Submitted:
19 December 2025
Posted:
22 December 2025
You are already at the latest version
Abstract
Gynura segetum, a traditional Chinese medicinal herb, has been increasingly recognized for its hepatotoxic potential due to its content of PAs. These compounds are among the most common causes of HILI in China and are strongly associated with HSOS. This review systematically explores the pathogenesis, diagnostic evolution, and therapeutic strategies of PAs-induced HSOS. We detail the molecular mechanisms underlying PA metabolism, including cytochrome P450-mediated bioactivation and the formation of pyrrole–protein adducts, which initiate sinusoidal endothelial cell injury and hepatocyte apoptosis. Advances in diagnostic criteria, such as the Nanjing Criteria and Drum Tower Severity Scoring System, are discussed alongside emerging biomarkers like circulating microRNAs and pyrrole–protein adducts. Imaging modalities, including contrast-enhanced CT and Gd-EOB-DTPA MRI, have transitioned from descriptive tools to quantitative and prognostic instruments. Therapeutic approaches have evolved from supportive care to precision interventions, including anticoagulation, TIPS, and autophagy-modulating agents. This review highlights the need for integrated diagnostic and therapeutic frameworks and calls for enhanced public awareness and regulatory oversight to mitigate PAs-related liver injury.
Keywords:
1. Introduction
2. Food and Pharmaceutical Safety Recommendations Regarding PAs
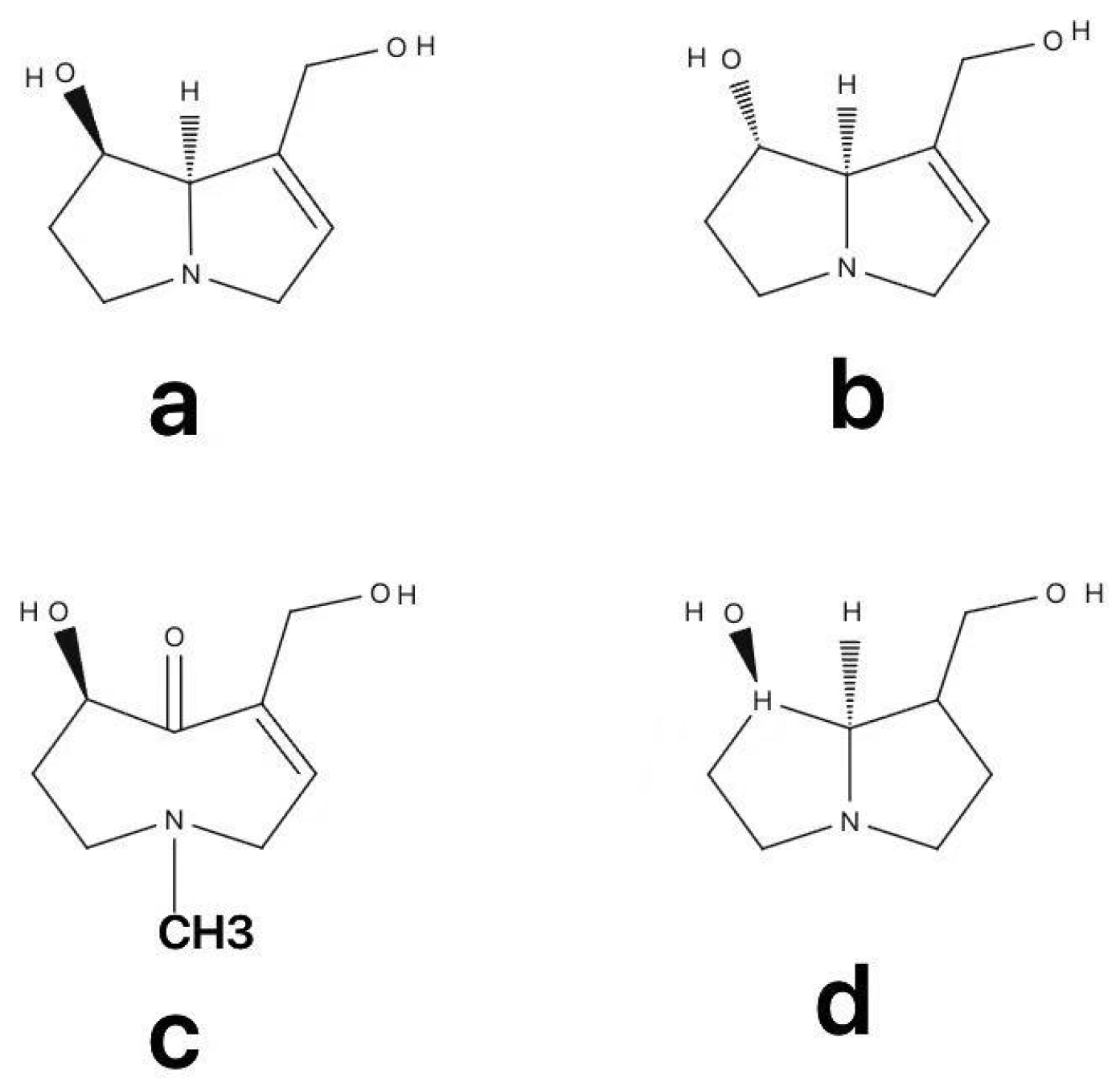
3. Metabolism of PAs
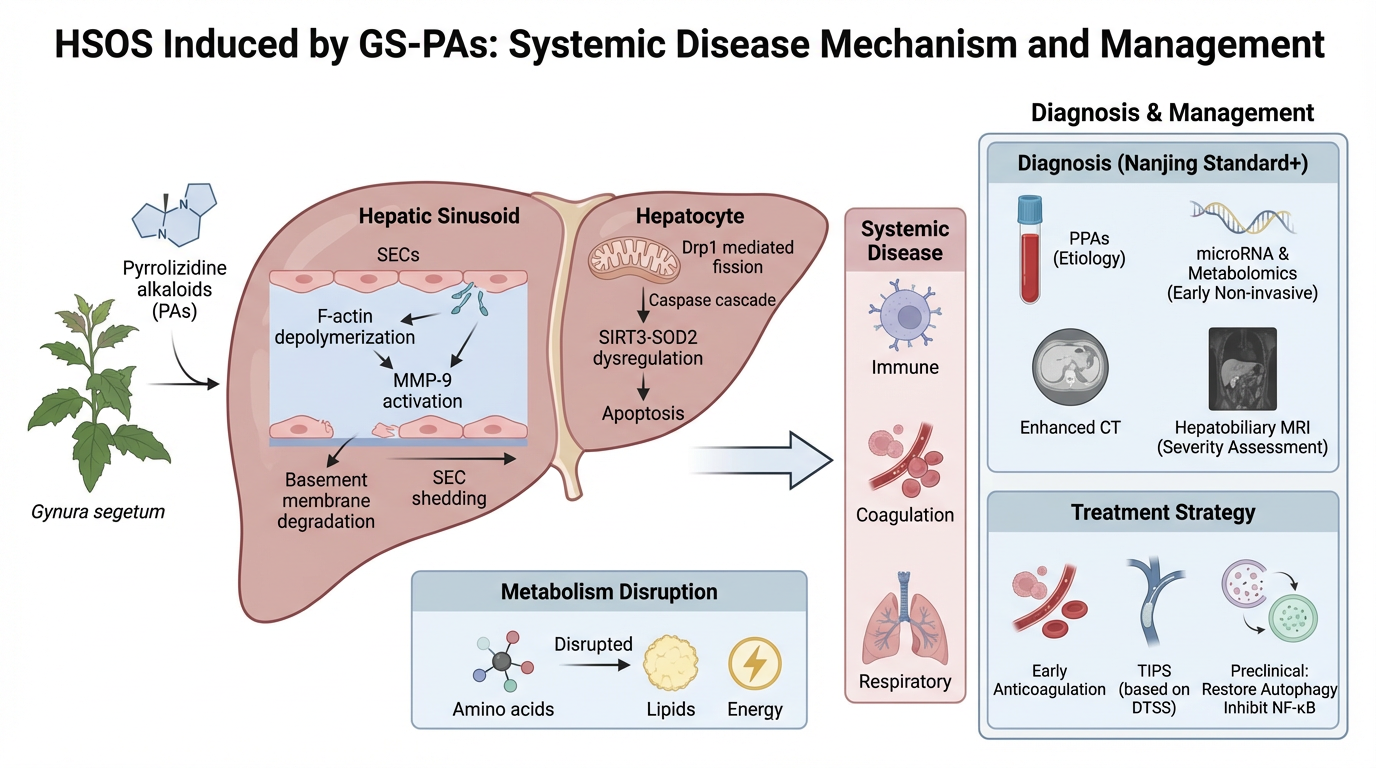
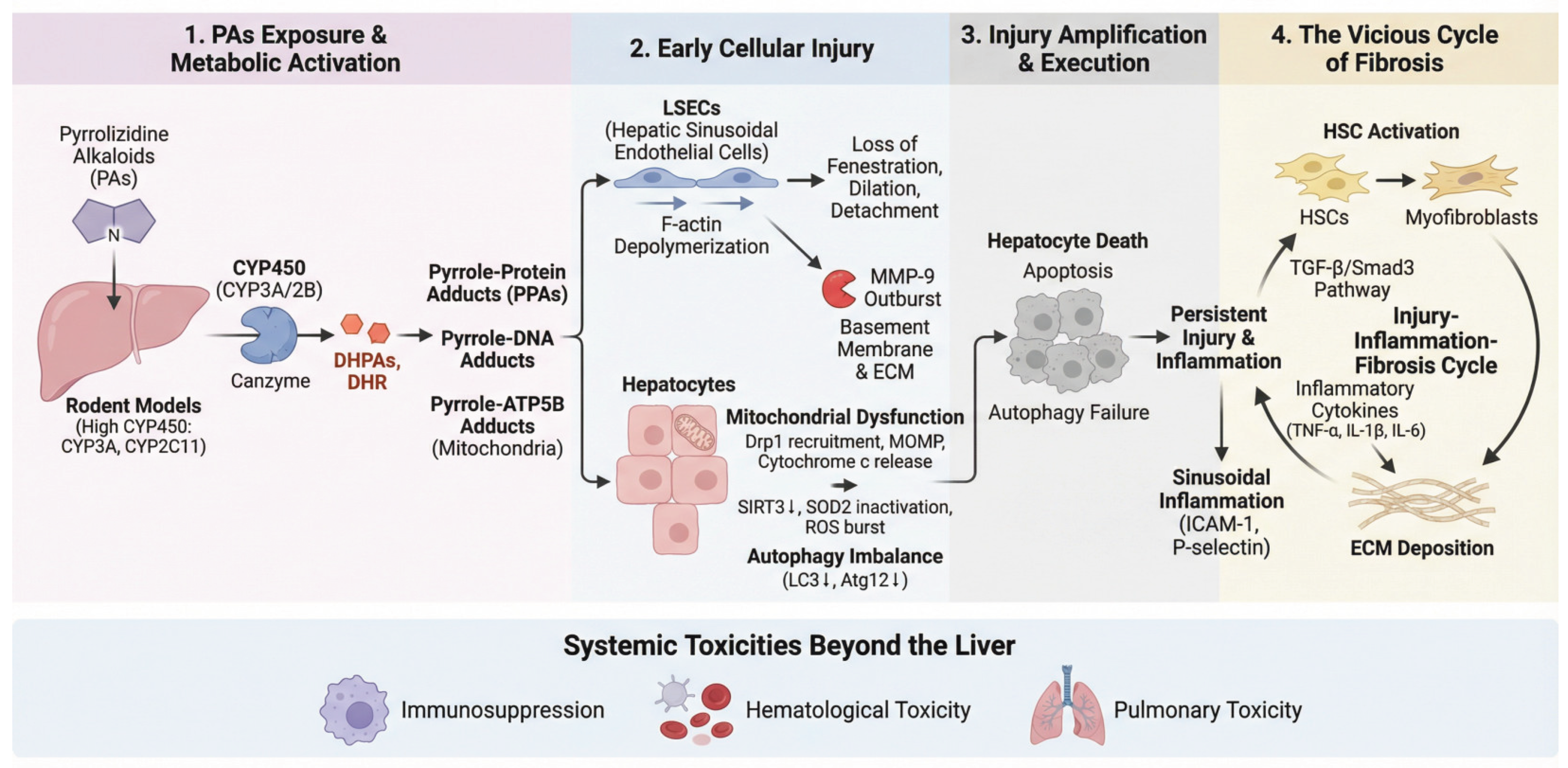
4. Pathogenesis: From Molecular Events to Pathological Outcomes
3.1. Establishment of Experimental Models
3.2. Initiation of Toxic Metabolism and Core Molecular Events
3.3. Selective Injury to Hepatic Sinusoidal Endothelial Cells
3.4. Amplification and Execution of Injury: MMP-9 Outburst and Hepatocyte Death
3.5. Hepatocyte Death: Mitochondrial Apoptosis and Autophagy Imbalance
3.6. The Vicious Cycle of Fibrosis
3.7. Systemic Toxicities Beyond the Liver in PAs-HSOS
3.7.1. Immunosuppression
