Submitted:
19 December 2025
Posted:
23 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
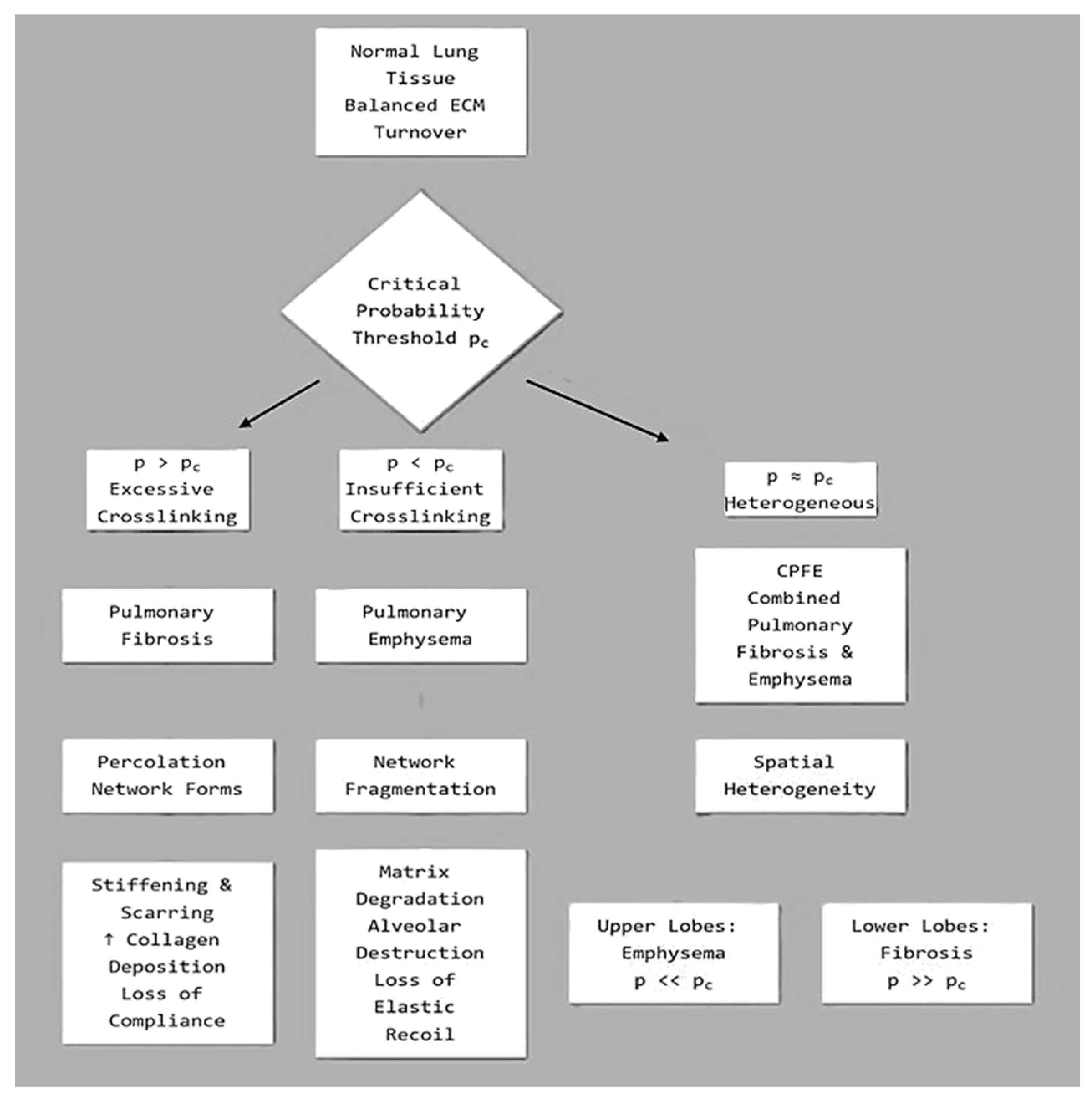
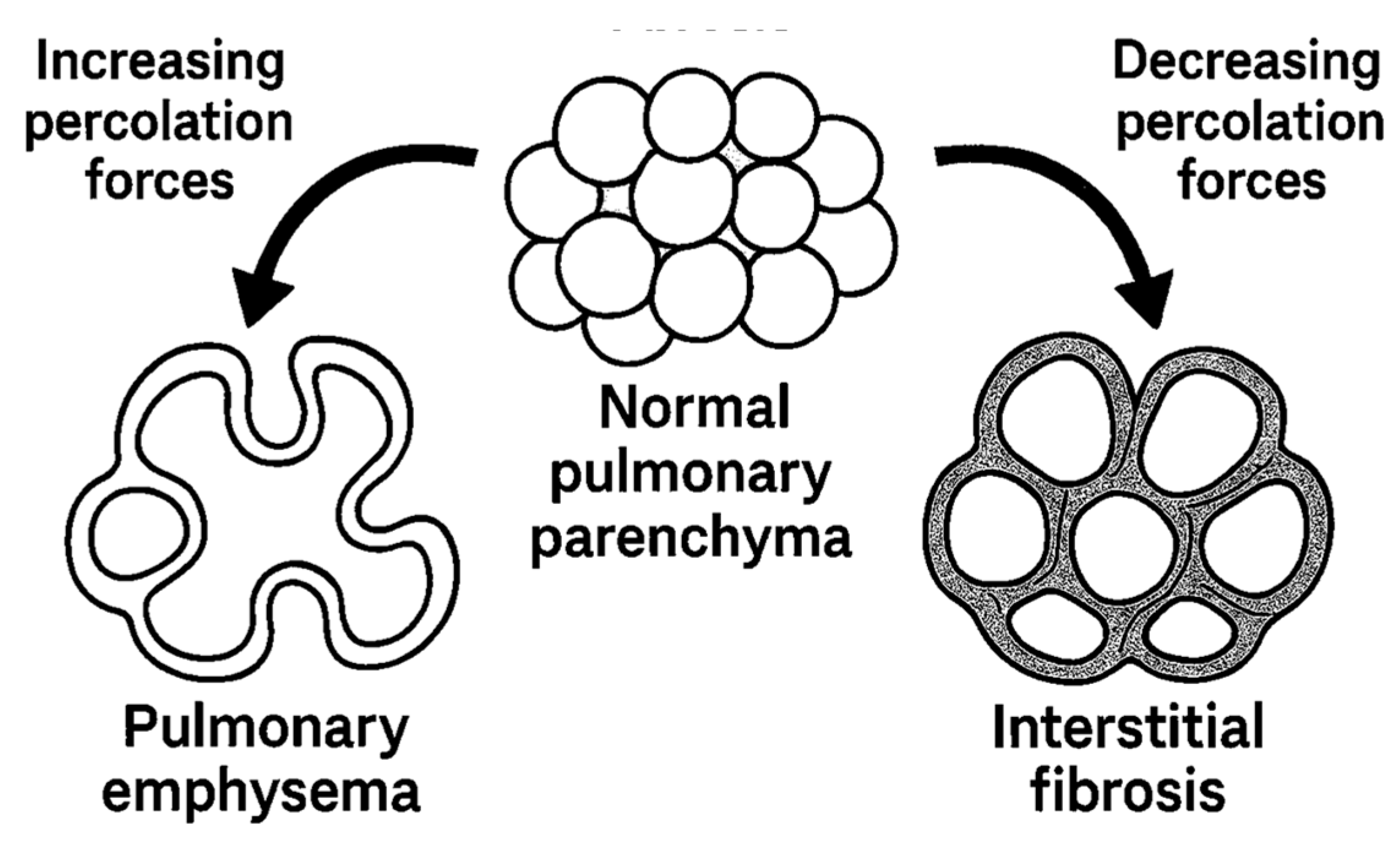
2. Percolation Theory and Biological Networks
3. Extracellular Matrix Crosslinking: Molecular Mechanisms and Types
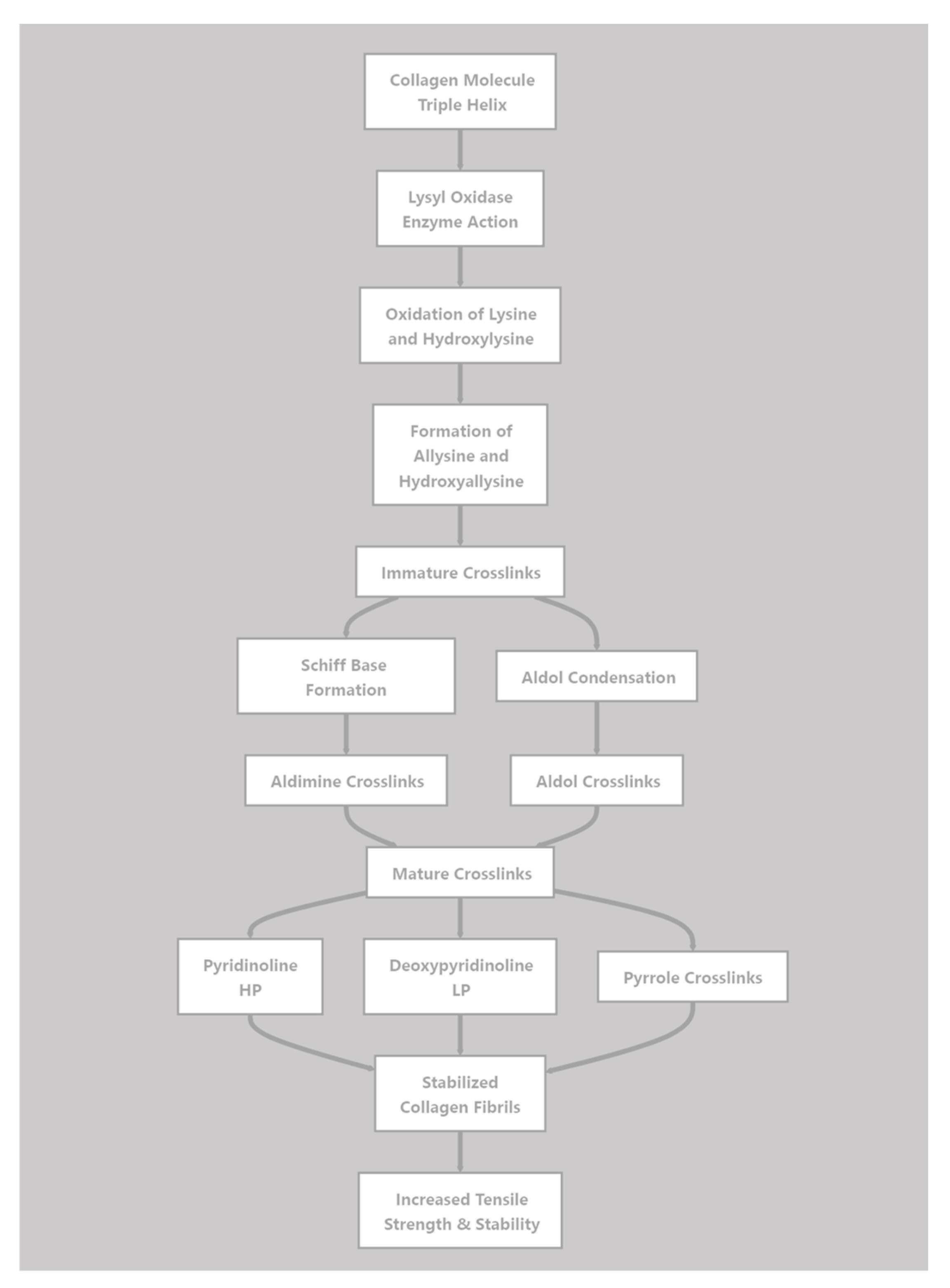
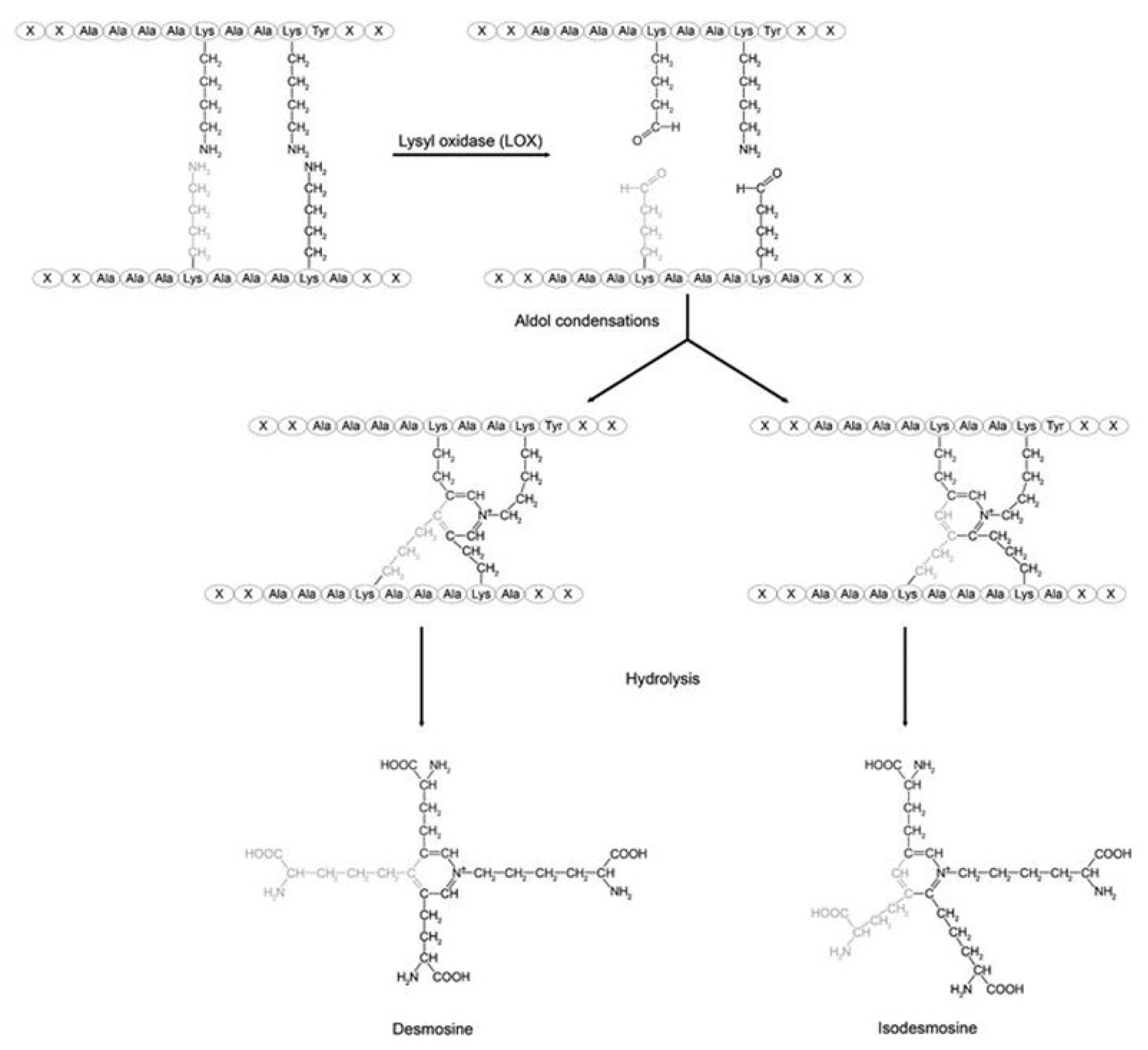
3.1. Enzymatic Crosslinking
3.2. Non-Enzymatic Crosslinking
3.3. Crosslink Dynamics and Tissue Mechanics
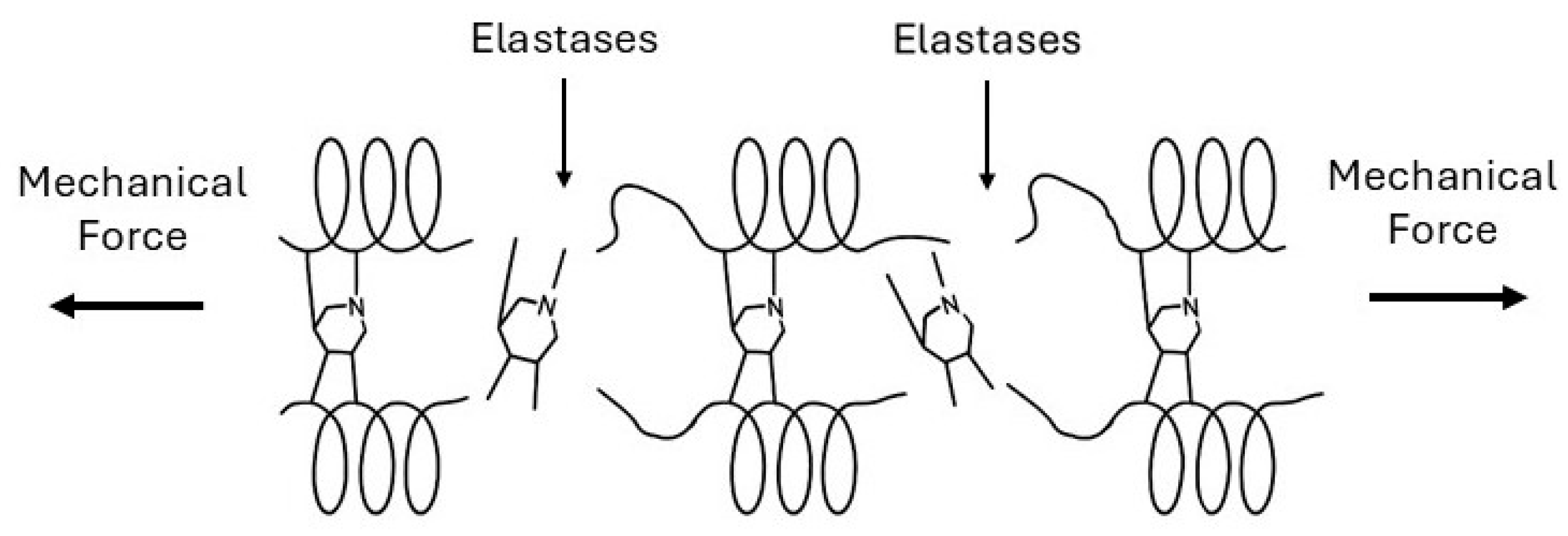
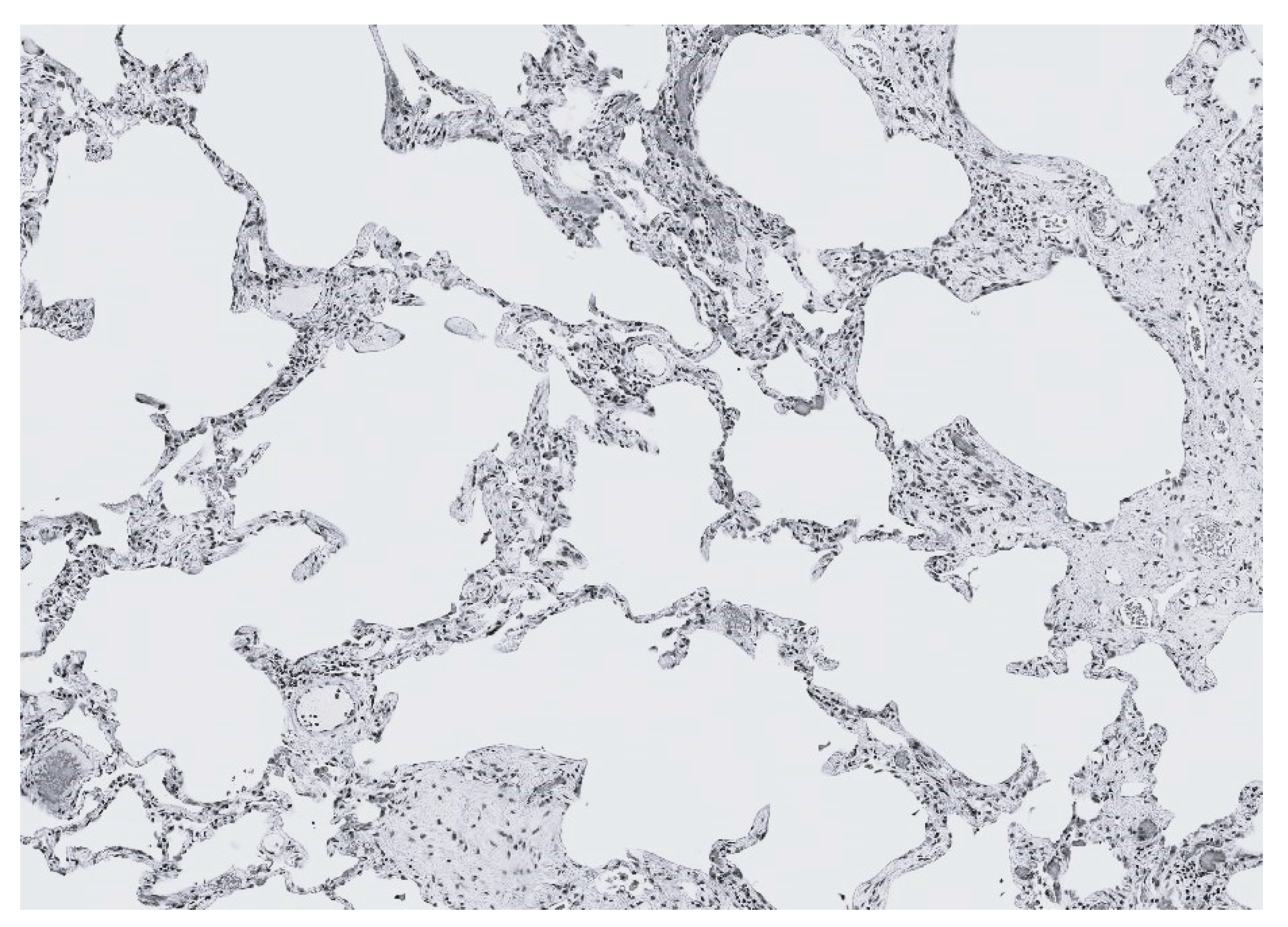
4. Emphysema: Degradative Percolation and Crosslink Loss
4.1. Initiation: Local Crosslink Disruption
4.2. Percolation Transition: Loss of Network Connectivity
4.3. Propagation: Mechanical Amplification
4.4. Crosslink Repair Failure
5. Fibrosis: Rigidity Percolation and Excessive Crosslinking
5.1. Initiation: Aberrant Repair and Crosslinking
5.2. Percolation Transition: Rigidity Threshold
5.3. Crosslink Types in Fibrosis
5.4. Propagation: Mechanical and Biochemical Feedback
6. Determinants of Pathway Selection: Why Emphysema or Fibrosis?
6.1. Balance of Proteolysis and Crosslinking
6.2. Matrix Composition and Pre-Existing Crosslink Density
6.3. Cell Populations and Inflammatory Milieu
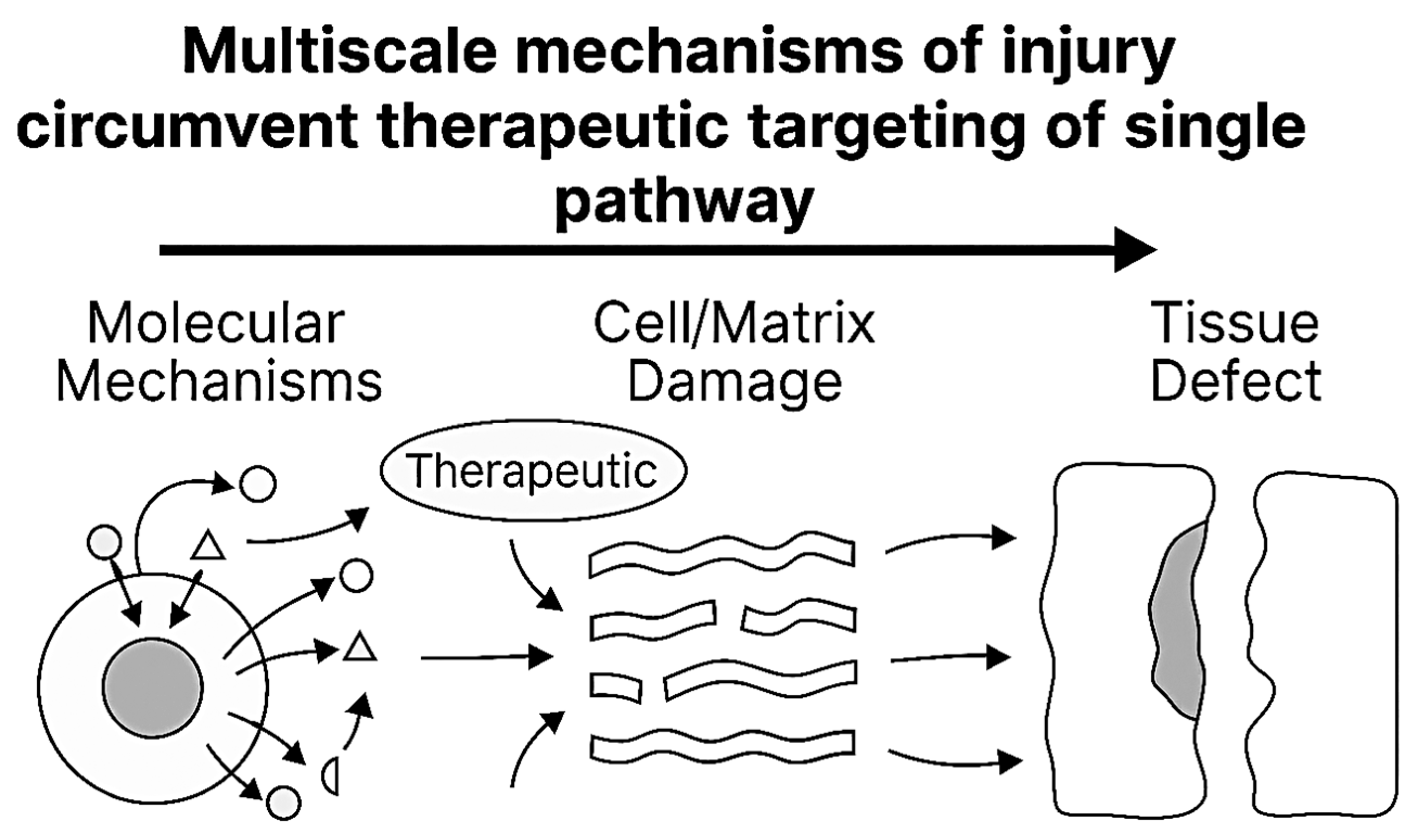
7. Therapeutic Implications: Targeting Crosslinking and Percolation
7.1. Emphysema: Preventing Degradative Percolation
7.2. Fibrosis: Preventing Rigidity Percolation
7.3. Therapeutic Limitations
8. Conclusions
References
- Medzhitov, R. The spectrum of inflammatory responses. Science 2021, 374, 1070–1075. [Google Scholar] [CrossRef]
- Gusev, E.; Zhuravleva, Y. Inflammation: A new look at an old problem. International Journal of Molecular Sciences 2022, 23, 4596. [Google Scholar] [CrossRef]
- Margraf, A.; Perretti, M. Immune cell plasticity in inflammation: Insights into description and regulation of immune cell phenotypes. Cells 2022, 11, 1824. Available online: https://www.mdpi.com/2073-4409/11/11/1824. [CrossRef]
- D’Souza, R.M.; Nagler, J. Anomalous critical and supercritical phenomena in explosive percolation. Nature Physics 2015, 11, 531–538. [Google Scholar] [CrossRef]
- Guo, T.; He, C.; Venado, A.; Zhou, Y. Extracellular matrix stiffness in lung health and disease. Comprehensive Physiology 2022, 12, 3523–3558. [Google Scholar] [CrossRef] [PubMed]
- Niewoehner, D.E.; Hoidal, J.R. Lung fibrosis and emphysema: Divergent responses to a common injury? Science 1982, 217, 359–360. [Google Scholar] [CrossRef] [PubMed]
