Submitted:
23 December 2025
Posted:
24 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
- Law of Dominance : When two different alleles are present in an organism, one may mask the expression of the other. The expressed trait is called dominant, while the hidden trait is recessive.
- Law of Segregation :An organism possesses two alleles for a particular trait, which segregate during gamete formation. Consequently, each gamete receives only one allele from each pair, Ensures genetic variation in the offspring.
- Law of Independent Assortment : The alleles of different genes are distributed to gametes independently of one another. Consequently, the inheritance of one trait generally does not influence the pattern of inheritance of another, given that the genes are on dissimilar chromosomes. or are far apart on the same chromosome.
2. Methodology
2.1. Basis of Population Growth
2.1.1. Initial Population Data
| RR | Homozygous dominant |
| Rr | Heterozygous |
| rr | Homozygous recessive |
2.1.2. Birth Rate (Natality)
- Fertility of the population: Every individual has a reproductive capacity determined by reproductive health. If a greater fraction of individuals are infertile, the birth rate per pair decreases.
- Resource availability: When a population demands more resources than the ecosystem can provide, resources are shared thinly. Individuals receive less than needed to remain healthy, indirectly lowering birth rate.
- Sex ratio: In populations where polygamy is common, a higher number of females allows growth even with fewer males (tending towards a 1:1 ratio). Where polygamy is not common, birth rates collapse if too few pairs can form.
2.1.3. Death Rate (Mortality)
- Resource consumption of the population: If the population demands more than the ecosystem can supply, health declines and mortality increases.
- Diseases: Diseases or genetic disorders can significantly increase mortality overall or for specific genotypes.
- Disasters: Stochastic disasters can cause sudden population declines.
2.1.4. Carrying Capacity (K)
2.1.5. Resource Consumption per Capita (RCPC)

2.2. Assumptions Taken for the Model
- Each Individual has one mate only.
- Generation n will have lower death rates than generation n-1.
- Generation n-2 will die out completely as they are too old to survive.
- Birth rate depends on how much resources are present per individual in the population after reaching carrying capacity it becomes constant.
- Death rate also varies with resource consumption per capita of the population.
- Infant mortality rates, deaths due to diseases, etc are all included in the in death rate of the population.
2.3. Algorithm
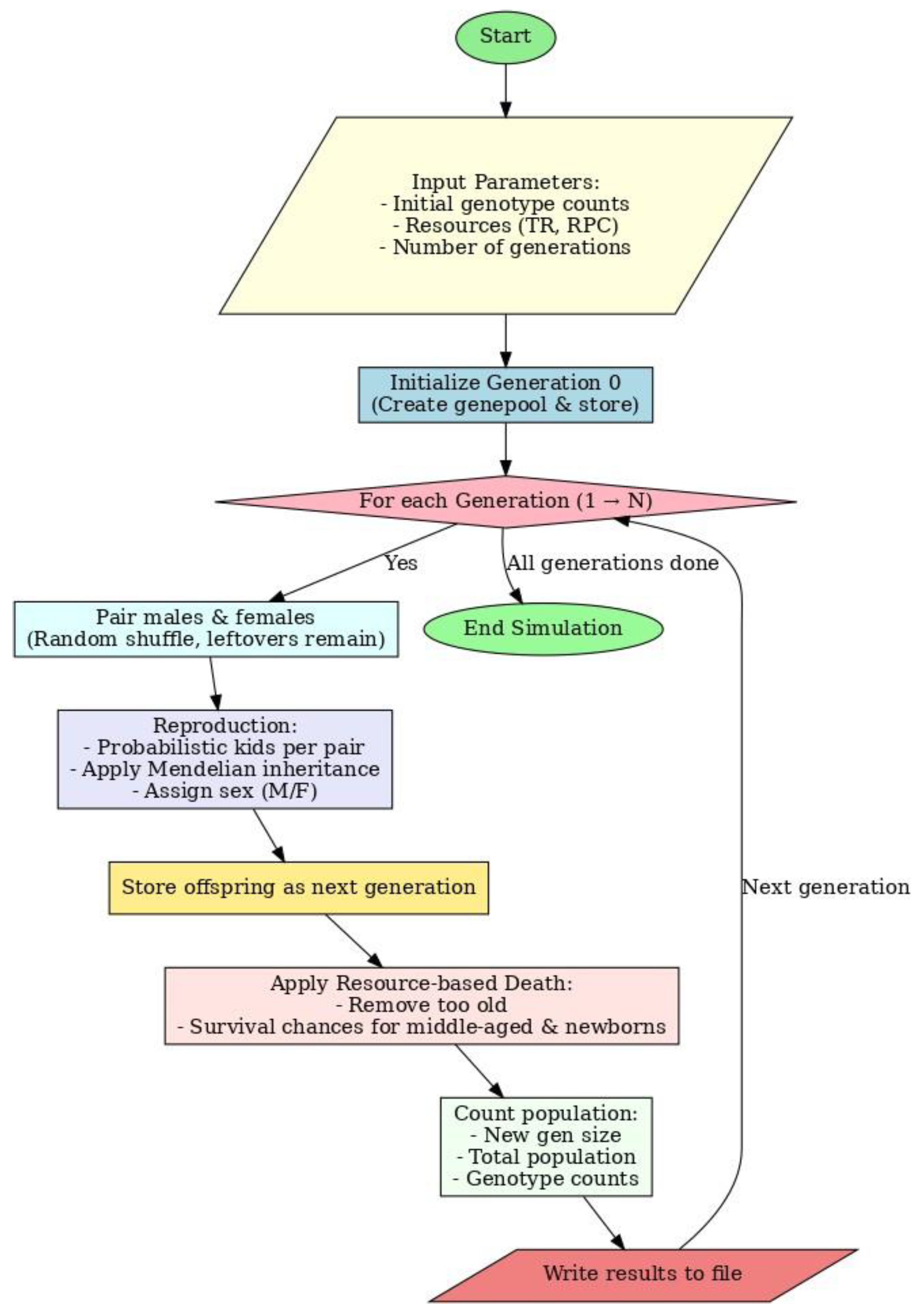
2.4. Input

2.5. Birth Probability Calculation
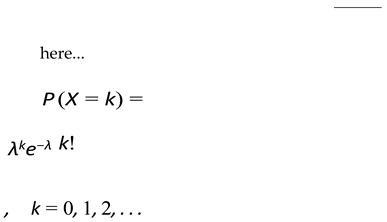

2.6. Pairing

2.7. Crossing

2.8. Updation

2.9. Death Rules
- Once reproduction is over, the population experiences mortality due to age and resource utilization.
