Submitted:
13 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Type of Study
2.2. Review Question
- Population: In vivo models of AD, dementia, cognitive decline, aging, oxidative stress or mitochondrial dysfunction.
- Intervention: Natural vitamin antioxidants and mitochondria-targeted antioxidants.
- Comparison: Control or usual care.
- Outcome: Cognition, inflammation, mitochondrial function or other cognitive health markers
2.3. Eligibility Criteria
- Inclusion criteria: English language, in vivo, specified antioxidants, outcome: dementia, cognition, cognitive or mitochondrial health.
- Exclusion criteria: Non- English language, non in vivo, multivitamin supplementations or vitamin pre-cursors, irrelevant outcomes.
2.4. Information Sources and Search Strategy
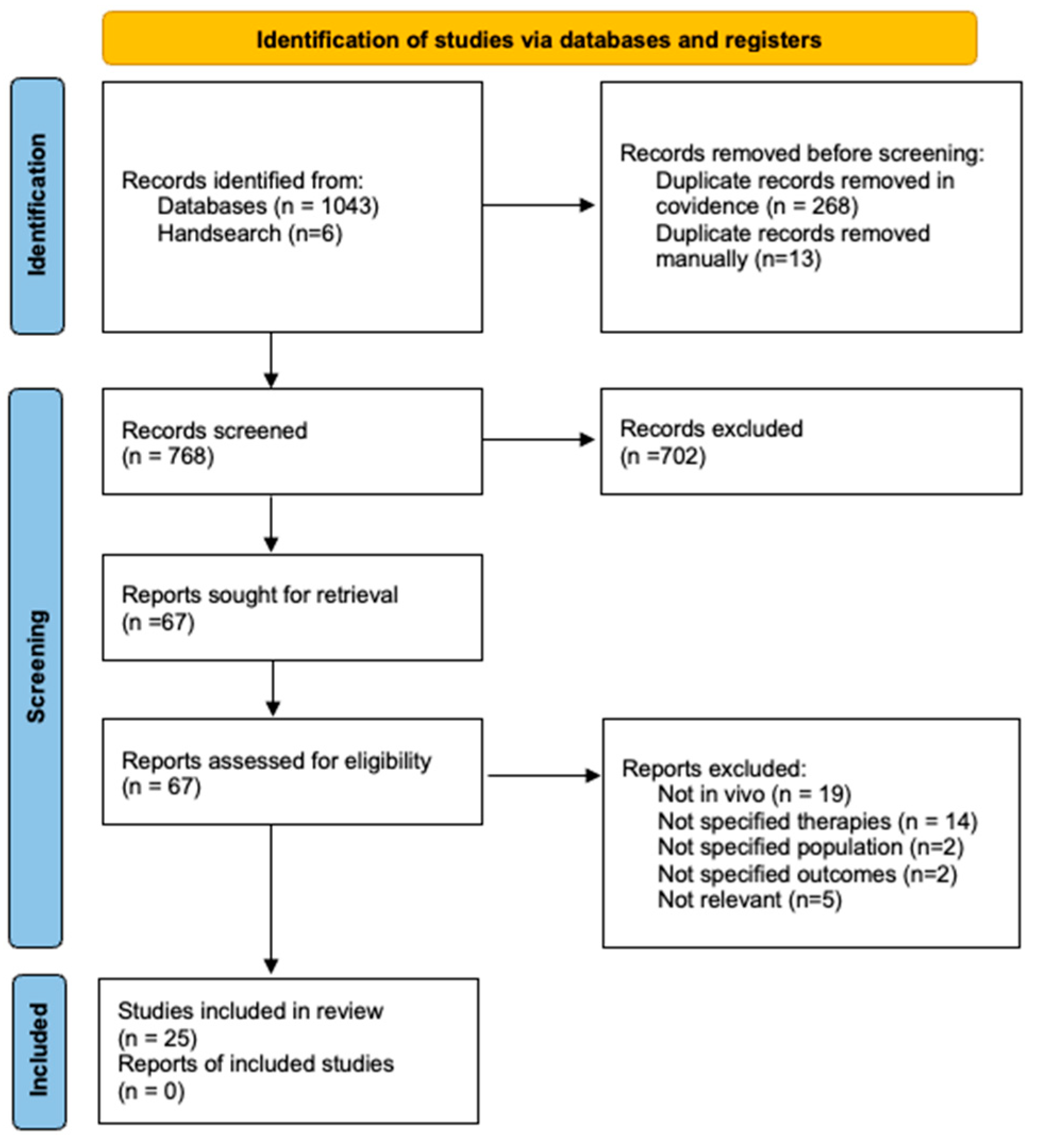
2.5. Selection of Sources of Evidence
2.6. Data Charting Process and Items
2.7. Synthesis of Results
2.8. Patients and Participants Involvement
3. Results
3.1. Natural Vitamin Antioxidants
3.1.1. Vitamin E
3.1.2. Vitamin A
3.1.3. Methylcobalamin (Vitamin B12)
3.1.4. Folate (Vitamin B9)
3.1.5. Niacin (Vitamin B3)
3.1.6. Thiamine (Vitamin B1)
3.1.7. Pantetheine (Vitamin B5)
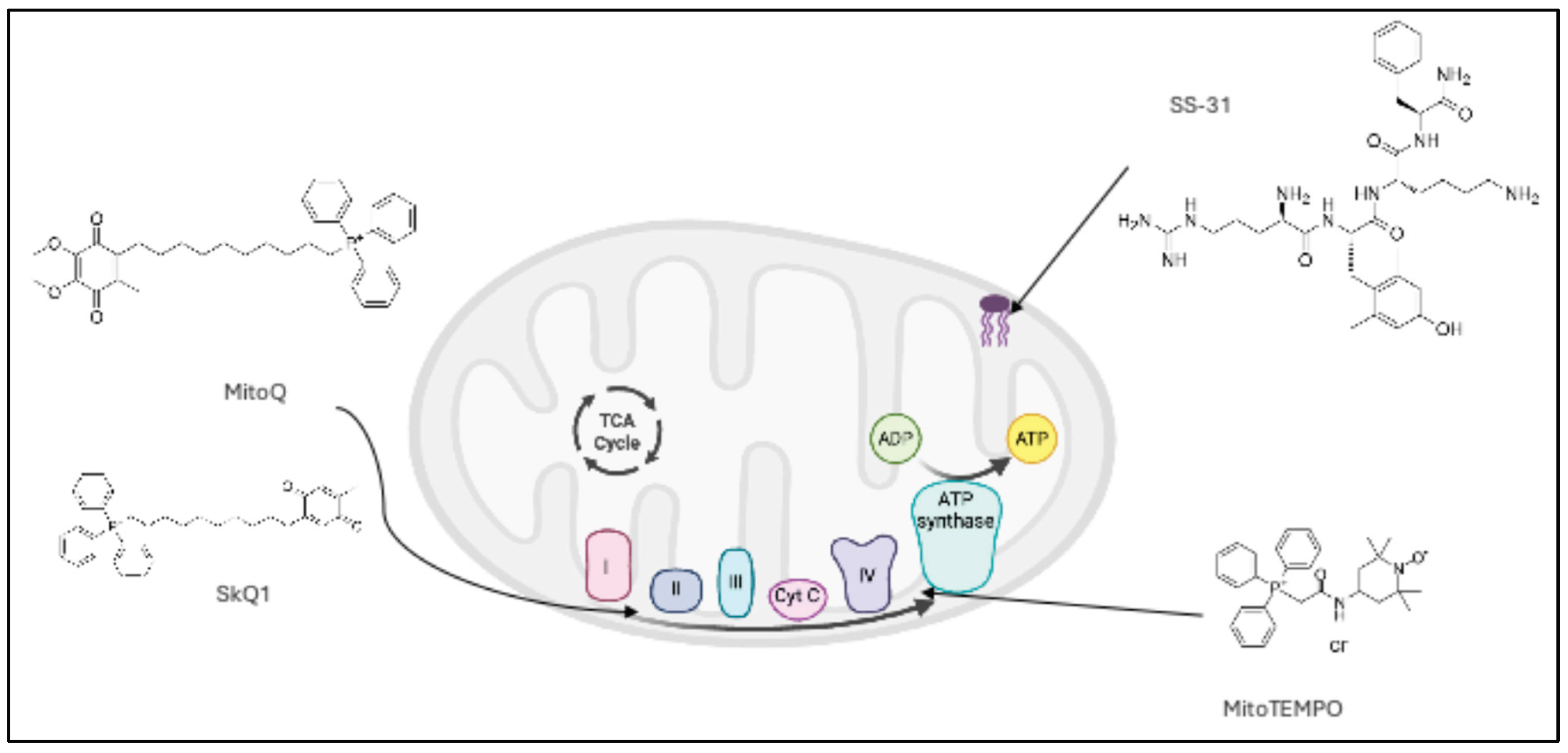
3.2. Mitochondria Targeted Antioxidants
3.2.1. MitoQ
3.2.2. MitoTEMPO
3.2.3. SS31
3.2.4. SkQ1
4. Discussion
4.1. Natural Vitamin Antioxidants
4.2. Mitochondria Targeted Antioxidants
4.3. Comparative Outcomes and Clinical Relevance
4.4. Strengths & Limitations
4.5. Implications & Future Recommendations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jones, A.; Ali, M.U.; Kenny, M.; Mayhew, A.; Mokashi, V.; He, H.; Lin, S.; Yavari, E.; Paik, K.; Subramanian, D. Potentially modifiable risk factors for dementia and mild cognitive impairment: an umbrella review and meta-analysis. In Dementia and Geriatric Cognitive Disorders; 2024. [Google Scholar]
- Steinmetz, J.D.; Seeher, K.M.; Schiess, N.; Nichols, E.; Cao, B.; Servili, C.; Cavallera, V.; Cousin, E.; Hagins, H.; Moberg, M.E. Global, regional, and national burden of disorders affecting the nervous system, 1990–2021: a systematic analysis for the Global Burden of Disease Study 2021. The Lancet Neurology 2024, 23, 344–381. [Google Scholar] [CrossRef]
- Australian Government. Dementia in Australia; 2024. [Google Scholar]
- Langa, K.M.; Levine, D.A. The diagnosis and management of mild cognitive impairment: a clinical review. Jama 2014, 312, 2551–2561. [Google Scholar] [CrossRef]
- Stanga, S.; Caretto, A.; Boido, M.; Vercelli, A. Mitochondrial Dysfunctions: A Red Thread across Neurodegenerative Diseases. International Journal of Molecular Sciences 2020, 21, 3719. [Google Scholar] [CrossRef]
- Fields, M.; Marcuzzi, A.; Gonelli, A.; Celeghini, C.; Maximova, N.; Rimondi, E. Mitochondria-targeted antioxidants, an innovative class of antioxidant compounds for neurodegenerative diseases: perspectives and limitations. International journal of molecular sciences 2023, 24, 3739. [Google Scholar] [CrossRef] [PubMed]
- Misrani, A.; Tabassum, S.; Yang, L. Mitochondrial dysfunction and oxidative stress in Alzheimer’s disease. Frontiers in aging neuroscience 2021, 13, 57. [Google Scholar] [CrossRef] [PubMed]
- Johri, A. Disentangling Mitochondria in Alzheimer’s Disease. International Journal of Molecular Sciences 2021, 22, 11520. [Google Scholar] [CrossRef]
- Smith, R.A.J.; Hartley, R.C.; Cochemé, H.M.; Murphy, M.P. Mitochondrial pharmacology. Trends in Pharmacological Sciences 2012, 33, 341–352. [Google Scholar] [CrossRef]
