Submitted:
05 January 2026
Posted:
06 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
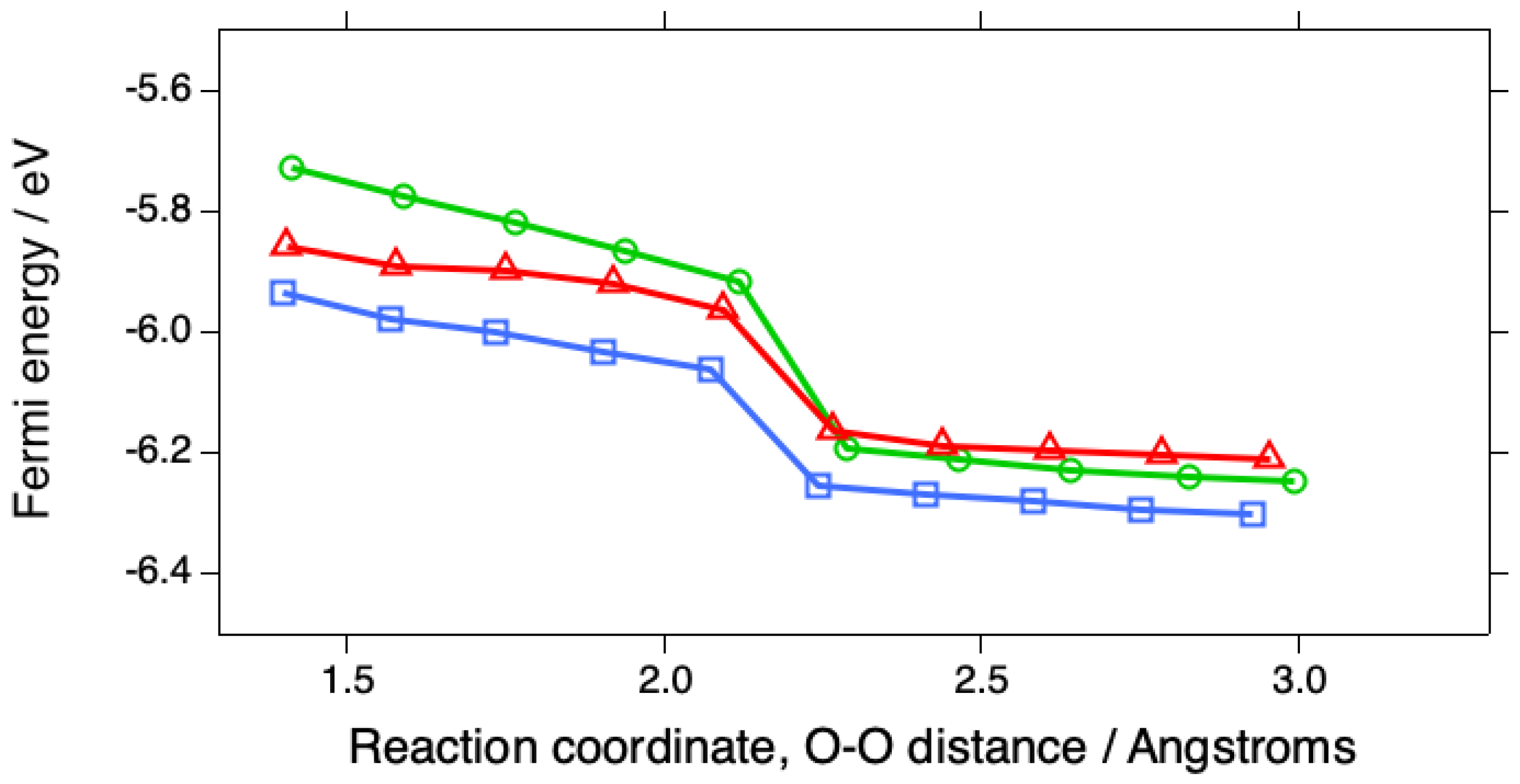
2. Results
3. Discussion
4. Materials and Methods
Supplementary Materials
Funding
Data Availability Statement
Conflicts of Interest
References
- Calle-Vallejo, F.; Loffreda, D.; KoperMarc, T.M.; Sautet, P. Introducing structural sensitivity into adsorption–energy scaling relations by means of coordination numbers. Nat Chem 2015, 7, 403–410. [Google Scholar] [CrossRef]
- Greeley, J.; Mavrikakis, M. Surface and Subsurface Hydrogen: Adsorption Properties on Transition Metals and Near-Surface Alloys. J. Phys. Chem. B 2005, 109, 3460–3471. [Google Scholar] [CrossRef] [PubMed]
- Shi, G.; Yano, H.; Tryk, D.A.; Nohara, S.; Uchida, H. High hydrogen evolution activity and suppressed H2O2 production on Pt-skin/PtFe alloy