Submitted:
01 January 2026
Posted:
05 January 2026
You are already at the latest version
Abstract
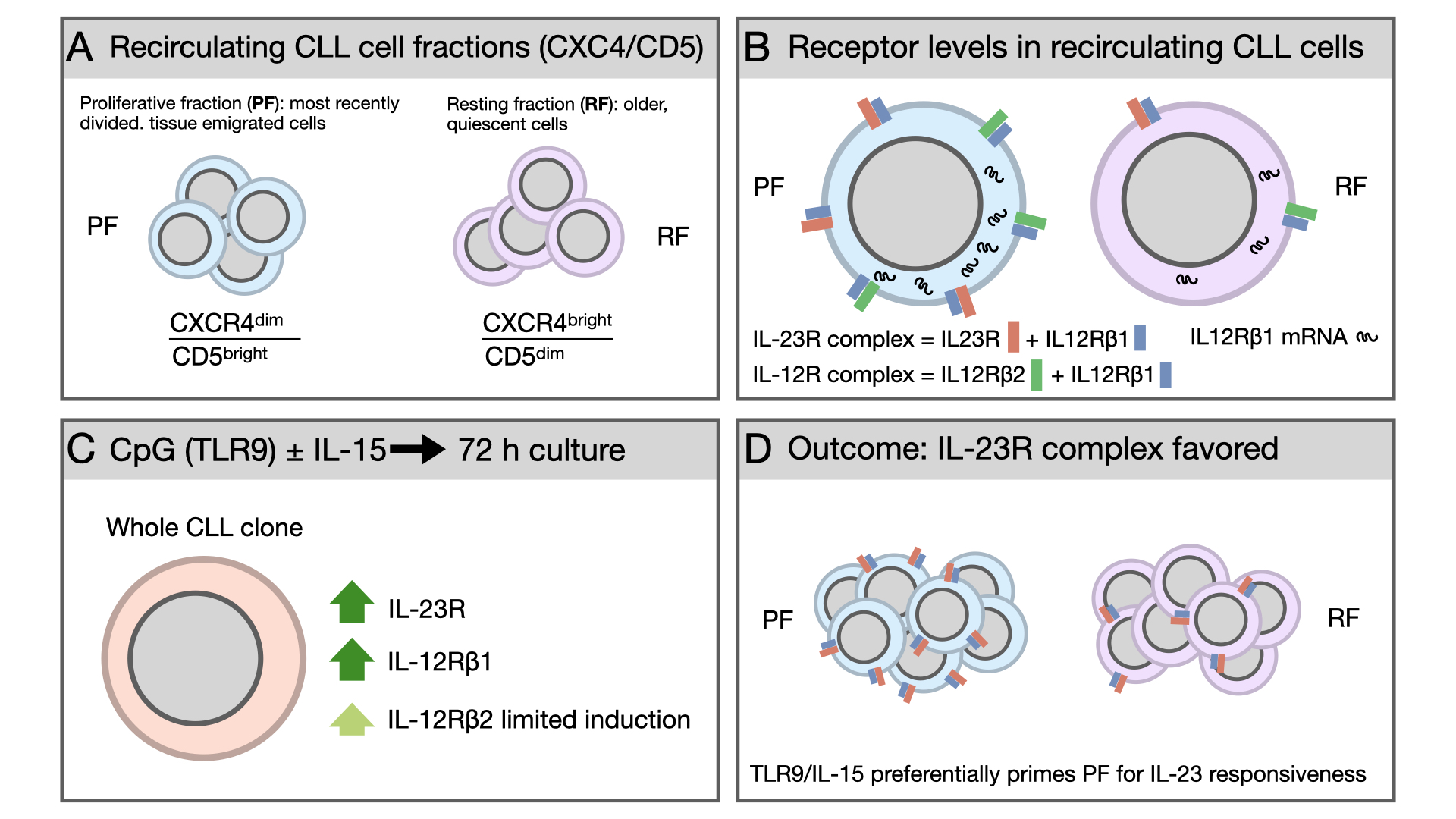
Chronic lymphocytic leukemia (CLL) is a dynamic malignancy in which intraclonal subfractions differ in activation history and responsiveness to microenvironmental signals. Here, we investigated the expression and inducibility of IL-12 family receptor subunits (IL-23R, IL-12Rβ1, IL-12Rβ2) and the related receptor complexes in recirculating CLL cells, with a focus on CXCR4/CD5-defined fractions: the proliferative fraction (PF; CXCR4^dim/CD5^bright; most recently divided, tissue-emigrated cells) and the resting fraction (RF; CXCR4^bright/CD5^dim; older, quiescent cells). At baseline, IL-12Rβ1 was enriched in the PF and was associated with a higher proportion of cells expressing IL-23R and IL-12R receptor complexes. Concomitantly, RT-qPCR disclosed higher IL-12Rβ1 mRNA levels. Following antigen-independent activation with CpG or CpG + IL-15, there was a marked increase of IL-23R and IL-12Rβ1 but not of IL-12Rβ2 surface expression, resulting in preferential upregulation of the IL-23R complex over the IL-12R complex. Fraction-specific analyses showed stronger induction of IL-23R and IL-23R complex expression in PF compared with RF. These findings identify an intraclonal bias toward IL-23 responsiveness in the CLL cells with a phenotype of recently divided, tissue-emigrated cells and suggest the IL-23/IL-23R axis as a potential therapeutic target.
