Submitted:
25 December 2025
Posted:
25 December 2025
You are already at the latest version
Abstract

Keywords:
1. Introduction
2. Results
2.1. Nutritional Composition
2.2. Mineral Profile
2.3. Fatty Acid Profile by GC-FID
2.4. Vitamin E Profile by HPLC-DAD-FLD
2.5. Bioactive Compounds
2.5.1. Total Phenolics, Flavonoids and Saponins Contents
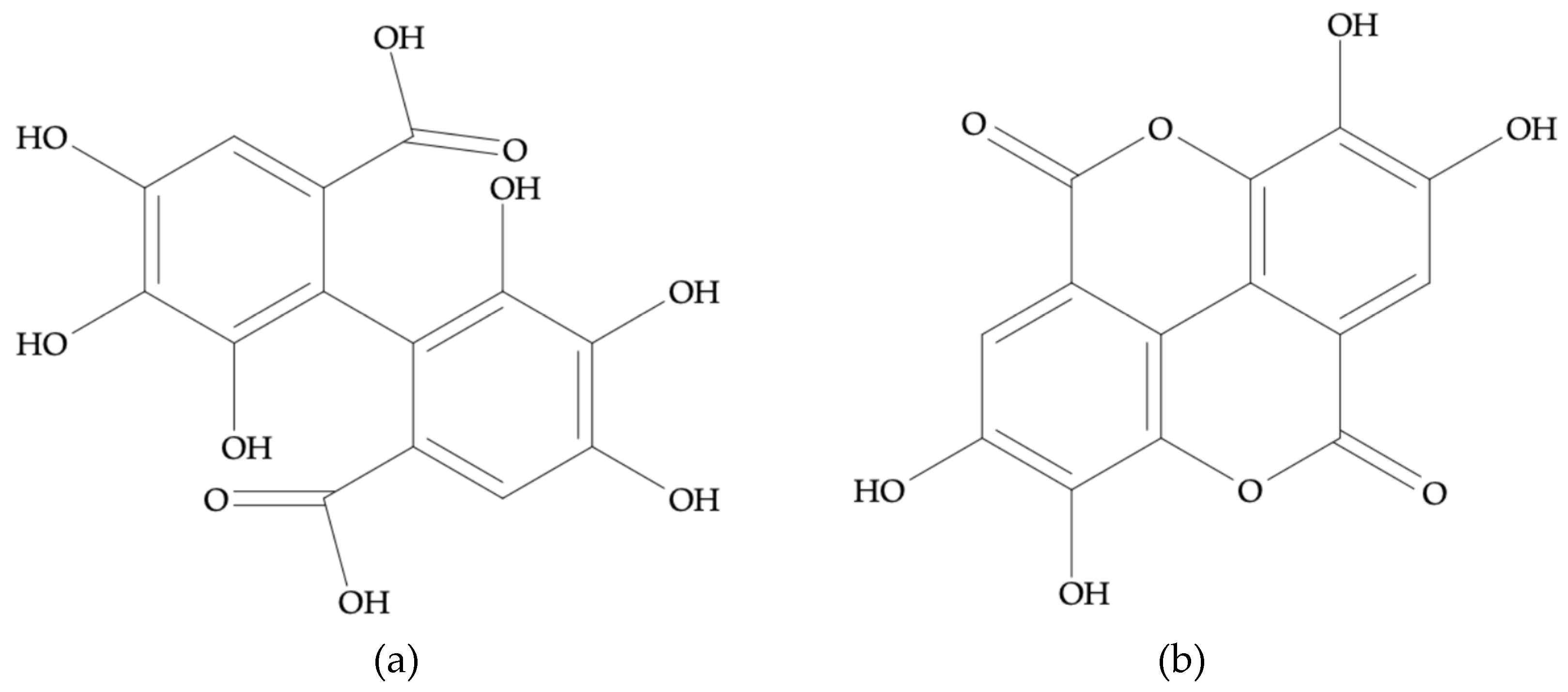
2.5.2. Phenolic Profile by HPLC-DAD
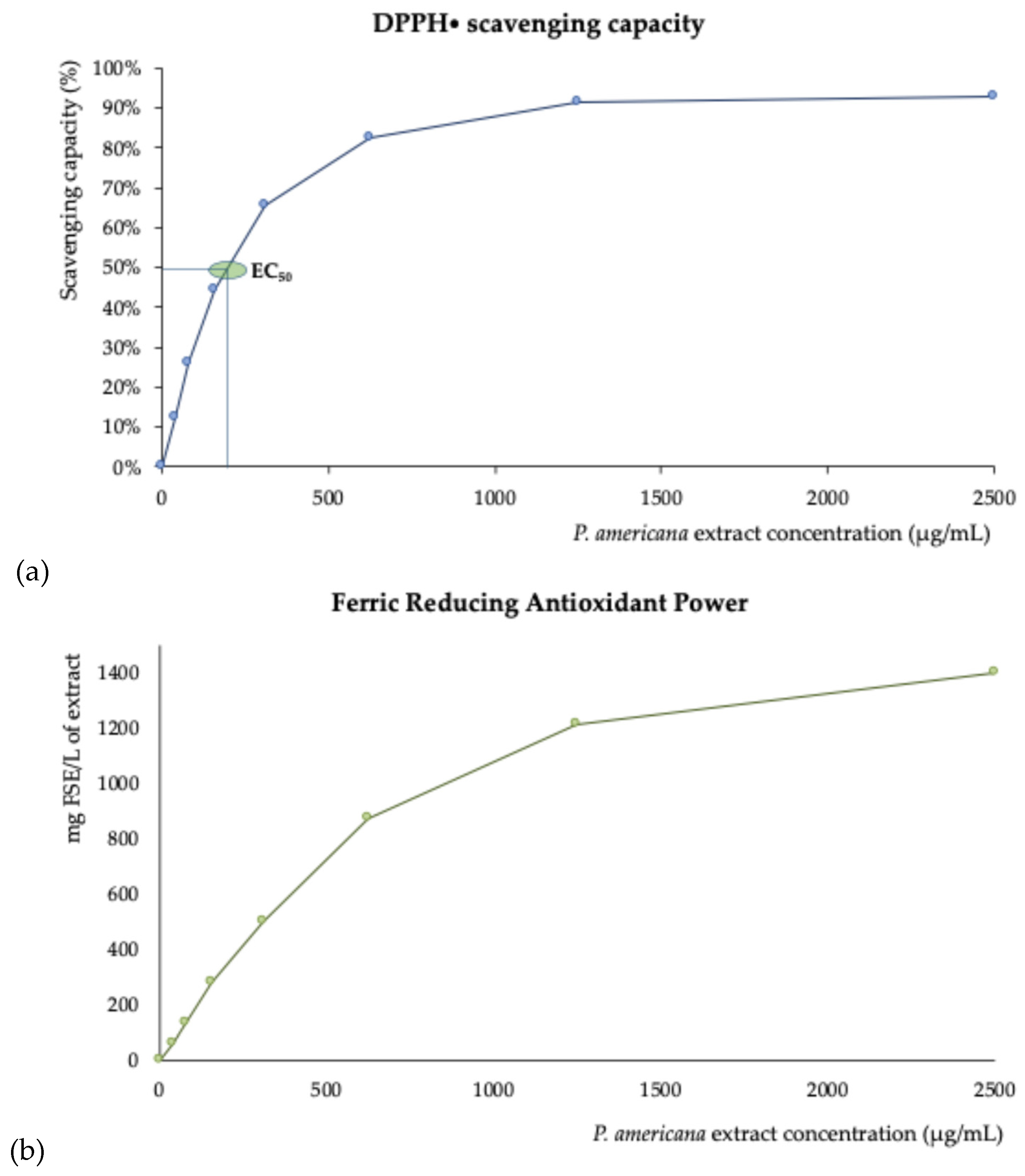
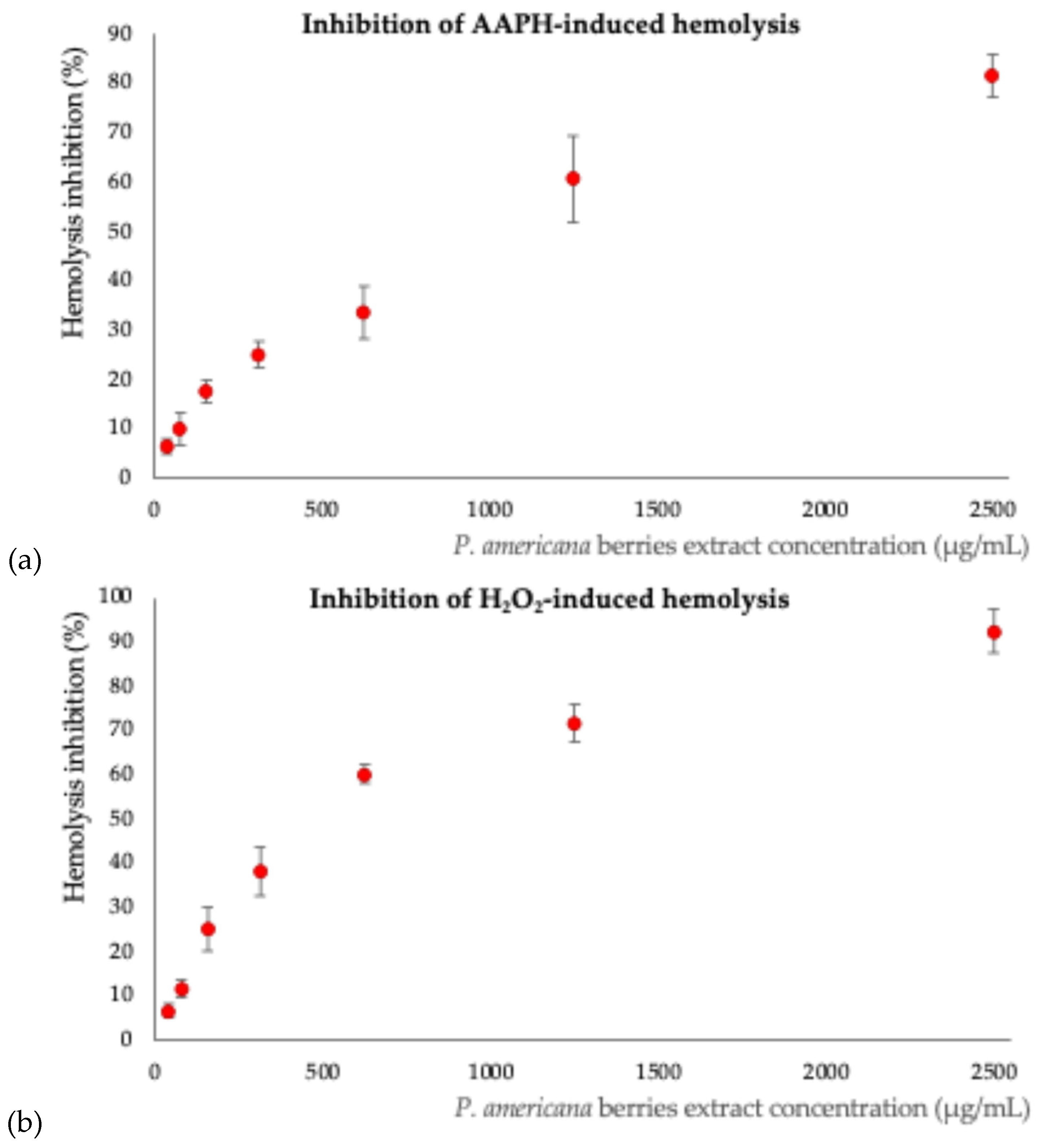
2.6. Antioxidant Activity
3. Discussion
3.1. Nutritional Composition and Mineral Profile
3.2. Fatty Acids and Vitamin E Profiles
3.3. Bioactive Compounds and Phenolic Profile
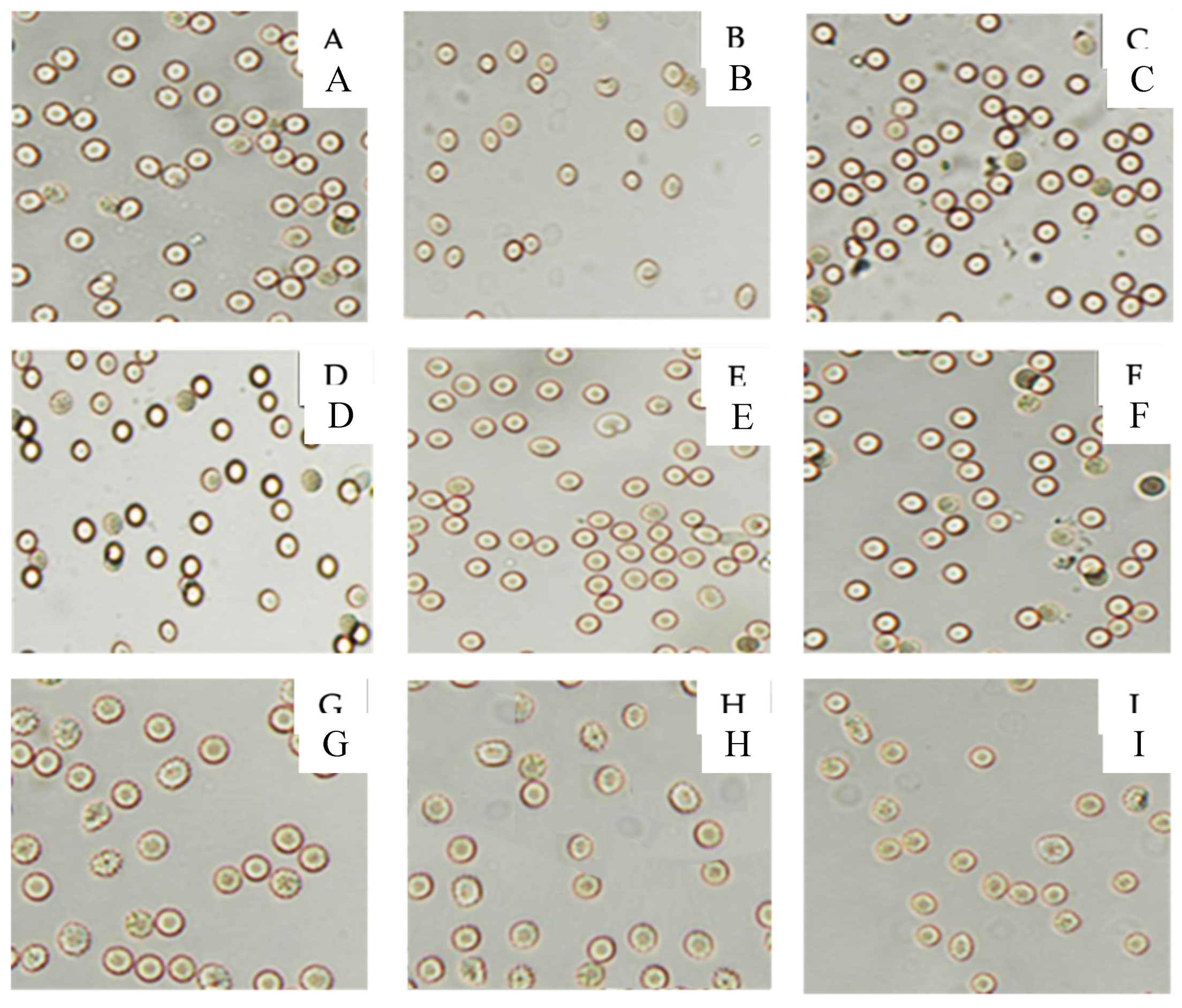
3.4. Antioxidant Behavior
4. Materials and Methods
4.1. Material and Sample Preparation
4.2. Nutritional Analysis
4.3. Macro and Trace Elements Composition
4.4. Lipidic Fraction Extraction
4.4.1. Vitamin E Profile by HPLC-DAD-FLD
4.4.2. Fatty Acid Profile
4.5. Bioactive Contents and Antioxidant Activity
4.5.1. Extracts
4.5.2. Total Phenolic Content
4.5.3. Total Flavonoids Content
4.5.4. Total Saponin Content
4.5.5. Individual Phenolics Profile
4.5.6. Antioxidant Activity
4.5.6.1. DPPH Free Radical Scavenging
4.5.6.2. Ferric Reducing Antioxidant Power (FRAP)
4.5.6.3. Effect on Erythrocyte Oxidative-Induced Hemolysis.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Follak, S.; Schwarz, M.; Essl, F. Notes on the Occurrence of Phytolacca americana L. in Crop Fields and Its Potential Agricultural Impact. Bioinvasions Rec 2022, 11, 620–630. [Google Scholar] [CrossRef]
