Submitted:
12 December 2025
Posted:
15 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. The Systematic Review
2.2. Statistics
3. Results
3.1. Menstruation and CRP
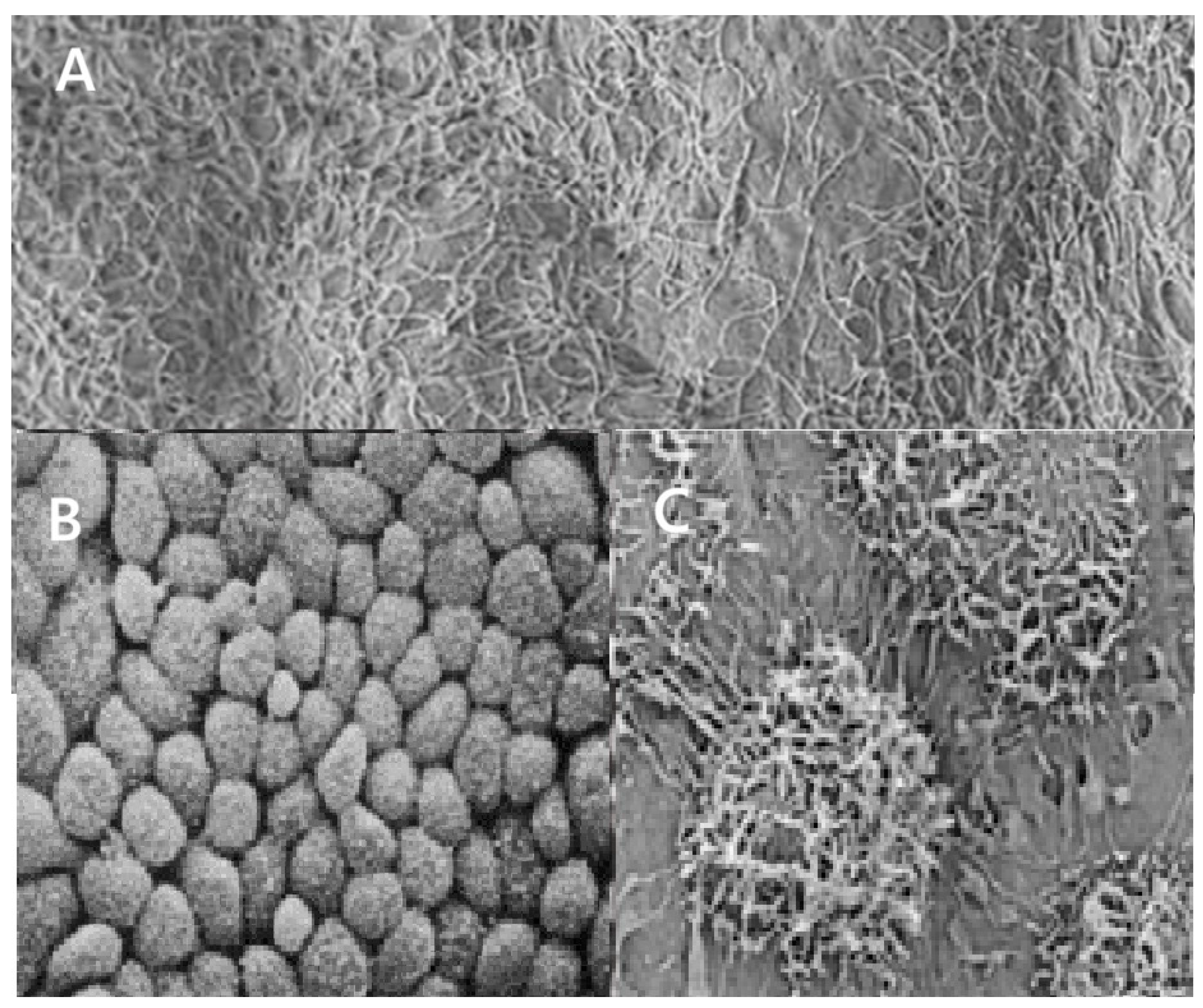
3.2. Menstruation, Inflammation and Adhesion Formation
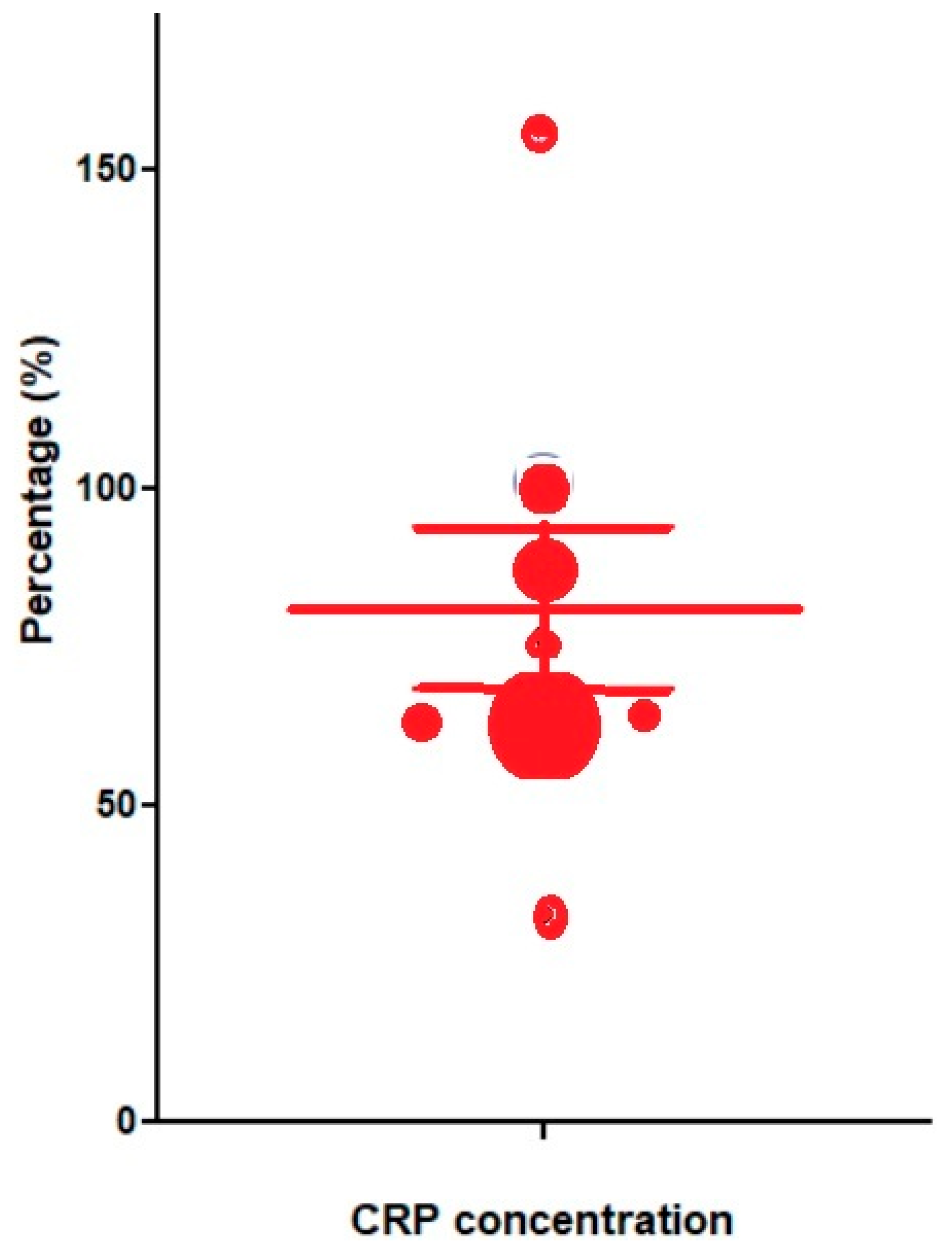
3.3. Logarithmic Distribution of CRP Concentrations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CRP | C-reactive protein |
| SD | Standard deviation |
| PMS | Premenstrual syndrome |
| EF | Early follicular |
| LF | Late follicular |
| LL | Late luteal |
| SQR | Semi Quartile Range |
| PCOS | Polycystic ovarian syndrome |
References
- Koninckx, P.R.; Ide, P.; Vandenbroucke, W.; Brosens, I.A. New aspects of the pathophysiology of endometriosis and associated infertility. J Reprod. Med 1980, 24, 257-260, https://pubmed.ncbi.nlm.nih.gov/7420327/.
