Submitted:
03 May 2026
Posted:
04 May 2026
Read the latest preprint version here
Abstract
Keywords:
Lay Summary
1. Introduction
2. Epidemiological Evidence: Parental Autoimmune Diseases and Autism Risk
2.1. Large-Scale Studies
2.2. A Pro-Inflammatory Cytokine Common Denominator
- Psoriasis: TNF-α drives keratinocyte proliferation and inflammatory cascade; anti-TNF biologics are first-line therapy [2]
- Type 1 Diabetes: TNF-α directly induces β-cell apoptosis and promotes autoimmune destruction of pancreatic islets [7]
- Rheumatoid Arthritis: TNF-α orchestrates synovial inflammation and joint destruction; anti-TNF therapy revolutionized treatment [5]
- Normal-Tension Glaucoma: pro-inflammatory and neurodegenerative signaling has been implicated in NTG pathogenesis, and experimental glaucoma models support cytokine-related mechanisms of retinal ganglion cell loss [8]
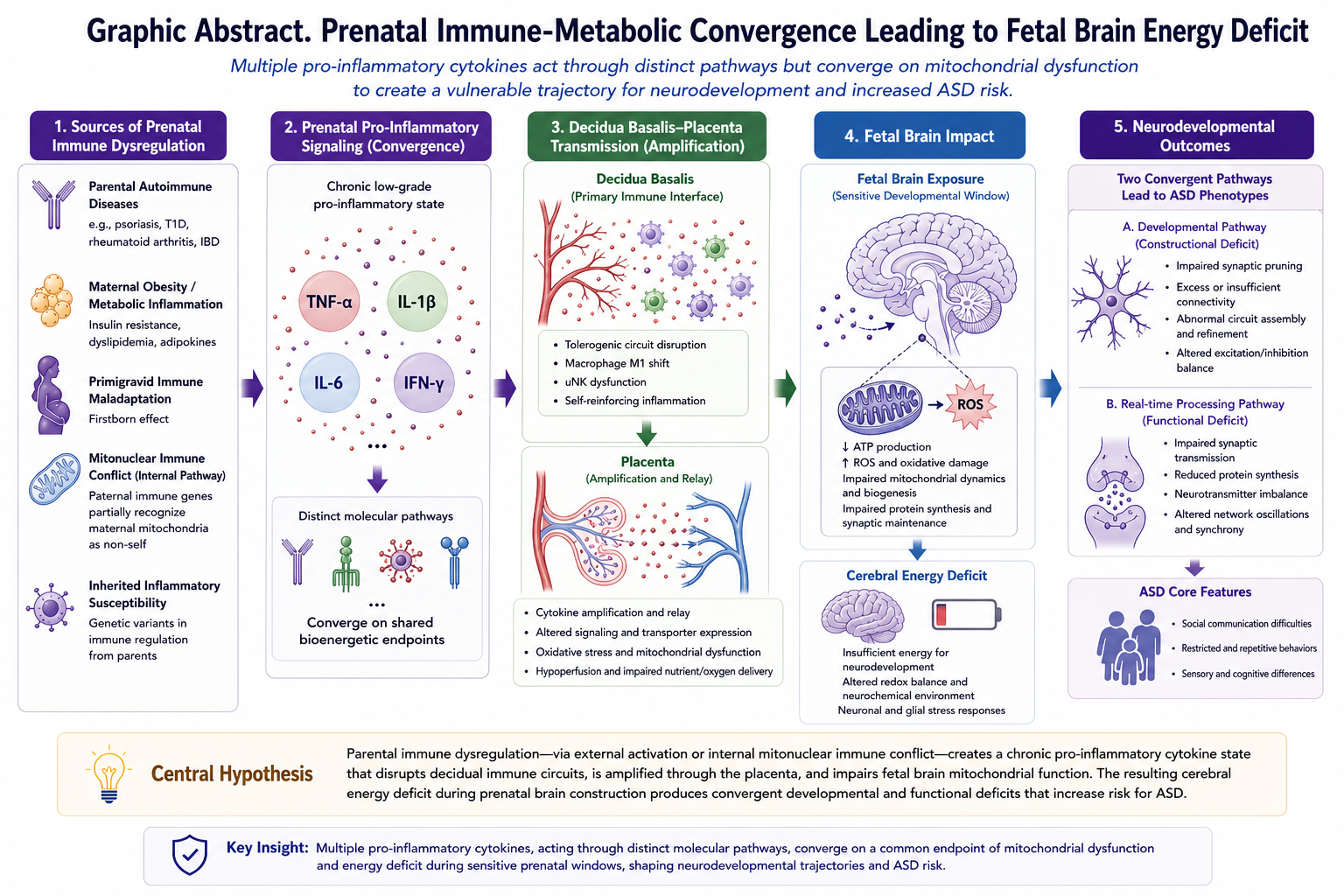
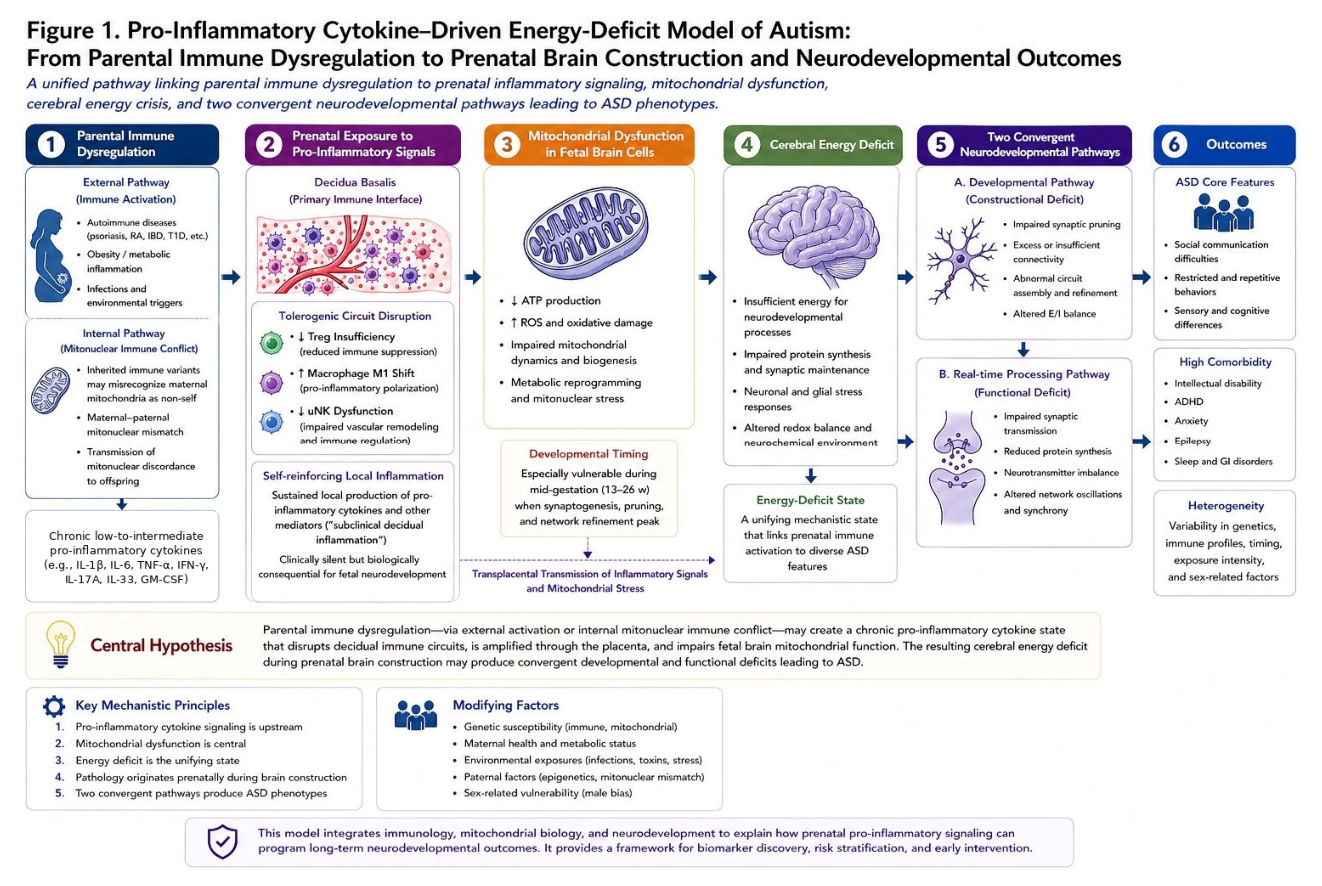
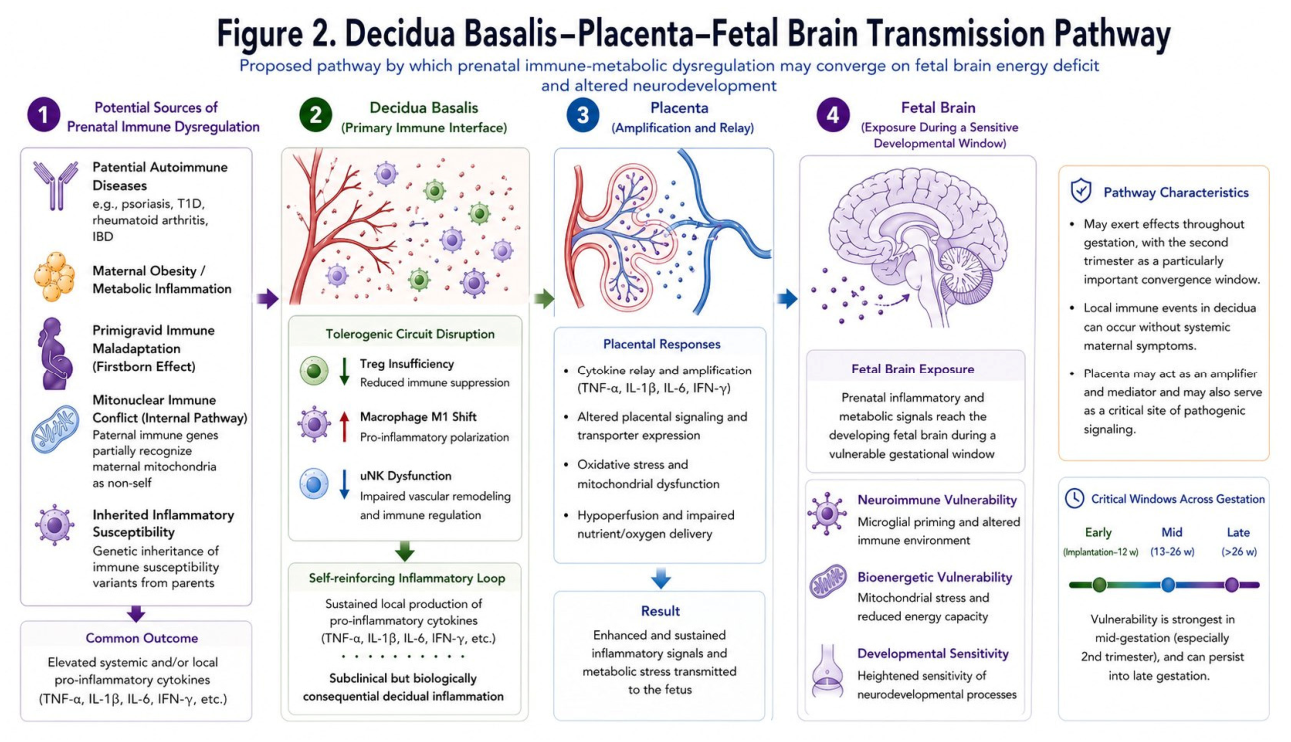
3. Sources of Prenatal Pro-Inflammatory Cytokine Exposure
3.1. Parental Autoimmune Diseases
3.2. Maternal Obesity: An Additional Source of Prenatal Pro-Inflammatory Cytokine Exposure
3.2.1. Epidemiological Evidence
3.2.2. Obesity as a Chronic Inflammatory State
- Adipose tissue is a major source of pro-inflammatory cytokine production, including TNF-α; obese individuals express approximately 2.5-fold more TNF-α mRNA in adipose tissue than lean controls [12]
- Other inflammatory markers (IL-6, CRP, leptin) are also elevated
- Inflammation correlates with degree of adiposity [11]
3.2.3. Mechanism of Fetal Exposure
- Transplacental passage: Maternal inflammatory cytokines can cross the placenta and directly affect fetal brain development
- Placental inflammation: The placenta itself becomes inflamed in obese pregnancies, producing additional local cytokines
- Metabolic stress: Maternal hyperglycemia and insulin resistance further compromise fetal mitochondrial function
- Oxidative stress: Obesity-associated oxidative stress damages both maternal and fetal mitochondria
3.2.4. Convergence with the Energy-Deficit Model
3.3. The Birth Order Effect: Maternal Immune Maladaptation
