Submitted:
05 January 2026
Posted:
07 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Death Signals in the Reprogramming of Differentiated Cells at Wound Sites
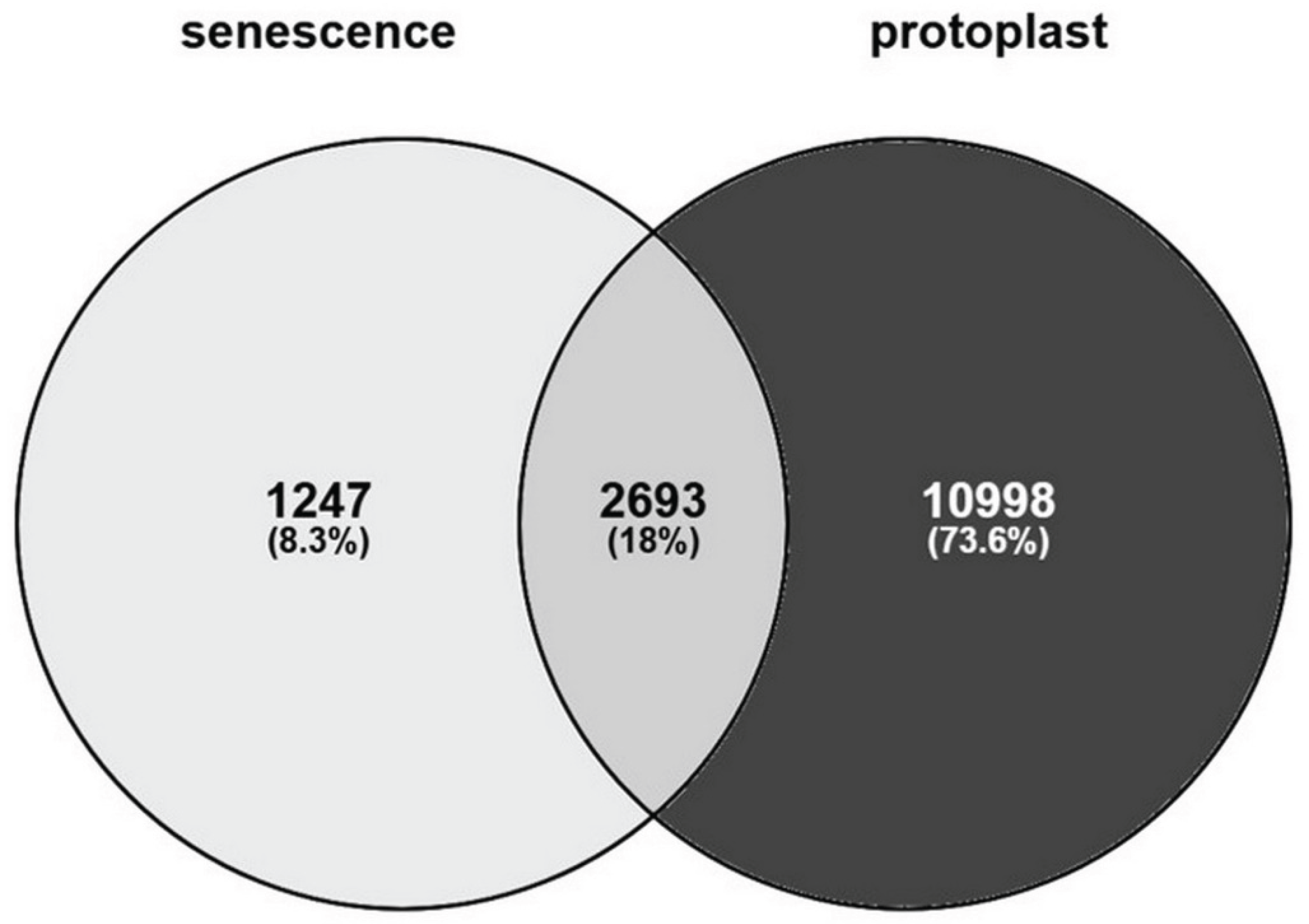
3. The Senescence-like State of Protoplast-Derived Cells
4. The Formation and Nature of Callus Tissues
5. Summary
6. Conclusions
Acknowledgments
References
- Birnbaum, K.D.; Alvarado, A.S. Slicing across Kingdoms: Regeneration in Plants and Animals. Cell 2008, 132, 697–710. [Google Scholar] [CrossRef] [PubMed]
- Christiaens, F.; Canher, B.; Lanssens, F.; Bisht, A.; Stael, S.; De Veylder, L.; Heyman, J. Pars Pro Toto: Every Single Cell Matters. Front. Plant Sci. 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Mironova, V.; Xu, J. A Single-Cell View of Tissue Regeneration in Plants. Curr. Opin. Plant Biol. 2019, 52, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Fehér, A. Somatic Embryogenesis - Stress-Induced Remodeling of Plant Cell Fate. Biochim. Biophys. Acta - Gene Regul. Mech. 2015, 1849. [Google Scholar] [CrossRef]
- Lup, S.D.; Tian, X.; Xu, J.; Pérez-Pérez, J.M. Wound Signaling of Regenerative Cell Reprogramming. Plant Sci. 2016, 250, 178–187. [Google Scholar] [CrossRef]
- Jopling, C.; Boue, S.; Belmonte, J.C.I. Dedifferentiation, Transdifferentiation and Reprogramming: Three Routes to Regeneration. Nat. Rev. Mol. Cell Biol. 2011, 12, 79–89. [Google Scholar] [CrossRef]
- Fehér, A. Callus, Dedifferentiation, Totipotency, Somatic Embryogenesis: What These Terms Mean in the Era of Molecular Plant Biology? Front. Plant Sci. 2019, 10, 536. [Google Scholar] [CrossRef]
- Sugimoto, K.; Gordon, S.P.; Meyerowitz, E.M. Regeneration in Plants and Animals: Dedifferentiation, Transdifferentiation, or Just Differentiation? Trends Cell Biol. 2011, 21, 212–218. [Google Scholar] [CrossRef]
- Jiang, F.; Zhu, J.; Liu, H.-L. Protoplasts: A Useful Research System for Plant Cell Biology, Especially Dedifferentiation. Protoplasma 2013, 250, 1231–1238. [Google Scholar] [CrossRef]
- Ikeuchi, M.; Sugimoto, K.; Iwase, A. Plant Callus: Mechanisms of Induction and Repression. Plant Cell 2013, 25, 3159–3173. [Google Scholar] [CrossRef]
- Fehér, A. A Common Molecular Signature Indicates the Pre-Meristematic State of Plant Calli. Int. J. Mol. Sci. 2023, 24, 13122. [Google Scholar] [CrossRef] [PubMed]
- Kopeina, G.S.; Efimenko, A.Yu.; Tkachuk, V.A.; Zhivotovsky, B. Programmed Cell Death and Tissue Regeneration: A Link That Should Be Resolved. Physiol. Rev. 2025, 105, 2377–2412. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, Y.; Steller, H. Live to Die Another Way: Modes of Programmed Cell Death and the Signals Emanating from Dying Cells. Nat. Rev. Mol. Cell Biol. 2015, 16, 329–344. [Google Scholar] [CrossRef]
- Vriz, S.; Reiter, S.; Galliot, B. Cell Death: A Program to Regenerate. Curr. Top. Dev. Biol. 2014, 108, 121–151. [Google Scholar] [CrossRef] [PubMed]
- Guerin, D.J.; Kha, C.X.; Tseng, K.A.-S. From Cell Death to Regeneration: Rebuilding After Injury. Front. Cell Dev. Biol. 2021, 9. [Google Scholar] [CrossRef]
