Submitted:
30 December 2025
Posted:
01 January 2026
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
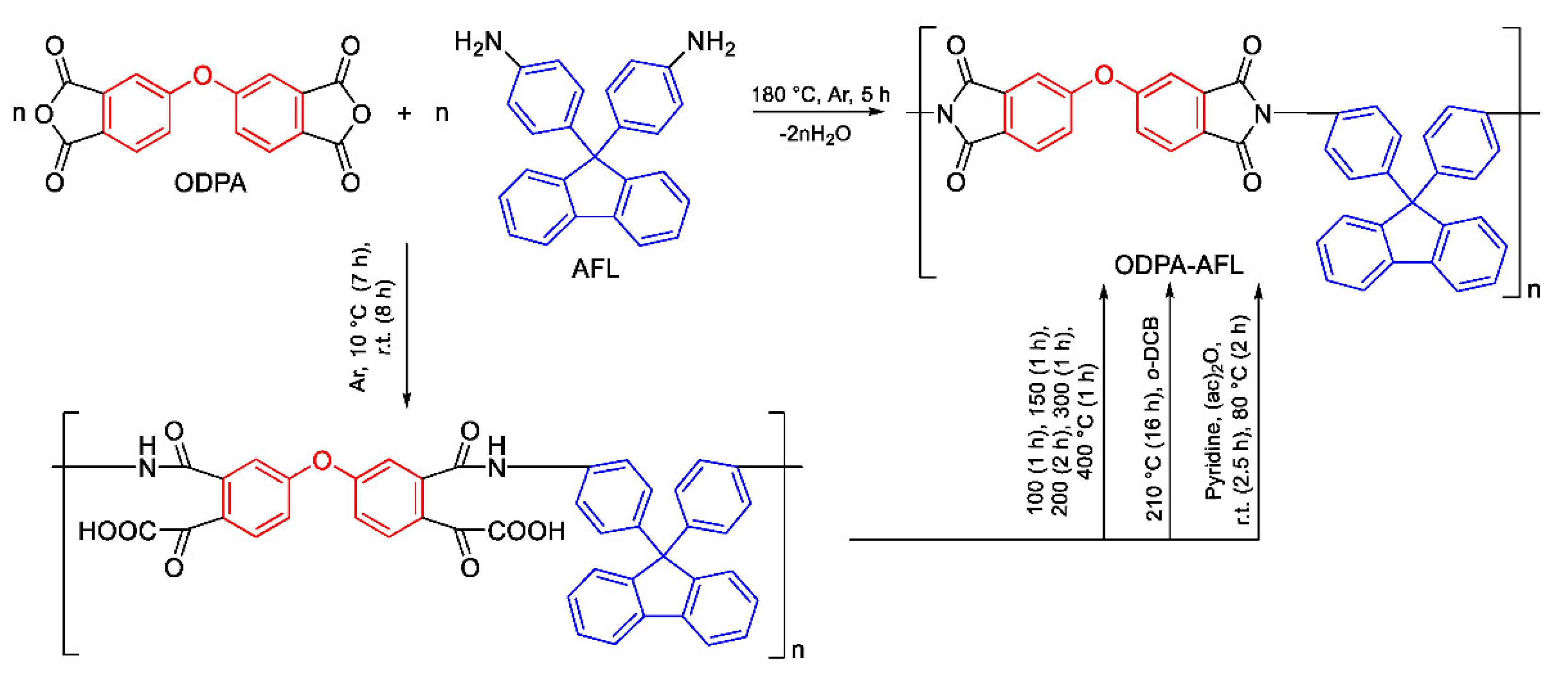
2.1. Polyimide Synthesis
2.1.1. Comparison of Two- and One-Step Methods
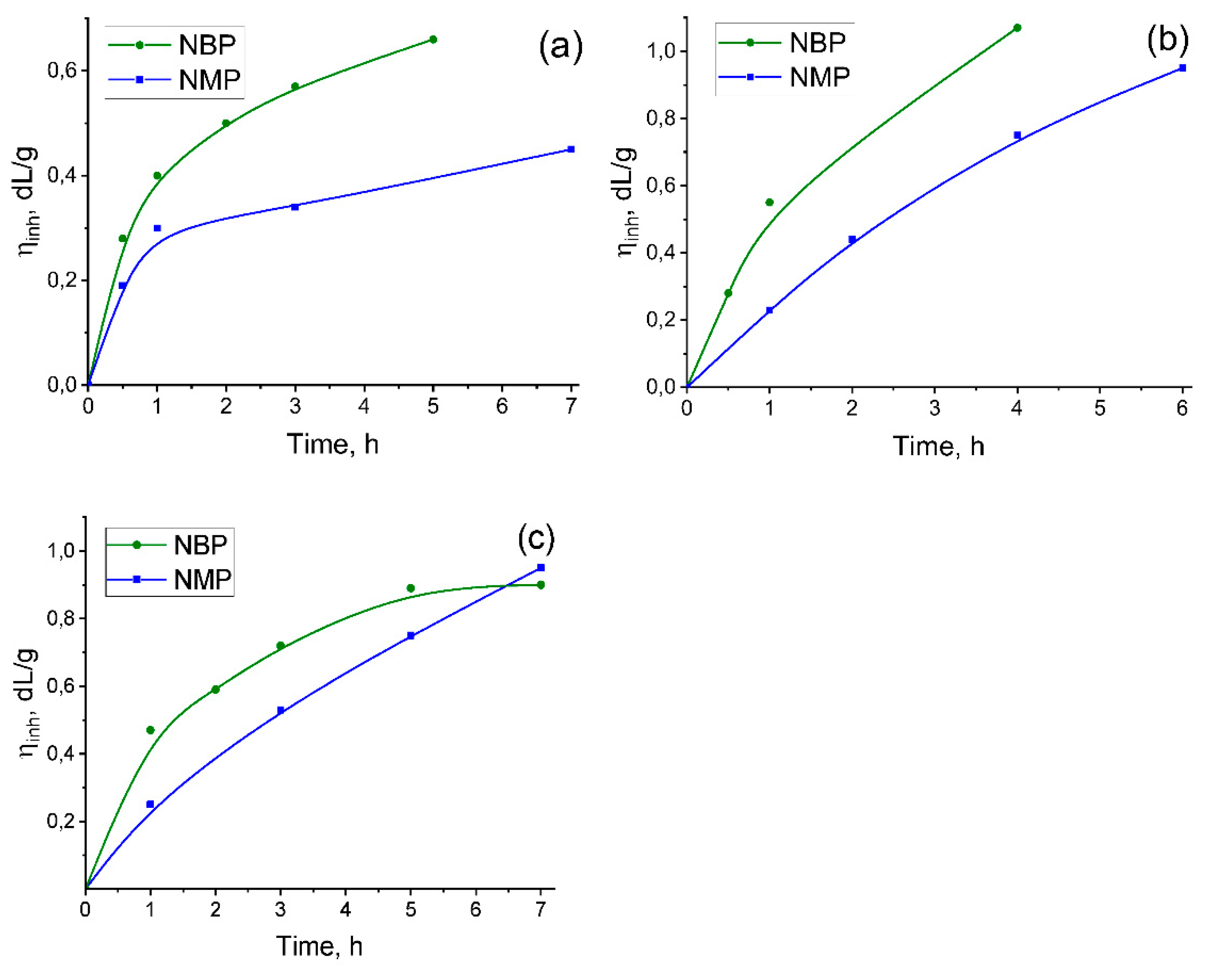
2.1.2. Comparison of Pi Synthesis in Nmp and Nbp
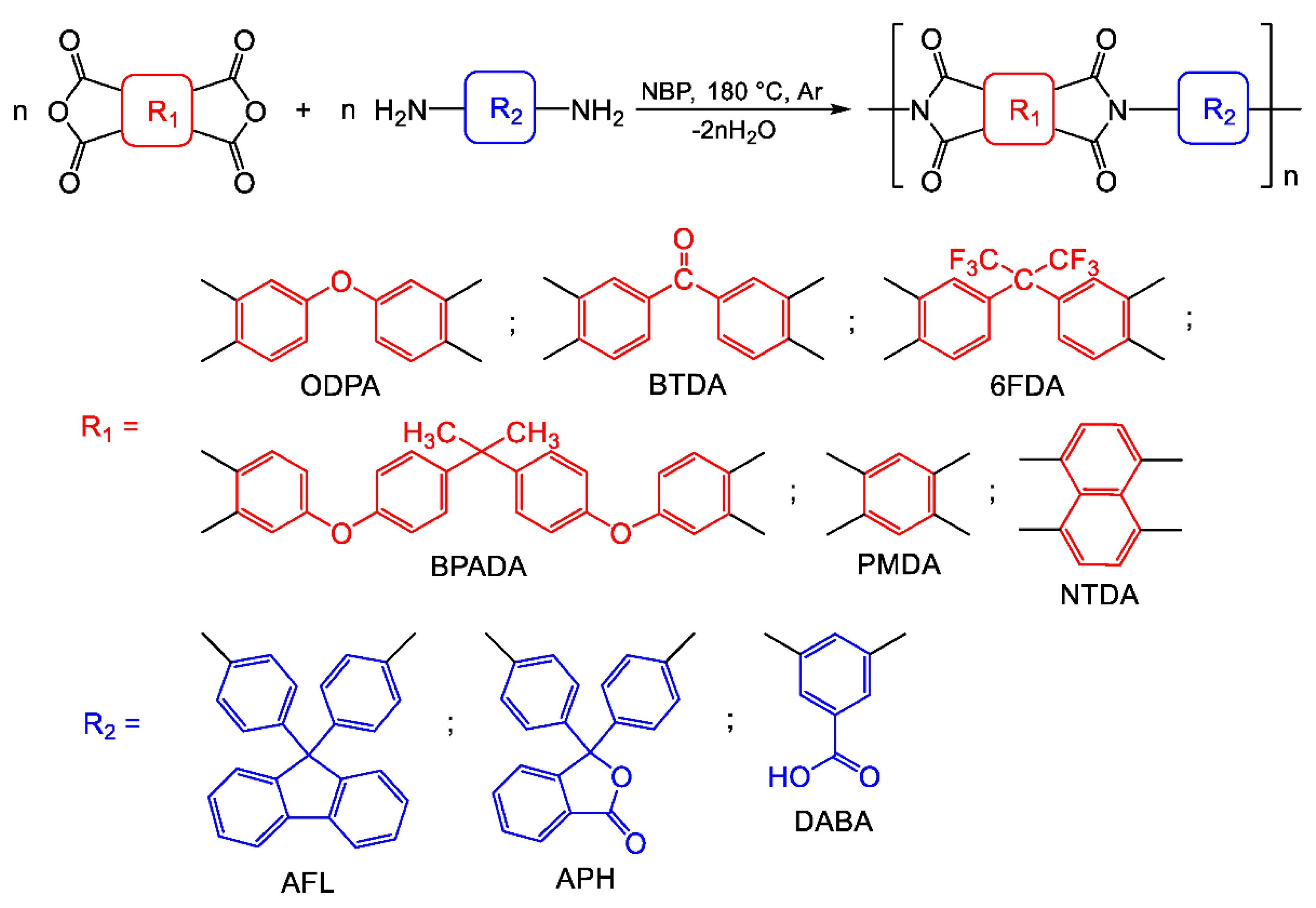
2.1.3. Versatility of Nbp for the Synthesis of Different Pis
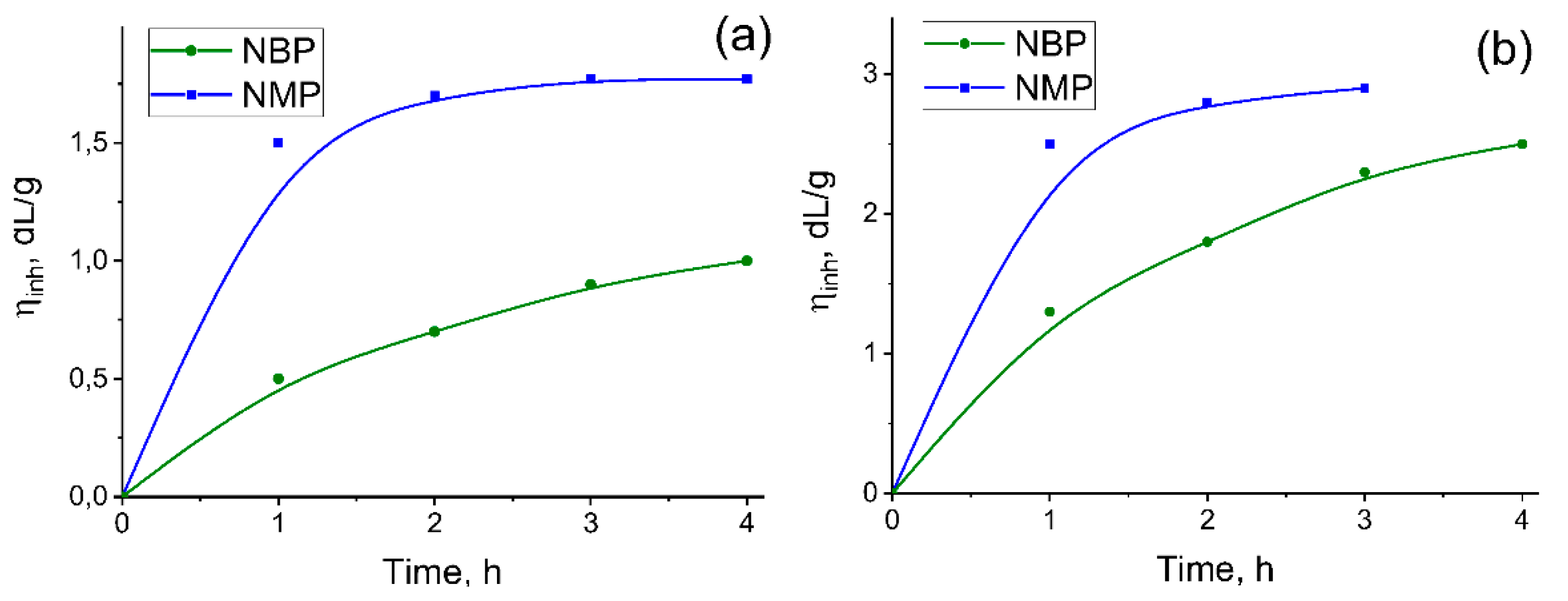
2.2. Polyamide Synthesis
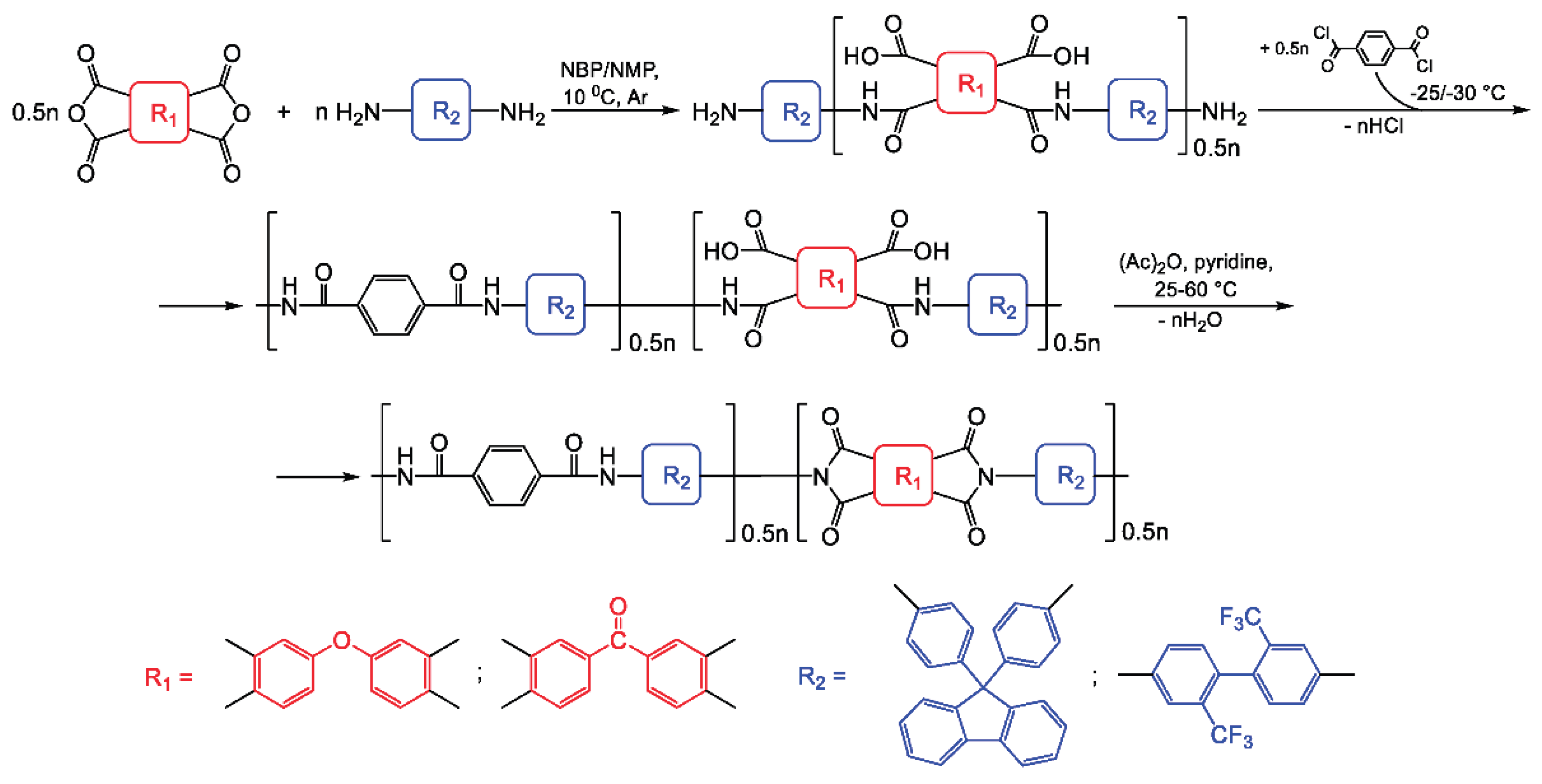
2.3. Poly(amide-Imide) Synthesis
3. Materials and Methods
3.1. Materials
3.2. Polyimide Synthesis
3.2.1. One-Step Method
3.2.2. Two-Step Methods
Synthesis of PAA
Chemical imidization
Thermal imidization
Thermal imidization with an azeotropic mixture
3.3. Polyamide Synthesis
3.4. Poly(amide-Imide) Synthesis
3.5. Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gouzman, I.; Grossman, E.; Verker, R.; Atar, N.; Bolker, A.; Eliaz, N. Advances in Polyimide-Based Materials for Space Applications. Adv Mater 2019, 31, e1807738. [Google Scholar] [CrossRef]
- Liaw, D.-J.; Wang, K.-L.; Huang, Y.-C.; Lee, K.-R.; Lai, J.-Y.; Ha, C.-S. Advanced polyimide materials: Syntheses, physical properties and applications. Progress in Polymer Science 2012, 37, 907–974. [Google Scholar] [CrossRef]
- Sapozhnikov, D.A.; Baiminov, B.A.; Vygodskii, Y.S. Highly Heat-Resistant Polymeric Coatings of Optical Fibers. Polymer Science, Series C 2020, 62, 165–171. [Google Scholar] [CrossRef]
- Wu, Z.; He, J.; Yang, H.; Yang, S. Progress in Aromatic Polyimide Films for Electronic Applications. Polymers (Basel) 2022, 14. [Google Scholar] [CrossRef]
- Zabegaeva, O.N.; Kosolapov, A.F.; Semjonov, S.L.; Ezernitskaya, M.G.; Afanasyev, E.S.; Godovikov, I.A.; Chuchalov, A.V.; Sapozhnikov, D.A. Polyamide-imides as novel high performance primary protective coatings of silica optical fibers: Influence of the structure and molecular weight. Reactive and Functional Polymers 2024, 194. [Google Scholar] [CrossRef]
- Zhang, T.; Chai, Y.; Wang, S.; Yu, J.; Jiang, S.; Zhu, W.; Fang, Z.; Li, B. Recent Study Advances in Flexible Sensors Based on Polyimides. Sensors (Basel) 2023, 23. [Google Scholar] [CrossRef] [PubMed]
