2. Data Description
2.1. Overview of the Dataset
This dataset contains individual-level behavioral measurements obtained from four experimental groups of mice at six months of age:
1. WT without DOX (wild-type mice without doxycycline treatment)
2. WT with DOX (wild-type mice with doxycycline treatment)
3. rTg4510 without DOX (tauopathy model without doxycycline treatment)
4. rTg4510 with DOX (tauopathy model with doxycycline treatment)
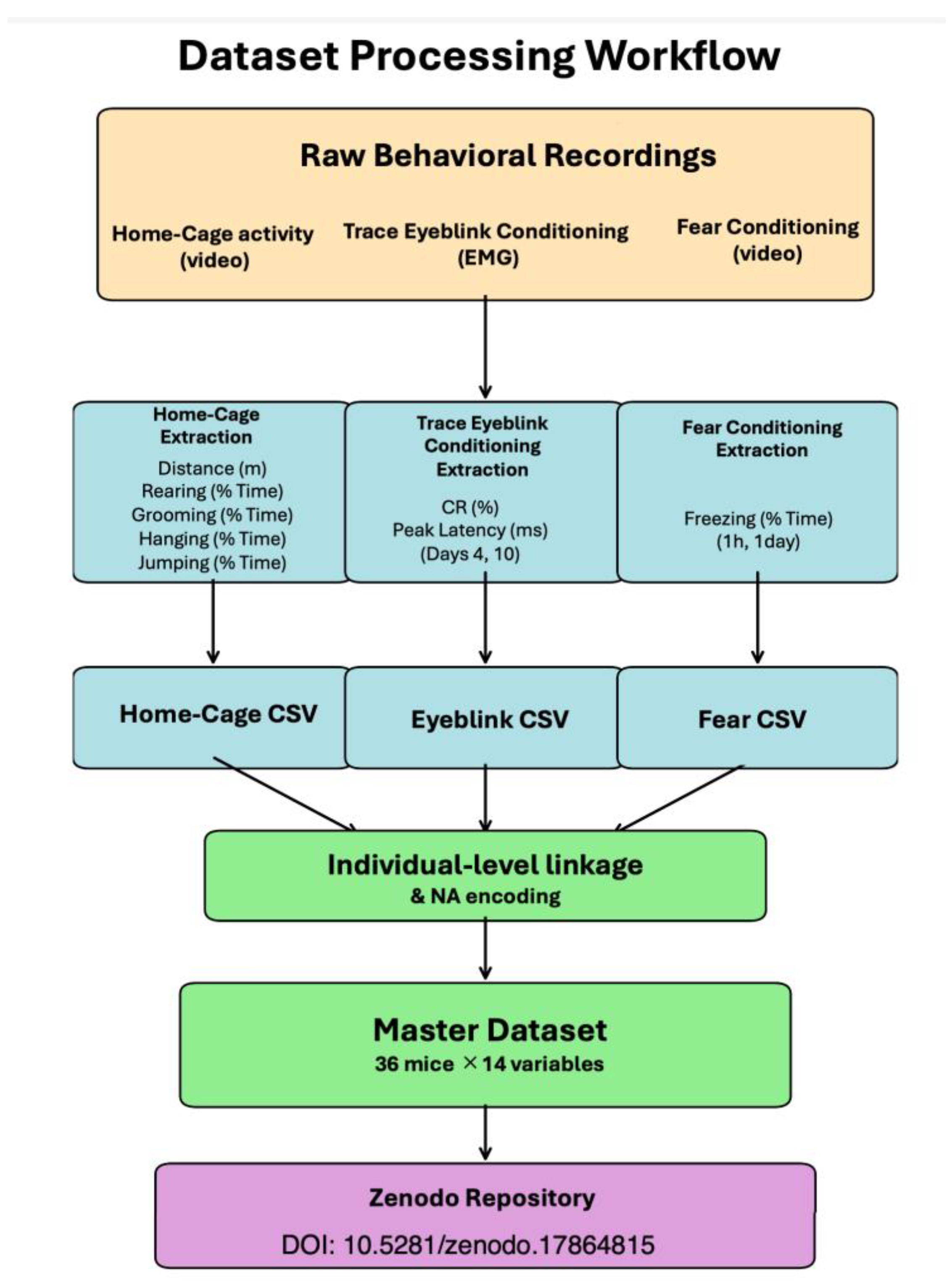
Initial analyses of group-level behavioral outcomes from this cohort have been published . The present data descriptor provides complete individual-level data to enable secondary analyses, cross-task correlation studies, and alternative analytical approaches. The dataset architecture and file organization are illustrated in
Figure 1, which shows the progression from raw behavioral recordings through standardized CSV files to the unified master dataset. This hierarchical structure ensures transparency in data processing and facilitates reuse across different analytical frameworks.
The complete dataset contains 36 mice (n=9 per group). However, two mice have incomplete data across behavioral domains: one WT without DOX mouse lacks eyeblink conditioning data, and one WT with DOX mouse lacks fear conditioning data. For cross-task analyses requiring complete individual-level linkage across all three behavioral modalities, the dataset was restricted to 34 mice with measurements in all domains (n=8, 8, 9, 9 for WT without DOX, WT with DOX, rTg4510 without DOX, and rTg4510 with DOX, respectively). All figures and statistical summaries in this descriptor are based on these 34 mice unless otherwise specified.
This figure illustrates the data transformation pipeline from raw behavioral recordings to the publicly available dataset. Raw data from three independent behavioral paradigms (home-cage activities monitored by video, trace eyeblink conditioning recorded via electromyography, and contextual fear conditioning assessed by video) were extracted into structured CSV files for each modality. Individual-level identifiers were used to link measurements across tasks, and missing values were encoded as NA. The three domain-specific files were integrated into a master dataset containing 36 mice and 14 behavioral variables. The complete dataset is deposited in Zenodo (DOI: 10.5281/zenodo.17864815) under a CC BY 4.0 license. For cross-task analyses requiring complete measurements across all behavioral domains, the dataset can be restricted to 34 mice (n=8, 8, 9, 9 for WT without DOX, WT with DOX, rTg4510 without DOX, and rTg4510 with DOX, respectively).
2.2. Behavioral Modalities
The dataset integrates measurements from three independent behavioral domains:
Spontaneous home-cage activity
One-hour continuous monitoring yielded five behavioral metrics:
• Distance traveled (meters)
• Rearing duration (time ratio)
• Grooming duration (time ratio)
• Hanging duration (time ratio)
• Jumping duration (time ratio)
All home-cage measures were extracted from video recordings and analyzed using HomeCageScan without any additional smoothing or preprocessing.