- Almikhlafi, M.A.; Karami, M.M.; Jana, A.; Alqurashi, T.M.; Majrashi, M.; Alghamdi, B.S.; Ashraf, G.M. Mitochondrial medicine: A promising therapeutic option against various neurodegenerative disorders. Current Neuropharmacology 2023, 21, 1165. [Google Scholar] [CrossRef]
- Bordoni, L.; Gabbianelli, R. Mitochondrial DNA and Neurodegeneration: Any Role for Dietary Antioxidants? Antioxidants 2020, 9, 764. [Google Scholar] [CrossRef]
- Gil Martínez, V.; Avedillo Salas, A.; Santander Ballestín, S. Vitamin supplementation and dementia: a systematic review. Nutrients 2022, 14, 1033. [Google Scholar] [CrossRef] [PubMed]
- Altieri, F.; Turco, E.M.; Vinci, E.; Torres, B.; Ferrari, D.; De Jaco, A.; Mazzoccoli, G.; Lamorte, G.; Nardone, A.; Della Monica, M.; et al. Production and characterization of CSSI003 (2961) human induced pluripotent stem cells (iPSCs) carrying a novel puntiform mutation in RAI1 gene, Causative of Smith-Magenis syndrome. Stem Cell Research 2018, 28, 153–156. [Google Scholar] [CrossRef]
- Rossman, M.J.; Santos-Parker, J.R.; Steward, C.A.C.; Bispham, N.Z.; Cuevas, L.M.; Rosenberg, H.L.; Woodward, K.A.; Chonchol, M.; Gioscia-Ryan, R.A.; Murphy, M.P.; et al. Chronic Supplementation With a Mitochondrial Antioxidant (MitoQ) Improves Vascular Function in Healthy Older Adults. Hypertension 2018, 71, 1056–1063. [Google Scholar] [CrossRef]
- Petrov, A.; Perekhvatova, N.; Skulachev, M.; Stein, L.; Ousler, G. SkQ1 ophthalmic solution for dry eye treatment: results of a phase 2 safety and efficacy clinical study in the environment and during challenge in the controlled adverse environment model. Advances in therapy 2016, 33, 96–115. [Google Scholar] [CrossRef]
- Tapias, V.; Jainuddin, S.; Ahuja, M.; Stack, C.; Elipenahli, C.; Vignisse, J.; Gerges, M.; Starkova, N.; Xu, H.; Starkov, A.A.; et al. Benfotiamine treatment activates the Nrf2/ARE pathway and is neuroprotective in a transgenic mouse model of tauopathy. Human Molecular Genetics 2018, 27(16), 2874–2892. [Google Scholar] [CrossRef]
- Anton-Fernandez, A.; Cuadros, R.; Peinado-Cahuchola, R.; Hernandez, F.; Avila, J. The role of folate receptor alpha in the partial rejuvenation of dentate gyrus cells. Improvement of cognitive function in elderly mice. bioRxiv 2023, 03. [Google Scholar]
- Sun, S.; Lu, W.; Zhang, C.; Wang, G.; Hou, Y.; Zhou, J.; Wang, Y. Folic acid and S-adenosylmethionine reverse Homocysteine-induced Alzheimer’s disease-like pathological changes in rat hippocampus by modulating PS1 and PP2A methylation levels. Brain Research 2024, 1841, 149095. [Google Scholar] [CrossRef]
- Ji, X.; Li, C.; Yu, W.; Cai, Y.; Lu, L.; Qian, Q.; Hu, Y.; Zhu, X.; Wang, H. Methylcobalamin Alleviates Neuronal Apoptosis and Cognitive Decline Induced by PM2.5 Exposure in Mice. Journal of Alzheimer’s Disease 2022, 86(4), 1783–1796. [Google Scholar] [CrossRef] [PubMed]
- Gong, B.; Pan, Y.; Vempati, P.; Zhao, W.; Knable, L.; Ho, L.; Wang, J.; Sastre, M.; Ono, K.; Sauve, A.A.; et al. Nicotinamide riboside restores cognition through an upregulation of proliferator-activated receptor-gamma coactivator 1alpha regulated beta-secretase 1 degradation and mitochondrial gene expression in Alzheimer’s mouse models. Neurobiology of Aging 2013, 34(6), 1581–1588. [Google Scholar] [CrossRef] [PubMed]
- Marmolejo-Garza, A.; Chatre, L.; Croteau, D.L.; Herron-Bedoya, A.; Luu, M.D.A.; Bernay, B.; Pontin, J.; Bohr, V.A.; Boddeke, E.; Dolga, A.M. Nicotinamide riboside modulates the reactive species interactome, bioenergetic status and proteomic landscape in a brain-region-specific manner. Neurobiology of Disease 2024, 200, 106645. [Google Scholar] [CrossRef] [PubMed]
