1. Introduction
Mitochondria are highly compartmentalized organelles in which numerous metabolic reactions occur, requiring the regulated uptake of substrates from the cytosol and the export of products formed within the matrix [
1]. Foundational studies conducted with isolated and coupled mitochondria have led to a general paradigm of mitochondrial metabolite transport summarized in
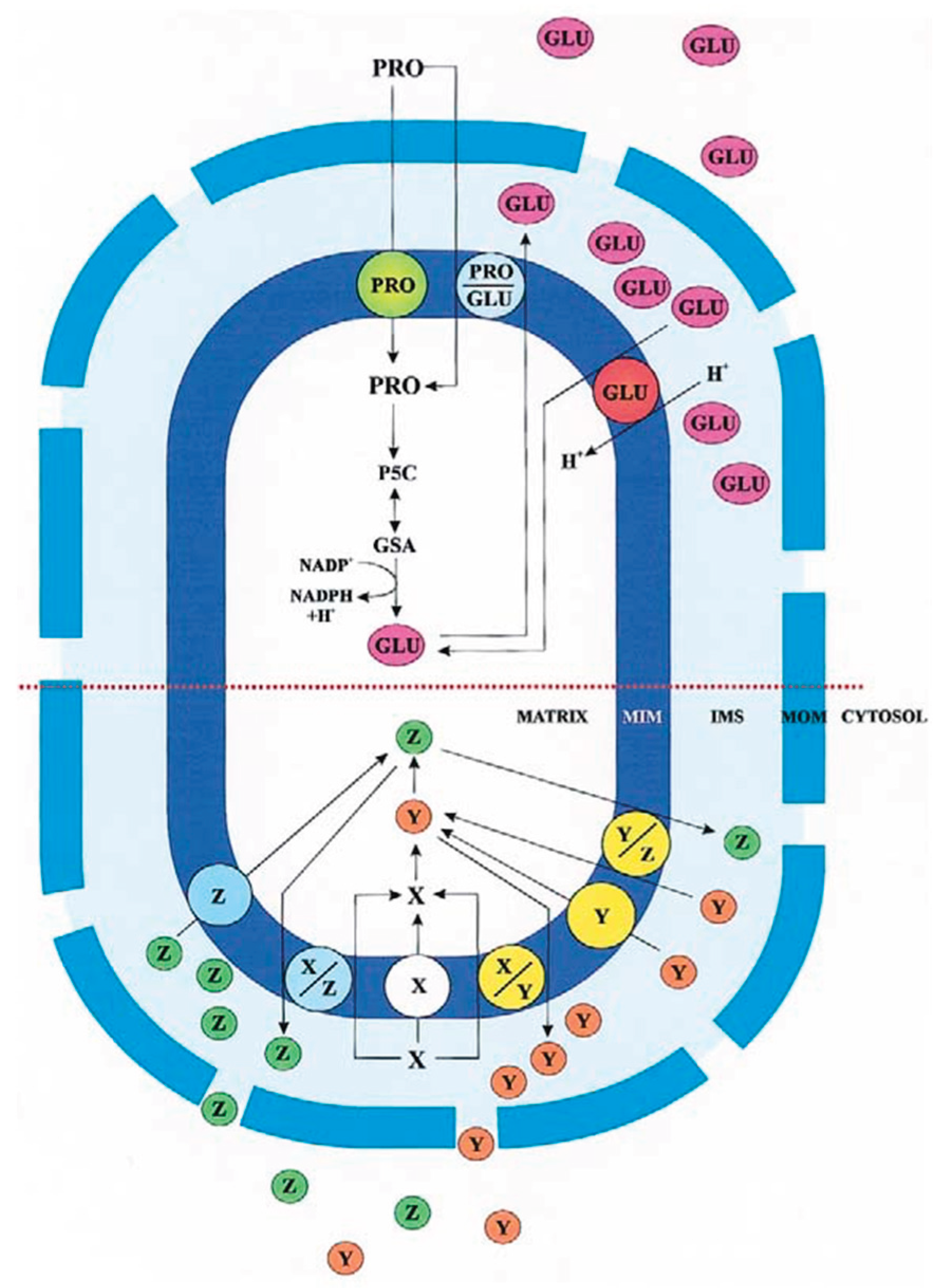
Figure 1 (lower panel; from [
1]).
According to this model, metabolite translocation across the mitochondrial membranes can be described as a two-step process. During the anaplerotic phase, substrates are imported into the matrix to sustain intramitochondrial reactions.
In the subsequent cataplerotic phase, products generated within the matrix are exported, typically in exchange for further substrate uptake. A given metabolite X may enter the matrix via uniport, proton-compensated symport, or antiport with non-carbon compounds. Once metabolized to generate products Y and Z, continued uptake of X may proceed by antiport with one of these newly formed metabolites.
Exported products may either remain in the cytosol or re-enter the organelle through alternative carriers, thereby establishing a tight coupling between electron transport, metabolite exchange, and cytosolic metabolism. Notably, no major exceptions to this paradigm have been reported to date.
2. The Role of the Intermembrane Space in Metabolite Trafficking
Despite its conceptual importance, the functional contribution of the intermembrane space (IMS) to metabolite distribution remains insufficiently explored. For example, whether metabolites newly exported from the matrix equilibrate rapidly with cytosolic concentrations is still unclear and rarely discussed in textbooks. Evidence for the presence of distinct IMS pools was provided in a study investigating the energy dependence of the proline/glutamate antiporter in rat kidney mitochondria. The exchange was shown to be driven by the electrochemical proton gradient, with a ΔΨ-dependent proline/glutamate translocator acting together with a ΔpH-dependent glutamate/OH⁻ carrier. High-performance liquid chromatography revealed a discrete glutamate pool in the IMS (
Figure 1, upper panel and [
2]), demonstrating that metabolites exported from the matrix may accumulate transiently in this compartment.
