Submitted:
11 December 2025
Posted:
14 December 2025
You are already at the latest version
Abstract
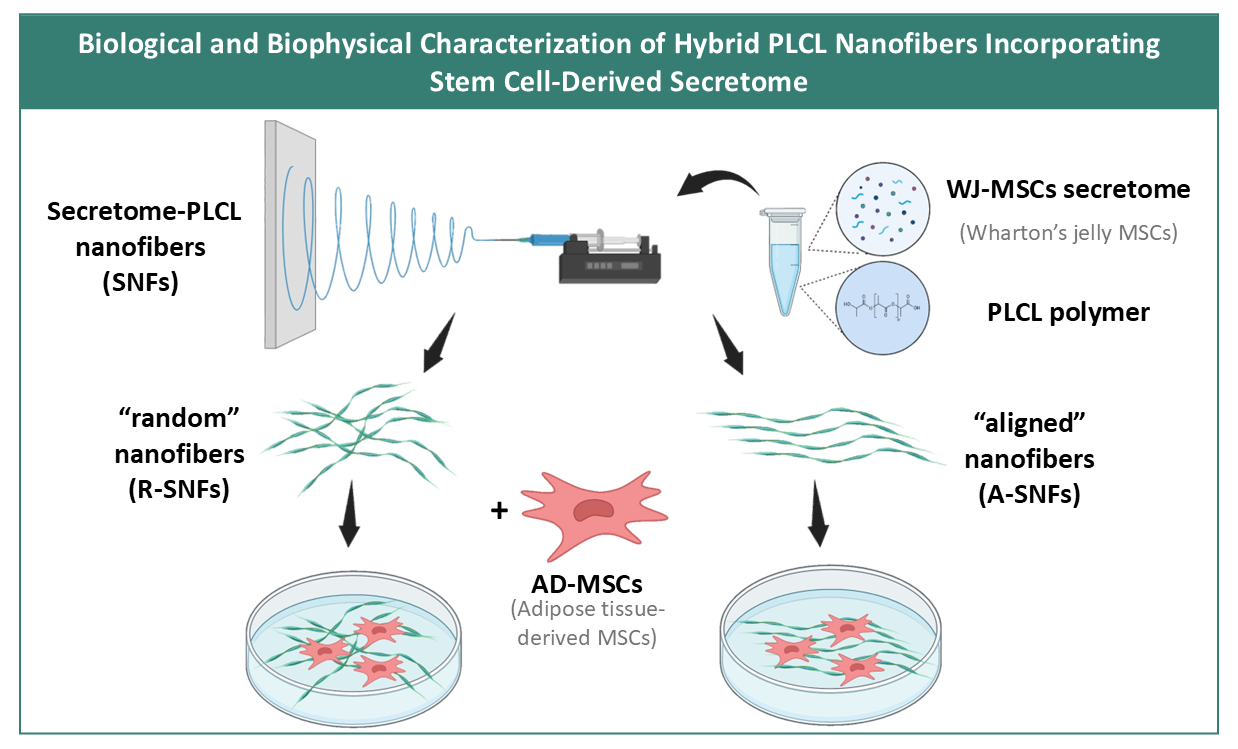
Keywords:
1. Introduction
2. Materials and Methods
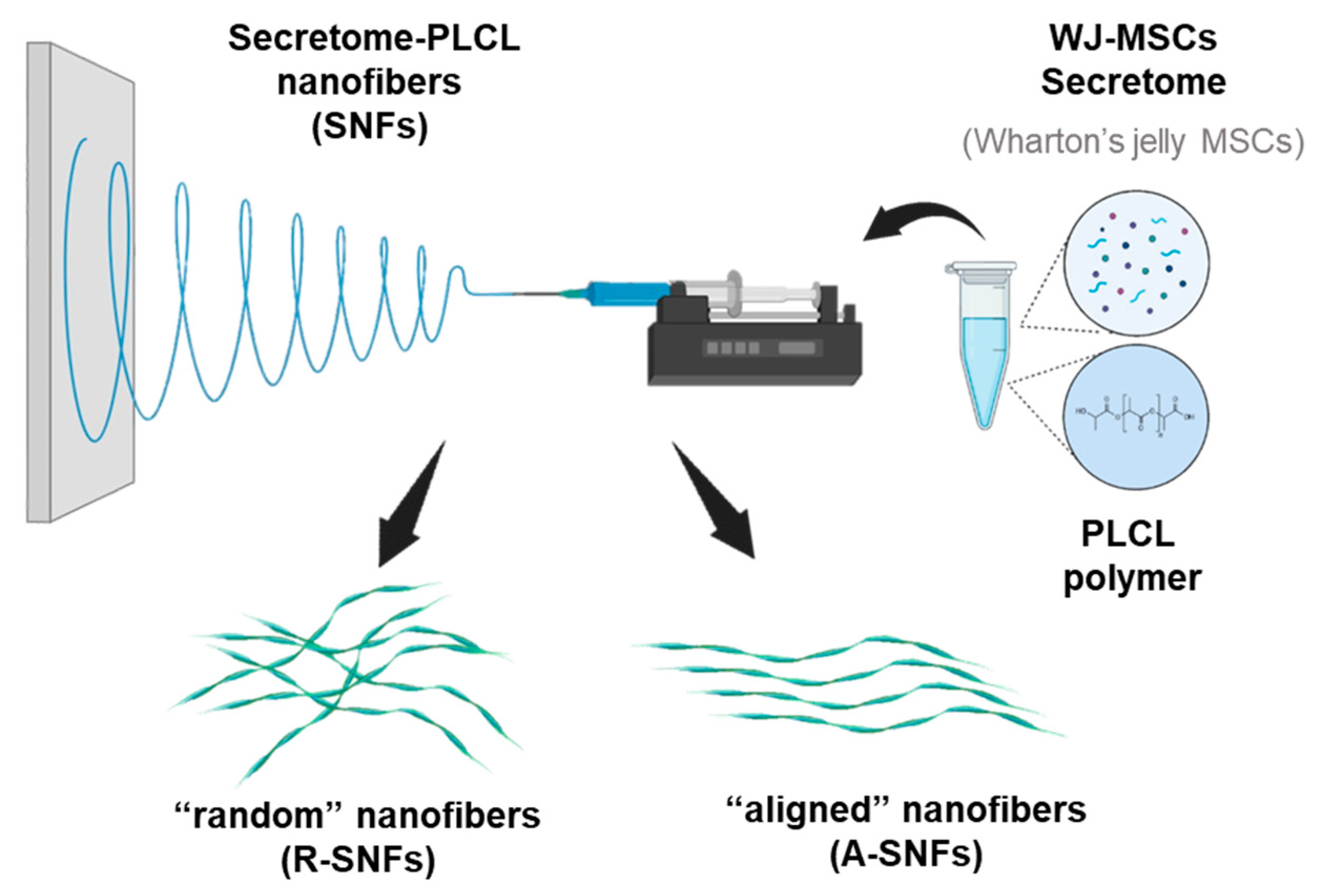
2.1. Eelectrospining
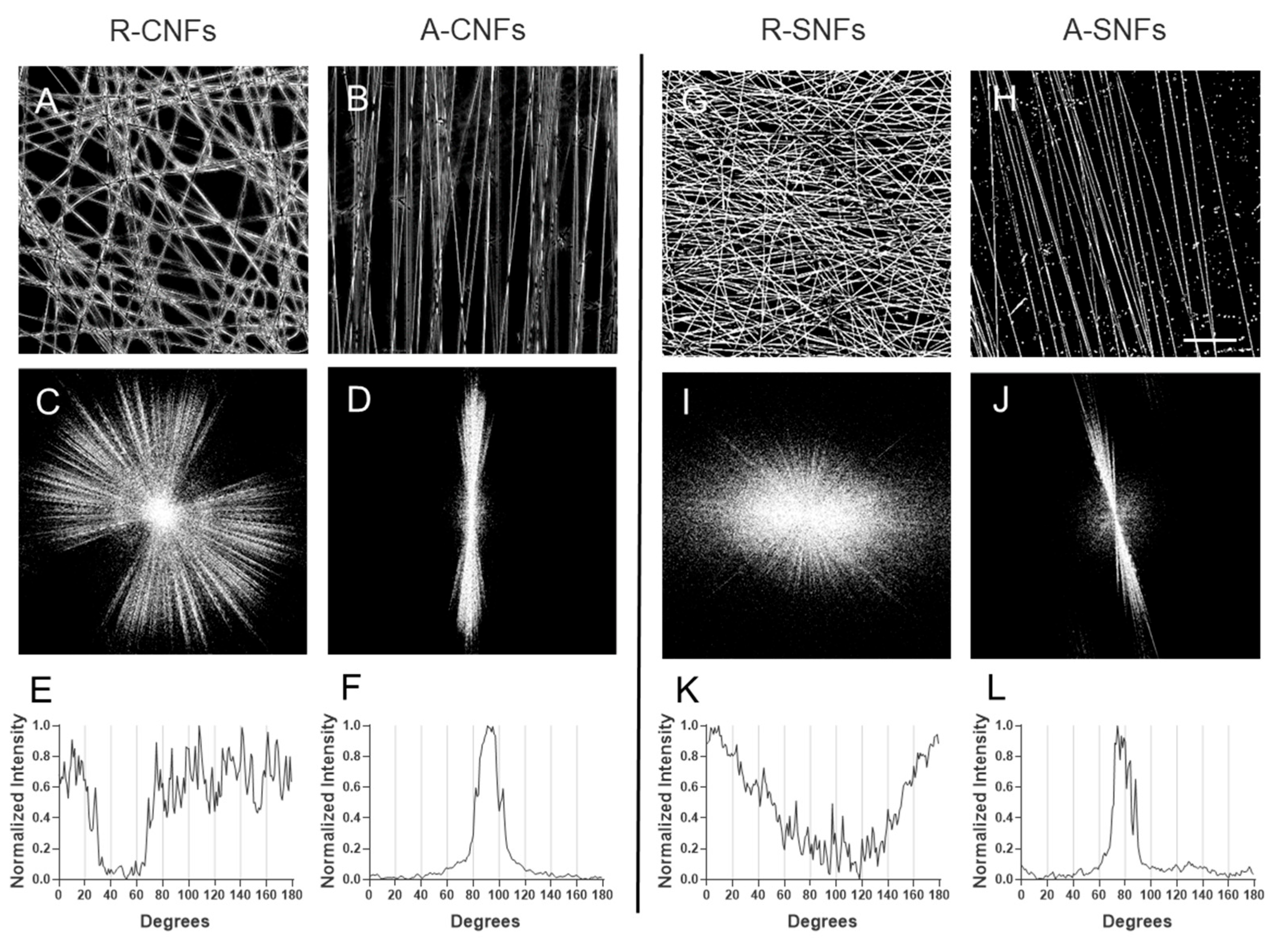
2.2. Fast Fourier Transformation Analysis (FFT) with ImageJ
2.3. Atomic Force Microscopy (AFM)
2.4. FITC Labeling of WJ-MSCs Bioactive Molecules and Purification by Gel Permeation Chromatography (GPC)
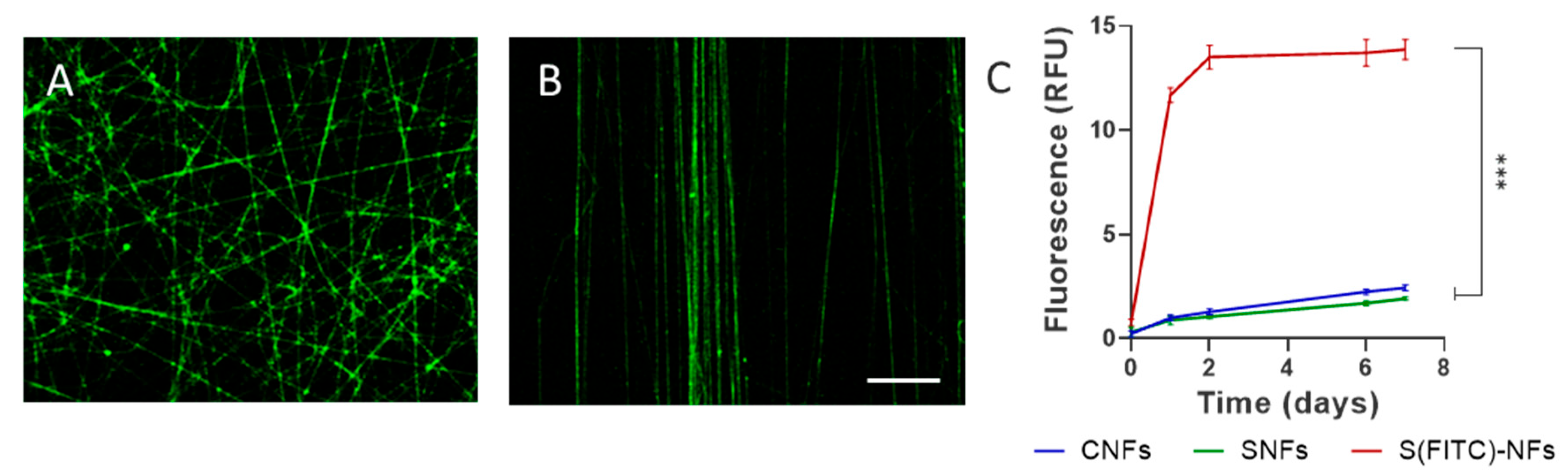
2.5. Protein Release Assay
2.6. Cell Culturing
2.7. Live-Dead Analysis
2.8. Cell Adhesion and Overall Morphology