- Sahimi, M. Percolation in Biological Systems. In Applications of Percolation Theory; Springer: Berlin/Heidelberg, Germany, 2023; Available online: https://link.springer.com/chapter/10.1007/978-3-031-20386-2_18.
- Mak, M. Impact of crosslink heterogeneity on extracellular matrix mechanics and remodeling. Comput. Struct. Biotechnol. J. 2020, 18, 3969–3976. [Google Scholar] [CrossRef]
- Vila Ellis, L.; Chen, J. A cell-centric view of lung alveologenesis. Developmental Dynamics 2021, 250, 482–496. [Google Scholar] [CrossRef]
- Burgess, J.K.; Harmsen, M.C. Chronic lung diseases: Entangled in extracellular matrix. European Respiratory Review 2022, 31. [Google Scholar] [CrossRef]
- Lloyd, S.M.; He, Y. Exploring Extracellular Matrix Crosslinking as a Therapeutic Approach to Fibrosis. Cells 2024, 13, 438. Available online: https://www.mdpi.com/2073-4409/13/5/438. [CrossRef]
- Levy, A.; Goldstein, H.; Brenman, D.; Diesendruck, C.E. Effect of intramolecular crosslinker properties on the mechanochemical fragmentation of covalently folded polymers. Journal of Polymer Science 2020, 58, 692–703. [Google Scholar] [CrossRef]
- Kong, W.; Lyu, C.; Liao, H.; Du, Y. Collagen crosslinking: Effect on structure, mechanics and fibrosis progression. Biomedical Materials 2021, 16, 062005. [Google Scholar] [CrossRef] [PubMed]
- Zaffryar-Eilot, S.; Hasson, P. Lysyl oxidases: Orchestrators of cellular behavior and ECM remodeling and homeostasis. International Journal of Molecular Sciences 2022, 23, 11378. [Google Scholar] [CrossRef] [PubMed]
- Schmelzer, C.E.; Hedtke, T.; Heinz, A. Unique molecular networks: Formation and role of elastin cross-links. Iubmb Life 2020, 72, 842–854. [Google Scholar] [CrossRef]
- Trębacz, H.; Barzycka, A. Mechanical properties and functions of elastin: An overview. Biomolecules 2023, 13, 574. [Google Scholar] [CrossRef]
- Soltani, F.; Kaartinen, M.T. Transglutaminases in fibrosis—Overview and recent advances. American Journal of Physiology-Cell Physiology 2023, 325, C1601–C1614. [Google Scholar] [CrossRef]
- Hitomi, K. Role of Transglutaminase 2 in Cell Death, Survival, and Fibrosis. Cells 2021, 10, 1842. [Google Scholar] [CrossRef] [PubMed]
- Gaar, J.; Naffa, R.; Brimble, M. Enzymatic and non-enzymatic crosslinks found in collagen and elastin and their chemical synthesis. Organic Chemistry Frontiers 2020, 7, 2789–2814. [Google Scholar] [CrossRef]