- Baranger, K.; van Gijsel-Bonnello, M.; Stephan, D.; Carpentier, W.; Rivera, S.; Khrestchatisky, M.; Gharib, B.; De Reggi, M.; Benech, P. Long-Term Pantethine Treatment Counteracts Pathologic Gene Dysregulation and Decreases Alzheimer’s Disease Pathogenesis in a Transgenic Mouse Model. Neurotherapeutics 2019, 16(4), 1237–1254. [Google Scholar] [CrossRef]
- Etchamendy, N.; Enderlin, V.; Marighetto, A.; Vouimba, R.M.; Pallet, V.; Jaffard, R.; Higueret, P. Alleviation of a selective age-related relational memory deficit in mice by pharmacologically induced normalization of brain retinoid signaling. Journal of Neuroscience 2001, 21, 6423–6429. [Google Scholar] [CrossRef]
- Takamura, R.; Watamura, N.; Nikkuni, M.; Ohshima, T. All-trans retinoic acid improved impaired proliferation of neural stem cells and suppressed microglial activation in the hippocampus in an Alzheimer’s mouse model. Journal of Neuroscience Research 2017, 95(3), 897–906. [Google Scholar] [CrossRef]
- Kolosova, N.G.; Shcheglova, T.V.; Sergeeva, S.V.; Loskutova, L.V. Long-term antioxidant supplementation attenuates oxidative stress markers and cognitive deficits in senescent-accelerated OXYS rats. Neurobiology of Aging 2017, 27, 1289–1297. [Google Scholar] [CrossRef]
- Garcia-Alloza, M.; Dodwell, S.A.; Meyer-Luehmann, M.; Hyman, B.T.; Bacskai, B.J. Plaque-derived oxidative stress mediates distorted neurite trajectories in the Alzheimer mouse model. Journal of Neuropathology & Experimental Neurology 2006, 65, 1082–1089. [Google Scholar] [CrossRef]
- Liu, X.; Zhang, Y.; Yang, X. PM2.5 induced neurodegenerative-like changes in mice and the antagonistic effects of vitamin E. Toxicology Research 2019, 8, 172–179. [Google Scholar] [CrossRef] [PubMed]
- Cowan, C.M.; Sealey, M.A.; Mudher, A. Suppression of tau-induced phenotypes by vitamin E demonstrates the dissociation of oxidative stress and phosphorylation in mechanisms of tau toxicity. Journal of Neurochemistry 2021, 157, 684–694. [Google Scholar] [CrossRef] [PubMed]
- McManus, M.J.; Murphy, M.P.; Franklin, J.L. The mitochondria-targeted antioxidant mitoq prevents loss of spatial memory retention and early neuropathology in a transgenic mouse model of Alzheimer’s disease. Journal of Neuroscience 2011, 31(44), 15703–15715. [Google Scholar] [CrossRef]
- Ng, L.F.; Gruber, J.; Cheah, I.K.; Goo, C.K.; Cheong, W.F.; Shui, G.; Sit, K.P.; Wenk, M.R.; Halliwell, B. The mitochondria-targeted antioxidant MitoQ extends lifespan and improves healthspan of a transgenic Caenorhabditis elegans model of Alzheimer disease. Free Radical Biology and Medicine 2014, 71, 390–401. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.; Chen, Z.; Huang, C.; Wu, Y.; Huang, L.; Wang, L.; Ke, S.; Liu, L. Mito-TEMPO, a mitochondria-targeted antioxidant, improves cognitive dysfunction due to hypoglycemia: an association with reduced pericyte loss and blood-brain barrier leakage. Molecular Neurobiology 2023, 60, 672–686. [Google Scholar] [CrossRef] [PubMed]
- Kapay, N.A.; Popova, O.V.; Isaev, N.K.; Stelmashook, E.V.; Kondratenko, R.V.; Zorov, D.B.; Skrebitsky, V.G.; Skulachev, V.P. Mitochondria-targeted plastoquinone antioxidant SkQ1 prevents amyloid-beta-induced impairment of long-term potentiation in rat hippocampal slices. Journal of Alzheimer’s Disease 2013, 36, 377–383. [Google Scholar] [CrossRef]