On the other hand, a variety of static approaches to investigate the role of mitochondrial transport in energy metabolism have been used in this century. In this case, a detailed description of the molecules involved in transport and metabolism is made, but no/minimal information on how certain transports and reactions can play a role in the metabolic pathways [see [
3]].
These considerations underscore the need to re-evaluate the integration of mitochondrial transport within major metabolic pathways. in this regard, glycolysis (including mitochondrial shuttles and the transport/metabolism of oxaloacetate and L-lactate) and gluconeogenesis (involving L-lactate, D-lactate, as a glucose precursor), have already been investigated [for refs see [
1,
3,
4]]. In contrast, other pathways remain insufficiently addressed, including the urea and purine nucleotide cycles (especially the fate of cytosolic fumarate), glutamine bioenergetics, and fatty acid synthesis (notably the roles of phosphoenolpyruvate and mitochondrial pyruvate kinase). These areas merit renewed investigation in light of the evolving understanding of mitochondrial transport and its integration with cellular metabolism.
3. Fumarate Transport in Mitochondria
A long-standing question in the study of the urea cycle concerns the metabolic fate of fumarate generated in the cytosol. Fumarate is released into the cytoplasm not only during the urea and purine nucleotide cycles but also during NAD⁺ deamination in ammoniogenesis and the metabolism of several amino acids. Yet its subsequent handling, particularly whether mitochondria play a direct role in its metabolism, has been insufficiently described in standard biochemistry textbooks.
Although low levels of cytosolic fumarase activity have been reported in several tissues, including heart, liver, skeletal muscle, and kidney [
5], these activities appear inadequate to support the metabolic requirements of highly active tissues. This suggests that cytosolic fumarate must enter mitochondria to be metabolized. However, because of its trans-configuration, fumarate has traditionally been considered non-permeant across the inner mitochondrial membrane since the discovery of the mitochondrial metabolite carriers [
6].
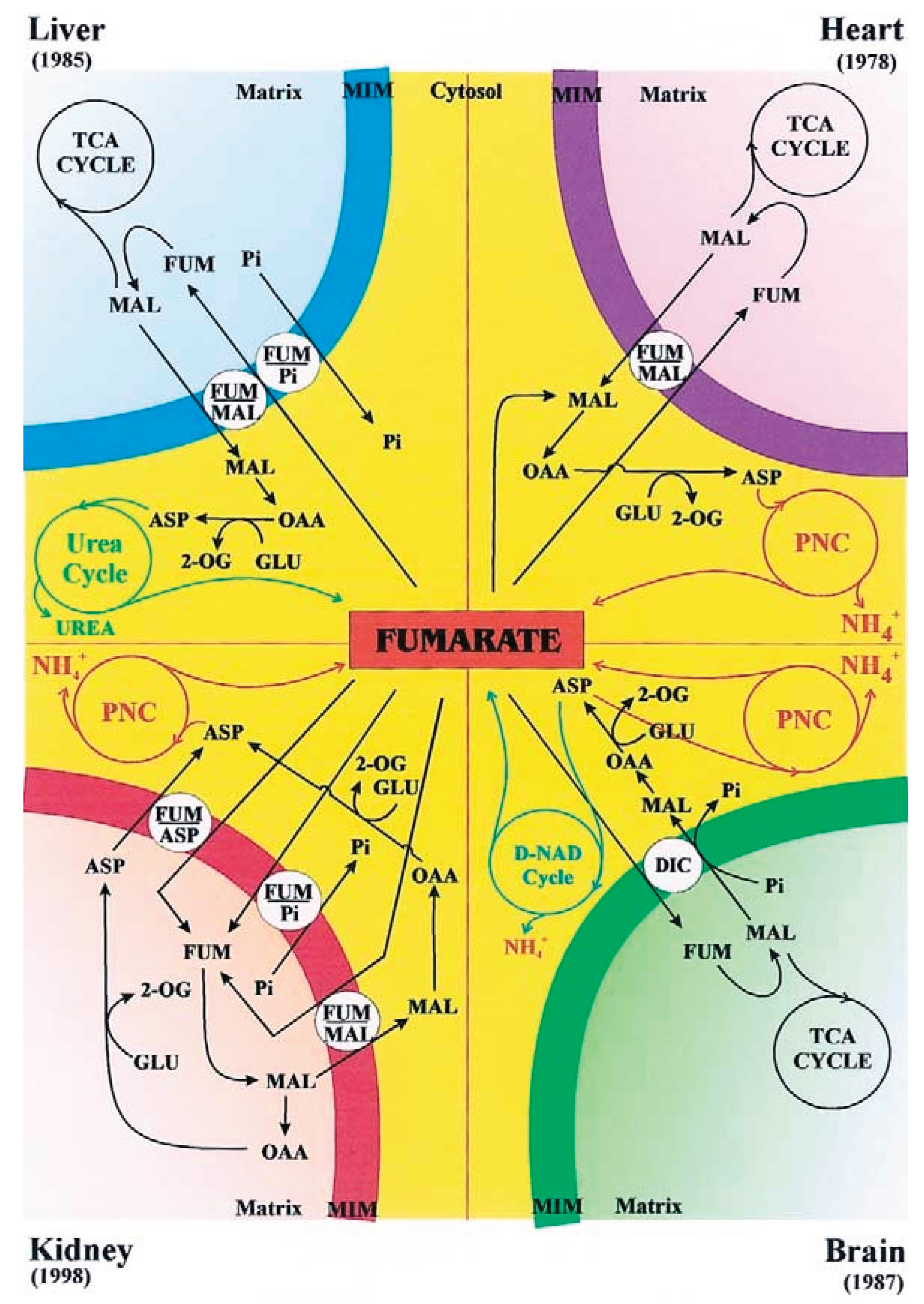
The first experimental evidence for mitochondrial fumarate uptake was provided in 1978, when a fumarate/malate exchange system was identified in rat heart mitochondria [
7,
8]. Subsequent investigations revealed the presence of two distinct transport systems in rat liver mitochondria: a fumarate/malate antiporter and a fumarate/phosphate (Pi) antiporter [
9]. Additional studies showed that fumarate is also taken up by rat brain mitochondria via the dicarboxylate carrier [
10]. Collectively, these findings demonstrate that directed fumarate transport occurs in multiple tissues and is mediated by specific carrier proteins.
