Submitted:
15 December 2025
Posted:
16 December 2025
You are already at the latest version
Abstract
Paris polyphylla (Chonglou), a medicinal herb documented in Shennong’s Classic of Materia Medica and a key component of formulas such as Yunnan Baiyao, is a rare and endangered plant prized for its bioactive steroidal saponins, notably polyphyllin I (PPI) and II (PPII). However, its pharmacological potential is hampered by inefficient extraction and unreliable compound identification. Herein, we developed a sustainable and efficient extraction strategy using ultrasound-assisted deep eutectic solvents (DES), optimized via an L9(34) orthogonal experimental design. Extraction efficiencies across the seven Paris species ranged from 2.04% to 16.51%, achieved by systematically optimizing key parameters such as the choline chloride-to-ethanol molar ratio (1:1.8), material-to-liquid ratio (1:20 g mL-1), and extraction time (100 min). By ultra high-performance liquid chromatography-tandem mass spectrometry (UHPLC-MS/MS) analysis, PPI and PPII were quantified using specific retention times and characteristic fragment ions, revealing content ranges of 3.282–21.452 mg g-1 and 4.201–17.975 mg g-1, respectively. This methodology provides a robust platform for quality control and standardization of Paris-derived medicines, while paving the way for sustainable utilization and in-depth study of its steroidal saponins.
Keywords:
1. Introduction
2. Materials and methodology
2.1. Materials and Instruments
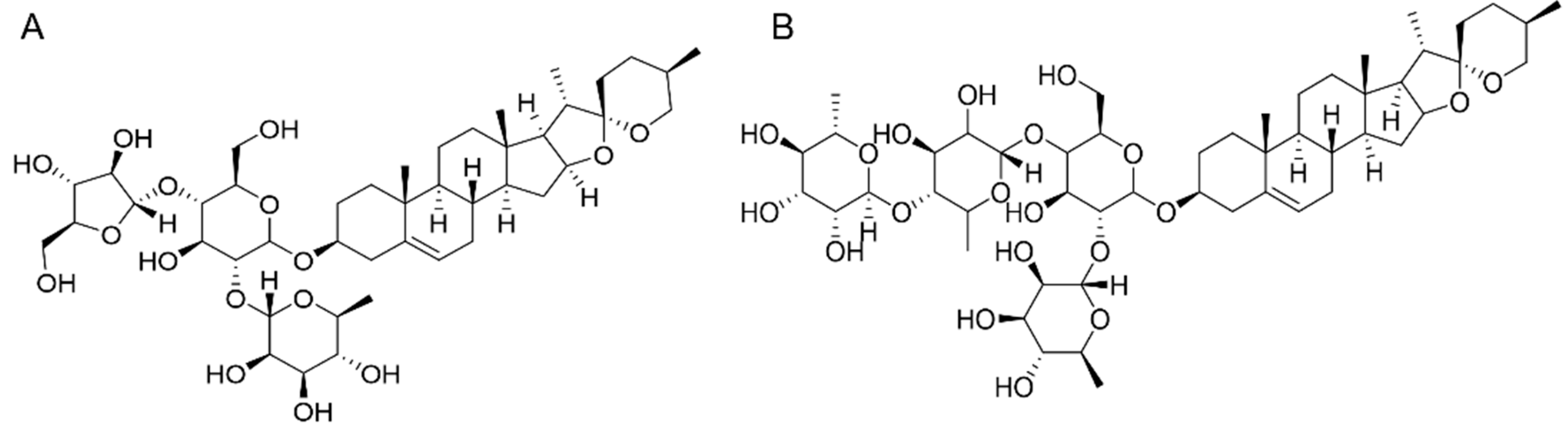
2.2. Basic Information of PPI and PPII
