Submitted:
12 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Issues pertaining to malnutrition are largely addressed by the consumption of fish meat, as it is both affordable and accessible to economically weaker sections of the population. Therefore, challenges observed in the aquaculture and fishery sectors, such as the detection of environmental changes, disease outbreaks, hindered growth, and poor fish health management, need to be addressed to increase production. Employment of modern technologies, such as (bio)sensors, is helpful for enhanced production in artisanal and large aquaculture systems. Because these can timely detect challenges, including climate change factors, sea-level rise-induced salinity load, changes in inland temperatures, ocean acidification, changes in precipitation patterns, ammonia toxicity, infectious diseases, and stress factors in water bodies or among their inhabitants, as a result, appropriate and timely measures can be taken at various stages of fish culture to address common problems. Using major scientific electronic databases, we comprehensively reviewed the topic of emerging needs, expanding applications, and recent technological advances in biosensors, with a particular focus on pisciculture. We highlight the biosensor technology used in the fisheries industry, which represents a pivotal step towards addressing its various aspects.
Keywords:
1. Introduction
2. Search Strategy
3. Current Status in Aquaculture
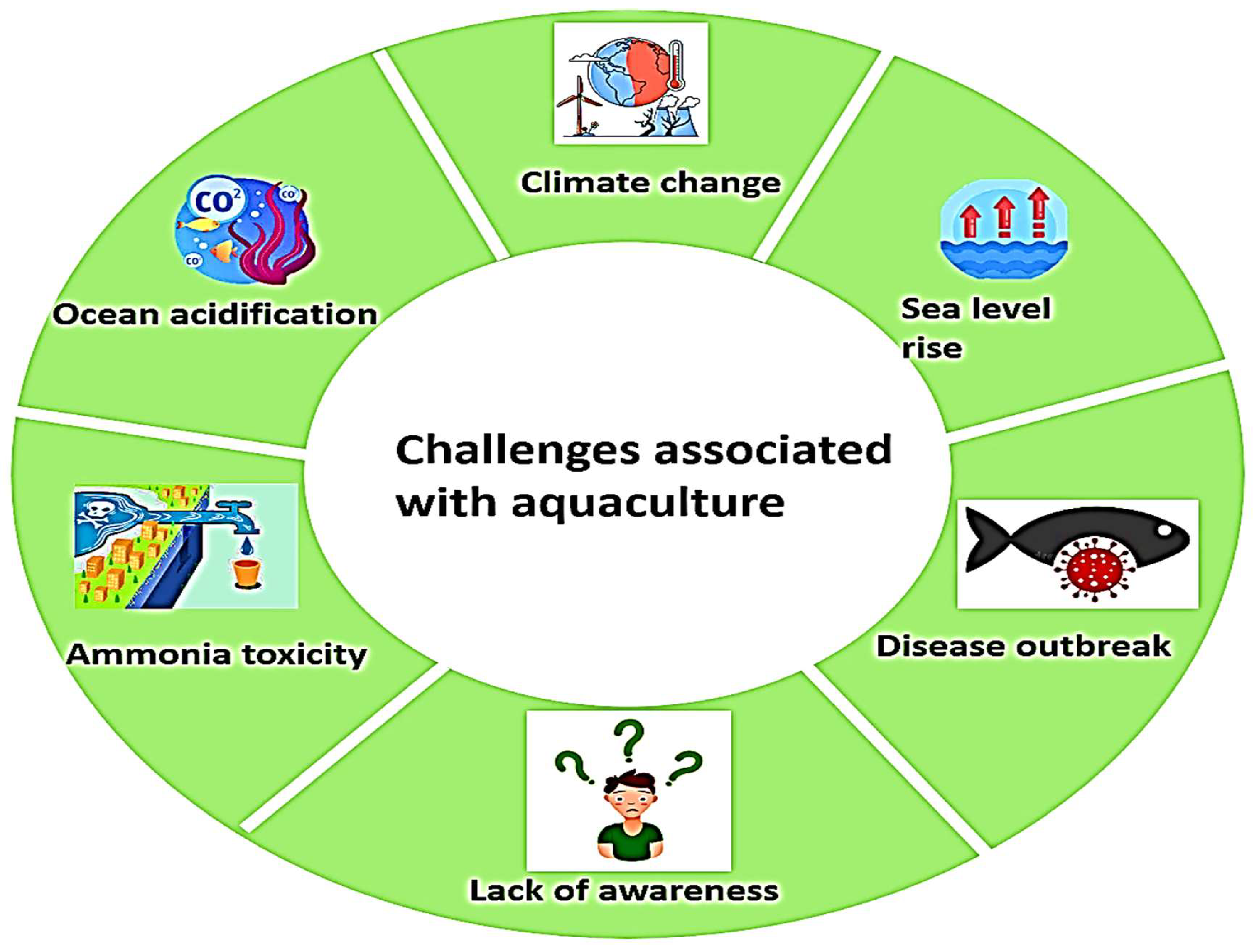
4. Challenges Associated with Aquaculture
5. Biosensors
5.1. Types of Biosensors Used in Pisciculture
5.2. Mode of Detection
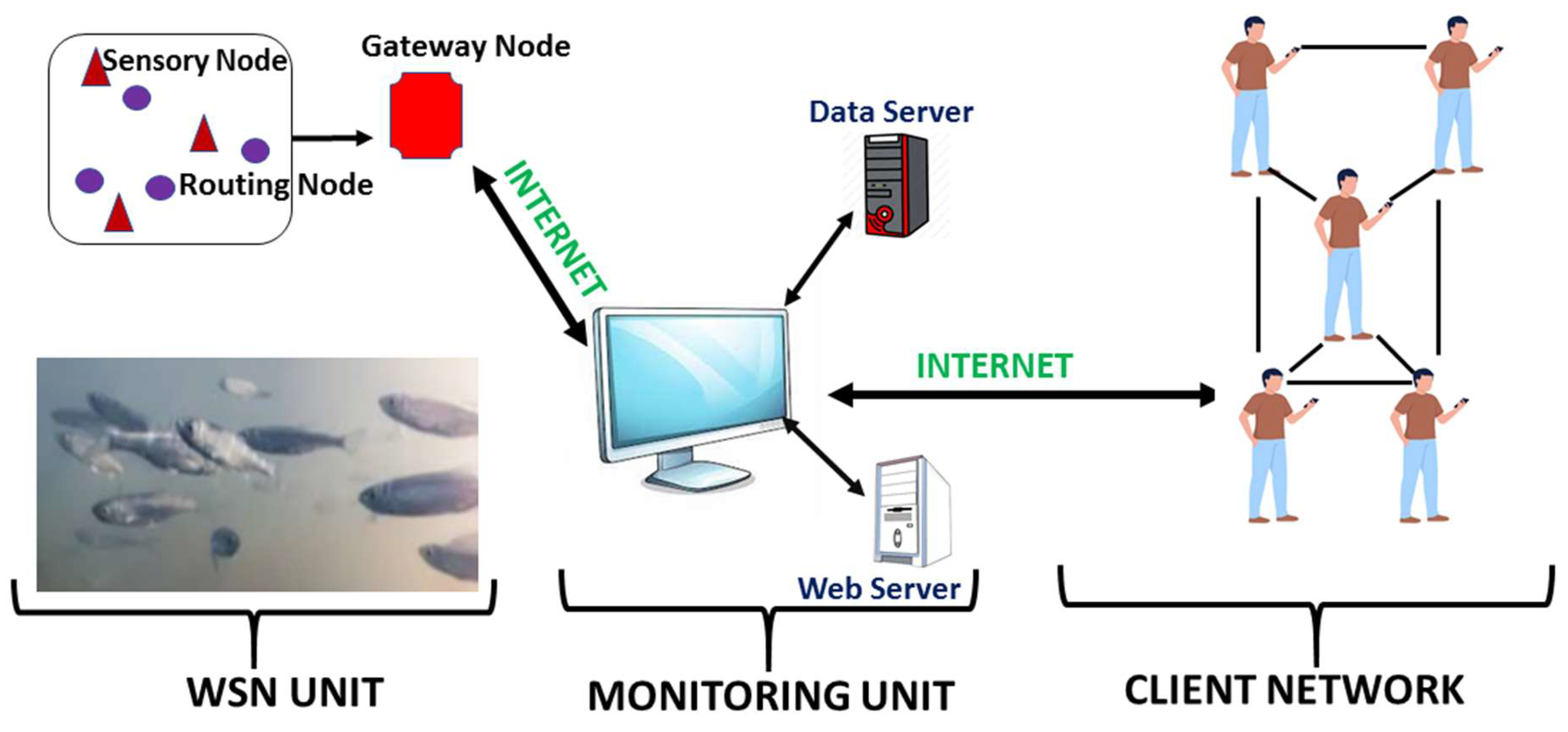
5.2.1. Wireless Sensor Network
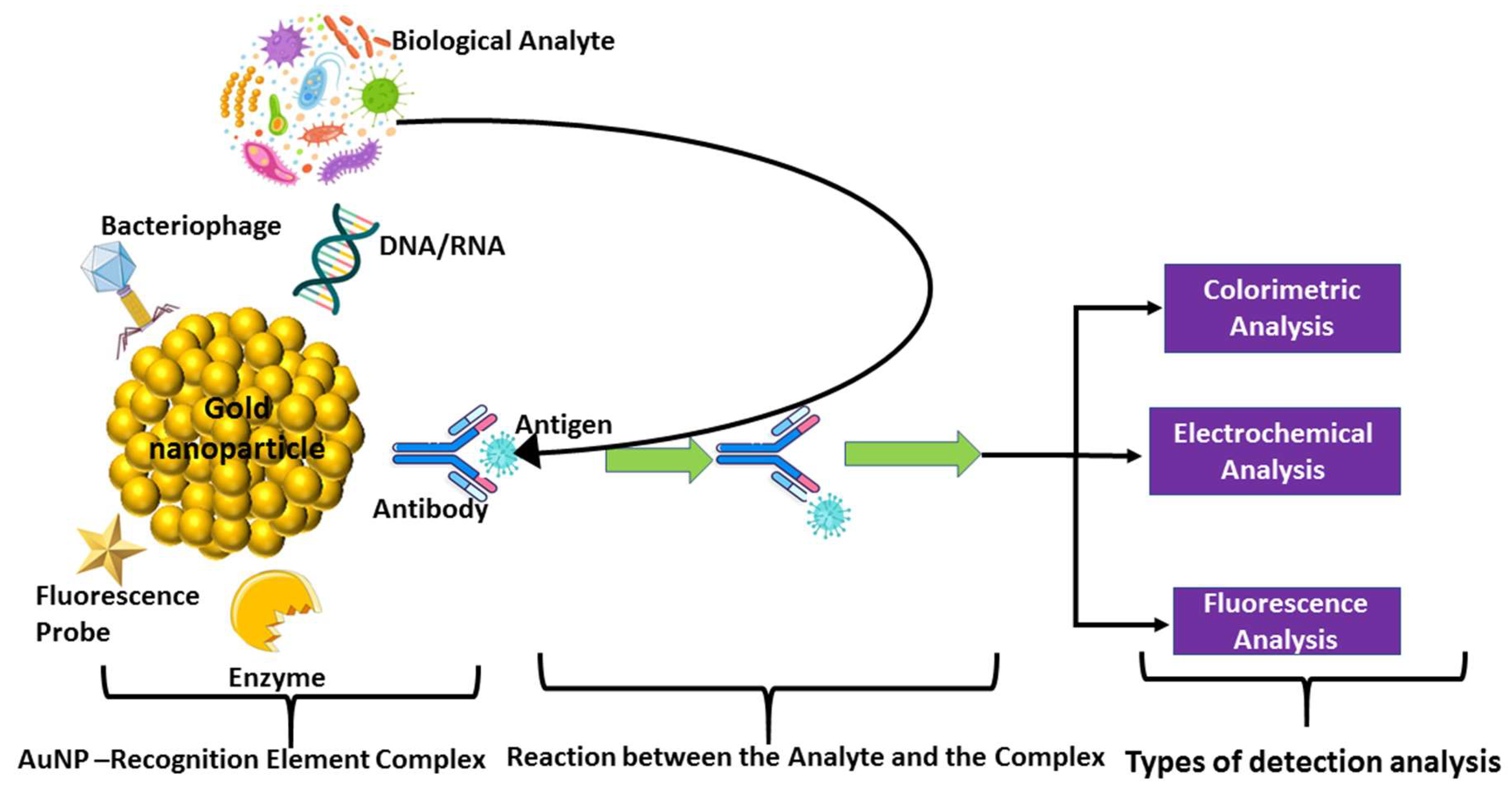
5.2.2. Nanoparticle Biosensor
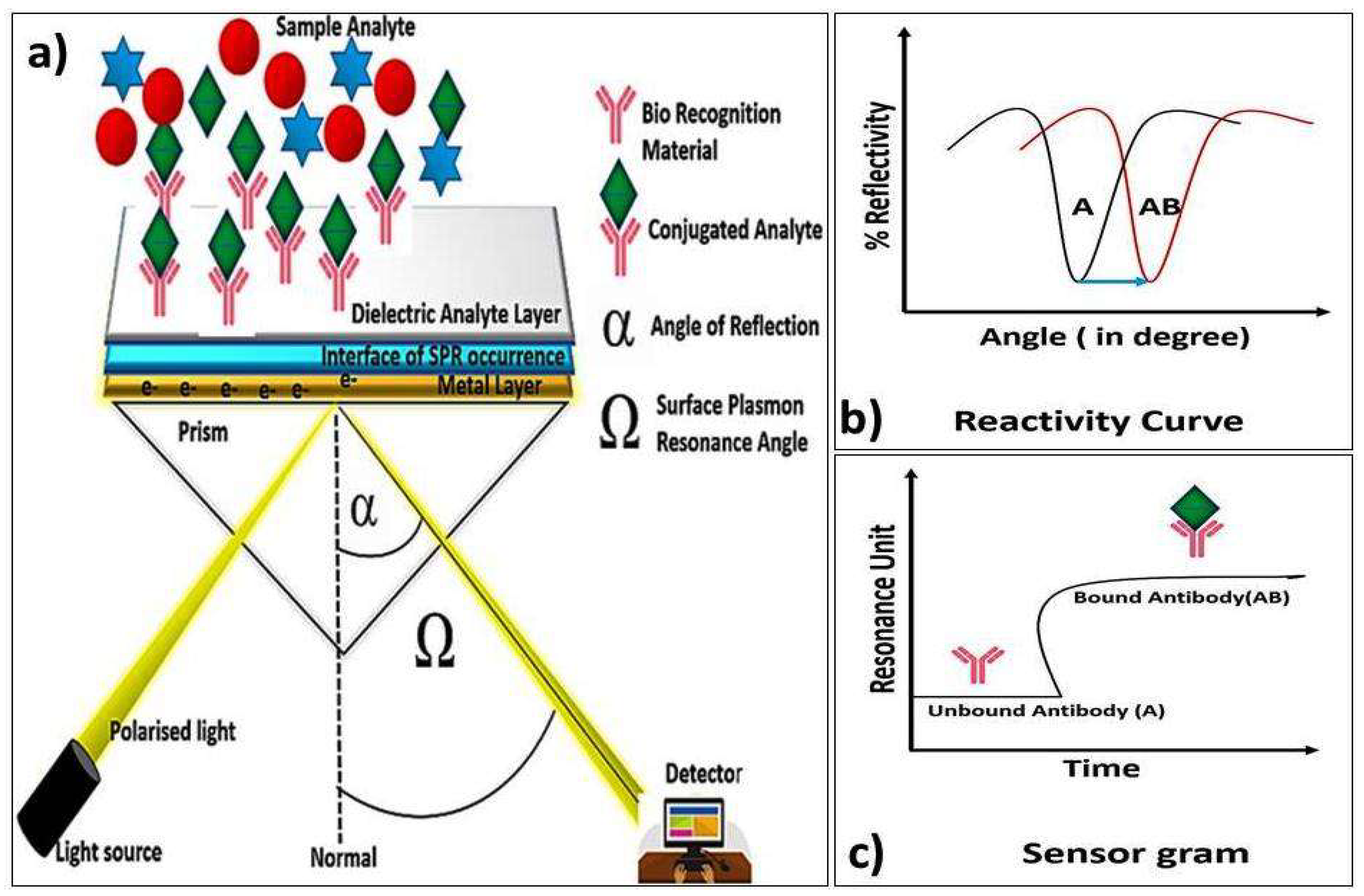
5.2.3. Surface Plasmon Resonance (SPR)
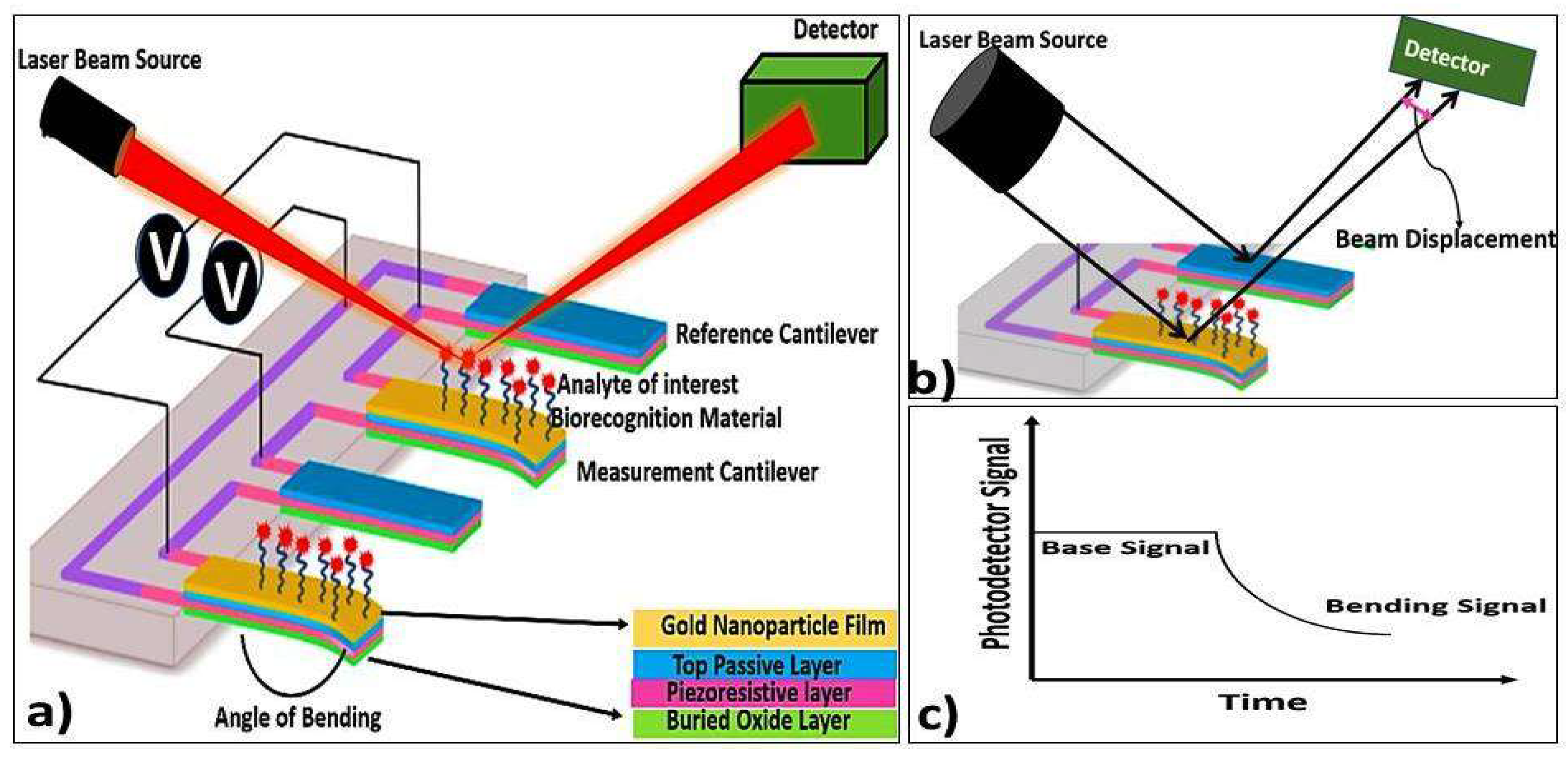
5.2.4. Microcantilever Biosensor
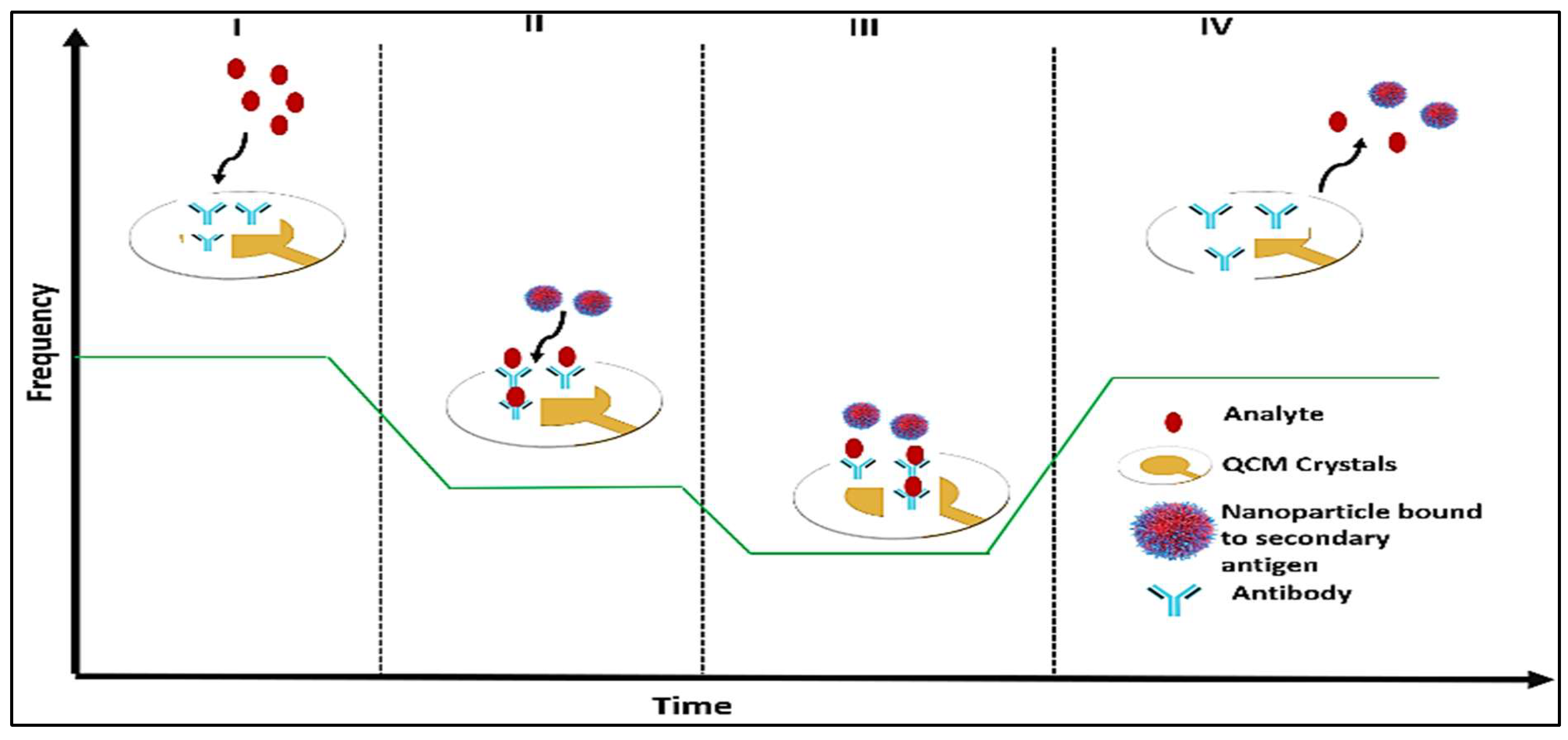
5.2.5. Quartz Crystal Microbalance Biosensor
5.2.6. Electrochemical Biosensor
6. Application of Biosensors in Aquaculture
6.1. Biosensors for Fish Health Management
6.2. Biosensors in Monitoring Stress Response
6.3. Biosensors in the Detection of Harmful Pathogens in Aquaculture
6.4. Biosensors in the Detection of Adulterations in Fish
6.5. Biosensors for the Detection of Fresh Aquaculture Products
6.6. Physiological Factors for Fish Health Management
6.7. Integration of Biosensors and AI
7. Conclusion and Prospective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AFM | Atomic Force Microscope |
| AuNP | gold nanoparticles |
| BOD | Biological Oxygen Demand |
| BSA | Bovine Serum Albumin |
| COVID-19 | Coronavirus Disease 2019 |
| CRISPR-Cas | Clustered Regularly Interspaced Short Palindromic Repeats |
| DFM | Dynamic Force Microscopy |
| DHP | Dihydroprogesterone |
| DMA | Dimethylamine |
| DO | Diamine Oxidase |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| FAO | Food and Agriculture Organization of the United Nations |
| FCM | Flow Cytometry |
| GBM | Gradient Boosting Machines |
| HIV | Human Immunodeficiency Virus |
| IEEE | Institute of Electrical and Electronics Engineers |
| LC50 | Lethal Concentration 50 |
| LCD | Liquid Crystal Display |
| MGO | Magnetic Graphene Oxide |
| MIPs | Molecularly-Imprinted Polymers |