- Li, F.; Huang, Q.; Chen, J.; Peng, Y.; Roop, D.R.; Bedford, J.S.; Li, C.-Y. Apoptotic Cells Activate the “Phoenix Rising” Pathway to Promote Wound Healing and Tissue Regeneration. Sci. Signal. 2010, 3, ra13–ra13. [Google Scholar] [CrossRef]
- Ikeuchi, M.; Ogawa, Y.; Iwase, A.; Sugimoto, K. Plant Regeneration: Cellular Origins and Molecular Mechanisms. Development 2016, 143, 1442–1451. [Google Scholar] [CrossRef]
- Sugimoto, K.; Temman, H.; Kadokura, S.; Matsunaga, S. To Regenerate or Not to Regenerate: Factors That Drive Plant Regeneration. Curr. Opin. Plant Biol. 2019, 47, 138–150. [Google Scholar] [CrossRef]
- Ibáñez, S.; Carneros, E.; Testillano, P.S.; Pérez-Pérez, J.M. Advances in Plant Regeneration: Shake, Rattle and Roll. Plants 2020, 9, 897. [Google Scholar] [CrossRef]
- Daneva, A.; Gao, Z.; Durme, M.V.; Nowack, M.K. Functions and Regulation of Programmed Cell Death in Plant Development. Annu. Rev. Cell Dev. Biol. 2016, 32, 441–468. [Google Scholar] [CrossRef]
- Xu, D.; Yang, L. Regeneration and Defense: Unveiling the Molecular Interplay in Plants. New Phytol. 2025, 246, 2484–2494. [Google Scholar] [CrossRef] [PubMed]
- Vega-Muñoz, I.; Duran-Flores, D.; Fernández-Fernández, Á.D.; Heyman, J.; Ritter, A.; Stael, S. Breaking Bad News: Dynamic Molecular Mechanisms of Wound Response in Plants. Front. Plant Sci. 2020, 11. [Google Scholar] [CrossRef]
- Hoermayer, L.; Montesinos, J.C.; Marhava, P.; Benková, E.; Yoshida, S.; Friml, J. Wounding-Induced Changes in Cellular Pressure and Localized Auxin Signalling Spatially Coordinate Restorative Divisions in Roots. Proc. Natl. Acad. Sci. 2020, 117, 15322–15331. [Google Scholar] [CrossRef]
- Heil, M.; Ibarra-Laclette, E.; Adame-Álvarez, R.M.; Martínez, O.; Ramirez-Chávez, E.; Molina-Torres, J.; Herrera-Estrella, L. How Plants Sense Wounds: Damaged-Self Recognition Is Based on Plant-Derived Elicitors and Induces Octadecanoid Signaling. PLOS ONE 2012, 7, e30537. [Google Scholar] [CrossRef] [PubMed]
- Cutler, S.R.; Somerville, C.R. Imaging Plant Cell Death: GFP-Nit1 Aggregation Marks an Early Step of Wound and Herbicide Induced Cell Death. BMC Plant Biol. 2005, 5, 4. [Google Scholar] [CrossRef]
- Heyman, J.; Cools, T.; Canher, B.; Shavialenka, S.; Traas, J.; Vercauteren, I.; Van den Daele, H.; Persiau, G.; De Jaeger, G.; Sugimoto, K.; et al. The Heterodimeric Transcription Factor Complex ERF115–PAT1 Grants Regeneration Competence. Nat. Plants 2016, 2, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Iakimova, E.T.; Woltering, E.J. The Wound Response in Fresh-Cut Lettuce Involves Programmed Cell Death Events. Protoplasma 2018, 255, 1225–1238. [Google Scholar] [CrossRef]
- Fagundes, D.; Bohn, B.; Cabreira, C.; Leipelt, F.; Dias, N.; Bodanese-Zanettini, M.H.; Cagliari, A. Caspases in Plants: Metacaspase Gene Family in Plant Stress Responses. Funct. Integr. Genomics 2015, 15, 639–649. [Google Scholar] [CrossRef]
- Hander, T.; Fernández-Fernández, Á.D.; Kumpf, R.P.; Willems, P.; Schatowitz, H.; Rombaut, D.; Staes, A.; Nolf, J.; Pottie, R.; Yao, P.; et al. Damage on Plants Activates Ca2+-Dependent Metacaspases for Release of Immunomodulatory Peptides. Science 2019, 363, eaar7486. [Google Scholar] [CrossRef]