2.9. Cell Proliferation Assay
2.10. Visualization of Focal Adhesions
2.11. Artificial Wound Healing (Scratch) Assay
2.12. Statistical Analysis
3. Results
3.1. Characterization of Hybrid Secretome-PLCL Nanofibers
3.1.1. Fast Fourier Transform (FFT) Analysis
3.1.2. Release Kinetics of Secretome from Nanofibers
3.1.3. Atomic Force Microscopy (AFM) Analysis of Nanofiber Morphology and Mechanical Properties
3.2. Cellular Interaction
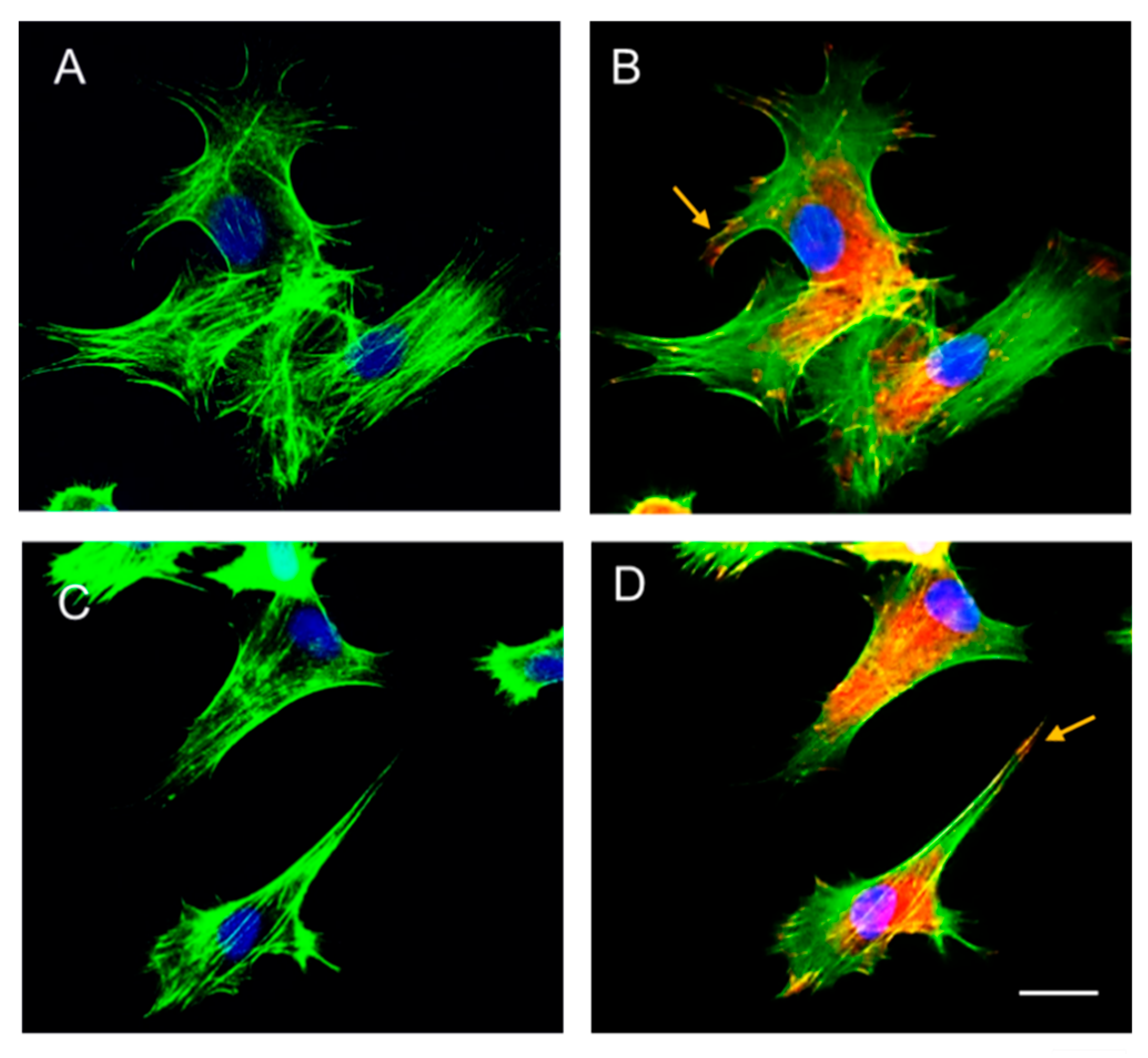
3.2.1. Overall Morphology and Spreading of AD-MSCs Adhering to Secretome-Containing Nanofibers
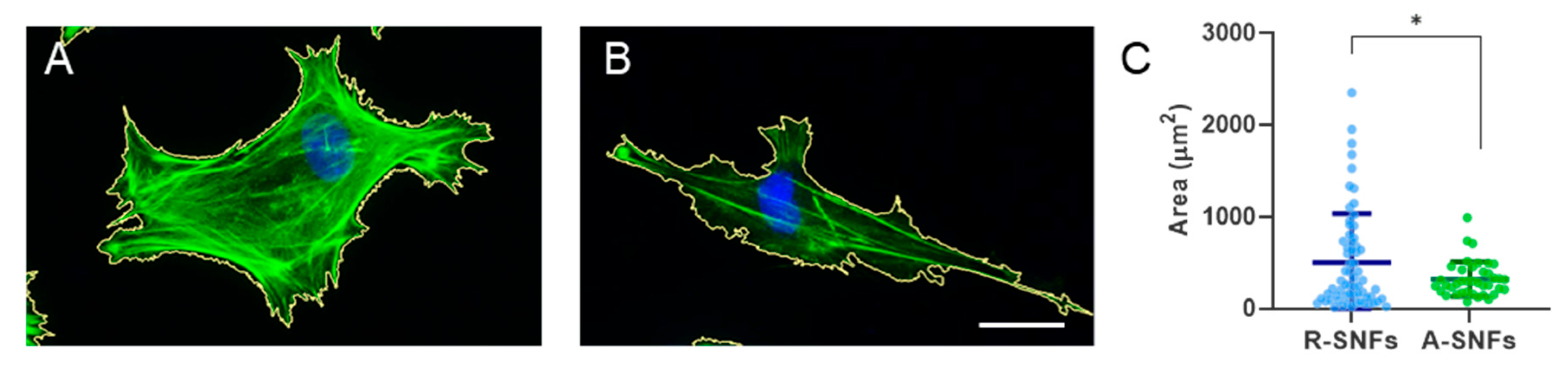
