Submitted:
12 December 2025
Posted:
16 December 2025
You are already at the latest version
Abstract
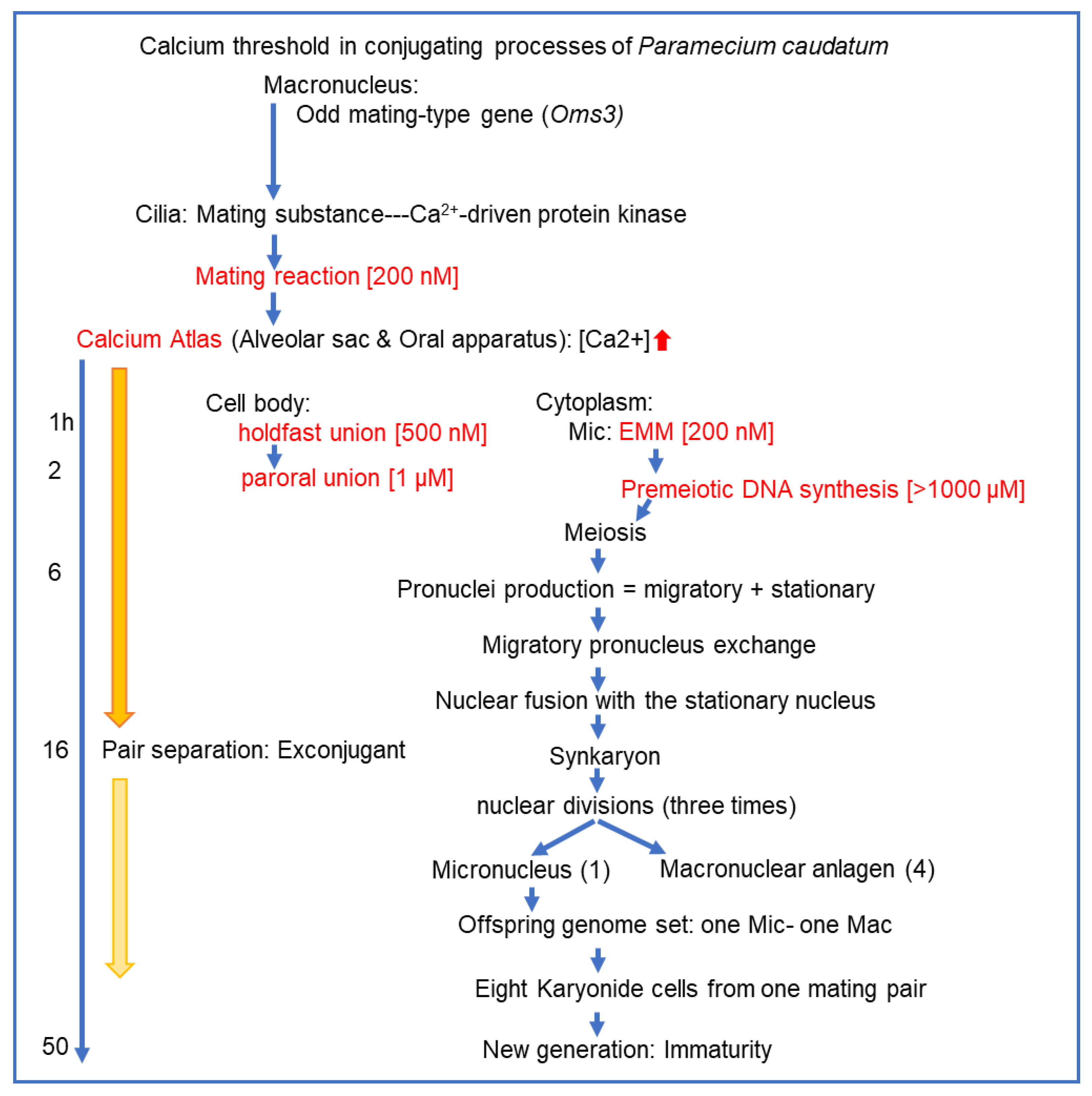
The unicellular ciliate Paramecium caudatum undergoes a developmental transition from asexual binary fission to sexual reproduction during its mature stage. This transition is triggered by mating interactions between cells of complementary mating types, leading to aggregate formation, mating pairs, and the meiotic division of micronuclei. Although calcium-driven EF-hand kinases have been implicated as mating type proteins, the spatiotemporal dynamics of calcium signaling during conjugation have not been comprehensively characterized. In this study, we established a behavioral assay to isolate committed cells from aggregates immediately after mating onset, and developed an experimental system to monitor intracellular calcium fluctuations specifically expressed in these cells. By combining Ca2+/EGTA buffering and microinjection approaches, we manipulated extracellular and intracellular calcium levels and confirmed the continuous requirement of calcium ions for conjugation-specific functions. Two significant findings emerged. First, we identified, for the first time, a calcium atlas covering the entire cell, with ascending centers localized in