- Yang, W.; Zhai, H.; Wu, F.; Deng, L.; Chao, Y.; Meng, X.; Chen, Q.; Liu, C.; Bie, X.; Sun, C.; et al. Peptide REF1 Is a Local Wound Signal Promoting Plant Regeneration. Cell 2024, 187, 3024–3038.e14. [Google Scholar] [CrossRef]
- Geng, X.; Zhang, X.; Zhou, W. Triggering Regeneration: A New Recognized Local Wound Signal in Plants. Innov. Life 2025, 3, 100124–2. [Google Scholar] [CrossRef]
- Iwase, A.; Mitsuda, N.; Koyama, T.; Hiratsu, K.; Kojima, M.; Arai, T.; Inoue, Y.; Seki, M.; Sakakibara, H.; Sugimoto, K.; et al. The AP2/ERF Transcription Factor WIND1 Controls Cell Dedifferentiation in Arabidopsis. Curr. Biol. 2011, 21, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Seo, S.; Sano, H.; Ohashi, Y. Jasmonic Acid in Wound Signal Transduction Pathways. Physiol. Plant. 1997, 101, 740–745. [Google Scholar] [CrossRef]
- Reinbothe, C.; Springer, A.; Samol, I.; Reinbothe, S. Plant Oxylipins: Role of Jasmonic Acid during Programmed Cell Death, Defence and Leaf Senescence. FEBS J. 2009, 276, 4666–4681. [Google Scholar] [CrossRef]
- Kim, J.; Chang, C.; Tucker, M.L. To Grow Old: Regulatory Role of Ethylene and Jasmonic Acid in Senescence. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef]
- Ikeuchi, M.; Favero, D.S.; Sakamoto, Y.; Iwase, A.; Coleman, D.; Rymen, B.; Sugimoto, K. Molecular Mechanisms of Plant Regeneration. Annu. Rev. Plant Biol. 2019, 70, 377–406. [Google Scholar] [CrossRef]
- Ikeuchi, M.; Rymen, B.; Sugimoto, K. How Do Plants Transduce Wound Signals to Induce Tissue Repair and Organ Regeneration? Curr. Opin. Plant Biol. 2020, 57, 72–77. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, F.; Chen, L.; Pan, Y.; Sun, L.; Bao, N.; Zhang, T.; Cui, C.-X.; Qiu, Z.; Zhang, Y.; et al. Jasmonate-Mediated Wound Signalling Promotes Plant Regeneration. Nat. Plants 2019, 5, 491–497. [Google Scholar] [CrossRef]
- Zhang, G.; Liu, W.; Gu, Z.; Wu, S.; E, Y.; Zhou, W.; Lin, J.; Xu, L. Roles of the Wound Hormone Jasmonate in Plant Regeneration. J. Exp. Bot. 2021, erab508. [Google Scholar] [CrossRef] [PubMed]
- Zhou, W.; Lozano-Torres, J.L.; Blilou, I.; Zhang, X.; Zhai, Q.; Smant, G.; Li, C.; Scheres, B. A Jasmonate Signaling Network Activates Root Stem Cells and Promotes Regeneration. Cell 2019, 177, 942–956.e14. [Google Scholar] [CrossRef] [PubMed]
- Ikeuchi, M.; Iwase, A.; Rymen, B.; Lambolez, A.; Kojima, M.; Takebayashi, Y.; Heyman, J.; Watanabe, S.; Seo, M.; De Veylder, L.; et al. Wounding Triggers Callus Formation via Dynamic Hormonal and Transcriptional Changes1[OPEN]. Plant Physiol. 2017, 175, 1158–1174. [Google Scholar] [CrossRef]
- Maruyama, K.; Ikeuchi, M. Multifaceted Controls on Auxin Metabolism during Cellular Reprogramming and Organ Regeneration in Plants. J. Exp. Bot. 2026, 77, 141–148. [Google Scholar] [CrossRef]
- Omary, M.; Matosevich, R.; Efroni, I. Systemic Control of Plant Regeneration and Wound Repair. New Phytol. 2023, 237, 408–413. [Google Scholar] [CrossRef]