3.3.1. Epidemiological Evidence for the Firstborn Effect
| Finding | Effect Size | Reference |
| Firstborn autism risk (Utah) | OR 1.8 | Bilder et al. (2009) [14] |
| Short interpregnancy interval (<12 mo) | OR 3.39 | Cheslack-Postava et al. (2011) [15] |
| Preeclampsia in nulliparous women | Significantly higher | Robillard et al. (1994) [16] |
| Partner change resets preeclampsia risk | Returns to primigravid | Dekker & Robillard (2007) [17] |
3.3.2. Primigravid Immune Maladaptation Mechanism
3.3.3. Preeclampsia as Parallel Paradigm
3.4. Mitonuclear Immune Conflict: An Endogenous Source of Pro-Inflammatory Signaling
3.4.1. The Gap in the Cytokine-Mitochondrial Hypothesis
3.4.2. The Unique Inheritance Pattern of Mitochondria
3.4.3. The Conflict Hypothesis: Paternal Immune Genes vs. Maternal Mitochondria
- Immune misrecognition: The paternal contribution to immune recognition machinery (HLA genes, innate immune pathways) may be calibrated to recognize mitochondrial signatures that differ from those inherited from the mother.
- Chronic immune attack: The immune system may mount persistent inflammatory responses against the individual’s own mitochondria, treating them as partially foreign.
- Endogenous cytokine production: This chronic immune activation would result in sustained pro-inflammatory cytokine release—activating the same pathogenic cascade described in previous sections, even without external inflammatory exposure from parental autoimmune disease.
3.4.4. Two Pathways to the Same Outcome
| Pathway 1: External | Pathway 2: Internal | |
| Source of Inflammatory Signaling | Parental autoimmune disease | Mitonuclear immune conflict |
| Mechanism | Genetic inheritance + fetal exposure during pregnancy | Paternal immune attack on maternal mitochondria |
| Parental disease required? | Yes | No |
| Final common pathway | Pro-inflammatory cytokine signaling → Mitochondrial dysfunction → Energy deficit → Autism | |
- Parental autoimmune disease is associated with elevated autism prevalence (Pathway 1)
- Autism also occurs without parental autoimmune disease (Pathway 2)
- Only a subset of children with autoimmune parents develop autism (variable mitonuclear compatibility may be protective or additive)