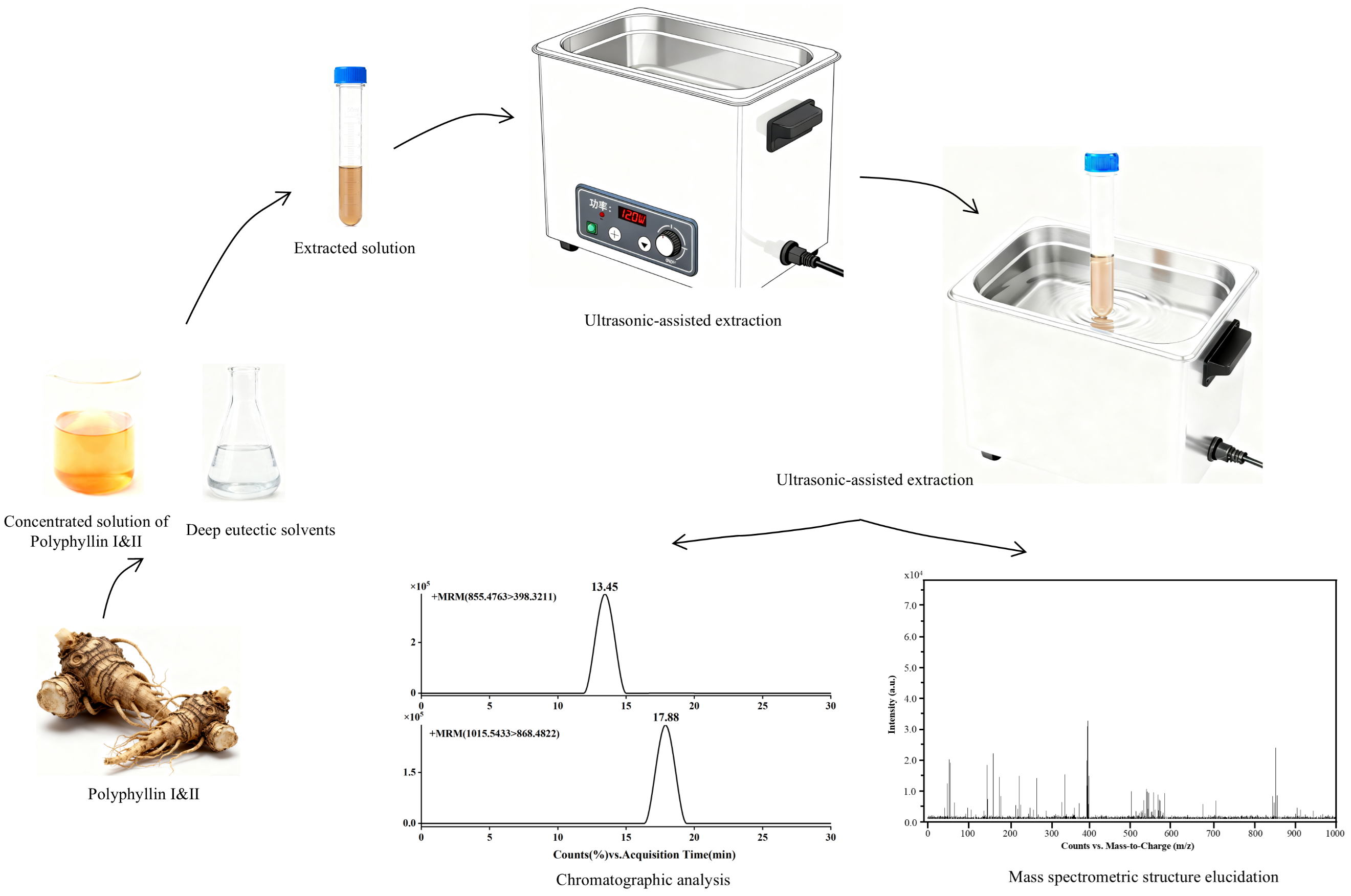
2.3. Ultrasonic-Assisted Preparation of Low Eutectic Solvents
2.4. Extraction Methods for Paris Polyphylla Medicinal Material
2.5. Optimization of Extraction Process Using Orthogonal Experimental Design
2.6. Chromatographic Parameter Conditions
2.7. Mass Spectrometry Parameter Conditions
2.8. Preparation of Standard Solutions
3. Results and discussions
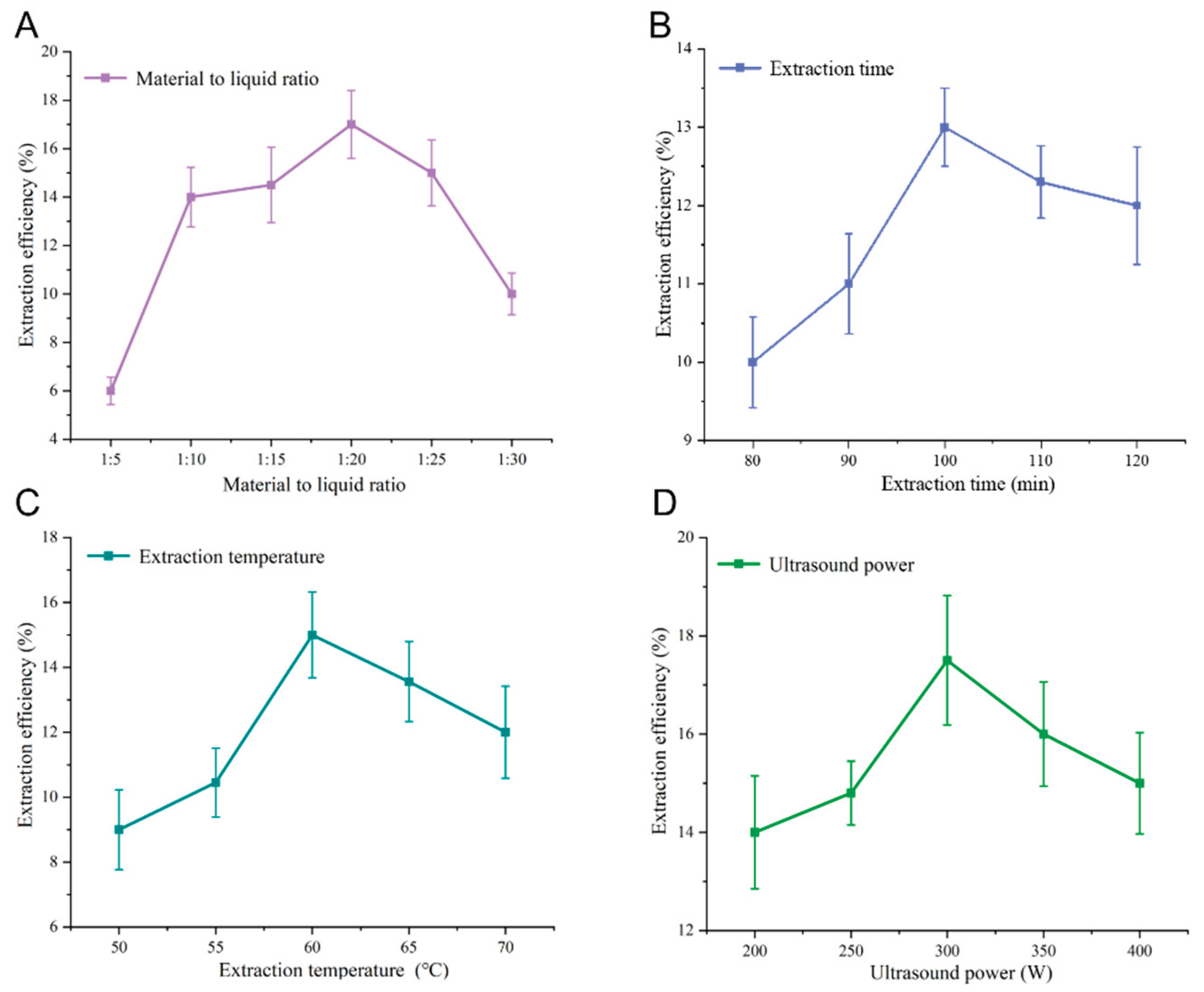
3.1. Optimization of Extraction Process
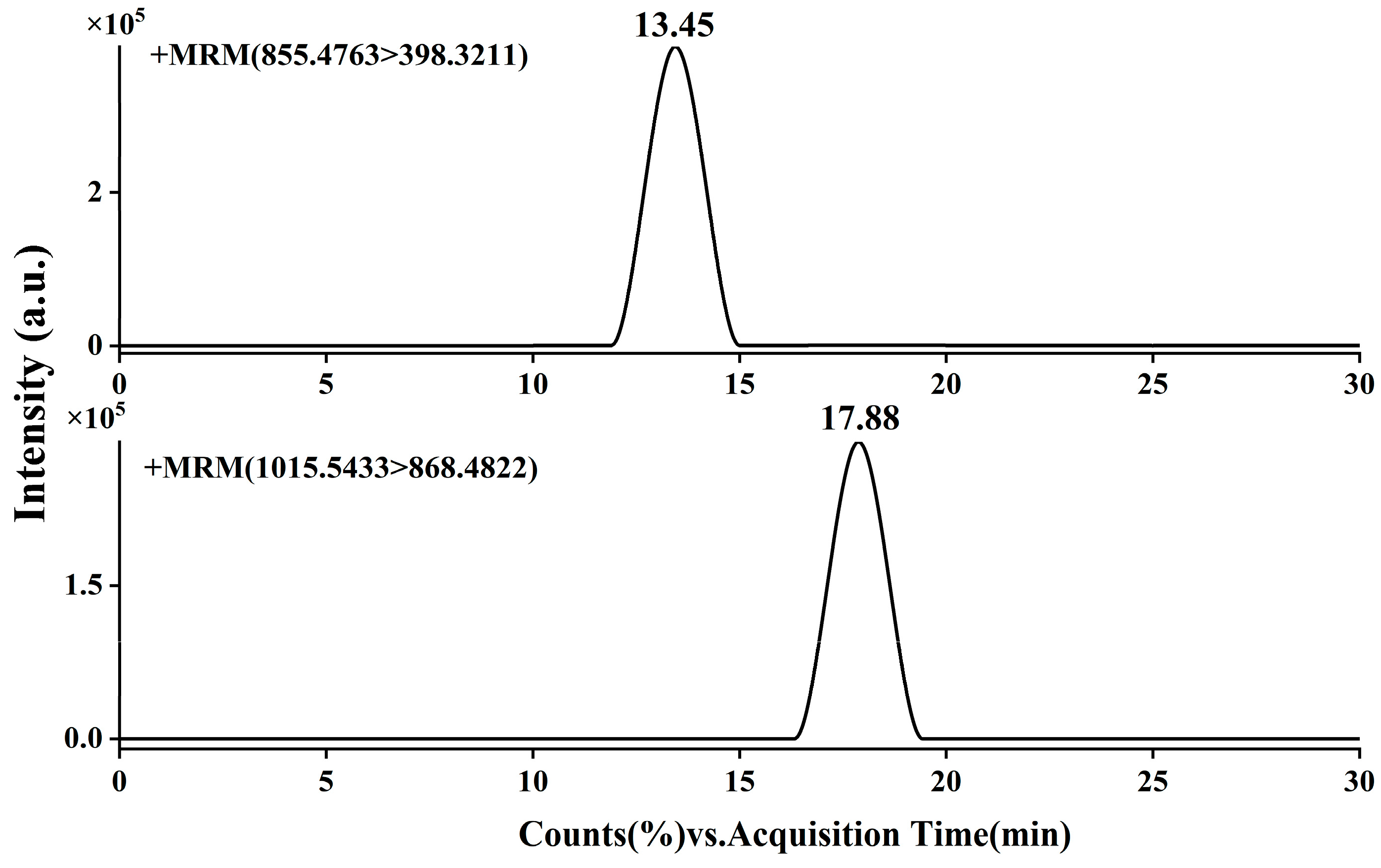
3.2. Optimization of Chromatography and Mass Spectrometry Parameters
3.3. Investigation of Methodological Verification Indicators
3.4. Analysis of Actual Samples
4. Conclusions
Acknowledgments
References
- Zhang, Y.; Fan, Y.C.; Zhang, Y.C.; Li, Q.; Su, Y.Y.; Xu, C.S.; Yu, H.L.; Wang, C.; Zhang, J.; Liao, Z.X. Antitumor activity and mechanistic study of steroidal saponins from the rhizomes of Paris polyphylla var. yunnanensis. Phytochemistry 2025, 235, 114455. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Liao, B.; Miao, J.; Chen, X. Paris spp (Liliaceae): A review of its botany, ethnopharmacology, phytochemistry, pharmacological activities, and practical applications. Front. Pharmacol. 2025, 16, 1570818. [Google Scholar] [CrossRef] [PubMed]
- Ye, X.; Yang, T.; Pu, X.; Hu, H.; Chen, J.; Tan, C.; Tan, X.; Li, S.; Liu, Y. The genus Paris: A fascinating resource for medicinal and botanical studies. Hortic. Res. 2025, 12, uhae327. [Google Scholar] [CrossRef] [PubMed]