| MOS-FET | Metal Oxide Semiconductor |
| NNV | Nervous Necrosis Virus |
| PCR | Polymerase Chain Reaction |
| PDMS | Polydimethylsiloxane |
| QCM | Quartz Crystal Microbalance |
| QCM-D | QCM with Dissipation Monitoring |
| RSV | Respiratory Syncytial Virus |
| SOFIA | State of World Fisheries and Aquaculture |
| SPR | Surface Plasmon Resonance |
| SVS | Support Vector Systems |
| SWCNT | Single-Walled Carbon Nanotubes |
| TMA | Trimethylamine |
| TVB | Total Volatile Bases |
| WHO | World Health Organization |
| WSN | Wireless Sensor Networks |
| WSSV | White Spot Syndrome Virus |
| XOD | Xanthine Oxidase |
References
- Seid, A.; Debebe, Z.; Ayelign, A.; Abeje, M.; Endris, B.S.; Assefa, M.; Jemal, A. Malnutrition Diagnosed by Patient-Generated Subjective Global Assessment and the Risk of All-Cause Mortality in Adults With Gastrointestinal Cancer: A Systematic Review and Meta-Analysis. Journal of Human Nutrition and Dietetics 2025, 38, e70012. [CrossRef]
- Yee Tan, P.; Ling Chan, C.; Vicheth Som, S.; Dye, L.; Bernadette Moore, J.; Caton, S.; Gong, Y.; Yee tan, P.; ling chan, chong; vicheth Som, S.; et al. Prevalence and Key Determinants of the Triple Burden of Childhood Malnutrition in Southeast Asian Countries: A Systematic Review and Meta-Analysis within an Adapted Socio-Ecological Framework. 2024. [CrossRef]
- FAO Report: Global Fisheries and Aquaculture Production Reaches a New Record High Available online: https://www.fao.org/newsroom/detail/fao-report-global-fisheries-and-aquaculture-production-reaches-a-new-record-high (accessed on 29 April 2025).
- Trinh, T.N.D.; Nguyen, H.A.; Thi, N.P.A.; Nam, N.N.; Tran, N.K.S.; Trinh, K.T.L. Biosensors for Seafood Safety Control—A Review. Micromachines 2024, Vol. 15, Page 1509 2024, 15, 1509. [CrossRef]
- Ying, R.; Li, N.; Zhang, Y.; Chen, H.; Zhang, S.; Feng, R.; Li, J.; Wang, S.; Mao, X. Innovations in Smart Enzyme Biosensors: Advancing the Detection of Antibiotic Residues in Aquaculture. Biotechnol Adv 2025, 83, 108607. [CrossRef]
- In Brief to The State of World Fisheries and Aquaculture 2024. 2024. [CrossRef]
- Beveridge, M.C.M.; Thilsted, S.H.; Phillips, M.J.; Metian, M.; Troell, M.; Hall, S.J. Meeting the Food and Nutrition Needs of the Poor: The Role of Fish and the Opportunities and Challenges Emerging from the Rise of Aquaculturea. J Fish Biol 2013, 83, 1067–1084. [CrossRef]
- Garlock, T.; Asche, F.; Anderson, J.; Bjørndal, T.; Kumar, G.; Lorenzen, K.; Ropicki, A.; Smith, M.D.; Tveterås, R.; Garlock, T.; et al. A Global Blue Revolution: Aquaculture Growth Across Regions, Species, and Countries. RvFSA 2020, 28, 107–116. [CrossRef]
- Naylor, R.L.; Hardy, R.W.; Buschmann, A.H.; Bush, S.R.; Cao, L.; Klinger, D.H.; Little, D.C.; Lubchenco, J.; Shumway, S.E.; Troell, M. A 20-Year Retrospective Review of Global Aquaculture. Nature 2021 591:7851 2021, 591, 551–563. [CrossRef]
- In Report of the FAO/MSU/WB First Multi-Stakeholder Consultation on a Progressive Management Pathway to Improve Aquaculture Biosecurity (PMP/AB); Washington, D.C; Washington, D.C., United States of America, 10-12 April 2018; 10. Washington, D.C. Report of the FAO/MSU/WB First Multi-Stakeholder Consultation on a Progressive Management Pathway to Improve Aquaculture Biosecurity (PMP/AB). 10–12 April 2018, Washington, D.C., United States of America.