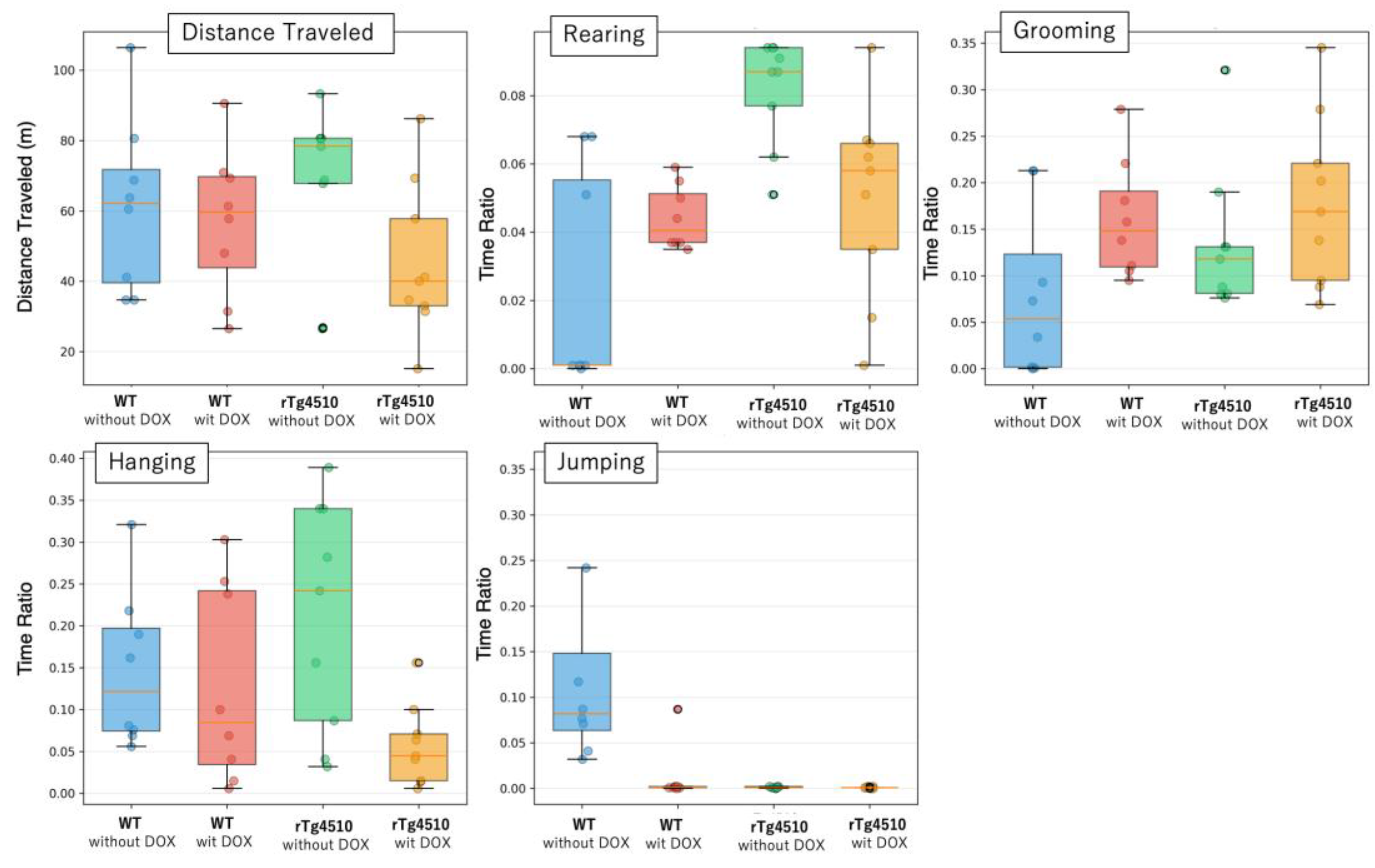
Figure 2 displays the distributions of these five metrics across the four experimental groups, illustrating both central tendencies and within-group variability.
Box plots with overlaid scatter points show raw individual values for five spontaneous behaviors: distance traveled (m), rearing (time ratio), grooming (time ratio), hanging (time ratio), and jumping (time ratio). Each panel displays data from the four experimental groups (WT without DOX, WT with DOX, rTg4510 without DOX, rTg4510 with DOX). Box plots represent the interquartile range with median line; whiskers extend to 1.5 × the IQR. Individual data points are overlaid to show within-group variability. This visualization illustrates group-level central tendencies and individual variability without statistical comparison.
Trace eyeblink conditioning
Animals underwent ten consecutive daily training sessions. For each session, the following variables were recorded:
• Conditioned response percentage (CR%, Days 1–10)
• Peak latency of eyelid closure (milliseconds, extracted for Days 4 and 10)
Peak latency was defined as the temporal delay from conditioned stimulus (CS) onset to the maximum amplitude of the conditioned eyelid response. One WT without DOX mouse lacks eyeblink data due to a technical issue during acquisition; this animal is included in all other measurements.
Contextual fear conditioning
Following a single training session, freezing behavior was assessed at two retention intervals:
• Freezing percentage at 1-hour retention test (Fear_1h)
• Freezing percentage at 1-day retention test (Fear_1day)
Freezing was quantified using an automated motion-threshold algorithm with consistent parameters across all animals. One WT with DOX mouse lacks fear conditioning data; all other variables for this animal are complete.
2.3. Data Structure and File Organization
The deposited dataset consists of the following files:
Master_Dataset.csv
• Wide-format table containing all behavioral variables for every mouse
• Each row represents one individual animal
• Missing values are explicitly encoded as NA
• Includes group labels (Genotype, DOX) and unique animal identifiers (Mouse_ID)
HomeCage_Activity.csv
• Mouse_ID, Genotype, DOX
• Distance, Rearing, Grooming, Hanging, Jumping
TraceEBC_CR.csv
• Mouse_ID, Genotype, DOX
• CR_Day1, CR_Day2, ..., CR_Day10
TraceEBC_Latency.csv
• Mouse_ID, Genotype, DOX
• Latency_Day4, Latency_Day10
FearConditioning.csv
• Mouse_ID, Genotype, DOX
• Fear_1h, Fear_1day
Data_Dictionary.csv
• Machine-readable version of
Table 1
• Variable names, descriptions, units, per-group sample sizes, and notes
All files use UTF-8 encoding and comma-separated value (CSV) format. Column names correspond exactly to the variable names listed in
Table 1.
2.4. Variable Definitions and Data Dictionary
Table 1 provides a complete data dictionary listing all variables, their units, per-group sample sizes, missing value status, and methodological notes. Key variable categories include:
Table 1.
Data Dictionary for the Behavioral Dataset.