- Sha, Y. Li, Preparation, structural characterization, bioactivities, and potential clinical applications of the polysaccharides from Paris polyphylla: A review. Front Pharmacol 2025, 16, 1539237. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Liu, D.; Cheng, H.; Bussmann, R.W.; He, H.; Guo, Z.; Liu, B. Ethnobotanical study of medicinal plants used by Miao people in Jijiezi, Yunnan, China. Ethnobotany Res. Appl. 2019, 18, 1–14. [Google Scholar] [CrossRef]
- Rawat, J.M.; Pandey, S.; Rawat, B.; Rai, N.; Preeti, P.; Thakur, A.; Butola, J.S.; Bachheti, R.K.; Wang, C. Traditional uses, active ingredients, and biological activities of Paris Polyphylla Smith: A comprehensive review of an important Himalayan medicinal plant. J. Chem. 2023, 2023, 1–18. [Google Scholar] [CrossRef]
- Liu, J.; Mu, Y.; Qi, K.; Li, J.; Hu, Y. Regulation of anti-tumour effects of Paris polyphylla saponins via ROS: Molecular mechanisms and therapeutic potentials. Front. Pharmacol. 2025, 16, 1611911. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Chen, B.; Wang, H.; Pan, F. Anti-neurodegenerative potential of polyphyllin: Mechanisms involving inflammation and oxidative stress modulation. Pharmacol. Discovery 2025, 5 10. [Google Scholar] [CrossRef]
- Li, X.; Guo, J.; Xu, Y.; Li, S.; Li, N.; Liu, Q. Effects of heat reflux extraction on the content, antioxidant, and immune activity of polyphenols and flavonoids from hempseed threshing residues. PLoS One 2025, 20, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Deng, L.; Huang, G. Ultrasound-assisted extraction and value of active substances in Muxu, Ultrason. Sonochem 2025, 113, 107220. [Google Scholar] [CrossRef] [PubMed]