- Old adults (generation n-2) are eliminated entirely (aging-related death).
- Middle-aged adults (generation n-1) survive probabilistically via the resource() function.
- Newborn adults (generation n) survive by the resource1() function, which provides them with slightly greater survival probabilities than old adults.

2.10. Resource Functions


3. Simulation Observations
3.1. Variation with Change in Birth Rates
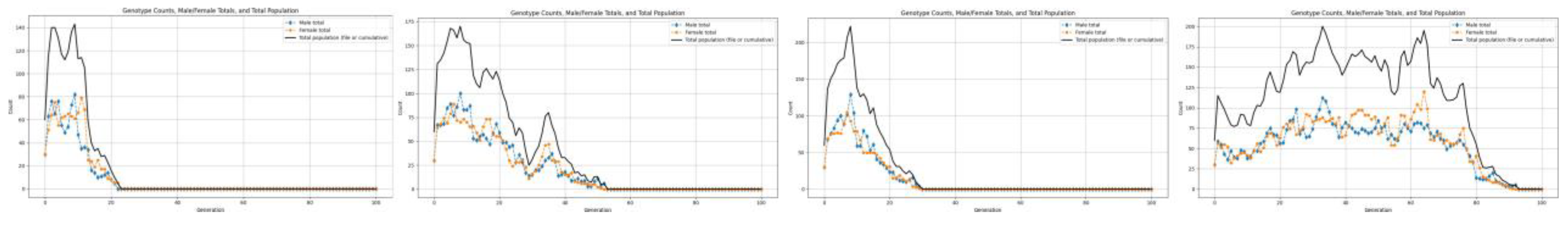
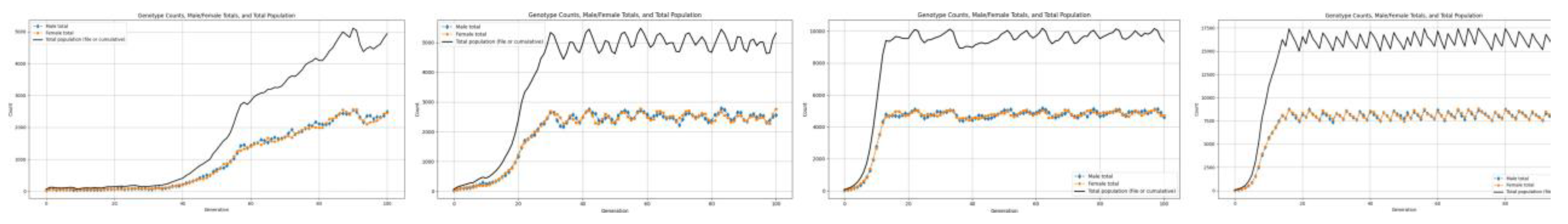
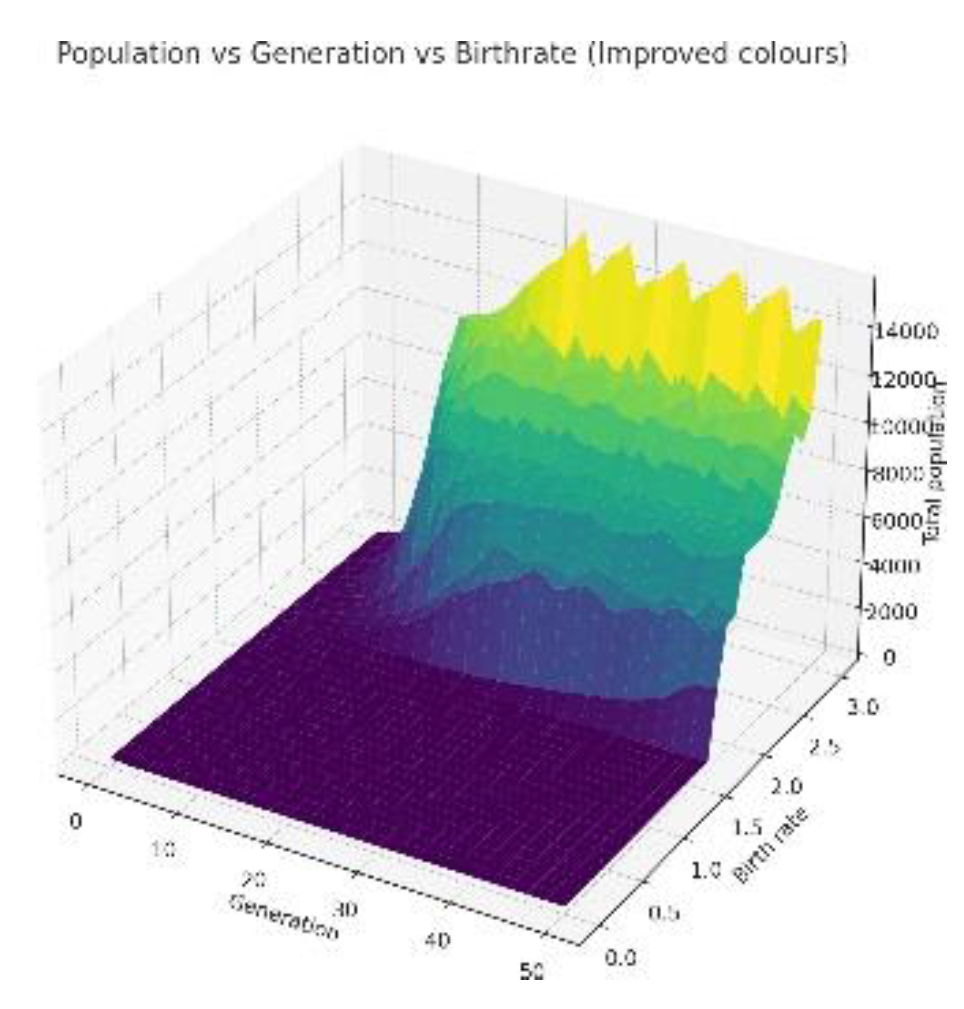
3.2. Low Resources
