Submitted:
14 December 2025
Posted:
17 December 2025
You are already at the latest version
Abstract
It is becoming evident that forgetting is an active and adaptive mechanism that facilitates cognitive flexibility through the regulation of the stability of the stored experiences. The complex and wide spectrum of molecular catalyses and circuit-based activities determines the preservation of memories, their impairment, or availability for updating. Similar studies have been done on memory reconsolidation with the idea that retrieval with moderate prediction error creates a window of temporary destabilisation where the underlying engram can be modified. Nevertheless, none of the existing frameworks combine active forgetting mechanisms, accessibility of engrams, dynamics of reconsolidation and emerging neurotechnology in a single model of targeted memory deletion. This summary presents the multi-scale findings, such as synaptic destabilisation and engram remodelling, prediction-error gating, and non-invasive neuromodulation, to describe the conceptual bases of engineered memory modification. It is based on this platform that we suggest that Targeted Memory Deletion System (TMDS) can be implemented in four phases consisting of engram identification, controlled destabilisation, focused interference during reconsolidation and validation of deletion versus suppression. The structure puts into the limelight the biological possibility as well as the ethical limitation of selective memory editing. Combined, these observations put active forgetting, rather than a failure of retention, as a mechanistic gateway in which the clinically precise adjustment of memory might be enabled.
Keywords:
1. Introduction: The Biological Imperative of Erasure
2. Molecular Mechanisms of Active Forgetting
2.1. The Rac1-Scribble-Cofilin Signalosome: The Structural Eraser
2.2. The Ubiquitin-Proteasome System (UPS): The Synaptic Cleaner
2.3. GluA2 Endocytosis: The Gateway to Lability
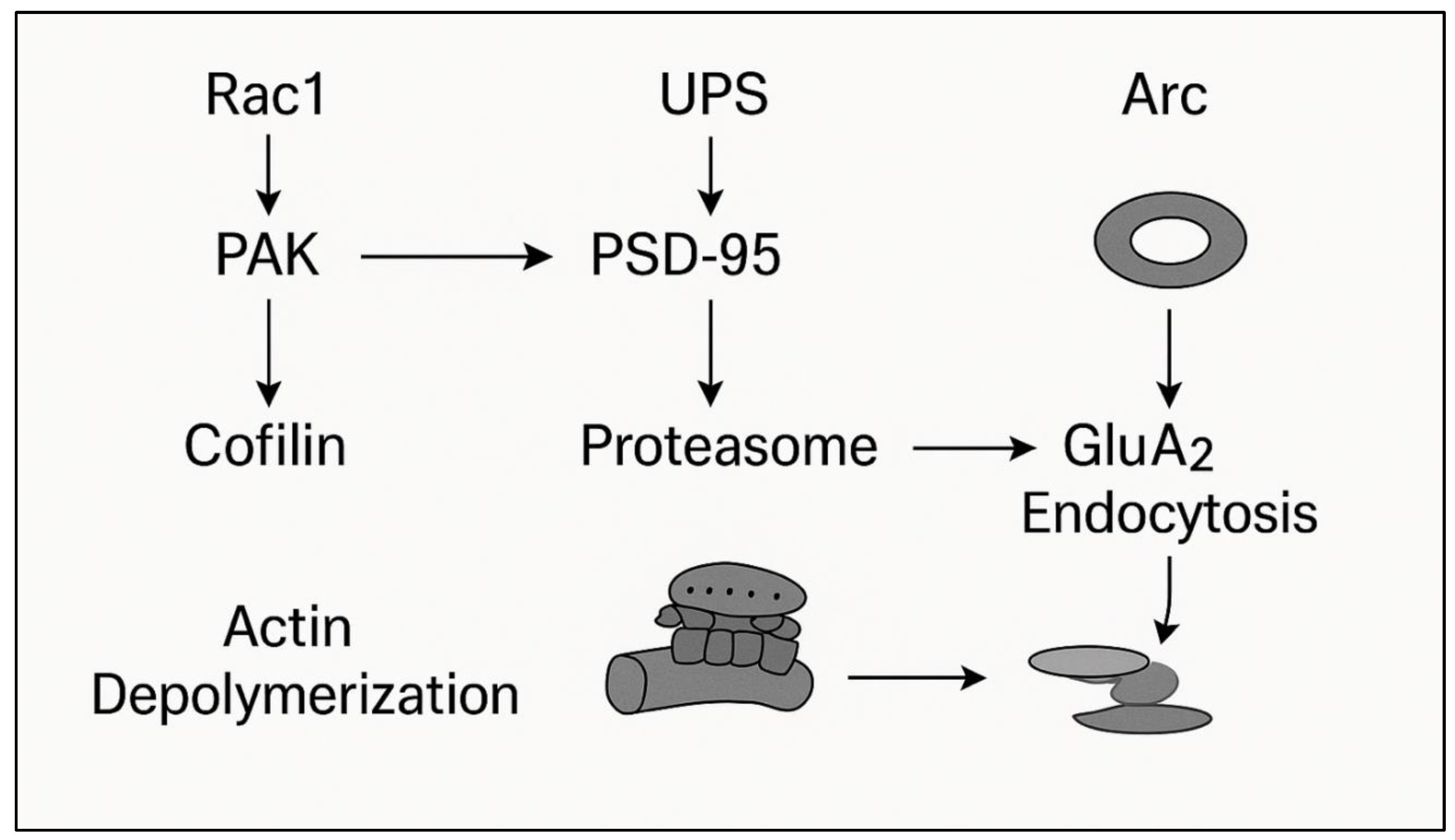
2.4. Arc: The Immediate Early Gene of Forgetting
2.5. Summary
- causing controlled destabilisation,
- maintaining local proteolysis, and
- Inhibition of synaptic restabilisation at reconsolidation.
3. Circuit Dynamics: The Engram and Its Regulation
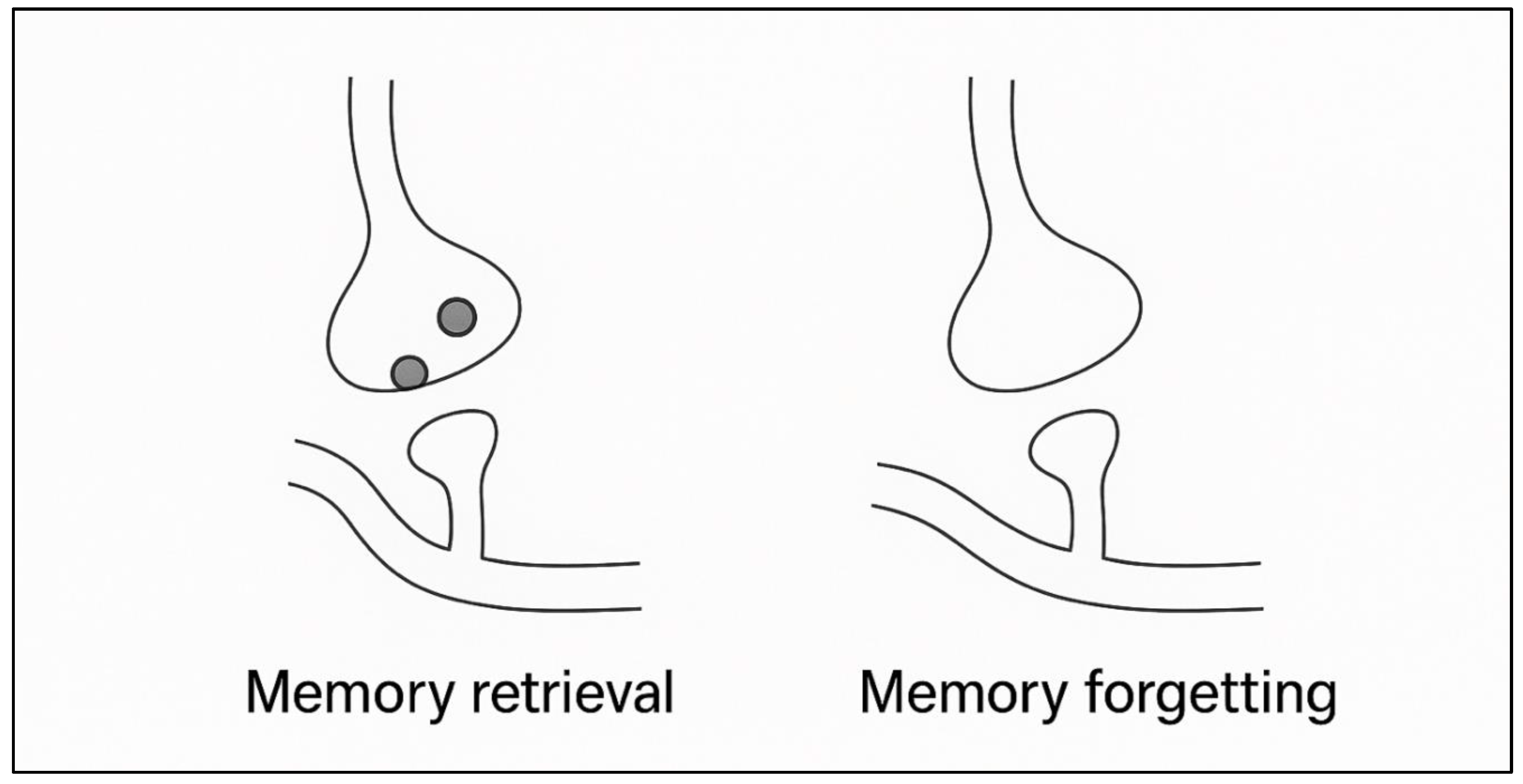
3.1. Silent Engrams vs. True Erasure
- Engram Connectivity- structural wiring of ensemble neurons
- Synaptic Potentiation - the weight pattern that is needed to do natural recall
3.2. Neurogenesis-Induced Forgetting
3.3. The Nucleus Reuniens: Orchestrating Specificity
3.4. Pattern Separation and Sparse Coding
3.5. Summary
- distinguish between erased and silent engrams,
- minimise disruption of the off-target circuit,
- distort retrieval pathways temporarily to destabilise it,
- should be able to achieve engram-level accuracy with sparse coding.