the anterior, oral apparatus, and posterior regions. The calcium/Indo-1-AM fluorescence peaked at six h after mating initiation and declined gradually, but persisted until conjugation was completed at ~48 h. Second, we demonstrated that distinct intracellular calcium thresholds are required for each stage of mating, including maintenance of mating activity, commitment of micronuclei to meiosis, and two-stepwise formation of mating pairs. These thresholds function as regulatory checkpoints that coordinate subcellular localization and stage synchronization. Collectively, our findings highlight calcium ions as pivotal regulators of conjugation in Paramecium and propose a novel framework, the Paramecium calcium atlas, for understanding the cellular and molecular mechanisms underlying sexual reproduction in ciliates.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Paramecium Stocks and Culturing Methods
2.2. Detection of Intracellular Calcium Ions Using Fluorescent Calcium Indicator, Indo-1 and Indo-1-AM
2.3. Quantifying Relative Fluorescence Intensity Through Computer Image Analysis
2.4. Calcium Ion Concentration Clamp Method
2.5. Microinjection of Ca2+/EGTA Buffers
2.6. Statistical Analysis
3. Results
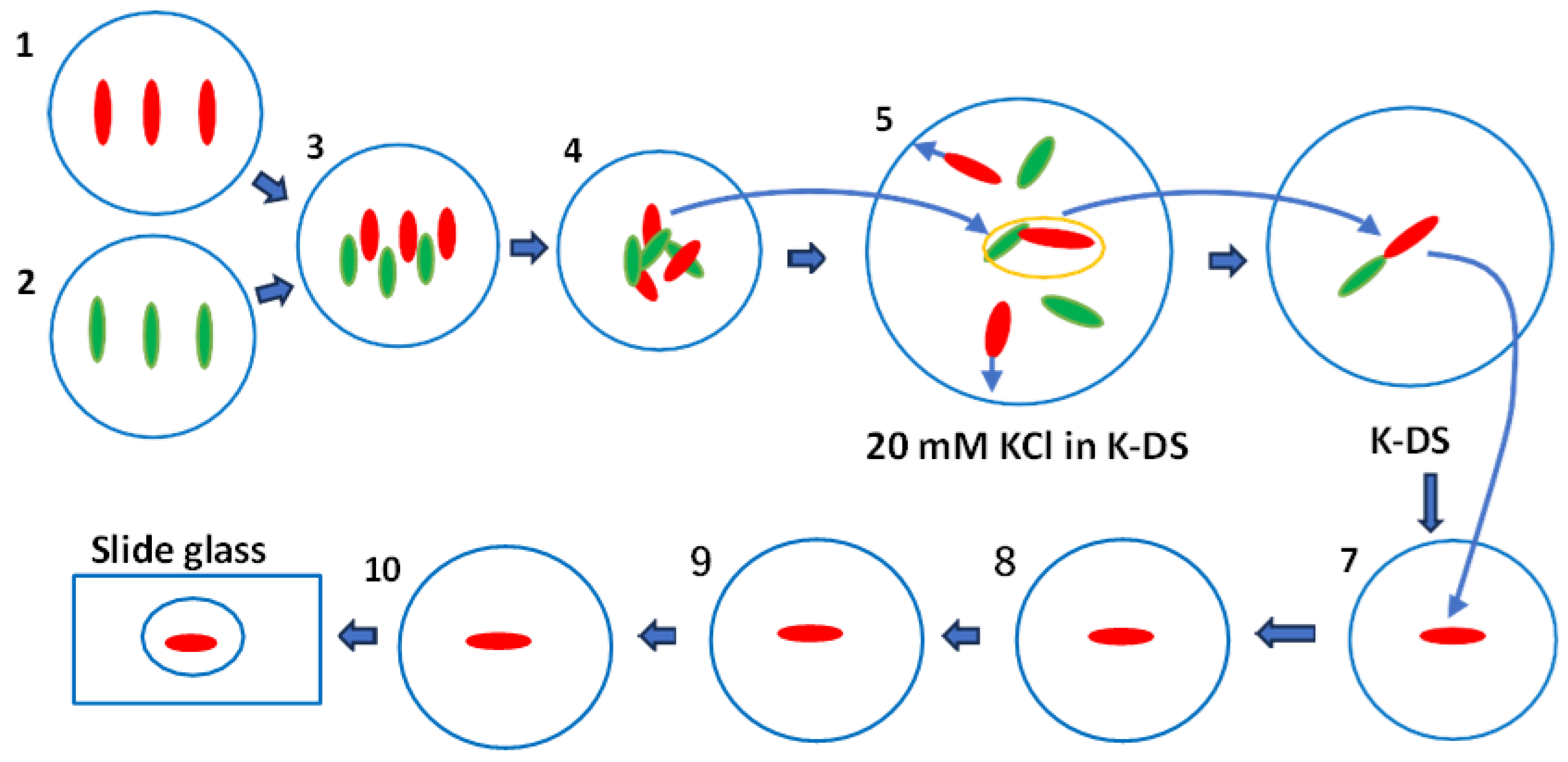
3.1. The Method for the Isolation of Mating-Competent Cells