3.2.2. Morphometry Analysis of Cell Spreading and Elongation
3.3. Stem Cells Culture on Secretome PLCL Nanofibers
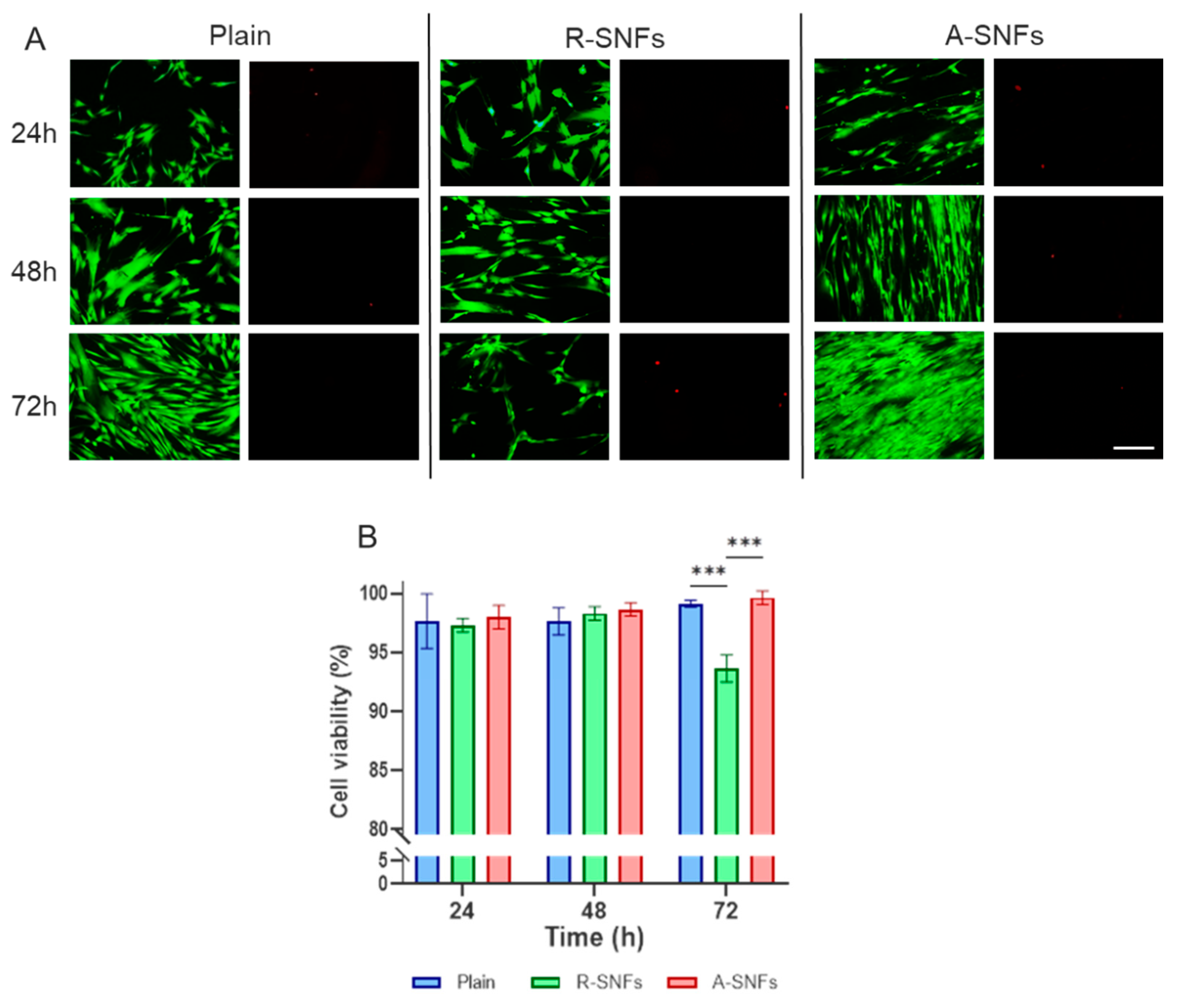
3.3.1. Aligned Nanofibers Sustain AD-MSC Viability During Extended Culture
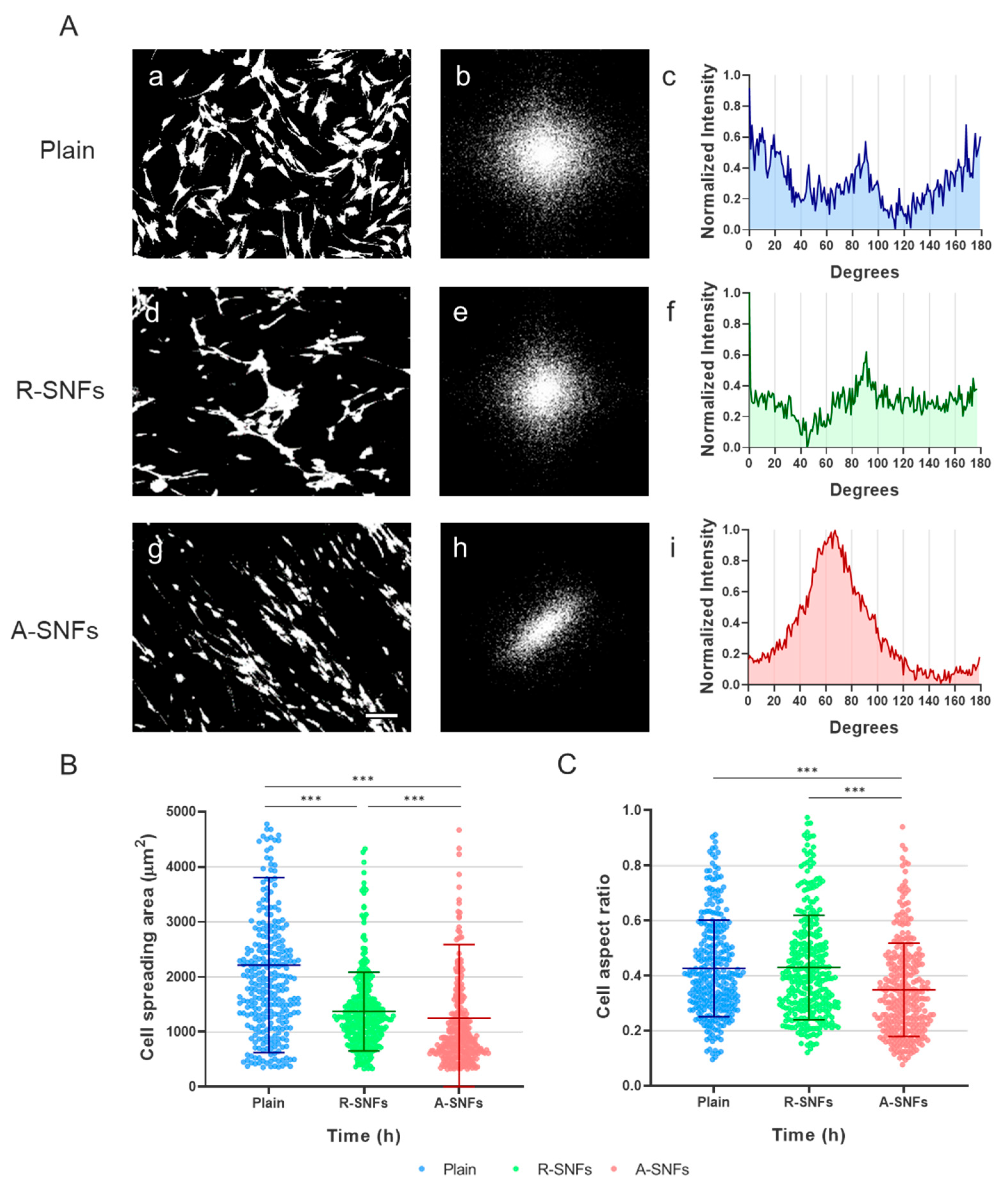
3.3.2. FFT Analysis of Cell Orientation
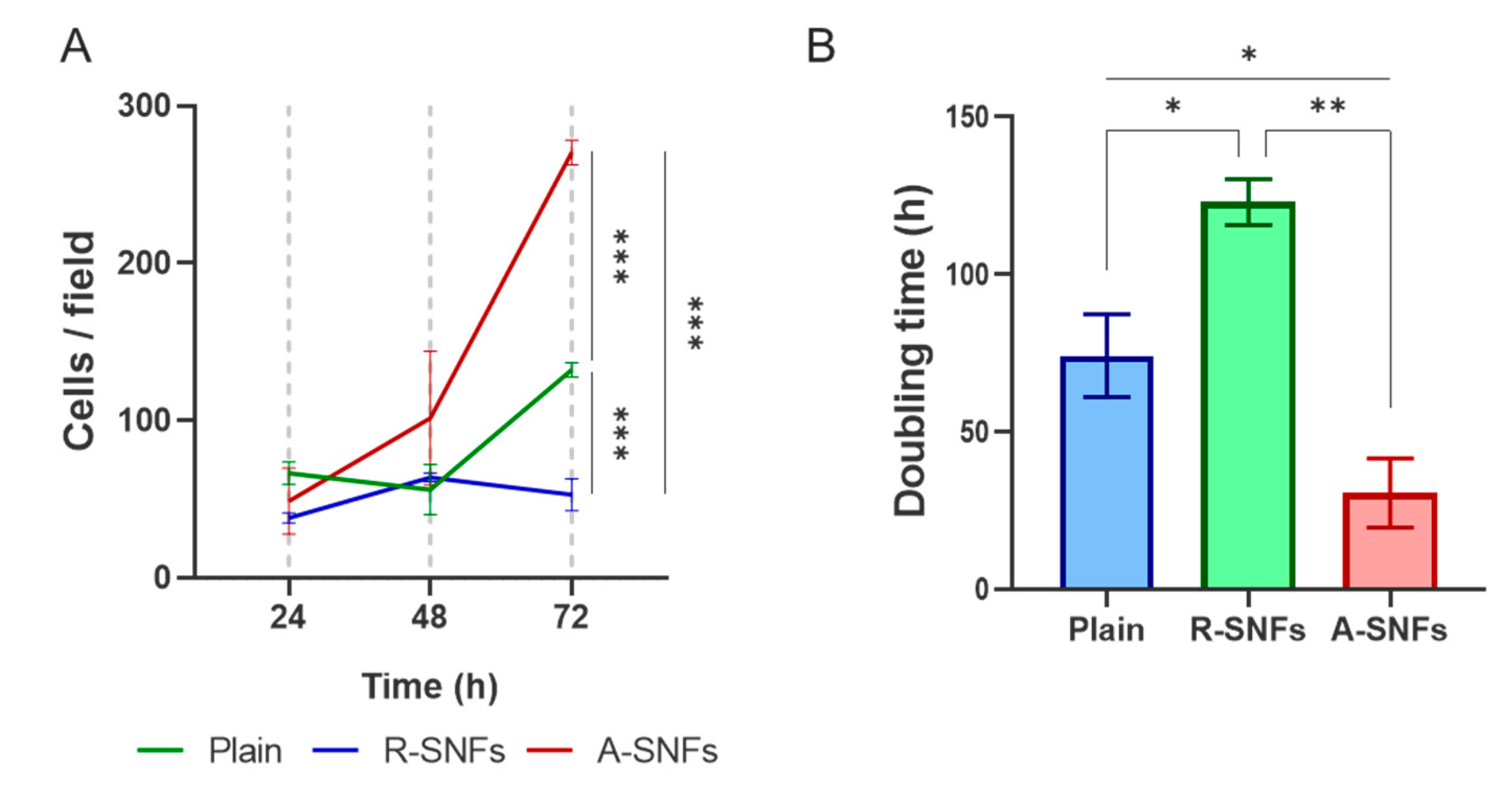
3.3.3. Nanofiber Alignment Supports AD-MSC Proliferation
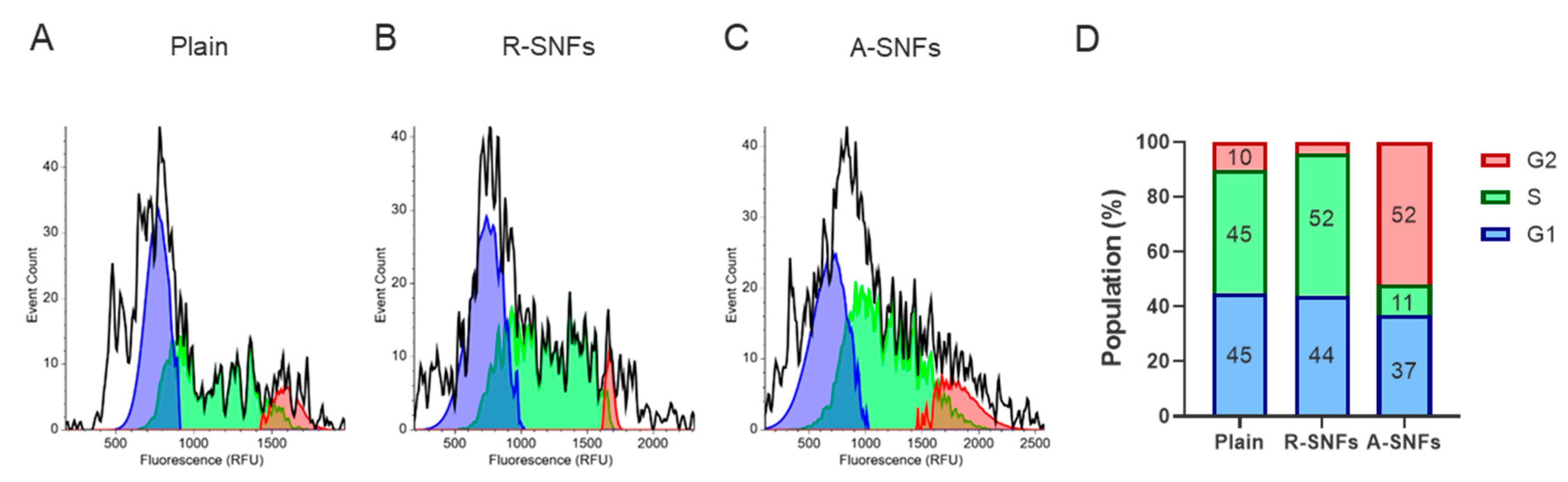
3.3.4. The Nanofiber Configuration Modulates Cell Cycle Progression of AD-MSC
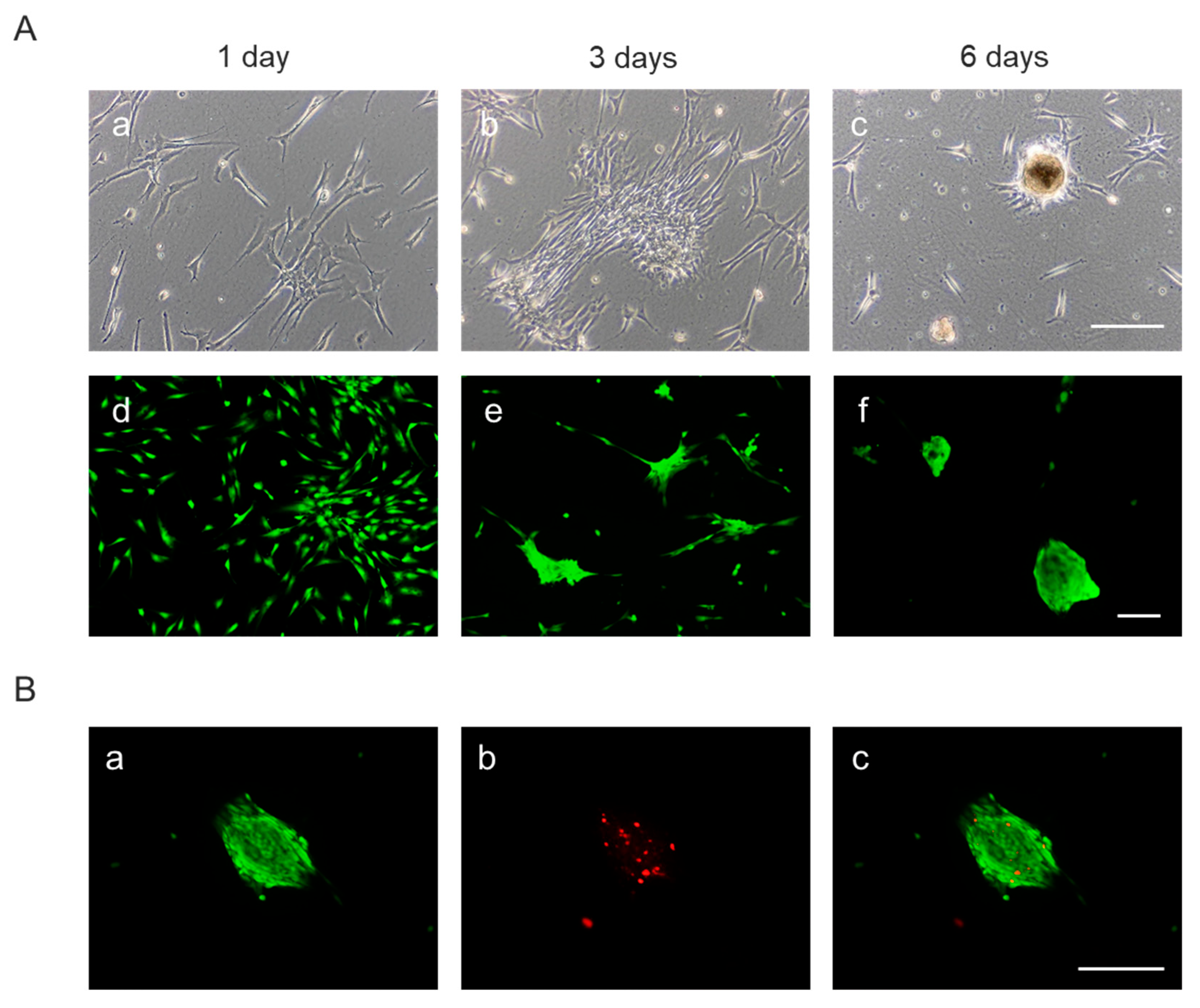
3.3.5. Spheroids Formation
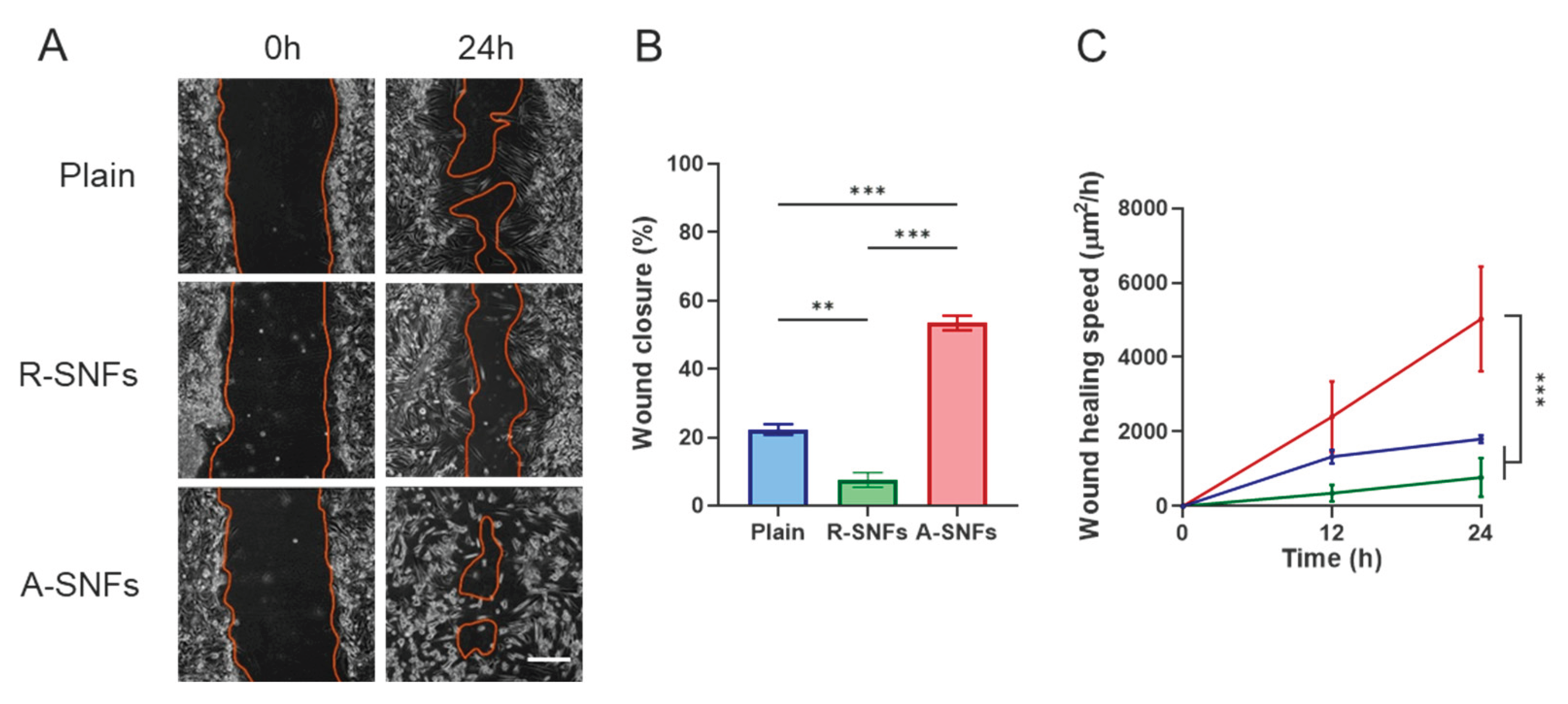
3.4. In Vitro Wound Healing Assay