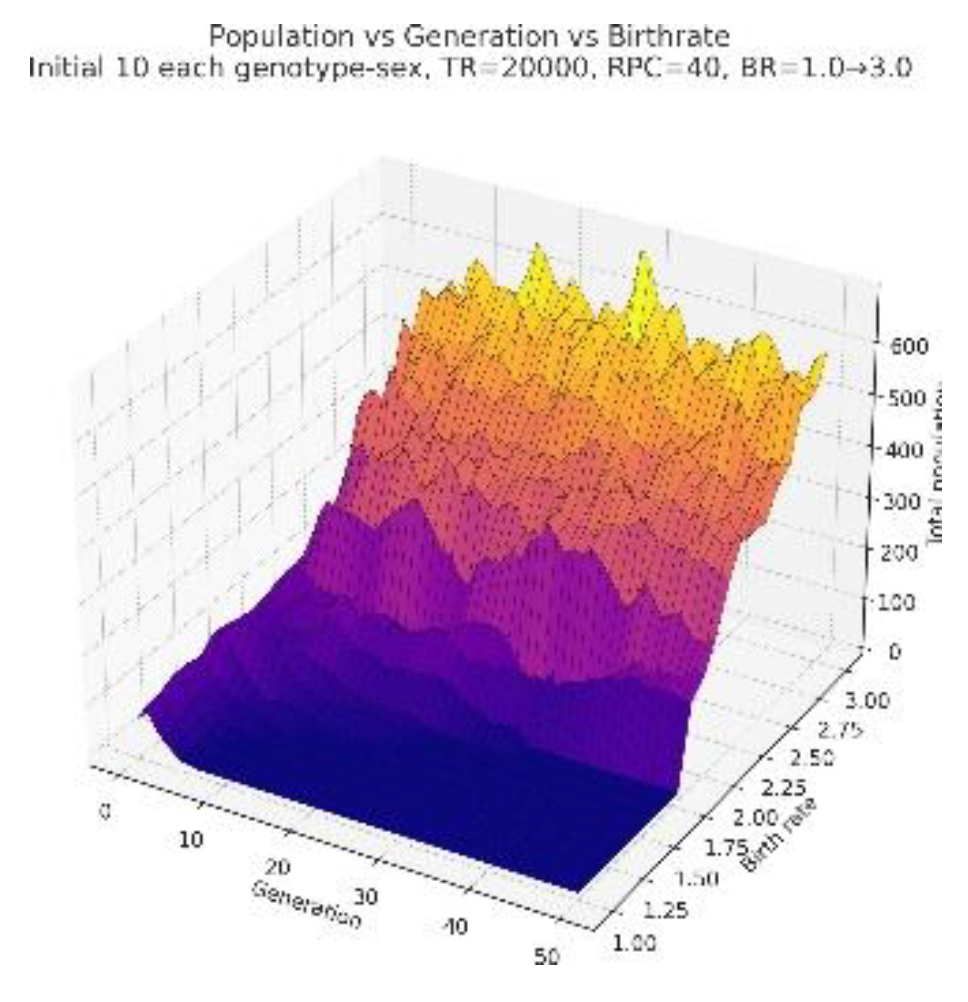
3.3. Genotypic Variation Observations
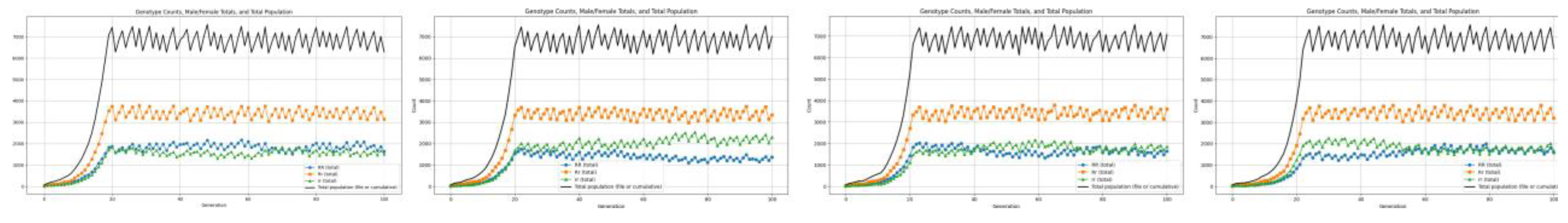
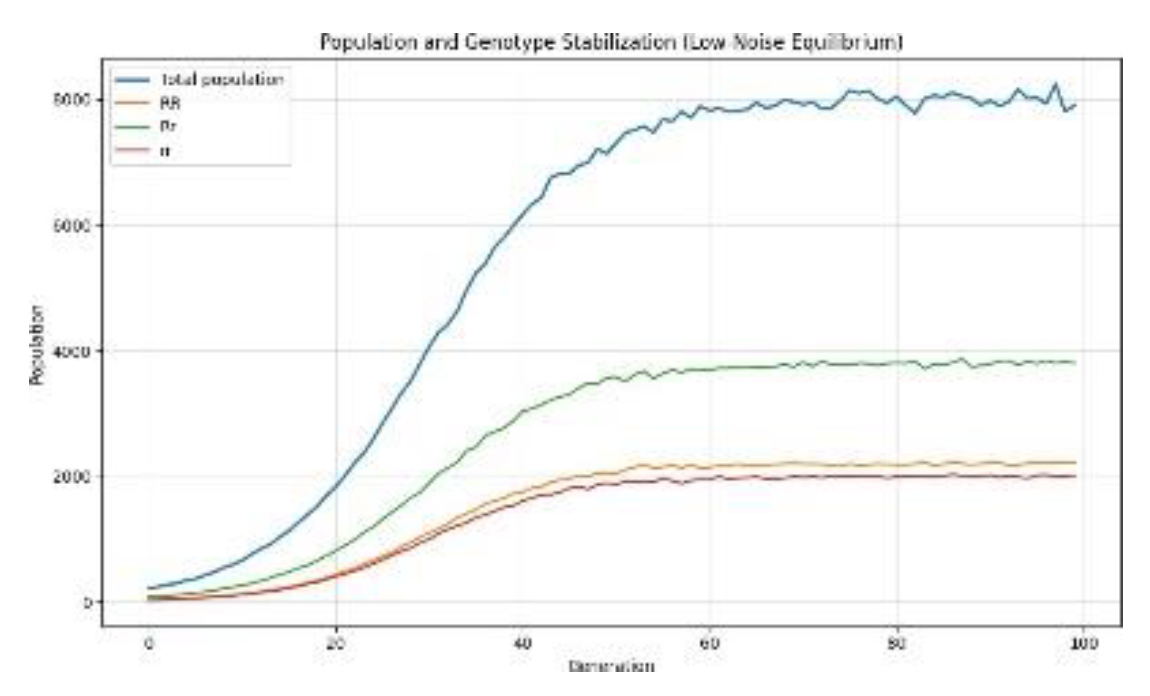
3.4. Defective Genotypes
3.4.1. Reproductive Disorder
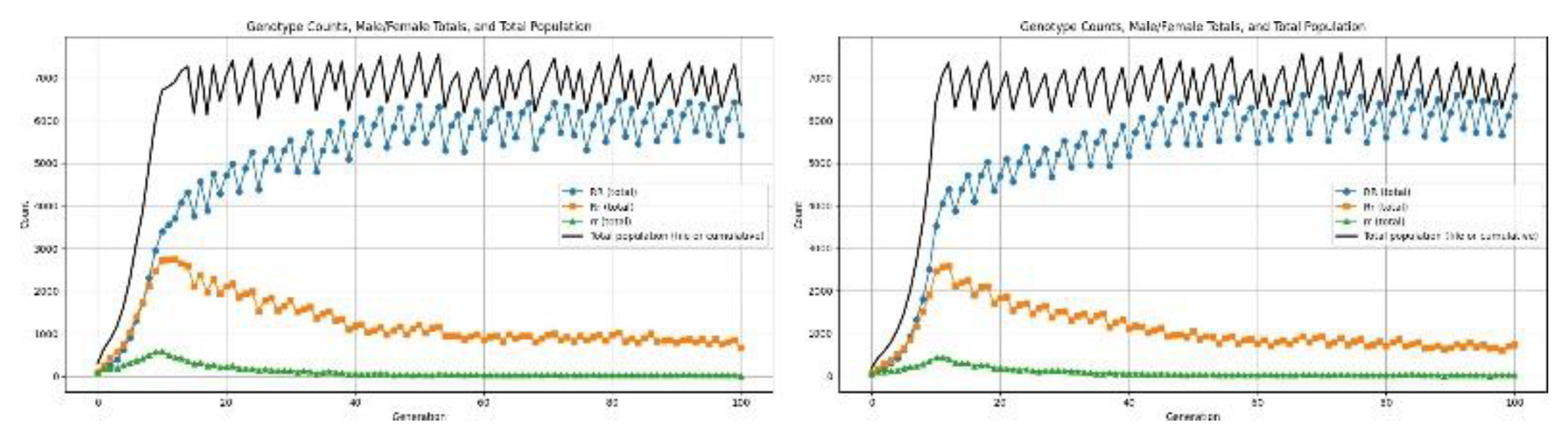