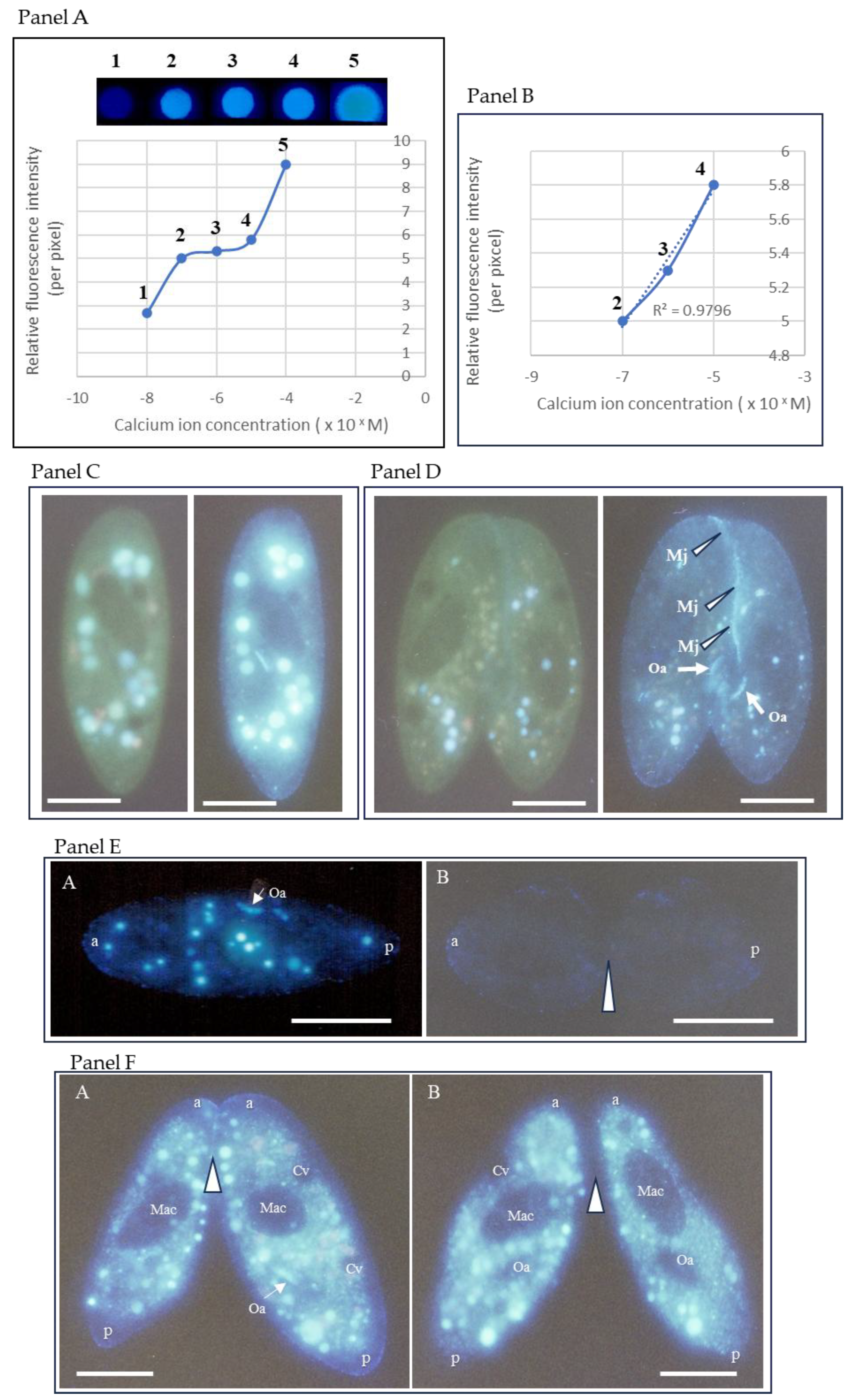
3.2. Calcium Indo-1-AM Measurement and Quantitative Assay
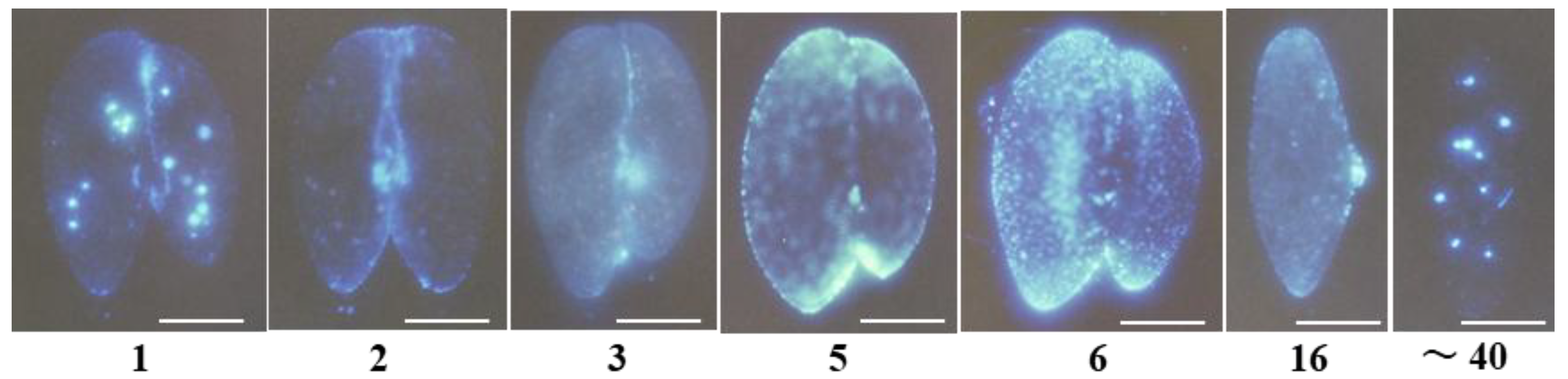
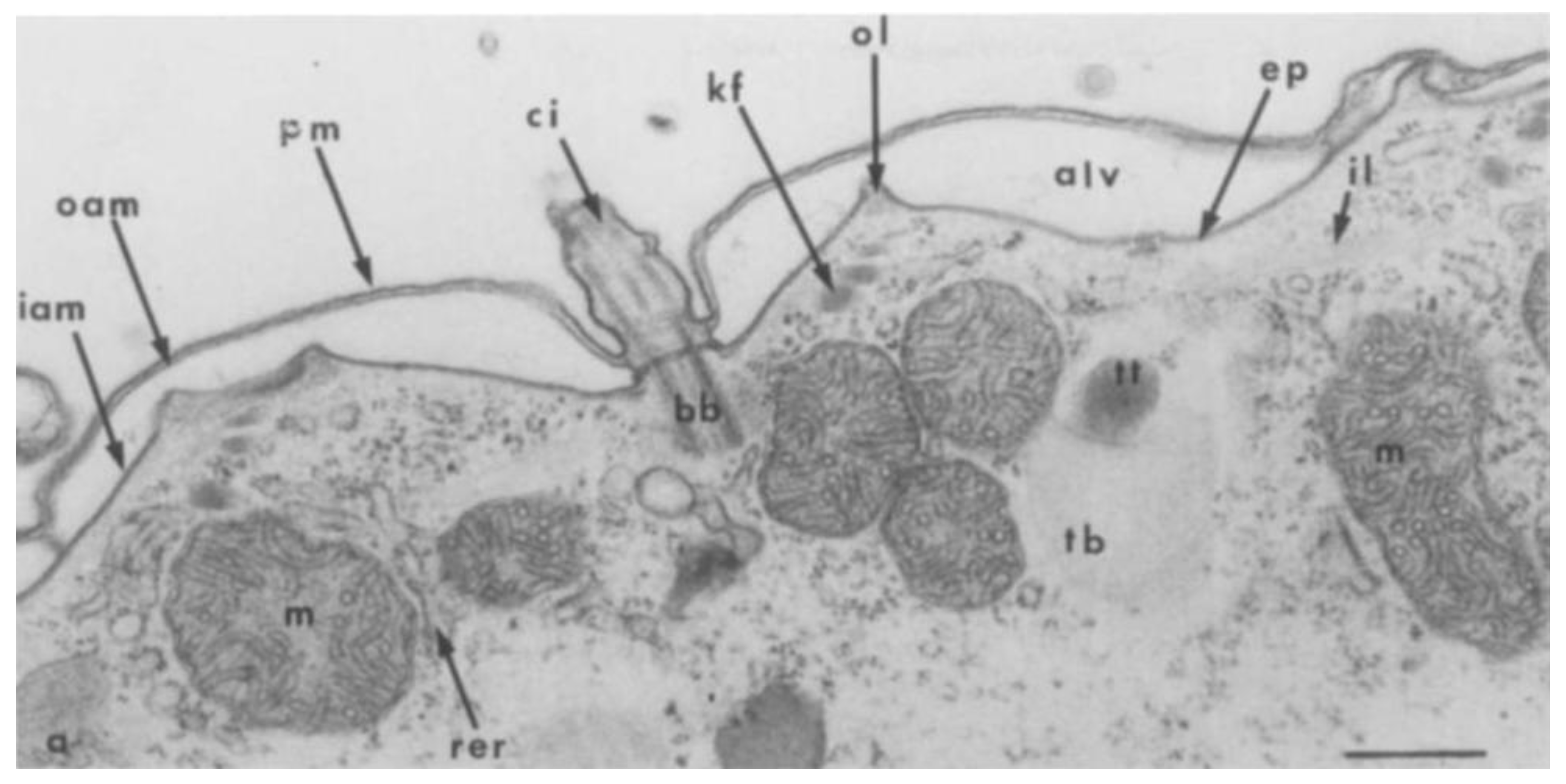
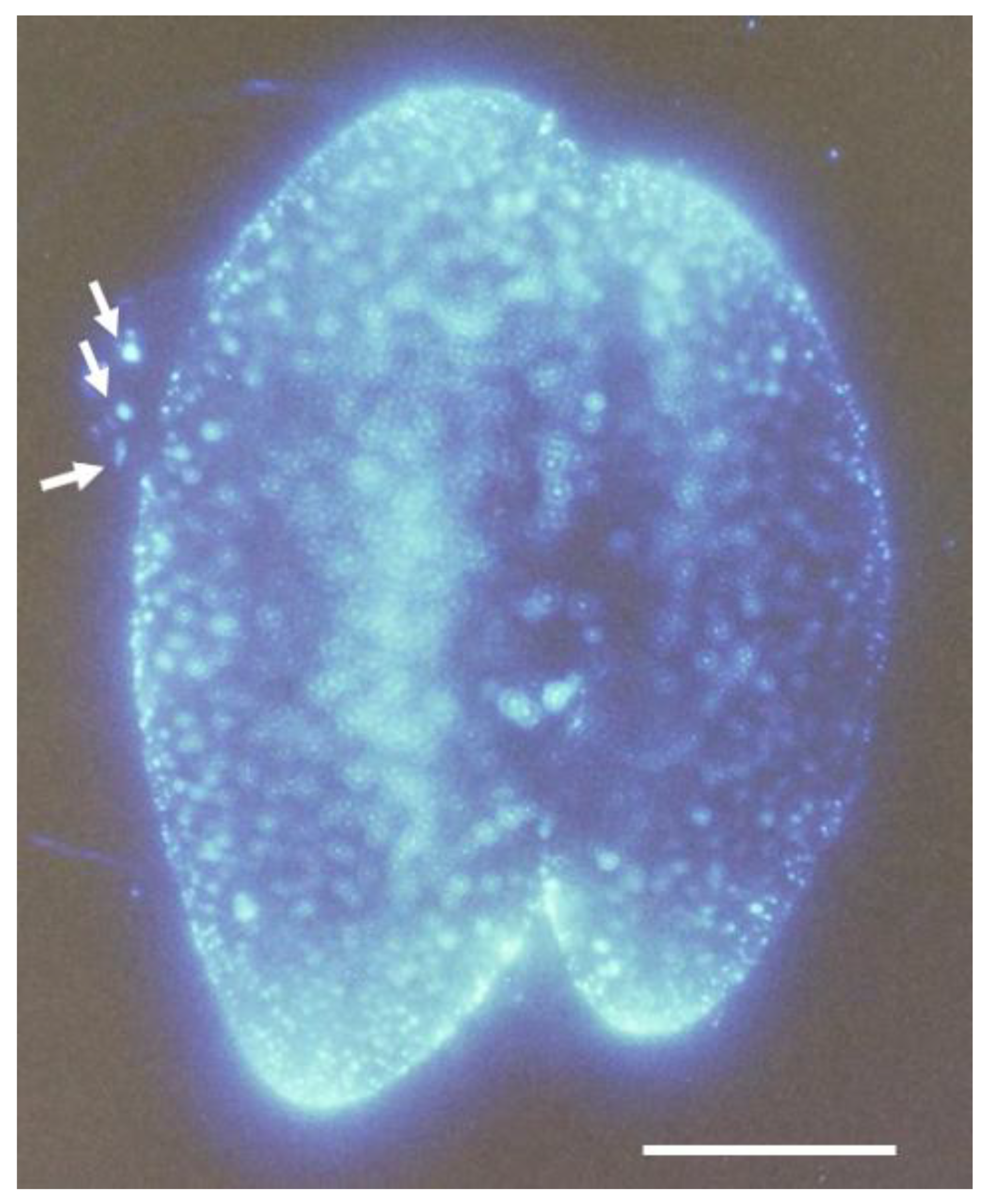
3.3. Characterization of the Paramecium Calcium Atlas
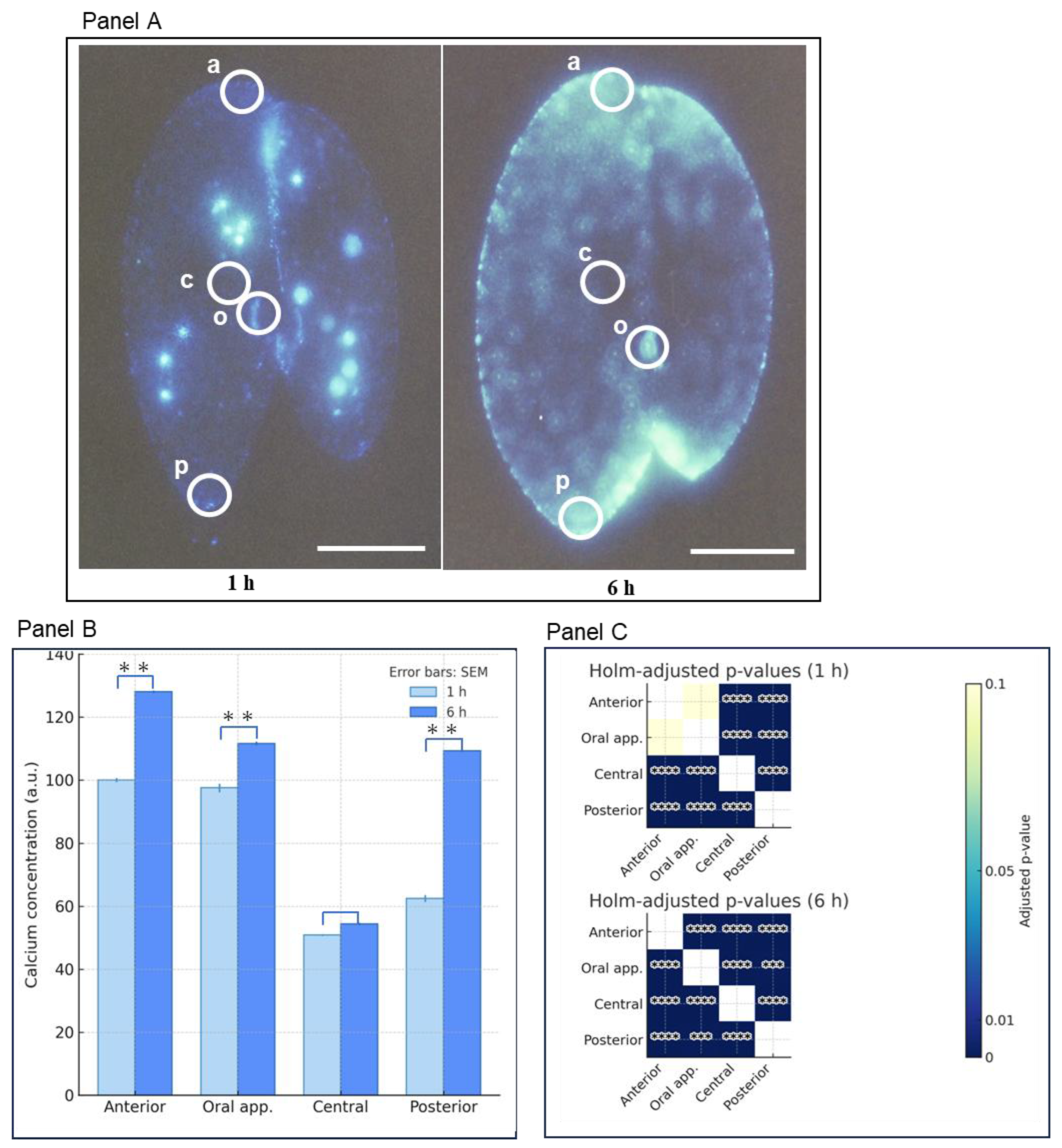
3.4. Comparison Between the Characterization of the Calcium Atlas at 1 and 6 Hours Conjugating Pairs
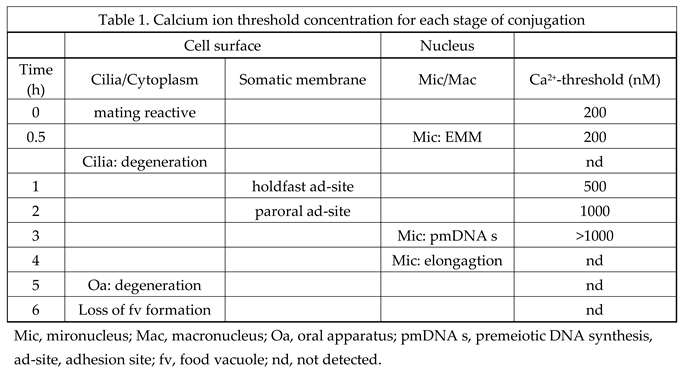
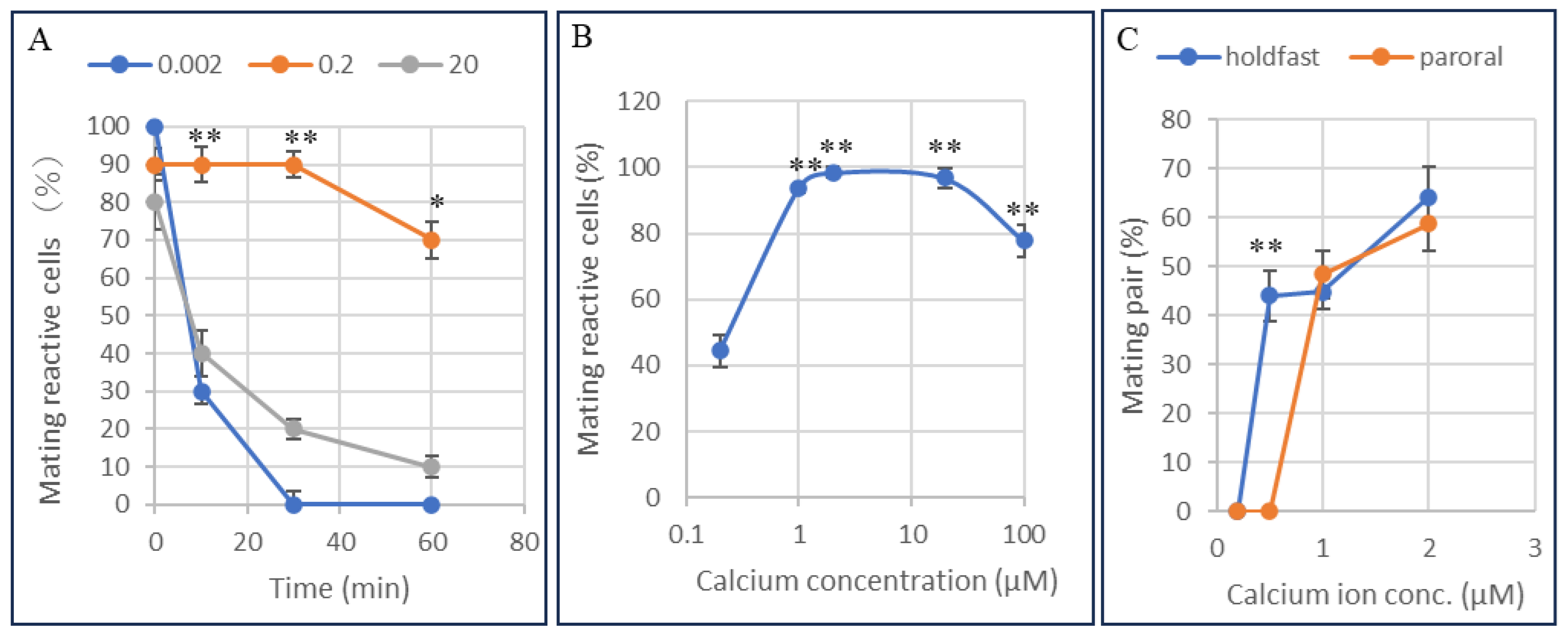
3.5. Determination of the Calcium Ion Threshold for the Conjugation Process Using the Calcium Ion Fixation Method with Ca2+/EGTA Buffers
3.6. Summary of the Paramecium Calcium Atlas
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- Yazaki, Y.; Shiratori, T.; Inagaki, Y. Protists with uncertain phylogenetic affiliations for resolving a deep tree of eukaryotes. Micro-organisms 2025, 13, 1–12. [Google Scholar] [CrossRef]
- Adl, S.M.; Simpson, A.G.B.; Lan2, C.E.; Lukes, J.; Bass, D.; Bowser, S.S.; Brown, M.W.; Burki, F.; Dunthorn, M.; Hampl, V.; et al. The revised Classification of Eukaryotes. J. Eukaryot. Microbiol. 2012, 59, 429–514. [Google Scholar] [CrossRef] [PubMed]