- Tian, X.; Liu, J.; Jiang, L.; Kong, W.; Fu, Y.; Qin, L.; Cui, Q. Efficient extraction and optimization procedures of polyphyllins from Paris polyphylla var chinensis by deep eutectic solvent coupled with ultrasonic-assisted extraction. Microchem. J. 2024, 196, 109692. [Google Scholar] [CrossRef]
- Vo, T.P.; Ho, T.A.T.; Truong, K.V.; Ha, N.M.H.; Nguyen, D.Q. Combining novel extraction techniques with natural deep eutectic solvent to acquire phenolic and terpenoid compounds from Paris polyphylla roots. J. Agric. Food Res. 2024, 18, 101544. [Google Scholar] [CrossRef]
- Xiao, X.; Yuan, Z.; Li, G. Separation and purification of steroidal saponins from Paris polyphylla by microwave-assisted extraction coupled with countercurrent chromatography using evaporative light scattering detection. J. Sep. Sci. 2014, 37, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Zhang, J.; Xu, F.; Wang, Y.; Zhang, J. Rapid and simple determination of polyphyllin I, II, VI, and VII in different harvest times of cultivated Paris polyphylla Smith var. yunnanensis (Franch.) Hand.-Mazz by UPLC-MS/MS and FT-IR. J. Nat. Med. 2017, 71, 139–147. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.X.; Xu, L.; Wang, X.; Zhang, K.K.; Zhang, C.Y.; Liu, H.Y.; Ding, P.P.; Shi, W.; Liu, Z.Y. Paris polyphylla ethanol extract and polyphyllin I ameliorate adenomyosis by inhibiting epithelial-mesenchymal transition. Phytomedicine 2024, 127, 155461. [Google Scholar] [CrossRef] [PubMed]