- Stefanova, N.A.; Muraleva, N.A.; Skulachev, V.P.; Kolosova, N.G. Alzheimer’s disease-like pathology in senescence-accelerated OXYS rats can be partially retarded with mitochondria-targeted antioxidant SkQ1. Journal of Alzheimer’s disease 2013, 38, 681–694. [Google Scholar] [CrossRef] [PubMed]
- Stefanova, N.A.; Muraleva, N.A.; Maksimova, K.Y.; Rudnitskaya, E.A.; Kiseleva, E.; Telegina, D.V.; Kolosova, N. An antioxidant specifically targeting mitochondria delays progression of Alzheimer’s disease-like pathology. Aging (Albany NY) 2016, 8, 2713. [Google Scholar] [CrossRef]
- Kolosova, N.G.; Tyumentsev, M.A.; Muraleva, N.A.; Kiseleva, E.; Vitovtov, A.O.; Stefanova, N.A. Antioxidant SkQ1 alleviates signs of Alzheimer’s disease-like pathology in old OXYS rats by reversing mitochondrial deterioration. Current Alzheimer Research 2017, 14, 1283–1292. [Google Scholar] [CrossRef]
- Jia, Y.-L.; Sun, S.-J.; Chen, J.-H.; Jia, Q.; Huo, T.-T.; Chu, L.-F.; Bai, J.-T.; Yu, Y.-J.; Yan, X.-X.; Wang, J.-H. SS31, a small molecule antioxidant peptide, attenuates β-amyloid elevation, mitochondrial/synaptic deterioration and cognitive deficit in SAMP8 mice. Current Alzheimer Research 2016, 13, 297–306. [Google Scholar] [CrossRef]
- Reddy, P.H.; Manczak, M.; Kandimalla, R. Mitochondria-targeted small molecule SS31: a potential candidate for the treatment of Alzheimer’s disease. Human molecular genetics 2017, 26, 1483–1496. [Google Scholar] [CrossRef]
- Zhao, W.; Xu, Z.; Cao, J.; Fu, Q.; Wu, Y.; Zhang, X.; Long, Y.; Zhang, X.; Yang, Y.; Li, Y. Elamipretide (SS-31) improves mitochondrial dysfunction, synaptic and memory impairment induced by lipopolysaccharide in mice. Journal of Neuroinflammation 2019, 16, 1–19. [Google Scholar] [CrossRef]
- Jia, Y.-L.; Wang, W.; Han, N.; Sun, H.-L.; Dong, F.-M.; Song, Y.-X.; Feng, R.-F.; Wang, J.-H. The mitochondria-targeted small molecule SS31 delays progression of behavioral deficits by attenuating β-amyloid plaque formation and mitochondrial/synaptic deterioration in APP/PS1 mice. Biochemical and Biophysical Research Communications 2023, 658, 36–43. [Google Scholar] [CrossRef]
- Calvo-Rodriguez, M.; Kharitonova, E.K.; Snyder, A.C.; Hou, S.S.; Sanchez-Mico, M.V.; Das, S.; Fan, Z.; Shirani, H.; Nilsson, K.P.R.; Serrano-Pozo, A.; et al. Real-time imaging of mitochondrial redox reveals increased mitochondrial oxidative stress associated with amyloid beta aggregates in vivo in a mouse model of Alzheimer’s disease. Molecular Neurodegeneration 2024, 19(1), no pagination. [Google Scholar] [CrossRef]
- Murphy, M.P.; Smith, R.A.J. Targeting antioxidants to mitochondria by conjugation to lipophilic cations. Annual review of pharmacology and toxicology 2007, 47, 629–656. [Google Scholar] [CrossRef] [PubMed]
- Skulachev, V.P.; Anisimov, V.N.; Antonenko, Y.N.; Bakeeva, L.E.; Chernyak, B.V.; Erichev, V.P.; Filenko, O.F.; Kalinina, N.I.; Kapelko, V.I.; Kolosova, N.G.; et al. An attempt to prevent senescence: A mitochondrial approach. Biochimica et Biophysica Acta (BBA) - Bioenergetics 2009, 1787, 437–461. [Google Scholar] [CrossRef] [PubMed]
- Vetoshkina, D.V.; Nikolaev, A.A.; Borisova-Mubarakshina, M.M. Antioxidant Properties of Plant Plastoquinone in Vivo and in Vitro. Biophysics 2024, 69, 445–458. [Google Scholar] [CrossRef]
- Yamada, Y.; Satrialdi; Hibino, M.; Sasaki, D.; Abe, J.; Harashima, H. Power of mitochondrial drug delivery systems to produce innovative nanomedicines. Advanced Drug Delivery Reviews 2020, 154-155, 187–209. [Google Scholar] [CrossRef]