- Adl, S.M.; Bass, D.; Lukes, J.; Schoch, C.L.; Smirnov, A.; Agatha, S.; Berney, C.; Brown, M.W.; Burki, F.; et al. Revision to the Classification, Nomenclature, and Diversity of Eukaryotes. J. Eukaryot. Microbiol. 2019, 66, 4–119. [Google Scholar] [CrossRef]
- Hiwatashi, K. Determination and inheritance of mating type in Paramecium caudatum. Genetics 1968, 58, 373–386. [Google Scholar] [CrossRef]
- Singh, D.P.; Saudemont, B.; Guglielmi, G.; Arnaiz, O.; Gout, J.F.; Prajer, M.; Potekhin, A.; Przybos, E.; Aubusson-Fleury, A.; Bhullar, S.; et al. Genome-defence small RNAs exapted for epigenetic mating-type inheritance. Nature 2014, 509, 447–452. [Google Scholar] [CrossRef]
- Chiba, T.; Takenaka, Y.; Haga, N. Identification and characterization of the gene responsible for the O3 mating type substance in Paramecium caudatum. Microorganisms 2024, 12, 588. [Google Scholar] [CrossRef]
- Marchadier, E.; Oates, M.O.; Fang, H.; Donoghue, P.C.J.; Hetherington, A.M.; Gough, J. Evolution of the Calcium-Based Intracellular Signaling System. Genome Biol Evol. 2016, 8, 2118–2132. [Google Scholar] [CrossRef]
- Schaffer, D.E.; Iyer, L.M.; Burroughs, A.M.; Aravind, L. Functional Innovation in the Evolution of the Calcium-Dependent System of the Eukaryotic Endoplasmic Reticulum. Front Genet. 2020, 6, 11:34. [Google Scholar] [CrossRef] [PubMed]