- Passarelli, M.; Machado, U.F. AGEs-induced and endoplasmic reticulum stress/inflammation-mediated regulation of GLUT4 expression and atherogenesis in diabetes mellitus. Cells 2021, 11, 104. [Google Scholar] [CrossRef]
- Kamml, J.; Ke, C.Y.; Acevedo, C.; Kammer, D.S. The influence of AGEs and enzymatic cross-links on the mechanical properties of collagen fibrils. J. Mech. Behav. Biomed. Mater. 2023, 143, 105870. [Google Scholar] [CrossRef]
- Cross, K.; Vetter, S.W.; Alam, Y.; Hasan, M.Z.; Nath, A.D.; Leclerc, E. Role of the receptor for advanced glycation end products (RAGE) and its ligands in inflammatory responses. Biomolecules 2024, 14, 1550. [Google Scholar] [CrossRef] [PubMed]
- Gatin, A.; Billault, I.; Duchambon, P.; Van der Rest, G.; Tabet, J.C. Oxidative radicals (HO• or N3•) induce several di-tyrosine bridge isomers at the protein scale. Free Radical Biology and Medicine 2021, 162, 1–10. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, H.; Song, L. Novel drug delivery systems targeting oxidative stress in chronic obstructive pulmonary disease: A review. Journal of nanobiotechnology 2020, 18, 145. [Google Scholar] [CrossRef]
- Fagiola, M.; Reznik, S.; Riaz, M.; Qyang, Y.; Lee, S.; Avella, J.; Turino, G.; Cantor, J. The relationship between elastin cross linking and alveolar wall rupture in human pulmonary emphysema. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2023, 324, L747–L755. [Google Scholar] [CrossRef]
- Obernolte, H.; Niehof, M.; Braubach, P.; Fieguth, H.G.; Jonigk, D.; Pfennig, O.; Tschernig, T.; Warnecke, G.; Braun, A.; Sewald, K. Cigarette smoke alters inflammatory genes and the extracellular matrix—Investigations on viable sections of peripheral human lungs. Cell and tissue research 2022, 387, 249–260. [Google Scholar] [CrossRef]
- Umeda, H.; Nakamura, F.; Suyama, K. Oxodesmosine and isooxodesmosine, candidates of oxidative metabolic intermediates of pyridinium cross-links in elastin. Arch Biochem Biophys. 2001, 385, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Casey, D.T.; Bou Jawde, S.; Herrmann, J.; Mori, V.; Mahoney, J.M.; Suki, B.; Bates, J.H. Percolation of collagen stress in a random network model of the alveolar wall. Scientific reports 2021, 11, 16654. [Google Scholar] [CrossRef]
- Depalle, B.; Qin, Z.; Shefelbine, S.J.; Buehler, M.J. Influence of cross-link structure, density and mechanical properties in the mesoscale deformation mechanisms of collagen fibrils. J. Mech. Behav. Biomed. Mater. 2015, 52, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Nelson, T.M.; Quiros, K.A.M.; Dominguez, E.C.; Ulu, A.; Nordgren, T.M.; Nair, M.G.; Eskandari, M. Healthy and diseased tensile mechanics of mouse lung parenchyma. Results in Engineering 2024, 22, 102169. [Google Scholar] [CrossRef]