- Schirmel, J. Differential Effects of American Pokeweed (Phytolacca americana) Invasion on Ground-Dwelling Forest Arthropods in Southwest Germany. Biol Invasions 2020, 22, 1289–1298. [Google Scholar] [CrossRef]
- Veleșcu, I.D.; Crivei, I.C.; Balint, A.B.; Arsenoaia, V.N.; Robu, A.D.; Stoica, F.; Rațu, R.N. Valorization of Betalain Pigments Extracted from Phytolacca americana L. Berries as Natural Colorant in Cheese Formulation. Agriculture 2025, 15, 86. [Google Scholar] [CrossRef]
- Jasińska, J.M.; Michalska, K.; Szuwarzyński, M.; Mazur, T.; Cholewa-Wójcik, A.; Kopeć, M.; Juszczak, L.; Kamińska, I.; Nowak, N.; Jamróz, E. Phytolacca americana Extract as a Quality-Enhancing Factor for Biodegradable Double-Layered Films Based on Furcellaran and Gelatin – Property Assessment. Int J Biol Macromol 2024, 279, 135155. [Google Scholar] [CrossRef] [PubMed]
- Bailly, C. Medicinal Properties and Anti-Inflammatory Components of Phytolacca (Shanglu). Digital Chinese Medicine 2021, 4, 159–169. [Google Scholar] [CrossRef]
- Díaz, R.; Yáñez-Sánchez, M.; de la Fuente, F.; Ortega, A.; Figueroa-Carvajal, A.; Gangitano, D.; Scholz-Wagenknecht, O. Toxic and Hallucinogenic Plants of Southern Chile of Forensic Interest: A Review. Plants 2025, 14, 2196. [Google Scholar] [CrossRef]