The fumarate/malate exchange is thought to play a key role in providing cytosolic malate, which supports gluconeogenesis and contributes to the closure of the urea cycle through aspartate formation via malate dehydrogenase and aspartate aminotransferase. Conversely, fumarate uptake in exchange for phosphate represents an anaplerotic route for carbon replenishment in the tricarboxylic acid (TCA) cycle, thereby linking amino acid catabolism to oxidative phosphorylation. In rat brain mitochondria, fumarate addition induces a hyperbolic efflux of malate, further supporting its role in mitochondrial–cytosolic metabolite exchange. A similar fumarate/malate antiporter has also been demonstrated in
Saccharomyces cerevisiae mitochondria [
11].
In rat kidney mitochondria, both under normal and acidotic conditions, fumarate, whether generated
in vitro from externally added adenyl-succinate via adenyl-succinate lyase or supplied exogenously, was shown to enter the matrix in exchange for malate or aspartate [
12]. These findings highlight that fumarate transport systems are not uniform across tissues but instead reflect the specific metabolic demands of each organ. For instance, while kidney mitochondria display fumarate/aspartate exchange, fumarate uptake in brain mitochondria occurs via the dicarboxylate carrier [
10].
Figure 2 (from [
13]) summarizes the known fumarate transport pathways in mitochondria from liver, heart, kidney, and brain, and highlights the specific carriers involved in each tissue type.
It is remarkable that, nearly a century after the discovery of the urea cycle, the fate of cytosolic fumarate remains largely overlooked, not only in biochemistry textbooks, but even in recent literature intended to clarify metabolic pathways. In one such article, the author states: “Similar to the urea cycle, the carbon skeleton of aspartate leaves the purine nucleotide cycle as fumarate in the cytosol. Even if the role of this compound is to perform the anaplerotic function of replenishing the citric acid cycle by being converted to oxaloacetate, to do so it must first enter the mitochondrial matrix. Because the mitochondrial inner membrane does not possess a transporter for fumarate, it must first be converted to malate in the cytosol” [
14].
In another paper reporting multiple functions of fumarate [
15], metabolite traffic across the mitochondrial membrane is described as “an essential process coordinating both mitochondrial and cytosolic metabolism… our coverage of the entire mitochondrial family remains incomplete and further investigations are required in order to gain a comprehensive understanding of organic acid transport. It is well established that the exchange of TCA cycle intermediates, such as fumarate, across the mitochondrial membrane connects the TCA cycle with diverse metabolic processes including gluconeogenesis, the glyoxylate cycle, and the biosynthesis of amino acids and nucleic acids.” Yet fumarate transporters are not discussed in this paper, nor in another study examining the role of fumarate in cancer [
16]. The authors note: “The phenomenon of enhanced glycolysis in tumors has been acknowledged for decades, but biochemical evidence to explain it is only just beginning to emerge… A significant hint as to the triggers and advantages of enhanced glycolysis in tumors was supplied by the recent discovery that succinate dehydrogenase (SDH) and fumarate hydratase (FH) are tumor suppressors… Further steps forward showed that the substrates of SDH (succinate dehydrogenase) and FH, (fumarate hydratase) succinate and fumarate, respectively, can mediate a ‘metabolic signaling’ pathway. Succinate or fumarate, which accumulate in mitochondria owing to the inactivation of SDH or FH, leak out to the cytosol, where they inhibit a family of prolyl hydroxylase enzymes.” However, no mechanism is provided to explain how succinate and fumarate “leak out to the cytosol.”
Similarly, in a review describing major changes in the expression of urea-cycle enzymes and metabolites in different cancers at various stages [
17], the generation of fumarate is proposed to integrate the urea cycle with the TCA cycle. Fumarate is suggested to enter mitochondria, yet no indication is given of the carrier(s) that would mediate such transport reported also in
Figure 3.
In another paper discussing the
“rewiring” of urea-cycle metabolism in cancer to support anabolism [
18], two schemes illustrating “urea cycle enzymes, substrates and intermediates” depict mitochondrial uptake of fumarate, again without identifying the corresponding transporter(s). The same omission appears in [
19].
Finally, among the “Key Facts” listed in a paper devoted to fumarate as “Metabolite of the Month” [
20], it is stated that “although fumarate is present in different cellular compartments, its transporters have not yet been identified in mammals, unlike in plants and bacteria.” This statement epitomizes the enduring enigma surrounding fumarate transport in mammalian mitochondria and highlights the need for renewed biochemical and physiological investigations into this overlooked aspect of intermediary metabolism.