- Burgstaller, G.; Oehrle, B.; Gerckens, M.; White, E.S.; Schiller, H.B.; Eickelberg, O. The instructive extracellular matrix of the lung: Basic composition and alterations in chronic lung disease. European Respiratory Journal 2017, 50. [Google Scholar] [CrossRef]
- Bhatt, S.P.; Bodduluri, S.; Reinhardt, J.M.; Nakhmani, A.; COPDGene Investigators. Mechanically Affected Lung and Progression of Emphysema. Am J Respir Crit Care Med. 2025, 211, 1409–1417. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chambers, R.C.; Mercer, P.F. Mechanisms of alveolar epithelial injury, repair, and fibrosis. Annals of the American Thoracic Society 2015, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tu, M.; Wei, T.; Jia, Y.; Wang, Y.; Wu, J. Molecular mechanisms of alveolar epithelial cell senescence and idiopathic pulmonary fibrosis: A narrative review. Journal of Thoracic Disease 2022, 15, 186. [Google Scholar] [CrossRef]
- Jiang, D.; Dey, T.; Liu, G. Recent developments in the pathobiology of lung myofibroblasts. Expert review of respiratory medicine 2021, 15, 239–247. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, A.; Yanagi, S.; Tsubouchi, H.; Miura, A.; Shigekusa, T.; Matsumoto, N.; Nakazato, M. Significance of nuclear upregulation of LOXL2 in fibroblasts and myofibroblasts in the fibrotic process of acute respiratory distress syndrome. 2020. [Google Scholar] [CrossRef]
- Andugulapati, S.B.; Gourishetti, K.; Tirunavalli, S.K.; Nayak, P.G. Biochanin-A ameliorates pulmonary fibrosis by suppressing the TGF-β mediated EMT, myofibroblasts differentiation and collagen deposition in in vitro and in vivo models. Phytomedicine 2020, 77, 153270. [Google Scholar] [CrossRef]
- Inui, N.; Sakai, S.; Kitagawa, M. Molecular pathogenesis of pulmonary fibrosis, with focus on pathways related to TGF-β and the ubiquitin-proteasome pathway. International journal of molecular sciences 2021, 22, 6107. [Google Scholar] [CrossRef]
- Nizamoglu, M.; de Hilster, R.H.; Zhao, F.; Sharma, P.K.; Borghuis, T.; Harmsen, M.C.; Burgess, J.K. An in vitro model of fibrosis using crosslinked native extracellular matrix-derived hydrogels to modulate biomechanics without changing composition. Acta biomaterialia 2022, 147, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Suki, B.; Bates, J.H. Pulmonary Fibrosis. In Mathematical Modeling of the Healthy and Diseased Lung: Linking Structure, Biomechanics, and Mechanobiology; Springer Nature: Cham, Switzerland, 2025; pp. 193–224. [Google Scholar]