- Popovici, L.F.; Brinza, I.; Gatea, F.; Badea, G.I.; Vamanu, E.; Oancea, S.; Hritcu, L. Enhancement of Cognitive Benefits and Anti-Anxiety Effects of Phytolacca americana Fruits in a Zebrafish (Danio Rerio) Model of Scopolamine-Induced Memory Impairment. Antioxidants 2025, 14, 97. [Google Scholar] [CrossRef]
- Trunjaruen, A.; Luecha, P.; Taratima, W. Micropropagation of Pokeweed (Phytolacca americana L.) and Comparison of Phenolic, Flavonoid Content, and Antioxidant Activity between Pokeweed Callus and Other Parts. PeerJ 2022, 10, e12892. [Google Scholar] [CrossRef]
- Han, H.Y.; Han, K.H.; Ahn, J.H.; Park, S.M.; Kim, S.; Lee, B.S.; Min, B.S.; Yoon, S.; Oh, J.H.; Kim, T.W. Subchronic Toxicity Assessment of Phytolacca americana L. (Phytolaccaceae) in F344 Rats. Nat Prod Commun 2020, 15. [Google Scholar] [CrossRef]
- Sydor, B.G.; Ramos-Milaré, Á.C.F.H.; Pereira, M.B.; Brustolin, A.Á.; Montaholi, D.C.; Lera-Nonose, D.S.S.L.; Negri, M.; de Lima Scodro, R.B.; Teixeira, J.J.V.; Lonardoni, M.V.C. Plants of the Phytolaccaceae Family with Antimicrobial Activity: A Systematic Review. Phytotherapy Research 2022, 36, 3505–3528. [Google Scholar] [CrossRef] [PubMed]
- Ravikiran, G.; Raju, A.B.; Venugopal, Y. Phytolacca americana: A Review. Int J Res Pharm Biomed Sci 2011, 3, 942–946. [Google Scholar]
- Wang, P.C.; Wang, Q.H.; Zhao, S.; Sun, X.; Kuang, H.X. Research Progress on Chemical Constituents, Pharmacological Effects, and Clinical Applications of Phytolaccae Radix. Chin Tradit Herb Drugs 2014, 45, 2722–2731. [Google Scholar] [CrossRef]