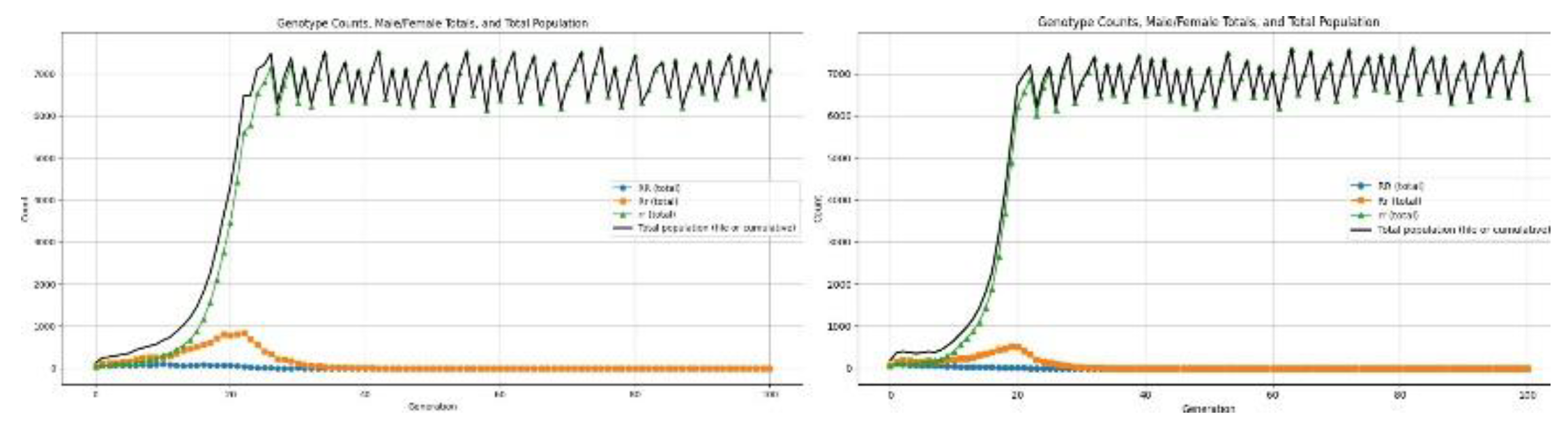
3.4.2. Health Disorders
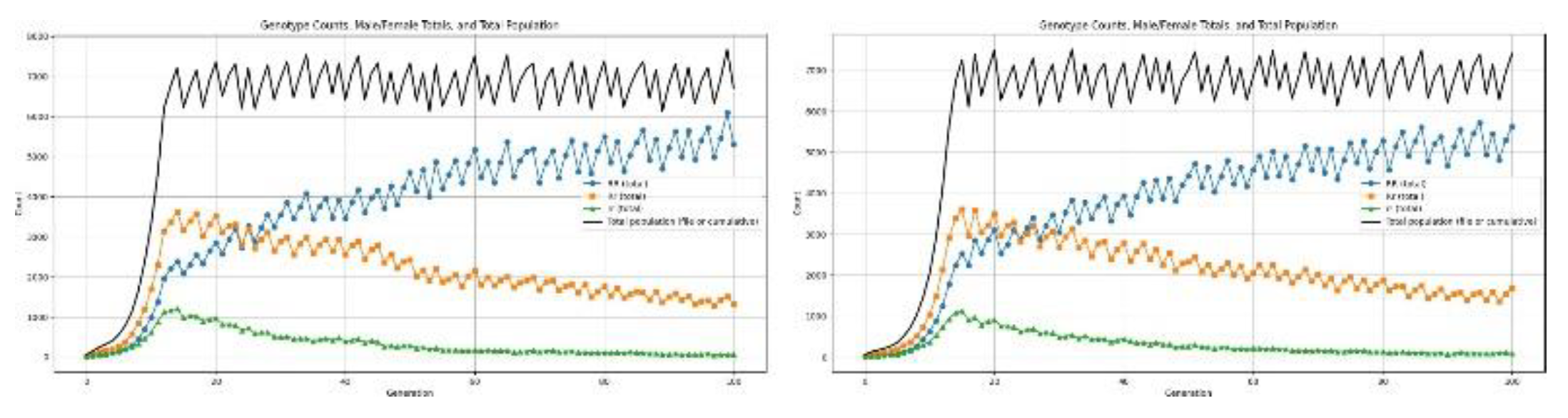
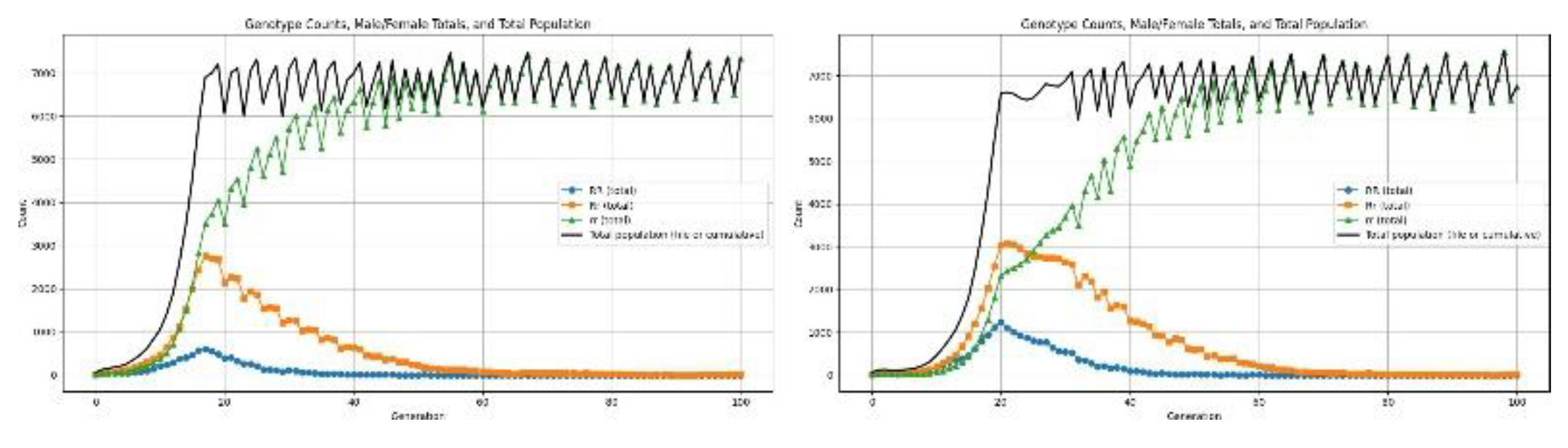
3.5. Disasters
3.5.1. Type-1 Disaster: Moderate Collapse
- -
- Multiyear regional droughts
- -
- Long-term crop failures resulting in chronic famine.