Table 1.
Data Dictionary for the Behavioral Dataset.
| Variable |
Description |
Unit |
N (per group) |
Missing |
Notes |
| Group |
Experimental condition: WT without DOX, WT with DOX, rTg4510 without DOX, rTg4510 with DOX |
– |
9/9/9/9 |
None |
Categorical |
| Distance |
Distance traveled in 1-h home-cage monitoring |
meters |
9/9/9/9 |
None |
General locomotor activity |
| Rearing |
Fraction of time spent rearing during home-cage monitoring |
% (time) |
9/9/9/9 |
None |
Vertical exploration |
| Grooming |
Fraction of time spent grooming during home-cage monitoring |
% (time) |
9/9/9/9 |
None |
Self-care behavior |
| Hanging |
Fraction of time spent in hanging posture during home-cage monitoring |
% (time) |
9/9/9/9 |
None |
Cage-top behavior |
| Jumping |
Fraction of time spent jumping during home-cage monitoring |
% (time) |
9/9/9/9 |
None |
Vertical locomotion |
| CR_day4 |
Percentage of conditioned responses on Day 4 of tEBC |
% |
8/9/9/9 |
1 (WT without DOX) |
Early-phase learning |
| CR_day10 |
Percentage of conditioned responses on Day 10 of tEBC |
% |
8/9/9/9 |
1 (WT without DOX) |
Late-phase learning |
Latency_
day4
|
Peak latency of eyelid response on Day 4 |
ms |
8/9/9/9 |
1 (WT without DOX) |
Early temporal processing |
Latency_
day10
|
Peak latency of eyelid response on Day 10 |
ms |
8/9/9/9 |
1 (WT without DOX) |
Late temporal refinement |
| Fear_1h |
Freezing during 1-hour retention test |
% |
9/8/9/9 |
1 (WT with DOX) |
Short-term memory retention |
| Fear_day1 |
Freezing during 1-day retention test |
% |
9/8/9/9 |
1 (WT with DOX) |
Long-term memory retention |
| Mouse_ID |
Individual identifier linking all tasks |
– |
8–9 |
None |
Individual identifier |
| Age |
Age at testing |
months |
6 |
None |
Fixed at 6 months |
Complete list of all variables with descriptions, units, per-group sample sizes (N in order: WT without DOX / WT with DOX / rTg4510 without DOX / rTg4510 with DOX), missing value status, and functional notes. Variables include group identifiers, five home-cage activities measures, eyeblink conditioning metrics at two time points (Days 4 and 10), and contextual fear memory at two retention intervals (1 hour and 1 day). Sample sizes reflect the complete dataset (36 mice total). Missing values: one WT without DOX mouse lacks eyeblink conditioning data; one WT with DOX mouse lacks fear conditioning data. For cross-task analyses requiring complete measurements across all behavioral domains, the dataset can be restricted to 34 mice (n=8, 8, 9, 9 for the four groups, respectively).
Group identifiers:
• Genotype: WT or rTg4510
• DOX: without DOX or with DOX
• Mouse_ID: unique identifier linking all measurements
Home-cage activities:
• Distance: total distance traveled during one-hour monitoring (meters)
• Rearing, Grooming, Hanging, Jumping: proportion of time engaged in each behavior (0–1 scale)
Trace eyeblink conditioning:
• CR_Day1 to CR_Day10: percentage of trials with conditioned responses (0–100%)
• Latency_Day4, Latency_Day10: peak latency from CS onset to maximum eyelid closure (milliseconds)
Contextual fear conditioning:
• Fear_1h: freezing percentage during 1-hour retention test (0–100%)
• Fear_1day: freezing percentage during 1-day retention test (0–100%)
Note: Some automated freezing scores exceed 100% due to detection algorithm sensitivity; these values are retained without normalization to preserve data authenticity.
2.5. Data Distributions and Quality
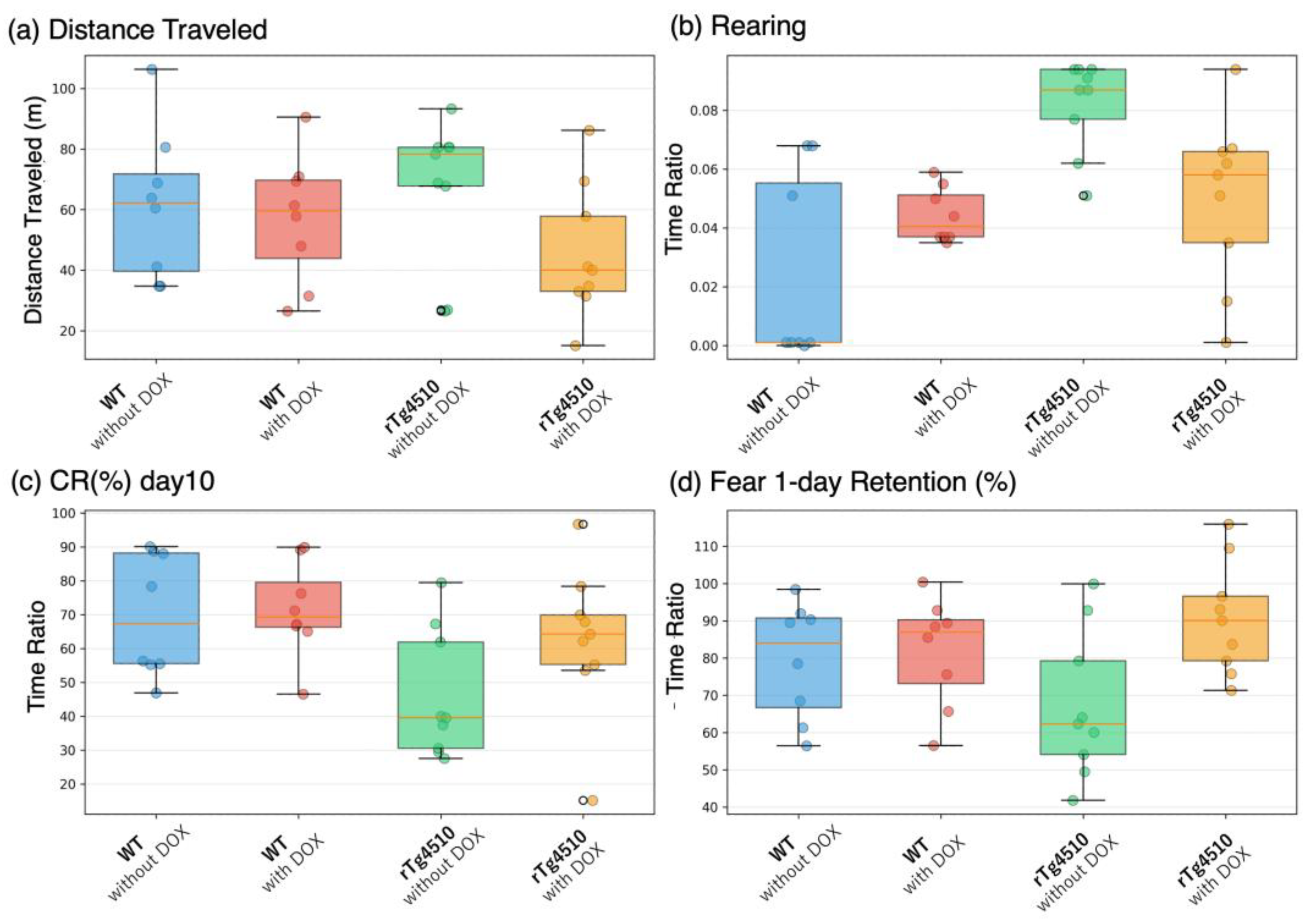
Figure 3 presents the distributions of four primary behavioral variables across the four experimental groups: distance traveled, rearing duration, CR percentage on Day 10 (CR10), and freezing percentage on the 1-day retention test (Fear_1day). Box plots illustrate the range, median, and quartiles for each measure, with individual data points overlaid to show within-group variability. This visualization is provided for descriptive purposes only and does not include inferential statistics.