Keywords:
1. Introduction
2. Results
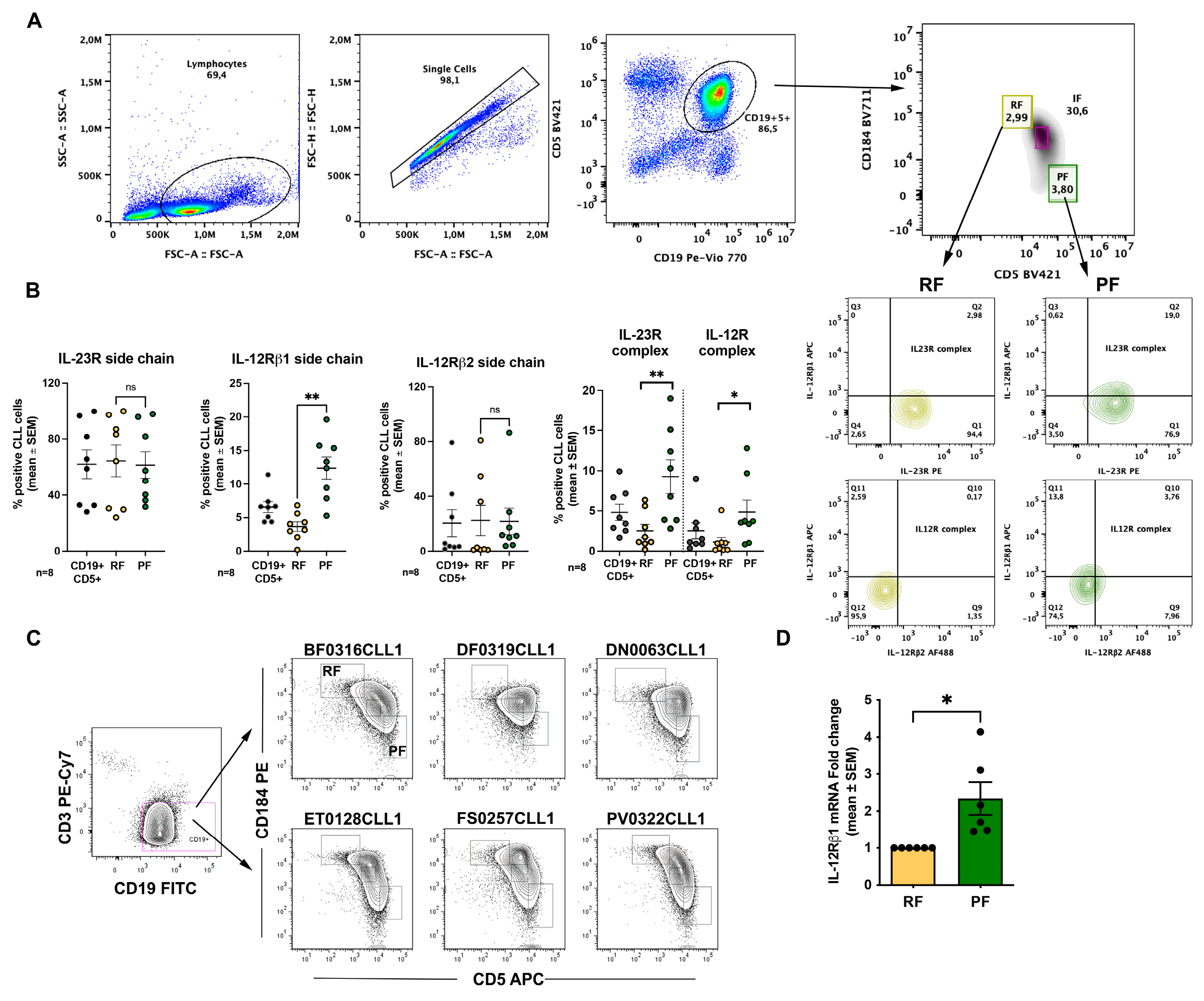
2.1. The Proliferative Fraction Shows Higher Expression of IL-23R and IL-12R Receptor Complexes in Recirculating CLL Cells
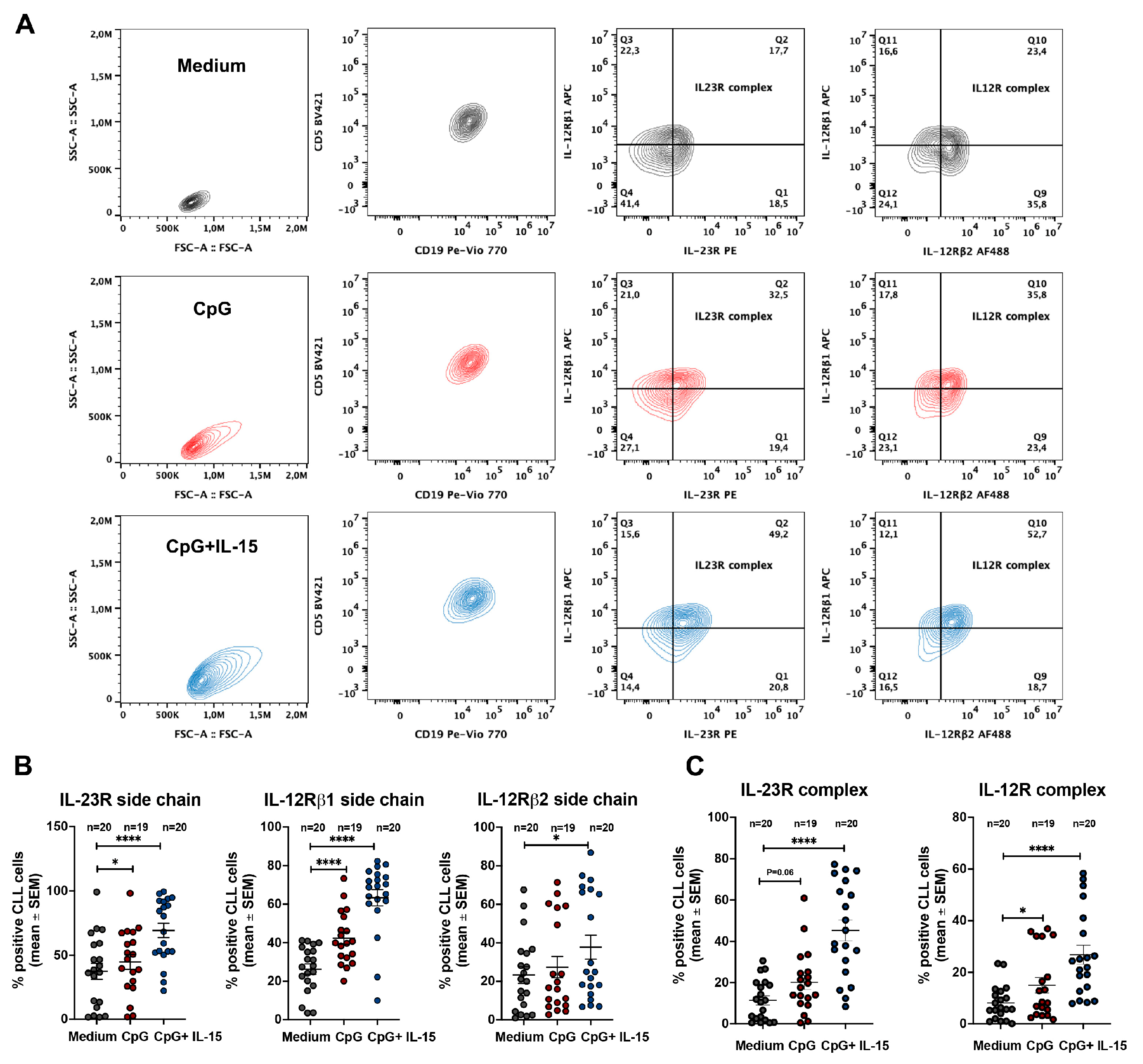
2.2. CpG + IL-15 Stimulation Induces IL-23R and IL-12R Receptor Complexes in CLL Cells
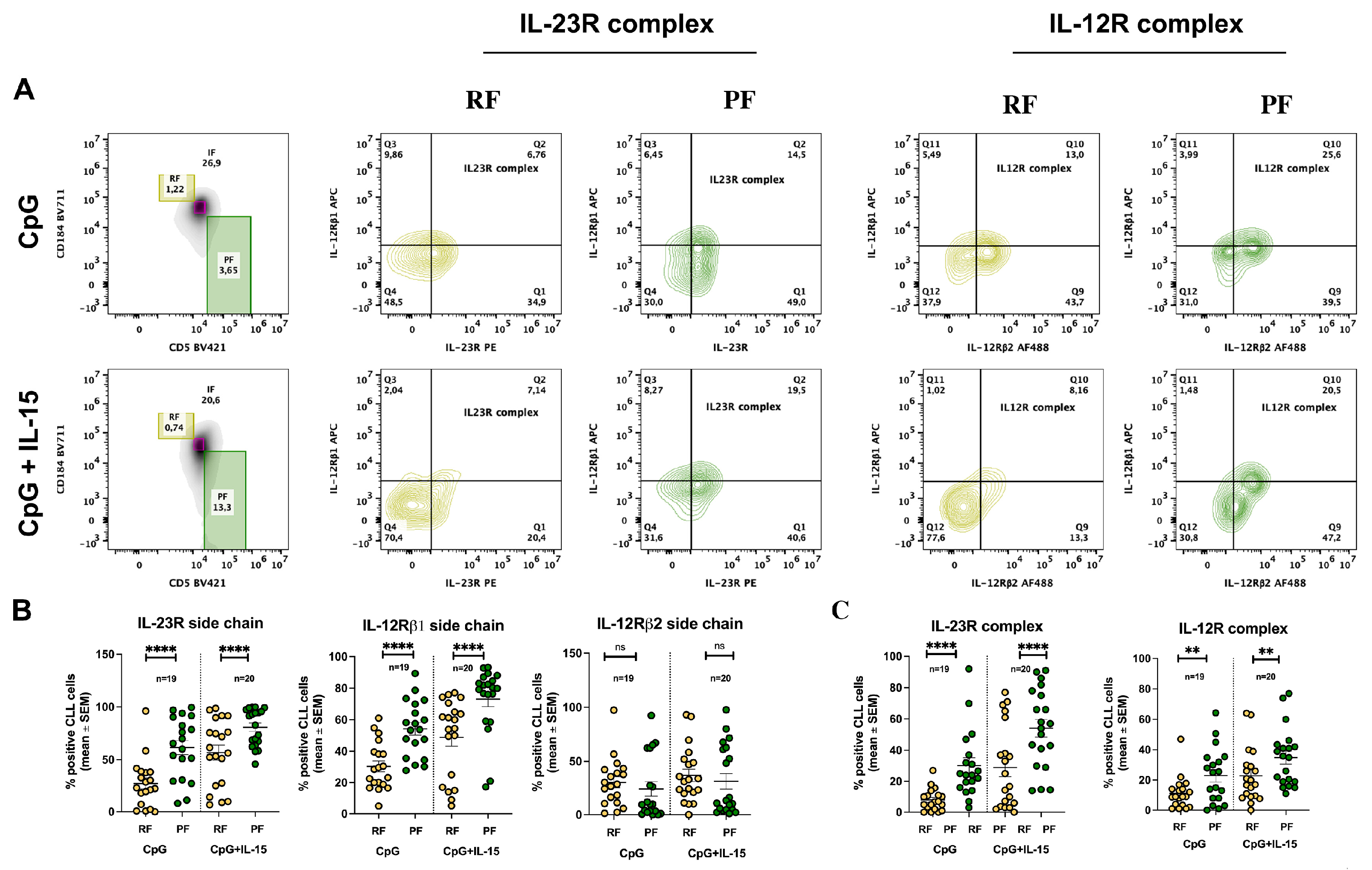
2.3. CpG + IL-15 Stimulation Differentially Upregulates IL-23R and IL-12R Receptor Subunits in Resting and Proliferative Fractions
3. Discussion
4. Materials and Methods
4.1. CLL Cell Samples
4.2. CLL Cell Isolation
4.3. CLL Cell Culture and Stimulation
4.4. Flow Cytometry: Cytokine Receptor Detection in Bulk CLL Cells
4.5. Flow Cytometry: Identification of RF/PF/IF and Receptor Detection in Intraclonal Fractions
4.6. RT-qPCR for Detection of IL-12Rβ1 RNA
4.7. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Hallek M, Cheson BD, Catovsky D, Caligaris-Cappio F, Dighiero G, Döhner H, Hillmen P, Keating M, Montserrat E, Chiorazzi N, Stilgenbauer S, Rai KR, Byrd JC, Eichhorst B, O’Brien S, Robak T, Seymour JF, Kipps TJ. iwCLL guidelines for diagnosis, indications for treatment, response assessment, and supportive management of CLL. Blood 2018, 131, 2745–2760. [CrossRef]
- Chiorazzi, N.; Ferrarini, M. B cell chronic lymphocytic leukemia: lessons learned from studies of the B cell antigen receptor. Annu. Rev. Immunol. 2003, 21, 841–894. [CrossRef]
- Chiorazzi, N.; Rai, K.R.; Ferrarini, M. Chronic lymphocytic leukemia. N. Engl. J. Med. 2005, 352, 804–815. [CrossRef]
