Submitted:
11 December 2025
Posted:
12 December 2025
You are already at the latest version
Abstract
Bacteriophages, traditionally viewed solely as antibacterial agents, are increasingly being studied for their immunomodulatory properties. In this study, we demonstrate that PM16 phage therapy not only effectively controls subcutaneous Proteus mirabilis infection in mice, but also induces long-term specific humoral immunity against subsequent reinfection. This immunomodulatory effect was dose-dependent. In vitro, PM16 directly activates macrophages, leading to increased production of proinflammatory cytokines (tumor necrosis factor-α and interleukin-1β) and inducible nitric oxide synthase, and enhances macrophage bactericidal activity against P. mirabilis. We assume that the enhancement of the adaptive immune response is mediated not by the phage acting as a classical antigenic adjuvant, but by its ability to prime innate immune cells, specifically macrophages. This priming leads to more efficient bacterial clearance, antigen presentation, and the formation of protective immunological memory.
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. P. mirabilis and its Podophage PM16
2.3. Limulus Amebocyte Lysate Assay
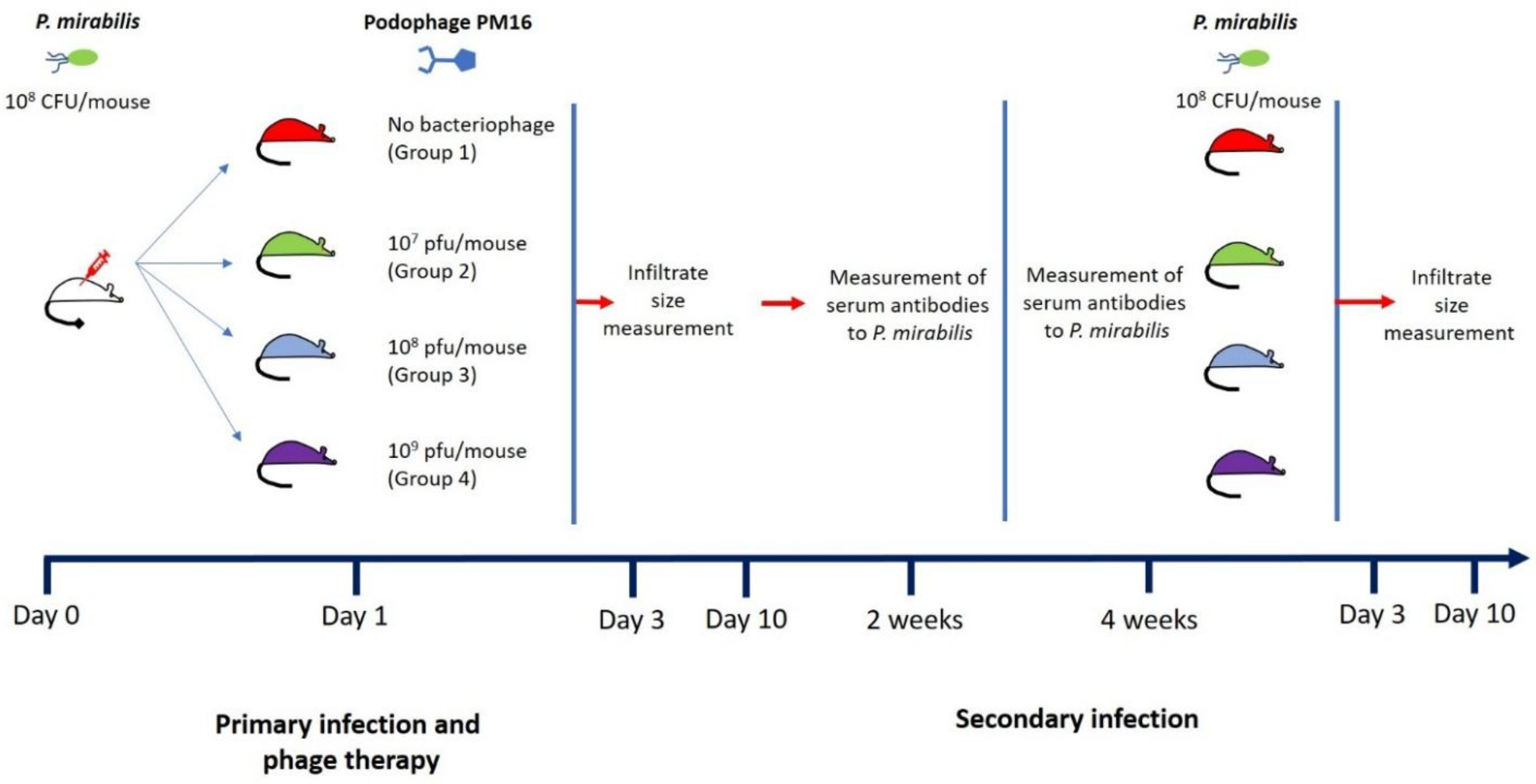