Box plots show raw individual values for four key behavioral variables: (A) distance traveled in the home cage (meters), (B) rearing time (proportion of recording time), (C) conditioned response percentage on Day 10 of trace eyeblink conditioning (CR10), and (D) freezing percentage during the 1-day retention test in contextual fear conditioning. Each panel displays data from the four experimental groups (WT without DOX, WT with DOX, rTg4510 without DOX, rTg4510 with DOX). Box plots represent the interquartile range with median; whiskers extend to 1.5× the IQR, and individual data points are overlaid. No statistical comparisons are performed; the figure illustrates the shape and distribution of raw behavioral data for each modality.
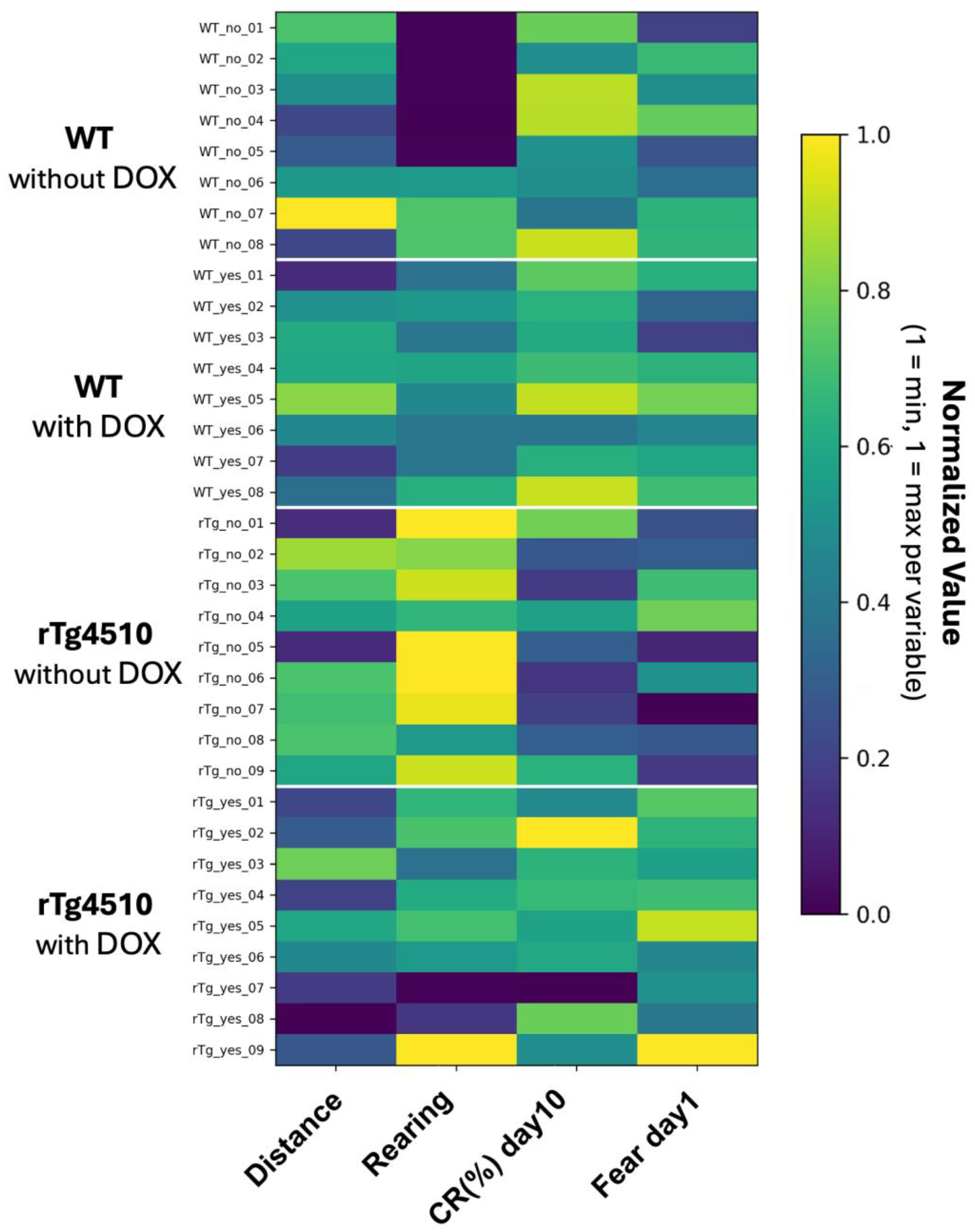
Figure 4 displays a subject × measure heatmap showing raw values for the four primary variables across all individual animals. Rows correspond to animals ordered by experimental group, and columns correspond to behavioral measures. Missing values appear as blank cells. This representation enables rapid inspection of data completeness and individual-level correspondence across behavioral domains.
Each row corresponds to a single mouse, ordered by group (WT without DOX, WT with DOX, rTg4510 without DOX, rTg4510 with DOX). Columns represent the four primary behavioral measures: distance traveled, rearing time, conditioned response percentage on Day 10 (CR10), and freezing percentage during the 1-day retention test. Raw values are visualized without normalization or scaling; color intensity reflects magnitude. Missing data appear as blank entries. This heatmap provides an overview of dataset completeness, within-group and between-group variability, and cross-task linkage at the individual level.