3.7.2. Hematological Toxicity
3.7.3. Pulmonary Toxicity
4. Evolution of the Diagnostic Framework: From Clinical Criteria to Precision Biomarkers
4.1. Establishment and Optimization of Clinical Diagnostic Standards
4.2. Advances in Quantitative and Functional Imaging Diagnosis
4.3. Emerging Frontiers in Precision Biomarkers
4.3.1. Etiology-Specific Biomarker: PPAs
4.3.2. Early Diagnostic and Prognostic Biomarkers: microRNAs and Metabolomics
4.3.3. Systems Biology Perspective: The Gut-Liver Axis
5. Evolution of Therapeutic Strategies: From Supportive Care to Multimodal Precision Intervention
5.1. Management of PA-HSOS
5.2. Precision Application of Interventional and Surgical Therapies and Advances in Prognostic Evaluation
5.2.1. Anticoagulation Therapy: From Exploratory Use to Established Role and Risk Management
5.2.2. Interventional Therapy: Earlier Application and Development of Prognostic Models
5.2.3. Combined Procedures and the Expanding Role of Liver Transplantation
6. Summary and Perspectives
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HSOS | Hepatic Sinusoidal Obstruction Syndrome |
| PAs | Pyrrolizidine Alkaloids |
| PPAs | pyrrole-protein adducts |
| GS | Gynura segetum |
| HILI | Herb-Induced Liver Injury |
| DILI | Drug-Induced Liver Injury |
| LSECs | Liver Sinusoidal Endothelial Cells |
| DTSS | Drum Tower Severity Score |
| HSCT | hematopoietic stem cell transplantation |
| VOD | veno-occlusive disease |
| HSCT | hematopoietic stem cell transplantation |
| IARC | The International Agency for Research on Cancer |
| EFSA | The European Food Safety Authority |
| MHRA | Medicines and Healthcare Products Regulatory Agency |
| DHPAS | dehydropyrrolizidine alkaloids |
| MMP-9 | metalloproteinase-9 |
| MMPi | MMP inhibitors |
| DRP1 | dynamin-related protein 1 |
| MOMP | mitochondrial outer membrane permeabilization |
| ECM | excessive deposition of extracellular matrix |
| PT | prothrombin time |
| APTT | activated partial thromboplastin time |
| TT | thrombin time |
| FIB | alongside decreased fibrinogen |
| DIPS | direct intrahepatic portocaval shunt |