- Kacprzyk, J.; Burke, R.; Schwarze, J.; McCabe, P.F. Plant Programmed Cell Death Meets Auxin Signalling. FEBS J. 2022, 289, 1731–1745. [Google Scholar] [CrossRef]
- Heyman, J.; Cools, T.; Vandenbussche, F.; Heyndrickx, K.S.; Van Leene, J.; Vercauteren, I.; Vanderauwera, S.; Vandepoele, K.; De Jaeger, G.; Van Der Straeten, D.; et al. ERF115 Controls Root Quiescent Center Cell Division and Stem Cell Replenishment. Science 2013, 342, 860–863. [Google Scholar] [CrossRef]
- Geem, K.R.; Kim, H.; Ryu, H. SCFFBS1 Regulates Root Quiescent Center Cell Division via Protein Degradation of APC/CCCS52A2. Mol. Cells 2022, 45, 695–701. [Google Scholar] [CrossRef]
- Zhang, Y.; Zheng, L.; Hong, J.H.; Gong, X.; Zhou, C.; Pérez-Pérez, J.M.; Xu, J. TOPOISOMERASE1α Acts through Two Distinct Mechanisms to Regulate Stele and Columella Stem Cell Maintenance. Plant Physiol. 2016, 171, 483–493. [Google Scholar] [CrossRef] [PubMed]
- Raya-González, J.; Oropeza-Aburto, A.; López-Bucio, J.S.; Guevara-García, Á.A.; de Veylder, L.; López-Bucio, J.; Herrera-Estrella, L. MEDIATOR18 Influences Arabidopsis Root Architecture, Represses Auxin Signaling and Is a Critical Factor for Cell Viability in Root Meristems. Plant J. 2018, 96, 895–909. [Google Scholar] [CrossRef]
- Kong, X.; Tian, H.; Yu, Q.; Zhang, F.; Wang, R.; Gao, S.; Xu, W.; Liu, J.; Shani, E.; Fu, C.; et al. PHB3 Maintains Root Stem Cell Niche Identity through ROS-Responsive AP2/ERF Transcription Factors in Arabidopsis. Cell Rep. 2018, 22, 1350–1363. [Google Scholar] [CrossRef] [PubMed]
- Larsen, P.B.; He, S.; Meyer, T.J.; Szurman-Zubrzycka, M.; Alfs, C.; Kwasniewska, J.; Pervis, A.; Gajecka, M.; Veerabahu, A.; Beaulieu, T.R.; et al. The Stem Cell Niche Transcription Factor ETHYLENE RESPONSE FACTOR 115 Participates in Aluminum-Induced Terminal Differentiation in Arabidopsis Roots. Plant Cell Environ. 2024, 47, 4432–4448. [Google Scholar] [CrossRef] [PubMed]
- Canher, B.; Heyman, J.; Savina, M.; Devendran, A.; Eekhout, T.; Vercauteren, I.; Prinsen, E.; Matosevich, R.; Xu, J.; Mironova, V.; et al. Rocks in the Auxin Stream: Wound-Induced Auxin Accumulation and ERF115 Expression Synergistically Drive Stem Cell Regeneration. Proc. Natl. Acad. Sci. U. S. A 2020, 117, 16667–16677. [Google Scholar] [CrossRef]
- Chang, X.; Riemann, M.; Liu, Q.; Nick, P. Actin as Deathly Switch? How Auxin Can Suppress Cell-Death Related Defence. PLOS ONE 2015, 10, e0125498. [Google Scholar] [CrossRef]
- Hong, J.H.; Savina, M.; Du, J.; Devendran, A.; Kannivadi Ramakanth, K.; Tian, X.; Sim, W.S.; Mironova, V.V.; Xu, J. A Sacrifice-for-Survival Mechanism Protects Root Stem Cell Niche from Chilling Stress. Cell 2017, 170, 102–113.e14. [Google Scholar] [CrossRef]
- Xuan, W.; Band, L.R.; Kumpf, R.P.; Van Damme, D.; Parizot, B.; De Rop, G.; Opdenacker, D.; Möller, B.K.; Skorzinski, N.; Njo, M.F.; et al. Cyclic Programmed Cell Death Stimulates Hormone Signaling and Root Development in Arabidopsis. Science 2016, 351, 384–387. [Google Scholar] [CrossRef]
- Sheldrake, A.R. The Production of Auxin by Dying Cells. J. Exp. Bot. 2021, 72, 2288–2300. [Google Scholar] [CrossRef]