- Tiskratok, W.; Chuinsiri, N.; Limraksasin, P.; Kyawsoewin, M.; Jitprasertwong, P. Extracellular Matrix Stiffness: Mechanotransduction and Mechanobiological Response-Driven Strategies for Biomedical Applications Targeting Fibroblast Inflammation. Polymers 2025, 17, 822. [Google Scholar] [CrossRef]
- Liu, L.; Yu, H.; Zhao, H.; Wu, Z.; Long, Y.; Zhang, J.; Du, Y. Matrix-transmitted paratensile signaling enables myofibroblast–fibroblast cross talk in fibrosis expansion. Proc. Natl. Acad. Sci. USA 2020, 117, 10832–10838. [Google Scholar] [CrossRef]
- Lu, J.; Wang, Z.; Zhang, L. Single-cell transcriptome analysis revealing mechanotransduction via the Hippo/YAP pathway in promoting fibroblast-to-myofibroblast transition and idiopathic pulmonary fibrosis development. Gene 2025, 943, 149271. [Google Scholar] [CrossRef]
- Yang, S.; Plotnikov, S.V. Mechanosensitive regulation of fibrosis. Cells 2021, 10, 994. [Google Scholar] [CrossRef]
- Aumiller, V.; Strobel, B.; Romeike, M.; Schuler, M.; Stierstorfer, B.E.; Kreuz, S. Comparative analysis of lysyl oxidase (like) family members in pulmonary fibrosis. Scientific reports 2017, 7, 149. [Google Scholar] [CrossRef]
- Lin, H.; Jiang, S. Combined pulmonary fibrosis and emphysema (CPFE): An entity different from emphysema or pulmonary fibrosis alone. J Thorac Dis. 2015, 7, 767–779. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cottin, V.; Nunes, H.; Brillet, P.; Delaval, P.; Devouassoux, G.; Tillie-Leblond, I.; Cordier, J.F. Combined pulmonary fibrosis and emphysema: A distinct underrecognised entity. European Respiratory Journal 2005, 26, 586–593. [Google Scholar] [CrossRef]
- Sicard, D.; Haak, A.J.; Choi, K.M.; Craig, A.R.; Fredenburgh, L.E.; Tschumperlin, D.J. Aging and anatomical variations in lung tissue stiffness. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2018, 314, L946–L955. [Google Scholar] [CrossRef] [PubMed]
- Mu, Q.; Wang, Q.; Yang, Y.; Wei, G.; Wang, H.; Li, Y. HMGB1 promotes M1 polarization of macrophages and induces COPD inflammation. Cell Biology International 2025, 49, 100–110. [Google Scholar] [CrossRef]
- Kishore, A.; Petrek, M. Roles of macrophage polarization and macrophage-derived miRNAs in pulmonary fibrosis. Frontiers in immunology 2021, 12, 678457. [Google Scholar] [CrossRef] [PubMed]