2.6. Missing Data
Missing values are limited to two animals and are explicitly documented:
One WT without DOX mouse: lacks all tEBC data (CR and latency values)
One WT with DOX mouse: lacks contextual fear conditioning data (Fear_1h and Fear_1day)
All other measurements are complete for these animals, and all other animals have complete datasets across all modalities. The transparent encoding of missing values (as NA) allows straightforward implementation of listwise or pairwise deletion strategies during analysis.
2.7. Cross-Task Structure and Validation
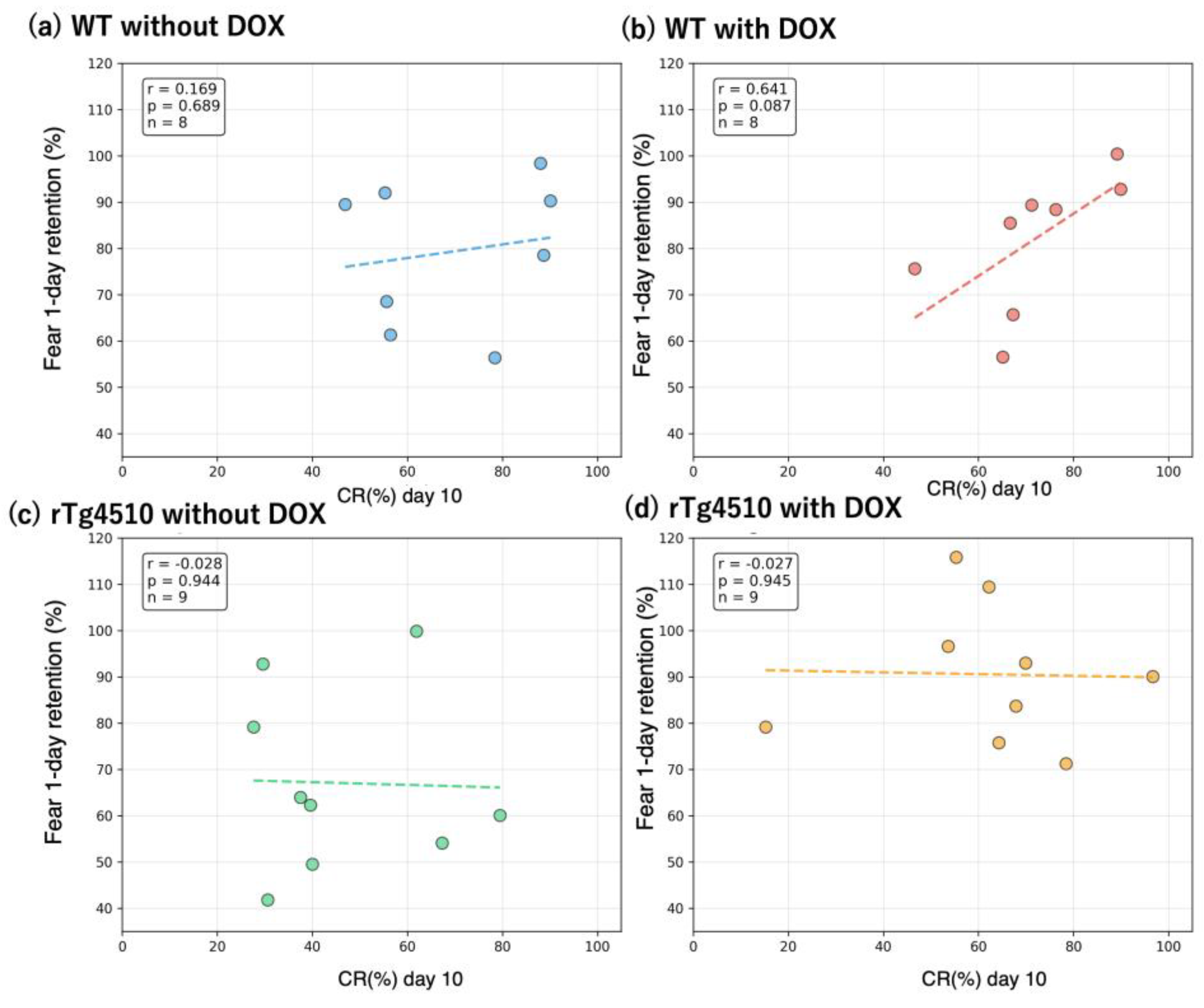
Because all behavioral measurements are linked at the individual level, the dataset enables investigation of cross-task relationships. As an example,
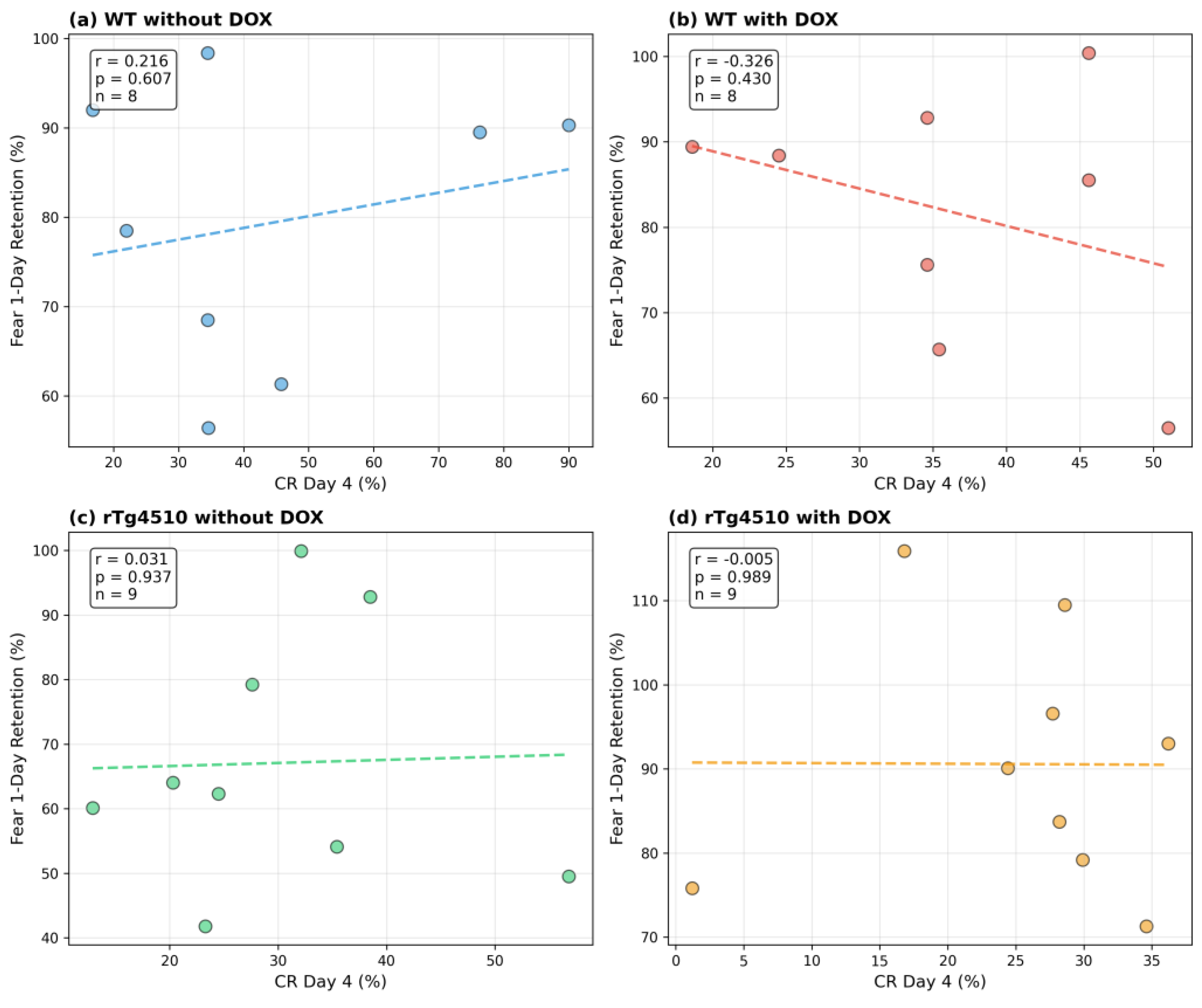
Figure 5 displays group-wise correlations between CR percentage on Day 10 (CR10) and freezing percentage on the 1-day retention test (Fear_1day). The four panels illustrate that the strength and direction of associations between eyeblink conditioning and contextual fear memory differ across genotype and treatment conditions.