- Bagnara, D.; Mazzarello, A.N.; Ghiotto, F.; Colombo, M.; Cutrona, G.; Fais, F.; Ferrarini, M. Old and New Facts and Speculations on the Role of the B Cell Receptor in the Origin of Chronic Lymphocytic Leukemia. Int. J. Mol. Sci. 2022, 23, 14249. [CrossRef]
- Messmer BT, Messmer D, Allen SL, Kolitz JE, Kudalkar P, Cesar D, Murphy EJ, Koduru P, Ferrarini M, Zupo S, Cutrona G, Damle RN, Wasil T, Rai KR, Hellerstein MK, Chiorazzi N. In vivo measurements document the dynamic cellular kinetics of chronic lymphocytic leukemia B cells. J. Clin. Invest. 2005, 115, 755–764. [CrossRef]
- Burger JA, Li KW, Keating MJ, Sivina M, Amer AM, Garg N, Ferrajoli A, Huang X, Kantarjian H, Wierda WG, O’Brien S, Hellerstein MK, Turner SM, Emson CL, Chen SS, Yan XJ, Wodarz D, Chiorazzi N. Leukemia cell proliferation and death in chronic lymphocytic leukemia patients on therapy with the BTK inhibitor ibrutinib. JCI Insight 2017, 2, e89904. [CrossRef]
- Burger, J.A.; Ghia, P.; Rosenwald, A.; Caligaris-Cappio, F. The microenvironment in mature B-cell malignancies: a target for new treatment strategies. Blood 2009, 114, 3367–3375. [CrossRef]
- Ghia, P.; Chiorazzi, N.; Stamatopoulos, K. Microenvironmental influences in chronic lymphocytic leukaemia: the role of antigen stimulation. J. Intern. Med. 2008, 264, 549–562. [CrossRef]