| TIPS | transjugular intrahepatic portosystemic shunt |
| Gd-EOBDTPA | gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid |
References
- Seow, L. J.; Beh, H. K.; Umar, M. I.; Sadikun, A.; Asmawi, M. Z. Anti-inflammatory and antioxidant activities of the methanol extract of Gynura segetum leaf. Int. Immunopharmacol. 2014, 23(1), 186–191. [Google Scholar] [CrossRef]
- Rautou, P. E. Management of hepatic vascular diseases. J. Hepatol. 2012, 56 Suppl 1, S25–S38. [Google Scholar] [CrossRef]
- Wang, J. Y.; Gao, H. Tusanqi and hepatic sinusoidal obstruction syndrome. J. Dig. Dis. 2014, 15(3), 105–107. [Google Scholar] [CrossRef]
- Lin, G.; Wang, J. Y.; Li, N.; Li, M.; Gao, H.; Ji, Y.; Zhang, F.; Wang, H.; Zhou, Y.; Ye, Y. Hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. J. Hepatol. 2011, 54(4), 666–673. [Google Scholar] [CrossRef]
- DeLeve, L. D.; Valla, D.-C.; Garcia-Tsao, G. Vascular disorders of the liver#†. Hepatology 2009, 49(5), 1729–1764. [Google Scholar] [CrossRef] [PubMed]
- Kikuta, A. Diagnosis and treatment of sinusoidal obstruction syndrome (veno-occlusive disease)]. Rinsho Ketsueki 2021, 62(8), 1256–1264. [Google Scholar] [CrossRef]
- Zhang, Z.; Zou, H.; Dai, Z.; Shang, J.; Sure, S.; Lai, C.; Shi, Y.; Yang, Q.; Xiang, G.; Yao, Y.; Feng, T.; Zhong, D.; Huang, X. Gynura segetum-induced liver injury leading to acute liver failure: a case report and literature review. BMC Complement Med Ther 2022, 22(1), 61. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Zhang, C. Y.; Li, D. P.; Chen, H. B.; Lin, G. Tu-San-Qi (Gynura japonica): the culprit behind pyrrolizidine alkaloid-induced liver injury in China. Acta Pharmacol. Sin. 2020, 1212–1222. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, H. Gynura segetum induces hepatic sinusoidal obstruction syndrome in a child: A case report. Medicine (Baltimore) 2024, 103(11), e37341. [Google Scholar] [CrossRef] [PubMed]
- Mackay, A. H. The Alkaloids of Senecio Jacobæa. Nature 1920, 106, 503. [Google Scholar] [CrossRef]
- Organization, W. H. Pyrrolizidine alkaloids: health and safety guide. In Geneva World Health Organization; 1989. [Google Scholar]