4. Reconsolidation: The Temporal Window of Vulnerability
4.1. Prediction Error (PE): The Boundary Condition
- No PE (Stability): A perfectly aligned retrieval generates no changes in the memory. The engram does not destabilise, and the continuation of the trace does not require the production of proteins.
- Moderate PE (Lability): The endocytosis of GluA2-AMPAR and ubiquitin-proteasome system-mediated proteolysis triggered by partial violations of expectation creates a reversible destabilisation window. This is the opportunity TMDS will not have.
- Excessive PE (Extinction): The existence of large discrepancies brings new learning of an inhibitory nature and not destabilisation of the original memory. The initial engram is retained, but through IL-ITC pathways, it is suppressed.
| PE Magnitude | Memory State | Outcome | Mechanism |
| Zero / Low | Stable | Persistence | No protein synthesis required. |
| Moderate | Labile | Update / Erasure | GluA2 endocytosis; UPS degradation. |
| High | Stable (Inhibited) | Extinction | New inhibitory circuit (IL-mPFC -> ITC). |
4.2. Molecular Blockade of Restabilisation
- GluA2 -AMPAR endocytosis, synaptic efficacy decreases;
- PSD scaffolds degenerate under the influence of UPS.
- Stabilisation proteins are removed depending on the proteasome.
- Activation of actin-remodelling pathways, including Rac1-Cofilin;
- De novo protein synthesis is required in restabilisation.