- Bera, D.; Chatterjee, R.; Banerjee, S. Aromatic polyamide nonporous membranes for gas separation application. e-Polymers 2021, 21, 108–130. [Google Scholar] [CrossRef]
- Kononova, S.V.; Gubanova, G.N.; Lebedeva, G.K.; Kruchinina, E.V.; Vlasova, E.N.; Popova, E.N.; Zakharova, N.V.; Vylegzhanina, M.E.; Novozhilova, E.A.; Danilova, K.V. Realization of Intermolecular Interactions as a Basis for Controlling Pervaporation Properties of Membranes Made of Aromatic Polyamide-Imides. Membranes (Basel) 2025, 15. [Google Scholar] [CrossRef]
- Pulyalina, A.; Rostovtseva, V.; Faykov, I.; Tataurov, M.; Dubovenko, R.; Shugurov, S. Development of Novel Polyamide-Imide/DES Composites and Their Application for Pervaporation and Gas Separation. Molecules 2021, 26. [Google Scholar] [CrossRef] [PubMed]
- Sapozhnikov, D.A.; Chuchalov, A.V.; Vygodskii, Y.S. Carboxylated polyimides for advanced functional materials: synthesis, modification, and various applications. European Polymer Journal 2025, 240. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, C.; Ling, Q.; Sang, L. Advances in pervaporation desalination based on polymer membranes. RSC Adv 2025, 15, 20985–21005. [Google Scholar] [CrossRef]
- Xu, Z.; Croft, Z.L.; Guo, D.; Cao, K.; Liu, G. Recent development of polyimides: Synthesis, processing, and application in gas separation. Journal of Polymer Science 2021, 59, 943–962. [Google Scholar] [CrossRef]
- Sapozhnikov, D.A.; Vygodskii, Y.S. Achievements in polycondensation and condensation polymers. Polymer Science Series B 2015, 57, 275–291. [Google Scholar] [CrossRef]
- Jalali, A. Synthesis and Characterization of Novel Polyamide-imides Containing Thioether Linkages, Thiazole, and Nitrophenyl Units with High Solubility and Processability for Optical Film Applications. Journal of Applied Material Science 2025, 1. [Google Scholar] [CrossRef]
- Yi, C.; Li, W.; Shi, S.; He, K.; Ma, P.; Chen, M.; Yang, C. High-temperature-resistant and colorless polyimide: Preparations, properties, and applications. Solar Energy 2020, 195, 340–354. [Google Scholar] [CrossRef]
- Chin, H.-S.; Wong, H.Q.; Yen, H.-J.; Kung, Y.-R. Synthesis and Optical Properties of Ion-Selective Polyamides Bearing a Phenanthrene Chromophore. ACS Applied Optical Materials 2025, 3, 383–391. [Google Scholar] [CrossRef]