3. Glutamine Transport in Mitochondria
The mitochondrial transport and metabolism of glutamine, extensively investigated throughout the last century, have been comprehensively summarized in earlier reviews [
1,
13]. A pivotal study published in 1994 examined glutamine transport in both normal and acidotic rat kidney mitochondria (RKM) using radiolabeled substrates and spectroscopic approaches under conditions permitting active glutamine metabolism [
21].
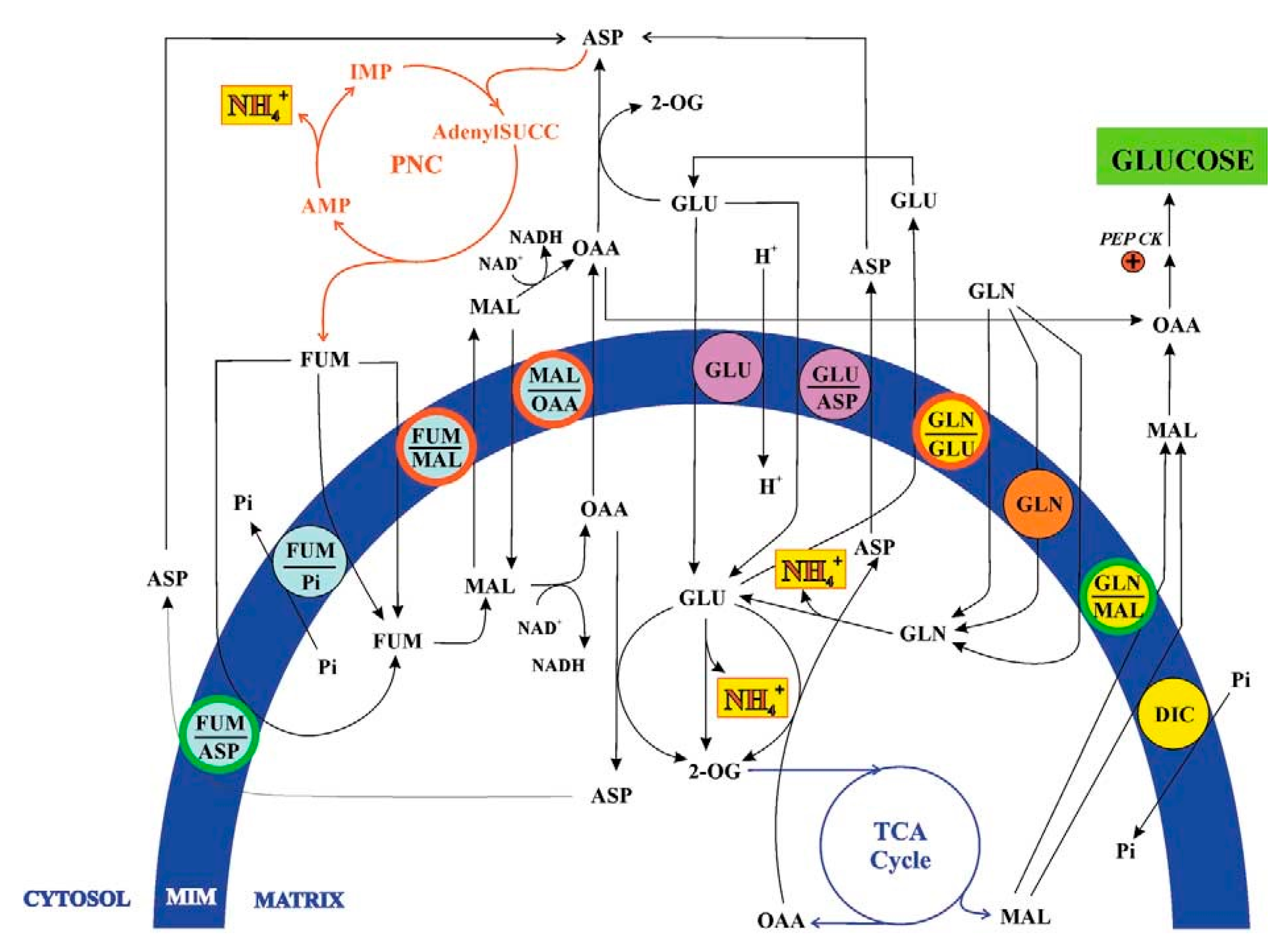
As illustrated in
Figure 3, glutamine crosses the inner mitochondrial membrane through three distinct transport mechanisms: a glutamine uniport, operative exclusively under acidic conditions; a glutamine/glutamate antiporter; and a glutamine/malate antiporter, both active in mitochondria from normal and acidotic rats. All transport systems satisfied the classical criteria for carrier-mediated translocation established in [
1].
Detailed kinetic analyses revealed that the uniport and both antiport systems differ in their Km and Vmax values, pH and temperature dependencies, and sensitivity to specific transport inhibitors. Together, these observations demonstrated the presence of five distinct glutamine translocators, whose differing activities align with the physiological requirements of renal ammoniogenesis. Under conditions of low ammonia demand, glutamate produced from intramitochondrial glutamine catabolism may exit the matrix. When ammonia production must increase, additional glutamate undergoes oxidative deamination by glutamate dehydrogenase, and malate derived from the tricarboxylic acid cycle is exported, likely to support renal gluconeogenesis.
Despite robust experimental evidence, later literature has frequently overlooked these transport systems. For example, a widely cited review on glutaminase regulation and glutamine metabolism [
22] mentions only a single glutamine uniporter. Another review addressing the role of glutamine in human carbohydrate metabolism [
23] highlights glutamine as an important gluconeogenic precursor yet does not discuss mitochondrial involvement or the glutamine/malate antiporter responsible for exporting malate, a key intermediate in renal gluconeogenesis.