- Zhang, L.; Li, Q.; Makamure, J.; Zhao, D.; Liu, Z.; Zheng, C.; Liang, B. Transjugular intrahepatic portosystemic shunt for hepatic sinusoidal obstruction syndrome associated with consumption of Gynura segetum. BMC Gastroenterol. 2021, 21(1), 26. [Google Scholar] [CrossRef]
- Lu, Y.; Ma, J.; Song, Z.; Ye, Y.; Fu, P. P.; Lin, G. The role of formation of pyrrole–ATP synthase subunit beta adduct in pyrrolizidine alkaloid-induced hepatotoxicity. Arch. Toxicol. 2018, 92(11), 3403–3414. [Google Scholar] [CrossRef]
- Rute, M.; David, P.; Patrícia, V. o.; Paula, A. Pyrrolizidine Alkaloids: Chemistry, Pharmacology, Toxicology and Food Safety. Int. J. Mol. Sci. 2018, 19(6), 1668. [Google Scholar] [CrossRef] [PubMed]
- Mattocks, A.R. The Occurrence and Analysis of Pyrrolizidine Alkaloid N-Oxides. Xenobiotica 1971, 1(4-5), 451–453. [Google Scholar] [CrossRef]
- Seeff, L.; Stickel, F.; Navarro, V. J. Hepatotoxicity of Herbals and Dietary Supplements - ScienceDirect. Drug-Induced Liver Disease (Third Edition) 2013, 631–657. [Google Scholar] [CrossRef]
- Knutsen, H. K.; Alexander, J.; Barregard, L.; Binaglia, M. Risks for human health related to the presence of pyrrolizidine alkaloids in honey, tea, herbal infusions and food supplements; John Wiley & Sons Ltd, 2017; pp. 1831–4732. [Google Scholar] [CrossRef]
- Knutsen, H. K.; Alexander, J.; Barregård, L.; Bignami, M.; Brüschweiler, B.; Ceccatelli, S.; Cottrill, B.; Dinovi, M.; Edler, L.; Grasl-Kraupp, B.; Hogstrand, C.; Hoogenboom, L. R.; Nebbia, C. S.; Oswald, I. P.; Petersen, A.; Rose, M.; Roudot, A. C.; Schwerdtle, T.; Vleminckx, C.; Vollmer, G.; Wallace, H.; Gomez Ruiz, J. A.; Binaglia, M. Risks for human health related to the presence of pyrrolizidine alkaloids in honey, tea, herbal infusions and food supplements. Efsa j 2017, 15(7), e04908. [Google Scholar] [CrossRef]
- (MHRA), M. a. H. p. R. A., Public consultation: proposals to prohibit the sale, supply or importation of unlicensed medicinal products for internal use which contain Senecio species and proposals to amend three existing orders. Agency, M. a. H. p. R., Ed. London, UK, 2007.