nanocatalysts for proton exchange membrane water electrolysis. Phys. Chem. Chem. Phys. 2019, 21, 2861–2865. [Google Scholar] [CrossRef]
- Tryk, D.A.; Shi, G.; Kakinuma, K.; Uchida, M.; Iiyama, A. Mechanisms for the Production and Suppression of Hydrogen Peroxide at the Hydrogen Electrode in Proton Exchange Membrane Fuel Cells and Water Electrolyzers: Theoretical Considerations. Catalysts 2024, 14, 890. [Google Scholar] [CrossRef]
- Maciá, M.D.; Campiña, J.M.; Herrero, E.; Feliu, J.M. On the kinetics of oxygen reduction on platinum stepped surfaces in acidic media. Journal of Electroanalytical Chemistry 2004, 564, 141–150. [Google Scholar] [CrossRef]
- Kuzume, A; Herrero, E; Feliu, J. M: Oxygen reduction on stepped platinum surfaces in acidic media. Journal of Electroanalytical Chemistry 2007, 599, 333–343. [Google Scholar] [CrossRef]
- Hitotsuyanagi, A.; Nakamura, M.; Hoshi, N. Structural effects on the activity for the oxygen reduction reaction on n(111)-(100) series of Pt: correlation with the oxide film formation. Electrochimica Acta 2012, 82, 512–516. [Google Scholar] [CrossRef]
- Hoshi, N.; Nakamura, M.; Hitotsuyanagi, A. Active sites for the oxygen reduction reaction on the high index planes of Pt. Electrochimica Acta 2013, 112, 899–904. [Google Scholar] [CrossRef]
- Greeley, J.; Rossmeisl, J.; Hellmann, A.; Norskov, J.K. Theoretical Trends in Particle Size Effects for the Oxygen Reduction Reaction. 2007, 221, 1209–1220. [Google Scholar] [CrossRef]
- Bandarenka, A.S.; Hansen, H.A.; Rossmeisl, J.; Stephens, I.E.L. Elucidating the activity of stepped Pt single crystals for oxygen reduction. Physical Chemistry Chemical Physics 2014, 16, 13625–13629. [Google Scholar] [CrossRef] [PubMed]
