, J. B.; Chang, S.; Maillot, B.; Gruissem, W. Vitamin B1 enhancement in the endosperm of rice through thiamine sequestration. Plant Biotechnology Journal 2024, 22(8), 2330–2332. [Google Scholar] [CrossRef]
- Garg, M.; Sharma, A.; Vats, S.; Tiwari, V.; Kumari, A.; Mishra, V.; Krishania, M. Vitamins in Cereals: A Critical Review of Content, Health Effects, Processing Losses, Bioaccessibility, Fortification, and Biofortification Strategies for Their Improvement. Frontiers in Nutrition 2021, 8. [Google Scholar] [CrossRef]
- Hellmann, H.; Mooney, S. Vitamin B6: A Molecule for Human Health? Molecules: Basel, Switzerland, 2010; Volume 15, 1, pp. 442–459. [Google Scholar] [CrossRef]
- Hrubša, M.; Siatka, T.; Nejmanová, I.; Vopršalová, M.; Kujovská Krčmová, L.; Matoušová, K.; Javorská, L.; Macáková, K.; Mercolini, L.; Remião, F.; Máťuš, M.; Mladěnka, P.; on behalf of the OEMONOM. Biological Properties of Vitamins of the B-Complex, Part 1: Vitamins B1, B2, B3, and B5. Nutrients 2022, 14(3), 484. [Google Scholar] [CrossRef] [PubMed]
- Kefale, H.; Segla Koffi Dossou, S.; Li, F.; Jiang, N.; Zhou, R.; Wang, L.; Zhang, Y.; Li, D.; You, J.; Wang, L. Widely targeted metabolic profiling provides insights into variations in bioactive compounds and antioxidant activity of sesame, soybean, peanut, and perilla. Food Research International 2023, 174, 113586. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, D. B Vitamins and the Brain: Mechanisms, Dose and Efficacy—A Review. Nutrients 2016, 8(2), 68. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.-H.; Jung, J. W.; Yu, O. S.; Lee, S. Y.; Kim, M. J.; Lee, S. G.; Park, H. M.; Jo, Y.; Joo, Y.; Oh, S. W. Natural variation in tocopherols, B vitamins, and isoflavones in seeds of 13 Korean conventional soybean varieties. Applied Biological Chemistry 2024, 67(1), 51. [Google Scholar] [CrossRef]
- Kim, G.-P.; Lee, J.; Ahn, K. G.; Hwang, Y. S.; Choi, Y.; Chun, J.; Chang, W. S.; Choung, M. G. Differential responses of B vitamins in black soybean seeds. Food Chemistry 2014, 153, 101–108. [Google Scholar] [CrossRef]
- Lebiedzińska, A.; Szefer, P. Vitamins B in grain and cereal–grain food, soy-products and seeds. Food Chemistry 2006, 95(1), 116–122. [Google Scholar] [CrossRef]
- Lee, M. C.; Hsu, Y. J.; Shen, S. Y.; Ho, C. S.; Huang, C. C. A functional evaluation of anti-fatigue and exercise performance improvement following vitamin B complex supplementation in healthy humans, a randomized double-blind trial. International Journal of Medical Sciences 2023, 20(10), 1272–1281. [Google Scholar] [CrossRef]
- Liu, Z.; Farkas, P.; Wang, K.; Kohli, M.; Fitzpatrick, T. B. B vitamin supply in plants and humans: The importance of vitamer homeostasis. The Plant Journal 2022, 111(3), 662–682. [Google Scholar] [CrossRef]
- Mangel, N.; Fudge, J. B.; Gruissem, W.; Fitzpatrick, T. B.; Vanderschuren, H. Natural Variation in Vitamin B1 and Vitamin B6 Contents in Rice Germplasm. Frontiers in Plant Science 2022, 13, 856880. [Google Scholar] [CrossRef]
- Lindschinger, M; Tatzber, F; Schimetta, W; Irene, S; Lindschinger; Cvirn, G; Stanger, Olaf; Lamont, E; Wonisch, W. A Randomized Pilot Trial to Evaluate the Bioavailability of Natural versus Synthetic Vitamin B Complexes in Healthy Humans and Their Effects on Homocysteine, Oxidative Stress, and Antioxidant Levels. Oxidative Medicine and Cellular Longevity 2019, 6082613. [Google Scholar] [CrossRef]
- Mkrtchyan, G. V.; Üçal, M.; Müllebner, A.; Dumitrescu, S.; Kames, M.; Moldzio, R.; Molcanyi, M.; Schaefer, S.; Weidinger, A.; Schaefer, U.; Hescheler, J.; Duvigneau, J. C.; Redl, H.; Bunik, V. I.; Kozlov, A. V. Thiamine preserves mitochondrial function in a rat model of traumatic brain injury, preventing inactivation of the 2-oxoglutarate dehydrogenase complex. Biochimica et Biophysica Acta (BBA) Bioenergetics 2018, 1859(9), 925–931. [Google Scholar] [CrossRef]
- Nguyen, T. T. L.; Pham, T. M. N.; Ho, T. B.; Ly-Nguyen, B. Optimization of Vitamin B1, B2, and B6 Absorption in Nang Tay Dum Floating Rice Grains. Foods 2024, 13(17), 2650. [Google Scholar] [CrossRef] [PubMed]
- Radchuk, V.; Borisjuk, L. Physical, metabolic and developmental functions of the seed coat. Frontiers in Plant Science 2014, 5, 510. [Google Scholar] [CrossRef]