- Liu, Y.-Y.; Cao, J.-H.; Wang, Y.; Shen, S.-G.; Liang, W.-H.; Wu, D.-Y. Colorless Polyamide–Imide Films with Enhanced Thermal and Dimensional Stability and Their Application in Flexible OLED Devices. ACS Applied Polymer Materials 2022, 4, 7664–7673. [Google Scholar] [CrossRef]
- Tapaswi, P.K.; Ha, C.S. Recent Trends on Transparent Colorless Polyimides with Balanced Thermal and Optical Properties: Design and Synthesis. Macromolecular Chemistry and Physics 2019, 220. [Google Scholar] [CrossRef]
- Xia, X.; He, X.; Zhang, S.; Zheng, F.; Lu, Q. Short-side-chain regulation of colorless and transparent polyamide-imides for flexible transparent displays. European Polymer Journal 2023, 191. [Google Scholar] [CrossRef]
- Zhuang, Y.; Seong, J.G.; Lee, Y.M. Polyimides containing aliphatic/alicyclic segments in the main chains. Progress in Polymer Science 2019, 92, 35–88. [Google Scholar] [CrossRef]
- Kothavade, P.; Kafi, A.; Shanmuganathan, K.; Bateman, S. High performance polyimides for additive manufacturing: A critical review. Progress in Polymer Science 2026, 172. [Google Scholar] [CrossRef]
- Mrinalini, M.; Prasanthkumar, S. Recent Advances on Stimuli-Responsive Smart Materials and their Applications. Chempluschem 2019, 84, 1103–1121. [Google Scholar] [CrossRef]
- Zhai, M.; Lv, X.; Zhang, C.; Fu, X.; Zhang, Q.; Huang, R.; Cai, W.; Niu, H.; Wang, W. Multi-Stimuli-Responsive Polyamides Containing Benzophenothiazine Being Sensitive to TNP, Acid, Voltage, and Light. ACS Applied Polymer Materials 2023, 5, 9466–9476. [Google Scholar] [CrossRef]
- Bejan, A.-E.; Constantin, C.-P.; Damaceanu, M.-D. ProDOT-containing polyamides: On the road from energy storage materials to perspective electrochromic capacitive windows. Journal of Power Sources 2024, 613. [Google Scholar] [CrossRef]
- Nimkar, A.; Bergman, G.; Ballas, E.; Tubul, N.; Levi, N.; Malchik, F.; Kukurayeve, I.; Chae, M.S.; Sharon, D.; Levi, M.; et al. Polyimide Compounds For Post-Lithium Energy Storage Applications. Angewandte Chemie 2023, 135. [Google Scholar] [CrossRef]
- Zhang, Y.; Wu, X.; Xia, L.; Liu, J.; Yao, L.; Xie, H.; Luo, J.; Pan, Z. Molecular engineering of polyamide-imide copolymers for high-temperature energy storage. Journal of Materials Chemistry A 2025. [Google Scholar] [CrossRef]