4.3. Pharmalogical Blockade of Restabilisation
4.4. Constraints and Failure Modes of Reconsolidation-Based Interventions
- Insufficient PE, preventing destabilisation.
- Overlong retrieval causes extinction rather than reconsolidation.
- Memory age or strength which can raise the PE threshold for lability.
- Boundary conditions that vary by memory type (episodic vs procedural vs emotional).
- Inconsistent mapping between retrieval cues and engram activation, especially in humans.
5. Technological Vectors for Delivery
5.1. Decoded Neurofeedback (DecNef): Implicit Modulation of Engram Probability Distributions
5.2. Targeted Memory Reactivation (TMR) In Sleep: Cue-Driven Modulation of Consolidation and Updating
5.3. Optogenetically Inspired Principles: Proof-of-Concept for Engram-Specific Modulation
- that activating an engram is sufficient to recall;
- that long-term depression (LTD) or silencing of engram neurons prevents recall
5.4. Non-Invasive Neuromodulation: Interfering with Coordinated Activity to Impair Restabilisation
5.5. Further Research on Focused Ultrasound (FUS): Deep and Focal Neuromodulation with Translational Potential
6. The Conceptual Architecture of the TMDS (4-Phase Framework)
| Phase | Objective | Biological State | Technological Vector | Success Metric |
| 1. Mapping | Identify Trace | Stable Engram | fMRI + MVPA | >90% Classifier Accuracy |
| 2. Destabilization | Open Window | Labile (GluA2 Endocytosis) | VR + Prediction Error | Moderate Arousal Spike |
| 3. Intervention | Block Restabilization | Protein Synthesis Inhibition | FUS + Rapamycin / DecNef | Synaptic Depotentiation |
| 4. Verification | Confirm Deletion | Silent / Erased | Spontaneous Recovery Test | No Reinstatement |
7. Ethical Analysis: The Rights to Memory and Mind
8. Conclusion
References
- Shuai Y, Lu B. Hippocampal Activation of Rac1 Regulates the Forgetting of Object Recognition Memory. Nat Commun. 2016;7:10838. Available from: https://pubmed.ncbi.nlm.nih.gov/27593377/.
- Shuai Y, Lu B. Roles of Rac1-Dependent Intrinsic Forgetting in Memory-Related Brain Disorders: Demon or Angel? Front Mol Neurosci. 2023;16:1172371. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC10341513/.
- Nader K, Schafe GE, LeDoux JE. Fear memories require protein synthesis in the amygdala for reconsolidation after retrieval. Nature. 2000;406(6797):722–726. [CrossRef]
- Sinclair AH, Barense MD. Prediction Error and Memory Reactivation: How Incomplete Reminders Drive Reconsolidation. Trends Neurosci. 2019;42(10):733–745. Available from: https://barense.psych.utoronto.ca/wp-content/uploads/2019/11/2019_Sinclair_Barense_TINs.pdf. [CrossRef]
- Merlo E, Milton AL, Goozée ZY, Theobald DE, Everitt BJ. Reconsolidation and extinction are dissociable and mutually inhibitory processes: Behavioral and molecular evidence. J Neurosci. 2014;34(7):2422–2431. [CrossRef]
- Penn AC, Zhang CL, Georges F, Royer L, Breillat C, Hosy E, et al. GluA2-dependent AMPA receptor endocytosis and the decay of long-term potentiation. Philos Trans R Soc Lond B Biol Sci. 2014;369(1633):20130141. Available from: https://royalsocietypublishing.org/doi/10.1098/rstb.2013.0141. [CrossRef]
- Shuai Y, Hu Y, Qin H, Campbell RA, Zhong Y. Scribble scaffolds a signalosome for active forgetting. Nature. 2015;520(7548):420–423. Available from: https://pubmed.ncbi.nlm.nih.gov/27263975/.