4. Discussion
4.1. Cell Adhesion, Morphology, and Spreading
4.2. Cell Alignment, Viability, and Proliferation
4.3. In Vitro Wound Healing and Migration
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PLCL | Poly(L-lactide-co-ε-caprolactone) |
| WJ-MSCs | Wharton’s jelly-derived mesenchymal stem cells |
| AFM | Atomic Force Microscopy |
| FFT | Fast Fourier Transform |
| AD-MSCs | Adipose-Derived MSCs |
| ECM | Extracellular Matrix |
| PLA | Poly(Lactic Acid) |
| FDA | Food and Drug Administration |
| MSCs | Mesenchymal Stem Cells |
| VEGF | Vascular Endothelial Growth Factor |
| bFGF | Basic Fibroblast Growth Factor |
| EGF | Epidermal Growth Factor |
| TGF-β | Transforming Growth Factor-β |
| mRNA | Messenger RNA |
| HFIP | Hexafluoroisopropanol |
| R-NFs | Random Nanofibers |
| A-NFs | Aligned Nanofibers |
| NCM | Non-Contact Mode |
| FITC | Fluorescein Isothiocyanate |
| GPC | Gel Permeation Chromatography |
| PBS | Phosphate-Buffered Saline |
| FBS | Fetal Bovine Serum |
References
- J. S. Boateng and K. H. Matthews, “Wound healing dressings and drug delivery systems: a review,” J Pharm Sci, vol. 97, no. 8, pp. 2892–2923, Aug. 2008. [CrossRef]
- S. Guo and L. A. DiPietro, “Factors Affecting Wound Healing,” J Dent Res, vol. 89, no. 3, pp. 219–229, Feb. 2010. [CrossRef]