- Herishanu Y, Pérez-Galán P, Liu D, Biancotto A, Pittaluga S, Vire B, Gibellini F, Njuguna N, Lee E, Stennett L, Raghavachari N, Liu P, McCoy JP, Raffeld M, Stetler-Stevenson M, Yuan C, Sherry R, Arthur DC, Maric I, White T, Marti GE, Munson P, Wilson WH, Wiestner A. The lymph node microenvironment promotes B-cell receptor signaling, NF-κB activation, and tumor proliferation in chronic lymphocytic leukemia. Blood 2011, 117, 563–574. [CrossRef]
- Kipps TJ, Stevenson FK, Wu CJ, Croce CM, Packham G, Wierda WG, O’Brien S, Gribben J, Rai K. Chronic lymphocytic leukaemia. Nat. Rev. Dis. Primers 2017, 3, 16096. [CrossRef]
- Herndon TM, Chen SS, Saba NS, Valdez J, Emson C, Gatmaitan M, Tian X, Hughes TE, Sun C, Arthur DC, Stetler-Stevenson M, Yuan CM, Niemann CU, Marti GE, Aue G, Soto S, Farooqui MZH, Herman SEM, Chiorazzi N, Wiestner A. Direct in vivo evidence for increased proliferation of CLL cells in lymph nodes compared to bone marrow and peripheral blood. Leukemia 2017, 31, 1340–1347. [CrossRef]
- Calissano C, Damle RN, Hayes G, Murphy EJ, Hellerstein MK, Moreno C, Sison C, Kaufman MS, Kolitz JE, Allen SL, Rai KR, Chiorazzi N. In vivo intraclonal and interclonal kinetic heterogeneity in B-cell chronic lymphocytic leukemia. Blood 2011, 117, 3059–3066. [CrossRef]
- Calissano, C.; Damle, R.N.; Marsilio, S.; Yan, X.-J.; Yancopoulos, S.; Hayes, G.; Emson, C.; Murphy, E.J.; Hellerstein, M.K.; Sison, C.; Kaufman, M.S.; Kolitz, J.E.; Allen, S.L.; Rai, K.R.; Ivanovic, I.; Dozmorov, I.M.; Roa, S.; Scharff, M.D.; Li, W.; Chiorazzi, N. Intraclonal complexity in chronic lymphocytic leukemia: Fractions enriched in recently born/divided and older/quiescent cells. Mol. Med. 2011, 17, 1374–1382. [CrossRef]