- Halme, J.; Hammond, M.G.; Hulka, J.F.; Raj, S.G.; Talbert, L.M. Retrograde menstruation in healthy women and in patients with endometriosis. Obstet Gynecol 1984, 64, 151-154.
- Sampson, J.A. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol 1927, 14, 422-469. [CrossRef]
- Kruitwagen, R.F. Menstruation as the pelvic aggressor. Baillieres Clin Obstet Gynaecol 1993, 7, 687-700. [CrossRef]
- Raimondo, D.; Raffone, A.; Aru, A.C.; Salucci, P.; Travaglino, A.; Maletta, M.; Ambrosio, M.; Borghese, G.; Iodice, R.; Casadio, P.; et al. C-reactive Protein for Predicting Early Postoperative Complications in Patients Undergoing Laparoscopic Shaving for Deep Infiltrating Endometriosis. Journal of Minimally Invasive Gynecology 2022, 29, 135-143. [CrossRef] [PubMed]
- Raimondo, D.; Raffone, A.; Aru, A.C.; Salucci, P.; Travaglino, A.; Maletta, M.; Ambrosio, M.; Borghese, G.; Iodice, R.; Casadio, P.; et al. C-reactive Protein for Predicting Early Postoperative Complications in Patients Undergoing Laparoscopic Shaving for Deep Infiltrating Endometriosis. J Minim Invasive Gynecol 2021. [CrossRef]
- De Cicco, C.; Schonman, R.; Ussia, A.; Koninckx, P.R. Extensive peritoneal lavage decreases postoperative C-reactive protein concentrations: a RCT. Gynecol Surg 2015, 12, 271-274. [CrossRef]
- Koninckx, P.R.; Gomel, V.; Ussia, A.; Adamyan, L. Role of the peritoneal cavity in the prevention of postoperative adhesions, pain, and fatigue. Fertil Steril 2016, 106, 998-1010. [CrossRef]
- Koninckx, P.R.; Ussia, A.; Adamyan, L.; Wattiez, A.; Vigano, P. Understanding Peritoneal Fluid Estrogen and Progesterone Concentrations Permits Individualization of Medical Treatment of Endometriosis-Associated Pain with Lower Doses, Especially in Adolescents Not Requiring Contraception. Journal of Clinical Medicine 2025, 14, 7196. [CrossRef] [PubMed]
- Mutsaers, S.E.; Pixley, F.J.; Prêle, C.M.; Hoyne, G.F. Mesothelial cells regulate immune responses in health and disease: role for immunotherapy in malignant mesothelioma. Curr. Opin. Immunol. 2020, 64, 88-109. [CrossRef] [PubMed]
- Koninckx, P.R.; Ussia, A.; Tahlak, M.; Adamyan, L.; Wattiez, A.; Martin, D.C.; Gomel, V. Infection as a potential cofactor in the genetic-epigenetic pathophysiology of endometriosis: a systematic review. Facts Views Vis Obgyn 2019, 11, 209-216.
- Tang, F.; Deng, M.; Xu, C.; Yang, R.; Ji, X.; Hao, M.; Wang, Y.; Tian, M.; Geng, Y.; Miao, J. Unraveling the microbial puzzle: exploring the intricate role of gut microbiota in endometriosis pathogenesis. Frontiers in cellular and infection microbiology 2024, 14, 1328419. [CrossRef]
- Guo, C.; Zhang, C. Role of the gut microbiota in the pathogenesis of endometriosis: a review. Front Microbiol 2024, 15, 1363455. [CrossRef]
- nervus vagus microbiota. 2024.