2.4. Subcutaneous Infection Model
2.5. Statistics
2.6. Assessment of Serum IgG to P. mirabilis Infection
2.7. Preparation of Stimulated Macrophages
2.8. Real-Time PCR
2.9. Microscopy and Bacterial Enumeration
2.10. Measurement of Nitric Oxide Production
2.11. Analysis of Bacterial Survival Within Macrophages
3. Results
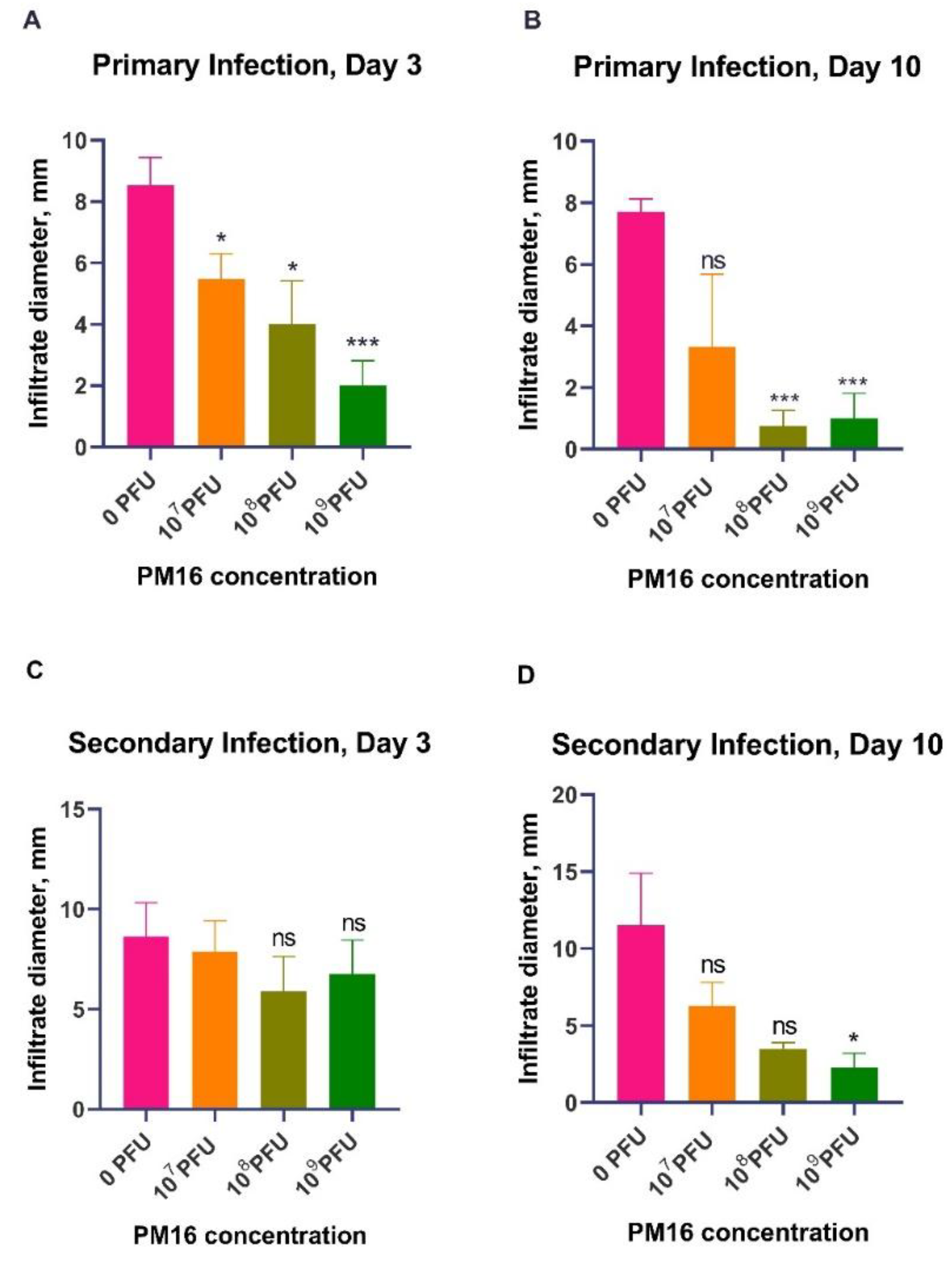
3.1. PM16 Phage Therapy Attenuates P. mirabilis Infection and Induces Long-Term Protective Immunity