- Alshamar, H.A.; Hatem, N.A.; Dapson, R.W. Betacyanins Are Plant-Based Dyes with Potential as Histological Stains. Biotech Histochem 2022, 97, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Xjung, S.Y. Technical Approaches of a Natural Dye Extracted from Phytolacca americana L.-Berries with Chemical Mordants. Technol Health Care 2014, 22, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, A.K.; Al-Rowaily, S.L.; Faisal, M.; Alatar, A.A.; El-Bana, M.I.; Assaeed, A.M. Nutritive Value and Antioxidant Activity of Some Edible Wild Fruits in the Middle East. J Med Plants Res 2013, 7, 938–946. [Google Scholar] [CrossRef]
- Subedi, T. An Assessment of Mineral Contents in Fruits. Prithvi Academic Journal 2023, 6, 21–31. [Google Scholar] [CrossRef]
- Islam, M.N.; Shahriar, S.M.S.; Amin, M.R.; Khan, F.; Hassan, M.T.; Hasan, M.M.; Hanif, M.A.; Salam, S.M.A. Heavy Metal Contamination in Fruits and Human Health Risk Assessment in Northwestern Bangladesh. J Food Compos Anal 2025, 148, 108159. [Google Scholar] [CrossRef]
- Neblea, M.A.; Marian, M.C.; Aydin, T. A Comprehensive Review of the Invasive Species Phytolacca acinosa Roxb. Sustainability 2025, 17, 4826. [Google Scholar] [CrossRef]
- Szewczyk, K.; Chojnacka, A.; Górnicka, M. Tocopherols and Tocotrienols—Bioactive Dietary Compounds; What Is Certain, What Is Doubt? Int J Mol Sci 2021, 22, 6222. [Google Scholar] [CrossRef]
- Breda, C.; Nascimento, A.; Meghwar, P.; Lisboa, H.; Aires, A.; Rosa, E.; Ferreira, L.; Barros, A.N. Phenolic Composition and Antioxidant Activity of Edible Flowers: Insights from Synergistic Effects and Multivariate Analysis. Antioxidants 2025, 14, 282. [Google Scholar] [CrossRef]