- Matosevich, R.; Cohen, I.; Gil-Yarom, N.; Modrego, A.; Friedlander-Shani, L.; Verna, C.; Scarpella, E.; Efroni, I. Local Auxin Biosynthesis Is Required for Root Regeneration after Wounding. Nat. Plants 2020, 6, 1020–1030. [Google Scholar] [CrossRef]
- Ribeiro, C.; de Melo, B.P.; Lourenço-Tessutti, I.T.; Ballesteros, H.F.; Ribeiro, K.V.G.; Menuet, K.; Heyman, J.; Hemerly, A.; de Sá, M.F.G.; De Veylder, L.; et al. The Regeneration Conferring Transcription Factor Complex ERF115-PAT1 Coordinates a Wound-Induced Response in Root-Knot Nematode Induced Galls. New Phytol. 2024, 241, 878–895. [Google Scholar] [CrossRef] [PubMed]
- Bisht, A.; Eekhout, T.; Canher, B.; Lu, R.; Vercauteren, I.; De Jaeger, G.; Heyman, J.; De Veylder, L. PAT1-Type GRAS-Domain Proteins Control Regeneration by Activating DOF3.4 to Drive Cell Proliferation in Arabidopsis Roots. Plant Cell 2023, 35, 1513–1531. [Google Scholar] [CrossRef] [PubMed]
- Chupeau, M.-C.; Granier, F.; Pichon, O.; Renou, J.-P.; Gaudin, V.; Chupeau, Y. Characterization of the Early Events Leading to Totipotency in an Arabidopsis Protoplast Liquid Culture by Temporal Transcript Profiling. Plant Cell 2013, 25, 2444–2463. [Google Scholar] [CrossRef]
- Grosset, J.; Marty, I.; Chartier, Y.; Meyer, Y. mRNAs Newly Synthesized by Tobacco Mesophyll Protoplasts Are Wound-Inducible. Plant Mol. Biol. 1990, 15, 485–496. [Google Scholar] [CrossRef] [PubMed]
- Papadakis, A.K.; Roubelakis-Angelakis, K.A. The Generation of Active Oxygen Species Differs in Tobacco and Grapevine Mesophyll Protoplasts. Plant Physiol. 1999, 121, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Suzuki, K.; Kawasaki, H.; Watanabe, Y. Differential Responses of Brassica Napus and Petunia Hybrida to Leaf Protoplast Isolation Stress. Physiol. Plant. 2002, 114, 645–651. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Kawasaki, H.; Itho, Y.; Watanabe, Y. Senescence Development of Brassica Napus Leaf Protoplast during Isolation and Subsequent Culture. J. Plant Physiol. 1998, 152, 487–493. [Google Scholar] [CrossRef]
- Watanabe, M.; Setoguchi, D.; Uehara, K.; Ohtsuka, W.; Watanabe, Y. Apoptosis-like Cell Death of Brassica Napus Leaf Protoplasts. New Phytol. 2002, 156, 417–426. [Google Scholar] [CrossRef]
- Damri, M.; Granot, G.; Ben-Meir, H.; Avivi, Y.; Plaschkes, I.; Chalifa-Caspi, V.; Wolfson, M.; Fraifeld, V.; Grafi, G. Senescing Cells Share Common Features with Dedifferentiating Cells. Rejuvenation Res. 2009, 12, 435–443. [Google Scholar] [CrossRef]
- Grafi, G.; Chalifa-Caspi, V.; Nagar, T.; Plaschkes, I.; Barak, S.; Ransbotyn, V. Plant Response to Stress Meets Dedifferentiation. Planta 2011, 233, 433–438. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, Y.; Li, S.; Tan, S.; Cao, J.; Wang, H.-L.; Luo, J.; Guo, H.; Zhang, Z.; Li, Z. Leaf Senescence Database v5.0: A Comprehensive Repository for Facilitating Plant Senescence Research. J. Mol. Biol. 2024, 436, 168530. [Google Scholar] [CrossRef]
- Xu, M.; Du, Q.; Tian, C.; Wang, Y.; Jiao, Y. Stochastic Gene Expression Drives Mesophyll Protoplast Regeneration. Sci. Adv. 2021. [Google Scholar] [CrossRef]