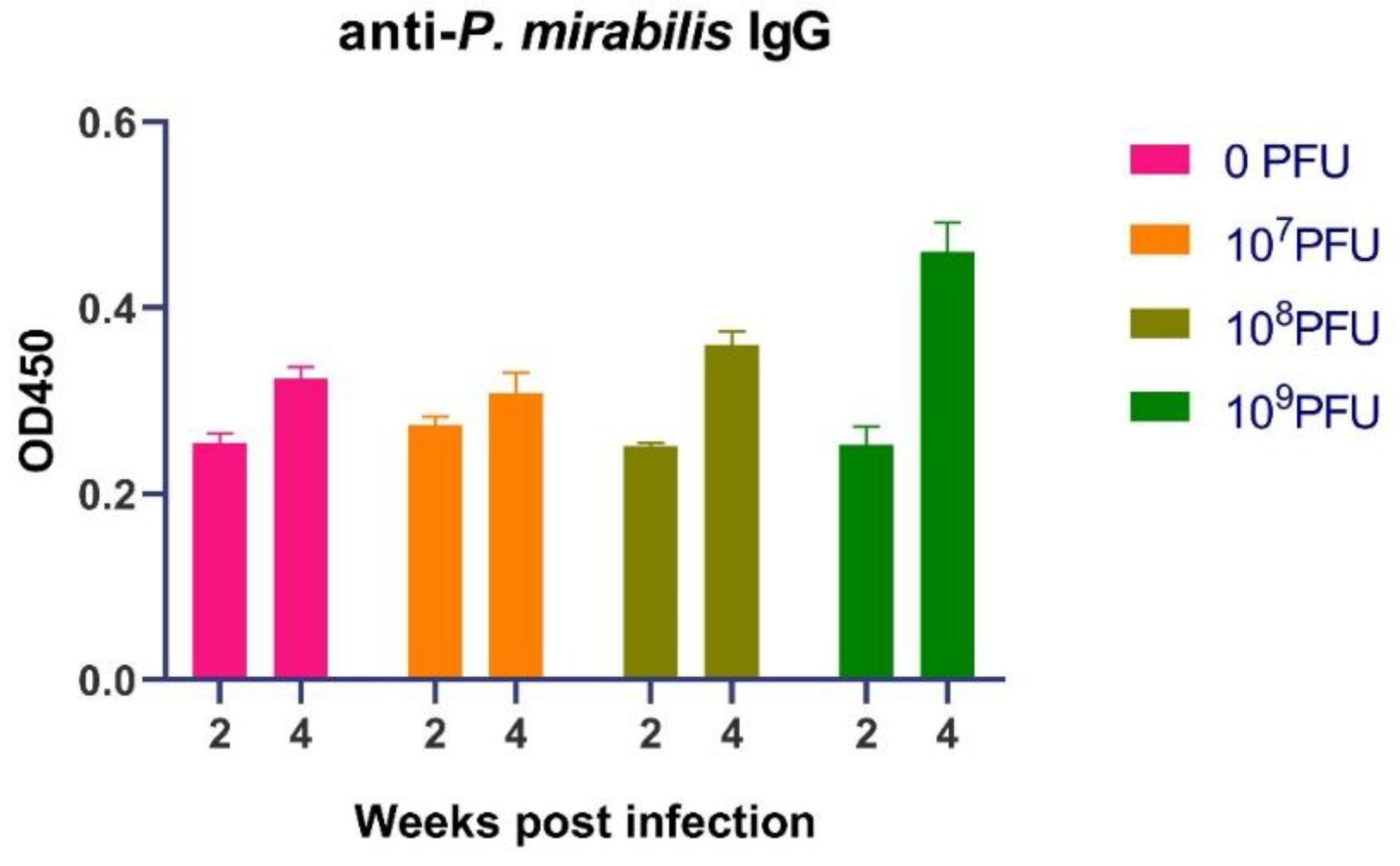
3.2. PM16 Phage Therapy Potentiates a Robust and Sustained Humoral Immune Response against P. mirabilis
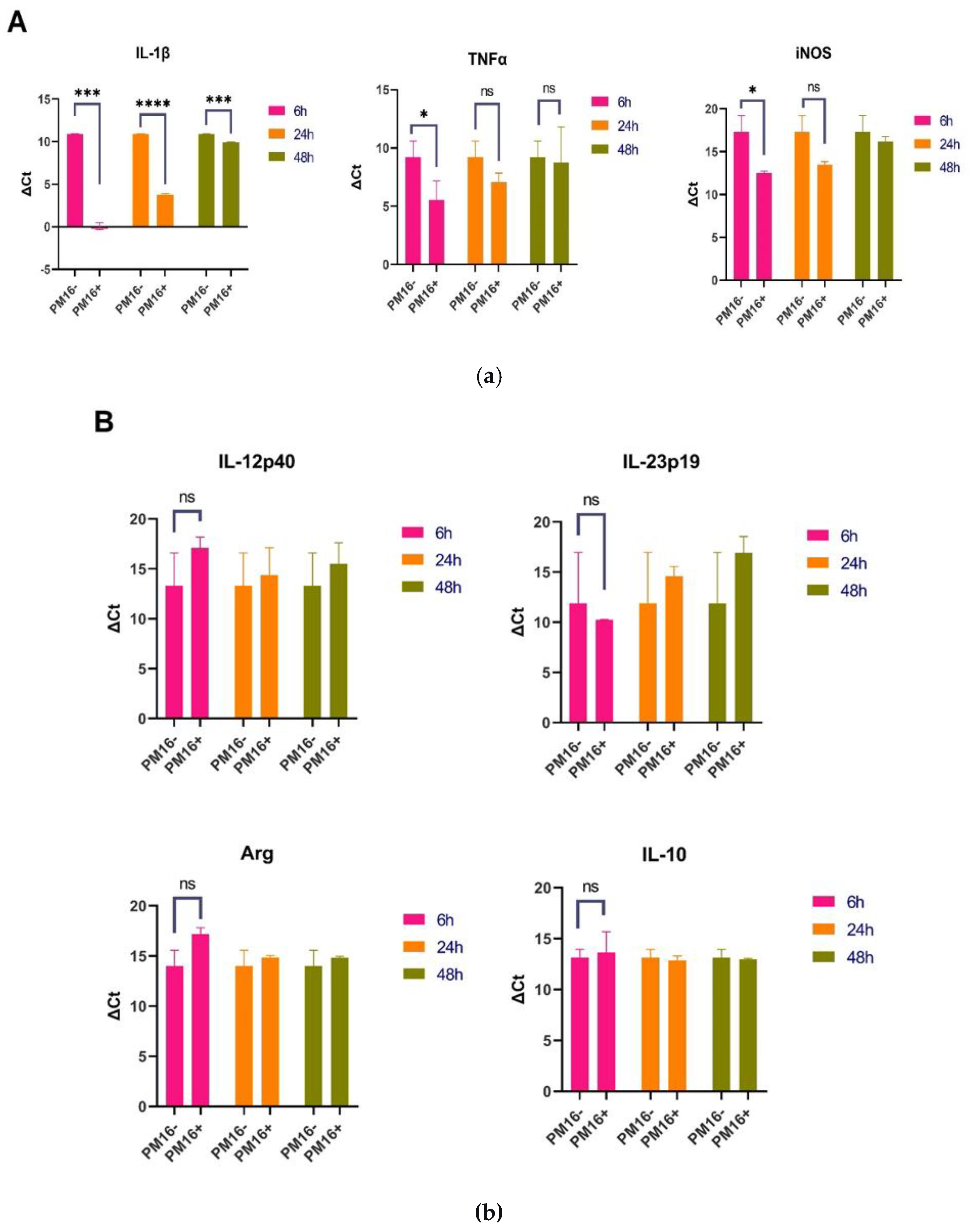
3.3. PM16 Phage Activates Macrophages, Inducing Proinflammatory Cytokines and iNOS
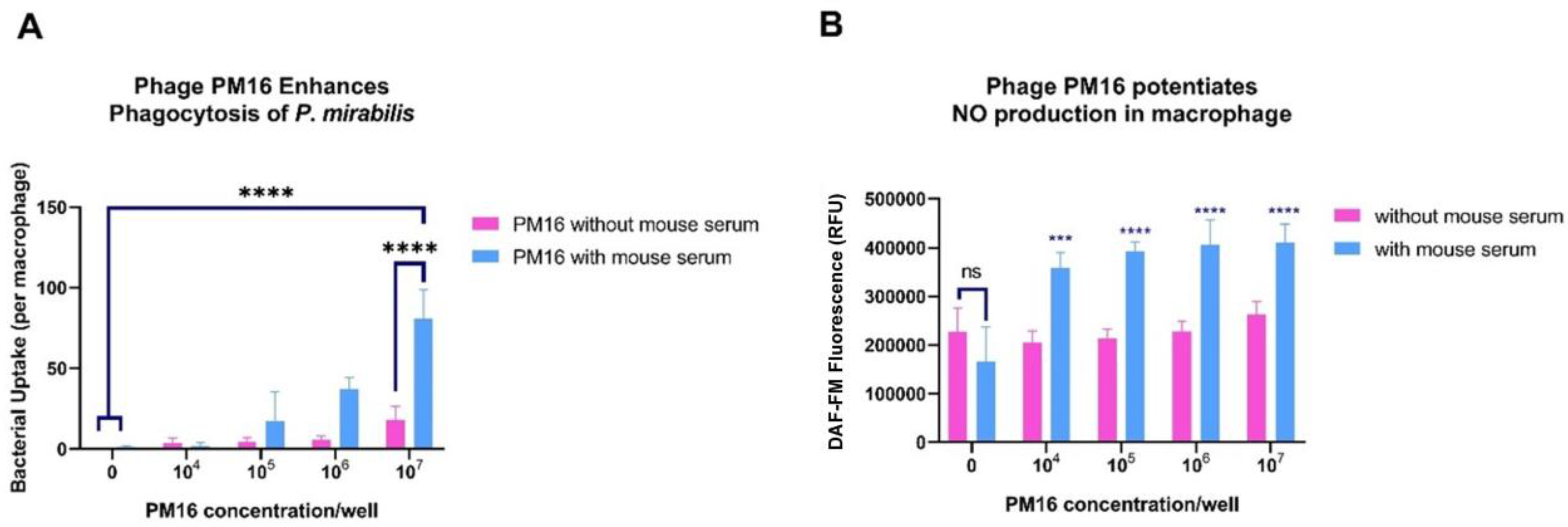
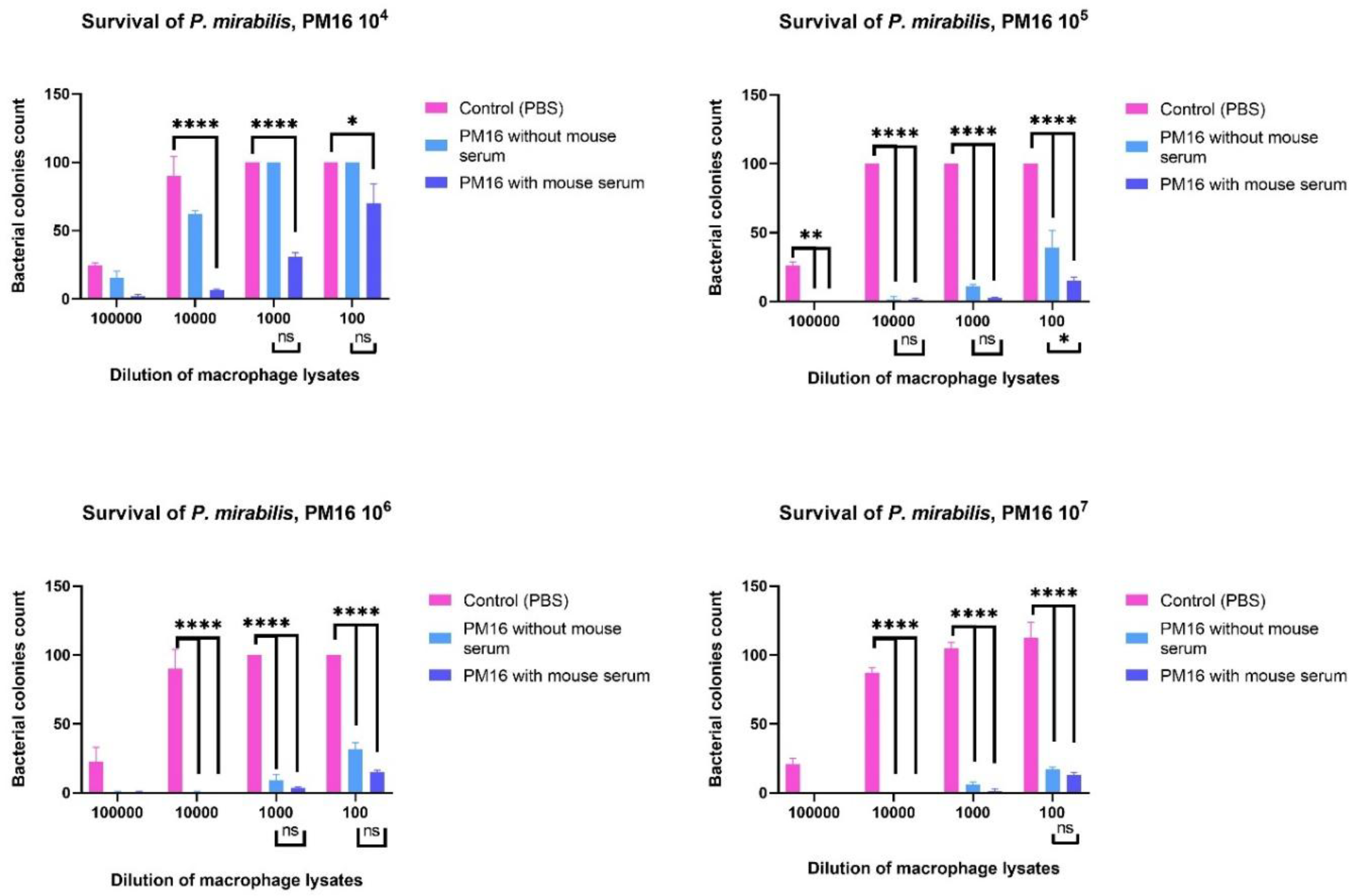
3.4. PM16 Enhances the Bactericidal Activity of Macrophages against P. mirabilis
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Schaffer, J.N.; Pearson, M.M. Proteus Mirabilis and Urinary Tract Infections. Microbiol Spectr 2015, 3. [CrossRef]
- Complicated Catheter-Associated Urinary Tract Infections Due to Escherichia Coli and Proteus Mirabilis | Clinical Microbiology Reviews Available online: https://journals.asm.org/doi/10.1128/cmr.00019-07 (accessed on 4 December 2025).