- Vollmannová, A.; Bojňanská, T.; Musilová, J.; Lidiková, J.; Cifrová, M. Quercetin as One of the Most Abundant Represented Biological Valuable Plant Components with Remarkable Chemoprotective Effects - A Review. Heliyon 2024, 10, e33342. [Google Scholar] [CrossRef]
- Wang, J.; Zhao, F.; Wu, W.; Lyu, L.; Li, W.; Zhang, C. Ellagic Acid from Hull Blackberries: Extraction, Purification, and Potential Anticancer Activity. Int J Mol Sci 2023, 24, 15228. [Google Scholar] [CrossRef] [PubMed]
- Kwok, H.; Wagner, A.; Alharbi, C.; Alshebremi, H.O.A.; Babiker, M.; Rahmani, A.Y.; Obaid, H.; Alharbi, A.; Alshebremi, M.; Yousif Babiker, A.; et al. The Role of Quercetin, a Flavonoid in the Management of Pathogenesis Through Regulation of Oxidative Stress, Inflammation, and Biological Activities. Biomolecules 2025, 15, 151. [Google Scholar] [CrossRef] [PubMed]
- Hadidi, M.; Liñán-Atero, R.; Tarahi, M.; Christodoulou, M.C.; Aghababaei, F. The Potential Health Benefits of Gallic Acid: Therapeutic and Food Applications. Antioxidants 2024, 13, 1001. [Google Scholar] [CrossRef]
- Sheng, Y.; Sun, Y.; Tang, Y.; Yu, Y.; Wang, J.; Zheng, F.; Li, Y.; Sun, Y. Catechins: Protective Mechanism of Antioxidant Stress in Atherosclerosis. Front Pharmacol 2023, 14, 1144878. [Google Scholar] [CrossRef]