- Gao Y, Li M, Chen W, Li H, Li W. Inhibition of Rac1-dependent forgetting alleviates memory deficits in Alzheimer’s disease models. Mol Neurodegener. 2019;14(1):20. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC6776562/.
- Frankland PW, Köhler S, Josselyn SA. Hippocampal neurogenesis and forgetting. Trends Neurosci. 2013;36(9):497–503.
- Jarome TJ, Helmstetter FJ. The ubiquitin–proteasome system as a critical regulator of synaptic plasticity and long-term memory formation. Neurobiol Learn Mem. 2013;105:107–116. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC3786694/. [CrossRef]
- Reichelt AC, Lee JLC. Memory reconsolidation in humans: A sensitive period for updating memory. Psychon Bull Rev. 2013;20(1):168–176.
- Stemerding S, Stibbe A. Demarcating the Boundary Conditions of Memory Reconsolidation: An Unsuccessful Replication. Learn Mem. 2024. Available from: https://www.semanticscholar.org/paper/Demarcating-the-boundary-conditions-of-memory-An-Stemerding-Stibbe/ec945dcf2895a79df4ea5545131d9971a11dc1f0.
- Kroes MCW, Schiller D, LeDoux JE, Phelps EA. Targeting reconsolidation: A route to modify emotional memories. Ann N Y Acad Sci. 2016;1369(1):17–28. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC5424072/.
- Ryan TJ, Roy DS, Pignatelli M, Arons A, Tonegawa S. Memory. Engram cells retain memory under retrograde amnesia. Science. 2015;348(6238):1007–1013. [CrossRef]
- Nabavi S, Fox R, Proulx CD, Lin JY, Tsien RY, Malinow R. Engineering a memory with LTD and LTP. Nature. 2014;511(7509):348–352. [CrossRef]
- Josselyn SA, Frankland PW. Memory Allocation: Mechanisms and Function. Annu Rev Neurosci. 2018;41:389–413. [CrossRef]
- Shepherd JD, Bear MF. New views of Arc, a master regulator of synaptic plasticity. Nat Neurosci. 2011;14(3):279–284. [CrossRef]
- Pastuzyn ED et al. The neuronal gene Arc encodes a repurposed retrotransposon Gag protein that mediates intercellular RNA transfer. Cell. 2018;172(1–2):275–288.e18. [CrossRef]
- Kim J, Kwon JT, Kim HS. Engram competition and systems consolidation as mediators of forgetting. Nat Commun. 2020;11:3587.
- Guo N, Soden ME, Herber C, Kim MT, Besnard A, Lin P, et al. Robustness of retrieval and updating of fear memory via optogenetic rewiring of hippocampal engram circuits. Neuron. 2018;97(1):1–17.
- Dunsmoor JE, Niv Y, Daw ND, Phelps EA. Rethinking Extinction. Neuron. 2015;88(1):47–63. [CrossRef]
- Kroes MCW, Fernández G. Dynamic memory updating: prediction error and reconsolidation. Curr Opin Neurobiol. 2012;23(3):1–7.
- Gisquet-Verrier P, Riccio DC. Memory integration: An alternative reconsolidation hypothesis. Front Behav Neurosci. 2018;12:21.
- Schiller D, Monfils MH, Raio CM, Johnson DC, LeDoux JE, Phelps EA. Preventing the return of fear in humans using reconsolidation update mechanisms. Nature. 2010;463(7277):49–53. [CrossRef]
- Barrett AB, Barnett L, Seth AK. Multivoxel pattern analysis for engram identification: methodological considerations. Trends Cogn Sci. 2016;20(8):542–554.
- Rasch B, Born J. About sleep’s role in memory. Physiol Rev. 2013;93(2):681–766.
- Oudiette D, Paller KA. Upgrading memory replay during sleep. Nat Neurosci. 2013;16(2):145–147.