- Schramm, S.; Köhler, N.; Rozhon, W. Pyrrolizidine Alkaloids: Biosynthesis, Biological Activities and Occurrence in Crop Plants. Molecules 2019, 24(3), 498. [Google Scholar] [CrossRef] [PubMed]
- Schrenk, D. Toxicology of pyrrolizidine alkaloids. Food Chem. Toxicol. 2020, 135, 110938. [Google Scholar] [CrossRef] [PubMed]
- Xiong, A.; Shao, Y.; Fang, L.; Yang, X.; Zhang, S.; Zheng, J.; Ding, W.; Yang, L.; Wang, Z. Comparative analysis of toxic components in different medicinal parts of Gynura japonica and its toxicity assessment on mice. Phytomedicine 2019, 54, 77–88. [Google Scholar] [CrossRef]
- Wang, W.; Yang, X.; Chen, Y.; Ye, X.; Wang, Z. Seneciphylline, a main pyrrolizidine alkaloid in Gynura japonica, induces hepatotoxicity in mice and primary hepatocytes via activating mitochondria﹎ediated apoptosis. J. Appl. Toxicol. 2020, 40(11), 1534–1544. [Google Scholar] [CrossRef]
- Ma, J.; Xia, Qingsu; Fu, Peter P.; Lin, Ge. Pyrrole-protein adducts – A biomarker of pyrrolizidine alkaloid-induced hepatotoxicity. Journal of Food and Drug Analysis 2018, 26(3), 965–972. [Google Scholar] [CrossRef]
- Neuman, M. G.; Lawrence, C.; Mihai, O.; Nanau, R. M.; Hyunjin, J. Hepatotoxicity of Pyrrolizidine Alkaloids. J. Pharm. Pharm. Sci. 2015, 18(4), 825–843. [Google Scholar] [CrossRef]
- Chojkier, M. Hepatic sinusoidal-obstruction syndrome: toxicity of pyrrolizidine alkaloids. J. Hepatol. 2003, 39(3), 437–446. [Google Scholar] [CrossRef] [PubMed]
- Ruan, J.; Yang, M.; Fu, P.; Ye, Y.; Lin, G. Metabolic activation of pyrrolizidine alkaloids: insights into the structural and enzymatic basis. Chem. Res. Toxicol. 2014, 27(6), 1030–1039. [Google Scholar] [CrossRef]
- Ruan, J.; Liao, C.; Ye, Y.; Lin, G. Lack of metabolic activation and predominant formation of an excreted metabolite of nontoxic platynecine-type pyrrolizidine alkaloids. Chem. Res. Toxicol. 2014, 27(1), 7–16. [Google Scholar] [CrossRef]
- He, Y.; Zhu, L.; Ma, J.; Lin, G. Metabolism-mediated cytotoxicity and genotoxicity of pyrrolizidine alkaloids. Arch. Toxicol. 2021, 95(3), 1917–1942. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Li, S.; Lu, M.; Li, Y.; Wang, Q.; Chen, L.; Jia, Y.; Cao, S.; Zhang, T.; Zhou, M.; Gou, X. Investigation of Gynura segetum root extract (GSrE) induced hepatotoxicity based on metabolomic signatures and microbial community profiling in rats. Front. Microbiol. 2022, 13, 947757. [Google Scholar] [CrossRef] [PubMed]
- Yang; Ji-Rong; Huo; Hong-Yi; Zhu; Zhe; Chen; Xiang-Yang; Wang. The effect of Salvia miltiorrhiza in a mouse model of hepatic sinusoidal obstruction syndrome induced by Gynura segetum. Revista espanola de enfermedades digestivas: organo oficial de la Sociedad Espanola de Patologia Digestiva 2019, 111(11), 823–827. [Google Scholar] [CrossRef]
- Zhang, F.; Zhou, Y.; Yang, X.; Xiong, A. Z.; Wang, Z. T.; Yang, L. Gynura Rhizoma containing pyrrolizidine alkaloids induces the hepatic sinusoidal obstruction syndrome in mice via upregulating fibrosis-related factors. Acta Pharmacol. Sin. 2019, 40(6), 781–789. [Google Scholar] [CrossRef]
- Yang, M.; Ruan, J.; Fu, P. P.; Lin, G. Cytotoxicity of pyrrolizidine alkaloid in human hepatic parenchymal and sinusoidal endothelial cells: Firm evidence for the reactive metabolites mediated pyrrolizidine alkaloid-induced hepatotoxicity. Chem. Biol. Interact. 2016, 243, 119–126. [Google Scholar] [CrossRef]
- Zhu, L.; Xue, J.; Xia, Q.; Fu, P. P.; Lin, G. The long persistence of pyrrolizidine alkaloid-derived DNA adducts in vivo: kinetic study following single and multiple exposures in male ICR mice. Arch. Toxicol. 2017, 91(2), 1–17. [Google Scholar] [CrossRef]
- Kolrep, F.; Numata, J.; Kneuer, C.; Preiss-Weigert, A.; Lahrssen-Wiederholt, M.; Schrenk, D.; These, A. In vitro biotransformation of pyrrolizidine alkaloids in different species. Part I: Microsomal degradation. Arch. Toxicol. 2018, 92(3), 1089–1097. [Google Scholar] [CrossRef]
- He, Y. Q.; Yang, L.; Liu, H. X.; Zhang, J. W.; Liu, Y.; Fong, A.; Xiong, A. Z.; Lu, Y. L.; Yang, L.; Wang, C. H. Glucuronidation, a new metabolic pathway for pyrrolizidine alkaloids. Chem. Res. Toxicol. 2010, 23(3), 591–599. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, W.; Yang, Y.; Chen, Y.; Wang, Z. Blood microRNA Signatures Serve as Potential Diagnostic Biomarkers for Hepatic Sinusoidal Obstruction Syndrome Caused by Gynura japonica Containing Pyrrolizidine Alkaloids. Front. Pharmacol. 12, 627126. [CrossRef] [PubMed]