- CandidateList of Substances of Very High Concern for Authorisation. Available online: https://echa.europa.eu/candidate-list-table (accessed on.
- San José, E.; de la Viuda, M.R.; Carmona, F.J.; Soto, C.; Palacio, L.; Prádanos, P.; Hernández, A.; Tena, A. Green dipolar aprotic solvents for the dynamic polycondensation of high-performance polyimide membranes. Green Chemistry 2024, 26, 11984–12007. [Google Scholar] [CrossRef]
- Sheldon, R.A. The greening of solvents: Towards sustainable organic synthesis. Current Opinion in Green and Sustainable Chemistry 2019, 18, 13–19. [Google Scholar] [CrossRef]
- Bisz, E.; Koston, M.; Szostak, M. N-Butylpyrrolidone (NBP) as a non-toxic substitute for NMP in iron-catalyzed C(sp2)–C(sp3) cross-coupling of aryl chlorides. Green Chemistry 2021, 23, 7515–7521. [Google Scholar] [CrossRef]
- Jiang, X.; Yong, W.F.; Gao, J.; Shao, D.-D.; Sun, S.-P. Understanding the role of substrates on thin film composite membranes: A green solvent approach with TamiSolve® NxG. Journal of Membrane Science 2021, 635. [Google Scholar] [CrossRef]
- Sherwood, J.; Parker, H.L.; Moonen, K.; Farmer, T.J.; Hunt, A.J. N-Butylpyrrolidinone as a dipolar aprotic solvent for organic synthesis. Green Chemistry 2016, 18, 3990–3996. [Google Scholar] [CrossRef]
- Voon, B.K.; Yap, Y.J.; Yong, W.F. Green solvents in membrane separation: progress, challenges, and future perspectives for sustainable industrial applications. Green Chemistry 2025, 27, 11705–11738. [Google Scholar] [CrossRef]
- Ivanković, A. Review of 12 Principles of Green Chemistry in Practice. International Journal of Sustainable and Green Energy 2017, 6. [Google Scholar] [CrossRef]
- Marino, T.; Russo, F.; Criscuoli, A.; Figoli, A. TamiSolve® NxG as novel solvent for polymeric membrane preparation. Journal of Membrane Science 2017, 542, 418–429. [Google Scholar] [CrossRef]
- Xie, W.; Li, T.; Tiraferri, A.; Drioli, E.; Figoli, A.; Crittenden, J.C.; Liu, B. Toward the Next Generation of Sustainable Membranes from Green Chemistry Principles. ACS Sustainable Chemistry & Engineering 2020, 9, 50–75. [Google Scholar] [CrossRef]
- De Schouwer, F.; Adriaansen, S.; Claes, L.; De Vos, D.E. Bio-based N-alkyl-2-pyrrolidones by Pd-catalyzed reductive N-alkylation and decarboxylation of glutamic acid. Green Chemistry 2017, 19, 4919–4929. [Google Scholar] [CrossRef]