- Trinchieri, G. Interleukin-12 and the regulation of innate resistance and adaptive immunity. Nat. Rev. Immunol. 2003, 3, 133–146. [CrossRef]
- 15 Oppmann B, Lesley R, Blom B, Timans JC, Xu Y, Hunte B, Vega F, Yu N, Wang J, Singh K, Zonin F, Vaisberg E, Churakova T, Liu M, Gorman D, Wagner J, Zurawski S, Liu Y, Abrams JS, Moore KW, Rennick D, de Waal-Malefyt R, Hannum C, Bazan JF, Kastelein RA. Novel p19 protein engages IL-12p40 to form a cytokine, IL-23, with biological activities similar as well as distinct from IL-12. Immunity 2000, 13, 715–725. [CrossRef]
- Parham C, Chirica M, Timans J, Vaisberg E, Travis M, Cheung J, Pflanz S, Zhang R, Singh KP, Vega F, To W, Wagner J, O’Farrell AM, McClanahan T, Zurawski S, Hannum C, Gorman D, Rennick DM, Kastelein RA, de Waal Malefyt R, Moore KW. A receptor for the heterodimeric cytokine IL-23 is composed of IL-12Rβ1 and a novel cytokine receptor subunit, IL-23R. J. Immunol. 2002, 168, 5699–5708. [CrossRef]
- Szabo, S.J.; Sullivan, B.M.; Peng, S.L.; Glimcher, L.H. Molecular mechanisms regulating Th1 immune responses. Annu. Rev. Immunol. 2003, 21, 713–758. [CrossRef]
- Langrish CL, Chen Y, Blumenschein WM, Mattson J, Basham B, Sedgwick JD, McClanahan T, Kastelein RA, Cua DJ. IL-23 drives a pathogenic T cell population that induces autoimmune inflammation. J. Exp. Med. 2005, 201, 233–240. [CrossRef]