- DeLeve, L. D.; McCuskey, R. S.; Wang, X.; Hu, L.; McCuskey, M. K.; Epstein, R. B.; Kanel, G. C. Characterization of a reproducible rat model of hepatic veno-occlusive disease. Hepatology 1999, 29(6), 1779–91. [Google Scholar] [CrossRef] [PubMed]
- DeLeve; Laurie, D.; Wang; Xiangdong; Tsai; Jeffrey; Kanel; Gary; Strasberg; Steven. Sinusoidal obstruction syndrome (veno-occlusive disease) in the rat is prevented by matrix metalloproteinase inhibition. Gastroenterology 2003, 125(3), 882–890. [Google Scholar] [CrossRef]
- Yu, X. Z.; Ji, T.; Bai, X. L.; Liang, L.; Wang, L. Y.; Chen, W.; Liang, T. B. Expression of MMP-9 in hepatic sinusoidal obstruction syndrome induced by Gynura segetum. J Zhejiang Univ Sci B 2013, 14(1), 68–75. [Google Scholar] [CrossRef]
- Wen, C.; Zhou, T.; Chang, Y.; Wei, Y.; Zhang, H.; Yang, Z. Exposure to Gynura japonica (Thunb.) Juel plants induces hepatoxicity in rats and Buffalo rat liver cells. J. Ethnopharmacol. 2024, 335, 118692. [Google Scholar] [CrossRef]
- Yang, X.; Wang, H.; Ni, H. M.; Xiong, A.; Wang, Z.; Sesaki, H.; Ding, W. X.; Yang, L. Inhibition of Drp1 protects against senecionine-induced mitochondria-mediated apoptosis in primary hepatocytes and in mice. Redox Biol 2017, 12, 264–273. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Yang, X.; Chen, Y.; Ye, X.; Jiang, K.; Xiong, A.; Yang, L.; Wang, Z. Seneciphylline, a main pyrrolizidine alkaloid in Gynura japonica, induces hepatotoxicity in mice and primary hepatocytes via activating mitochondria-mediated apoptosis. J. Appl. Toxicol. 2020, 40(11), 1534–1544. [Google Scholar] [CrossRef]
- Yao, J.; Wu, J.; Jia, S.; Shao, J.; Zhang, X.; Xu, Z.; Zhang, H.; Li, H.; Yao, X. Effects of bicyclol on hepatic sinusoidal obstruction syndrome induced by Gynura segetum. J. Clin. Lab. Anal. 2022, 36(12), e24793. [Google Scholar] [CrossRef]
- Zhao, Y. Q.; Deng, X. W.; Xu, G. Q.; Lin, J.; Lu, H. Z.; Chen, J. Mechanical homeostasis imbalance in hepatic stellate cells activation and hepatic fibrosis. Front Mol Biosci 2023, 10, 1183808. [Google Scholar] [CrossRef] [PubMed]
- Yuandani; Jantan, I.; Husain, K. 4,5,4’-Trihydroxychalcone, 8,8’-(ethene-1,2-diyl)-dinaphtalene-1,4,5-triol and rutin from Gynura segetum inhibit phagocytosis, lymphocyte proliferation, cytokine release and nitric oxide production from phagocytic cells. BMC Complement. Altern. Med. 2017, 17(1), 211. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Zhang, G.; Teng, X.; Zhang, Z.; Pan, J.; Shou, Q.; Chen, M. Hematologic toxicity of Gynura segetum and effects on vascular endothelium in a rat model of hepatic veno-occlusive disease. Zhonghua Gan Zang Bing Za Zhi 2015, 23(1), 59–63. [Google Scholar] [CrossRef] [PubMed]
- Song, Z.; He, Y.; Ma, J.; Fu, P. P.; Lin, G. Pulmonary toxicity is a common phenomenon of toxic pyrrolizidine alkaloids. J Environ Sci Health C Toxicol Carcinog 2020, 38(2), 124–140. [Google Scholar] [CrossRef]
- Gastroenterology, H. C. G. o. C. S. o., Expert consensus on diagnosis and treatment of pyrrolidine alkaloid-related hepatic sinusoidal obstruction syndrome (Nanjing, 2017). Chinese Journal of Digestion 2017, (8), 513–522. [CrossRef]
- Danan, G.; Teschke, R. RUCAM in Drug and Herb Induced Liver Injury: The Update. Int. J. Mol. Sci. 2015, 17(1), 14. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, W.; Zhang, M.; Zhang, F.; Xiao, J.; Yin, Q.; Han, H.; Li, T.; Lin, G.; Zhuge, Y. Development of a Drum Tower Severity Scoring (DTSS) system for pyrrolizidine alkaloid-induced hepatic sinusoidal obstruction syndrome. Hepatol. Int. 2022, 16(3), 669–679. [Google Scholar] [CrossRef]
- Wang, C.; Wu, X.; Xie, W.; Ren, X.; Zhang, W.; Xu, J. Quantitative Analysis of CT Images in Patients with Pyrrolizidine Alkaloid-Induced Sinusoidal Obstruction Syndrome. Sci. Rep. 2019, (1). [Google Scholar] [CrossRef]
- Guo, T.; Li, X.; Yang, X.; Kong, X.; Liu, H.; Bai, T.; Xu, K.; Ye, J.; Song, Y. Gadoxetic Acid-Enhanced Hepatobiliary-Phase Magnetic Resonance Imaging for Pyrrolizidine Alkaloid-Induced Hepatic Sinusoidal Obstruction Syndrome and Association with Liver Function. Sci. Rep. 2019, 9(1), 1231. [Google Scholar] [CrossRef]
- Ma, J.; Xia, Q.; Fu, P. P.; Lin, G. Pyrrole-protein adducts - A biomarker of pyrrolizidine alkaloid-induced hepatotoxicity. J Food Drug Anal 2018, 26(3), 965–972. [Google Scholar] [CrossRef]
- Gao, H.; Li, N.; Wang, J. Y.; Zhang, S. C.; Lin, G. Definitive diagnosis of hepatic sinusoidal obstruction syndrome induced by pyrrolizidine alkaloids. J. Dig. Dis. 2015, 13(1), 33–39. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhang, H.; Shi, J.; Qiu, S.; Fei, Q.; Zhu, F.; Wang, J.; Huang, Y.; Tang, D.; Chen, B. Metabolomics Based Comparison on the Biomarkers between Panax Notoginseng and its Counterfeit Gynura Segetum in Rats. Current Pharmaceutical Analysis 2020. [Google Scholar] [CrossRef]