- Diorazio, L.J.; Hose, D.R.J.; Adlington, N.K. Toward a More Holistic Framework for Solvent Selection. Organic Process Research & Development 2016, 20, 760–773. [Google Scholar] [CrossRef]
- Gao, F.; Bai, R.; Ferlin, F.; Vaccaro, L.; Li, M.; Gu, Y. Replacement strategies for non-green dipolar aprotic solvents. Green Chemistry 2020, 22, 6240–6257. [Google Scholar] [CrossRef]
- Lopez, J.; Pletscher, S.; Aemissegger, A.; Bucher, C.; Gallou, F. N-Butylpyrrolidinone as Alternative Solvent for Solid-Phase Peptide Synthesis. Organic Process Research & Development 2018, 22, 494–503. [Google Scholar] [CrossRef]
- Ohlander, A.; Ludtke, C.; Sahakjan, A.; Johnsson, R.E. N-Butylpyrrolidinone is an equally good solvent as N,N-dimethylformamide for microwave assisted solid phase peptide synthesis. J Pept Sci 2024, 30, e3612. [Google Scholar] [CrossRef] [PubMed]
- Russo, F.; Marino, T.; Galiano, F.; Gzara, L.; Gordano, A.; Organji, H.; Figoli, A. Tamisolve((R)) NxG as an Alternative Non-Toxic Solvent for the Preparation of Porous Poly (Vinylidene Fluoride) Membranes. Polymers (Basel) 2021, 13. [Google Scholar] [CrossRef]
- Russo, F.; Ursino, C.; Sayinli, B.; Koyuncu, I.; Galiano, F.; Figoli, A. Advancements in Sustainable PVDF Copolymer Membrane Preparation Using Rhodiasolv® PolarClean As an Alternative Eco-Friendly Solvent. Clean Technologies 2021, 3, 761–786. [Google Scholar] [CrossRef]
- Depuydt, S.; Van der Bruggen, B. Green Synthesis of Cation Exchange Membranes: A Review. Membranes (Basel) 2024, 14. [Google Scholar] [CrossRef]
- Goh, W.H.D.; Lau, H.S.; Yong, W.F. An integrated life cycle assessment and techno-economic analysis: Evaluation on the production of polymers of intrinsic microporosity (PIM-1) and UiO-66-NH(2) as membrane materials. Sci Total Environ 2023, 892, 164582. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, A.M.; Thür, R.; van Duffel, B.; Vankelecom, I.F.J. Use of green polar aprotic solvents TamiSolve® NxG, DMSO and methyl-THF for the synthesis of asymmetric polyimide-based biogas purification membranes. Journal of Membrane Science 2025, 717. [Google Scholar] [CrossRef]