- DeJulius, C.R.; Dollinger, B.R.; Kavanaugh, T.E.; Dailing, E.; Yu, F.; Gulati, S.; Miskalis, A.; Zhang, C.; Uddin, J.; Dikalov, S.; et al. Optimizing an Antioxidant TEMPO Copolymer for Reactive Oxygen Species Scavenging and Anti-Inflammatory Effects in Vivo. Bioconjugate Chemistry 2021, 32, 928–941. [Google Scholar] [CrossRef]
- Szeto, H.H. Mitochondria-targeted peptide antioxidants: Novel neuroprotective agents. The AAPS Journal 2006, 8, 62. [Google Scholar] [CrossRef]
- Zhao, K.; Zhao, G.-M.; Wu, D.; Soong, Y.; Birk, A.V.; Schiller, P.W.; Szeto, H.H. Cell-permeable peptide antioxidants targeted to inner mitochondrial membrane inhibit mitochondrial swelling, oxidative cell death, and reperfusion injury. Journal of Biological Chemistry 2004, 279, 34682–34690. [Google Scholar] [CrossRef] [PubMed]
- Kontush, A.; Schekatolina, S. Vitamin E in neurodegenerative disorders: Alzheimer’s disease. Annals of the New York Academy of Sciences 2004, 1031, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Varesi, A.; Campagnoli, L.I.M.; Carrara, A.; Pola, I.; Floris, E.; Ricevuti, G.; Chirumbolo, S.; Pascale, A. Non-Enzymatic Antioxidants against Alzheimer’s Disease: Prevention, Diagnosis and Therapy. Antioxidants 2023, 12, 180. [Google Scholar] [CrossRef]
- Caldwell, C.C.; Yao, J.; Brinton, R.D. Targeting the Prodromal Stage of Alzheimer’s Disease: Bioenergetic and Mitochondrial Opportunities. Neurotherapeutics 2015, 12, 66–80. [Google Scholar] [CrossRef]
- Teixeira, J.; Silva, T.; Andrade, P.B.; Borges, F. Alzheimer’s disease and antioxidant therapy: How long how far? Current Medicinal Chemistry 2013, 20(24), 2939–2952. [Google Scholar] [CrossRef]
- Ferreira, M.E.; de Vasconcelos, A.S.; da Costa Vilhena, T.; da Silva, T.L.; da Silva Barbosa, A.; Gomes, A.R.; Dolabela, M.F.; Percário, S. Oxidative Stress in Alzheimer’s Disease: Should We Keep Trying Antioxidant Therapies? Cell Mol Neurobiol 2015, 35, 595–614. [Google Scholar] [CrossRef]
- Mecocci, P.; Polidori, M.C. Antioxidant clinical trials in mild cognitive impairment and Alzheimer’s disease. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease 2012, 1822, 631–638. [Google Scholar] [CrossRef] [PubMed]
- Mecocci, P.; Polidori, M.C.; Cherubini, A.; Ingegni, T.; Mattioli, P.; Catani, M.; Rinaldi, P.; Cecchetti, R.; Stahl, W.; Senin, U.; et al. Lymphocyte oxidative DNA damage and plasma antioxidants in Alzheimer disease. Archives of Neurology 2022, 59, 794–798. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Apostolopoulos, V. B Vitamins and Ageing. In Biochemistry and Cell Biology of Ageing: Part I Biomedical Science; Harris, J.R., Korolchuk, V.I., Eds.; Springer Singapore: Singapore, 2018; pp. 451–470. [Google Scholar]
- Wang, Z.; Zhu, W.; Xing, Y.; Jia, J.; Tang, Y. B vitamins and prevention of cognitive decline and incident dementia: a systematic review and meta-analysis. Nutrition Reviews 2021, 80, 931–949. [Google Scholar] [CrossRef]
- Weissig, V. Drug Development for the Therapy of Mitochondrial Diseases. Trends in Molecular Medicine 2020, 26, 40–57. [Google Scholar] [CrossRef]
- Kolosova, N.G.; Kozhevnikova, O.S.; Muraleva, N.A.; Rudnitskaya, E.A.; Rumyantseva, Y.V.; Stefanova, N.A.; Telegina, D.V.; Tyumentsev, M.A.; Fursova, A.Z. SkQ1 as a Tool for Controlling Accelerated Senescence Program: Experiments with OXYS Rats. Biochemistry (Moscow) 2022, 87, 1552–1562. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Search results for “mitochondria-targeted antioxidant AND dementia OR cognition”. Available online: https://clinicaltrials.gov (accessed on 15 May 2025).