- Salmeri, N.; Sinagra, E.; Dolci, C.; Buzzaccarini, G.; Sozzi, G.; Sutera, M.; Candiani, M.; Ungaro, F.; Massimino, L.; Danese, S.; et al. Microbiota in Irritable Bowel Syndrome and Endometriosis: Birds of a Feather Flock Together-A Review. Microorganisms 2023, 11. [CrossRef]
- Lin, Q.; Duan, H.; Wang, S.; Guo, Z.; Wang, S.; Chang, Y.; Chen, C.; Shen, M.; Shou, H.; Zhou, C. Endometrial microbiota in women with and without adenomyosis: A pilot study. Front Microbiol 2023, 14, 1075900. [CrossRef]
- Kobayashi, H. Gut and reproductive tract microbiota: Insights into the pathogenesis of endometriosis (Review). Biomed Rep 2023, 19, 43. [CrossRef]
- Yuan, W.; Wu, Y.; Chai, X.; Wu, X. The colonized microbiota composition in the peritoneal fluid in women with endometriosis. Archives of Gynecology and Obstetrics 2022. [CrossRef] [PubMed]
- Binda, M.M.; Riiskjaer, M.; Koninckx, P.R. Pneumoperitoneum induced mesothelial cell changes in a laparoscopic mouse model. European Journal of Obstetrics & Gynecology and Reproductive Biology 2021, 265, 107-112. [CrossRef] [PubMed]
- Volz, J.; Koster, S.; Spacek, Z.; Paweletz, N. Characteristic alterations of the peritoneum after carbon dioxide pneumoperitoneum. Surg. Endosc 1999, 13, 611-614.
- Mutsaers, S.E.; Prele, C.M.; Pengelly, S.; Herrick, S.E. Mesothelial cells and peritoneal homeostasis. Fertil Steril 2016, 106, 1018-1024. [CrossRef]
- Binda, M.M.; Corona, R.; Amant, F.; Koninckx, P.R. Conditioning of the abdominal cavity reduces tumor implantation in a laparoscopic mouse model. Surg Today 2014, 44, 1328-1335. [CrossRef]
- Corona, R.; Verguts, J.; Schonman, R.; Binda, M.M.; Mailova, K.; Koninckx, P.R. Postoperative inflammation in the abdominal cavity increases adhesion formation in a laparoscopic mouse model. Fertil. Steril 2011, 95, 1224-1228. [CrossRef]
- Ryan, G.B.; Majno, G. Acute inflammation. A review. Am J Pathol 1977, 86, 183-276.
- Özdemir-Van Brunschot, D.M.D.; Van Laarhoven, K.C.J.H.M.; Scheffer, G.-J.; Pouwels, S.; Wever, K.E.; Warlé, M.C. What is the evidence for the use of low-pressure pneumoperitoneum? A systematic review. Surg. Endosc. 2016, 30, 2049-2065. [CrossRef] [PubMed]
- Breborowicz, A.; Oreopoulos, D.G. Is normal saline harmful to the peritoneum? Perit. Dial. Int 2005, 25 Suppl 4, S67-S70. [CrossRef]
- Corona, R.; Binda, M.M.; Mailova, K.; Verguts, J.; Koninckx, P.R. Addition of nitrous oxide to the carbon dioxide pneumoperitoneum strongly decreases adhesion formation and the dose-dependent adhesiogenic effect of blood in a laparoscopic mouse model. Fertil. Steril 2013, 100, 1777-1783. [CrossRef] [PubMed]
- Viganò, P.; Caprara, F.; Giola, F.; Di Stefano, G.; Somigliana, E.; Vercellini, P. Is retrograde menstruation a universal, recurrent, physiological phenomenon? A systematic review of the evidence in humans and non-human primates. Hum Reprod Open 2024, 2024, hoae045. [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. PLoS Med. 2021, 18, e1003583. [CrossRef] [PubMed]
- Koninckx, P.R.; Heyns, W.; Verhoeven, G.; Van, B.H.; Lissens, W.D.; De, M.P.; Brosens, I.A. Biochemical characterization of peritoneal fluid in women during the menstrual cycle. J Clin. Endocrinol. Metab 1980, 51, 1239-1244. [CrossRef]