- Astori, E.; Garavaglia, M.L.; Colombo, G.; Landoni, L.; Portinaro, N.M.; Milzani, A.; Dalle-Donne, I. Antioxidants in smokers. Nutrition Research Reviews 2022, 35, 70–97. [Google Scholar] [CrossRef]
- Christopoulou, M.E.; Papakonstantinou, E.; Stolz, D. Matrix metalloproteinases in chronic obstructive pulmonary disease. Int. J. Mol. Sci. 2023, 24, 3786. [Google Scholar] [CrossRef]
- Cabral-Pacheco, G.A.; Garza-Veloz, I.; Castruita-De la Rosa, C.; Ramirez-Acuña, J.M.; Perez-Romero, B.A.; Guerrero-Rodriguez, J.F.; Martinez-Avila, N.; Martinez-Fierro, M.L. The roles of matrix metalloproteinases and their inhibitors in human diseases. Int. J. Mol. Sci. 2020, 21, 9739. [Google Scholar] [CrossRef]
- Khedoe, P.P.P.; Wu, X.; Gosens, R.; Hiemstra, P.S. Repairing damaged lungs using regenerative therapy. Current Opinion in Pharmacology 2021, 59, 85–94. [Google Scholar] [CrossRef]
- Wang, L.; Feng, M.; Zhao, Y.; Chen, B.; Zhao, Y.; Dai, J. Biomimetic scaffold-based stem cell transplantation promotes lung regeneration. Bioengineering & Translational Medicine 2023, 8, e10535. [Google Scholar]
- Chen, L.; Li, S.; Li, W. LOX/LOXL in pulmonary fibrosis: Potential therapeutic targets. Journal of drug targeting 2019, 27, 790–796. [Google Scholar] [CrossRef]
- Findlay, A.; Turner, C.; Schilter, H.; Deodhar, M.; Zhou, W.; Perryman, L.; Foot, J.; Zahoor, A.; Yao, Y.; Hamilton, R.; et al. An activity-based bioprobe differentiates a novel small molecule inhibitor from a LOXL2 antibody and provides renewed promise for anti-fibrotic therapeutic strategies. Clin Transl Med 2021, 11, e572. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Raghu, G.; Brown, K.K.; Collard, H.R.; Cottin, V.; Gibson, K.F.; Kaner, R.J.; Lederer, D.J.; Martinez, F.J.; Noble, P.W.; Song, J.W.; et al. Efficacy of simtuzumab versus placebo in patients with idiopathic pulmonary fibrosis: A randomised, double-blind, controlled, phase 2 trial. Lancet Respir Med 2017, 5, 22–32. [Google Scholar] [CrossRef]
- Fell, S.; Wang, Z.; Blanchard, A.; Nanthakumar, C.; Griffin, M. Transglutaminase 2: A novel therapeutic target for idiopathic pulmonary fibrosis using selective small molecule inhibitors. Amino Acids 2021, 53, 257–272. [Google Scholar] [CrossRef]
- Schultz, C. Targeting the extracellular matrix for delivery of bioactive molecules to sites of arthritis. British journal of pharmacology 2019, 176, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Sourris, K.C.; Watson, A.; Jandeleit-Dahm, K. Inhibitors of advanced glycation end product (AGE) formation and accumulation. In Reactive Oxygen Species: Network Pharmacology and Therapeutic Applications; Springer International Publishing: Cham, 2020; pp. 395–423. [Google Scholar]