- Folloni D, Verhagen L, Mars RB, Fouragnan E, Constans C, Aubry JF, et al. Manipulation of subcortical and deep cortical activity in humans with transcranial focused ultrasound stimulation. Neuron. 2020;109(2):236–248.e5. [CrossRef]
- Redondo RL, Morris RG. Making memories last: the synaptic tagging and capture hypothesis. Nat Rev Neurosci. 2011;12(1):17–30. [CrossRef]
- Liu X et al. Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature. 2012;484:381–385.
- Besnard A, Sahay A. Adult hippocampal neurogenesis and mental health: A systematic review. Neuron. 2021;109(10):1507–1528.
- Davis RL, Zhong Y. The Biology of Forgetting—A Perspective. Neuron. 2017;95(3):490–503. Available from: https://pubmed.ncbi.nlm.nih.gov/28772119/.
- Hardt O, Nader K, Nadel L. Decay happens: The role of active forgetting in memory. Trends Cogn Sci. 2013;17(3):111–120. [CrossRef]
- Lee JLC, Nader K, Schiller D. An update on memory reconsolidation updating. Trends Cogn Sci. 2017;21(7):531–545. [CrossRef]
- Ji J, Maren S. Hippocampal involvement in contextual modulation of fear extinction. Hippocampus. 2007;17(9):749–758. [CrossRef]
- Likhtik E, Paz R. Amygdala–prefrontal interactions in (mal)adaptive learning. Trends Neurosci. 2015;38(3):158–166. [CrossRef]
- Deisseroth K. Optogenetics: 10 years of microbial opsins in neuroscience. Nat Neurosci. 2015;18(9):1213–1225. [CrossRef]
- Hayashi-Takagi A et al. Labelling and optical erasure of synaptic memory traces in the motor cortex. Nature. 2015;525:333–338. [CrossRef]
- O’Donnell C, Nolan MF. Tuning synaptic weights for biasing memory specificity. Nat Neurosci. 2020;23(1):9–10.
- Ito HT et al. A prefrontal–thalamo–hippocampal circuit for goal-directed spatial navigation. Nature. 2015;522:50–55.
- McAvoy K, Sahay A. Adult hippocampal neurogenesis and memory homeostasis. Neuropsychopharmacology. 2017;42(1):233–255.
- Richards BA, Frankland PW. The persistence and transience of memory. Nat Rev Neurosci. 2017;18(2):109–118.
- Anderson MC, Hulbert JC. Active forgetting: Adaptation of memory by prefrontal control. Annu Rev Psychol. 2021;72:1–36. [CrossRef]
- Anderson MC, Hanslmayr S. Neural mechanisms of motivated forgetting. Trends Cogn Sci. 2014;18(6):279–292. [CrossRef]
- Kroes MCW, Fernández G. How to erase memory: A practical review of reconsolidation interference. Neurosci Biobehav Rev. 2019;100:17–31.
- Shapiro ML, Eichenbaum H. Hippocampus as a memory map: Neural representation of space in rats. Nature. 1997;387:272–276.
- Oudiette D, Antony JW, Creery JD, Paller KA. The role of memory reactivation during wakefulness and sleep in strengthening memory. Curr Biol. 2013;23(19):R848–R850.
- Paller KA et al. Memory reactivation and consolidation during sleep. Learn Mem. 2021;28(11):395–403. [CrossRef]
- Ngo HV et al. Auditory closed-loop stimulation of sleep slow oscillations enhances memory. Neuron. 2013;78(3):545–553.
- Tuszynski MH, Weiner L, Fraenkel D, et al. FUS-mediated blood-brain barrier opening enhances neuroplasticity. J Neurosci. 2022;42(1):68–79.
- Sadtler PT et al. Neural constraints on learning. Nature. 2014;512(7515):423–426. [CrossRef]
- Shibata K, Watanabe T, Sasaki Y, Kawato M. Perceptual learning incepted by decoded fMRI neurofeedback. Science. 2011;334(6061):1413–1415. [CrossRef]
- Taschereau-Dumouchel V, Kawato M, Lau H. Multivoxel neurofeedback modulates confidence judgments while avoiding stimulus presentation. Nat Commun. 2022;13:2695.