- D. Chouhan and B. B. Mandal, “Silk biomaterials in wound healing and skin regeneration therapeutics: From bench to bedside,” Acta Biomater, vol. 103, pp. 24–51, Feb. 2020. [CrossRef]
- K. Ye and H. Kuang, “Electrospun Nanofibers for Tissue Engineering with Drug Loading and Release,” Pharmaceutics, vol. 11, no. 4, p. 182, Apr. 2019. [CrossRef]
- J. Xue and T. Wu, “Electrospinning and Electrospun Nanofibers: Methods, Materials, and Applications,” Chem Rev, vol. 119, Mar. 2019. [CrossRef]
- M. Zhang and Z. Chang, “Synthesis of Poly(l-lactide-co-ε-caprolactone) Copolymer: Structure, Toughness, and Elasticity,” Polymers (Basel), vol. 13, no. 8, Apr. 2021. [CrossRef]
- M. Dhayer and V. Barral, “Material and biological characterization of 3D knitted bioresorbable poly (D,L-lactide) (PLA) and polycaprolactone (PCL) scaffolds for soft tissue regeneration: from fabrication to in vivo performance,” J Biol Eng, vol. 53, Jun. 2025. [CrossRef]
- M. Eryildiz and A. Karakus, “Development and Characterization of PLA/PCL Blend Filaments and 3D Printed Scaffolds,” Journal of Materials Engineering and Performance , vol. 34, no. 14, Jul. 2025. [CrossRef]
- C. for D. and R. H. U.S. Food and Drug Administration, “Polycaprolactone (PCL) Safety Profile,” 2021.