- -
- Prolonged disease endemics with moderate lethality—for example, historical waves of malaria or cholera.
- -
- Sustained environmental degradation: soil nutrient loss, habitat fragmentation Type-1 events mimic chronic but not catastrophic pressures—enough to significantly reshape population structure over many generations.
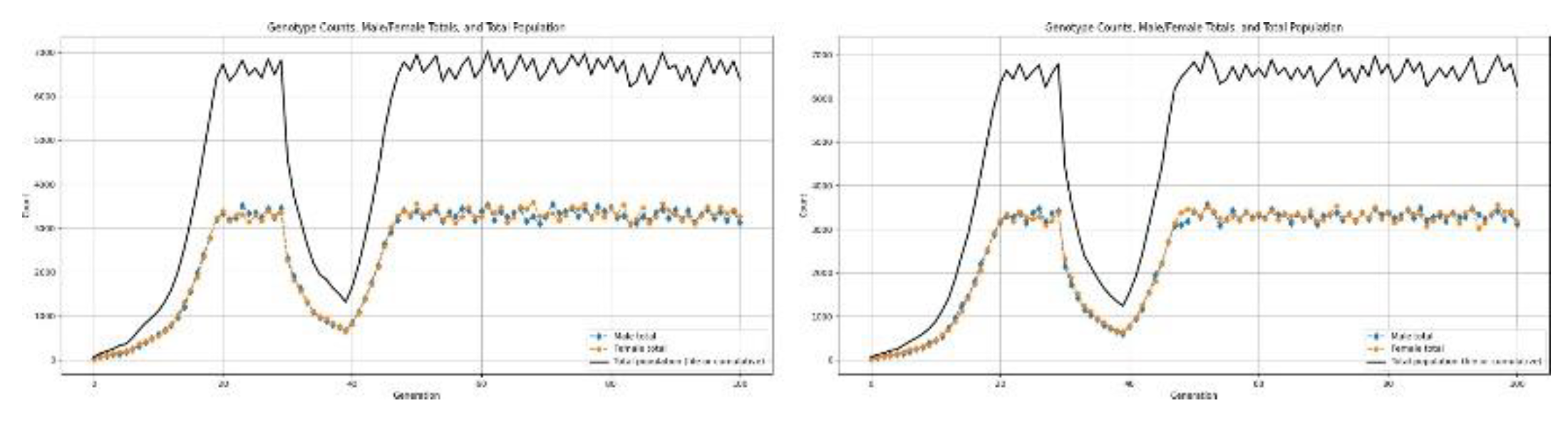
3.5.2. Type-2 Disaster: Severe Collapse
- -
- Sudden climate changes, such as rapid cooling or multicontinent monsoon failures
- -
- Highly lethal infectious disease outbreaks, such as smallpox or measles in immunolog- ically naïve populations
- -
- Large-scale wildfires or volcanic eruptions affecting multi-year resource availability
- -
- Oceanic or freshwater ecosystem crashes; i.e., fishery collapses, oxygen depletion events Type-2 disasters exert extreme selective pressure and can accelerate changes in allele frequencies.