- Lushchak, O.; Gospodaryov, D.; Strilbytska, O.; Bayliak, M. Chapter Five - Changing ROS, NAD and AMP: A path to longevity via mitochondrial therapeutics. In Advances in Protein Chemistry and Structural Biology; Çakatay, U., Atayik, M.C., Eds.; Academic Press, 2023; Volume 136, pp. 157–196. [Google Scholar]
- Antonenko, Y.N.; Avetisyan, A.V.; Bakeeva, L.E.; Chernyak, B.V.; Chertkov, V.A.; Domnina, L.V.; Ivanova, O.Y.; Izyumov, D.S.; Khailova, L.S.; Klishin, S.S.; et al. Mitochondria-targeted plastoquinone derivatives as tools to interrupt execution of the aging program. 1. Cationic plastoquinone derivatives: Synthesis and in vitro studies. Biochemistry (Moscow) 2008, 73, 1273–1287. [Google Scholar] [CrossRef] [PubMed]
| Title | Author/s | Year | Country | Aims | Population (Tx) | Tx, Dose, Duration | Methods | Results |
|---|---|---|---|---|---|---|---|---|
| Benfotiamine treatment activates the Nrf2/ARE pathway and is neuroprotective in a transgenic mouse model of tauopathy [16] | Tapias et al. | 2018 | USA | To investigate whether benfotiamine prevents the formation of NFTs and confers neuroprotection in a mouse model of tauopathy. | P301S TG mice 1mo (F:M 1:1) | Benfotiamine, 200mg/kg PO, 9 mo | Mice were treated as described from 1 month of age and cognitively tested at 5,7 and 9 months of age. They were sacrificed at 10 months of age for histopathological analysis. | Benfotiamine reduced NFTs, was neuroprotective and reduced mitochondrial dysfunction, resulting in improved cognition and lifespan in a mouse model of tauopathy. |
| The role of folate receptor alpha in the partial rejuvenation of dentate gyrus cells. Improvement of cognitive function in elderly mice [17]. | Antón- Fernández et al. | 2024 | Spain | To explore whether aged mice brain cells can be rejuvenated through use of methyl donors, folate and methionine. | C57BL/6 wild-type mice 21mo (M n=1, F n=6) | Folate, 0.25mg/ml, Once | Treatment as listed was injected into the DG of mice brain hemispheres. Mice then underwent cognitive testing following sacrifice and extraction of brain tissue. | Infusion with folate in aged mice was able to improve some aspects of cognition and rejuvenate DG cells, independent of sex. |
| Folic acid and S-adenosylmethionine reverse Homocysteine-induced Alzheimer’s disease-like pathological changes in rat hippocampus by modulating PS1 and PP2A methylation levels [18] | Sun et al. | 2024 | China | To elucidate the mechanism by which elevated homocysteine levels cause AD like pathology and whether folic acid and S-adenosylmethionine improve this in vitro and in vivo. | Wistar rats (M, n=32) | FA , 2mg/kg, IP, 4 wks | Rats were treated with IP Hcy +/- FA as described, compared to controls then sacrificed for plasma and hippocampal analyses. | Rats exposed to Hcy and treated with FA showed decreased plasma Hcy. Rat hippocampal neurons showed decreeased burden and markers of both Aβ and tau. |
| Methylcobalamin Alleviates Neuronal Apoptosis and Cognitive Decline Induced by PM2.5 Exposure in Mice [19] | Ji et al. | 2022 | China | To explore whether Vitamin B12 is neuroprotective against cognitive impairment and apoptosis induced by chronic PM2.5 exposure. | C57BL/6 mice, 6wk (M, n=8) | MeCbl 1.25mg/L, PO, 6 mo | Mice were exposed to either filtered air or PM2.5 12 hours per day, 6 days per week and treated with MeCbl compared to negative controls. They were then subjected to cognitive testing and sacrificed for brain tissue and blood analysis. | MeCbl treatment in mice was able to reduce PM2.5 induced neuronal apoptosis, via improving markers of oxidative stress and mitochondrial function, resulting in improved cognition. |
| Nicotinamide riboside restores cognition through an upregulation of proliferator-activated receptor-γ coactivator 1α regulated β-secretase 1 degradation and mitochondrial gene expression in Alzheimer’s mouse models [20] | Gong et al. | 2013 | USA | To investigate if NR treatment in an AD mouse model could attenuate Aβ toxicity through activation of PGC-1a-mediated BACE1 degradation. | PGC-1α/Tg2576 mice, 5-6mo | NR, 250mg/kg PO, 3 mo | Mice were treated as described, compared to controls. Then subjected to cognitive testing and sacrificed for brain tissue analysis. | NR attenuated Aβ toxicity, improved synaptic plasticity, abolished reduced LTP and improved cognitive function in a mouse model of AD. |
| Nicotinamide riboside modulates the reactive species interactome, bioenergetic status and proteomic landscape in a brain-region-specific manner [21] |
Marmolejo-Garza et al. | 2024 | Netherlands | To investigate the effects of nicotinamide riboside (NR) on apoptosis, inflammation and protein expression for both in vitro and in vivo models of AD. | 3xPB mice, 16-18mo | NR, 12mM , 8 wks | Mice were treated as described, compared to controls. They were then sacrificed and brain tissue was analysed. | NR treatment reduced oxidative stress, apoptosis and some proteomic changes in an AD mouse model. |
| Long-Term Pantetheine Treatment Counteracts Pathologic Gene Dysregulation and Decreases Alzheimer’s Disease Pathogenesis in a Transgenic Mouse Model [22] | Baranger et al. | 2019 | France | To investigate the potential in vivo benefits of pantetheine supplementation in a mouse model of AD. | 5XFAD mice 1.5mo (n=10) | Pantetheine 15mg, IP, 3x/ wk 5.5 mo | Mice were treated as described at 1.5 months of age, subjected to behavioural testing at 7 months, then sacrificed for brain histopathological analysis. | Pantetheine supplementation in a mouse model reversed AD induced astrogliosis, microgliosis, Aβ plaque burden and aggressive behaviour. |
| Alleviation of a Selective Age-Related Relational Memory Deficit in Mice by Pharmacologically Induced Normalization of Brain Retinoid Signaling [23] | Etchamendy et al. |
2001 | France | To determine the effect of aging on LTP in the brain of aged mice and whether RA has a promnesic effect. | Male mice C57BL/6 Jico inbred strain 21-23mo | RA (150ug/kg) (n=10) or RA + CD3106 (n=6) | Aged or control mice were injected w RA as described with or without RAR antagonist before undergoing cognitive testing followed by sacrifice and brain tissue analysis. | RA treatment partially improved LTP, increased brain expression of RAR and improved cognitive function in aged mice. |