- Yang, J.; Bhassu, S.; Rajamanikam, A. Charting the Future: Advanced Technologies for Sustainable Parasite Control in Aquaculture. International Journal of Molecular Sciences 2025, Vol. 26, Page 10738 2025, 26, 10738. [CrossRef]
- Renuka, S.; Poopal, R.K.; Ramesh, M.; Clara-Bindu, F. Responses of Labeo Rohita Fingerlings to N-Acetyl-p-Aminophenol Toxicity. Ecotoxicol Environ Saf 2018, 157, 73–80. [CrossRef]
- Sheikhlangi, Z.; Gharaei, A.; Mirdar Harijani, J.; Davari, S.A.; Hassanein, P.; Rahdari, A. Toxicological Effects of Meloxicam on Physiological and Antioxidant Status of Common Carp (Cyprinus Carpio). Vet Med Sci 2023, 9, 2085–2094. [CrossRef]
- Edwards, P.; Zhang, W.; Belton, B.; Little, D.C. Misunderstandings, Myths and Mantras in Aquaculture: Its Contribution to World Food Supplies Has Been Systematically over Reported. Mar Policy 2019, 106, 103547. [CrossRef]
- Naylor, R.L.; Kishore, A.; Sumaila, U.R.; Issifu, I.; Hunter, B.P.; Belton, B.; Bush, S.R.; Cao, L.; Gelcich, S.; Gephart, J.A.; et al. Blue Food Demand across Geographic and Temporal Scales. Nat Commun 2021, 12, 1–14. [CrossRef]
- Görs, M.; Schumann, R.; Hepperle, D.; Karsten, U. Quality Analysis of Commercial Chlorella Products Used as Dietary Supplement in Human Nutrition.. [CrossRef]
- Rodríguez-Morales, A. Challenges for Fish Foodborne Parasitic Zoonotic Diseases. Recent Pat Antiinfect Drug Discov 2015, 10, 3–5. [CrossRef]
- Adams, A. Progress, Challenges and Opportunities in Fish Vaccine Development. Fish Shellfish Immunol 2019, 90, 210–214. [CrossRef]
- Jenny, J.P.; Anneville, O.; Arnaud, F.; Baulaz, Y.; Bouffard, D.; Domaizon, I.; Bocaniov, S.A.; Chèvre, N.; Dittrich, M.; Dorioz, J.M.; et al. Scientists’ Warning to Humanity: Rapid Degradation of the World’s Large Lakes. J Great Lakes Res 2020, 46, 686–702. [CrossRef]
- Bhalla, N.; Jolly, P.; Formisano, N.; Estrela, P. Introduction to Biosensors. Essays Biochem 2016, 60, 1–8. [CrossRef]
- Mehrotra, P. Biosensors and Their Applications - A Review. J Oral Biol Craniofac Res 2016, 6, 153–159. [CrossRef]
- Wang, Y.; Sun, W.; Li, Y.; Zhuang, X.; Tian, C.; Luan, F.; Fu, X. Imidazole Metal-Organic Frameworks Embedded in Layered Ti3C2Tx Mxene as a High-Performance Electrochemiluminescence Biosensor for Sensitive Detection of HIV-1 Protein. Microchemical Journal 2021, 167, 106332. [CrossRef]
- Velusamy, K.; Periyasamy, S.; Kumar, P.S.; Rangasamy, G.; Nisha Pauline, J.M.; Ramaraju, P.; Mohanasundaram, S.; Nguyen Vo, D.V. Biosensor for Heavy Metals Detection in Wastewater: A Review. Food and Chemical Toxicology 2022, 168. [CrossRef]
- Jayanthi, V.S.P.K.S.A.; Das, A.B.; Saxena, U. Recent Advances in Biosensor Development for the Detection of Cancer Biomarkers. Biosens Bioelectron 2017, 91, 15–23. [CrossRef]
- Derin, E.; Inci, F. Advances in Biosensor Technologies for Acute Kidney Injury. ACS Sens 2022, 7, 358–385. [CrossRef]
- SiouNing, A.S.; Seong, T.S.; Kondo, H.; Bhassu, S. MicroRNA Regulation in Infectious Diseases and Its Potential as a Biosensor in Future Aquaculture Industry: A Review. Molecules 2023, 28. [CrossRef]
- Maldonado, G.E.; Luna-Moreno, J.; Rodríguez-Delgado, D.; Villarreal-Chiu, J.M.; Quintanilla-Villanueva, G.E.; Maldonado, J.; Luna-Moreno, D.; Manuel Rodríguez-Delgado, J.; Francisco Villarreal-Chiu, J.; Rodríguez-Delgado, M.M. Progress in Plasmonic Sensors as Monitoring Tools for Aquaculture Quality Control. Biosensors 2023, Vol. 13, Page 90 2023, 13, 90. [CrossRef]
- Bostock, J.; McAndrew, B.; Richards, R.; Jauncey, K.; Telfer, T.; Lorenzen, K.; Little, D.; Ross, L.; Handisyde, N.; Gatward, I.; et al. Aquaculture: Global Status and Trends. Philosophical Transactions of the Royal Society B: Biological Sciences 2010, 365, 2897–2912. [CrossRef]
- Pradeepkiran, J.A. Aquaculture Role in Global Food Security with Nutritional Value: A Review. Transl Anim Sci 2019, 3, 903–910. [CrossRef]
- You, K.; Ma, C.; Gao, H.; Li, F.; Zhang, M.; Qiu, Y.; Wang, B. Research on the Jellyfish (Rhopilema Esculentum Kishinouye) and Associated Aquaculture Techniques in China: Current Status. Aquaculture International 2007, 15, 479–488. [CrossRef]
- Shetty, S.; Pai, R.M.; Pai, M.M.M. Design and Implementation of Aquaculture Resource Planning Using Underwater Sensor Wireless Network. Cogent Eng 2018, 5, 1–23. [CrossRef]
- The State of World Fisheries and Aquaculture 2020. In Brief. The State of World Fisheries and Aquaculture 2020. In brief 2020. [CrossRef]
- Handisyde, N.T.; Ross, L.G.; Badjeck, M.-C.; Allison, E.H.; Department for International Development, L. The Effects of Climate Change on World Aquaculture: A Global Perspective.
- Cochrane, K.; De Young, C.; Soto, D. Climate Change Implications for Fisheries and Aquaculture Overview of Current Scientific Knowledge FOOD AND AGRICULTURE ORGANIZATION OF THE UNITED NATIONS. 2009.
- S, A.; Ajit Keshav, C.; Barlaya, G.; Rathod, R.; RN, M.; Ikmail, S.; GS, S.; HK, D.; I, S.; AS, M.; et al. Adaptation and Mitigation Strategies of Climate Change Impact in Freshwater Aquaculture in Some States of India. J Fishscicom 2018, 12. [CrossRef]
- Tewabe, D.; Tewabe, D. Climate Change Challenges on Fisheries and Aquaculture. Int J Aquac Fish Sci 2014, 1, 006–011. [CrossRef]
- Merino, G.; Barange, M.; Blanchard, J.L.; Harle, J.; Holmes, R.; Allen, I.; Allison, E.H.; Badjeck, M.C.; Dulvy, N.K.; Holt, J.; et al. Can Marine Fisheries and Aquaculture Meet Fish Demand from a Growing Human Population in a Changing Climate? Global Environmental Change 2012, 22, 795–806. [CrossRef]
- Bell, J.D.; Ganachaud, A.; Gehrke, P.C.; Griffiths, S.P.; Hobday, A.J.; Hoegh-Guldberg, O.; Johnson, J.E.; Borgne, R. Le; Lehodey, P.; Lough, J.M.; et al. Mixed Responses of Tropical Pacific Fisheries and Aquaculture to Climate Change. Nat Clim Chang 2013, 3, 591–599. [CrossRef]
- Cheung, W.W.L.; Watson, R.; Pauly, D. Signature of Ocean Warming in Global Fisheries Catch. Nature 2013, 497, 365–368. [CrossRef]
- Sae-Lim, P.; Kause, A.; Mulder, H.A.; Olesen, I. Breeding and Genetics Symposium: Climate Change and Selective Breeding in Aquaculture. J Anim Sci 2017, 95, 1801–1812. [CrossRef]
- Barange; Bahri; Beveridge; MCM; Cochrane; KL; Funge-Smith; Poulain; eds Impacts of Climate Change on Fisheries and Aquaculture: Synthesis of Current Knowledge, Adaptation and Mitigation Options.
- Masson-Delmotte, V.; Zhai, P.; Pörtner, H.-O.; Roberts, D.; Skea, J.; Shukla, P.R.; Pirani, A.; Moufouma-Okia, W.; Péan, C.; Pidcock, R.; et al. Global Warming of 1.5 °C An IPCC Special Report on the Impacts of Global Warming of 1.5 °C above Pre-Industrial Levels and Related Global Greenhouse Gas Emission Pathways, in the Context of Strengthening the Global Response to the Threat of Climate Change, Sustainable Development, and Efforts to Eradicate Poverty Edited by Science Officer Science Assistant Graphics Officer Working Group I Technical Support Unit. 2019.
- In Global Adaptation and Resilience to Climate Change; 2019; Global Adaptation and Resilience to Climate Change. 2019. [CrossRef]
- Maulu, S.; Hasimuna, O.J.; Haambiya, L.H.; Monde, C.; Musuka, C.G.; Makorwa, T.H.; Munganga, B.P.; Phiri, K.J.; Nsekanabo, J.D.M. Climate Change Effects on Aquaculture Production: Sustainability Implications, Mitigation, and Adaptations. Front Sustain Food Syst 2021, 5, 609097. [CrossRef]
- Richards, R.G.; Davidson, A.T.; Meynecke, J.O.; Beattie, K.; Hernaman, V.; Lynam, T.; van Putten, I.E. Effects and Mitigations of Ocean Acidification on Wild and Aquaculture Scallop and Prawn Fisheries in Queensland, Australia. Fish Res 2015, 161, 42–56. [CrossRef]
- Hoegh-Guldberg, O.; Mumby, P.J.; Hooten, A.J.; Steneck, R.S.; Greenfield, P.; Gomez, E.; Harvell, C.D.; Sale, P.F.; Edwards, A.J.; Caldeira, K.; et al. Coral Reefs under Rapid Climate Change and Ocean Acidification. Science 2007, 318, 1737–1742. [CrossRef]
- Weatherdon, L. V.; Magnan, A.K.; Rogers, A.D.; Sumaila, U.R.; Cheung, W.W.L. Observed and Projected Impacts of Climate Change on Marine Fisheries, Aquaculture, Coastal Tourism, and Human Health: An Update. Front Mar Sci 2016, 3, 179990. [CrossRef]
- Kibria, G.; Haroon, Y. Climate Change Impacts on Tropical and Temperate Fisheries, Aquaculture, and Seafood Security and Implications-A Review.
- Blanchard, J.L.; Watson, R.A.; Fulton, E.A.; Cottrell, R.S.; Nash, K.L.; Bryndum-Buchholz, A.; Büchner, M.; Carozza, D.A.; Cheung, W.W.L.; Elliott, J.; et al. Linked Sustainability Challenges and Trade-Offs among Fisheries, Aquaculture and Agriculture. Nat Ecol Evol 2017, 1, 1240–1249. [CrossRef]
- Alam, M.R.; Sharmin, S.; Islam, S.M.; Alam, M.A.; Ehiguese, F.O.; Pattadar, S.N.; Shahjahan, M. Salinity Intrusion Affects Early Development of Freshwater Aquaculture Species Pabda, Ompok Pabda. Aquac Rep 2020, 18, 100476. [CrossRef]
- Hua, H.H.; Cremin, E.; Huynh, D. Van; Long, G.; Renaud, F.G. Impacts of Aquaculture Practices on the Sustainability of Social-Ecological Systems in Coastal Zones of the Mekong Delta. Ocean Coast Manag 2024, 258, 107392. [CrossRef]
- Ammonia Nitrogen Dynamics in Aquaculture - Responsible Seafood Advocate Available online: https://www.globalseafood.org/advocate/ammonia-nitrogen-dynamics-in-aquaculture/ (accessed on 14 May 2025).
- Dopazo, C.P. The Infectious Pancreatic Necrosis Virus (IPNV) and Its Virulence Determinants: What Is Known and What Should Be Known. Pathogens 2020, 9, 94. [CrossRef]
- Cvetkovikj, A.; Radeski, M.; Blazhekovikj-Dimovska, D.; Kostov, V.; Stevanovski, V. Factors Affecting Fin Damage of Farmed Rainbow Trout. Maced Vet Rev 2015, 38, 61–71. [CrossRef]
- Budd, A.M.; Banh, Q.Q.; Domingos, J.A.; Jerry, D.R. Sex Control in Fish: Approaches, Challenges and Opportunities for Aquaculture. Journal of Marine Science and Engineering 2015, Vol. 3, Pages 329-355 2015, 3, 329–355. [CrossRef]
- Ridha, M. Evaluation of Monosex Culture of GIFT and Non-Improved Strains of Nile Tilapia Oreochromis Niloticus in Recirculating Tanks. 2011.
- Rehman, S.; Gora, A.H.; Ahmad, I.; Rasool, S.I. Stress in Aquaculture Hatcheries: Source, Impact and Mitigation. Int J Curr Microbiol Appl Sci 2017, 6, 3030–3045. [CrossRef]
- Canosa, L.F.; Bertucci, J.I. The Effect of Environmental Stressors on Growth in Fish and Its Endocrine Control. Front Endocrinol (Lausanne) 2023, 14, 1109461. [CrossRef]
- Sánchez-Velázquez, J.; Peña-Herrejón, G.A.; Aguirre-Becerra, H. Fish Responses to Alternative Feeding Ingredients under Abiotic Chronic Stress. Animals 2024, Vol. 14, Page 765 2024, 14, 765. [CrossRef]
- Akalu, B. The Main Factors Affecting Growth Performance of Oreochromis Niloticus L., (1758) in Aquaculture System. Journal of Fisheries & Livestock Production 2021, 9, 1. [CrossRef]
- Harmon, T.S. Methods for Reducing Stressors and Maintaining Water Quality Associated with Live Fish Transport in Tanks: A Review of the Basics. Rev Aquac 2009, 1, 58–66. [CrossRef]
- Islam, R.; Ahmed, R.; Dey, B.; Haque, M.S.; Aktar, S.; Bhuiyan, M.S.; Arif, M.S.; Habib Ador, M.A.; Ul Haque, M.M.; Saha, N. Salinity Hazard Drives the Alteration of Occupation, Land Use and Ecosystem Service in the Coastal Areas: Evidence from the South-Western Coastal Region of Bangladesh. Heliyon 2023, 9, e18512. [CrossRef]
- Ammonia | US EPA Available online: https://www.epa.gov/caddis/ammonia (accessed on 14 May 2025).