- M. Wang and T. Dai, “BMP-2 loaded Bioactive PLLA/PCL Blended nanofibers for synergistic influences on osteogenic differentiation of rat bone marrow-derived mesenchymal stem cells via TGF-β/Smad2/3 signaling pathway,” J Drug Deliv Sci Technol, vol. 105, Mar. 2025. [CrossRef]
- R. Margiana and A. Markov, “Clinical application of mesenchymal stem cell in regenerative medicine: a narrative review,” Stem Cell Res Ther, vol. 13, no. 1, Jul. 2022. [CrossRef]
- B. Zhang and X. Tian, “Mesenchymal Stem Cell-Derived Extracellular Vesicles in Tissue Regeneration,” Cell Transplant, Mar. 2020. [CrossRef]
- J. W. Prado-Yupanqui and L. Ramírez-Orrego, “The Hidden Power of the Secretome: Therapeutic Potential on Wound Healing and Cell-Free Regenerative Medicine-A Systematic Review,” Int J Mol Sci, vol. 26, no. 5, Feb. 2025. [CrossRef]
- E. Novoseletskaya and O. Grigorieva, “Mesenchymal Stromal Cell-Produced Components of Extracellular Matrix Potentiate Multipotent Stem Cell Response to Differentiation Stimuli,” Front Cell Dev Biol, vol. 8, Sep. 2020. [CrossRef]
- A. Krasnodembskaya and Y. Song, “Antibacterial Effect of Human Mesenchymal Stem Cells Is Mediated in Part from Secretion of the Antimicrobial Peptide LL-37,” Stem Cells, vol. 12, Dec. 2010. [CrossRef]
- F. Alcayaga-Miranda and J. Cuenca, “Antimicrobial Activity of Mesenchymal Stem Cells: Current Status and New Perspectives of Antimicrobial Peptide-Based Therapies,” Front Immunol, Mar. 2017. [CrossRef]
- T. Stoyanova and L. Topalova, “Selective Paracrine Modulation of Stromal Cells: Wharton’s Jelly MSC Secretome Enhances Adipose-Derived MSC Functionality While Maintaining Dermal Fibroblast Quiescence,” Int J Mol Sci, vol. 26, no. 20, Oct. 2025. [CrossRef]
- D. R. Stirling and M. J. Swain-Bowden, “CellProfiler 4: improvements in speed, utility and usability,” BMC Bioinformatics, vol. 22, Sep. 2021. [CrossRef]
- A. Suarez-Arnedo and F. T. Figueroa, “An image J plugin for the high throughput image analysis of in vitro scratch wound healing assays,” PLoS One, vol. 15, no. 7, Jul. 2020. [CrossRef]
- D. Gugutkov and J. Gustavsson, “Electrospun fibrinogen–PLA nanofibres for vascular tissue engineering,” J Tissue Eng Regen Med, vol. 25, May 2016. [CrossRef]
- S. Wang and S. Zhong, “Effects of fiber alignment on stem cells–fibrous scaffold interactions,” J Mater Chem B, no. 16, Mar. 2015. [CrossRef]
- C. A. Bashur and L. A. Dahlgren, “Effect of fiber diameter and orientation on fibroblast morphology and proliferation on electrospun poly(D,L-lactic-co-glycolic acid) meshes,” Biomaterials, vol. 27, no. 33, Nov. 2006. no. 33). [CrossRef]
- C.-Y. Huang and K.-H. Hu, “Comparison of cell behavior on pva/ pva-gelatin electrospun nanofibers with random and aligned configuration,” Sci Rep, Dec. 2016.
- N.-E. Ryu and S.-H. Lee, “Spheroid Culture System Methods and Applications for Mesenchymal Stem Cells,” Cells, vol. 8, no. 12, Dec. 2019. [CrossRef]
- M. C. Amores de Sousa and C. A. V Rodrigues, “Functionalization of Electrospun Nanofibers and Fiber Alignment Enhance Neural Stem Cell Proliferation and Neuronal Differentiation,” Front Bioeng Biotechnol, Oct. 2020. [CrossRef]
- F. Yang and R. Murugan, “Electrospinning of nano/micro scale poly(L-lactic acid) aligned fibers and their potential in neural tissue engineering,” Biomaterials, vol. 15, May 2005. [CrossRef]
- D. Gugutkov and C. Gonzalez-Garcia, “Fibrinogen organization at the cell-material interface directs endothelial cell behavior,” J Bioact Compat Polym, vol. 26, no. 4, Jul. 2011. [CrossRef]

| Young’s modulus (GPa) | Size Hight (µm) | Size Width (µm) | Aspect ratio | |
|---|---|---|---|---|
| CNFs | 0.99 ± 0.18 | 2.89 ± 0.73 | 3.88 ± 0.29 | 0.75 ± 0.21 |
| SNFs | 1.49 ± 0.22 | 0.12 ± 0.04 | 0.81 ± 0.16 | 0.15 ± 0.06 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