3.4.5. Testable Predictions and Preliminary Evidence
- Anti-mitochondrial antibodies or mitochondria-targeted immune markers may be elevated in autistic individuals without parental autoimmune history
- Inflammatory cytokines may be elevated even in autism cases without parental autoimmune disease
- Specific HLA haplotype combinations from parents may show associations with autism risk
3.5. The Decidual-Placental Transmission Pathway: How Pro-Inflammatory Signals Reach the Fetal Brain
3.5.1. The Decidual Immune Tolerance Circuit
3.5.2. Disruption of the Tolerogenic Circuit
3.5.3. Placental Amplification and Fetal Exposure
3.5.4. Subclinical Decidual Inflammation: Why Healthy-Appearing Mothers Can Have Affected Offspring
3.5.5. Microglial Reprogramming: The Bridge from Placental Inflammation to Aberrant Synaptic Pruning
4. Pro-Inflammatory Cytokines and Mitochondrial Dysfunction: The Mechanistic Link
4.1. Illustrative Effects of TNF-α on Mitochondrial Function
4.2. Rapid Neurotoxicity as an Illustrative Cytokine Example
- Reduction in mitochondrial basal respiration within 1.5 hours of TNF-α exposure
- Decreased ATP production preceding neuronal cell death
- Effects mediated specifically through TNF-R1 receptor signaling
- Cascade involving caspase-8 activation, membrane potential collapse, and cytochrome c release
4.3. Beyond TNF-α: Convergent Mitochondrial Effects of Other Pro-Inflammatory Cytokines
4.3.1. IL-6: JAK-STAT3 Pathway and Mitochondrial Respiration
4.3.2. IL-1β: NLRP3 Inflammasome and Mitochondrial-Immune Crosstalk
4.3.3. IFN-γ: Metabolic Reprogramming and Complex II Inhibition
| Cytokine | Primary Signaling Pathway | Mitochondrial Target | Functional Consequence |
| TNF-α | NF-κB / caspase cascade | Complex I, III; membrane potential | ETC inhibition, ROS ↑, ATP ↓ |
| IL-6 | gp130-JAK1-STAT3-HIF1α | Complex I (via mitoSTAT3); respiration | Respiration suppression, metabolic shift |
| IL-1β | NLRP3 inflammasome / NF-κB | mtROS/mtDNA feedback loop | Self-amplifying inflammation, BBB permeability ↑ |
| IFN-γ | JAK-STAT1 / itaconate | Complex II (SDH); OXPHOS → glycolysis | Metabolic reprogramming, OXPHOS collapse |