- Fan, B.; Li, J.; Wang, X.; Chen, J.; Gao, X.; Li, W.; Ai, S.; Cui, L.; Gao, S.; Liu, Z. Ammonia Spatiotemporal Distribution and Risk Assessment for Freshwater Species in Aquatic Ecosystem in China. Ecotoxicol Environ Saf 2021, 207, 111541. [CrossRef]
- Ahmed, I.; Ishtiyaq, S.; Sayed, S.F. An Overview on Understanding the Major Bacterial Fish Diseases in Freshwater Salmonids. Frontiers in Aquaculture 2025, 4, 1515831. [CrossRef]
- Dopazo, C.P. The Infectious Pancreatic Necrosis Virus (IPNV) and Its Virulence Determinants: What Is Known and What Should Be Known. Pathogens 2020, 9. [CrossRef]
- Roberts, R.J.; Pearson, M.D. Infectious Pancreatic Necrosis in Atlantic Salmon, Salmo Salar L. J Fish Dis 2005, 28, 383–390. [CrossRef]
- Rimsta, E. Infectious Pancreatic Necrosis. CABI Compendium 2019. [CrossRef]
- Portz, D.E.; Woodley, C.M.; Cech, J.J. Stress-Associated Impacts of Short-Term Holding on Fishes. Rev Fish Biol Fish 2006, 16, 125–170. [CrossRef]
- Vijayan, M.M.; Leatherland, J.F. Effect of Stocking Density on the Growth and Stress-Response in Brook Charr, Salvelinus Fontinalis. Aquaculture 1988, 75, 159–170. [CrossRef]
- Sarkar, B.; Mahanty, A.; Gupta, S.K.; Choudhury, A.R.; Daware, A.; Bhattacharjee, S. Nanotechnology: A next-Generation Tool for Sustainable Aquaculture. Aquaculture 2022, 546, 737330. [CrossRef]
- Mandal, S.; Chandra Banik, G. Chemical Science Review and Letters Biosensor: Potential and Scope in Agriculture. Chem Sci Rev Lett 2021. [CrossRef]
- Oliveira, S.; Sharifuzzaman, M.; Moro, G.; Sinibaldi, A.; Altintas, Z.; Kumar, S.; Chiavaioli, F.; Marques, C. Advances in Optical Devices for the Detection of Contaminants in Food and Water. TrAC Trends in Analytical Chemistry 2025, 184, 118139. [CrossRef]
- Singh, S.; Kumar, V.; Dhanjal, D.S.; Datta, S.; Prasad, R.; Singh, J. Biological Biosensors for Monitoring and Diagnosis. Microbial Biotechnology: Basic Research and Applications 2020, 317. [CrossRef]
- Huang, X.; Zhu, Y.; Kianfar, E. Nano Biosensors: Properties, Applications and Electrochemical Techniques. Journal of Materials Research and Technology 2021, 12, 1649–1672. [CrossRef]
- Das, S.; Singh, S.; Chawla, V.; Chawla, P.A.; Bhatia, R. Surface Plasmon Resonance as a Fascinating Approach in Target-Based Drug Discovery and Development. TrAC Trends in Analytical Chemistry 2024, 171, 117501. [CrossRef]
- Quadrini, L.; Laschi, S.; Ciccone, C.; Catelani, F.; Palchetti, I. Electrochemical Methods for the Determination of Urea: Current Trends and Future Perspective. TrAC Trends in Analytical Chemistry 2023, 168, 117345. [CrossRef]
- Jaffrezic-Renault, N.; Dzyadevych, S. V. Conductometric Microbiosensors for Environmental Monitoring. Sensors 2008, Vol. 8, Pages 2569-2588 2008, 8, 2569–2588. [CrossRef]
- Nehru, R.; Chen, C.W.; Dong, C. Di A Review of Smart Electrochemical Devices for Pesticide Detection in Agricultural Food and Runoff Contaminants. Science of The Total Environment 2024, 935, 173360. [CrossRef]
- Chai, C.; Oh, S.W. Electrochemical Impedimetric Biosensors for Food Safety. Food Sci Biotechnol 2020, 29, 879–887. [CrossRef]
- Marrazza, G. Piezoelectric Biosensors for Organophosphate and Carbamate Pesticides: A Review. Biosensors 2014, Vol. 4, Pages 301-317 2014, 4, 301–317. [CrossRef]
- Shah, M.M.; Ren, W.; Irudayaraj, J.; Sajini, A.A.; Ali, M.I.; Ahmad, B. Colorimetric Detection of Organophosphate Pesticides Based on Acetylcholinesterase and Cysteamine Capped Gold Nanoparticles as Nanozyme. Sensors 2021, 21, 8050. [CrossRef]
- Shi, B.; Sreeram, V.; Zhao, D.; Duan, S.; Jiang, J. A Wireless Sensor Network-Based Monitoring System for Freshwater Fishpond Aquaculture. Biosyst Eng 2018, 172, 57–66. [CrossRef]
- Jianqing, H.; Weixing, W.; Sheng, J.; Daozong, S.; Guocheng, O.; Kanju, L.; Jianqing, H.; Weixing, W.; Sheng, J.; Daozong, S.; et al. Development and Test of Aquacultural Water Quality Monitoring System Based on Wireless Sensor Network. Transactions of the Chinese Society of Agricultural Engineering (Transactions of the CSAE), 2013, Vol. 29, Issue 4, Pages: 183-190 2013, 29, 183–190. [CrossRef]
- Koubâa, A.; Alves, M.; Tovar, E.; Koubaa, A. IEEE 802.15.4 for Wireless Sensor Networks: A Technical Overview IEEE 802.15.4 for Wireless Sensor Networks: A Technical Overview IEEE 802.15.4 for Wireless Sensor Networks: A Technical Overview. 2005.
- Abdel-Ghany, H.M.; El-Sayed, A.F.M.; Ezzat, A.A.; Essa, M.A.; Helal, A.M. Dietary Lipid Sources Affect Cold Tolerance of Nile Tilapia (Oreochromis Niloticus). J Therm Biol 2019, 79, 50–55. [CrossRef]
- Jiang, R.; Zhang, J.; Liao, Z.; Zhu, W.; Su, H.; Zhang, Y.; Su, J. Temperature-Regulated Type II Grass Carp Reovirus Establishes Latent Infection in Ctenopharyngodon Idella Brain. Virol Sin 2023, 38, 440–447. [CrossRef]
- Berry, C.C.; Curtis, A.S.G. Functionalisation of Magnetic Nanoparticles for Applications in Biomedicine. J Phys D Appl Phys 2003, 36, R198. [CrossRef]
- Abdelwahab, A.A.; Eid, S.; Naggar, A.H.; Emran, M.Y.; El-Feky, H.H. A Novel Robust Biosensor Using Oxidized Copolymer Diaminonaphtalene/Remazol Blue R Capped Zinc Oxide Nanoparticles/Carbon Nanotubes Nanocomposite for Monitoring Adrenaline and Paracetamol in Bio-Fluids. Microchemical Journal 2025, 215, 114160. [CrossRef]
- Emran, M.Y.; El-Safty, S.A.; Elmarakbi, A.; Reda, A.; El Sabagh, A.; Shenashen, M.A. Chipset Nanosensor Based on N-Doped Carbon Nanobuds for Selective Screening of Epinephrine in Human Samples. Adv Mater Interfaces 2022, 9, 2101473. [CrossRef]
- Youssef, F.S.; El-Banna, H.A.; Elzorba, H.Y.; Galal, A.M. Application of Some Nanoparticles in the Field of Veterinary Medicine. Int J Vet Sci Med 2019, 7, 78. [CrossRef]
- Feng, S.; Zuo, C.; Qian, J.; Han, J.; Li, Y.; Tao, T.; Chen, Q. Single-Shot Absolute 3D Shape Measurement with Deep-Learning-Based Color Fringe Projection Profilometry. Optics Letters, Vol. 45, Issue 7, pp. 1842-1845 2020, 45, 1842–1845. [CrossRef]
- Zhou, W.; Gao, X.; Liu, D.; Chen, X. Gold Nanoparticles for in Vitro Diagnostics. Chem Rev 2015, 115, 10575–10636. [CrossRef]
- Subara, D.; Jaswir, I. Gold Nanoparticles: Synthesis and Application for Halal Authentication in Meat and Meat Products. Int J Adv Sci Eng Inf Technol 2018, 8, 1633–1641. [CrossRef]
- Kneipp, K.; Wang, Y.; Kneipp, H.; Perelman, L.T.; Itzkan, I.; Dasari, R.R.; Feld, M.S. Single Molecule Detection Using Surface-Enhanced Raman Scattering (SERS). Phys Rev Lett 1997, 78, 1667. [CrossRef]
- Nie, S.; Emory, S.R. Probing Single Molecules and Single Nanoparticles by Surface-Enhanced Raman Scattering. Science (1979) 1997, 275, 1102–1106. [CrossRef]
- Huang, F.; Zhang, Y.; Lin, J.; Liu, Y. Biosensors Coupled with Signal Amplification Technology for the Detection of Pathogenic Bacteria: A Review. Biosensors 2021, Vol. 11, Page 190 2021, 11, 190. [CrossRef]
- Thwala, L.N.; Ndlovu, S.C.; Mpofu, K.T.; Lugongolo, M.Y.; Mthunzi-Kufa, P. Nanotechnology-Based Diagnostics for Diseases Prevalent in Developing Countries: Current Advances in Point-of-Care Tests. Nanomaterials 2023, Vol. 13, Page 1247 2023, 13, 1247. [CrossRef]
- Lightner, D. V.; Redman, R.M.; Pantoja, C.R.; Tang, K.F.J.; Noble, B.L.; Schofield, P.; Mohney, L.L.; Nunan, L.M.; Navarro, S.A. Historic Emergence, Impact and Current Status of Shrimp Pathogens in the Americas. J Invertebr Pathol 2012, 110, 174–183. [CrossRef]
- Valenzuela-Amaro, H.M.; Aguayo-Acosta, A.; Meléndez-Sánchez, E.R.; de la Rosa, O.; Vázquez-Ortega, P.G.; Oyervides-Muñoz, M.A.; Sosa-Hernández, J.E.; Parra-Saldívar, R. Emerging Applications of Nanobiosensors in Pathogen Detection in Water and Food. Biosensors 2023, Vol. 13, Page 922 2023, 13, 922. [CrossRef]
- Dasari, R.; Vankara, A.P.; Khateef, R.; Shegu, V.R.T.; Degati, V.; Thummala, C. Role of Nanoparticles in Fish Disease Management: A Review. Biocatal Agric Biotechnol 2024, 58, 103218. [CrossRef]
- Hegde, M.; Pai, P.; Shetty, M.G.; Babitha, K.S. Gold Nanoparticle Based Biosensors for Rapid Pathogen Detection: A Review. Environ Nanotechnol Monit Manag 2022, 18, 100756. [CrossRef]
- Piliarik, M.; Vaisocherová, H.; Homola, J. Surface Plasmon Resonance Biosensing. Methods Mol Biol 2009, 503, 65–88. [CrossRef]
- Hassan, M.M.; Sium, F.S.; Islam, F.; Choudhury, S.M. A Review on Plasmonic and Metamaterial Based Biosensing Platforms for Virus Detection. Sens Biosensing Res 2021, 33. [CrossRef]
- Fattahi, Z.; Khosroushahi, A.Y.; Hasanzadeh, M. Recent Progress on Developing of Plasmon Biosensing of Tumor Biomarkers: Efficient Method towards Early Stage Recognition of Cancer. Biomedicine & Pharmacotherapy 2020, 132, 110850. [CrossRef]
- Akter, S.; Abdur Razzak, S.M. Highly Sensitive Open-Channels Based Plasmonic Biosensor in Visible to near-Infrared Wavelength. Results Phys 2019, 13, 102328. [CrossRef]
- Naresh, V.; Lee, N. A Review on Biosensors and Recent Development of Nanostructured Materials-Enabled Biosensors. Sensors 2021, Vol. 21, Page 1109 2021, 21, 1109. [CrossRef]
- Díaz, V.; Ibáñez, R.; Gómez, P.; Urtiaga, A.M.; Ortiz, I. Kinetics of Nitrogen Compounds in a Commercial Marine Recirculating Aquaculture System. Aquac Eng 2012, 50, 20–27. [CrossRef]
- Vráblová, M.; Koutník, I.; Smutná, K.; Marková, D.; Veverková, N. Combined SPRi Sensor for Simultaneous Detection of Nitrate and Ammonium in Wastewater. Sensors 2021, Vol. 21, Page 725 2021, 21, 725. [CrossRef]
- Endo, H.; Wu, H. Biosensors for the Assessment of Fish Health: A Review. Fisheries Science 2019, 85, 641–654. [CrossRef]
- Li, M.; Singh, R.; Soares, M.S.; Marques, C.; Zhang, B.; Zhang, B.; Kumar, S.; Kumar, S. Convex Fiber-Tapered Seven Core Fiber-Convex Fiber (CTC) Structure-Based Biosensor for Creatinine Detection in Aquaculture. Optics Express, Vol. 30, Issue 8, pp. 13898-13914 2022, 30, 13898–13914. [CrossRef]
- Leitão, C.; Leal-Junior, A.; Almeida, A.R.; Pereira, S.O.; Costa, F.M.; Pinto, J.L.; Marques, C. Cortisol AuPd Plasmonic Unclad POF Biosensor. Biotechnology Reports 2021, 29, e00587. [CrossRef]
- Yakes, B.J.; Buijs, J.; Elliott, C.T.; Campbell, K. Surface Plasmon Resonance Biosensing: Approaches for Screening and Characterising Antibodies for Food Diagnostics. Talanta 2016, 156–157, 55–63. [CrossRef]
- Surface Plasmon Resonance (SPR) - Lifeasible Available online: https://www.lifeasible.com/custom-solutions/plant/analytical-services/gene-function-analysis/surface-plasmon-resonance-spr/ (accessed on 7 December 2025).
- Cantilever | Construction, Engineering, Design | Britannica Available online: https://www.britannica.com/technology/cantilever (accessed on 29 April 2025).
- Thundat, T.; Wachter, E.A.; Sharp, S.L.; Warmack, R.J. Detection of Mercury Vapor Using Resonating Microcantilevers. Appl Phys Lett 1995, 66, 1695–1697. [CrossRef]
- Alunda, B.O.; Lee, Y.J. Review: Cantilever-Based Sensors for High Speed Atomic Force Microscopy. Sensors 2020, Vol. 20, Page 4784 2020, 20, 4784. [CrossRef]
- Lang, H.P.; Hegner, M.; Meyer, E.; Gerber, C. Nanomechanics from Atomic Resolution to Molecular Recognition Based on Atomic Force Microscopy Technology. INSTITUTE OF PHYSICS PUBLISHING NANOTECHNOLOGY Nanotechnology 2002, 13, 29–36.