More recent studies relating glutamine metabolism to cancer biology have also contributed to this conceptual gap. A paper examining metabolic reprogramming in cancer [
24] briefly mentions metabolite traffic but provides no details on the transporters required for glutamine-dependent mitochondrial functions. Notably, the authors identified 2-oxoglutarate, rather than malate, as the precursor of phosphoenolpyruvate in renal gluconeogenesis, contradicting established biochemical pathways. The same article states: “the mitochondrial glutamine transporter has long been unknown,” despite the earlier characterization of five such transport systems.
Equally surprising is the statement found in a paper published five years ago [
25]: “The identification of the glutamine transporter comes at an exciting time in understanding the transport of molecules across membranes. While older methods relied on relatively low-throughput membrane-reconstitution studies or genetic screens in yeast, new techniques such as insertional mutagenesis now permit the identification of cell-surface and mitochondrial transporters.”
The authors seem to disregard the long-established history of mitochondrial metabolite transport studies, in which isolated coupled mitochondria played a central role. Techniques used for decades after the discovery of mitochondrial carriers, described in [
3], are dismissed as “old,” despite their effectiveness in studying metabolic transport. The situation is comparable to a virtuoso violinist rejecting Stradivari or Guarneri del Gesù instruments in favor of newly built ones.
Other reviews continue to perpetuate the misconception that mitochondrial glutamine transport remains poorly characterized. In [
26], the importance of renal gluconeogenesis under ketogenic or diabetic conditions is emphasized, yet the mechanistic basis for glutamine-derived carbon export is not addressed. In [
27], it is claimed that glutamine metabolic activity interferes with the characterization of mitochondrial carriers; however, as clarified in [
3], the original work in [
21] directly detected export products in intact mitochondria, overcoming such limitations.
Even in recent years, statements persist that the structural and functional characterization of mitochondrial glutamine transporters is still “at infancy” [
28], or that “the network of proteins involved in glutamine flux to the mitochondrial matrix is still underneath” [
29]. These claims overlook the experimentally demonstrated existence of five glutamine transport systems, knowledge that could provide critical insight into cancer cell metabolism and metabolic reprogramming.
Finally, it is noteworthy that a recent study on glutamine transport and prostate cancer radiosensitivity [
30] focuses exclusively on plasma membrane transporters (SLC1A5, SLC7A5, SLC38A1), without addressing the mitochondrial carriers essential for glutamine utilization, even though disruption of glutamine flux is central to the work’s conclusions.
Collectively, these omissions highlight a persistent gap between classical biochemical knowledge and contemporary interpretations of glutamine metabolism. Renewed attention to established transport mechanisms may significantly advance our understanding of mitochondrial function in renal physiology, cancer metabolism, and nutrient signaling.
4. Phosphoenolpyruvate Transport and Mitochondrial Pyruvate Kinase
During a biochemistry lecture, a debate arose concerning two key principles relevant to the field: the principle of molecular economy in living organisms, as formulated by Albert Lehninger (i.e., the principle of maximum economy of parts and processes), and Le Chatelier’s principle, which states that “if a dynamic equilibrium is disturbed by changing the conditions, the position of equilibrium shifts to counteract the change.”
A student then posed the following question: “Why should ATP continue to be formed during glycolysis under high-energy cellular conditions? Specifically, why should ATP be generated in the pyruvate kinase (PK) reaction when the cell is already energy-rich? Would it not be more consistent with the principles mentioned above for phosphoenolpyruvate (PEP) to enter the mitochondria instead, perhaps in exchange for citrate, which is required in the cytosol for fatty-acid synthesis?
Another student followed up by asking what reactions PEP might undergo inside mitochondria, given the apparent absence of intramitochondrial PEP-metabolizing enzymes. Could mitochondria possess their own pyruvate kinase?
PEP transport in mitochondria has already been described in [
3], however both transport and intramitochondrial metabolism of this substrate have been overlooked in the biochemistry textbooks. On the other hand, still PK remains to be considered a cytosolic enzyme.
The first evidence for the presence of PK in both animal and plant mitochondria dates back to 2007, when it was investigated whether and how mitochondria from Jerusalem artichoke (JAM) could metabolize externally added PEP, potentially giving rise to a previously unrecognized mitochondrion–cytosol crosstalk [
31]. This enzyme was identified both immunologically and functionally. It is localized within the inner mitochondrial compartments and can be differentiated from the cytosolic isoform based on distinct pH optima and inhibitor sensitivity profiles.
Accordingly, it was shown that mitochondria isolated from pig liver (PLM) contain PK located in their inner compartments [
32,
33].