4.4. Tissue-Specific Vulnerability: Why the Brain Bears the Burden
4.4.1. Divergent Energy Dependencies of Brain and Skeletal Muscle
4.4.2. Hypotonia and Hyperlactatemia: Evidence That Muscle Is Not Entirely Spared
5. Consequences of Cerebral Energy Deficit
5.1. Impaired Synaptic Pruning
- Microglia actively phagocytose synapses, requiring substantial ATP
- The infant brain consumes 40% of total body energy [32]—far exceeding adult proportions
- Complement cascade activation and autophagy pathways require ATP
| Parameter | Neurotypical | Autism |
| Synaptic density reduction (childhood→adolescence) | ~50% | ~16% |
| Dendritic spine density | Normal | Elevated |
| mTOR pathway activity | Normal | Hyperactive |
| Autophagy function | Normal | Impaired |
- Local over-connectivity: Excess short-range connections creating “neural noise”
- Long-distance under-connectivity: Insufficient resources for developing major “highway” connections between brain regions
- Reduced signal-to-noise ratio: Difficulty filtering relevant from irrelevant information
- Sensory overload: Heightened sensitivity due to failure to attenuate sensory inputs
5.2. Impaired Social Cognition and Gaze Avoidance
- Fusiform Face Area (FFA): Face identity processing
- Superior Temporal Sulcus (STS): Gaze direction and biological motion
- Amygdala: Emotional salience and threat detection
- Prefrontal Cortex: Social context integration and decision-making
| Experience Category | Representative Quote |
| Energy Exertion | “Eye contact feels like I’m using up a lot of energy. Maximum 2-6 seconds.” |
| Audiovisual Integration Failure | “I cannot listen to someone while making eye contact at the same time.” |
| Cognitive Trade-off | “When I focus on eye contact, I can’t process what’s being said.” |
| Recovery Requirement | “The longer I maintain eye contact, the more recovery time I need afterward.” |
5.3. Epilepsy Comorbidity as Supporting Evidence
- TNF-α, for example, has been reported to increase AMPA receptor surface expression, enhancing excitatory transmission
- Pro-inflammatory signaling may promote GABA receptor internalization, reducing inhibitory tone