- Stock, I. Natural Antibiotic Susceptibility of Proteus Spp., with Special Reference to P. Mirabilis and P. Penneri Strains. J Chemother 2003, 15, 12–26. [CrossRef]
- Kynshi, M.A.L., Kharkamni, E., Borah, V.V. Proteus mirabilis: Insights into biofilm formation, virulence mechanisms, and novel therapeutic strategies. The Microbe 2025, 8, 100450. [CrossRef]
- Wang, Y., Yu, Y. Phage therapy as a revitalized weapon for treating clinical diseases. Microbiome Res Rep 2025, 4, 35. [CrossRef]
- Youssef, R.A., Sakr, M., Shebl, R.I., Aboshanab, K.M. Recent insights on challenges encountered with phage therapy against gastrointestinal-associated infections. Gut Pathogens 2026, 17, 60. [CrossRef]
- Levin, B.R.; Bull, J.J. Population and Evolutionary Dynamics of Phage Therapy. Nat Rev Microbiol 2004, 2, 166–173. [CrossRef]
- Marchi, J.; Zborowsky, S.; Debarbieux, L.; Weitz, J.S. The Dynamic Interplay of Bacteriophage, Bacteria and the Mammalian Host during Phage Therapy. iScience 2023, 26, 106004. [CrossRef]
- Weissfuss, C., Li, J., Behrendt, U., Hoffmann, K., Bürkle, M., Tan, C., Krishnamoorthy, G., Korf, I., Rohde, C., Gaborieau, B., Debarbieux, L., Ricard, J-D., Witzenrath, M., Felten, M., Nouailles, G. Adjunctive phage therapy improves antibiotic treatment of ventilator-associated-pneumonia with Pseudomonas aeruginosa. Nature Communications 2025, 16, 4500. [CrossRef]
- Górski, A., Dąbrowska, K., Międzybrodzki, R., Weber-Dąbrowska, B., Łusiak-Szelachowska, M., Jończyk-Matysiak, E., Borysowski, J. Phages and immunomodulation. Future Microbiol 2017, 12, 905-914. [CrossRef]
- Górski, A., Międzybrodzki, R., Łobocka, L., Głowacka-Rutkowska, A., Bednarek, A., Borysowski, J., Jończyk-Matysiak, E., Łusiak-Szelachowska, M., Weber-Dąbrowska, B., Bagińska, N., Letkiewicz, S., Dąbrowska, K., Scheres, J. Phage Therapy: What Have We Learned? Viruses 2018, 10, 288. [CrossRef]
- Van Belleghem, J.; Dąbrowska, K.; Vaneechoutte, M.; Barr, J.; Bollyky, P. Interactions between Bacteriophage, Bacteria, and the Mammalian Immune System. Viruses 2018, 11, 10. [CrossRef]
- Jończyk-Matysiak, E.; Weber-Dąbrowska, B.; Owczarek, B.; Międzybrodzki, R.; Łusi-ak-Szelachowska, M.; Łodej, N.; Górski, A. Phage-Phagocyte Interactions and Their Implications for Phage Application as Therapeutics. Viruses 2017, 9, 150. [CrossRef]
- Podlacha, M.; Grabowski, Ł.; Kosznik-Kawśnicka, K.; Zdrojewska, K.; Stasiłojć, M.; Węgrzyn, G.; Węgrzyn, A. Interactions of Bacteriophages with Animal and Human Organisms—Safety Issues in the Light of Phage Therapy. Int J Mol Sci 2021, 22, 8937. [CrossRef]
- Schaffer, J.N.; Pearson, M.M. Proteus Mirabilis and Urinary Tract Infections. Microbiol Spectr 2015, 3. [CrossRef]
- Complicated Catheter-Associated Urinary Tract Infections Due to Escherichia Coli and Proteus Mirabilis | Clinical Microbiology Reviews Available online: https://journals.asm.org/doi/10.1128/cmr.00019-07 (accessed on 4 December 2025).
- Stock, I. Natural Antibiotic Susceptibility of Proteus Spp., with Special Reference to P. Mirabilis and P. Penneri Strains. J Chemother 2003, 15, 12–26. [CrossRef]
- Chechushkov, A.; Kozlova, Y.; Baykov, I.; Morozova, V.; Kravchuk, B.; Ushakova, T.; Bardasheva, A.; Zelentsova, E.; Allaf, L.A.; Tikunov, A.; et al. Influence of Caudovirales Phages on Humoral Immunity in Mice. Viruses 2021, 13, 1241. [CrossRef]
- Morozova, V.; Kozlova, Yu.; Shedko, E.; Kurilshikov, A.; Babkin, I.; Tupikin, A.; Yunusova, A.; Chernonosov, A.; Baykov, I.; Кondratov, I.; et al. Lytic Bacteriophage PM16 Specific for Proteus Mirabilis: A Novel Member of the Genus Phikmvvirus. Arch Virol 2016, 161, 2457–2472. [CrossRef]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual; 3rd ed.; Cold Spring Harbor Laboratory Press: Cold Spring Harbor, N.Y, 2001; Vol. 1; ISBN 978-0-87969-577-4.