- Śliwińska, A.A.; Tomiczak, K. Advancing the Potential of Polyscias fruticosa as a Source of Bioactive Compounds: Biotechnological and Pharmacological Perspectives. Molecules 2025, 30, 3460. [Google Scholar] [CrossRef]
- Zheleva-Dimitrova, D.Z. Antioxidant and Acetylcholinesterase Inhibition Properties of Amorpha fruticosa L. and Phytolacca americana L. Pharmacogn Mag 2013, 9, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Zhao, W.; Yang, Z.; Subbiah, V.; Suleria, H.A.R. Extraction and Characterization of Phenolic Compounds and Their Potential Antioxidant Activities. Environ Sci Pollut Res Int 2022, 29, 81112. [Google Scholar] [CrossRef]
- Albano, G.D.; Gagliardo, R.P.; Montalbano, A.M.; Profita, M. Overview of the Mechanisms of Oxidative Stress: Impact in Inflammation of the Airway Diseases. Antioxidants 2022, 11, 2237. [Google Scholar] [CrossRef]
- Vinha, A.F.; Sousa, C.; Costa, C. Oxidative Stress, Antioxidants and Biomarkers: Appreciation for Analysis Methods for Health Promotion. Int Aca Res J Int Med Pub Heath 2023, 4, 47–55. [Google Scholar]
- Daraghmeh, D.N.; Karaman, R. The Redox Process in Red Blood Cells: Balancing Oxidants and Antioxidants. Antioxidants 2025, 14, 36. [Google Scholar] [CrossRef]
- Shahidi, F.; Samarasinghe, A. How to Assess Antioxidant Activity? Advances, Limitations, and Applications of in Vitro, in Vivo, and Ex Vivo Approaches. Food Production, Processing and Nutrition 2025, 7, 50. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists Official Methods of Analysis of AOAC INTERNATIONAL. AOAC 2012, 19st ed.