Scatterplots show the relationship between conditioned response percentage on Day 10 of trace eyeblink conditioning (CR10, x-axis) and freezing percentage during the 1-day retention test in contextual fear conditioning (y-axis). Each panel represents one experimental group: (a) WT without DOX, (b) WT with DOX, (c) rTg4510 without DOX, and (d) rTg4510 with DOX. Each point represents a single mouse. Pearson correlation coefficients (r) are indicated within each panel after listwise deletion of missing values. No inferential comparisons are performed; this figure illustrates an example of cross-task relationships that can be explored using this dataset.
•
Appendix Figure A1: Correlations between CR percentage on Day 4 and 1-day freezing
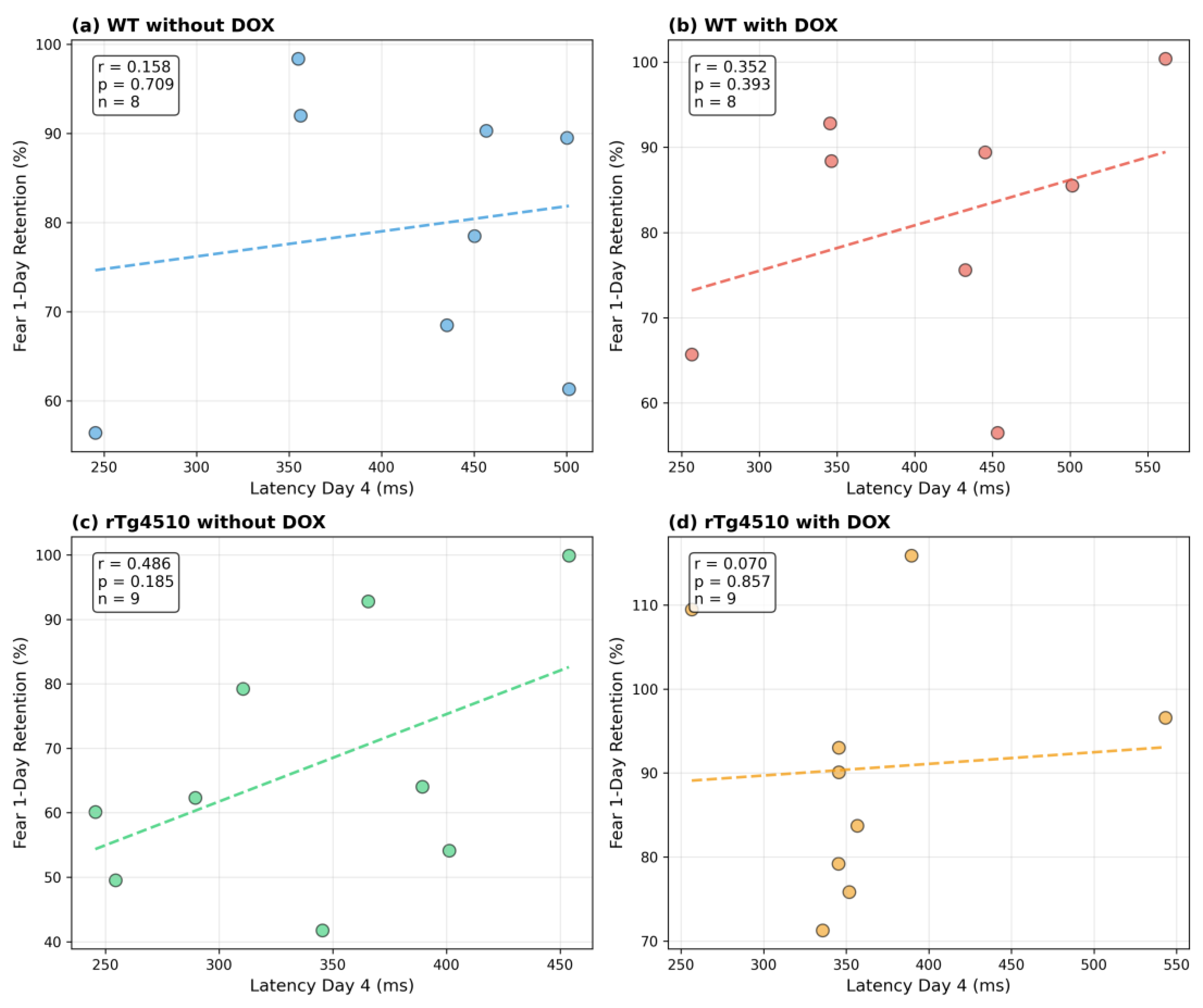
•
Appendix Figure A2: Correlations between peak latency on Day 4 and 1-day freezing
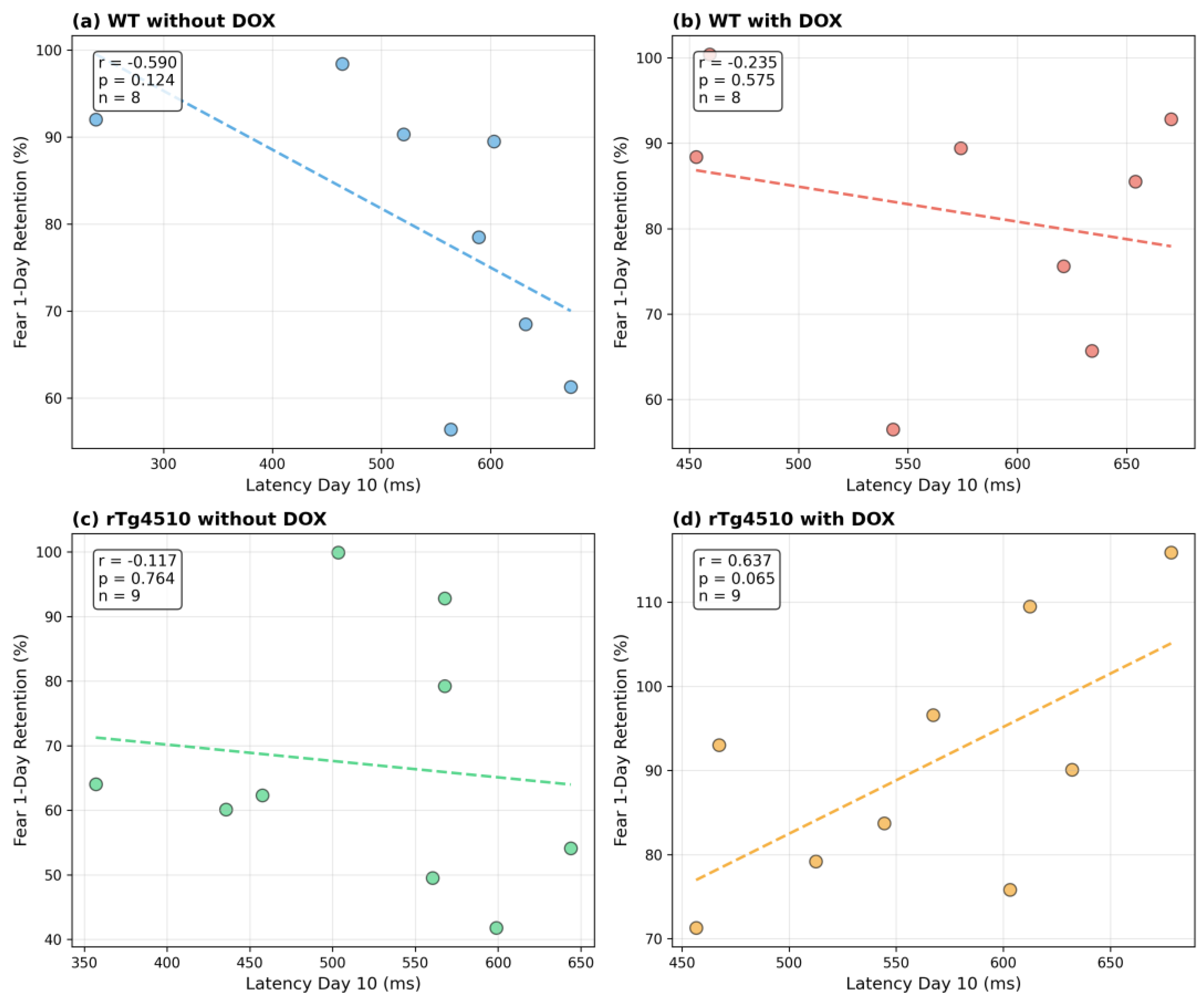
•
Appendix Figure A3: Correlations between peak latency on Day 10 and 1-day freezing
These analyses are included solely to demonstrate the dataset's capacity for cross-paradigm investigation and are not presented as inferential findings. Detailed interpretation appears in
Section 3 (Technical Validation).
2.8. Data Formats and Compatibility
All data are provided in widely compatible CSV format with the following specifications:
• Encoding: UTF-8
• Delimiter: comma (,)
• Decimal separator: period (.)
• Missing values: NA
• Row structure: Each row = one animal
• Column structure: Each column = one variable or one measurement day
This format ensures compatibility with R, Python, MATLAB, Excel, and other statistical or data analysis environments. No proprietary software is required to access or analyze the dataset.
2.9. Reproducibility and Reusability
The dataset is structured to support multiple analytical approaches:
• Cross-task correlation analyses: Individual-level linkage enables examination of relationships between independent learning systems
• Individual difference modeling: Complete behavioral profiles support clustering, classification, and latent variable analyses
• Treatment and genotype comparisons: Four-group design allows examination of DOX effects and genotype-dependent phenotypes