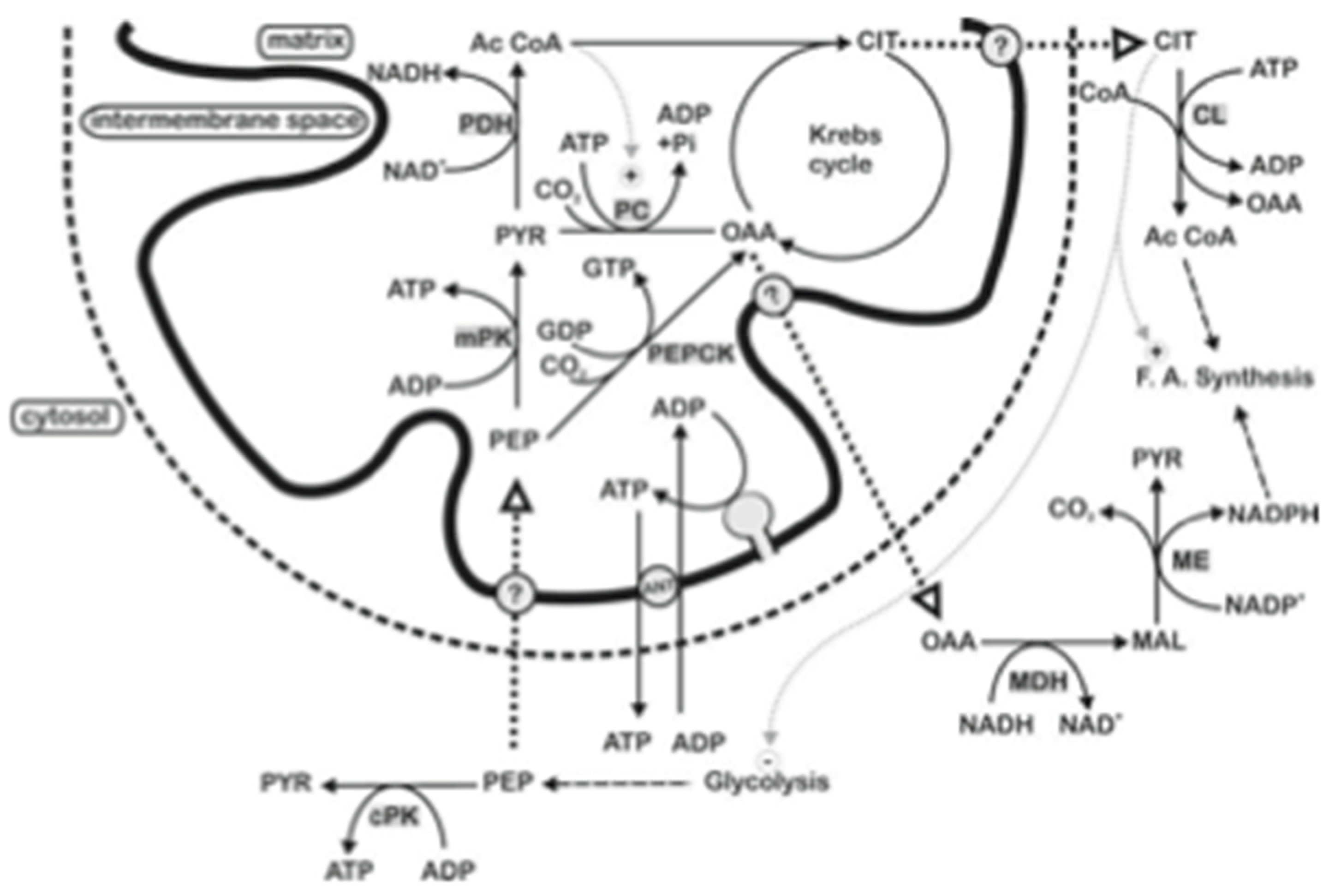
Figure 4.
From [
33]. PEP metabolism in pig liver mitochondria. For an explanation see the text. Abbreviation: AcCoA, acetylCoA; ANT, adenine nucleotide transporter; CIT, citrate; CL, citrate lyase; CoA, Coenzyme A; cPK, cytosolic pyruvate kinase; F.A. synthesis, fatty acid synthesis; MAL, malate; MDH, malate dehydrogenase; ME, malic enzyme; mPK, mitochondrial pyruvate kinase; OAA, oxaloacetate; PC, pyruvate carboxylase; PDH, pyruvate dehydrogenase complex; PEP, phosphoenolpyruvate; PEPCK, PEP carboxykinase; PYR, pyruvate; , inhibition; +, activation.
Figure 4.
From [
33]. PEP metabolism in pig liver mitochondria. For an explanation see the text. Abbreviation: AcCoA, acetylCoA; ANT, adenine nucleotide transporter; CIT, citrate; CL, citrate lyase; CoA, Coenzyme A; cPK, cytosolic pyruvate kinase; F.A. synthesis, fatty acid synthesis; MAL, malate; MDH, malate dehydrogenase; ME, malic enzyme; mPK, mitochondrial pyruvate kinase; OAA, oxaloacetate; PC, pyruvate carboxylase; PDH, pyruvate dehydrogenase complex; PEP, phosphoenolpyruvate; PEPCK, PEP carboxykinase; PYR, pyruvate; , inhibition; +, activation.
As described in [
3], PEP enters mitochondria in a carrier mediated manner; inside PLM, PEP can give pyruvate via mPK likely with ATP synthesis from endogenous ADP. Since citrate and oxaloacetate are exported outside mitochondria as a result of PEP addition to PLM, we are forced to assume that they are newly synthesized as a result of the intramitochondrial metabolism of PEP and exported. This may occur as follows: inside the matrix pyruvate, newly synthesized from taken up PEP via PK, gives acetyl-CoA via the pyruvate dehydrogenase complex, oxaloacetate is formed via the acetyl-CoA activated pyruvate carboxylase and/or the mitochondrial PEP-CK; in turn oxaloacetate can be condensed with acetyl-CoA to produce citrate as well as being exported outside mitochondria, likely via a putative PEP/oxaloacetate antiporter. The newly synthesized citrate is transported outside mitochondria, perhaps in exchange with PEP, thus starting fatty acid synthesis, by providing acetyl-CoA via the ATP dependent citrate lyase, by regulating the cytosolic metabolism with inhibition of phosphofructokinase and activation of the acetyl-CoA carboxylase. The effluxed oxaloacetate is assumed to be reduced by the cytosolic NADH via malate dehydrogenase, to malate which in turn could produce NADPH via malic enzyme to be used in fatty acid synthesis.