- Ruwanpura, S.M.; Thomas, B.J.; Bardin, P.G. Pirfenidone: Molecular mechanisms and potential clinical applications in lung disease. Am. J. Respir. Cell Mol. Biol. 2020, 62, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Papavassiliou, K.A.; Sofianidi, A.A.; Spiliopoulos, F.G.; Gogou, V.A.; Gargalionis, A.N.; Papavassiliou, A.G. YAP/TAZ signaling in the pathobiology of pulmonary fibrosis. Cells 2024, 13, 1519. [Google Scholar] [CrossRef] [PubMed]
- Leslie, M.N.; Chou, J.; Young, P.M.; Traini, D.; Bradbury, P.; Ong, H.X. How do mechanics guide fibroblast activity? complex disruptions during emphysema shape cellular responses and limit research. Bioengineering 2021, 8, 110. [Google Scholar] [CrossRef] [PubMed]
- Cantor, J. Desmosine: The Rationale for Its Use as a Biomarker of Therapeutic Efficacy in the Treatment of Pulmonary Emphysema. Diagnostics 2025, 15, 578. [Google Scholar] [CrossRef]
- Stankovic, S.; Stjepanovic, M.; Asanin, M. Biomarkers in Idiopathic Pulmonary Fibrosis. In Idiopathic Pulmonary Fibrosis; IntechOpen: London, UK, 2021. [Google Scholar]
- Sack, I. Magnetic resonance elastography from fundamental soft-tissue mechanics to diagnostic imaging. Nature Reviews Physics 2023, 5, 25–42. [Google Scholar] [CrossRef]
- Suki, B.; Majumdar, A.; Nugent, M.A.; Bates, J.H.T. In silico modeling of interstitial lung mechanics: Implications for disease development and repair. Drug Discov. Today Dis. Mech. 2007, 4, 23–28. [Google Scholar] [CrossRef]
- Neelakantan, S.; Xin, Y.; Gaver, D.P.; Cereda, M.; Rizi, R.; Smith, B.J.; Avazmohammadi, R. Computational lung modelling in respiratory medicine. Journal of The Royal Society Interface 2022, 19, 20220062. [Google Scholar] [CrossRef]
- Bentaher, A.; Glehen, O.; Degobert, G. Pulmonary Emphysema: Current Understanding of Disease Pathogenesis and Therapeutic Approaches. Biomedicines 2025, 13, 2120. [Google Scholar] [CrossRef] [PubMed]
- Doroudian, M.; O’Neill, A.; Mac Loughlin, R.; Prina-Mello, A.; Volkov, Y.; Donnelly, S.C. Nanotechnology in pulmonary medicine. Current opinion in pharmacology 2021, 56, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Duan, X.P.; Qin, B.D.; Jiao, X.D.; Liu, K.; Wang, Z.; Zang, Y.S. New clinical trial design in precision medicine: Discovery, development and direction. Signal Transduction and Targeted Therapy 2024, 9, 57. [Google Scholar] [CrossRef]