- Sungkanak, U.; Sappat, A.; Wisitsoraat, A.; Promptmas, C.; Tuantranont, A. Ultrasensitive Detection of Vibrio Cholerae O1 Using Microcantilever-Based Biosensor with Dynamic Force Microscopy. Biosens Bioelectron 2010, 26, 784–789. [CrossRef]
- Loh, S.H.; Cheah, W.J. Optical Beam Deflection Based AFM with Integrated Hardware and Software Platform for an Undergraduate Engineering Laboratory. Applied Sciences 2017, Vol. 7, Page 226 2017, 7, 226. [CrossRef]
- Fathy, J.; Lai, Y. A V-Shaped Microcantilever Sensor Based on a Gap Method for Real-Time Detection of E. Coli Bacteria. Biosensors 2022, Vol. 12, Page 194 2022, 12, 194. [CrossRef]
- Liu, M.; Zhu, C.; Dong, Z.; Wang, Z.; Yang, H.; Li, J.; Li, K.; Shen, B.; Li, X.; Leng, P.; et al. Aptamer Proximal Enzyme Cascade Reactions for Ultrafast Detection of Glucose in Human Blood Serum. Microchimica Acta 2025 192:2 2025, 192, 71-. [CrossRef]
- Sin, M.L.; Mach, K.E.; Wong, P.K.; Liao, J.C. Advances and Challenges in Biosensor-Based Diagnosis of Infectious Diseases. Expert Rev Mol Diagn 2014, 14, 225–244. [CrossRef]
- Allain, J.P.; Opare-Sem, O. Screening and Diagnosis of HBV in Low-Income and Middle-Income Countries. Nat Rev Gastroenterol Hepatol 2016, 13, 643–653. [CrossRef]
- Palladino, P.; Minunni, M.; Scarano, S. Cardiac Troponin T Capture and Detection in Real-Time via Epitope-Imprinted Polymer and Optical Biosensing. Biosens Bioelectron 2018, 106, 93–98. [CrossRef]
- Ogi, H. Wireless-Electrodeless Quartz-Crystal-Microbalance Biosensors for Studying Interactions among Biomolecules: A Review. Proc Jpn Acad Ser B Phys Biol Sci 2013, 89, 401. [CrossRef]
- Cooper, M.A.; Singleton, V.T. A Survey of the 2001 to 2005 Quartz Crystal Microbalance Biosensor Literature: Applications of Acoustic Physics to the Analysis of Biomolecular Interactions. Journal of Molecular Recognition 2007, 20, 154–184. [CrossRef]
- Tai, D.F.; Lin, C.Y.; Wu, T.Z.; Chen, L.K. Recognition of Dengue Virus Protein Using Epitope-Mediated Molecularly Imprinted Film. Anal Chem 2005, 77, 5140–5143. [CrossRef]
- Li, D.; Wang, J.; Wang, R.; Li, Y.; Abi-Ghanem, D.; Berghman, L.; Hargis, B.; Lu, H. A Nanobeads Amplified QCM Immunosensor for the Detection of Avian Influenza Virus H5N1. Biosens Bioelectron 2011, 26, 4146–4154. [CrossRef]
- Zhang, L.Q.; Shen, H.X.; Cheng, Q.; Liu, L.C. Sensitive Electrogravimetric Immunoassay of Hepatitis B Surface Antigen through Hyperbranched Polymer Bridge Linked to Multiple Secondary Antibodies. Chemical Papers 2016, 70, 1031–1038. [CrossRef]
- Vasapollo, G.; Sole, R. Del; Mergola, L.; Lazzoi, M.R.; Scardino, A.; Scorrano, S.; Mele, G. Molecularly Imprinted Polymers: Present and Future Prospective. Int J Mol Sci 2011, 12, 5908–5945. [CrossRef]
- Buchatip, S.; Ananthanawat, C.; Sithigorngul, P.; Sangvanich, P.; Rengpipat, S.; Hoven, V.P. Detection of the Shrimp Pathogenic Bacteria, Vibrio Harveyi, by a Quartz Crystal Microbalance-Specific Antibody Based Sensor. Sens Actuators B Chem 2010, 145, 259–264. [CrossRef]
- Lim, H.J.; Saha, T.; Tey, B.T.; Tan, W.S.; Ooi, C.W. Quartz Crystal Microbalance-Based Biosensors as Rapid Diagnostic Devices for Infectious Diseases. Biosens Bioelectron 2020, 168, 112513. [CrossRef]
- Singh, P.; Pandey, S.K.; Singh, J.; Srivastava, S.; Sachan, S.; Singh, S.K. Biomedical Perspective of Electrochemical Nanobiosensor. Nanomicro Lett 2016, 8, 193–203. [CrossRef]
- Upasham, S.; Banga, I.K.; Jagannath, B.; Paul, A.; Lin, K.C.; Muthukumar, S.; Prasad, S. Electrochemical Impedimetric Biosensors, Featuring the Use of Room Temperature Ionic Liquids (RTILs): Special Focus on Non-Faradaic Sensing. Biosens Bioelectron 2021, 177, 112940. [CrossRef]
- Belluzo, M.S.; Ribone, M.É.; Lagier, C.M. Assembling Amperometric Biosensors for Clinical Diagnostics. Sensors (Basel) 2008, 8, 1366. [CrossRef]
- Mikkelsen, S.R.; Rechnitz, G.A. Conductometric Transducers for Enzyme-Based Biosensors. Anal Chem 1989, 61, 1737–1742. [CrossRef]
- Su, X.; Sutarlie, L.; Loh, X.J. Sensors, Biosensors, and Analytical Technologies for Aquaculture Water Quality. Research (Wash D C) 2020, 2020. [CrossRef]
- Wang, C.; Wang, T.; Li, Z.; Xu, X.; Zhang, X.; Li, D. An Electrochemical Enzyme Biosensor for Ammonium Detection in Aquaculture Using Screen-Printed Electrode Modified by Gold Nanoparticle/Polymethylene Blue. Biosensors 2021, Vol. 11, Page 335 2021, 11, 335. [CrossRef]
- Das, J.; Mishra, H.N. Electrochemical Biosensor for Monitoring Fish Spoilage Based on Nanocellulose as Enzyme Immobilization Matrix. Journal of Food Measurement and Characterization 2023 17:4 2023, 17, 3827–3844. [CrossRef]
- Fletcher, D.J. Plasma Glucose and Plasma Fatty Acid Levels of Limanda Limanda (L.) in Relation to Season, Stress, Glucose Loads and Nutritional State. J Fish Biol 1984, 25, 629–648. [CrossRef]
- Maita, M.; Aoki, H.; Yamagata, Y.; Satoh, S.; Okamoto, N.; Watanabe, T. Plasma Biochemistry and Disease Resistance in Yellowtail Fed a Non-Fish Meal Diet. Fish Pathol 1998, 33, 59–63. [CrossRef]
- Maita, M.; Satoh, K.I.; Fukuda, Y.; Lee, H.K.; Winton, J.R.; Okamoto, N. Correlation between Plasma Component Levels of Cultured Fish and Resistance to Bacterial Infection. Fish Pathol 1998, 33, 129–133. [CrossRef]
- Oliveira, J.; Oliva-Teles, A.; Couto, A. Tracking Biomarkers for the Health and Welfare of Aquaculture Fish. Fishes 2024, Vol. 9, Page 289 2024, 9, 289. [CrossRef]
- Lemos, L.S.; Angarica, L.M.; Hauser-Davis, R.A.; Quinete, N. Cortisol as a Stress Indicator in Fish: Sampling Methods, Analytical Techniques, and Organic Pollutant Exposure Assessments. Int J Environ Res Public Health 2023, 20, 6237. [CrossRef]
- Ravariu, C.; Parvulescu, C.C.; Manea, E.; Tucureanu, V. Optimized Technologies for Cointegration of MOS Transistor and Glucose Oxidase Enzyme on a Si-Wafer. Biosensors 2021, Vol. 11, Page 497 2021, 11, 497. [CrossRef]
- Inoue, K.; Wada, M.; Higuchi, T.; Oshio, S.; Umeda, T.; Yoshimura, Y.; Nakazawa, H. Application of Liquid Chromatography–Mass Spectrometry to the Quantification of Bisphenol A in Human Semen. Journal of Chromatography B 2002, 773, 97–102. [CrossRef]
- Tschmelak, J.; Proll, G.; Gauglitz, G. Verification of Performance with the Automated Direct Optical TIRF Immunosensor (River Analyser) in Single and Multi-Analyte Assays with Real Water Samples. Biosens Bioelectron 2004, 20, 743–752. [CrossRef]
- Chilmonczyk, S.; Monge, D. Flow Cytometry as a Tool for Assessment of the Fish Cellular Immune Response to Pathogens. Fish Shellfish Immunol 1999, 9, 319–333. [CrossRef]
- González, S.F.; Krug, M.J.; Nielsen, M.E.; Santos, Y.; Call, D.R. Simultaneous Detection of Marine Fish Pathogens by Using Multiplex PCR and a DNA Microarray. J Clin Microbiol 2004, 42, 1414. [CrossRef]
- Umasankar, Y.; Chen, S.M. A Review on the Electrochemical Sensors and Biosensors Composed of Nanowires as Sensing Material. Sensors 2008, Vol. 8, Pages 290-313 2008, 8, 290–313. [CrossRef]
- Windmiller, J.R.; Wang, J. Wearable Electrochemical Sensors and Biosensors: A Review. Electroanalysis 2013, 25, 29–46. [CrossRef]
- Grieshaber, D.; MacKenzie, R.; Vörös, J.; Reimhult, E. Electrochemical Biosensors - Sensor Principles and Architectures. Sensors (Basel) 2008, 8, 1400. [CrossRef]
- Muramatsu, T.; Ohnuki, H.; Ushio, H.; Hibi, K.; Igarashi, M.; Hayashi, T.; Ren, H.; Endo, H. Electrochemical Flow Injection Immunoassay for Cortisol Using Magnetic Microbeads. Int J Environ Anal Chem 2011, 91, 161–173. [CrossRef]
- Wu, H.; Ohnuki, H.; Murata, M.; Endo, H. Flow Immunosensor System with an Electrode Replacement Unit for Continuous Cortisol Monitoring for Fish. Sens Biosensing Res 2017, 13, 122–127. [CrossRef]
- Wu, H.; Ohnuki, H.; Ren, H.; Endo, H. Carbon-Nanotube-Enhanced Label-Free Immunosensor for Highly Sensitive Detection of Plasma Cortisol Level in Fish. Sensors and Materials 2015, 27, 793–803.
- Wu, H.; Ohnuki, H.; Hibi, K.; Ren, H.; Endo, H. Development of a Label-Free Immunosensor System for Detecting Plasma Cortisol Levels in Fish. Fish Physiol Biochem 2016, 42, 19–27. [CrossRef]
- Wu, H.; Ohnuki, H.; Ota, S.; Murata, M.; Yoshiura, Y.; Endo, H. New Approach for Monitoring Fish Stress: A Novel Enzyme-Functionalized Label-Free Immunosensor System for Detecting Cortisol Levels in Fish. Biosens Bioelectron 2017, 93, 57–64. [CrossRef]
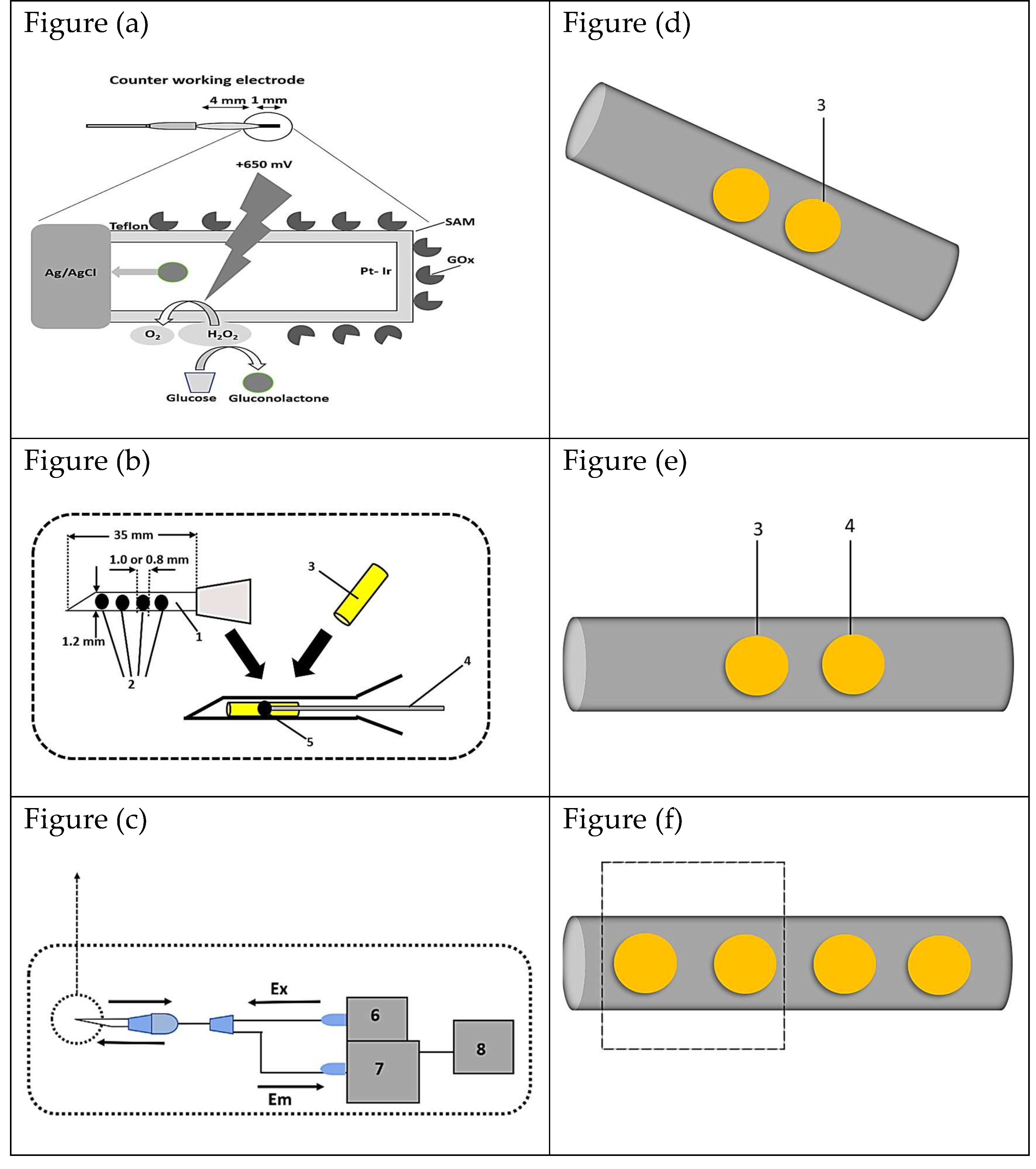
- Endo, H.; Yonemori, Y.; Hibi, K.; Ren, H.; Hayashi, T.; Tsugawa, W.; Sode, K. Wireless Enzyme Sensor System for Real-Time Monitoring of Blood Glucose Levels in Fish. Biosens Bioelectron 2009, 24, 1417–1423. [CrossRef]
- Endo, H.; Takahashi, E.; Murata, M.; Ohnuki, H.; Ren, H.; Tsugawa, W.; Sode, K. Wireless Monitoring of Blood Glucose Levels in Flatfish with a Needle Biosensor. Fisheries Science 2010, 76, 687–694. [CrossRef]
- Endo, H.; Yonemori, Y.; Musiya, K.; Maita, M.; Shibuya, T.; Ren, H.; Hayashi, T.; Mitsubayashi, K. A Needle-Type Optical Enzyme Sensor System for Determining Glucose Levels in Fish Blood. Anal Chim Acta 2006, 573–574, 117–124. [CrossRef]
- Wu, H.; Aoki, A.; Arimoto, T.; Nakano, T.; Ohnuki, H.; Murata, M.; Ren, H.; Endo, H. Fish Stress Become Visible: A New Attempt to Use Biosensor for Real-Time Monitoring Fish Stress. Biosens Bioelectron 2015, 67, 503–510. [CrossRef]
- Endo, H.; Muramatsu, T.; Yoshizaki, G.; Ren, H.; Ohnuki, H. Development of a Label-Free Immunosensor System for Detecting Oocyte Maturation-Inducing Hormone in Fish. Fisheries Science 2011, 78, 391–398. [CrossRef]
- Hirai, M.; Muramatsu, T.; Ohnuki, H.; Hibi, K.; Ren, H.; Endo, H. Carbon Nanotube Enhanced Label-Free Immunosensor for Amperometric Determination of Oocyte Maturation-Inducing Hormone in Fish. Fish Physiol Biochem 2013, 39, 299–308. [CrossRef]
- Hibi, K.; Hatanaka, K.; Takase, M.; Ren, H.; Endo, H. Wireless Biosensor System for Real-Time L-Lactic Acid Monitoring in Fish. Sensors 2012, Vol. 12, Pages 6269-6281 2012, 12, 6269–6281. [CrossRef]
- Hibi, K.; Yoshiura, Y.; Ushio, H.; Ren, H.; Endo, H. Rapid Detection of Flavobacterium Psychrophilum Using Fluorescent Magnetic Beads and Flow Cytometry. Sensors and Materials 2012, 24, 311–322.