Notice that this work confirmed experimentally the existence of mPK already predicted from interrogation of the NCBI genome database. Interestingly transcript variant for Homo sapiens mitochondrial PK was reported (
http://www.ncbi.nlm.nih.gov/nuc core/NM_000298). Whether mitochondrial PK exists also in other mammalian mitochondria remains to be established. However, the evidence that PEP itself can produce citrate, and that 3-mercaptopicolinate, an inhibitor of the mitochondrial PEP-CK, can prevent only partially the citrate synthesis [see 33] is in favor of the occurrence of mitochondrial PK in rat, rabbit and pigeon mitochondria.
The discovery of mitochondrial PK necessitates a revision of current models of mammalian PEP metabolism. The existence of mitochondrial PEP uptake, its conversion within mitochondria, and the export of derived metabolites imply an additional regulatory node connecting glycolysis, lipid biosynthesis, and gluconeogenesis. Interestingly, no mention of mitochondrial pyruvate kinase is made in [
34], where the structure, function, and regulation of pyruvate kinases were comprehensively reviewed, underscoring the need for further biochemical and structural characterization. In light of these findings, the current view of PEP metabolism in mammals, at least in pigs, must be revised.
In conclusion, studies of PEP metabolism should carefully consider mitochondrial PEP uptake, its intramitochondrial conversion, and the export of newly synthesized metabolites. The presence of mitochondrial PK provides new insights into the integration of cytosolic and mitochondrial metabolism. Its characterization may reveal additional mechanisms of metabolic regulation, energy distribution, and biosynthetic coordination in both plant and animal cells.
5. Conclusions
Mitochondrial metabolite transport remains one of the most fascinating yet underexplored aspects of cellular bioenergetics. Despite extensive biochemical evidence demonstrating specific transport systems for fumarate, glutamine, and other key intermediates, modern literature often fails to acknowledge or build upon these earlier findings. The integration of transport processes with metabolic fluxes, linking anaplerotic and cataplerotic reactions, constitutes a dynamic equilibrium central to mitochondrial function. Revisiting and expanding classical experimental data using modern analytical tools will be essential for a renewed understanding of how mitochondrial transport orchestrates energy metabolism, redox balance, and biosynthetic capacity across tissues.
References
- Passarella, S.; Atlante, A.; Valenti, D.; de Bari, L. The role of mitochondrial transport in energy metabolism. Mitochondrion 2003, 2, 319–343. [Google Scholar] [CrossRef]
- Atlante, A.; Passarella, S.; Pierro, P.; Di Martino, C.; Quagliariello, E. The mechanism of proline/glutamate antiport in rat kidney mitochondria. Energy dependence and glutamate-carrier involvement. Eur J Biochem 1996, 241, 171–177. [Google Scholar] [CrossRef]
- Passarella, S.; Schurr, A.; Portincasa, P. Mitochondrial Transport in Glycolysis and Gluconeogenesis: Achievements and Perspectives. Int. J. Mol. Sci. 2021, 22, 12620. [Google Scholar] [CrossRef]
- Passarella, S. Revisiting concepts of mitochondrial transport and energy metabolism in health and cancer. Academia Biology in press. [Google Scholar]
- Akiba, T.; Hiraga, K.; Tuboi, S. Intracellular distribution of fumarase in various animals. J Biochem 1984, 96, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Chappell, J.B.; Haarhoff, K.N. The Penetration of the Mitochondrial Membrane by Anions and Cations; Slater, E.C., Kaniuga, Z., Wojtczak, L., Eds.; Academic Press: London, UK, 1966; pp. 75–91. [Google Scholar]
- Passarella, S.; Fasano, E.; Carrieri, S.; Quagliariello, E. Prime evidenze sperimentali del trasporto del fumarato in mitocondri di cuore di ratto [Initial experimental evidence of fumarate transport in rat heart mitochondria]. Boll Soc Ital Biol Sper. 1978, 54, 40–45. [Google Scholar] [PubMed]
- Passarella, S.; Fasano, E.; Carrieri, S.; Quagliariello, E. Fumarate permeation in rat heart mitochondria. Biochem Biophys Res Commun 1979, 90, 498–505. [Google Scholar] [CrossRef]
- Atlante, A.; Passarella, S.; Giannattasio, S.; Quagliariello, E. Fumarate permeation in rat liver mitochondria: fumarate/malate and fumarate/phosphate translocators. Biochem Biophys Res Commun. 1985, 132, 8–18. [Google Scholar] [CrossRef]
- Passarella, S.; Atlante, A.; Barile, M.; Quagliariello, E. Anion transport in rat brain mitochondria: fumarate uptake via the dicarboxylate carrier. Neurochem Res 1987, 12, 255–264. [Google Scholar] [CrossRef]