- Tan, Y.; Zhou, X. Anticoagulant therapy likely increases risk of bleeding in Gynura segetum-induced hepatic sinus obstruction syndrome. Medicine (Baltimore) 2024, 103(6), e35914. [Google Scholar] [CrossRef]
- Wang, Y.; Qiao, D.; Li, Y.; Xu, F. Risk factors for hepatic veno-occlusive disease caused by Gynura segetum: a retrospective study. BMC Gastroenterol. 2018, 18(1), 156. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Zheng, S. Clinicopathological characteristics and diagnosis of hepatic sinusoidal obstruction syndrome caused by Tusanqi - Case report and literature review. Open Med (Wars) 2023, 18(1), 20230737. [Google Scholar] [CrossRef]
- Sun, Z.; Kang, J.; Zhang, Y. Hepatic veno-occlusive disease related to Gynura segetum: A case report. Medicine (Baltimore) 2018, 97(17), e0552. [Google Scholar] [CrossRef]
- Xiao, J.; Tu, J.; Zhang, H.; Zhang, F.; Zhang, W.; Xu, H.; Yin, Q.; Yang, J.; Han, H.; Wang, Y.; Zhang, B.; Peng, C.; Zou, X.; Zhang, M.; Zhuge, Y. Risk factors of poor prognosis in patients with pyrrolidine alkaloid-induced hepatic sinusoidal obstruction syndrome after transjugular intrahepatic portosystemic shunt. Hepatol. Int. 2021, 15(3), 720–729. [Google Scholar] [CrossRef]
- Xiao, J.; Tu, J.; Zhang, H.; Zhang, F.; Zhuge, Y. Risk factors of poor prognosis in patients with pyrrolidine alkaloid-induced hepatic sinusoidal obstruction syndrome after transjugular intrahepatic portosystemic shunt. Hepatol. Int. DOI. [CrossRef]
- Shihua, L.; Jianguo, C.; He, H.; Kechun, Y. Direct Intrahepatic Portocaval Shunt for Sinusoidal Obstruction Syndrome Associated with Hepatotoxicity of Pyrrolizidine Alkaloids. BioMed Res. Int. 2018, 2018, 1–8. [Google Scholar] [CrossRef]
- Gao, Y.; Zhang, X.; Zhou, X.; Wang, J.; Gong, L.; Chen, G. A rare case of Gynura-segetum-related hepatic sinus obstruction syndrome complicated with alcoholic liver disease. J. Pak. Med. Assoc. 2024, 74(7), 1355–1357. [Google Scholar] [CrossRef] [PubMed]
- Cen, P.; Ding, J.; Jin, J. Hepatic sinusoidal obstruction syndrome caused by the ingestion ofGynura segetumin a patient with alcoholic cirrhosis: a case report. J. Int. Med. Res. 2021, 49(4), 157–168. [Google Scholar] [CrossRef] [PubMed]
| Period | Country/Region | Key Regulatory Actions/Events | Core Content and Impact |
|---|---|---|---|
| 1920s–1950s Initial Recognition | South Africa | Report and Confirmation of “Seneciosis” in Livestock | First scientific confirmation of a direct link between consumption of PAs-containing plants (Crotalaria) and animal hepatic veno-occlusive disease (VOD), laying the foundation for PAs toxicology research. |
| 1950s–1970s Widespread Discovery & Local Response | Jamaica, India, Afghanistan | Documentation of Mass Human Poisonings, e.g., “Jamaican Vomiting Sickness” | Outbreaks of VOD in multiple regions linked to PAs-contaminated grains confirmed severe human toxicity, triggering international concern. |
| 1970s–1990s Systematic Regulatory Beginnings | Germany | Early National Restrictions on Herbal Products | Based on toxicological research, Germany’s Federal Ministry of Health issued an ordinance in 1992 limiting specific plants for herbal teas and proposing a provisional tolerable daily intake (TDI), forming one of the earliest national PA regulatory frameworks. |
| 1990s–Present Establishment of Global Standards | European Union (EMA) | EMA/HMPC/893108/2011 Guideline on Pyrrolizidine Alkaloids in Herbal Medicinal Products | Adopted on 21 July 2016, this guideline sets unified limits (e.g., maximum daily intake of 1.0 μg for adults) for PAs in all herbal medicines marketed in the EU, mandating risk assessment and profoundly impacting the global industry. |
| 1990s–Present Establishment of Global Standards | United Kingdom (MHRA) | 2007 Public Consultation: Proposal to Prohibit Unlicensed Oral Medicines Containing Senecio | This consultation led to a ban on specific PAs-containing plants in unlicensed medicines, exemplifying a targeted national action with a precautionary principle. |
| 1990s–Present Establishment of Global Standards | United States (FDA) | Enforcement via Warning Letters & Import Alerts (e.g., Import Alert 54–10) | The FDA monitors PAs risks primarily through enforcement tools like warning letters and import alerts for non-compliant products (e.g., herbal teas, supplements), without setting a unified federal limit. |