- Regulation - 1169/2011 - EN - Food Information to Consumers Regulation - EUR-Lex. Available online: https://eur-lex.europa.eu/eli/reg/2011/1169/oj/eng (accessed on 29 November 2025).
- Pinto, E.; Almeida, A.A.; Aguiar, A.A.R.M.; Ferreira, I.M.P.L.V.O. Changes in Macrominerals, Trace Elements and Pigments Content during Lettuce (Lactuca Sativa L.) Growth: Influence of Soil Composition. Food Chem 2014, 152, 603–611. [Google Scholar] [CrossRef]
- Alves, R.C.; Casal, S.; Oliveira, M.B.P.P. Determination of Vitamin e in Coffee Beans by HPLC Using a Micro-Extraction Method. Food Sci Technol Int 2009, 15, 57–63. [Google Scholar] [CrossRef]
- ISO 12966-2 Animal and Vegetable Fats and Oils Gas Chromatography of Fatty Acid Methyl Esters Part 2: Preparation of Methyl Esters of Fatty Acids. ISO Standards 2017.
- Vinha, A.F.; Costa, A.S.G.; Barreira, J.C.M.; Pacheco, R.; Oliveira, M.B.P.P. Chemical and Antioxidant Profiles of Acorn Tissues from Quercus Spp.: Potential as New Industrial Raw Materials. Ind Crops Prod 2016, 94, 134–151. [Google Scholar] [CrossRef]
- Costa, A.S.G.; Alves, R.C.; Vinha, A.F.; Barreira, S.V.P.; Nunes, M.A.; Cunha, L.M.; Oliveira, M.B.P.P. Optimization of Antioxidants Extraction from Coffee Silverskin, a Roasting by-Product, Having in View a Sustainable Process. Ind Crops Prod 2014, 53, 350–357. [Google Scholar] [CrossRef]
- Helaly, F.M.; Soliman, H.S.M.; Soheir, A.D.; Ahmed, A.A. Controlled Release of Migration of Molluscicidal Saponin from Different Types of Polymers Containing Calendula officinalis. Adv Poly Technol 2001, 20, 305–311. [Google Scholar] [CrossRef]
- Costa, A.S.G.; Alves, R.C.; Vinha, A.F.; Costa, E.; Costa, C.S.G.; Nunes, M.A.; Almeida, A.A.; Santos-Silva, A.; Oliveira, M.B.P.P. Nutritional, Chemical and Antioxidant/Pro-Oxidant Profiles of Silverskin, a Coffee Roasting By-Product. Food Chem 2018, 267, 28–35. [Google Scholar] [CrossRef] [PubMed]
| Nutrient | P. americana berries |
| Moisture | 8.42 ± 0.37 |
| Ash | 7.90 ± 0.21 |
| Crude protein | 10.71 ± 0.18 |
| Fat | 14.07 ± 0.53 |
| Carbohydrates | 67.31 ± 0.61 |
| Fibre | 35.12 ± 2.19 |
| Insoluble Fibre | 23.18 ± 0.85 |
| Soluble Fibre | 11.94 ± 1.34 |
| Remaining Carbohydrates | 32.19 ± 2.32 |
| Energy (Kcal/ 100g) | 368.53 ± 1.51 |
| Macro elements | |
| Ca (mg/ g)) | 1.15 ± 72 |
| K (mg/ g) | 21.63 ± 0.28 |
| Mg (mg /g) | 1.69 ± 115 |
| Na (mg/ g) | 0.023 ± 0.005 |