• Temporal dynamics: Daily CR measurements and dual retention timepoints enable investigation of learning trajectories
• Machine learning applications: Multivariate structure supports supervised and unsupervised learning algorithms
All measurements are provided in their original, unprocessed form without smoothing, normalization, or imputation. This approach maximizes transparency and allows researchers to apply their own preprocessing pipelines according to specific analytical needs.
3. Methods
3.1. Animals and Experimental Design
Six-month-old male and female rTg4510 mice and wild-type (WT) mice were used in this study. The rTg4510 line expresses human P301L mutant tau under control of a tetracycline-responsive promoter, enabling doxycycline (DOX)-mediated suppression of transgenic tau expression. Animals were assigned to four experimental groups based on genotype and DOX treatment:
1. WT without DOX (n = 9)
2. WT with DOX (n = 9)
3. rTg4510 without DOX (n = 9)
4. rTg4510 with DOX (n = 9)
Mice in DOX-treated groups received doxycycline-containing chow (200 mg/kg) ad libitum from 4 to 6 months of age. All animals were housed under a 12:12 h light–dark cycle at 22–24°C with food and water available ad libitum. Behavioral testing was conducted in the following sequence to minimize task interference: (1) home-cage activity monitoring, (2) tEBC, and (3) contextual fear conditioning. All procedures were approved by the Institutional Animal Care and Use Committee of Teikyo University and conducted in accordance with institutional and national guidelines for animal research.
3.2. Home-Cage Activity Monitoring
Spontaneous home-cage behavior was recorded for one hour using a standard overhead video camera, and all recordings were saved as MPEG video files. Behavioral categories were quantified using HomeCageScan (CleverSys Inc.), which automatically classifies behaviors from video recordings [
13,
14,
15]. All metrics (distance traveled, rearing, grooming, hanging, and jumping) represent the direct output of HomeCageScan without any additional smoothing or preprocessing applied by the experimenters.