- Pallotta, ML.; Fratianni, A.; Passarella, S. Metabolite transport in isolated yeast mitochondria: fumarate/malate and succinate/malate antiports. FEBS Lett 1999, 462, 313–316. [Google Scholar] [CrossRef]
- Atlante, A.; Gagliardi, S.; Passarella, S. Fumarate permeation in normal and acidotic rat kidney mitochondria: fumarate/malate and fumarate/aspartate translocators. Biochem Biophys Res Commun 1998, 24, 711–718. [Google Scholar] [CrossRef] [PubMed]
- Passarella, S.; Atlante, A. Teaching the role of mitochondrial transport in energy metabolism. Biochem Mol Biol Educ 2007, 35, 125–132. [Google Scholar] [CrossRef]
- Arinze, I.J. Facilitating understanding of the purine nucleotide cycle and the one-carbon pool: Part I: The purine nucleotide cycle. Biochem Mol Biol Educ. 2005, 33, 165–168. [Google Scholar]
- Araújo, W.L.; Nunes-Nesi, A.; Fernie, A.R. Fumarate: Multiple functions of a simple metabolite. Phytochemistry 2011, 72, 838–843. [Google Scholar] [CrossRef] [PubMed]
- King, A.; Selak, M.A.; Gottlieb, E. Succinate dehydrogenase and fumarate hydratase: linking mitochondrial dysfunction and cancer. Oncogene 2006, 25, 4675–4682. [Google Scholar] [CrossRef] [PubMed]
- Hajaj, E.; Sciacovelli, M.; Frezza, C.; Erez, A. The context-specific roles of urea cycle enzymes in tumorigenesis. Mol Cell 2021, 81, 3749–3759. [Google Scholar] [CrossRef]
- Keshet, R.; Szlosarek, P.; Carracedo, A.; Erez, A. Rewiring urea cycle metabolism in cancer to support anabolism. Nat Rev Cancer 2018, 18, 634–645. [Google Scholar]
- Ghosh, N.; Mahalanobish, S.; Sil, P.C. Reprogramming of urea cycle in cancer: Mechanism, regulation and prospective therapeutic scopes. Biochem Pharmacol 2024, 228, 116326. [Google Scholar] [CrossRef]
- Schatton, D.; Frezza, C. Fumarate. Trends Endocrinol Metab 2025, S1043-2760(24)00332-1. [Google Scholar] [CrossRef]
- Atlante, A.; Passarella, S.; Minervini, G.M.; Quagliariello, E. Glutamine transport in normal and acidotic rat kidney mitochondria. Arch Biochem Biophys 1994, 15, 369–381. [Google Scholar] [CrossRef]
- Curthoys, N.P.; Watford, M. Regulation of glutaminase activity and glutamine metabolism. Annu Rev Nutr 1995, 15, 133–159. [Google Scholar] [CrossRef]
- Stumvoll, M.; Perriello, G.; Meyer, C.; Gerich, J. Role of glutamine in human carbohydrate metabolism in kidney and other tissues. Kidney Int 1999, 55, 778–792. [Google Scholar] [CrossRef]
- Yoo, H.C.; Park, S.J.; Nam, M.; Kang, J.; Kim, K.; Yeo, J.H.; Kim, J.K.; Heo, Y.; Lee, H.S.; Lee, M.Y.; Lee, C.W.; Kang, J.S.; Kim, Y.H.; Lee, J.; Choi, J.; Hwang, G.S.; Bang, S.; Han, J.M. A Variant of SLC1A5 Is a Mitochondrial Glutamine Transporter for Metabolic Reprogramming in Cancer Cells. Cell Metab 2020, 31, 267–283. [Google Scholar] [CrossRef]
- Stine, Z.E.; Dang, C.V. Glutamine Skipping the Q into Mitochondria. Trends Mol Med 2020, 26, 6–7. [Google Scholar] [CrossRef]
- Newsholme, P.; Lima, M.M.; Procopio, J.; Pithon-Curi, T.C.; Doi, S.Q.; Bazotte, R.B.; Curi, R. Glutamine and glutamate as vital metabolites. Braz J Med Biol Res 2003, 36, 153–163. [Google Scholar] [CrossRef]
- Matés, J.M.; Segura, J.A.; Campos-Sandoval, J.A.; Lobo, C.; Alonso, L.; Alonso, F.J.; Márquez, J. Glutamine homeostasis and mitochondrial dynamics. Int J Biochem Cell Biol 2009, 41, 2051–2061. [Google Scholar] [CrossRef]
- Scalise, M.; Pochini, L.; Galluccio, M.; Indiveri, C. Glutamine transport. From energy supply to sensing and beyond. Biochim Biophys Acta 2016, 1857, 1147–1157. [Google Scholar] [CrossRef]
- Scalise, M.; Pochini, L.; Galluccio, M.; Console, L.; Indiveri, C. Glutamine transport and Mitochondrial Metabolism in Cancer Cell Growth. Front Oncol 2017, 7, 306. [Google Scholar] [CrossRef]
- Kahya, U.; Lukiyanchuk, V.; Gorodetska, I.; Weigel, MM.; Köseer, AS.; Alkan, B.; Savic, D.; Linge, A.; Löck, S.; Peitzsch, M.; Skvortsova, II; Krause, M.; Dubrovska, A. Disruption of glutamine transport uncouples the NUPR1 stress-adaptation program and induces prostate cancer radiosensitivity. Cell Commun Signal 2025, 23, 351–375. [Google Scholar] [CrossRef]
- de Bari, L.; Valenti, D.; Pizzuto, R.; Atlante, A.; Passarella, S. Phosphoenolpyruvate metabolism in Jerusalem artichoke mitochondria. Biochim Biophys Acta 2007, 1767, 281–294. [Google Scholar] [CrossRef]
- Pizzuto, R.; Paventi, G.; Chieppa, G.; Atlante, A.; Passarella, S. Is there a pyruvate kinase in pig liver mitochondria? Ital J Biochem 2007, 56, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Pizzuto, R.; Paventi, G.; Atlante, A.; Passarella, S. Pyruvate kinase in pig liver mitochondria. Arch Biochem Biophys 2010, 495, 42–48. [Google Scholar] [CrossRef] [PubMed]
- Schormann, N.; Hayden, K.L.; Lee, P.; Banerjee, S.; Chattopadhyay, D. An overview of structure, function, and regulation of pyruvate kinases. Protein Sci 2019, 28, 1771–1784. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).