- Thakur, U.; Shashni, S.; Thakur, N.; Rana, S.K.; Singh, A. A review on Paris polyphylla Smith: A vulnerable medicinal plant species of a global significance. J. Appl. Res. Med. Aromat. Plants 2023, 33. [Google Scholar] [CrossRef]
- Han, M.; Wang, Y. Mining the potential quality marker and predicting the total flavonoid content of Paris polyphylla var. Yunnanensis based on information fusion. Microchem. J. 2025, 212, 113415. [Google Scholar] [CrossRef]
- Pei, Y.; Zuo, Z.; Zhang, Q.; Wang, Y. Data fusion of Fourier transform mid-infrared (MIR) and near-infrared (NIR) spectroscopies to identify geographical origin of wild Paris polyphylla var. yunnanensis. Molecules 2019, 24, 2559. [Google Scholar] [CrossRef] [PubMed]
- Zhong, C.; Li, L.; Wang, Y. Comparative analysis of flavonoids in Paris polyphylla var. yunnanensis under different climatic zones using FT-NIR spectroscopy, UPLC-ESI-MS/MS, and chemometrics. J. Appl. Res. Med. Aromat. Plants 2025, 49, 100676. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Jin, H.; Zhang, J.; Wang, Y. Quantitative analysis in combination with fingerprint technology and chemometric analysis applied for evaluating six species of wild Paris using UHPLC-UV-MS. J. Anal. Methods Chem. 2016, 2016, 3182796. [Google Scholar] [CrossRef] [PubMed]
| Name (English) | Molecular formula | Molecular weight | Specification | Quantity | Purity | Storage conditions |
| Polyphyllin I | C44H70O16 | 854.47 | 20 mg | 1 | 98% | 2-8℃, protect from moisture, airtight, light-proof |
| Polyphyllin II | C51H82O20 | 1014.54 | 20 mg | 1 | 98% | 2-8℃, protect from moisture, airtight, light-proof |