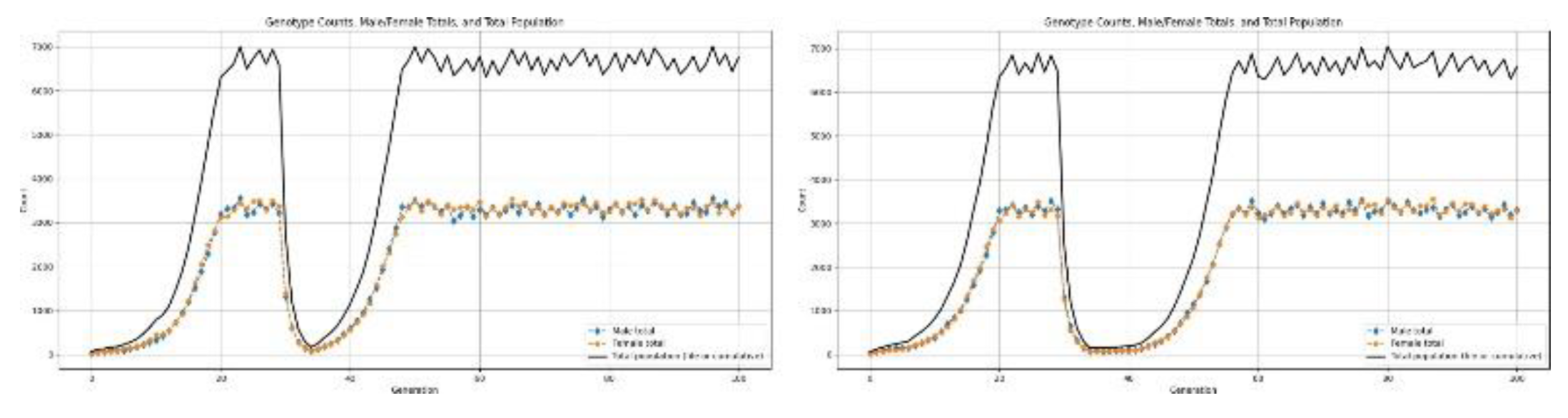
3.5.3. Type-3 Disaster: Catastrophic Collapse
- -
- Meteor impact or massive volcanic eruption causing near-instant ecosystem collapse
- -
- Super-virulent pandemics with extreme lethality
- -
- Acute toxic contamination events (Chemical spills, Radiation exposure)
- -
- Sudden habitat elimination: tsunami, large-scale glacier melt floods Type-3 disasters are near-extinction events and often determine whether a population survives at all.
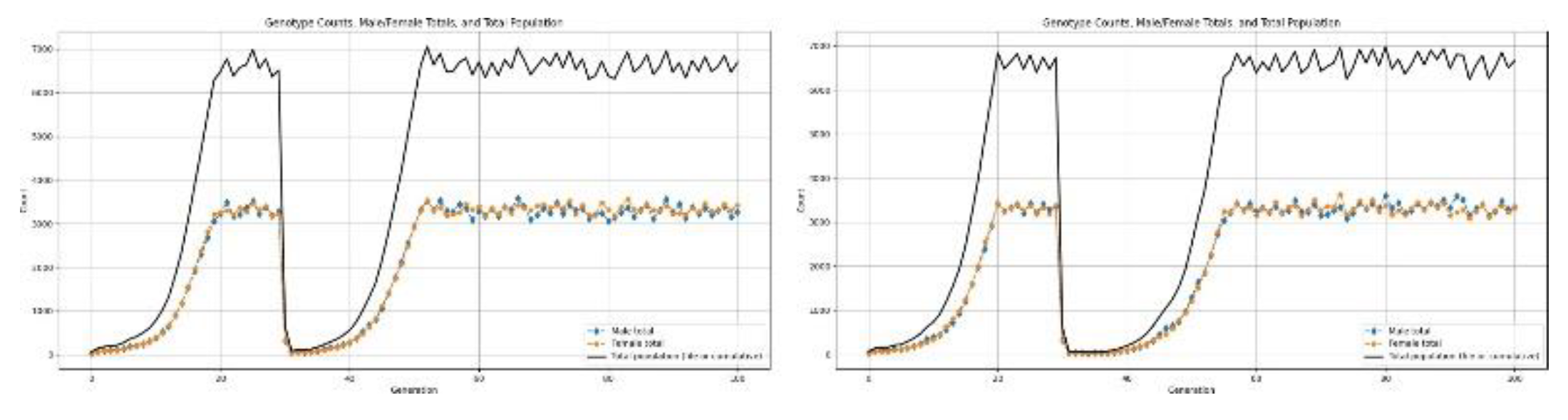
4. Results and Discussions
4.1. Population Collapse for Lower Birth Rates
- -
- Nt: Population size at generation t
- -
- N0: Initial population
- -
- TR: Total available resources
- -
- RPC: Resource consumption per capita
- -
- BR: Birth rate
- -
- µ(λ): Average number of children produced per pair
- -
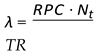 : Resource usage ratio
: Resource usage ratio
- Survival of existing individuals, given by probability Sold(λ)
- Survival of newborn individuals, given by probability Snew(λ)
- Average reproduction — each pair produces on average µ(λ) offspring. Since only half of the population can breed (assuming equal sexes), the fraction of new offspring relative to the total population is 1 .
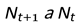
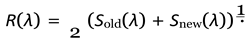
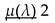
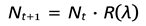
- -
- If R > 1, the population grows or survives.
- -
- If R < 1, the population shrinks and eventually collapses to extinction.
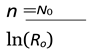
4.2. Stable Phase
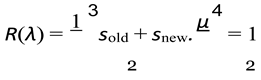
- -
- sold(λ) is a function that depends on λ and defines the survivability for older genera- tions.
- -
- snew(λ) is a function that depends on λ and defines the survivability for younger generations.
- -
- µ(λ) is a function that depends on λ and defines the birth rate of the population.
4.3. Birthrate as a Function of λ

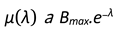
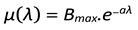
4.4. Behavior in Absence of Abundant Resources
4.5. Genotypic Analysis
- -
- p = frequency of the allele R.
- -
- q = frequency of the allele r.
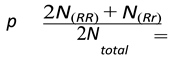
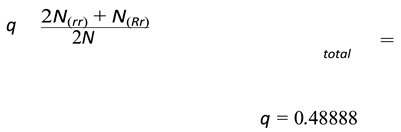
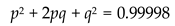
4.6. Genetic Disorders
4.6.1. Reproductive Recessive
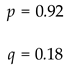
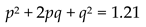
4.6.2. Reproductive Dominant
4.6.3. Health recessive
4.6.4. Health Dominant
4.7. Disasters
4.7.1. Type-1
4.7.2. Type-2
4.7.3. Type-3
4.8. Limitations
Acknowledgments
References
- Verhulst, P. F. (1838). Notice sur la loi que la population suit dans son accroissement. Correspondance Mathématique et Physique, 10, 113–121.