- Heyman, J.; Cools, T.; Vandenbussche, F.; Heyndrickx, K.S.; Van Leene, J.; Vercauteren, I.; Vanderauwera, S.; Vandepoele, K.; De Jaeger, G.; Van Der Straeten, D.; et al. ERF115 Controls Root Quiescent Center Cell Division and Stem Cell Replenishment. Science 2013, 342, 860–863. [Google Scholar] [CrossRef]
- Johnson, R.A.; Conklin, P.A.; Tjahjadi, M.; Missirian, V.; Toal, T.; Brady, S.M.; Britt, A.B. SUPPRESSOR OF GAMMA RESPONSE1 Links DNA Damage Response to Organ Regeneration. Plant Physiol. 2018, 176, 1665–1675. [Google Scholar] [CrossRef]
- Zhao, J.; Morozova, N.; Williams, L.; Libs, L.; Avivi, Y.; Grafi, G. Two Phases of Chromatin Decondensation during Dedifferentiation of Plant Cells: Distinction between Competence for Cell Fate Switch and a Commitment for S Phase. J. Biol. Chem. 2001, 276, 22772–22778. [Google Scholar] [CrossRef]
- Florentin, A.; Damri, M.; Grafi, G. Stress Induces Plant Somatic Cells to Acquire Some Features of Stem Cells Accompanied by Selective Chromatin Reorganization. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2013, 242, 1121–1133. [Google Scholar] [CrossRef]
- Grafi, G. How Cells Dedifferentiate: A Lesson from Plants. Dev. Biol. 2004, 268, 1–6. [Google Scholar] [CrossRef]
- Sakamoto, Y.; Kawamura, A.; Suzuki, T.; Segami, S.; Maeshima, M.; Polyn, S.; De Veylder, L.; Sugimoto, K. Transcriptional Activation of Auxin Biosynthesis Drives Developmental Reprogramming of Differentiated Cells. Plant Cell 2022, 34, 4348–4365. [Google Scholar] [CrossRef]
- Williams, L.; Zhao, J.; Morozova, N.; Li, Y.; Avivi, Y.; Grafi, G. Chromatin Reorganization Accompanying Cellular Dedifferentiation Is Associated with Modifications of Histone H3, Redistribution of HP1, and Activation of E2F-Target Genes. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2003, 228, 113–120. [Google Scholar] [CrossRef]
- Avivi, Y.; Morad, V.; Ben-Meir, H.; Zhao, J.; Kashkush, K.; Tzfira, T.; Citovsky, V.; Grafi, G. Reorganization of Specific Chromosomal Domains and Activation of Silent Genes in Plant Cells Acquiring Pluripotentiality. Dev. Dyn. Off. Publ. Am. Assoc. Anat. 2004, 230, 12–22. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, T.; Miskolczi, P.; Ayaydin, F.; Mészáros, T.; Dudits, D.; Fehér, A. Exogenous Auxin and Cytokinin Dependent Activation of CDKs and Cell Division in Leaf Protoplast-Derived Cells of Alfalfa. 2000, Vol. 32, 129–141. [Google Scholar] [CrossRef]
- Fehér, A.; Ötvös, K.; Pasternak, T.P.; Szandtner, A.P. The Involvement of Reactive Oxygen Species (ROS) in the Cell Cycle Activation (G 0-to-G 1 Transition) of Plant Cells. 2007, Vol. 51, 823–826. [Google Scholar]
- Lee, K.; Park, O.-S.; Jung, S.-J.; Seo, P.J. Histone Deacetylation-Mediated Cellular Dedifferentiation in Arabidopsis. J. Plant Physiol. 2016, 191, 95–100. [Google Scholar] [CrossRef]
- Zhai, N.; Xu, L. Pluripotency Acquisition in the Middle Cell Layer of Callus Is Required for Organ Regeneration. Nat. Plants 2021, 7, 1453–1460. [Google Scholar] [CrossRef] [PubMed]