| No. | Abbreviation | Solvent 1 | Solvent 2 | Volume Ratio |
| 1 | DES-a | Ethanol | Choline Chloride | 1.0:1.8 |
| 2 | DES-b | Butanediol | Choline Chloride | |
| 3 | DES-c | Ethylene Glycol | Choline Chloride | |
| 4 | DES-d | Ethanol | Methanol | |
| 5 | DES-e | Methanol | Choline Chloride |
| Level | Material-to-liquid ratio (g mL-1) | Extraction time (min) | Extraction temperature (°C) | Ultrasound power (W) |
| 1 | 1:16 | 96 | 60 | 300 |
| 2 | 1:18 | 98 | 62 | 310 |
| 3 | 1:20 | 100 | 64 | 320 |
| Analyte | Polyphyllin I | Polyphyllin II |
| Chromatographic column | Waters Acquity UPLC BEH C18 column | Waters Acquity UPLC BEH C18 column |
| Mobile phase | A: 0.1% Formic acid in H2O B: Formic acid in acetonitrile |
A: 0.1% Formic acid in H2O B: Formic acid in acetonitrile |
| Stationary phase | Waters Acquity HPLC BEH C18 column | Waters Acquity HPLC BEH C18 column |
| Flow rate (mL min-1) |
0.20 | 0.20 |
| Elution gradient | 0.0-1.0 min, 20%B; 2.0-3.0 min, 20%-50%B; 3.0-5.0 min, 50%-95%B; 5.0-28.0 min, 95%B; 28.0-29.0 min, 95%-40%B; 29.0-29.5 min, 40%-5%B 29.5-30.0 min, 5%-5%B |
0.0-1.0 min, 10%B; 1.0-1.5 min, 10%-40%B; 1.5-2.0 min, 40%-80%B; 2.0-3.0 min, 80%-90%B; 3.0-29.0 min, 90%B; 29.0-29.5 min, 90%-5%B 29.5-30.0 min, 5%B |
| Injection volume (μL) | 5 | 5 |
| Column temperature (℃) | 38 | 38 |
| Detection Wavelength (nm) | 203 | 203 |
| Retention Time (min) | 13.45 | 17.88 |
| Analyte | Polyphyllin I | Polyphyllin II |
| Ionization Mode | Positive (ESI+) | Positive (ESI+) |
| Precursor ion (m/z) | [M+H]⁺, m/z, 855.4763, C44H70O16 | [M+H]⁺, m/z,1015.5433, C51H82O20 |