- Mezghiche, H.; Yahia-Cherbal, H.; Rogge, L.; Bianchi, D. Interleukin-23 receptor: Expression and regulation in immune cells. Eur. J. Immunol. 2024, 54, e2250348. [CrossRef]
- Cocco C, Canale S, Frasson C, Di Carlo E, Ognio E, Ribatti D, Prigione I, Basso G, Airoldi I. Interleukin-23 acts as antitumor agent on childhood B-acute lymphoblastic leukemia cells. Blood 2010, 116, 3887–3898. [CrossRef]
- Pistoia V, Cocco C, Airoldi I. Interleukin-12 receptor beta2: from cytokine receptor to gatekeeper gene in human B-cell malignancies. J Clin Oncol. 2009 Oct 1;27(28):4809-16. [CrossRef]
- Giuliani N, Airoldi I. Novel insights into the role of interleukin-27 and interleukin-23 in human malignant and normal plasma cells. Clin Cancer Res. 2011 Nov 15;17(22):6963-70. [CrossRef]
- Cutrona G, Tripodo C, Matis S, Recchia AG, Massucco C, Fabbi M, Colombo M, Emionite L, Sangaletti S, Gulino A, Reverberi D, Massara R, Boccardo S, de Totero D, Salvi S, Cilli M, Pellicanò M, Manzoni M, Fabris S, Airoldi I, Valdora F, Ferrini S, Gentile M, Vigna E, Bossio S, De Stefano L, Palummo A, Iaquinta G, Cardillo M, Zupo S, Cerruti G, Ibatici A, Neri A, Fais F, Ferrarini M, Morabito F. Microenvironmental regulation of the IL-23R/IL-23 axis overrides chronic lymphocytic leukemia indolence. Sci Transl Med. 2018 Feb 14;10(428):eaal1571. [CrossRef]
- Airoldi I, Di Carlo E, Cocco C, Sorrentino C, Fais F, Cilli M, D’Antuono T, Colombo MP, Pistoia V. Lack of Il12rb2 signaling predisposes to spontaneous autoimmunity and malignancy. Blood. 2005 Dec 1;106(12):3846-53. [CrossRef]