- Koninckx, P.; De Hertogh, R.; Heyns, W.; Meulepas, E.; Brosens, I.; De Moor, P. Secretion rates of LH and FSH during infusion of LH-FSH/RH in normal women and in patients with secondary amenorrhea: suggestive evidence for two pools of LH and FSH. J Clin Endocrinol Metab 1976, 43, 159-167. [CrossRef] [PubMed]
- Sollberger, S.; Ehlert, U. How to use and interpret hormone ratios. Psychoneuroendocrinology 2016, 63, 385-397. [CrossRef]
- Blum, C.A.; Müller, B.; Huber, P.; Kraenzlin, M.; Schindler, C.; De Geyter, C.; Keller, U.; Puder, J.J. Low-grade inflammation and estimates of insulin resistance during the menstrual cycle in lean and overweight women. J Clin Endocrinol Metab 2005, 90, 3230-3235. [CrossRef] [PubMed]
- Talebpour, A.; Mohammadifard, M.; Reza; Mahmoudzadeh, S.; Rezapour, H.; Saharkhiz, M.; Tajik, M.; Ferns, G.A.; Bahrami, A. Effect of curcumin on inflammatory biomarkers and iron profile in patients with premenstrual syndrome and dysmenorrhea: A randomized controlled trial. Physiological Reports 2023, 11. [CrossRef]
- Yama, K.; Asari, Y.; Ono, A.; Machida, M.; Miura, J. Plasma Interleukin-10 Levels Are Altered in Women with Severe Premenstrual Syndrome: A Preliminary Study. Women’s Health Reports 2020, 1, 73-79. [CrossRef]
- Chaireti, R.; Lindahl, T.L.; Byström, B.; Bremme, K.; Larsson, A. Inflammatory and endothelial markers during the menstrual cycle. Scand. J. Clin. Lab. Invest. 2016, 76, 190-194. [CrossRef]
- Gursoy, A.Y.; Caglar, G.S.; Kiseli, M.; Pabuccu, E.; Candar, T.; Demirtas, S. CRP at early follicular phase of menstrual cycle can cause misinterpretation for cardiovascular risk assessment. Interventional Medicine and Applied Science 2015, 7, 143-146. [CrossRef]
- Gaskins, A.J.; Wilchesky, M.; Mumford, S.L.; Whitcomb, B.W.; Browne, R.W.; Wactawski-Wende, J.; Perkins, N.J.; Schisterman, E.F. Endogenous Reproductive Hormones and C-reactive Protein Across the Menstrual Cycle: The BioCycle Study. Am. J. Epidemiol. 2012, 175, 423-431. [CrossRef]
- Wander, K.; Brindle, E.; O’Connor, K.A. C-reactive protein across the menstrual cycle. Am. J. Phys. Anthropol. 2008, 136, 138-146. [CrossRef] [PubMed]
- Puder, J.J.; Blum, C.A.; Mueller, B.; De Geyter, C.; Dye, L.; Keller, U. Menstrual cycle symptoms are associated with changes in low-grade inflammation. Eur J Clin Invest 2006, 36, 58-64. [CrossRef]
- Hellebrekers, B.W.J.; Trimbos-Kemper, T.C.M.; Boesten, L.; Jansen, F.W.; Kolkman, W.; Trimbos, J.B.; Press, R.R.; Van Poelgeest, M.I.E.; Emeis, S.J.; Kooistra, T. Preoperative predictors of postsurgical adhesion formation and the Prevention of Adhesions with Plasminogen Activator (PAPA-study): results of a clinical pilot study. Fertility and Sterility 2009, 91, 1204-1214. [CrossRef]
- Berbic, M.; Ng, C.H.; Fraser, I.S. Inflammation and endometrial bleeding. Climacteric 2014, 17 Suppl 2, 47-53. [CrossRef]
- Evans, J.; Salamonsen, L.A. Inflammation, leukocytes and menstruation. Rev Endocr Metab Disord 2012, 13, 277-288. [CrossRef]
- Bricou, A.; Batt, R.E.; Chapron, C. Peritoneal fluid flow influences anatomical distribution of endometriotic lesions: why Sampson seems to be right. Eur. J. Obstet. Gynecol. Reprod. Biol 2008, 138, 127-134.