- Hardtke, C.S.; Berleth, T. The Arabidopsis Gene MONOPTEROS Encodes a Transcription Factor Mediating Embryo Axis Formation and Vascular Development. EMBO J. 1998, 17, 1405–1411. [Google Scholar] [CrossRef]
- Aida, M.; Beis, D.; Heidstra, R.; Willemsen, V.; Blilou, I.; Galinha, C.; Nussaume, L.; Noh, Y.-S.; Amasino, R.; Scheres, B. The PLETHORA Genes Mediate Patterning of the Arabidopsis Root Stem Cell Niche. Cell 2004, 119, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Schlereth, A.; Möller, B.; Liu, W.; Kientz, M.; Flipse, J.; Rademacher, E.H.; Schmid, M.; Jürgens, G.; Weijers, D. MONOPTEROS Controls Embryonic Root Initiation by Regulating a Mobile Transcription Factor. Nature 2010, 464, 913–916. [Google Scholar] [CrossRef] [PubMed]
- Hofhuis, H.; Laskowski, M.; Du, Y.; Prasad, K.; Grigg, S.; Pinon, V.; Scheres, B. Phyllotaxis and Rhizotaxis in Arabidopsis Are Modified by Three PLETHORA Transcription Factors. Curr. Biol. 2013, 23, 956–962. [Google Scholar] [CrossRef]
- Horstman, A.; Willemsen, V.; Boutilier, K.; Heidstra, R. AINTEGUMENTA-LIKE Proteins: Hubs in a Plethora of Networks. Trends Plant Sci. 2014, 19, 146–157. [Google Scholar] [CrossRef]
- Krogan, N.T.; Marcos, D.; Weiner, A.I.; Berleth, T. The Auxin Response Factor MONOPTEROS Controls Meristem Function and Organogenesis in Both the Shoot and Root through the Direct Regulation of PIN Genes. New Phytol. 2016, 212, 42–50. [Google Scholar] [CrossRef]
- QingQing, L.; WenBin, N.; HanMa, Z. Progress on the functions of ARF5/MONOPTEROS(MP). Acta Bot. Boreali-Occident. Sin. 2016, 36, 197–203. [Google Scholar]
- Du, Y.; Scheres, B. PLETHORA Transcription Factors Orchestrate de Novo Organ Patterning during Arabidopsis Lateral Root Outgrowth. Proc. Natl. Acad. Sci. 2017, 114, 11709–11714. [Google Scholar] [CrossRef] [PubMed]
- Burkart, R.C.; Strotmann, V.I.; Kirschner, G.K.; Akinci, A.; Czempik, L.; Dolata, A.; Maizel, A.; Weidtkamp-Peters, S.; Stahl, Y. PLETHORA-WOX5 Interaction and Subnuclear Localization Control Arabidopsis Root Stem Cell Maintenance. EMBO Rep. 2022, 23, e54105. [Google Scholar] [CrossRef]
- Grunewald, W.; De Smet, I.; Lewis, D.R.; Löfke, C.; Jansen, L.; Goeminne, G.; Vanden Bossche, R.; Karimi, M.; De Rybel, B.; Vanholme, B.; et al. Transcription Factor WRKY23 Assists Auxin Distribution Patterns during Arabidopsis Root Development through Local Control on Flavonol Biosynthesis. Proc. Natl. Acad. Sci. U. S. A 2012, 109, 1554–1559. [Google Scholar] [CrossRef]
- Grunewald, W.; De Smet, I.; De Rybel, B.; Robert, H.S.; van de Cotte, B.; Willemsen, V.; Gheysen, G.; Weijers, D.; Friml, J.; Beeckman, T. Tightly Controlled WRKY23 Expression Mediates Arabidopsis Embryo Development. EMBO Rep. 2013, 14, 1136–1142. [Google Scholar] [CrossRef]
- Makkena, S.; Lamb, R.S. The bHLH Transcription Factor SPATULA Regulates Root Growth by Controlling the Size of the Root Meristem. BMC Plant Biol. 2013, 13, 1. [Google Scholar] [CrossRef] [PubMed]
- Prát, T.; Hajný, J.; Grunewald, W.; Vasileva, M.; Molnár, G.; Tejos, R.; Schmid, M.; Sauer, M.; Friml, J. WRKY23 Is a Component of the Transcriptional Network Mediating Auxin Feedback on PIN Polarity. PLOS Genet. 2018, 14, e1007177. [Google Scholar] [CrossRef] [PubMed]