- Hibi, K.; Ushio, H.; Fukuda, H.; Mitsubayashi, K.; Hayashi, T.; Ren, H.; Endo, H. Immunomagnetic Separation Using Carbonyl Iron Powder and Flow Cytometry for Rapid Detection of Flavobacterium Psychrophilum. Anal Bioanal Chem 2008, 391, 1147–1152. [CrossRef]
- Hibi, K.; Abe, A.; Ohashi, E.; Mitsubayashi, K.; Ushio, H.; Hayashi, T.; Ren, H.; Endo, H. Combination of Immunomagnetic Separation with Flow Cytometry for Detection of Listeria Monocytogenes. Anal Chim Acta 2006, 573–574, 158–163. [CrossRef]
- Ryumae, U.; Hibi, K.; Yoshiura, Y.; Ren, H.; Endo, H. Rapid and Highly Sensitive Detection of Flavobacterium Psychrophilum Using High Gradient Immunomagnetic Separation with Flow Cytometry. Aquaculture 2010, 309, 125–130. [CrossRef]
- Ryumae, U.; Hibi, K.; Yoshiura, Y.; Ren, H.; Endo, H. Ultra Highly Sensitive Method for Detecting Flavobacterium Psychrophilum Using High-Gradient Immunomagnetic Separation with a Polymerase Chain Reaction. Aquac Res 2012, 43, 929–939. [CrossRef]
- Takase, M.; Murata, M.; Hibi, K.; Huifeng, R.; Endo, H. Development of Mediator-Type Biosensor to Wirelessly Monitor Whole Cholesterol Concentration in Fish. Fish Physiol Biochem 2014, 40, 385–394. [CrossRef]
- Taogesi; Wu, H.; Murata, M.; Ren, H.; Endo, H. Carbon Nanotube-Enhanced Enzyme Sensor for Real-Time Monitoring of Cholesterol Levels in Free-Swimming Fish. Sensors and Materials 2015, 27, 805–815. [CrossRef]
- Endo, H.; Maita, M.; Takikawa, M.; Ren, H.; Hayashi, T.; Urano, N.; Mitsubayashi, K. Enzyme Sensor System for Determination of Total Cholesterol in Fish Plasma. FISHERIES SCIENCE 2003, 69, 1194–1199. [CrossRef]
- Wu, H.; Fujii, Y.; Nakano, T.; Arimoto, T.; Murata, M.; Matsumoto, H.; Yoshiura, Y.; Ohnuki, H.; Endo, H. Development of a Novel Enhanced Biosensor System for Real-Time Monitoring of Fish Stress Using a Self-Assembled Monolayer. Sensors 2019, Vol. 19, Page 1518 2019, 19, 1518. [CrossRef]
- Matsuyama, M.; Adachi, S.; Nagahama, Y.; Kitajima, C.; Matsuura, S. Annual Reproductive Cycle of the Captive Female Japanese Sardine Sardinops Melanostictus: Relationship to Ovarian Development and Serum Levels of Gonadal Steroid Hormones. Mar Biol 1991, 108, 21–29. [CrossRef]
- Wu, H.; Yamada, K.; Murata, M.; Matsumoto, H.; Ohnuki, H.; Endo, H. A Novel Interactive Biosensor System for Real-Time Remote Stress Response Monitoring and Visualization by Using Bi-Directional Data Link. Biosens Bioelectron X 2022, 10, 100133. [CrossRef]
- Wu, H.; Aoki, A.; Arimoto, T.; Nakano, T.; Ohnuki, H.; Murata, M.; Ren, H.; Endo, H. Fish Stress Become Visible: A New Attempt to Use Biosensor for Real-Time Monitoring Fish Stress. Biosens Bioelectron 2015, 67, 503–510. [CrossRef]
- Bass, S.L.S.; Gerlai, R. Zebrafish (Danio Rerio) Responds Differentially to Stimulus Fish: The Effects of Sympatric and Allopatric Predators and Harmless Fish. Behavioural Brain Research 2008, 186, 107–117. [CrossRef]
- Barton, B.A.; Iwama, G.K. Physiological Changes in Fish from Stress in Aquaculture with Emphasis on the Response and Effects of Corticosteroids. Annu Rev Fish Dis 1991, 1, 3–26. [CrossRef]
- Yonemori, Y.; Takahashi, E.; Ren, H.; Hayashi, T.; Endo, H. Biosensor System for Continuous Glucose Monitoring in Fish. Anal Chim Acta 2009, 633, 90–96. [CrossRef]
- Wu, H.; Shinoda, R.; Murata, M.; Matsumoto, H.; Ohnuki, H.; Endo, H. Real-Time Fish Stress Visualization Came True:A Novel Multi-Stage Color-Switching Wireless Biosensor System. Biosens Bioelectron 2019, 130, 360–366. [CrossRef]
- Johari, S.A.; Kalbassi, M.R.; Soltani, M.; Yu, I.J. Application of Nanosilver-Coated Zeolite as Water Filter Media for Fungal Disinfection of Rainbow Trout (Oncorhynchus Mykiss) Eggs. Aquaculture International 2016, 24, 23–38. [CrossRef]
- Gupta, R.; Kulkarni, G.U. Removal of Organic Compounds from Water by Using a Gold Nanoparticle-Poly(Dimethylsiloxane) Nanocomposite Foam. ChemSusChem 2011, 4, 737–743. [CrossRef]
- Krishnani, K.K.; Srinives, S.; Mohapatra, B.C.; Boddu, V.M.; Hao, J.; Meng, X.; Mulchandani, A. Hexavalent Chromium Removal Mechanism Using Conducting Polymers. J Hazard Mater 2013, 252–253, 99–106. [CrossRef]
- Chakraborty, P.; Krishnani, K.K. Emerging Bioanalytical Sensors for Rapid and Close-to-Real-Time Detection of Priority Abiotic and Biotic Stressors in Aquaculture and Culture-Based Fisheries. Science of The Total Environment 2022, 838, 156128. [CrossRef]
- Pires, N.M.M.; Dong, T.; Yang, Z.; da Silva, L.F.B.A. Recent Methods and Biosensors for Foodborne Pathogen Detection in Fish: Progress and Future Prospects to Sustainable Aquaculture Systems. Crit Rev Food Sci Nutr 2021, 61, 1852–1876. [CrossRef]
- Vu, Q.K.; Tran, Q.H.; Vu, N.P.; Anh, T. Le; Dang, T.T. Le; Matteo, T.; Nguyen, T.H.H. A Label-Free Electrochemical Biosensor Based on Screen-Printed Electrodes Modified with Gold Nanoparticles for Quick Detection of Bacterial Pathogens. Mater Today Commun 2021, 26, 101726. [CrossRef]
- Hou, Y.; Zhu, L.; Hao, H.; Zhang, Z.; Ding, C.; Zhang, G.; Bi, J.; Yan, S.; Liu, G.; Hou, H. A Novel Photoelectrochemical Aptamer Sensor Based on Rare-Earth Doped Bi2WO6 and Ag2S for the Rapid Detection of Vibrio Parahaemolyticus. Microchemical Journal 2021, 165, 106132. [CrossRef]
- Nakama, K.; Sedki, M.; Mulchandani, A. Label-Free Chemiresistor Biosensor Based on Reduced Graphene Oxide and M13 Bacteriophage for Detection of Coliforms. Anal Chim Acta 2021, 1150, 338232. [CrossRef]
- Norouz Dizaji, A.; Ali, Z.; Ghorbanpoor, H.; Ozturk, Y.; Akcakoca, I.; Avci, H.; Dogan Guzel, F. Electrochemical-Based ‘”Antibiotsensor”’ for the Whole-Cell Detection of the Vancomycin-Susceptible Bacteria. Talanta 2021, 234, 122695. [CrossRef]
- Sheini, A. A Point-of-Care Testing Sensor Based on Fluorescent Nanoclusters for Rapid Detection of Septicemia in Children. Sens Actuators B Chem 2021, 328, 129029. [CrossRef]
- Ma, X.; wang, C.; Qin, M.; Tian, X.; Fan, S.; Zu, H.; Lyu, M.; Wang, S. Rapid Detection of Aeromonas Hydrophila with a DNAzyme-Based Sensor. Food Control 2021, 123. [CrossRef]
- Yin, L.; Duan, N.; Chen, S.; Yao, Y.; Liu, J.; Ma, L. Ultrasensitive Pathogenic Bacteria Detection by a Smartphone-Read G-Quadruplex-Based CRISPR-Cas12a Bioassay. Sens Actuators B Chem 2021, 347, 130586. [CrossRef]
- Gogola, J.L.; Martins, G.; Gevaerd, A.; Blanes, L.; Cardoso, J.; Marchini, F.K.; Banks, C.E.; Bergamini, M.F.; Marcolino-Junior, L.H. Label-Free Aptasensor for P24-HIV Protein Detection Based on Graphene Quantum Dots as an Electrochemical Signal Amplifier. Anal Chim Acta 2021, 1166. [CrossRef]
- Cao, M.; Sun, Q.; Zhang, X.; Ma, Y.; Wang, J. Detection and Differentiation of Respiratory Syncytial Virus Subgroups A and B with Colorimetric Toehold Switch Sensors in a Paper-Based Cell-Free System. Biosens Bioelectron 2021, 182, 113173. [CrossRef]
- Zhao, X.; Zhu, Z.; Zou, R.; Wang, L.; Gong, H.; Cai, C. An Enzyme-Free Three-Dimensional DNA Walker Powered by Catalytic Hairpin Assembly for H5N1 DNA Ratiometric Detection. Microchemical Journal 2021, 170. [CrossRef]
- Boonkaew, S.; Yakoh, A.; Chuaypen, N.; Tangkijvanich, P.; Rengpipat, S.; Siangproh, W.; Chailapakul, O. An Automated Fast-Flow/Delayed Paper-Based Platform for the Simultaneous Electrochemical Detection of Hepatitis B Virus and Hepatitis C Virus Core Antigen. Biosens Bioelectron 2021, 193. [CrossRef]
- Wang, C.; Wang, C.; Wang, X.; Wang, K.; Zhu, Y.; Rong, Z.; Wang, W.; Xiao, R.; Wang, S. Magnetic SERS Strip for Sensitive and Simultaneous Detection of Respiratory Viruses. ACS Appl Mater Interfaces 2019, 11, 19495–19505. [CrossRef]
- Mohanty, B.P. Fish as Health Food. Handbook of Fisheries and Aquaculture 2011, 843–861.
- Bandyopadhyay, K.; Misra, S.; Bhattacharya, S.; Mukherjee, A.; Shee, A. A Review on Utilization of Biosensors for Detection of Adulteration in Fish. International Journal for Modern Trends in Science and Technology 2020, 10, 50–55.
- Roberts, B.; Morris, B.A.; Clifford, M.N. Comparison of Radioimmunoassay and Spectrophotometric Analysis for the Quantitation of Hypoxanthine in Fish Muscle. Food Chem 1991, 42, 1–17. [CrossRef]
- Yano, Y.; Kataho, N.; Watanabe, M.; Nakamura, T.; Asano, Y. Evaluation of Beef Aging by Determination of Hypoxanthine and Xanthine Contents: Application of a Xanthine Sensor. Food Chem 1995, 52, 439–445. [CrossRef]
- Agüí, L.; Manso, J.; Yáñez-Sedeño, P.; Pingarrón, J.M. Amperometric Biosensor for Hypoxanthine Based on Immobilized Xanthine Oxidase on Nanocrystal Gold–Carbon Paste Electrodes. Sens Actuators B Chem 2006, 113, 272–280. [CrossRef]
- Lawal, A.T.; Adeloju, S.B. Progress and Recent Advances in Fabrication and Utilization of Hypoxanthine Biosensors for Meat and Fish Quality Assessment: A Review. Talanta 2012, 100, 217–228. [CrossRef]
- Lin, Z.; Sun, J.; Chen, J.; Guo, L.; Chen, Y.; Chen, G. Electrochemiluminescent Biosensor for Hypoxanthine Based on the Electrically Heated Carbon Paste Electrode Modified with Xanthine Oxidase. Anal Chem 2008, 80, 2826–2831. [CrossRef]
- Evelázio De Souza, N.; Nakatani, H.S.; Vieira, L.; Santos, D.; Peralta Pelegrine, C.; Terezinha, S.; Gomes, M.; Matsushita, M.; Vergilio Visentainer, J. Biosensor Based on Xanthine Oxidase for Monitoring Hypoxanthine in Fish Meat. Am J Biochem Biotechnol 2005, 1, 85–89. [CrossRef]
- Yan, Z.; Niu, Q.; Mou, M.; Wu, Y.; Liu, X.; Liao, S. A Novel Colorimetric Method Based on Copper Nanoclusters with Intrinsic Peroxidase-like for Detecting Xanthine in Serum Samples. Journal of Nanoparticle Research 2017, 19, 1–12. [CrossRef]
- Chang, L.Y.; Chuang, M.Y.; Zan, H.W.; Meng, H.F.; Lu, C.J.; Yeh, P.H.; Chen, J.N. One-Minute Fish Freshness Evaluation by Testing the Volatile Amine Gas with an Ultrasensitive Porous-Electrode-Capped Organic Gas Sensor System. ACS Sens 2017, 2, 531–539. [CrossRef]
- Kuske, M.; Romain, A.C.; Nicolas, J. Microbial Volatile Organic Compounds as Indicators of Fungi. Can an Electronic Nose Detect Fungi in Indoor Environments? Build Environ 2005, 40, 824–831. [CrossRef]
- Pais, G.L.; Meloni, D.; Mudadu, A.G.; Crobu, L.; Pulina, A.; Chessa, G. Colorimetric Analysis and Determination of Histamine in Samples of Yellowfin Tuna (Thunnus Albacares) Marketed in Sardinia (Italy) by a Combination of Rapid Screening Methods and LC-MS/MS. Foods 2022, 11. [CrossRef]
- Colombo, F.M.; Cattaneo, P.; Confalonieri, E.; Bernardi, C. Histamine Food Poisonings: A Systematic Review and Meta-Analysis. Crit Rev Food Sci Nutr 2018, 58, 1131–1151. [CrossRef]
- FDA Issues Draft Compliance Policy Guide for Decomposition and Histamine in Scombrotoxin (Histamine)-Forming Fish and Fishery Products | FDA Available online: https://www.fda.gov/food/hfp-constituent-updates/fda-issues-draft-compliance-policy-guide-decomposition-and-histamine-scombrotoxin-histamine-forming (accessed on 15 May 2025).