- Kyathanahalli, C.N.; Tu, F.F.; Hellman, K.M. Inflammatory Mechanisms of Dysmenorrhea: Novel Insights From Menstrual Effluent in an Adolescent Cohort. BJOG: An International Journal of Obstetrics & Gynaecology 2025, 132, 1626-1634. [CrossRef]
- Schonman, R.; Corona, R.; Bastidas, A.; De, C.C.; Koninckx, P.R. Effect of Upper Abdomen Tissue Manipulation on Adhesion Formation between Injured Areas in a Laparoscopic Mouse Model. J. Minim. Invasive. Gynecol 2009, 16, 307-312. [CrossRef]
- Burke, R.K.; Hertig, A.T.; Miele, C.A. Prognostic value of subacute focal inflammation of the endometrium, with special reference to pelvic adhesions as observed on laparoscopic examination: An eight-year review. J. Reprod. Med 1985, 30, 646-650.
- Prasad, K. C-Reactive Protein (CRP)-Lowering Agents. Cardiovasc. Drug Rev. 2006, 24, 33-50. [CrossRef] [PubMed]
- Donnez, O.; A., V.L.; Defrere, S.; Colette, S.; O., V.K.; Dehoux, J.P.; Squifflet, J.; Donnez, J. Induction of endometriotic nodules in an experimental baboon model mimicking human deep nodular lesions. Fertil Steril 2013, 99, 783-789. [CrossRef] [PubMed]
- Koninckx, P.R.; Verguts, J.; Corona, R.; Adamyan, L.; Brosens, I. A mixture of 86% of CO2, 10% of N2O, and 4% of oxygen permits laparoscopy under local anesthesia: a pilot study. Gynecol Surg 2015, 12, 57-60. [CrossRef] [PubMed]
- Cibula, D.; Widschwendter, M.; Májek, O.; Dusek, L. Tubal ligation and the risk of ovarian cancer: review and meta-analysis. Hum Reprod Update 2011, 17, 55-67. [CrossRef] [PubMed]
- Magarakis, L.; Idahl, A.; Särnqvist, C.; Strandell, A. Efficacy and safety of sterilisation procedures to reduce the risk of epithelial ovarian cancer: a systematic review comparing salpingectomy with tubal ligation. The European Journal of Contraception & Reproductive Health Care 2022, 27, 230-239. [CrossRef]
- Jahanfar, S.; Mortazavi, J.; Lapidow, A.; Cu, C.; Al Abosy, J.; Morris, K.; Becerra-Mateus, J.C.; Steinfeldt, M.; Maurer, O.; Bohang, J.; et al. Assessing the impact of contraceptive use on reproductive cancer risk among women of reproductive age—a systematic review. Frontiers in Global Women’s Health 2024, 5. [CrossRef]
- Arshadi, M.; Hesari, E.; Ahmadinezhad, M.; Yekta, E.M.; Ebrahimi, F.; Azizi, H.; Esfarjani, S.V.; Rostami, M.; Khodamoradi, F. The association between oral contraceptive pills and ovarian cancer risk: A systematic review and meta-analysis. Bull. Cancer 2024, 111, 918-929. [CrossRef] [PubMed]
| Menstrual or EF | Late follicular or luteal | P value |
% increase (Mean±SD) |
|||||
| N |
Mean (Median) |
SD (SQR) [min -max] |
Mean (median) |
SD (SQR) [min -max] |
||||
| Talebpour et al. 2023 [34] | 111 | (0.4) | [0.0-4.0] | LL | (0.2) | [0.0-2.0] | - | 100 |
| Yama et al 2020 [35] | 21 | (0.24) | (0.1-0.4) | LL | (0.18) | (0.1-0.6) | NS | 33 |
| Chiareti et al 2015 [36] | 102 | 1.65 | 3 | LL | 0.89 | 1 | 0.025 | 85 |
| Gursoy et al 2015 [37] | 27 | (1.8) | [0.3-7.7] | LF | (0.7) | [0.1-8.3] | <0.001 | 157 |
| Gaskins et al. 2012 [38] | 509 | (0.75) | (0.4-1.7) | LF | (0.45) | (0.3-1.2) | 0.001 | 66 |
| Wander et al 2008 [39] | 72 | 2.8 | - | LF | 1.7 | - | 0.01 | 64 |
| Puder et al 2006 [40] | 15 | 1.4 | 1.3 | LF | 0.8 | 0.74 | 0.007 | 75 |
| Blum et al. 2005 [33] | 15 | 130%£ | 50% | LF | 78% | 30% | 0.00001 | 67 |
| 80±36 | ||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