- Saïdi, S.; Macedonio, F.; Russo, F.; Hannachi, C.; Hamrouni, B.; Drioli, E.; Figoli, A. Preparation and characterization of hydrophobic P(VDF-HFP) flat sheet membranes using Tamisolve® NxG solvent for the treatment of saline water by direct contact membrane distillation and membrane crystallization. Separation and Purification Technology 2021, 275. [Google Scholar] [CrossRef]
- Hosseinabadi, S.R.; Caspers, S.; Shenoy, A.M.; Nevel, A.-S.V.; Rutgeerts, L.A.J.; Vankelecom, I.F.J. Solution casting temperature changing for crosslinked polyvinylidene difluoride solvent resistant nanofiltration membranes: understanding the phase inversion process and performance impact. Separation and Purification Technology 2025, 372. [Google Scholar] [CrossRef]
- Depuydt, S.; Bosko, M.L.; Eerdekens, T.; Van Leuven, W.; Brozzi, E.; Ruya, P.M.; Van der Bruggen, B. Utilizing green solvents to synthesize high performance crosslinked sulfonated PEEK cation exchange membranes. Journal of Membrane Science 2025, 727. [Google Scholar] [CrossRef]
- Bozorgmehr, Z.; Rutgeerts, L.A.J.; Van den Mooter, P.-R.; Vankelecom, I.F.J. Comparative analysis of degassing methods for preparation of pinhole-free membranes. Journal of Membrane Science 2025, 718. [Google Scholar] [CrossRef]
- Kluge, S.; Hartenauer, K.; Tutuş, M. Morphology Behavior of Polysulfone Membranes Made from Sustainable Solvents. Gases 2024, 4, 133–152. [Google Scholar] [CrossRef]
- Alqadhi, N.; Oldal, D.G.; Gopalsamy, K.; Abdulhamid, M.A.; Szekely, G. Poly(ether-ether-ketone) copolymers featuring sulfonyl moieties for organic solvent nanofiltration membranes. Separation and Purification Technology 2025, 355. [Google Scholar] [CrossRef]
- Park, S.-H.; Yang, C.; Ayaril, N.; Szekely, G. Solvent-Resistant Thin-Film Composite Membranes from Biomass-Derived Building Blocks: Chitosan and 2,5-Furandicarboxaldehyde. ACS Sustainable Chemistry & Engineering 2021, 10, 998–1007. [Google Scholar] [CrossRef]
- Tzoumani, I.; Iatridi, Z.; Fidelli, A.M.; Krassa, P.; Kallitsis, J.K.; Bokias, G. Room-Temperature Self-Healable Blends of Waterborne Polyurethanes with 2-Hydroxyethyl Methacrylate-Based Polymers. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef]
- Use of improved n-alkyl pyrrolidone solvents. WO2013107822A1.