- Haga, N. Micromanipulation in Paramecium: From non-mendelian inheritance to the outlook for versatile micromachines. J. Eukar-yot. Microbiol. 2022, 69, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Fujishima, M.; Hiwatashi, K. An early step in initiation of fertilization in Paramecium: early micronuclear migration. J. Exp. Zool 1977, 201, 127–134. [Google Scholar] [CrossRef]
- Yanagi, A. Positional control of the fates of nuclei produced after meiosis in Paramecium caudatum: Analysis by nuclear transplantation. Dev. Biol. 1987, 122, 535–539. [Google Scholar] [CrossRef]
- Takenaka, Y.; Yanagi, A.; Masuda, H.; Mitsui, Y.; Mizuno, H.; Haga, N. Direct observation of histone H2B-YFP fusion protein and transport of their mRNA between conjugating Paramecia. Gene 2007, 108–115. [Google Scholar] [CrossRef]
- Grynkiewicz, G.; Poenie, M.; Tsien, Y. R. A new generation of Ca2+ indicators with greatly improved fluorescence properties. J. Biol. Chem. 1985, 260, 3440–3450. [Google Scholar] [CrossRef] [PubMed]
- Koizumi, S. Microinjection and transfer of cytoplasm in Paramecium. Exp. Cell Res. 1974, 88, 74–78. [Google Scholar] [CrossRef]
- Haga, N.; Forte, M.; Saimi, Y.; Kung, C. Microinjection of cytoplasm as a test of complementation in Paramecium. J. Cell Biol. 1982, 82, 559–564. [Google Scholar] [CrossRef]