| All-Trans Retinoic Acid Improved Impaired Proliferation of Neural Stem Cells and Suppressed Microglial Activation in the Hippocampus in an Alzheimer’s Mouse Model [24] | Takamura et al. | 2017 | Japan | To determine whether the suppression of NSC proliferation in a mouse model of AD is impacted by ATRA (all trans retinoic acid). | 3xTg AD mice 12mo (F) | ATRA, 20mg/kg, IP 3x/wk for 4 wks | Mice were treated as described with ATRA or control, then sacrificed and brain tissue extracted for histological analysis. | ATRA improved NSC proliferation and reduced neuroinflammation in a mouse model of AD. |
| Long-term antioxidant supplementation attenuates oxidative stress markers and cognitive deficits in senescent-accelerated OXYS rats [25] | Kolosova et al. | 2006 | Russia | To verify the correlation between brain aging and oxidative stress and investigate the effect of long-term vitamin E or flavonoid supplementation in preventing cognitive deficits. | Male OXYs + Wistar rats | α-tocopherol, 30mg/kg, 4 courses | Rats were treated in 4 courses as described in 2 month increments with either vitamin E or a flavonoid starting at 3mo old and compared to controls. Cognitive testing was undertaken, and rats were sacrificed for brain and serum analyses. | α-tocopherol treated OXYS rats had improved short term memory and decreased markers of oxidative stress. |
| Plaque-Derived Oxidative Stress Mediates Distorted Neurite Trajectories in the Alzheimer Mouse Model [26] | Garcia-Alloza et al. | 2006 | USA | To determine if antioxidants vitamin E and ginko biloba diminish reactive oxygen species in a mouse model of AD. | APPswe/PS1d9 mice 6-10 mo | Trolox, 210mg/kg, 24 hrs/15 d | The acute treatment group received Trolox prior to cortical surgery, then were imaged. The chronic treatment group were treated as described for 15 days, sacrificed then imaged. | Trolox treatment decreased Aβ plaque-related oxidative stress and dystrophic neurites in a mouse model AD. |
| PM2.5 induced neurodegenerative-like changes in mice and the antagonistic effects of vitamin E [27] | Liu et al. | 2019 | China | To investigate the effects of PM2.5 exposure on the brain and whether vitamin E can ameliorate the pathology. | C57BL/6J mice (n=10) | Vitamin E, 50mg/kg, 2 wks | Mice were exposed to PM2.5 for 1 week and treated as described with concurrent vitamin E or control, sacrificed and brain tissue was analysed. | Mice exposed to PM2.5 recorded cognitive deficits and increased markers of neurodegeneration, which was blocked by Vitamin E treatment. |
| Suppression of tau-induced phenotypes by vitamin E demonstrates the dissociation of oxidative stress and phosphorylation in mechanisms of tau toxicity [28] | Cowan et al. | 2021 | UK | To investigate if Tau protein induces pathological changes in vivo and if these changes are rescued by treatment with Vitamin C or E. | Tg Tau drosphilia melanogaster (n=30/group) | Vitamin E: 0.5, 1.5 or 4.5mM, Vitamin C 100uM or 10mM | Flies were given food with listed concentrations of vitamins and their motor activity was analysed. Once they reached adulthood, they were sacrificed for brain tissue, with a small subgroup undergoing further testing. | Treatment with either vitamin E or C leads to reduced oxidative damage, improved neuronal function and improved behaviour in a d melanogaster model of tauopathy. |
| The Mitochondria-Targeted Antioxidant MitoQ Prevents Loss of Spatial Memory Retention and Early Neuropathology in a Transgenic Mouse Model of Alzheimer’s Disease [29] | McManus et al. | 2011 | USA, UK | To examine the ability of MitoQ to prevent AD like pathology in mouse cortical neurons and a mouse model of AD. | 3xTg-AD mice 2 mo (F, n=117) | MitoQ, 100uM, PO, 5 mo | Mice were treated with MitoQ, compared with negative controls. They were then subjected to cognitive testing, followed by sacrifice and analysis of brain tissue. | MitoQ treatment reduced oxidative stress, synaptic loss and astrogliosis in the cortex and decreased Aβ burden in the hippocampus and neocortex. This restored cognitive performance to the level of WT mice. |
| The mitochondria-targeted antioxidant MitoQ extends lifespan and improves healthspan of a transgenic Caenorhabditis elegans model of Alzheimer disease [30] | Ng et al. | 2014 | Singapore | To examine the efficacy of MitoQ in reducing Aβ induced pathology and oxidative stress in a C. elegans model of AD. | Tg C. elegans CL2006 (n=200) |
MitoQ, 0.1,1,5uM, 2 d | Treatment was administered as described and compared to controls, with concurrent behaviour analysis, followed by post-treatment tissue analysis. | MitoQ treatment prolonged the lifespan and healthspan in a C. elegans AD model, mediated through its interaction with complex I and IV of the ETC. |
| Mito-TEMPO, a Mitochondria-Targeted Antioxidant, Improves Cognitive Dysfunction due to Hypoglycemia: an Association with Reduced Pericyte Loss and Blood-Brain Barrier Leakage [31] | Lin et al. | 2022 | China | To investigate the mechanism of hypoglycemia induced cognitive dysfunction and whether this is reversed by MitoTEMPO in vivo. | C57BL/6J mice (M,n=100) | MitoTEMPO, 0.7/mg/kg, 10 d | Mice were induced to hypoglycaemia then treated as described, including controls and either sacrificed for histological testing or subjected to cognitive testing. | MitoTEMPO reduced oxidative stress, pericyte loss and apoptosis, protected against BBB leakage and neuron damage and this led to improved cognition in a mouse model of cognitive dysfunction. |
| Mitochondria-Targeted Plastoquinone Antioxidant SkQ1 Prevents Amyloid--Induced Impairment of Long-Term Potentiation in Rat Hippocampal Slices [32] | Kapay et al. | 2013 | Russia | To investigate whether SkQ1 attenuates Aβ -induced impairment of LTP in rat hippocampi. | Wistar rats (M) | SkQ1 250nmol/kg, IP, Once | Rats were injected with a single dose of SkQ1 as described, compared with controls, and sacrificed 24 hours later for incubation with Aβ and subsequent hippocampal tissue analysis. | SkQ1 treatment in rats rescues the inhibitory effect of Aβ peptide on LTP induction in hippocampus. |
| Alzheimer’s Disease-Like Pathology in Senescence-Accelerated OXYS Rats can be Partially Retarded with Mitochondria-Targeted Antioxidant SkQ1 [33] | Stefanova et al. | 2014 | Russia | To study the influence of long term treatment with SkQ1on the OXYS rat model of AD. | OXYS rats 1.5mo (M,n=15) | SkQ1, 250nmol/kg PO, 21.5 mo | Rats were treated as described and compared to healthy controls, subjected to behavioural testing at multiple time points then sacrificed at 23 mo for brain tissue analysis. | Long term SkQ1 treatment in the OXYs model of AD restored some domains of cognitive function and reduced Aβ burden. |