- Winters, J.; Dehaen, W.; Binnemans, K. N-butyl pyrrolidone/ionic liquid mixtures as benign alternative solvents to N-methyl pyrrolidone for the synthesis of polyaramids. Materials Today Communications 2021, 29. [Google Scholar] [CrossRef]
- Polyimide compositions and methods. US20160208097A1.
- Bayminov, B.A.; Zabegaeva, O.N.; Sapozhnikov, D.A.; Vygodskii, Y.S. Trends in the Synthesis of Polyimides and Their Nanocomposites. Ineos Open 2021, 4. [Google Scholar] [CrossRef]
- Chuchalov, A.V.; Bayminov, B.A.; Folomin, A.D.; Zabegaeva, O.N.; Godovikov, I.A.; Kononova, E.G.; Kosolapov, A.F.; Semjonov, S.L.; Vygodskii, Y.S.; Sapozhnikov, D.A. Autocatalytic one-step high-temperature synthesis of carboxylated polyimides for in-situ high performance applications. Chemical Engineering Journal 2023, 472. [Google Scholar] [CrossRef]
- Sing Soh, L.; Uyin Hong, S.; Zeng Liang, C.; Fen Yong, W. Green solvent-synthesized polyimide membranes for gas separation: Coupling Hansen solubility parameters and synthesis optimization. Chemical Engineering Journal 2023, 478. [Google Scholar] [CrossRef]
- García, C.; Lozano, Á.E.; de la Campa, J.G.; Vygoskii, Y.; Zolotukhin, M.; de Abajo, J.; Garrido, L.; Guzmán, J. Gas Transport Coefficients of Phthalide-Containing High-Tg Glassy Polymers Determined by Gas-Flux and NMR Measurements. Macromolecules 2015, 48, 2585–2592. [Google Scholar] [CrossRef]
- Kim, Y.J.; Glass, T.E.; Lyle, G.D.; McGrath, J.E. Kinetic and mechanistic investigations of the formation of polyimides under homogeneous conditions. Macromolecules 2002, 26, 1344–1358. [Google Scholar] [CrossRef]
- Ando, S.; Matsuura, T.; Sasaki, S. Coloration of Aromatic Polyimides and Electronic Properties of Their Source Materials. Polymer Journal 1997, 29, 69–76. [Google Scholar] [CrossRef]
- Novakov, I.A.; Orlinson, B.S.; Zavialov, D.V.; Mednikov, S.V.; Gurevich, L.M.; Bogdanov, A.I.; Saveliev, E.N.; Alykova, E.A.; Nakhod, M.A.; Pichugin, A.M.; et al. Optically transparent (co)polyimides based on alicyclic diamines with improved dielectric properties. Russian Chemical Bulletin 2023, 72, 1366–1376. [Google Scholar] [CrossRef]
- Hasegawa, M.; Horii, S. Low-CTE Polyimides Derived from 2,3,6,7-Naphthalenetetracarboxylic Dianhydride. Polymer Journal 2007, 39, 610–621. [Google Scholar] [CrossRef]
- Xu, R.; Li, L.; Jin, X.; Hou, M.; He, L.; Lu, Y.; Song, C.; Wang, T. Thermal crosslinking of a novel membrane derived from phenolphthalein-based cardo poly(arylene ether ketone) to enhance CO2/CH4 separation performance and plasticization resistance. Journal of Membrane Science 2019, 586, 306–317. [Google Scholar] [CrossRef]
- Vinogradova, S.V.; Korshak, V.V.; Vygodskii, Y.S.; Zaitsev, V.I. Study of certain principles of the low temperature polycondensation of 3,3-bis-(4-aminophenyl)phthalide and isophtahlic acid chloride in solution. Polymer Science U.S.S.R. 1967, 9, 731–736. [Google Scholar] [CrossRef]
- Karyakin, N.V.; Rabinovich, I.B.; Rusanov, A.L. Physical Chemistry of the Polycondensation of Polyfunctional Nucleophilic Compounds with Polycarboxylic Acid Derivatives. Russian Chemical Reviews 1985, 54, 594–608. [Google Scholar] [CrossRef]
- Jordan, A.; Hall, C.G.J.; Thorp, L.R.; Sneddon, H.F. Replacement of Less-Preferred Dipolar Aprotic and Ethereal Solvents in Synthetic Organic Chemistry with More Sustainable Alternatives. Chem Rev 2022, 122, 6749–6794. [Google Scholar] [CrossRef]