- Fox MD, Buckner RL. The human brain is intrinsically organized into dynamic, anticorrelated functional networks. J Neurophysiol. 2013;110(6):1528–1546.
- Reinhart RMG, Nguyen JA. Working memory revived in older adults by synchronizing rhythmic brain circuits. Nat Neurosci. 2019;22(5):820–827. [CrossRef]
- Grossmann T. The neurobiology of human infant memory. Trends Cogn Sci. 2013;17(7):307–313.
- Eichenbaum H. On the integration of space, time, and memory. Neuron. 2017;95(5):1007–1018. [CrossRef]
- Squire LR, Alvarez P. Retrograde amnesia and memory consolidation: A neurobiological perspective. Curr Opin Neurobiol. 1995;5(2):169–177. [CrossRef]
- Milad MR, Quirk GJ. Fear extinction as a model for psychotherapy. Biol Psychiatry. 2012;73(1):61–70.
- Tovote P, Fadok JP, Lüthi A. Neuronal circuits for fear and anxiety. Nat Rev Neurosci. 2015;16(6):317–331. [CrossRef]
- Armony JL, LeDoux JE. How the brain processes emotional information. J Cogn Neurosci. 2000;12(2):181–194. [CrossRef]
- Delgado MR, Olsson A, Phelps EA. Extending animal models of fear conditioning to humans. Biol Psychol. 2006;73(1):39–48. [CrossRef]
- Sah P, Faber ESL, Lopez De Armentia M, Power J. The amygdaloid complex: Anatomy and physiology. Physiol Rev. 2003;83(3):803–834. [CrossRef]
- Bechmann M, Brand M. Autobiographical memory disturbance in psychiatric disorders. Nat Rev Neurol. 2020;16(10):567–582.
- Parfit D. Reasons and Persons. Oxford University Press; 1984.
- Schechtman M. The Constitution of Selves. Cornell University Press; 1996. [CrossRef]
- Metzinger T. Minimal phenomenal experience and self-model theory. Philos Trans R Soc B. 2020;375:20190367.
- Floridi L. The Logic of Being Informed. Synthese. 2011;190(20):3513–3520.
- Ienca M, Andorno R. Towards new human rights in the age of neuroscience and neurotechnology. Life Sci Soc Policy. 2017;13(1):5. [CrossRef]
- Bublitz JC, Merkel R. Crimes against minds: Mental interference and human rights. Crim Law Philos. 2014;8:51–77.
- Lavazza A. Erasing traumatic memories: ethics of reconsolidation therapies. Front Psychol. 2015;6:1170.
- Farah MJ. Neuroethics: The ethical, legal, and societal impact of neuroscience. Annu Rev Psychol. 2012;63:571–591. [CrossRef]
- Chatterjee A. The ethics of neuroenhancement. Semin Neurol. 2004;24:435–444.
- President’s Council on Bioethics. Beyond Therapy: Biotechnology and the Pursuit of Happiness. Washington, DC; 2003.
- McKinnon MC, Palombo DJ, Nazarov A, Kumar N, Kaniasty K, Freiburger O, et al. Autobiographical memory and PTSD. Depress Anxiety. 2015;32(9):645–654.
- Dunn BD, Dalgleish T, Lawrence AD. The somatic marker hypothesis and memory. Psychol Bull. 2010;136(1):1–29.
- Brewin CR. Theoretical foundations of intrusive images in PTSD. Psychol Rev. 2014;121(3):338–363.
- Lonergan MH, Olivera-Figueroa LA, Pitman RK, Brunet A. Propranolol and trauma: a meta-analysis of emotional memory reconsolidation interference. J Psychiatr Res. 2013;47(1):1–10.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