| An antioxidant specifically targeting mitochondria delays progression of Alzheimer’s disease-like pathology [34] | Stefanova et al. | 2016 | Russia | To determine the role of mitochondrial damage in AD and whether SkQ1can alleviate this. | OXYS Rats 12mo (M, n=15) | SkQ1, 250nmol/kg PO, 6mo | Rats were treated as described, subjected to behavioural testing then sacrificed for brain histopathological analysis. | SkQ1 treatment in rats restores mitochondrial function, resulting in increased neurons and synapses, decrease in Aβ and tau burden and improved cognition. |
| Antioxidant SkQ1 Alleviates Signs of Alzheimer’s Disease-like Pathology in Old OXYS Rats by Reversing Mitochondrial Deterioration [35] |
Kolosova et al. | 2017 | Russia | To evaluate the efficacy of SkQ1 in an aged AD rat model in mitigating significant AD-induced pathology. | OXYS rats 19mo (M, n=15) | SkQ1, 250nmol/kg PO, 5 mo | Rats were treated as described and compared to healthy controls, subjected to behavioural testing or sacrificed for brain tissue analysis at 24mo old. | Treatment with SkQ1 in an aged OXYS rat model of AD resulted in some improved markers of mitochondrial function, Aβ burden and certain behavioural alterations. |
| SS31, a Small Molecule Antioxidant Peptide, Attenuates β-Amyloid Elevation, Mitochondrial/Synaptic Deterioration and Cognitive Deficit in SAMP8 Mice [36] | Jia et al. | 2016 | China | To assess mitochondrial and synaptic alterations in a mouse model of AD and whether SS31 can mitigate these and improved cognition. | SAMP8 mice 10mo (M,n=10) | SS31, 5mg/kg IP, 8 wks | Mice were treated as described compared to negative controls. They were subjected to behavioural testing after 8 weeks then sacrificed for brain tissue analysis. | SS31 treatment reduced hippocampal Aβ burden, protected mitochondrial dynamics and function, preserved synaptic function and improved cognitive performance in a mouse model of AD. |
| Mitochondria-targeted small molecule SS31: a potential candidate for the treatment of Alzheimer’s disease [37] | Reddy et al. | 2017 | USA | To improve understanding of the protective effects of SS31 in AD associated mitochondrial and synaptic dysfunction in a mouse model of AD. | APP Tg mice 12mo (n=5) | SS31 5mg/kg IP, 6 wks | Mice were treated as described and compared to negative controls. They were sacrificed for brain and plasma analysis at 6 weeks. | SS31 lowered markers of AD pathology such as Aβ production, mitochondrial dysfunction and impaired biogenesis and synaptic dysfunction in a mouse model of AD. |
| Elamipretide (SS-31) improves mitochondrial dysfunction, synaptic and memory impairment induced by lipopolysaccharide in mice [38] | Zhao et al. | 2019 | China | To assess neuroprotective effects of SS-31 against LPS-induced oxidative stress and cognitive dysfunction. | C57BL/6 mice 10-11wk old (M, n=96) | SS31, 5mg/lg, 4 d | Mice were induced to oxidative stress by LPS, treated as described, then subjected to various cognitive tests compared to controls. |
SS31 protected against mitochondrial dysfunction, oxidative stress, synaptic dysfunction and regulated signalling pathways in this mouse model of cognitive impairment, leading to reduced learning and memory deficits. |
| The mitochondria-targeted small molecule SS31 delays progression of behavioural deficits by attenuating b-amyloid plaque formation and mitochondrial/synaptic deterioration in APP/PS1 mice [39] |
Jia et al. | 2023 | China | To examine the effect of SS31 on a disease progression in an early mouse model of AD. | APP/PS1 Tg mice 8mo (M, n=12) | SS31, 3mg/kg IP, 3x/wk for 24 wks | The APP/PS1 mouse model was compared to control mice and treated as described. Behaviour was tested at 22 weeks, then mice were sacrificed at 24 weeks. | SS31 lowered markers of AD pathology such as apoptosis, Aβ burden, ROS, mitochondrial dysfunction and improved cognitive and behavioural deficits in a mouse model of AD. |
| Real-time imaging of mitochondrial redox reveals increased mitochondrial oxidative stress associated with amyloid β aggregates in vivo in a mouse model of Alzheimer’s disease [40] | Calvo-Rodriguez et al. | 2024 | USA | To visualise the effects of Aβ on mitochondrial oxidation in vivo and determine whether this can be attenuated using SS-31. | APP/PS1 Tg mice 8 MO (M,F n=3) | SS31, 5mg/kg, 2x/ wk for 8 wks | Mice were treated as described compared to controls. After treatment mice were imaged in real time using multiphoton microscopy and then sacrificed for brain tissue analysis. | Mitochondrial oxidative stress in a mouse model of AD imaged in real time was inhibited by treatment with SS31 and Aβ plaque- associated dystrophic neurites were decreased without any observed decrease in Aβ plaque burden. |
| MTA | Mechanisms of action |
|---|---|
| MitoQ | The anti-oxidant Ubiquinone is conjugated to TPP, a lipophilic cation which passes through the lipid bilayer and accumulates in mitochondria due to its electrostatic potential with the mitochondrial membrane. Ubiquinone is reduced primarily by complex II of the ETC into its active form ubiquinol which exerts its antioxidant effect by scavenging ROS and targeting lipid peroxidation [41]. |
| SkQ1 | The antioxidant plastoquinone, a component of the chloroplast ETC containing methyl groups in place of methoxy groups in ubiquinone, is conjugated to TPP. It accumulates in mitochondria in a similar fashion to MitoQ and once reduced to plastoquinol exerts its antioxidant effect, also scavenging ROS and targeting lipid peroxidation [42,43]. |
| MitoTEMPO | The antioxidant TEMPO is conjugated with TPP, again accumulating in mitochondria via the same mechanism as above [44]. TEMPO is a synthetic superoxide scavenger which acts as a SOD mimic, neutralising ROS and sustaining redox equilibrium [45]. |
| SS31 (Elamipretide) | The Szeto Schiller peptides are small hydrophilic aromatic-cationic tetrapeptides which penetrate cell membranes and associate with cardiolipin in the inner mitochondrial membrane, stabilising its structure [46]. Its anti-oxidant effects include inhibiting ROS generation and lipid peroxidation, and mitochondria- protective effects include preventing cytochrome C release and inhibiting MPT to prevent cell swelling and death [47]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