- The resulting excitation/inhibition imbalance may lower seizure threshold
- Impaired Na+/K+-ATPase function due to ATP deficit, destabilizing membrane potential
- Compromised GABAergic inhibition, which is highly energy-dependent
- Notably, primary mitochondrial diseases (e.g., MELAS, Leigh syndrome) frequently present with epilepsy
5.4. Impaired Protein Synthesis: The Critical Energy Bottleneck
5.4.1. Mitochondrial Protein Synthesis as Rate-Limiting Step
| Protein | Function | ASD Association | Energy Cost |
| SHANK3 | Synaptic scaffold | 0.5-2% of ASD cases | Large (1,731 aa) |
| NRXN/NLGN | Synaptic adhesion | Multiple variants | Transmembrane |
| PSD-95 | Postsynaptic density | Altered in ASD | Scaffold assembly |
| BDNF | Neuron survival | Reduced in ASD | Activity-dependent |
| FMRP | mRNA regulation | Fragile X syndrome | Translation control |
5.4.2. The SHANK3-Mitochondria Connection
6. Integrated Pathophysiological Model
7. Exploratory Prediction: Normal-Tension Glaucoma and Autism
7.1. NTG as a Condition with Reported Inflammatory Involvement
- Elevated TNF-α levels have been reported in aqueous humor and serum of NTG patients [8]
- TNF-α directly induces RGC apoptosis via TNF-R1 signaling [8]
- Immune-modulating interventions have shown protective effects in some animal models [8]
- Disease progression occurs despite normal intraocular pressure, suggesting the involvement of IOP-independent neurodegenerative mechanisms
7.2. The Untested Association
| Parental Condition | Reported Inflammatory Involvement | Autism Association Studied? |
| Psoriasis | Substantial | Yes (OR 1.59) |
| Type 1 Diabetes | Documented | Yes (OR 1.49-2.36) |
| Rheumatoid Arthritis | Documented | Yes (OR 1.51) |
| Normal-Tension Glaucoma | Reported | No studies identified |
8. Therapeutic Implications
8.1. Anti-Inflammatory Interventions
8.2. Early Identification
8.3. Toward Future Risk Stratification: Prenatal Cytokine Monitoring
9. Limitations and Future Directions
10. Conclusions
References
- Meltzer A, Van de Water J. The role of the immune system in autism spectrum disorder. Neuropsychopharmacology. 2017;42(1):284-298.