- Jinnouchi, R.; Kodama, K.; Nagoya, A.; Morimoto, Y. Simulated Volcano Plot of Oxygen Reduction Reaction on Stepped Pt Surfaces. Electrochimica Acta 2017, 230, 470–478. [Google Scholar] [CrossRef]
- Kodama, K.; Jinnouchi, R.; Takahashi, N.; Murata, H.; Morimoto, Y. Activities and Stabilities of Au-Modified Stepped-Pt Single-Crystal Electrodes as Model Cathode Catalysts in Polymer Electrolyte Fuel Cells. Journal of the American Chemical Society 2016, 138, 4194–4200. [Google Scholar] [CrossRef]
- Staszak-Jirkovský, J.; Subbaraman, R.; Strmcnik, D.; Harrison, K.L.; Diesendruck, C.E.; Assary, R.; Frank, O.; Kobr, L.; Wiberg, G.K.H.; Genorio, B.; et al. Water as a Promoter and Catalyst for Dioxygen Electrochemistry in Aqueous and Organic Media. ACS Catalysis 2015, 6600–6607. [Google Scholar] [CrossRef]
- Lu, F.; Zhang, Y.; Liu, S.; Lu, D.; Su, D.; Liu, M.; Zhang, Y.; Liu, P.; Wang, J.X.; Adzic, R.R.; et al. Surface Proton Transfer Promotes Four-Electron Oxygen Reduction on Gold Nanocrystal Surfaces in Alkaline Solution. Journal of the American Chemical Society 2017, 139, 7310–7317. [Google Scholar] [CrossRef]
- Rurigaki, T.; Hitotsuyanagi, A.; Nakamura, M.; Sakai, N.; Hoshi, N. Structural effects on the oxygen reduction reaction on the high index planes of Pt3Ni: n(111)-(111) and n(111)-(100) surfaces. Journal of Electroanalytical Chemistry 2014, 716, 58–62. [Google Scholar] [CrossRef]
- Stipe, B.C.; Rezaei, M.A.; Ho, W.; Gao, S.; Persson, M.; Lundqvist, B.I. Single-Molecule Dissociation by Tunneling Electrons. Physical Review Letters 1997, 78, 4410. [Google Scholar] [CrossRef]