| Essential trace elements | |
| Fe (µg/ g) | 87.15 ± 11.63 |
| Cu (µg/ g) | 4.33 ± 0.03 |
| Zn (µg/ g) | 10.8 ± 0.3 |
| Mn (µg/ g) | 13.75 ± 0.15 |
| Mo (µg/ g) | 0.29 ± 0.02 |
| Co (µg/ g) | 0.29 ± 0.004 |
| Se (µg/ g) | < LoD |
| Non-essential and toxic trace elements | |
| As (µg/ g) | 0.12 ± 0.01 |
| Ba (µg/ g) | 1.0 ± 0.1 |
| Be (µg/ g) | 0.009 ± 0.001 |
| Cd (µg/ g) | 0.03 ± 0.001 |
| Cs (µg/g) | 0.10 ± 0.01 |
| Li (µg/ g) | 0.88 ± 0.03 |
| Ni (µg/ g) | 0.37 ± 0.02 |
| Hg (µg/ g) | < LoD |
| Tl (ng/ g) | 6.0 ± 0.6 |
| Pb (µg/ g) | 0.103 ± 0.00 |
| Rb (µg/ g) | 11.83 ± 0.29 |
| Sb (µg/ g) | < LoD |
| Sr (µg/ g) | 4.9 ± 0.7 |
| Fatty acid | Structure | Relative percentage |
| Butyric | C4:0 | 0.0021 ± 0.001 |
| Hexanoic | C6:0 | 0.019 ± 0.003 |
| Octanoic | C8:0 | 0.030 ± 0.002 |
| Decanoic | C10:0 | 0.043 ± 0.001 |
| Lauric | C12:0 | 0.055 ± 0.004 |
| Myristic | C14:0 | 0.177 ± 0.004 |
| Pentadecanoic | C15:0 | 0.029 ± 0.003 |
| Palmitic | C16:0 | 9.5 ± 0.4 |
| Palmitoleic | C16:1 | 0.200 ± 0.002 |
| Heptadecanoic | C17:0 | 0.08 ± 0.01 |
| Stearic | C18:0 | 2.3 ± 0.1 |
| Oleic | C18:1n9c | 20 ± 1 |
| Linoleic1 | C18:2n6c1 | 37 ± 1 |
| Arachidic | C20:0 | 0.26 ± 0.01 |
| α-Linolenic1 | C18:3n31 | 30 ± 1 |
| cis-11-Eicosanoic | C20:1n9 | 0.19 ± 0.01 |
| Eicosatrienoic | C20:3n31 | 0.16 ± 0.01 |
| ΣSFA | 12.5 ± 0.4 | |
| ΣMUFA | 20 ± 1 | |
| ΣPUFA | 67 ± 1 |
| Vitamer | Content (mg/ 100 g) |
| α-tocopherol | 4.1 ± 0.5 |
| γ-tocopherol | 2.7 ± 0.3 |
| Total vitamin E | 6.8 ± 0.4 |
| Bioactive compounds | Hydroalcoholic extracts (mg/ g) |
| Total phenolics (TPC) | 91.0 ± 0.4 |
| Total flavonoids (TFC) | 52.1 ± 7.0 |
| Total saponins (TSC) | 63.4 ± 4.0 |
| Compound | Content (mg/ g extract) |
| Gallic acid | 1.37 ± 0.01 |
| Catechin | 0.88 ± 0.02 |
| Quercetin-3-O-rutinoside | 4.25 ± 0.05 |
| Quercetin | 18.5 ± 0.3 |
| Ellagic acid | 14.9 ± 0.5 |
| Analyte | Calibration equation | R2 | LOD (µg/L) |
LOQ (µg/L) |
Repeatability (% RSD) |
Intermediate precision (% RSD) |
| Ellagic acid | y = 257.3x + 0.07 | 0.9963 | 8.9 | 29.6 | 2.9 | 5.2 |
| Rutin | y = 37.0x + 0.06 | 0.9951 | 9.5 | 31.6 | 2.7 | 5.1 |
| Catechin | y = 31.9x + 0.01 | 0.9956 | 28.1 | 93.6 | 3.3 | 6.8 |
| Quercetin | y = 227.1x + 0.41 | 0.9986 | 23.3 | 77.6 | 2.5 | 3.1 |
| Gallic acid | y = 392.7x + 0.38 | 0.9922 | 46.8 | 155.8 | 4.7 | 7.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