- Wu S, Ding Y, Wu F, et al. Family history of autoimmune diseases is associated with an increased risk of autism in children: A systematic review and meta-analysis. Neurosci Biobehav Rev. 2015;55:322-332.
- Xiang AH, Wang X, Martinez MP, et al. Maternal Type 1 Diabetes and Risk of Autism in Offspring. JAMA. 2018;320(1):89-91.
- Persson M, et al. Maternal type 1 diabetes, pre-term birth and risk of autism spectrum disorder. Int J Epidemiol. 2023;52(2):377-386.
- Keil A, Daniels JL, Forssen U, et al. Parental autoimmune diseases associated with autism spectrum disorders in offspring. Epidemiology. 2010;21(6):805-808.
- Atladóttir HO, Pedersen MG, Thorsen P, et al. Association of family history of autoimmune diseases and autism spectrum disorders. Pediatrics. 2009;124(2):687-694.
- Quattrin T, Haller MJ, Steck AK, et al. Golimumab and Beta-Cell Function in Youth with New-Onset Type 1 Diabetes. N Engl J Med. 2020;383(21):2007-2017.
- Nakazawa T, et al. Tumor necrosis factor-alpha mediates oligodendrocyte death and delayed retinal ganglion cell loss in a mouse model of glaucoma. J Neurosci. 2006;26(49):12633-12641.
- Li YM, Ou JJ, Liu L, Zhang D, Zhao JP, Tang SY. Association between maternal obesity and autism spectrum disorder in offspring: a meta-analysis. J Autism Dev Disord. 2016;46(1):95-102.
- Wang Y, Tang S, Xu S, Weng S, Liu Z. Maternal body mass index and risk of autism spectrum disorders in offspring: a meta-analysis. Sci Rep. 2016;6:34248.
- Cawthorn WP, Sethi JK. TNF-α and adipocyte biology. FEBS Lett. 2008;582(1):117-131.
- Kern PA, Saghizadeh M, Ong JM, Bosch RJ, Deem R, Simsolo RB. The expression of tumor necrosis factor in human adipose tissue: regulation by obesity, weight loss, and relationship to lipoprotein lipase. J Clin Invest. 1995;95(5):2111-2119.
- Pantham P, Aye IL, Powell TL. Inflammation in maternal obesity and gestational diabetes mellitus. Placenta. 2015;36(7):709-715.
- Bilder D, Pinborough-Zimmerman J, Miller J, McMahon W. Prenatal, perinatal, and neonatal factors associated with autism spectrum disorders. Pediatrics. 2009;123(5):1293-1300.
- Cheslack-Postava K, Liu K, Bearman PS. Closely spaced pregnancies are associated with increased odds of autism in California sibling births. Pediatrics. 2011;127(2):246-253.
