Submitted:
10 December 2025
Posted:
11 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Plant and Soil Samples
2.2. Experimental Design
2.3. Data Analysis
3. Results
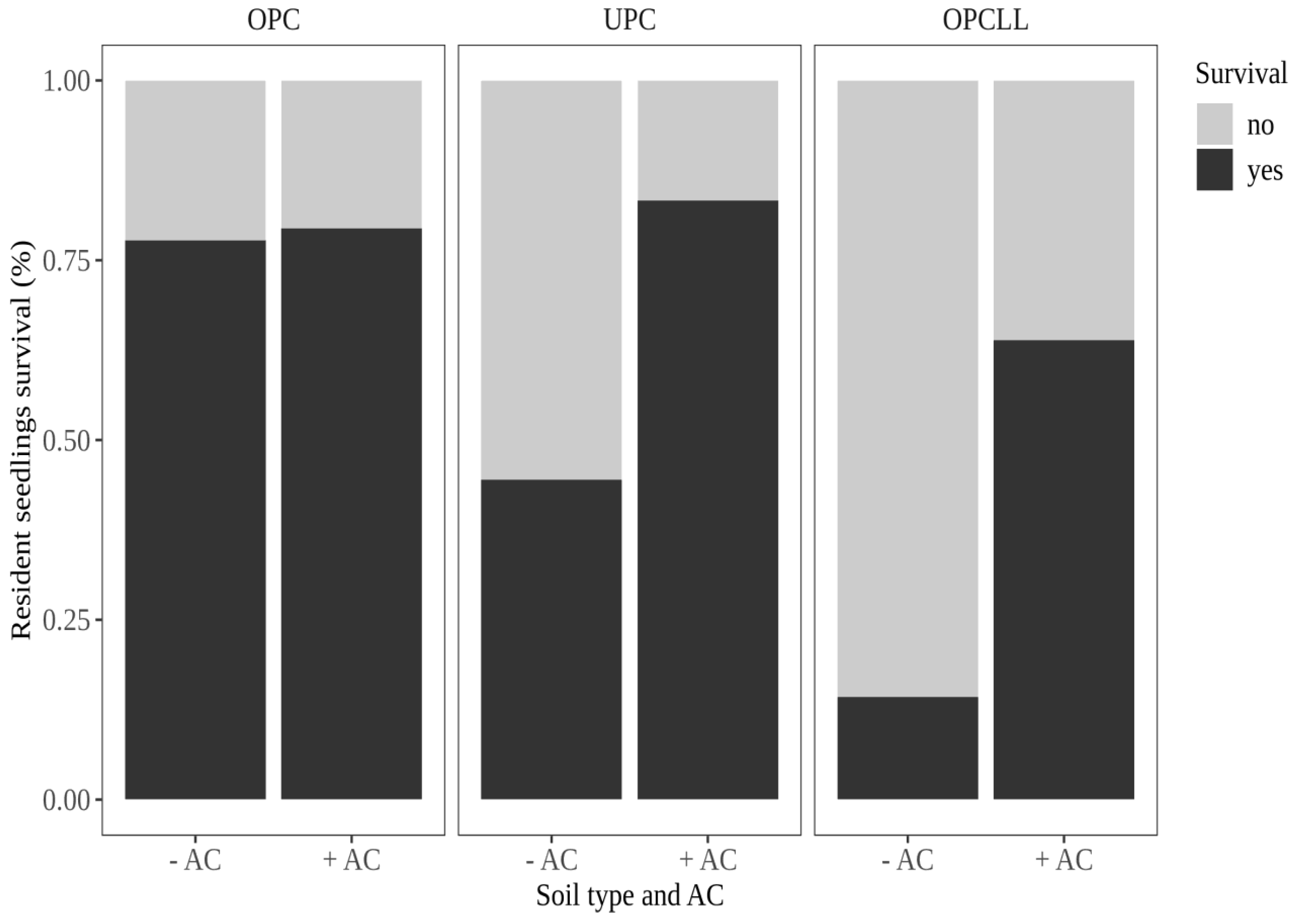