- Chlyah, A.; Van, M.T.T. Differential Reactivity in Epidermal Cells of Begonia Rex Excised and Grown in Vitro. Physiol. Plant. 1975, 35, 16–20. [Google Scholar] [CrossRef]
- Venverloo, C.J.; Koster, J.; Libbenga, K.R. The Formation of Adventitious Organs: IV. The Ontogeny of Shoots and Leaves From Epidermis Cells of Nautilocalyx Lynchii. Z. Für Pflanzenphysiol 1983, 109, 55–67. [Google Scholar] [CrossRef]
- Morinaka, H.; Coleman, D.; Sugimoto, K.; Iwase, A. Molecular Mechanisms of Plant Regeneration from Differentiated Cells: Approaches from Historical Tissue Culture Systems. Plant Cell Physiol. 2023, 64, 297–304. [Google Scholar] [CrossRef]
- Davey, M.R.; Freason, E.M.; Withers, L.A.; Power, J.B. Observations on the Morphology, Ultrastructure and Regeneration of Tobacco Leaf Epidermal Protoplasts. Plant Sci. Lett. 1974, 2, 23–27. [Google Scholar] [CrossRef]
- Javelle, M.; Vernoud, V.; Rogowsky, P.M.; Ingram, G.C. Epidermis: The Formation and Functions of a Fundamental Plant Tissue. New Phytol. 2011, 189, 17–39. [Google Scholar] [CrossRef]
- Atta, R.; Laurens, L.; Boucheron-Dubuisson, E.; Guivarc’h, A.; Carnero, E.; Giraudat-Pautot, V.; Rech, P.; Chriqui, D. Pluripotency of Arabidopsis Xylem Pericycle Underlies Shoot Regeneration from Root and Hypocotyl Explants Grown in Vitro. Plant J. Cell Mol. Biol. 2009, 57, 626–644. [Google Scholar] [CrossRef]
- Sugimoto, K.; Jiao, Y.; Meyerowitz, E.M. Arabidopsis Regeneration from Multiple Tissues Occurs via a Root Development Pathway. Dev. Cell 2010, 18, 463–471. [Google Scholar] [CrossRef]
- Liu, H.; Wang, G.-C.; Feng, Z.; Zhu, J. Screening of Genes Associated with Dedifferentiation and Effect of LBD29 on Pericycle Cells in Arabidopsis Thaliana. Plant Growth Regul. 2010, 62, 127–136. [Google Scholar] [CrossRef]
- Xiao, W.; Molina, D.; Wunderling, A.; Ripper, D.; Vermeer, J.E.M.; Ragni, L. Pluripotent Pericycle Cells Trigger Different Growth Outputs by Integrating Developmental Cues into Distinct Regulatory Modules. Curr. Biol. 2020, 30, 4384–4398.e5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Umeda, M.; Kakimoto, T. Pericycle Cell Division Competence Underlies Various Developmental Programs. Plant Biotechnol. 2022, 39, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Fehér, A.; Bernula, D.; Gémes, K. The Many Ways of Somatic Embryo Initiation. In Somatic Embryogenesis: Fundamental Aspects and Applications; Springer International Publishing, 2016; pp. 23–37. ISBN 978-3-319-33705-0. [Google Scholar]
| protoplasts versus leaf cells | 4-day-old versus 0-day-old protoplasts | ||||||
| GID | log2FC | P value | FDR | log2FC | P value | FDR | |
| ERF115 | AT5G07310 | 11,45 | 1,8251E-37 | 0,00 | -1,31 | 0,00135717 | 0,00205361 |
| PAT1 | AT5G48150 | 0,75 | 8,1593E-06 | 1,3381E-05 | -2,18 | 3,7995E-35 | 4,3449E-34 |
| ARF5 | AT1G19850 | 6,59 | 1,169E-142 | 2,779E-140 | -0,79 | 2,7827E-05 | 4,9201E-05 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