- Robillard PY, Hulsey TC, Perianin J, Janky E, Miri EH, Papiernik E. Association of pregnancy-induced hypertension with duration of sexual cohabitation before conception. Lancet. 1994;344(8928):973-975.
- Dekker G, Robillard PY. Pre-eclampsia: Is the immune maladaptation hypothesis still standing? J Reprod Immunol. 2007;76(1-2):8-16.
- Zhang Q, Raoof M, Chen Y, et al. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature. 2010;464(7285):104-107.
- Zhou R, Yazdi AS, Menu P, Tschopp J. A role for mitochondria in NLRP3 inflammasome activation. Nature. 2011;469(7329):221-225.
- Zhang B, Angelidou A, Alysandratos KD, et al. Mitochondrial DNA and anti-mitochondrial antibodies in serum of autistic children. J Neuroinflammation. 2010;7:80.
- Moffett A, Colucci F. Co-evolution of NK receptors and HLA ligands in humans is driven by reproduction. Immunol Rev. 2015;267(1):283-297.
- Erlebacher A. Immunology of the maternal-fetal interface. Annu Rev Immunol. 2013;31:387-411.
- Mor G, Aldo P, Alvero AB. The unique immunological and microbial aspects of pregnancy. Nat Rev Immunol. 2017;17(8):469-482.
- Paolicelli RC, Bolasco G, Pagani F, et al. Synaptic pruning by microglia is necessary for normal brain development. Science. 2011;333(6048):1456-1458.
- Cunningham CL, Martínez-Cerdeño V, Noctor SC. Microglia regulate the number of neural precursor cells in the developing cerebral cortex. J Neurosci. 2013;33(10):4216-4233.
- Vargas DL, Nascimbene C, Krishnan C, Zimmerman AW, Pardo CA. Neuroglial activation and neuroinflammation in the brain of patients with autism. Ann Neurol. 2005;57(1):67-81.
- Li X, Chauhan A, Sheikh AM, et al. Elevated immune response in the brain of autistic patients. J Neuroimmunol. 2009;207(1-2):111-116.
- Ashwood P, Krakowiak P, Hertz-Picciotto I, et al. Elevated plasma cytokines in autism spectrum disorders provide evidence of immune dysfunction and are associated with impaired behavioral outcome. Brain Behav Immun. 2011;25(1):40-45.
- Xie J, Huang L, Li X, et al. Immunological cytokine profiling identifies TNF-α as a key molecule dysregulated in autistic children. Oncotarget. 2017;8(47):82390-82398.
- Morris G, Berk M. The many roads to mitochondrial dysfunction in neuroimmune and neuropsychiatric disorders. BMC Med. 2015;13:68.
- Deng W, et al. Rapid mitochondrial dysfunction mediates TNF-alpha-induced neurotoxicity. J Neurochem. 2015;132(4):443-451.
- Kuzawa CW, Chugani HT, Grossman LI, et al. Metabolic costs and evolutionary implications of human brain development. Proc Natl Acad Sci U S A. 2014;111(36):13010-13015.
- Lopez-Espejo MA, Nuñez AC, Moscoso OC, Escobar RG. Clinical characteristics of children affected by autism spectrum disorder with and without generalized hypotonia. Eur J Pediatr. 2021;180(10):3243-3246.
- Rossignol DA, Frye RE. Mitochondrial dysfunction in autism spectrum disorders: a systematic review and meta-analysis. Mol Psychiatry. 2012;17(3):290-314.
- Tang G, Gudsnuk K, Kuo SH, et al. Loss of mTOR-dependent macroautophagy causes autistic-like synaptic pruning deficits. Neuron. 2014;83(5):1131-1143.
- Trevisan DA, Roberts N, Lin C, Birmingham E. How do adults and teens with self-declared Autism Spectrum Disorder experience eye contact? PLOS ONE. 2017;12(11):e0188446.
- Dalton KM, Nacewicz BM, Johnstone T, et al. Gaze fixation and the neural circuitry of face processing in autism. Nat Neurosci. 2005;8(4):519-526.
- Amiet C, Gourfinkel-An I, Bouzamondo A, et al. Epilepsy in autism is associated with intellectual disability and gender: evidence from a meta-analysis. Biol Psychiatry. 2008;64(7):577-582.