- Lou, A.; Pethica, B.A.; Somasundaran, P.; Fan, A. Surface and colloid properties of cyclic amides III. Surface activity and micellization of N-butyl-2-pyrrolidone in water. Journal of Dispersion Science and Technology 1999, 20, 569–580. [Google Scholar] [CrossRef]
- Zabegaeva, O.N.; Cherepkova, M.A.; Sapozhnikov, D.A. Synthesis of Polyamide-Imides by the Interaction of Terephthaloyl Chloride and 3,3’,4,4’-Diphenyl Ether Tetracarboxylic Acid Dianhydride with Various Diamines. Ineos Open 2023. [Google Scholar] [CrossRef]
- Lao, H.; Mushtaq, N.; Chen, G.; Jiang, H.; Jiao, Y.; Zhang, A.; Fang, X. Transparent polyamide-imide films with high Tg and low coefficient of thermal expansion: Design and synthesis. Polymer 2020, 206. [Google Scholar] [CrossRef]



| PI | Time, h | ηinh, dL/g | Mn, kDa | Mw, kDa | Mw / Mn | Тg, °С |
Т10%, °С |
|---|---|---|---|---|---|---|---|
| ODPA-AFL | 5 | 0.7 | 56 | 169 | 3.0 | 350 360 350 - |
550 550 540 550 |
| ODPA-AFL1 | 20 | 0.8 | 62 | 164 | 2.7 | ||
| ODPA-AFL2 | 31 | 0.7 | 37 | 133 | 3.6 | ||
| ODPA-AFL3 | 21 | - | - | - | - | ||
| ODPA-AFL4 | 7 | 0.4 | 17 | 69 | 4.2 | 350 | 510 |
| ODPA-AFL0.5:DABA0.5 | 4 | 1.1 | 254 | 462 | 1.8 | 335 | 530 |
| ODPA-AFL0.5:DABA0.54 | 6 | 1.0 | 227 | 449 | 2.0 | 340 | 540 |
| BTDA-AFL0.5:DABA0.5 | 7 | 0.9 | 91 | 398 | 4.4 | 340 | 520 |
| BTDA-AFL0.5:DABA0.54 | 7 | 1.0 | 146 | 421 | 2.9 | 340 | 510 |
| 6FDA-DABA | 6 | 0.7 | 275 | 430 | 1.6 | 330 | 500 |
| 6FDA-DABA4 | 6 | 0.8 | 346 | 537 | 1.6 | 335 | 490 |
| BPADA-DABA | 6 | 0.8 | 239 | 440 | 1.8 | 260 | 470 |
| PMDA-APH | 4 | 0.9 | 191 | 320 | 1.7 | - | 510 |
| NTDA-APH | 5 | 0.3 | 12 | 26 | 2.2 | - | 510 |
| NTDA-APH0.5:DABA0.5 | 4 | 0.2 | 12 | 56 | 4.8 | - | 495 |
| Polymer | Solvent | ηinh, dL/g | Mn, kDa | Mw, kDa | Mw/ Mn | Solubility1 | Tg, °С | T10% °С | |
|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | ||||||||
| TPC-AFL | NBP | 0.9 | 46 | 137 | 2.9 | + | ‒ | 380 | 470 |
| NBP2 | 0.7 | - | - | - | - | - | - | - | |
| NBP3 | 0.4 | - | - | - | - | - | - | - | |
| NMP | 1.8 | 165 | 354 | 2.1 | + | ‒ | 380 | 500 | |
| TPC-TFMB | NBP | 2.3 | 51 | 164 | 3.2 | ± | ‒ | 260 | 500 |
| NMP | 3.0 | 44 | 199 | 4.5 | + | ‒ | 260 | 480 | |
| Polymer | Solvent | ηinh, dL/g | |
|---|---|---|---|
| PAAA | PAI | ||
| ODPA:TPC-AFL | NBP | 0.7 | 0.6 |
| NMP | 1.2 | 0.9 | |
| BTDA:TPC-AFL | NBP | 0.4 | 0.3 |
| NMP | 0.7 | 0.6 | |
| ODPA:TPC-TFMB | NBP | 1.6 | 1.6 |
| NMP | 2.6 | 2.6 | |
| BTDA:TPC-TFMB | NBP | 1.4 | 1.3 |
| NMP | 2.0 | 2.0 | |
| Polymer | Solvent | ηinh of PAAA/PAI (excess of TPC, mol. %), dL/g | |||
|---|---|---|---|---|---|
| 0 | 3 | 7 | 10 | ||
| ODPA:TPC-AFL | NBP | 0.7/0.6 | 0.8/0.7 | 0.9/0.9 | 0.9/0.8 |
| NMP | 1.2/0.9 | 1.1/0.9 | 1.1/0.9 | - | |
| BTDA:TPC-AFL | NBP | 0.4/0.3 | 0.6/0.5 | 0.8/0.7 | 0.8/0.7 |
| NMP | 0.7/0.6 | 0.7/0.6 | 0.8/0.6 | - | |
| Polymer | Solvent | ηinh, dL/g | Mn, kDa | Mw, kDa | Mw/ Mn | Solubility1 | Tg, °С | T10%, °С | |
|---|---|---|---|---|---|---|---|---|---|
| S1 | S2 | ||||||||
| ODPA:TPC-AFL | NBP2 | 0.9 | 57 | 213 | 3.7 | + | ± | 370 | 515 |
| NMP | 0.9 | 91 | 250 | 2.7 | + | ± | 3703 | 5004 | |
| BTDA:TPC-AFL | NBP2 | 0.7 | 46 | 195 | 4.2 | + | ± | 380 | 500 |
| NMP | 0.6 | 30 | 129 | 4.2 | + | ± | 3803 | 4904 | |
| ODPA:TPC-TFMB | NBP | 1.6 | 119 | 316 | 2.7 | + | + | 270 | 500 |
| NMP | 2.6 | 211 | 472 | 3.0 | + | + | 2603 | 5104 | |
| BTDA:TPC-TFMB | NBP | 1.3 | 202 | 384 | 1.9 | ± | ± | 285 | 525 |
| NMP | 2.0 | 166 | 412 | 2.4 | + | + | 2803 | 5104 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.