- May, R. M. (1976). Simple mathematical models with very complicated dynamics. Nature, 261 (5560), 459–467. https://doi.org/10.1038/261459a0. [CrossRef]
- Fowler, C. W. (1981). Density dependence as related to life history strategy. Ecology, 62 (3), 602–610. https://doi.org/10.2307/1938559. [CrossRef]
- Mendel, G. (1866). Experiments on plant hybridization. Verhandlungen des Natur- forschenden Vereins in Brünn, 4, 3–47.
- Hardy, G. H. (1908). Mendelian proportions in a mixed population. Science, 28 (706), 49–50. https://doi.org/10.1126/science.28.706.49. [CrossRef]
- Weinberg, W. (1908). Über den Nachweis der Vererbung beim Menschen. Jahreshefte des Vereins für Vaterländische Naturkunde in Württemberg, 64, 368–382.
- Kendall, D. G. (1949). Stochastic processes and population growth. Journal of the Royal Statistical Society: Series B, 11 (2), 230–282. https://doi.org/10.1111/j.2517- 6161.1949.tb00023.x. [CrossRef]
- Dennis, B., Munholland, P. L., & Scott, J. M. (1991). Estimation of growth and extinction parameters for endangered species. Ecological Monographs, 61 (2), 115–143. https://doi.org/10.2307/1943211. [CrossRef]
- Lande, R., Engen, S., & Sæther, B.-E. (2003). Stochastic population dynamics in ecology and conservation. Ecology, 84 (9), 2267–2274. https://doi.org/10.1890/02-0399. [CrossRef]
- Cohen, J. E. (1995). Population growth and Earth’s human carrying capacity. Science, 269 (5222), 341–346. https://doi.org/10.1126/science.269.5222.341. [CrossRef]
- Nei, M., Maruyama, T., & Chakraborty, R. (1975). The bottleneck effect and genetic variability. Evolution, 29 (1), 1–10. https://doi.org/10.1111/j.1558-5646.1975.tb00807.x. [CrossRef]
- Frankham, R. (1995). Effective population size/adult population size ratios in wildlife. Genetical Research, 66 (2), 95–107. https://doi.org/10.1017/S0016672300034455. [CrossRef]
- Lynch, M., & Lande, R. (1998). The critical effective size for a genetically se- cure population. Animal Conservation, 1 (1), 70–72. https://doi.org/10.1111/j.1469- 1795.1998.tb00228.x. [CrossRef]
- Blau, N., van Spronsen, F. J., & Levy, H. L. (2010). Phenylketonuria. The Lancet, 376 (9750), 1417–1427. https://doi.org/10.1016/S0140-6736(10)60961-0. [CrossRef]
- Seminara, S. B., et al. (1998). The GPR54 gene as a regulator of puberty. The New England Journal of Medicine, 349 (17), 1614–1627. https://doi.org/10.1056/NEJMoa035322. [CrossRef]
- Layman, L. C. (2013). The genetic basis of gonadotropin-releasing hormone deficiency. Nature Reviews Endocrinology, 9 (10), 569–576. https://doi.org/10.1038/nrendo.2013.147. [CrossRef]
- Allee, W. C. (1931). Animal aggregations. The American Naturalist, 65 (699), 191–204. https://doi.org/10.1086/280363. [CrossRef]
- Courchamp, F., Clutton-Brock, T., & Grenfell, B. (1999). Inverse density de- pendence and the Allee effect. Trends in Ecology & Evolution, 14 (10), 405–410. https://doi.org/10.1016/S0169-5347(99)01683-3. [CrossRef]
- Kimura, M. (1983). The neutral theory of molecular evolution. Nature, 302 (5903), 546–550. https://doi.org/10.1038/302546a0. [CrossRef]
- Brook, B. W., Traill, L. W., & Bradshaw, C. J. A. (2006). Minimum vi- able population sizes and global extinction risk. Science, 312 (5772), 397–400. https://doi.org/10.1126/science.1127855. [CrossRef]
- Tilman, D., Isbell, F., & Cowles, J. M. (2014). Biodiversity and ecosystem functioning. Annual Review of Ecology, Evolution, and Systematics, 45, 471–493. https://doi.org/10.1146/annurev-ecolsys-120213-091917. [CrossRef]
- Willi, Y., Van Buskirk, J., & Hoffmann, A. A. (2006). Limits to the adaptive potential of small populations. Annual Review of Ecology, Evolution, and Systematics, 37, 433–458. https://doi.org/10.1146/annurev.ecolsys.37.091305.110145. [CrossRef]
- Melbourne, B. A., & Hastings, A. (2008). Extinction risk depends strongly on factors contributing to stochasticity. Nature, 454 (7200), 100–103. https://doi.org/10.1038/nature06922. [CrossRef]
- Lee, R. (2003). The demographic transition. Journal of Economic Perspectives, 17 (4), 167–190. https://doi.org/10.1257/089533003772034943. [CrossRef]
- Bradshaw, C. J. A., & Brook, B. W. (2014). Human population reduction is not a quick fix. Proceedings of the National Academy of Sciences, 111 (46), 16610–16615. https://doi.org/10.1073/pnas.1410465111. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