- Buttgereit F, Brand MD. A hierarchy of ATP-consuming processes in mammalian cells. Biochem J. 1995;312(Pt 1):163-167.
- Bülow P, Patgiri A, Faundez V. Mitochondrial protein synthesis and the bioenergetic cost of neurodevelopment. iScience. 2022;25(9):104920.
- Liu X, Liu H, Gu N, Pei J, Lin X, Zhao W. Preeclampsia promotes autism in offspring via maternal inflammation and fetal NFκB signaling. Life Sci Alliance. 2023;6(8):e202301957.
- Mahadevan U, Long MD, Kane SV, et al. Pregnancy and neonatal outcomes after fetal exposure to biologics and thiopurines among women with inflammatory bowel disease. Gastroenterology. 2021;160(4):1131-1139.
- Sarieva K, Kagermeier T, Khakipoor S, et al. Human brain organoid model of maternal immune activation identifies radial glia cells as selectively vulnerable. Mol Psychiatry. 2023;28(12):5077-5089.
- Smith SE, Li J, Garbett K, Mirnics K, Patterson PH. Maternal immune activation alters fetal brain development through interleukin-6. J Neurosci. 2007;27(40):10695-10702.
- Wegrzyn J, Potla R, Ber YJ, et al. Function of mitochondrial Stat3 in cellular respiration. Science. 2009;323(5915):793-797.
- Ahn J, Kim M, et al. The pro-inflammatory cytokine IL6 suppresses mitochondrial function via the gp130-JAK1/STAT1/3-HIF1α/ERRα axis. Cell Rep. 2025;44(3):115358.
- Shi J, Bhargava P, Zhang Q. NLRP3 inflammasome in neuroinflammation and central nervous system diseases. Cell Mol Immunol. 2025;22(4):341-355.
- Jessop F, Buntyn R, Schwarz B, Priest S, Bosio CM. Interferon gamma reprograms host mitochondrial metabolism through inhibition of complex II to control intracellular bacterial replication. Infect Immun. 2020;88(2):e00744-19.
- Garabatos N, Alvarez-Sierra D, et al. Pro-inflammatory macrophage activation does not require inhibition of oxidative phosphorylation. EMBO Rep. 2025;26(4):e59465.
- Kim S, et al. Chronic low-level IFN-γ expression disrupts mitochondrial complex I activity in renal macrophages: An early mechanistic driver of lupus nephritis pathogenesis. Int J Mol Sci. 2025;26(1):63.
| Parental Disease | Odds Ratio | 95% CI | Key Reference |
| Psoriasis | 1.59 | 1.21-2.10 | Wu et al. (2015) [2] |
| Type 1 Diabetes (T1D) | 1.49-2.36 | 1.21-4.12 | Xiang et al. (2018); Persson et al. (2023) [3,4] |
| Rheumatoid Arthritis | 1.51 | 1.14-2.00 | Keil et al. (2010) [5] |
| Hypothyroidism | 1.64 | 1.16-2.32 | Atladóttir et al. (2009) [6] |
| Any Autoimmune Disease | 1.28-1.50 | 1.11-1.75 | Wu et al. (2015) [2] |
| Mechanism | Effect on Energy Metabolism |
| ETC Complex I Inhibition | Blocks electron transfer at the first step of oxidative phosphorylation |
| ETC Complex III Inhibition | Disrupts cytochrome bc1 complex function |
| Cytochrome c Oxidase (COX) | Reduces terminal electron transfer and oxygen consumption |
| Membrane Depolarization | Collapses mitochondrial membrane potential (ΔΨm), halting ATP synthesis |
| PDH Suppression | Inhibits pyruvate dehydrogenase, blocking glucose entry into TCA cycle |
| ROS Overproduction | Increases reactive oxygen species, causing oxidative damage to mitochondrial components |
| Warburg Effect Induction | Shifts metabolism to inefficient aerobic glycolysis (2 vs 36 ATP per glucose) |
| Parameter | Brain (Neurons) | Skeletal Muscle |
| Primary ATP source | OXPHOS (~95%) | OXPHOS + glycolysis (variable) |
| Glycolytic capacity | Very limited | High (type II fibers) |
| Glycogen reserves | Minimal | Substantial |
| Tolerance to OXPHOS impairment | Very low | Moderate to high |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