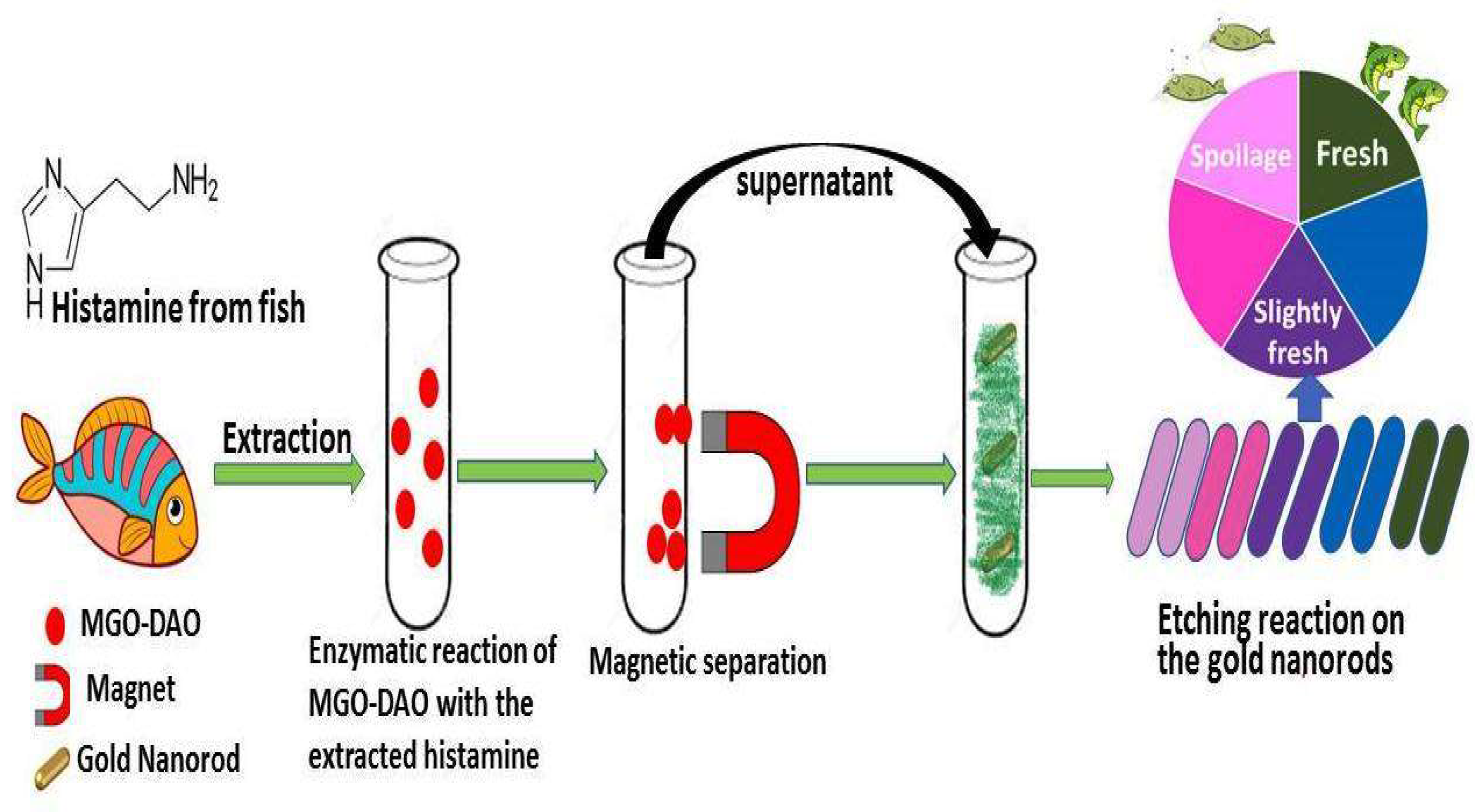
- Xu, X.; Wu, X.; Zhuang, S.; Zhang, Y.; Ding, Y.; Zhou, X. Colorimetric Biosensor Based on Magnetic Enzyme and Gold Nanorods for Visual Detection of Fish Freshness. Biosensors (Basel) 2022, 12, 135. [CrossRef]
- Vasconcelos, H.; Coelho, L.C.C.; Matias, A.; Saraiva, C.; Jorge, P.A.S.; de Almeida, J.M.M.M. Biosensors for Biogenic Amines: A Review. Biosensors (Basel) 2021, 11, 82. [CrossRef]
- Lingamdinne, L.P.; Koduru, J.R.; Karri, R.R. A Comprehensive Review of Applications of Magnetic Graphene Oxide Based Nanocomposites for Sustainable Water Purification. J Environ Manage 2019, 231, 622–634. [CrossRef]
- Fukuda, Y.; Maita, M.; Satoh, K.I.; Okamoto, N. Influence of Dissolved Oxygen Concentration on the Mortality of Yellowtail Experimentally Infected with Enterococcus Seriolicida. Fish Pathol 1997, 32, 129–130. [CrossRef]
- Davis, C.R.; Marty, G.D.; Adkison, M.A.; Freiberg, E.F.; Hedrick, R.P. Association of Plasma IgM with Body Size, Histopathologic Changes, and Plasma Chemistries in Adult Pacific Herring Clupea Pallasi. Dis Aquat Organ 1999, 38, 125–133. [CrossRef]
- Singh, R.; Singh, A.K.; Tripathi, M. Melatonin Induced Changes in Specific Growth Rate, Gonadal Maturity, Lipid and Protein Production in Nile Tilapia Oreochromis Niloticus (Linnaeus 1758). Asian-Australas J Anim Sci 2012, 25, 37–43. [CrossRef]
- Řehulka, J.; Minařík, B. Cholesterolaemia and Triacylglycerolaemia in Farmed Rainbow Trout, Oncorhynchus Mykiss. Aquac Res 2012, 43, 1651–1659. [CrossRef]
- Lahnsteiner, F.; Mansour, N.; McNiven, M.A.; Richardson, G.F. Fatty Acids of Rainbow Trout (Oncorhynchus Mykiss) Semen: Composition and Effects on Sperm Functionality. Aquaculture 2009, 298, 118–124. [CrossRef]
- Yoneyama, Y.; Yonemori, Y.; Murata, M.; Ohnuki, H.; Hibi, K.; Hayashi, T.; Ren, H.; Endo, H. Wireless Biosensor System for Real-Time Cholesterol Monitoring in Fish “Nile Tilapia.” Talanta 2009, 80, 909–915. [CrossRef]
- DICKS, J.; CARDOSI, M.; TURNER, A.; KARUBE, I. The Application of Ferrocene-Modified N-Type Silicon in Glucose Biosensors. Electroanalysis 1993, 5.
- van Staveren, D.R.; Metzler-Nolte, N. Bioorganometallic Chemistry of Ferrocene. Chem Rev 2004, 104, 5931–5985. [CrossRef]
- Sharma, D.; Kumar, R. Smart Aquaculture: Integration of Sensors, Biosensors, and Artificial Intelligence. 2021, 455–464. [CrossRef]
- Dhinakaran, D.; Gopalakrishnan, S.; Manigandan, M.D.; Anish, T.P. IoT-Based Environmental Control System for Fish Farms with Sensor Integration and Machine Learning Decision Support. International Journal on Recent and Innovation Trends in Computing and Communication 2023, 11, 203–217. [CrossRef]
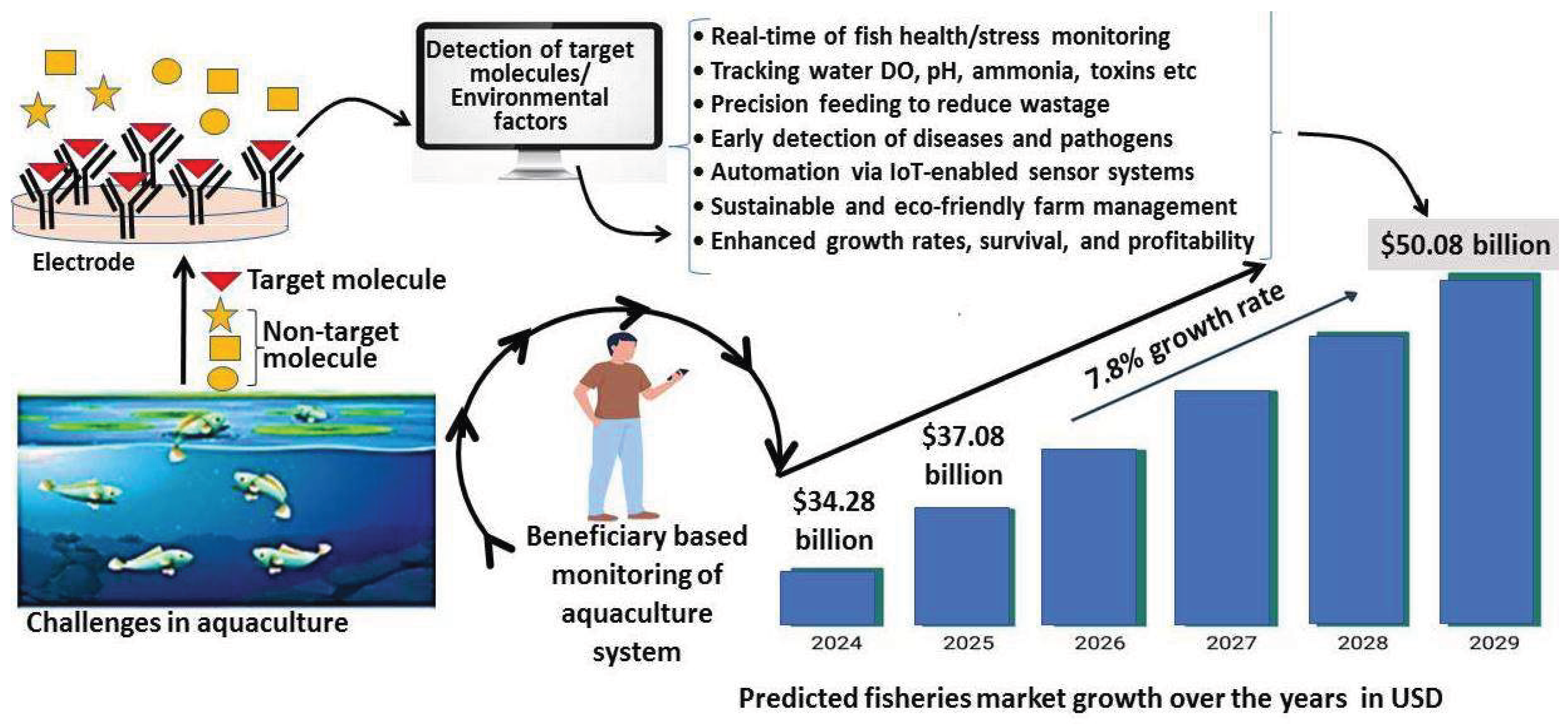
| Challenges | Effects | References |
| Climate Change | Alters physical and physiological aspects of finfish and shellfish; changes in primary and secondary productivity; ecosystem structure alterations. | [33,34,35] |
| Sea-Level Rise and Inland Temperature Changes | Damages coastal ecosystems, mangroves, and salt marshes; reduces fish abundance and distribution. | [36] |
| Geographical and Climatic Variations | Varying impacts on aquaculture based on location, climate zones, economy, production systems, and species. | [35,37,38,39,40,41,42,43] |
| Rising Temperatures | Affects growth and development of aquatic animals; impacts endocrine and osmoregulatory systems; leads to algal blooms reducing dissolved oxygen; coral bleaching affecting reef fisheries. | [36,44] |
| Ocean Acidification | Decline in pH affecting shell-forming organisms like shrimps, mussels, oysters, and corals; increased vulnerability to predation; higher production costs due to reliance on hatcheries. | [41,44,45,46,47,48,49] |
| Changes in Precipitation Patterns | Increased rainfall causing floods introducing invasive species; droughts leading to water stress, stock loss, and higher maintenance costs. | [36,44] |
| Sea-Level Rise and Natural Phenomena | Salinity intrusions threatening aquaculture; anthropogenic activities like shrimp farming exacerbating salinity intrusion affecting aquatic flora and fauna. | [50,51,62] |
| Ammonia Toxicity | Harmful effects on aquatic life; inverse relationship with dissolved oxygen; eutrophication due to imbalance in ammonia and nitrate levels. | [52,63,64] |
| Disease Outbreaks | Parasitic, bacterial, viral, and fungal diseases causing significant harm; examples include pancreatic necrosis in salmonids and fin rot in Atlantic salmon and rainbow trout. | [65,66,67,68] |
| Need for Sex Control | Ensures stability of mating systems; prevents precocious maturation; desire for mono-sex populations for better growth rates, e.g., male Nile tilapia grow faster. | [55,56] |
| Stress Factors | Negatively impacts growth, reproduction, immune function; influenced by diet, handling, and transport; poor water quality and oxygen levels exacerbate stress. | [60,69,70] |
| Lack of Education and Awareness | Aqua culturists often being poor are inadequately prepared to adapt to changes, making them vulnerable to impacts on fish resources. | [36] |
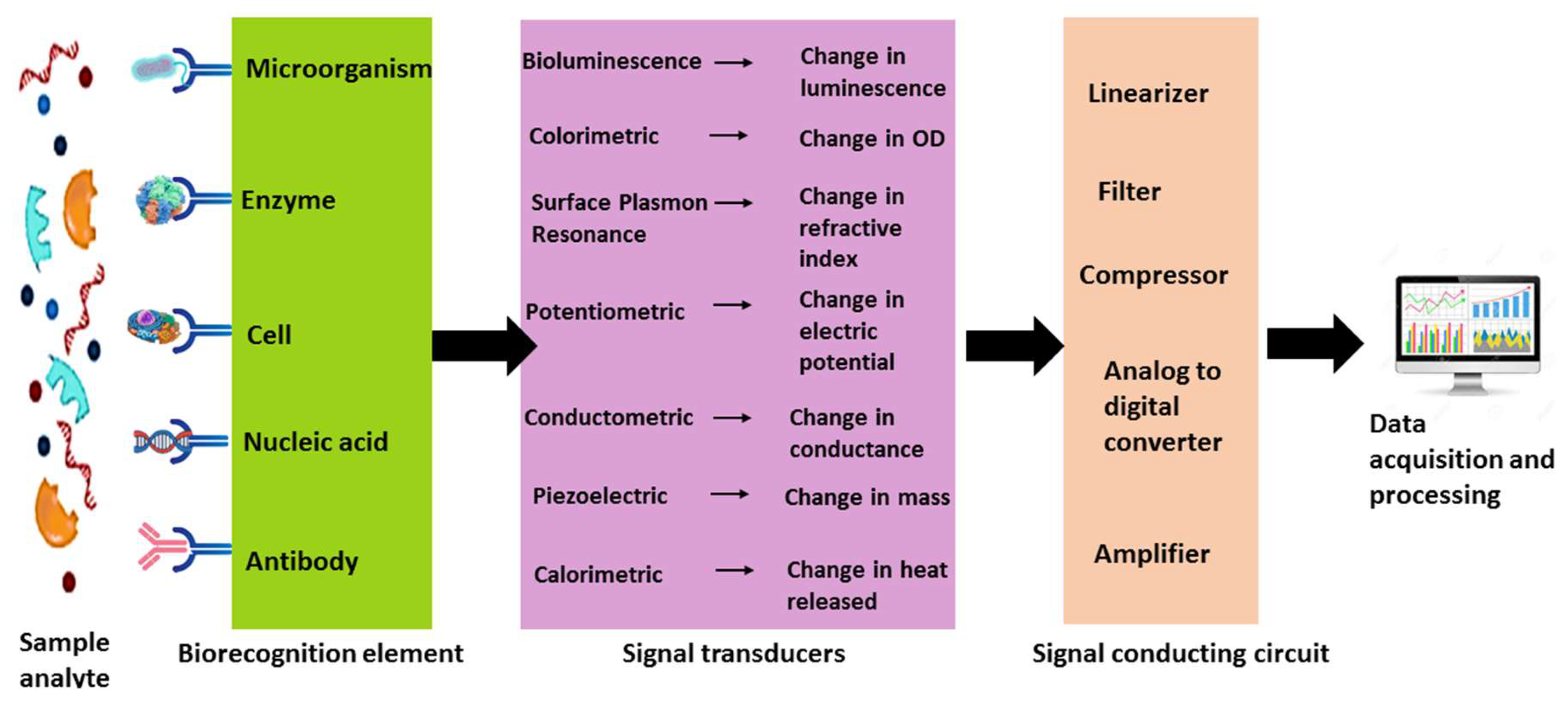
| Transducer type | Biosensor type | Working principle | Application | Reference |
| Optical | Bioluminescent | Luminescence variation | Detection of heavy metals, food toxicants, and pathogens | [73] |
| Fluorescence | Interaction with fluorescence-labeled biomolecules | BOD measurement, water availability assessment in plants, pathogen detection | [74] | |
| Colorimetric | Changes in optical density | Identification of waterborne and foodborne pathogens | [75] | |
| Surface Plasmon Resonance | Variation in refractive index due to bio analyte binding | Disease diagnosis, drug residue analysis, livestock monitoring, toxic gas detection | [76] | |
| Electrochemical | Potentiometric | Measures electric potential | Detection of urea, CO2, pesticides, sugars, and pH levels | [77] |
| Conductometric | Variation in electrical conductance | Monitoring environmental contamination, pesticide, and heavy metal detection | [78] | |
| Amperometric | Electron transfer due to redox reactions | Identification of organophosphate pesticides and pathogens | [79] | |
| Impedimetric | Measures electrochemical cell impedance | Detection of peptides, small proteins, milk toxins, and foodborne pathogens | [80] | |
| Piezoelectric | Quartz Crystal Microbalance | Mass variations in biological components | Used as a humidity sensor, food safety tool, and for detecting organophosphate and carbamate pesticides, as well as glucose measurement | [81] |
| Thermal | Calorimetric | Heat absorption and release | Detection of organophosphate pesticides and food and waterborne pathogens | [82] |
| Parameter | Electrochemical Sensors | Optical Sensors | QCM Sensors |
| Principle | Electrical changes in the form of current, voltage, impedance from biochemical reactions is measured | Changes in light signals in the form of absorbance, fluorescence, Raman, colorimetry is measured | Mass change on crystal surface via shift in resonance frequency is detcted |
| Typical Targets | Water parameters such as NH3, NO2−, pH, dissolved O2, metabolites, stress biomarkers are measured | Biological molecules such as proteins, enzymes, hormones, pathogens, dissolved O2, turbidity, toxins, physiological stress indicators can be measured. | Titer and level of infection of pathogens, proteins, toxins, mucus biomarkers, immune molecules can be measured |
| Sensitivity | High sensitivity as low analyte levels can be measured with modified electrodes | Very high because it uses fluorescence/Raman spectral signal and allows multiplexing | Extremely high because uses mass measurement and sensitive up to ng–pg level |