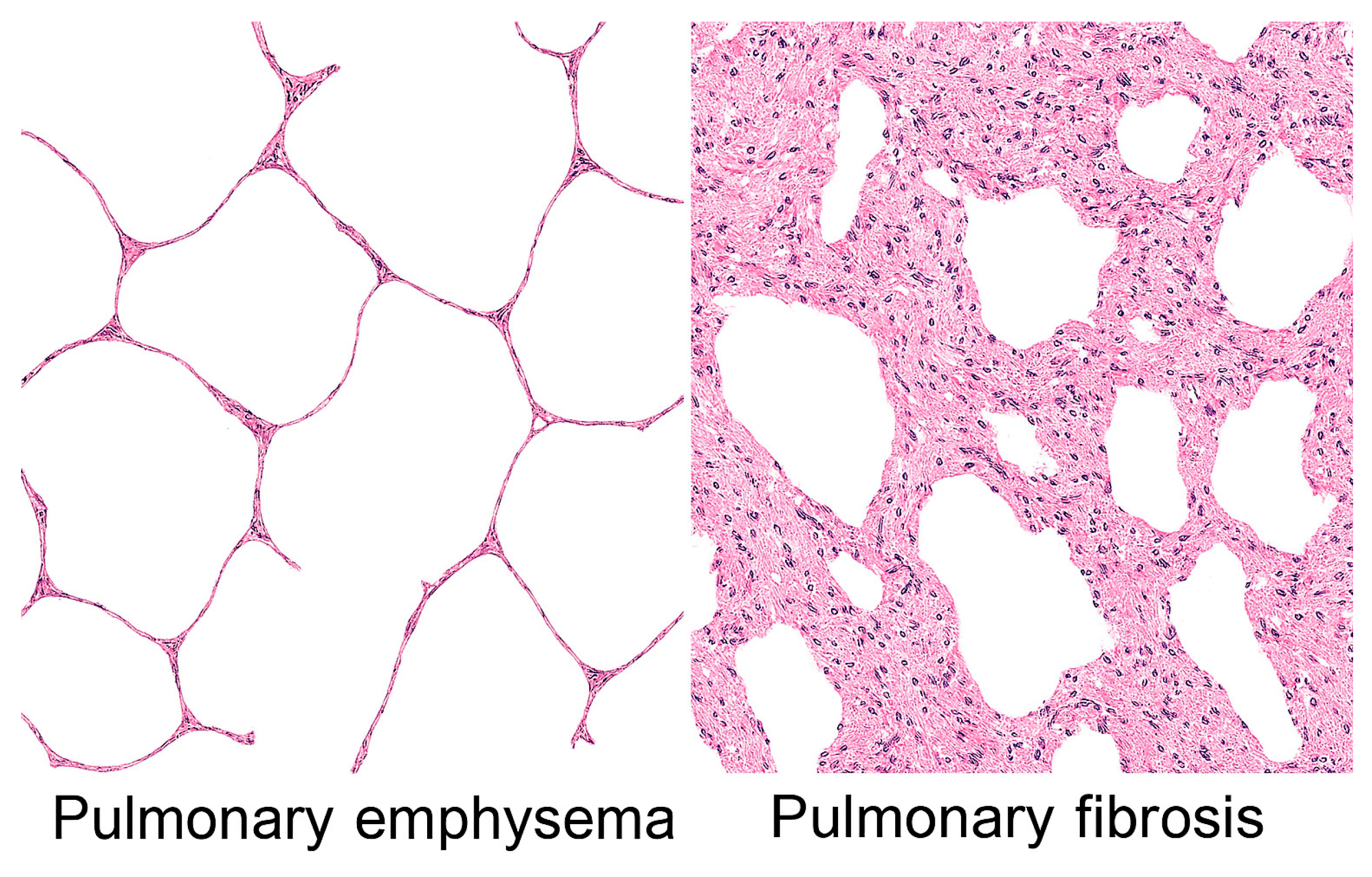


| Scale / Feature | Pulmonary Emphysema | Interstitial Fibrosis |
|---|---|---|
| Molecular Scale | ||
| Primary Process | Proteolytic degradation of ECM | Excessive ECM synthesis and deposition |
| Key Molecules | Matrix metalloproteinases (MMPs), elastases, oxidants | TGF-β, collagen I/III, fibronectin, α-SMA |
| Elastin Status | Degraded and fragmented | Preserved or increased |
| Collagen Content | Decreased or normal | Markedly increased (2-5x normal) |
| Cellular Scale | ||
| Cell Types Involved | Neutrophils, macrophages, inflammatory cells | Myofibroblasts, fibroblasts, epithelial cells |
| Cellular Response | Inflammation-driven destruction | Aberrant wound healing response |
| Alveolar Epithelium | Loss of capillary-alveolar interface | Type II pneumocyte hyperplasia, epithelial injury |
| Tissue Scale (Microscopic) | ||
| Alveolar Structure | Enlarged airspaces, wall destruction | Collapsed/obliterated airspaces, thickened walls |
| Interstitium | Thinned or absent | Markedly thickened with scar tissue |
| Architecture Pattern | Loss of normal architecture | Honeycombing, architectural distortion |
| Crosslink Percolation | Below threshold: isolated clusters | Above threshold: spanning network |
| Mechanical Properties | ||
| Tissue Compliance | Increased (hyperelastic, floppy) | Decreased (stiff, rigid) |
| Elastic Recoil | Reduced (loss of elastic fibers) | Reduced (increased stiffness) |
| Stress Distribution | Heterogeneous, stress concentrations | Transmitted through percolated network |
| Organ Scale (Macroscopic) | ||
| Lung Volume | Increased (hyperinflation) | Decreased (restrictive pattern) |
| CT Appearance | Low attenuation areas, bullae | Reticular opacities, ground-glass, honeycombing |
| Distribution | Upper lobe predominant (typically) | Lower lobe and peripheral predominant |
| Surface Appearance | Overinflated, pale, bullous | Shrunken, firm, nodular |
| Functional/Clinical Scale | ||
| Primary Defect | Obstructive (air trapping) | Restrictive (reduced expansion) |
| FEV1/FVC Ratio | Decreased (<0.70) | Normal or increased |
| DLCO | Decreased (loss of surface area) | Decreased (thickened membrane) |
| Gas Exchange | Impaired (V/Q mismatch) | Impaired (diffusion limitation) |
| Breathlessness Pattern | Progressive with air trapping | Rapid, shallow breathing |
| Reversibility | Largely irreversible structural damage | Progressive and irreversible scar |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