- Gambardella, P.; Sljivancanin, Z.; Hammer, B.; Blanc, M.; Kuhnke, K.; Kern, K. Oxygen dissociation at Pt steps. Physical Review Letters 2001, 87, 4. [Google Scholar] [CrossRef]
- Gee, A.T.; Hayden, B.E. The dynamics of O[sub 2] adsorption on Pt(533): Step mediated molecular chemisorption and dissociation. J. Chem. Phys. 2000, 113, 10333–10343. [Google Scholar] [CrossRef]
- Gland, J.L.; Sexton, B.A.; Fisher, G.B. Oxygen interactions with the Pt(111) surface. Surface Science 1980, 95, 587–602. [Google Scholar] [CrossRef]
- Gland, J.L. Molecular and atomic adsorption of oxygen on the Pt(111) and Pt(S)-12(111) ラ (111) surfaces. Surface Science 1980, 93, 487–514. [Google Scholar] [CrossRef]
- Eichler, A.; Hafner, J. Molecular Precursors in the Dissociative Adsorption of O2 on Pt(111). Phys. Rev. Lett. 1997, 79, 4481–4484. [Google Scholar] [CrossRef]
- Ou, L.; Yang, F.; Liu, Y.; Chen, S. First-Principle Study of the Adsorption and Dissociation of O2 on Pt(111) in Acidic Media. J. Phys. Chem. C 2009, 113, 20657–20665. [Google Scholar] [CrossRef]
- Li, R.; Li, H.; Liu, J. First principles study of O2 dissociation on Pt(111) surface: Stepwise mechanism. International Journal of Quantum Chemistry 2016, 116, 908–914. [Google Scholar] [CrossRef]
- Ungerer, M.J.; Santos-Carballal, D.; Cadi-Essadek, A.; van Sittert, C.G.C.E.; de Leeuw, N.H. Interaction of H2O with the Platinum Pt (001), (011), and (111) Surfaces: A Density Functional Theory Study with Long-Range Dispersion Corrections. The Journal of Physical Chemistry C 2019, 123, 27465–27476. [Google Scholar] [CrossRef]
- Hammer, B. Special sites at noble and late transition metal catalysts. Topics in Catalysis 2006, 37, 3–16. [Google Scholar] [CrossRef]
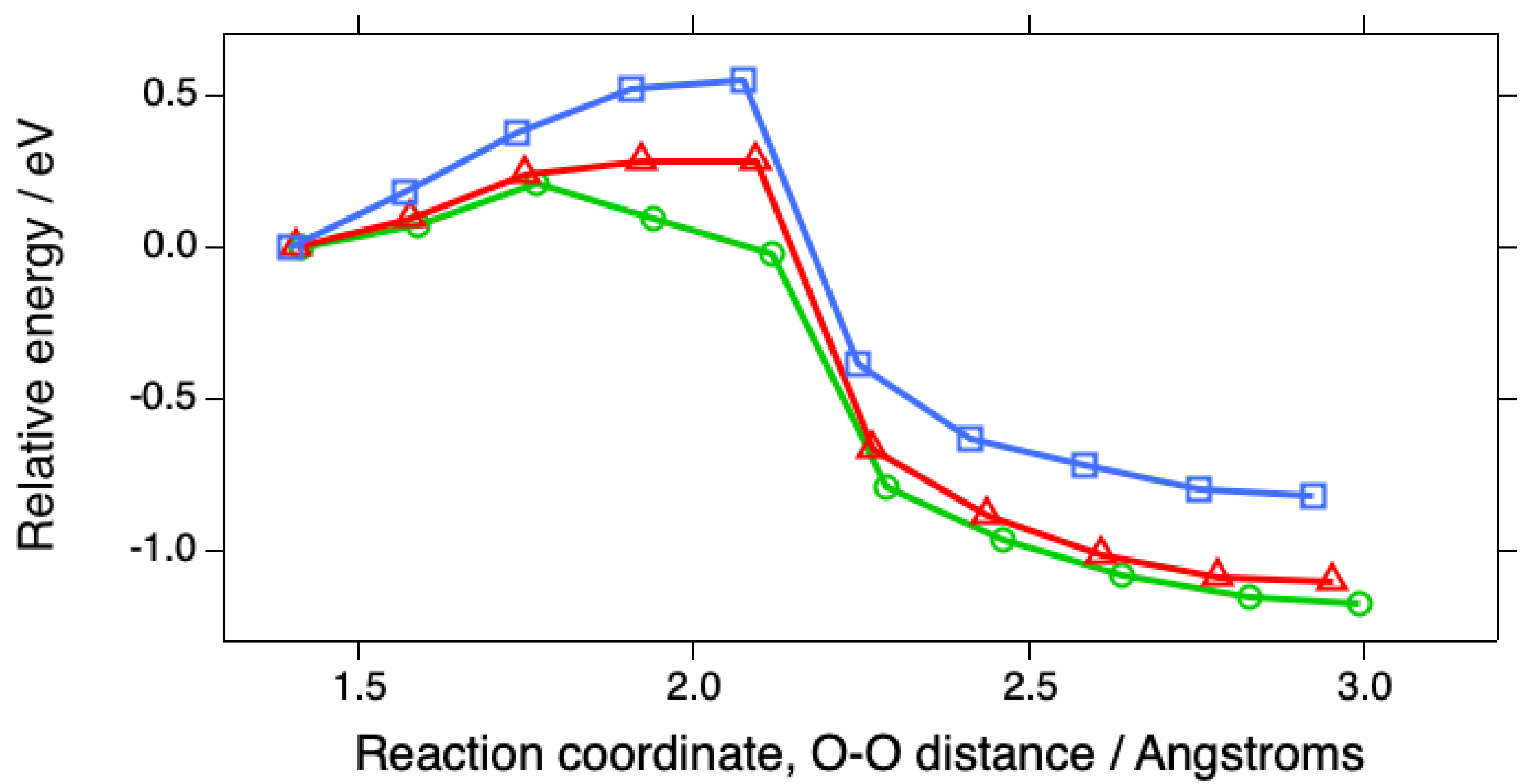