- Eisner, D.; Neher, E.; Taschenberger, H.; Smith, G. Physiology of intracellular calcium buffering. Physiol. Rev. 2023, 103, 2767–2845. [Google Scholar] [CrossRef]
- Plattner, H. Membrane Traffic and Ca²⁺ Signals in Ciliates. J. Eukaryot. Microbiol. 2022, 69, e12895. [Google Scholar] [CrossRef]
- Plattner, H. Trichocysts—Paramecium’s Projectile-like Secretory Organelles: Reappraisal of Their Biogenesis, Composition, Intra-cellular Transport, and Possible Functions. J. Eukaryot. Microbiol. 2017, 64, 106–133. [Google Scholar] [CrossRef]
- Schönemann, B.; et al. A Set of SNARE Proteins in the Contractile Vacuole Complex of Paramecium. Protist 2013, 164, 44–59. [Google Scholar]
- Plattner, H. Molecular Aspects of Calcium Signaling in Ciliates. J. Eukaryot. Microbiol. 2015, 62, 623–643. [Google Scholar]
- Ladenburger, E.-M.; Plattner, H. Calcium-Release Channels in Paramecium: Genomic Expansion, Differential Positioning and Partial Transcriptional Elimination. PLoS ONE 2011, 6, e27111. [Google Scholar] [CrossRef]
- Stelly, N.; Mauger, J-P.; Claret, M.; Adoutte, A. Cortical Alveoli of Paramecium: a Vast Submembranous Calcium Storage Compartment. J. Cell Biol. 1991, 113, 103–112. [Google Scholar] [CrossRef]
- Ramoino, P.; et al. GABA Release in the Ciliated Protozoon Paramecium Occurs by Neuronal-like Exocytosis. J. Exp. Biol. 2010, 213, 1251–1258. [Google Scholar] [CrossRef]