| 1990s–Present Establishment of Global Standards | Australia (TGA) | Limits Specified in the Therapeutic Goods (Standard for Medicinal Plants) Order (Schedule 14) | The Order lists permit PAs-containing plants with strict conditions, doses, and mandatory warning labels, establishing clear national standards. |
| Recent Developments: Extension to the Food Sector | European Union | Setting Maximum Levels in Food: Commission Regulation (EU) 2020/2040 | This regulation, amending Regulation (EC) No 1881/2006, sets specific PAs maximum levels for dried herbs, herbal infusions, and food supplements, marking a key expansion of PAs regulation into the food chain. |
| Recent Developments: Extension to the Food Sector | European Union, China, etc. | Enhanced Monitoring in Honey and Other Foods | Recognizing PAs contamination in honey, salads, etc., regulators have intensified monitoring and research. The EU and China include honey in official control plans, though a uniform EU-wide maximum level for honey is not yet established. |
| No. | Standard Name | Year of Publication/Proposal | Institution/Country | Applicable Type | Core Diagnostic Points |
|---|---|---|---|---|---|
| 1 | Seattle Criteria (Original) | 1984 | Seattle Bone Marrow Transplant Group (USA) | Transplant/Chemotherapy-related HSOS | Within 20 days post-transplant, presence of≥2 criteria: (1) Jaundice (elevated TBil); (2) Hepatomegaly or right upper quadrant pain; (3) Weight gain >2%. |
| 2 | Baltimore Criteria | 1987 | Johns Hopkins University (USA) | Transplant/Chemotherapy-related HSOS | (1) TBil ≥2 mg/dL; (2) Hepatomegaly/right upper quadrant pain; (3) Weight gain >5%; (4) Ascites–Diagnosis requires TBil plus at least one of the other criteria. |
| 3 | Modified Seattle Criteria | 1993 | Multicenter Revision (USA) | Transplant/Chemotherapy-related HSOS | Still requires ≥2 criteria within 20 days post-transplant, but now includes imaging and fluid balance assessments. |
| 4 | EBMT Diagnostic and Severity Criteria (Adult) | 2016 | EBMT (European Society for Blood and Marrow Transplantation) | Transplant/Chemotherapy-related HSOS | (1) Jaundice, hepatomegaly, weight gain, ascites; (2) Occurs within 21 days or may have late onset; (3) Incorporates ultrasound, hemodynamic, and organ function parameters; introduces severity grading. |
| Treatment category | Specific measures | Explanation/Action mechanism | Precautions |
|---|---|---|---|
| Basic and Supportive Care | Discontinuation of PAs-containing Plants and Products | Complete cessation and avoidance of re-exposure to plants containing pyrrolizidine alkaloids and related products | Control disease progression from the source |
| Salt Restriction and Diuresis | Restrict sodium intake (<2 g/day); rational use of diuretics (e.g., spironolactone combined with furosemide) | Basic measures for controlling ascites and alleviating symptoms | |
| Liver-protective Therapy | Administration of hepatoprotective drugs such as polyene phosphatidylcholine, silymarin compounds, and glycyrrhizin preparations | Reduce hepatocyte damage | |
| Albumin Supplementation | Infusion of human albumin to increase plasma colloid osmotic pressure | Applicable for patients with hypoalbuminemia; aids in ascites resolution | |
| Nutritional Support | Provide adequate calories and protein to maintain a positive nitrogen balance | Supports overall patient recovery | |
| Specific Drug Therapy | Low Molecular Weight Heparin Anticoagulation | Improves hepatic microcirculation and prevents microthrombus formation | Suitable for early-stage patients without bleeding tendency |
| Prostaglandin E1 | Vasodilation and inhibition of platelet aggregation | May help improve hepatic blood flow | |
| Corticosteroids | Suppresses early inflammatory response | Efficacy remains controversial, not recommended for routine use; requires careful benefit-risk assessment by experienced physicians | |
| Interventional Therapy | Transjugular Intrahepatic Portosystemic Shunt (TIPS) | Establishes an intrahepatic portal vein-hepatic vein shunt to reduce portal pressure and promote ascites absorption | Indicated for refractory ascites unresponsive to medical diuretic therapy; potential complications include hepatic encephalopathy and shunt stenosis |
| Surgical Treatment | Liver Transplantation | Replaces the diseased liver and restores liver function | Applicable to end-stage liver disease patients refractory to all medical and interventional therapies; suitable for irreversible damage such as liver failure and severe cirrhosis |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