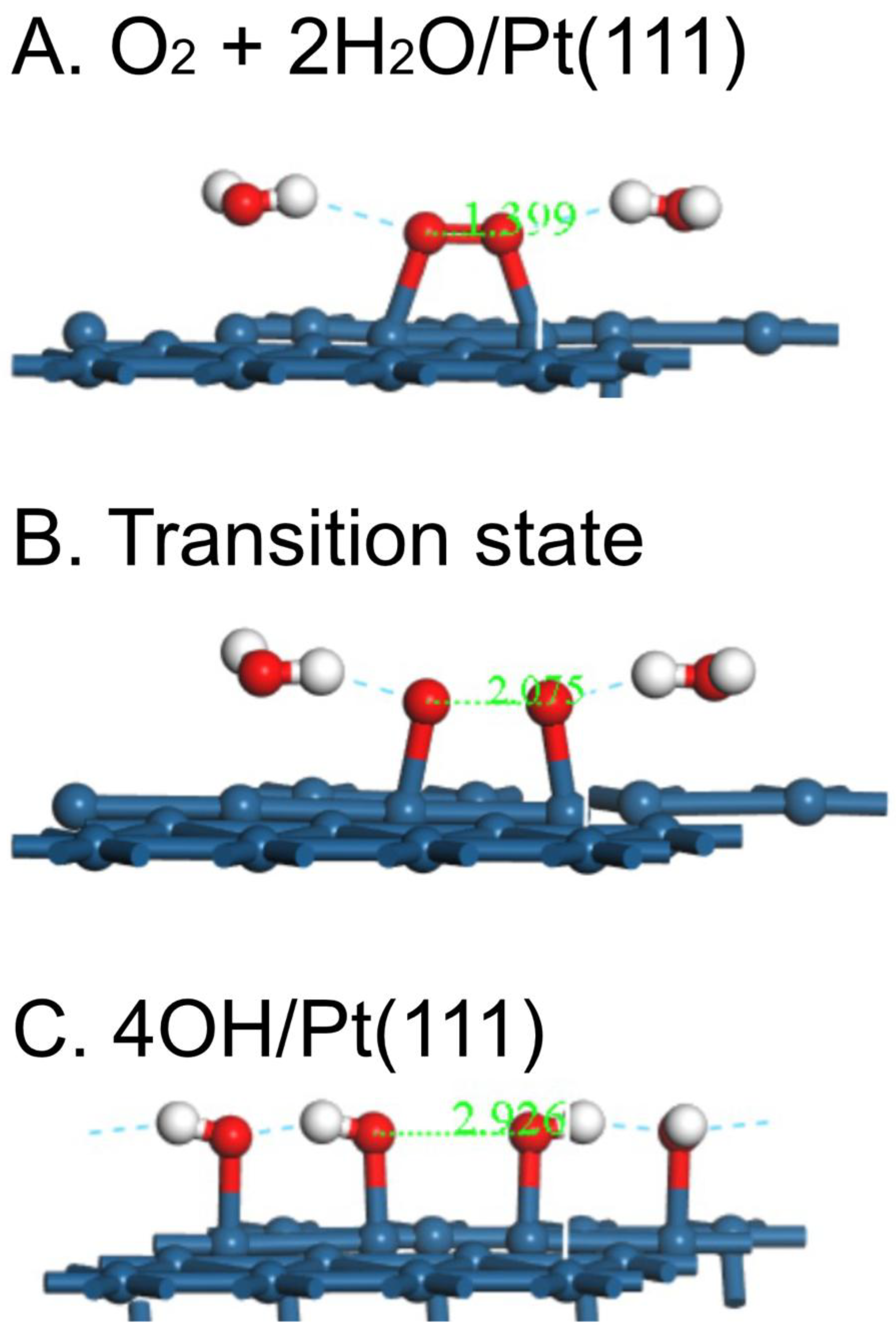
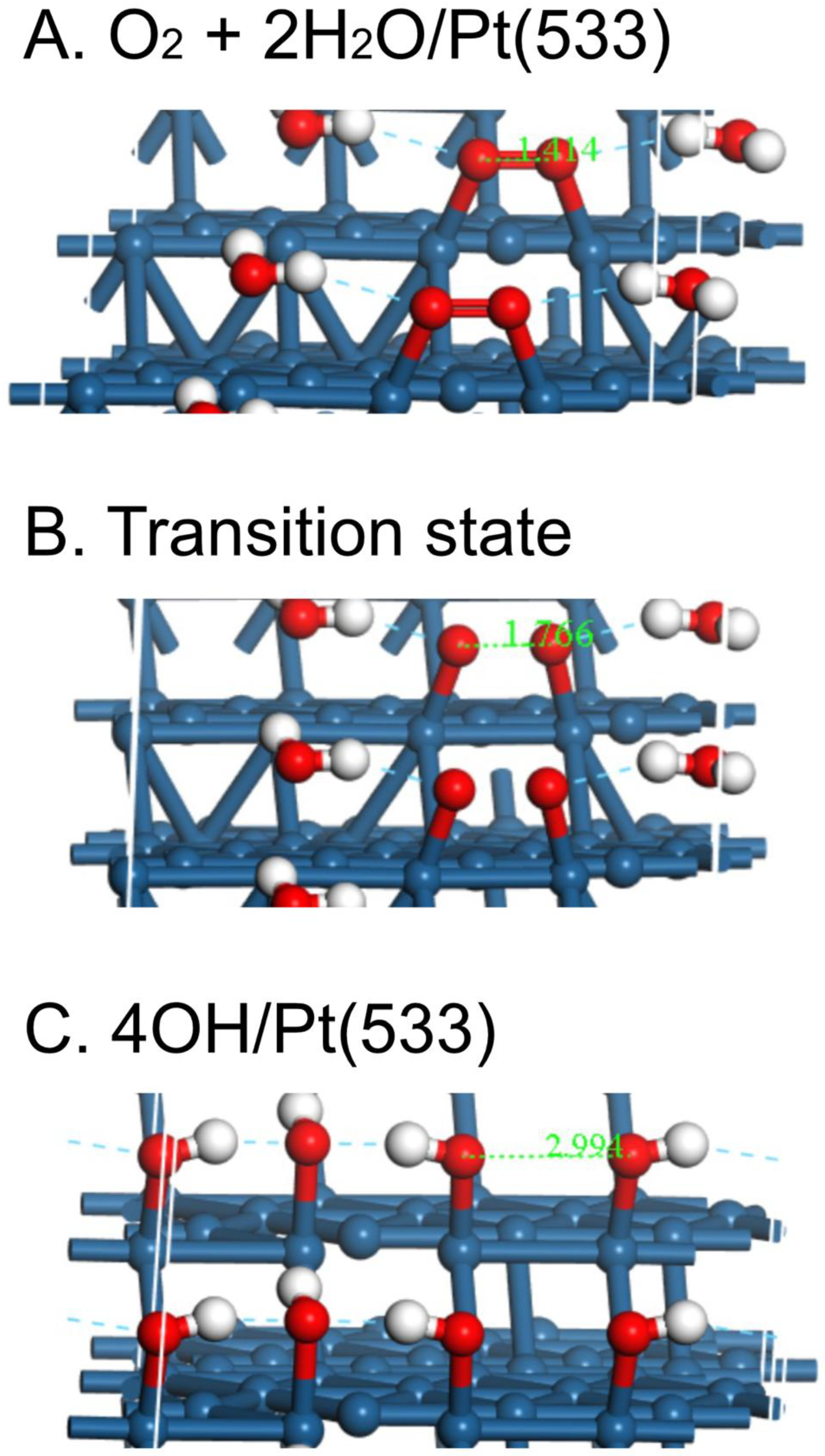
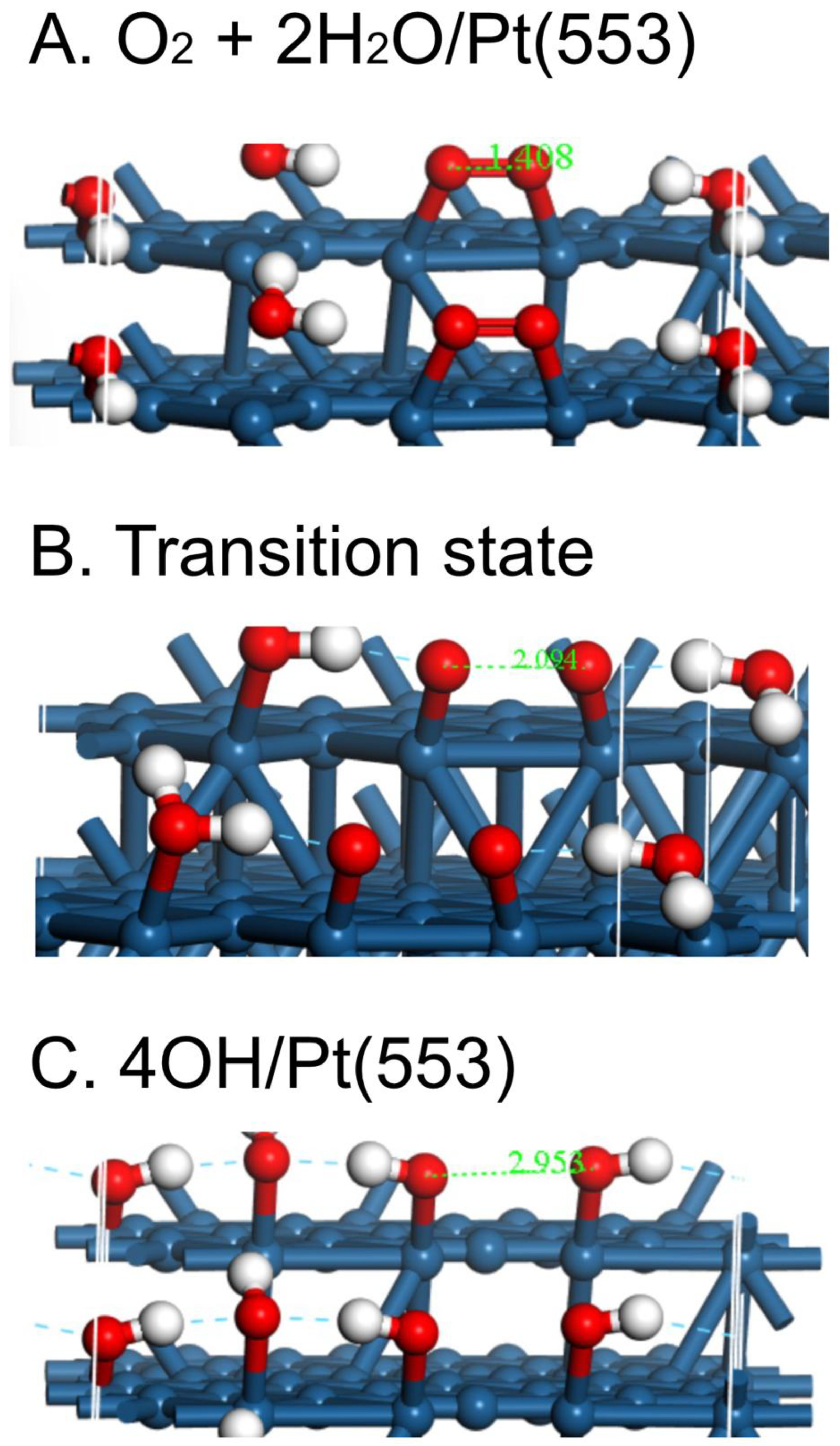
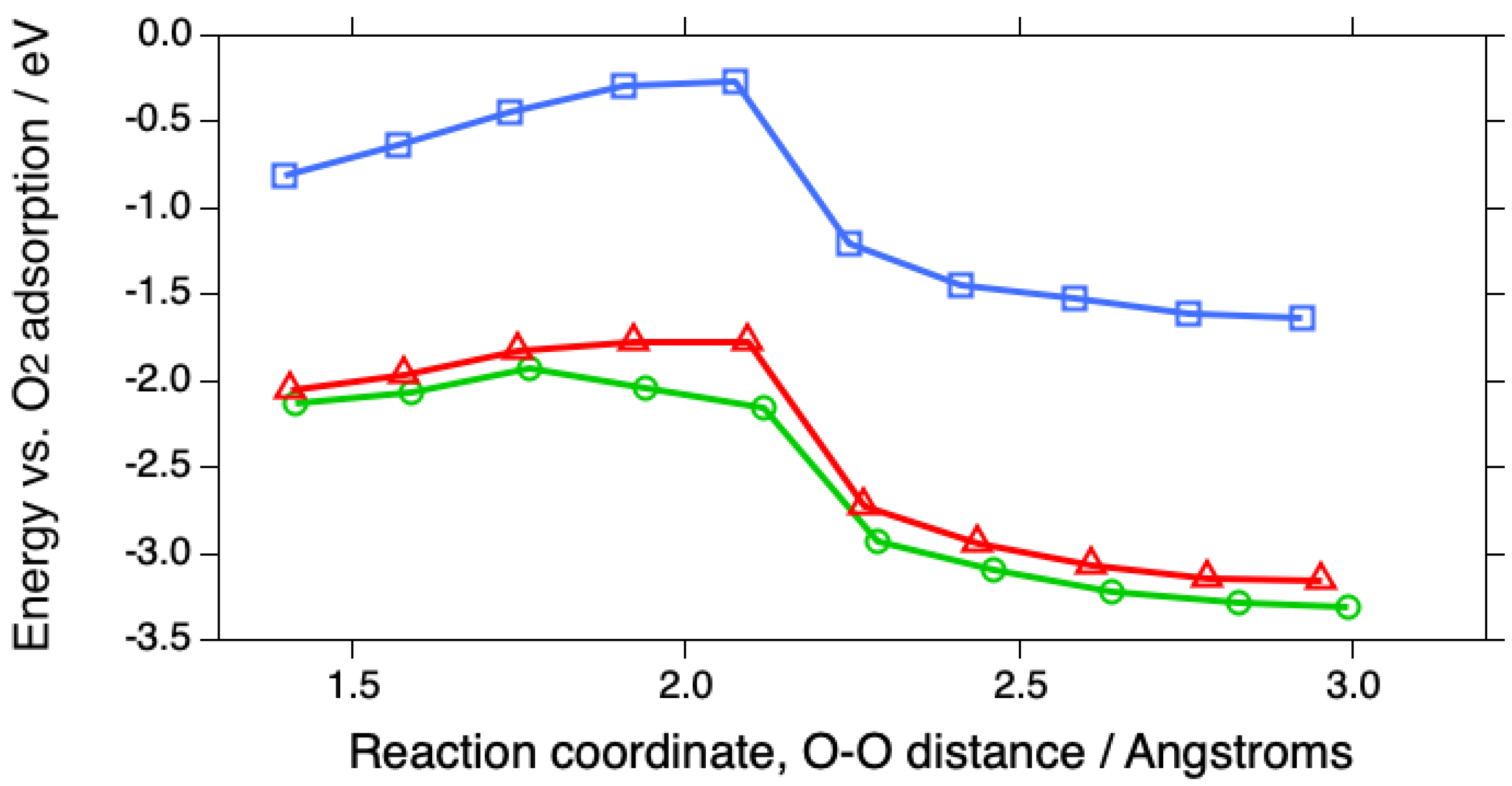
| Surface | O2 | O-O, Å | OH | H2O | Reference |
| Pt(111) exp. | -0.35 ~ -0.38 | [19,20] | |||
| Pt(111) calc. | -045 ~ -0.81 | [2,3,4,21,22,23] | |||
| Pt(111) calc. | -0.46 | [24] | |||
| Pt(111) calc. | -0.89 | 1.370 | -2.75 | -1.41 | pw |
| Pt(111)/H2O calc. | -0.81 | 1.399 | pw | ||
| Pt(533) calc. | -1.96 | 1.375 | -3.24 | -1.64 | pw |
| Pt(533)/H2O calc. | -2.05 | 1.414 | pw | ||
| Pt(553) calc. | -1.86 | 1.378 | -3.33 | -1.81 | pw |
| Pt(553)/H2O calc. | -2.13 | 1.408 | pw |
| Surface | Eact, eV | O-O, TS, Å | ΔE, eV |
| Pt(111) | 0.55 | 2.075 | -0.82 |
| Pt(533) | 0.21 | 1.766 | -1.17 |
| Pt(553) | 0.28 | 1.923 ~ 2.094 | -1.11 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