- Gould, S.B.; Tham, W.H.; Cowman, A.F.; McFadden, G.I.; Waller, R.F. Alveolins, a New Family of Cortical Proteins That Define the Alveolar Sacs in Apicomplexa, Dinoflagellates, and Ciliates. Mol. Biol. Evol. 2008, 25, 1219–1230. [Google Scholar] [CrossRef]
- Schilde, C.; Lutter, K.; Kissmehl, R.; Plattner, H. Molecular Identification of a SNAP-25-like SNARE Protein in Paramecium. Eukaryotic. Cell 2008, 7, 1387–1402. [Google Scholar] [CrossRef]
- Plattner, H.; Kissmehl, R. Dense-Core Secretory Vesicle Docking and Exocytotic Membrane Fusion in Paramecium Cells. Biochim. Biophys. Acta 2003, 1641, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Ladenburger, E.-M.; Plattner, H. Calcium Release Channels in Paramecium: In Situ Localization and Functional Characterization. J. Cell Sci. 2001, 114, 2653–2663. [Google Scholar]
- Plattner, H.; Flötenmeyer, M.; Kissmehl, R.; Hauser, K. Microdomain Arrangement of the SERCA-Type Ca²⁺-Pump (Ca²⁺-ATPase) in Subplasmalemmal Calcium Stores of Paramecium Cells. J. Eukaryot. Microbiol. 1999, 46, 311–324. [Google Scholar]
- Plattner, H. Microdomain Arrangement of the SERCA-Type Ca²⁺ Pump in Paramecium. J. Histochem. Cytochem. 1999, 47, 7–19. [Google Scholar]