- Popescu, M.; Van Belleghem, J.D.; Khosravi, A.; Bollyky, P.L. Bacteriophages and the Immune System. Annu. Rev. Virol. 2021, 8, annurev-virology-091919-074551. [CrossRef]
- Jończyk-Matysiak, E.; Łusiak-Szelachowska, M.; Kłak, M.; Bubak, B.; Międzybrodzki, R.; Weber-Dąbrowska, B.; Żaczek, M.; Fortuna, W.; Rogóż, P.; Letkiewicz, S.; et al. The Effect of Bacteriophage Preparations on Intracellular Killing of Bacteria by Phagocytes. J Immunol Res 2015, 2015, 482863. [CrossRef]
- Górski, A.; Międzybrodzki, R.; Borysowski, J.; Dąbrowska, K.; Wierzbicki, P.; Ohams, M.; Korczak-Kowalska, G.; Olszowska-Zaremba, N.; Łusiak-Szelachowska, M.; Kłak, M.; et al. Phage as a Modulator of Immune Responses: Practical Implications for Phage Therapy. Adv Virus Res 2012, 83, 41–71. [CrossRef]
- Międzybrodzki, R.; Borysowski, J.; Kłak, M.; Jończyk-Matysiak, E.; Obmińska-Mrukowicz, B.; Suszko-Pawłowska, A.; Bubak, B.; Weber-Dąbrowska, B.; Górski, A. In Vivo Studies on the Influence of Bacteriophage Preparations on the Autoimmune Inflammatory Process. Biomed Res Int 2017, 2017, 3612015. [CrossRef]
- Bacteriophage Preparation Inhibition of Reactive Oxygen Species Generation by Endotoxin-Stimulated Polymorphonuclear Leukocytes - PubMed Available online: https://pubmed.ncbi.nlm.nih.gov/17996972/ (accessed on 2 December 2025).
- T4 Phage Tail Adhesin Gp12 Counteracts LPS-Induced Inflammation In Vivo - Pub-Med Available online: https://pubmed.ncbi.nlm.nih.gov/27471503/ (accessed on 2 December 2025).
- Pro- and Anti-Inflammatory Responses of Peripheral Blood Mononuclear Cells Induced by Staphylococcus Aureus and Pseudomonas Aeruginosa Phages | Scientific Re-ports Available online: https://www.nature.com/articles/s41598-017-08336-9 (accessed on 2 December 2025).
- Roach, D.R.; Leung, C.Y.; Henry, M.; Morello, E.; Singh, D.; Di Santo, J.P.; Weitz, J.S.; Debarbieux, L. Synergy between the Host Immune System and Bacteriophage Is Essential for Successful Phage Therapy against an Acute Respiratory Pathogen. Cell Host Microbe 2017, 22, 38-47.e4. [CrossRef]
| Cytokine | Primer set | Tm |
| Murine IL-1b | Forward: 5’ TGCCACCTTTTGACAGTGATG 3’ Reverse: 5’ TGATGTGCTGCTGCGAGATT 3’ |
60 |
| Murine IL12p40 | Forward: 5’ GGAGGGGTGTAACCAGAAAGG 3’ Reverse: 5’ TAGCGATCCTGAGCTTGCAC 3’ |
59 |
| Murine IL23 | Forward: 5’ ACCAGCGGGACATATGAATCT 3’ Reverse: 5’ AGACCTTGGCGGATCCTTTG 3’ |
59 |
| Murine inducible NO-synthase | Forward: 5’ GCTCCCTATCTTGAAGCCCC 3’ Reverse: 5’ TGGAAGCCACTGACACTTCG 3’ |
58 |
| Murine IL10 | Forward: 5’ GTAGAAGTGATGCCCCAGGC 3’ Reverse: 5’ GACACCTTGGTCTTGGAGCTTATT 3’ |
60 |
| Murine Arginase 1 | Forward: 5’ TTTCTCAAAAGGACAGCCTCG 3’ Reverse: 5’ CAGACCGTGGGTTCTTCACA 3’ |
58 |
| Murine IL4 | Forward: 5’ TCACAGCAACGAAGAACACCA 3’ Reverse: 5’ CAGGCATCGAAAAGCCCGAA 3’ |
58 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