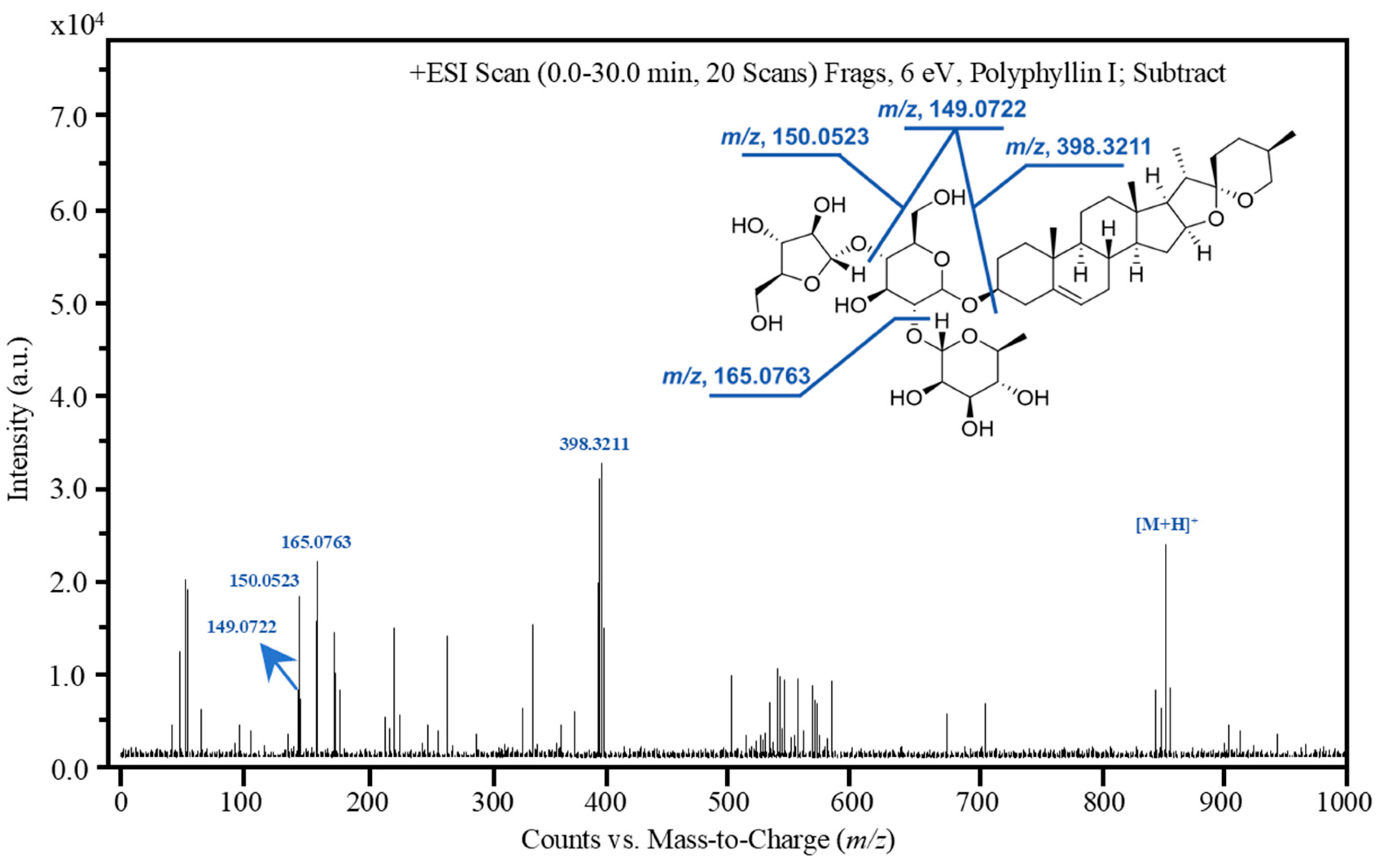
| Product ions | [C27H42O2]+, m/z, 398.3211; [C6H12O4+H]+, m/z, 149.0722; [C5H10O5]+, m/z, 150.0523; [C6H12O5+H]+, m/z, 165.0763; |
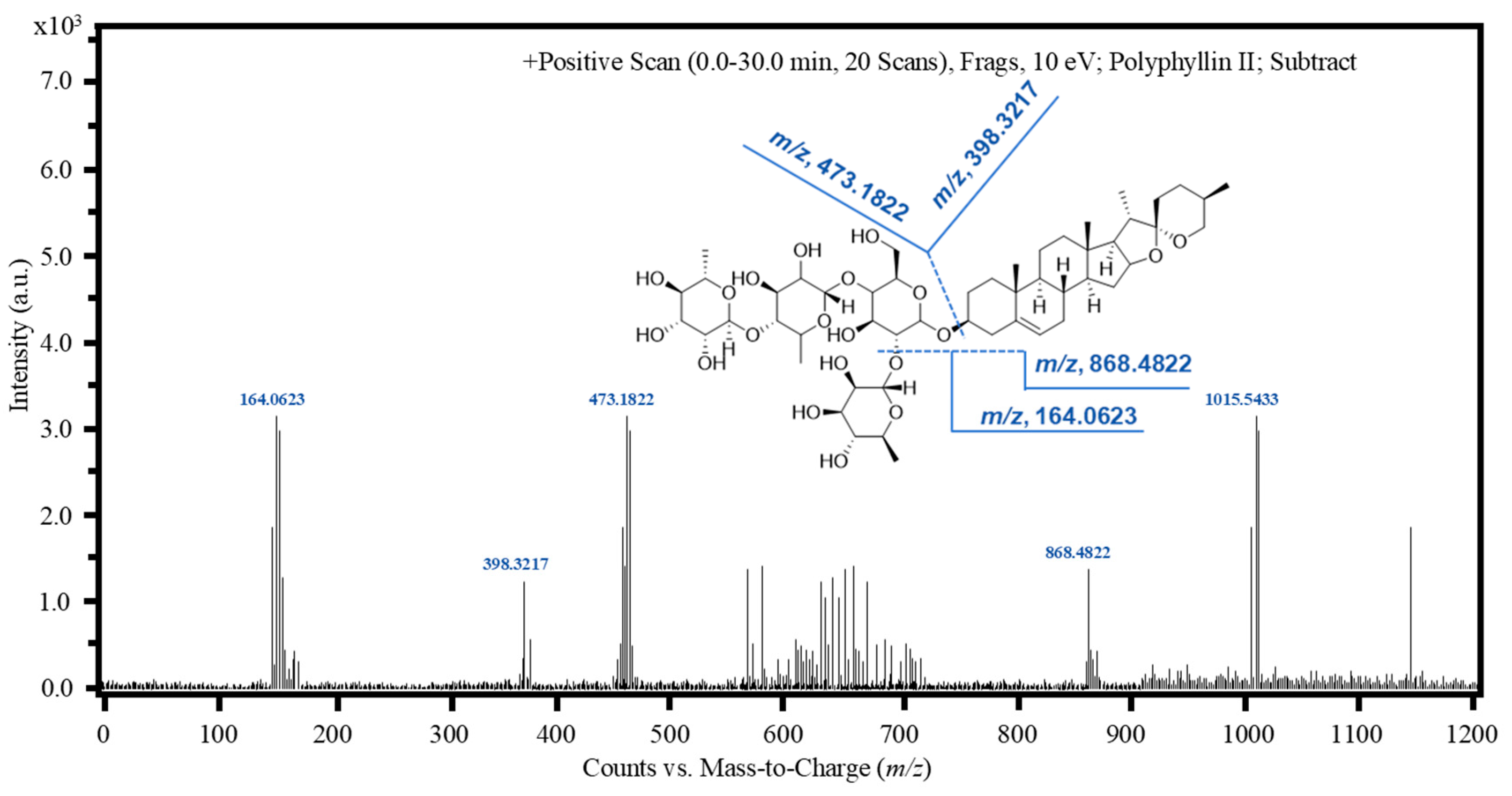
[M-C6H10O4]+, m/z, 868.4822; [C6H11O5+H]+, m/z, 164.0623; [C18H32O14+H]+, m/z, 473.1822; [C27H42O2]+, m/z, 398.3217; |
| Collision energy (eV) | 6 | 10 |
| Instrument | Waters ACQUITY UPLC system coupled with a quadrupole time-of-flight mass spectrometer (Waters Corporation, Milford, MA, USA) | Waters ACQUITY UPLC system coupled with a quadrupole time-of-flight mass spectrometer (Waters Corporation, Milford, MA, USA) |
| No. | Material-to-liquid ratio(g mL-1) | Extraction time (min) | Extraction temperature (°C) | Ultrasound power (W) | Overall separation efficiency (%) |
| 1 | 1 | 1 | 1 | 1 | 9.16 |
| 2 | 1 | 2 | 2 | 2 | 6.15 |
| 3 | 1 | 3 | 3 | 3 | 7.02 |
| 4 | 2 | 1 | 2 | 3 | 8.85 |
| 5 | 2 | 2 | 3 | 1 | 10.12 |
| 6 | 2 | 3 | 1 | 2 | 11.26 |
| 7 | 3 | 1 | 3 | 2 | 9.76 |
| 8 | 3 | 2 | 1 | 3 | 7.85 |
| 9 | 3 | 3 | 2 | 1 | 10.53 |
| K1 | 28.45 | 29.16 | 29.98 | 27.11 | |
| K2 | 28.16 | 31.45 | 28.64 | 31.66 | |
| K3 | 30.15 | 29.78 | 30.36 | 31.47 | |
| R | 1.25 | 1.06 | 0.62 | 3.15 |
| Source of variation | Sum of squares | F Value | Significance (p-value) |
| Material-to-liquid ratio (g mL-1) | 1.5783 | 0.9435 | <0.05 |
| Extraction time (min) | 1.3112 | 0.8624 | <0.05 |
| Extraction temperature (°C) | 0.4645 | 0.2645 | |
| Ultrasound power (W) | 3.0764 | 1.2305 | <0.01 |
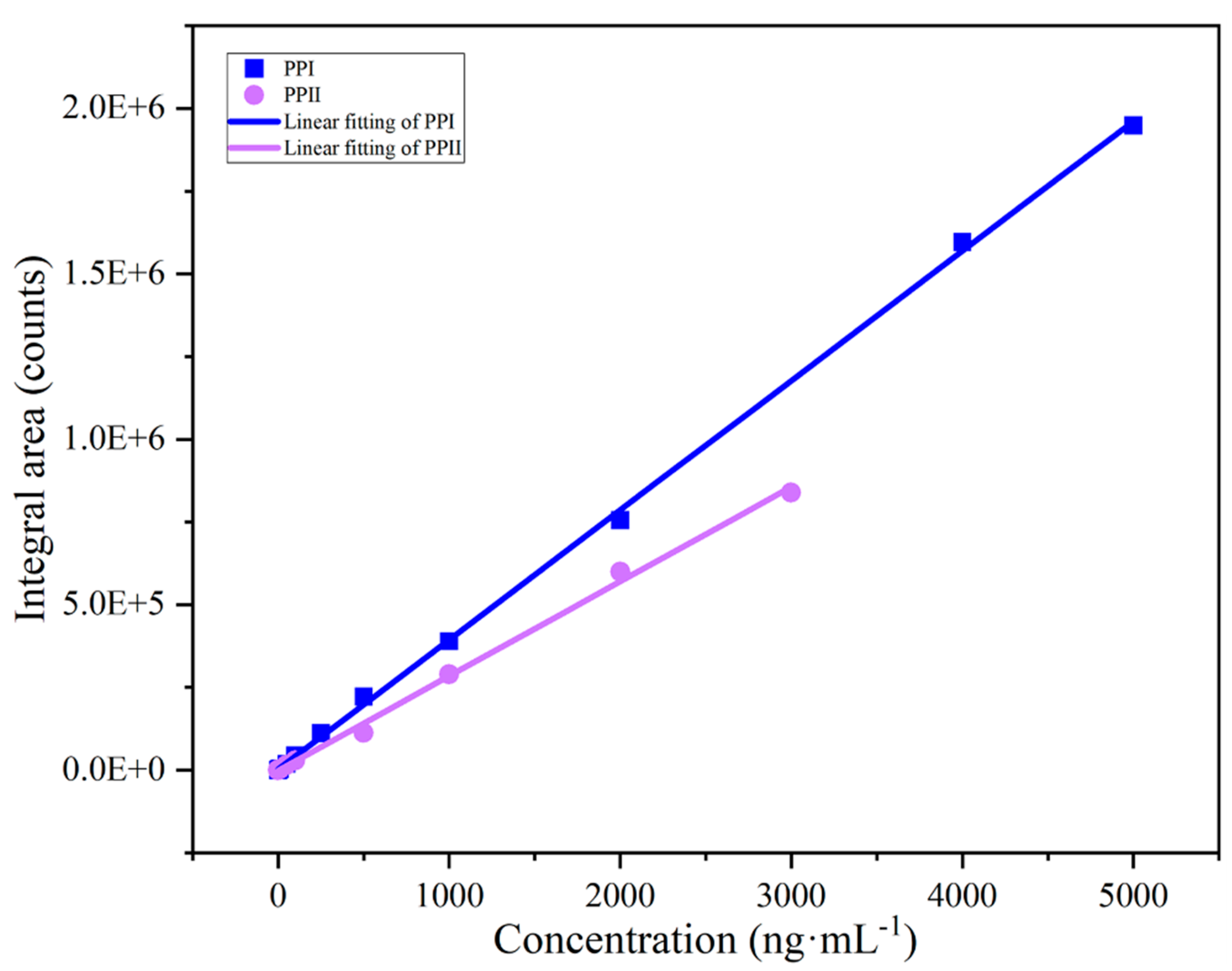