- Hauser, K.; Pavlovic, N.; Kissmehl, R.; Plattner, H. Molecular Characterization of a Sarco (Endo)plasmic Reticulum-Type Ca²⁺-ATPase (SERCA) from Paramecium tetraurelia. Biochem. J. 1998, 334, 31–38. [Google Scholar]
- Länge, S.; Klauke, N.; Plattner, H. Subplasmalemmal Ca²⁺ Stores of Probable Relevance for Exocytosis in Paramecium tetraurelia: Alveolar Sacs Share Some but Not All Characteristics with Sarcoplasmic Reticulum. Cell Calcium 1995, 17, 335–344. [Google Scholar] [CrossRef] [PubMed]
- Plattner, H.; Verkhratsky, A. The Ancient Roots of Calcium Signalling Evolutionary Tree. Cell Calcium 2015, 57, 123–132. [Google Scholar] [CrossRef]
- Marchadier, E.; Oates, M.E.; Fang, H.; Donoghue, P.C.J.; Hetherington, A.M.; Gough, J. Evolution of the Calcium-Based Intracellular Signaling System. Genome Biology and Evolution 2016, 8(7), 2118–2132. [Google Scholar] [CrossRef] [PubMed]
- Schulz, P.; Herde, M.; Romeis, T. Calcium-dependent protein kinases: hubs in plant stress signaling and development. Plant Physiology 2013, 163, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Wernimont, A.K.; et al. Structures of apicomplexan calcium-dependent protein kinases reveal the mechanism of activation by calcium. Nature Structural & Molecular Biology 2010, 17, 596–601. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