Five behavioral metrics were quantified:
• Distance traveled: Total distance (meters) computed from centroid displacement across video frames
• Rearing: Proportion of time spent in vertical exploration posture
• Grooming: Proportion of time engaged in self-grooming behavior
• Hanging: Proportion of time in cage-hanging posture
• Jumping: Proportion of time engaged in jumping movements
All proportional metrics are expressed as time ratios (0–1 scale). Missing values due to video occlusion or detection uncertainty were retained as NA. No smoothing, imputation, or normalization was applied to preserve data authenticity.
3.3. Trace Eyeblink Conditioning
Trace eyeblink conditioning (tEBC) was conducted over 10 consecutive days using a well-established trace paradigm [
3,
4].
Conditioning protocol:
• Conditioned stimulus (CS): 250 ms tone (1 kHz, 80 dB)
• Trace interval: 500 ms stimulus-free period
• Unconditioned stimulus (US): 100 ms corneal electrical shock (0.2 mA, 100 Hz, square pulses)
• Intertrial interval: 20–40 s (variable)
• Trials per session: 100 trials
•Training duration: 10 consecutive days (Days 1–10)
Response detection:
A conditioned response (CR) was defined as an eyelid closure exceeding a fixed amplitude threshold during the trace interval (between CS offset and US onset). Detection thresholds and filtering parameters were identical across all animals to ensure consistency.
Extracted variables:
• CR percentage (CR%): Proportion of trials with CRs for each training day (Days 1–10)
• CR peak latency: Time (milliseconds) from CS onset to maximum eyelid deflection during CR trials, extracted for Days 4 and 10
One WT without DOX mouse lacked reliable eyelid recordings due to technical failure during acquisition; all values for this animal are encoded as NA in the t tEBC dataset.
3.4. Contextual Fear Conditioning
Contextual fear conditioning was performed in a standard conditioning chamber (30 × 30 × 35 cm) equipped with a stainless steel grid floor and physical movement tracking system (FreezeScan).
Training protocol:
• Habituation: 2 min exploration period
• Footshock: Single 2 s shock at 0.7 mA
• Post-shock period: 30 s before removal from chamber
Retention testing:
Freezing behavior was assessed at two time points:
• 1-hour retention test: 2 min exposure to the same context
• 1-day retention test: 2 min exposure to the same context
Freezing was quantified automatically using a motion-threshold detection algorithm (FreezeScan) with consistent parameters across all animals. Freezing percentage was calculated as the proportion of time spent in a freezing posture (absence of movement except respiration) during the test period.
Extracted variables:
• Fear_1h: Freezing percentage during 1-hour retention test
• Fear_1day: Freezing percentage during 1-day retention test
One WT with DOX mouse lacks contextual fear conditioning data due to equipment malfunction; values for this animal are encoded as NA.
3.5. Data Integration and Organization
All behavioral measurements were linked at the individual animal level using unique mouse identifiers (Mouse_ID). This structure enables direct cross-task analyses without requiring data alignment or matching procedures.
File organization:
The dataset is provided as multiple CSV files to facilitate selective loading and analysis:
Master_Dataset.csv: Complete wide-format table with all variables
HomeCage_Activity.csv: Distance traveled and behavioral time ratios
TraceEBC_CR.csv: CR percentages for Days 1–10
TraceEBC_Latency.csv: Peak latency values for Days 4 and 10
FearConditioning.csv: Freezing percentages at 1 h and 1 day
Data_Dictionary.csv: Machine-readable variable reference corresponding to
Table 1
Data processing:
No preprocessing, smoothing, normalization, or imputation was applied to any behavioral measurements. All values represent direct outputs from acquisition systems or automated detection pipelines. Missing values are explicitly encoded as NA and documented in the Data Description section.
File format specifications:
Encoding: UTF-8
Delimiter: Comma (,)
Decimal separator: Period (.)
Missing values: NA
Row structure: Each row represents one individual animal
Column structure: Each column represents one variable or one measurement day
3.6. Statistical Analyses for Technical Validation
Cross-task correlation analyses were performed to validate internal consistency and demonstrate the dataset's capacity for cross-paradigm investigation. Pearson correlation coefficients were computed between eyeblink conditioning metrics (CR percentage, peak latency) and contextual fear memory (1-day freezing percentage) within each experimental group.
Analyses were conducted separately for:
CR percentage on Day 4 vs. Fear_1day (Appendix
Figure A1)
Peak latency on Day 4 vs. Fear_1day (Appendix
Figure A2)
Peak latency on Day 10 vs. Fear_1day (Appendix
Figure A3)
CR percentage on Day 10 vs. Fear_1day (
Figure 5)
Missing values were handled by listwise deletion on a per-analysis basis. Correlation analyses are presented solely to demonstrate data quality and cross-task relationships; they are not intended as inferential conclusions about mechanisms or treatment effects. All visualization and analysis code is available in the data repository.
3.7. Software and Equipment
Home-cage monitoring: Video acquisition and automated behavioral classification using [HomeCageScan system (CleverSys, Inc., Reston, VA]
Trace eyeblink conditioning: Trace eyeblink conditioning was performed using a fully custom-built EMG-based acquisition and stimulus-control system. Eyelid muscle activity was recorded using fine-wire EMG electrodes and amplified with a commercial differential amplifier. The amplified EMG signals were digitized via an A/D converter operating under a DOS/V environment. All stimulus timing, including tone CS presentation, trace-interval control, and periorbital electrical US delivery, was executed by in-house software running on a Microsoft Windows 3.1–based system. Conditioned responses (CRs) were defined as increases in EMG activity exceeding a fixed threshold within the trace interval. Detailed procedures for EMG recording, filtering, and CR detection have been described previously in our earlier report [
3,
4].
Fear conditioning: Standard conditioning chamber with automated freezing detection using FreezeScan TM1.0 system (Clever Sys, Inc., Reston, VA).
Data processing: Raw behavioral outputs from each system (HomeCageScan, EMG-based tEBC system, and contextual fear conditioning software) were manually exported and organized into CSV files. No additional preprocessing, smoothing, or normalization was applied
Statistical analysis: Technical validation analyses were performed by computing Pearson correlation coefficients between eyeblink conditioning measures and contextual fear memory within each experimental group. All correlation analyses (r values) and descriptive statistics were conducted using GraphPad Prism (GraphPad Software, San Diego, CA).
Visualization: Initial plots (e.g., scatterplots and summary graphs) were generated using GraphPad Prism. Final figure layouts, labels, and graphical adjustments were completed manually using Microsoft PowerPoint. No smoothing, transformation, or additional data preprocessing was applied during figure preparation.