| Selectivity | Moderate but can be enhanced with integration of additives such as nanomaterials | Optical probes provide highly selective molecules | Antibodies, aptamers provide high selectivity |
| Real-time Monitoring | Excellent and can be used routinely | Water transparency can hinder the signal and needs stable signal for real time monitoring | Usually limited for real tie assays |
| Environmental Interference | Can be affected by fouling and electrode change such as degradation | Turbidity, scattering, ambient light can influence the signal | Physical parameters such as temperature, viscosity, and water flow may affect |
| Cost | Usually low | Usually moderate | Usually high |
| Ease of use in fish farms | Easy | Moderately easy | Little difficult |
| Major Advantages | Low cost, fast response, portable, easy automation | Highly sensitive, non-invasive, supports remote sensing | Ultra-sensitive, label-free, ideal for pathogen detection |
| Major Limitations | Electrode fouling, routine calibration is required | Usually costly and can be affected by water transparency | Environmental changes may affect the signaling |
|
Example |
Ammonia (NH3/NH4+) Electrochemical Sensor, Cortisol Electrochemical Immunosensor, Dissolved Oxygen Electrochemical Sensor (Clark-type electrode), Heavy Metal Detection Sensor (Pb2+, Cd2+, Hg2+) sensors |
Fluorescent DO Sensor, Raman Spectroscopy for Fish Pathogens, Colorimetric Ammonia Sensor Strip, Fiber-Optic pH Sensor, Fluorescence-based Cortisol Detection | QCM Immunosensor for Fish Pathogens, QCM Biosensor for Fish Mucus Proteins, QCM for Toxin Detection (e.g., microcystins), Aptamer-functionalized QCM for Antibiotic Residues |
| Name of biosensor | Detected substance | Mechanism | References |
| Cortisol Biosensor | Cortisol levels in fish plasma | Electrochemical immunoassay using glucose oxidase (Gox) as a signal amplifier | [154,155,156,157,158] |
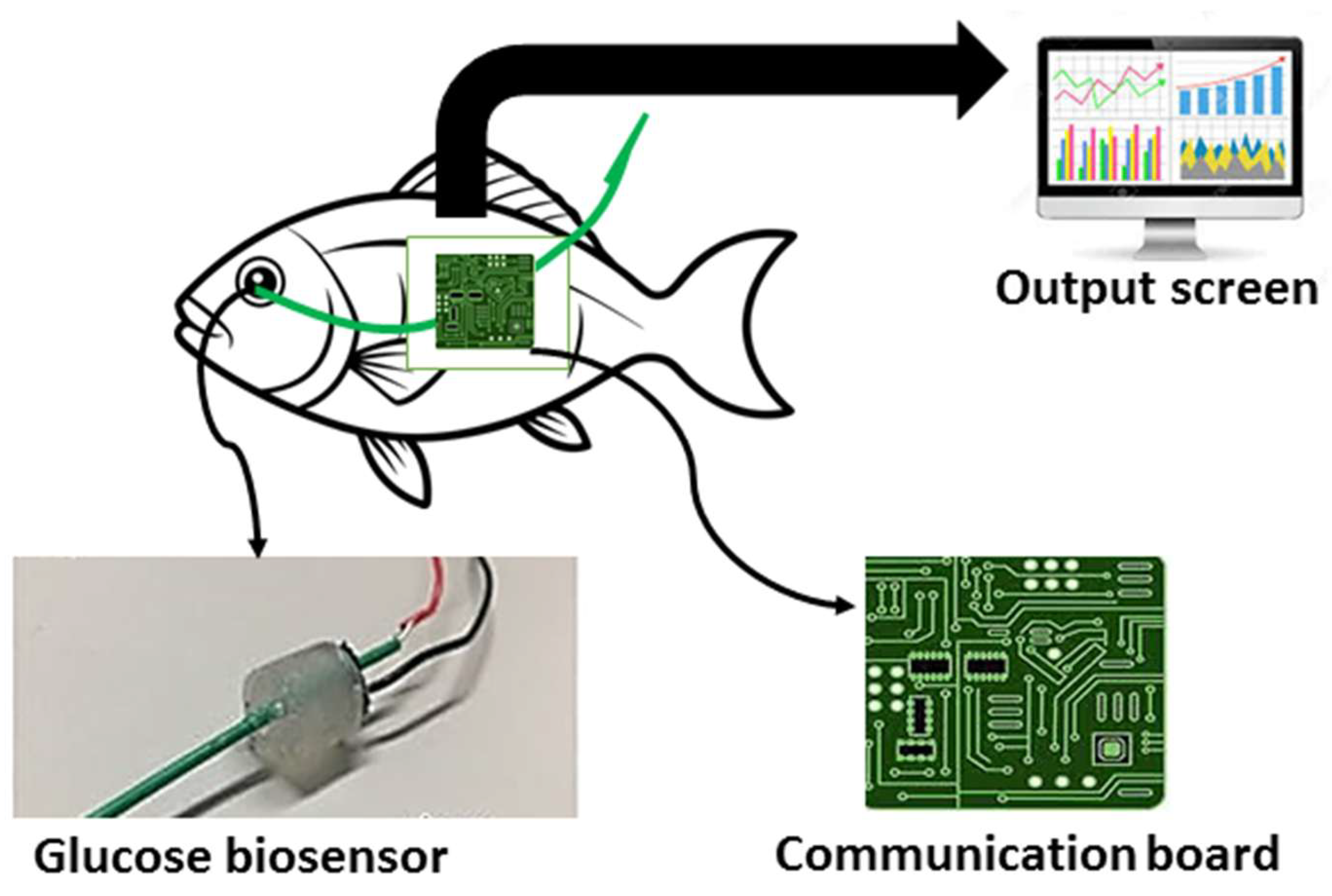
| Glucose Biosensor | Blood glucose levels | Needle-type optical enzyme sensor system | [159,160,161] |
| Wireless Glucose Biosensor | Real-time glucose monitoring in fish | Microelectrode with enzyme reaction, wireless transmission | [159,160,162] |
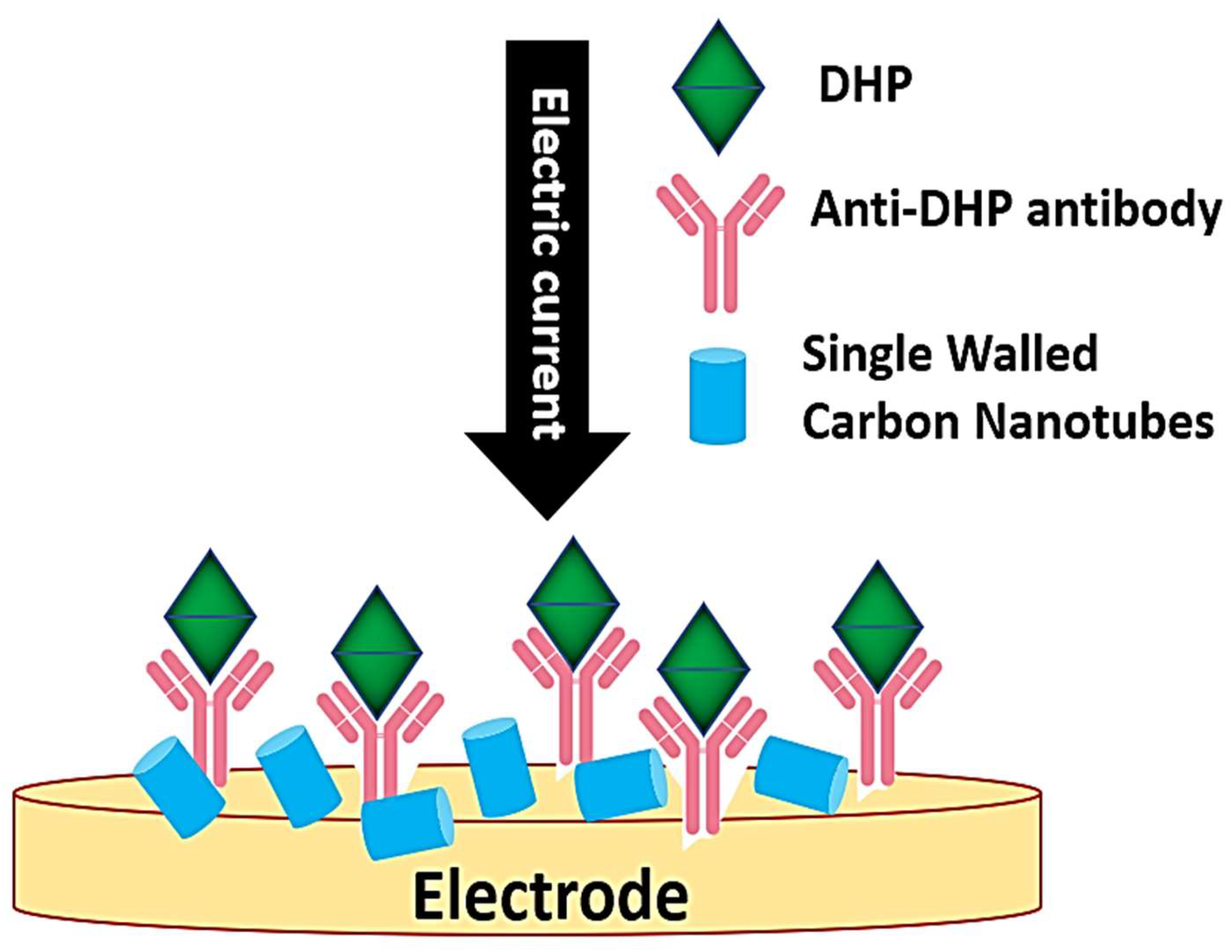
| Ovulation Prediction Biosensor (DHP Sensor) | 17,20β-Dihydroxy-4-pregnen-3-one (DHP) for predicting spawning time | Label-free immunosensor, carbon nanotube-enhanced electrochemical detection | [163,164] |
| Pathogen Detection Biosensor (HGIMS-FCM & HGIMS-PCR) | Detection of Flavobacterium psychrophilum in fish | High Gradient Immunomagnetic Separation (HGIMS) with Flow Cytometry (FCM) or Polymerase Chain Reaction (PCR) | [165,166,167,168,169,170] |
| Total Cholesterol Biosensor | Whole cholesterol concentration in fish | Wireless CNT-enhanced mediator-type sensor | [171,172] |
| L-Lactic Acid Biosensor | Blood L-lactic acid levels | Enzyme-based FIA biosensor | [159,165,173] |
| Visual Stress Monitoring Biosensor (LED Biosensor) | Fish stress response (glucose-based detection) | Color-switching biosensor using LED indicators | [174] |
| Name of biosensor | Stress detection | Mechanism | References |
| Silver nanoparticle-coated zeolite filter | Fungal infections in rainbow trout culture | Water filtration using silver nanoparticle-coated zeolite to reduce fungal infections | [182] |
| Gold-PDMS (Polydimethylsiloxane) filter | Removal of contaminants (thiophenol, thioanisole, toluene, sodium sulfide) from wastewater | Gold-PDMS-based filtration system for contaminant removal | [183] |
| Electrochemically synthesized conducting polymer-polyaniline sensor | Detection of heavy metal contamination (Cr VI) in water | Conducting polymer-based electrochemical sensor for chromium detection at low pH | [184] |
| Fluorescence-based biosensor | Detection of White Spot Syndrome Virus in shrimp and ammonia-oxidizing bacteria in aquaculture soil and biofilms | Fluorescence-based sensitive method for pathogen detection | [185] |
| Name of biosensor | Target pathogen | Mechanism | References |
| Electrochemical Biosensor | Escherichia coli O157 | Uses anti-E. coli O157 antibody on carbon screen-printed electrodes (SPEs) with gold nanoparticle modification for stability and sensitivity. | [187] |
| Photo-electrochemical Biosensor | Vibrio parahaemolyticus | Aptamer-based detection utilizing a layered assembly method for photocurrent response. | [188] |
| Chemiresistor Biosensor | Escherichia coli strains | M13 phage used to modify sensor material for F-pili selective detection via reduced graphene oxide. | [189] |
| Electrochemical Impedance Spectroscopy | Staphylococcus aureus | Uses vancomycin for bacterial binding. | [190] |
| Fluorescence Biosensor | Staphylococcus aureus, Streptococcus pyogenes, Escherichia coli, Pseudomonas aeruginosa | Uses fluorescence emission under UV light with gold and copper nanoclusters. | [191] |
| Fluorescence DNAzyme Sensor | Aeromonas hydrophila | DNAzyme-based fluorescence emission mechanism | [192] |
| Colorimetric CRISPR-Cas12a Sensor | Salmonella | Uses CRISPR-Cas12a technology for detection. | [193] |
| Name of biosensor | Target virus | Mechanism | References |
| Electrochemical Biosensor | HIV (Human Immunodeficiency Virus) | Uses graphene quantum dots to amplify electrochemical signal and immobilize p24-HIV aptamer for detection of p24 protein. | [194] |
| Toehold Switch Sensor | Respiratory Syncytial Virus (RSV) (Subgroups A & B) | Detects RSV RNA trigger sequences via a toehold switch mechanism producing a colorimetric output. | [195] |
| Fluorescence DNA Walker Sensor | H5N1 (Avian Influenza Virus) | Uses DNA walker mechanism for fluorescence-based viral DNA detection | [196] |
| Electrochemical Immunosensor | Hepatitis B and C Viruses | Hepatitis B and C Viruses Uses anti-HBV and anti-HCV antibodies to detect respective surface/core antigens. | [197] |
| SERS-Based Biosensor | Influenza A H1N1 & Human Adenovirus (HAdV) | Uses monoclonal antibodies with surface-enhanced Raman spectroscopy for detection. | [198] |
| Electrochemiluminescence Biosensor | HIV (HIV-1 Protein) | Uses metal-organic framework (ZIF-8) for sensitive HIV detection. | (Wang et al., 2021) |
| Name of biosensor | Freshness indicator | Mechanism | References |
| Enzymatic Biosensor | Hypoxanthine (ATP degradation product) | Xanthine oxidase (XOD) immobilized on electrodes detects hypoxanthine oxidation to xanthine and uric acid, producing H2O2. Colorimetric or electrochemical detection is used. | [201,202,203,204,205,206] |
| Colorimetric Sensor | Xanthine (byproduct of hypoxanthine oxidation) | Uses oxidation of 3,3’,5,5’-tetramethylbenzidine (TMB) dye enhanced by copper nanoclusters in the presence of H2O2 (produced from xanthine oxidation). | [207] |
| Gold Nanorods (GNRs)-Based Sensor | Hypoxanthine | Hypoxanthine oxidized by XOD produces H2O2, which undergoes a Fenton reaction in the presence of Fe2+, leading to a visible color change. Different hypoxanthine concentrations generate different colors. | [207] |
| Organic Semiconductor Gas Sensor | Total Volatile Bases (TVB) - Ammonia, Dimethylamine (DMA), Trimethylamine (TMA) | Uses a porous top metal electrode with a nanostructured surface to enhance gas adsorption, detecting volatile compounds linked to fish spoilage. | [208] |
| Electronic Nose (E-nose) with MOS-FET Gas Sensor | Volatile Compounds from Fish Spoilage | Uses metal oxide semiconductor (MOS-FET) sensors to detect volatile amines and microbial spoilage markers in fish. | [209] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