- Ray, A.; Dutta, S. Dietary Diversification and Its Impact on Human Health. In Nanotechnology in Functional Foods; 2022; Volume Chapter 9. [Google Scholar] [CrossRef]
- Safiul Azam, F. M.; Lian, T.; Liang, Q.; Wang, W.; Zhang, C.; Jiang, L. Variation of vitamin B contents in maize inbred lines: Potential genetic resources for biofortification. Frontiers in Nutrition 2022, 9, 1029119. [Google Scholar] [CrossRef]
- Shewry, P. R.; Van Schaik, F.; Ravel, C.; Charmet, G.; Rakszegi, M.; Bedo, Z.; Ward, J. L. Genotype and Environment Effects on the Contents of Vitamins B1, B2, B3, and B6 in Wheat Grain. Journal of Agricultural and Food Chemistry 2011, 59(19), 10564–10571. [Google Scholar] [CrossRef] [PubMed]
- Titcomb, T. J.; Tanumihardjo, S. A. Global Concerns with B Vitamin Statuses: Biofortification, Fortification, Hidden Hunger, Interactions, and Toxicity. Comprehensive Reviews in Food Science and Food Safety 2019, 18(6), 1968–1984. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Zhang, L.; Huang, X.; Wang, X.; Yang, R.; Mao, J.; Wang, X.; Wang, X.; Zhang, Q.; Li, P. Identification of Nutritional Components in Black Sesame Determined by Widely Targeted Metabolomics and Traditional Chinese Medicines. Molecules 2018, 23(5), 1180. [Google Scholar] [CrossRef]
- Watanabe, F.; Yabuta, Y.; Bito, T.; Teng, F. Vitamin B12-Containing Plant Food Sources for Vegetarians. Nutrients 2014, 6(5), 1861–1873. [Google Scholar] [CrossRef]
- Yang, Y.; Ke, Y.; Liu, X.; Zhang, Z.; Zhang, R.; Tian, F.; Zhi, L.; Zhao, G.; Lv, B.; Hua, S.; Wu, H. Navigating the B vitamins: Dietary diversity, microbial synthesis, and human health. Cell Host & Microbe 2024, 32(1), 12–18. [Google Scholar] [CrossRef] [PubMed]
Figure 1.
B-vitamins variations [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in seeds of five oilseed crops (Rs: rapeseed, Pl: perilla, So: soybean, Pe: peanut, and Se: sesame.
Figure 1.
B-vitamins variations [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in seeds of five oilseed crops (Rs: rapeseed, Pl: perilla, So: soybean, Pe: peanut, and Se: sesame.
Figure 2.
B vitamins variations [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in leaves of three oilseed crops (Rs: rapeseed, Pl: perilla, and Se: sesame) and leafy vegetables (Sp: spinach and Le: lettuce).
Figure 2.
B vitamins variations [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in leaves of three oilseed crops (Rs: rapeseed, Pl: perilla, and Se: sesame) and leafy vegetables (Sp: spinach and Le: lettuce).
Figure 3.
B-vitamins variations [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in seedlings of three oilseed crops (So: soybean, Pe: peanut, Se: sesame).
Figure 3.
B-vitamins variations [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in seedlings of three oilseed crops (So: soybean, Pe: peanut, Se: sesame).
Figure 4.
Comparison of B vitamins content among different tissues of five oilseed crops. Sesame (A), rapeseed (B), perilla (C), soybean (D), and peanut (E).
Figure 4.
Comparison of B vitamins content among different tissues of five oilseed crops. Sesame (A), rapeseed (B), perilla (C), soybean (D), and peanut (E).
Figure 5.
Comparison of the content of B vitamins [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in black and white sesame seeds. H: Heat map of relative content of seven B vitamins in different tissues of black and white sesame seed.
Figure 5.
Comparison of the content of B vitamins [B1 (A), B2 (B), B3 (C), B5 (D), B6 (E), B9 (F) and B12 (G)] in black and white sesame seeds. H: Heat map of relative content of seven B vitamins in different tissues of black and white sesame seed.
Figure 6.
Diagrammatical illustration of significant B vitamin contents across the studied materials.
Figure 6.
Diagrammatical illustration of significant B vitamin contents across the studied materials.
Table 1.
Variation in vitamin B3 content among sesame germplasms.
Table 1.
Variation in vitamin B3 content among sesame germplasms.
Seed coat color
|
Number |
|
Vitamin B3 (μg/g) content |
| Max |
Min |
Mean |
SD |
CV |
| White |
38 |
55.87 |
23.57 |
38.79 |
5.94 |
16.44% |
| Black |
18 |
45.14 |
25.37 |
38.70 |
5.40 |
13.93% |
| Yellow |
15 |
82.25 |
26.24 |
40.66 |
13.35 |
32.83% |
| Brown |
21 |
48.64 |
24.08 |
37.90 |
7.18 |
18.94% |
| Total |
92 |
82.25 |
23.57 |
39.28 |
7.86 |
15.12% |
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).