- Mongini PK, Gupta R, Boyle E, Nieto J, Lee H, Stein J, Bandovic J, Stankovic T, Barrientos J, Kolitz JE, Allen SL, Rai K, Chu CC, Chiorazzi N. Toll-like receptor 9 and interleukin-15 promote CLL B-cell survival and proliferation: implications for disease progression. J. Immunol. 2015, 194, 653–664. [CrossRef]
- Matis S, Grazia Recchia A, Colombo M, Cardillo M, Fabbi M, Todoerti K, Bossio S, Fabris S, Cancila V, Massara R, Reverberi D, Emionite L, Cilli M, Cerruti G, Salvi S, Bet P, Pigozzi S, Fiocca R, Ibatici A, Angelucci E, Gentile M, Monti P, Menichini P, Fronza G, Torricelli F, Ciarrocchi A, Neri A, Fais F, Tripodo C, Morabito F, Ferrarini M, Cutrona G. MiR-146b-5p regulates IL-23 receptor complex expression in chronic lymphocytic leukemia cells. Blood Adv. 2022 Oct 25;6(20):5593-5612. [CrossRef]
- Molica, S.; Giannarelli, D.; Mirabelli, R.; Levato, L.; Gentile, M.; Lentini, M.; Morabito, F. Changes in the incidence, pattern of presentation and clinical outcome of early chronic lymphocytic leukemia patients using the 2008 International Workshop on CLL guidelines. Expert Rev. Hematol. 2014, 7, 691–695. [CrossRef]
- Monti, P.; Menichini, P.; Speciale, A.; Cutrona, G.; Fais, F.; Taiana, E.; Neri, A.; Bomben, R.; Gentile, M.; Gattei, V.; Ferrarini, M.; Morabito, F.; Fronza, G. Heterogeneity of TP53 Mutations and P53 Protein Residual Function in Cancer: Does It Matter? Front. Oncol. 2020, 10, 593383. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