| Analyte | Linear range (ng mL-1) | R2 | Regression equation |
LOD (ng mL-1) |
LOQ (ng mL-1) |
| polyphyllin I | 0.5~4000.0 | 0.99952 | Y=2478.5592+391.6256x | 0.4391 | 0.4832 |
| polyphyllin II | 0.2~3000.0 | 0.99763 | Y=-1445.2323+285.7164x | 0.1874 | 0.2143 |
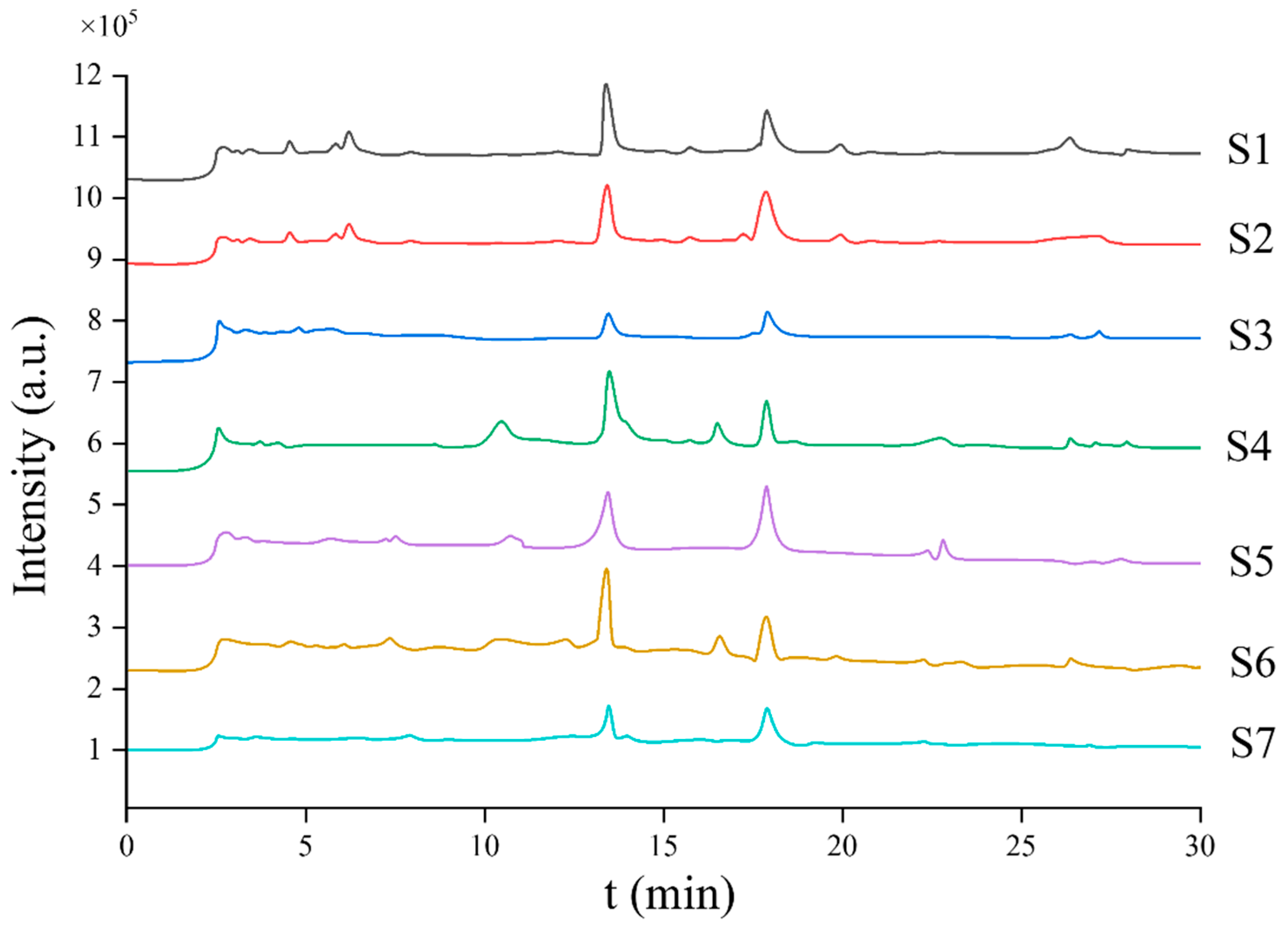
| Sample | Polyphyllin I | Polyphyllin II | ||||||||
| Content (mg·g-1) | Avg. Recovery (%) | Precision (RSD, %) | Accuracy (%) | Content (mg·g-1) | Avg. Recovery (%) | Precision (RSD, %) | Accuracy (%) | |||
| Intra-day | Inter-day | Intra-day | Inter-day | |||||||
| S1 | 7.983 | 93.28 | 1.27 | 1.33 | 97.26 | 8.921 | 97.16 | 1.09 | 1.22 | 91.99 |
| S2 | 3.282 | 98.12 | 0.47 | 0.33 | 97.36 | 4.201 | 92.17 | 0.78 | 0.66 | 93.28 |
| S3 | 7.391 | 90.18 | 1.42 | 1.22 | 97.27 | 9.721 | 83.10 | 2.07 | 1.87 | 94.27 |
| S4 | 14.298 | 88.29 | 3.91 | 3.78 | 91.03 | 11.298 | 90.17 | 1.09 | 1.21 | 92.38 |
| S5 | 9.276 | 95.28 | 2.10 | 2.07 | 94.67 | 5.292 | 96.18 | 0.99 | 1.03 | 98.71 |
| S6 | 21.452 | 99.18 | 5.01 | 4.78 | 95.27 | 17.975 | 94.78 | 4.78 | 4.97 | 91.28 |
| S7 | 17.294 | 97.91 | 4.57 | 4.23 | 96.35 | 16.392 | 99.10 | 3.89 | 3.76 | 98.13 |
| Traditional Chinese Medicine | Total Separation Efficiency (%) | Deviation of Efficiency (%) |
| Paris polyphylla Smith | 14.33 | 2.87 |
| Typhonium giganteum | 2.04 | 0.32 |
| Iphigenia indica | 6.98 | 1.02 |
| Chinese Paris Rhizome | 7.47 | 1.76 |
| Yunnan Paris Rhizome | 14.63 | 3.22 |
| Large-leaved Paris Rhizome | 4.92 | 0.89 |
| Root of Paris polyphylla | 16.51 | 4.04 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