3.1. Effect of Soil Origin and Activated Carbon on Seedling Survival
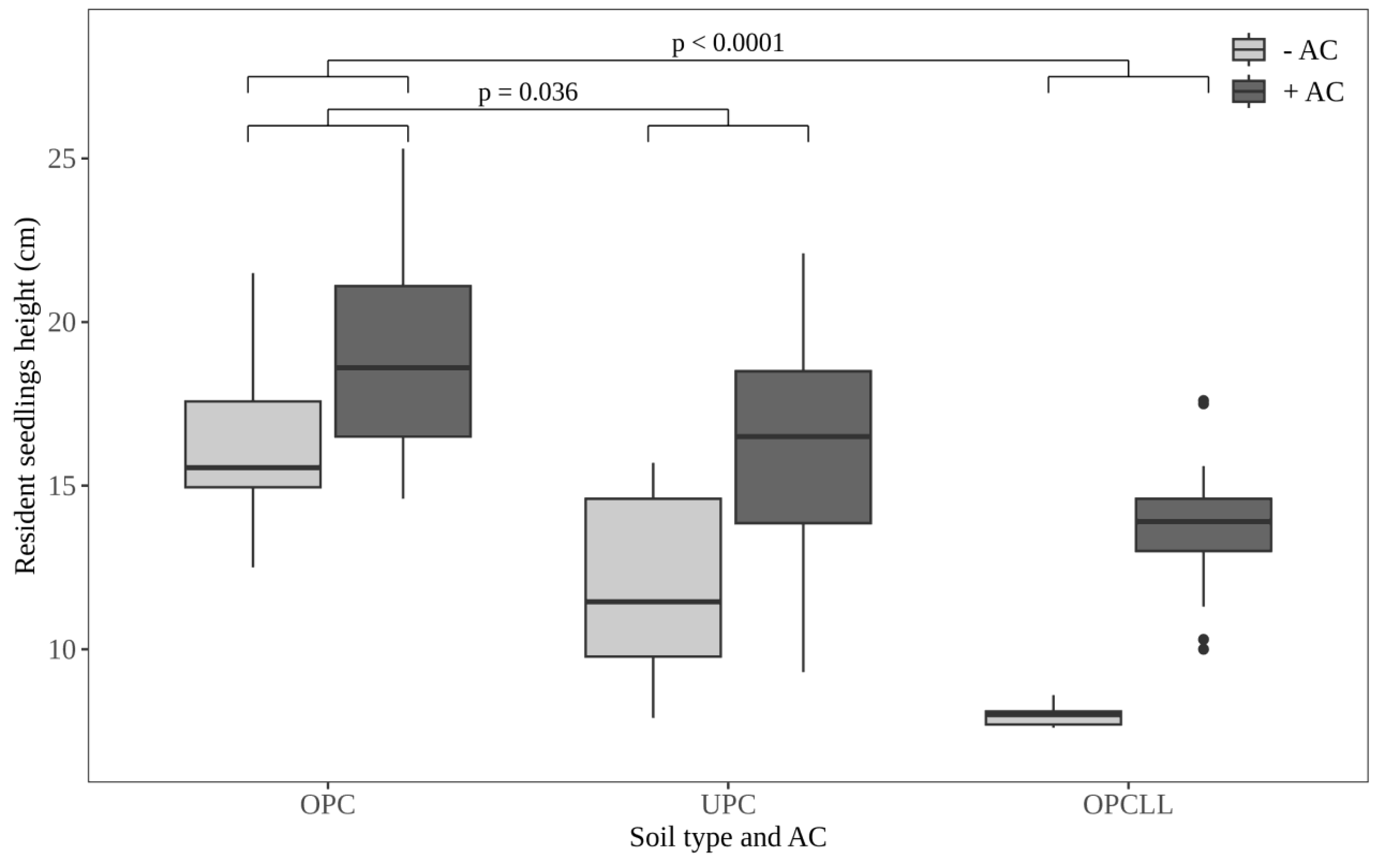
3.2. Effect of Soil Type and AC on Resident Seedlings Height
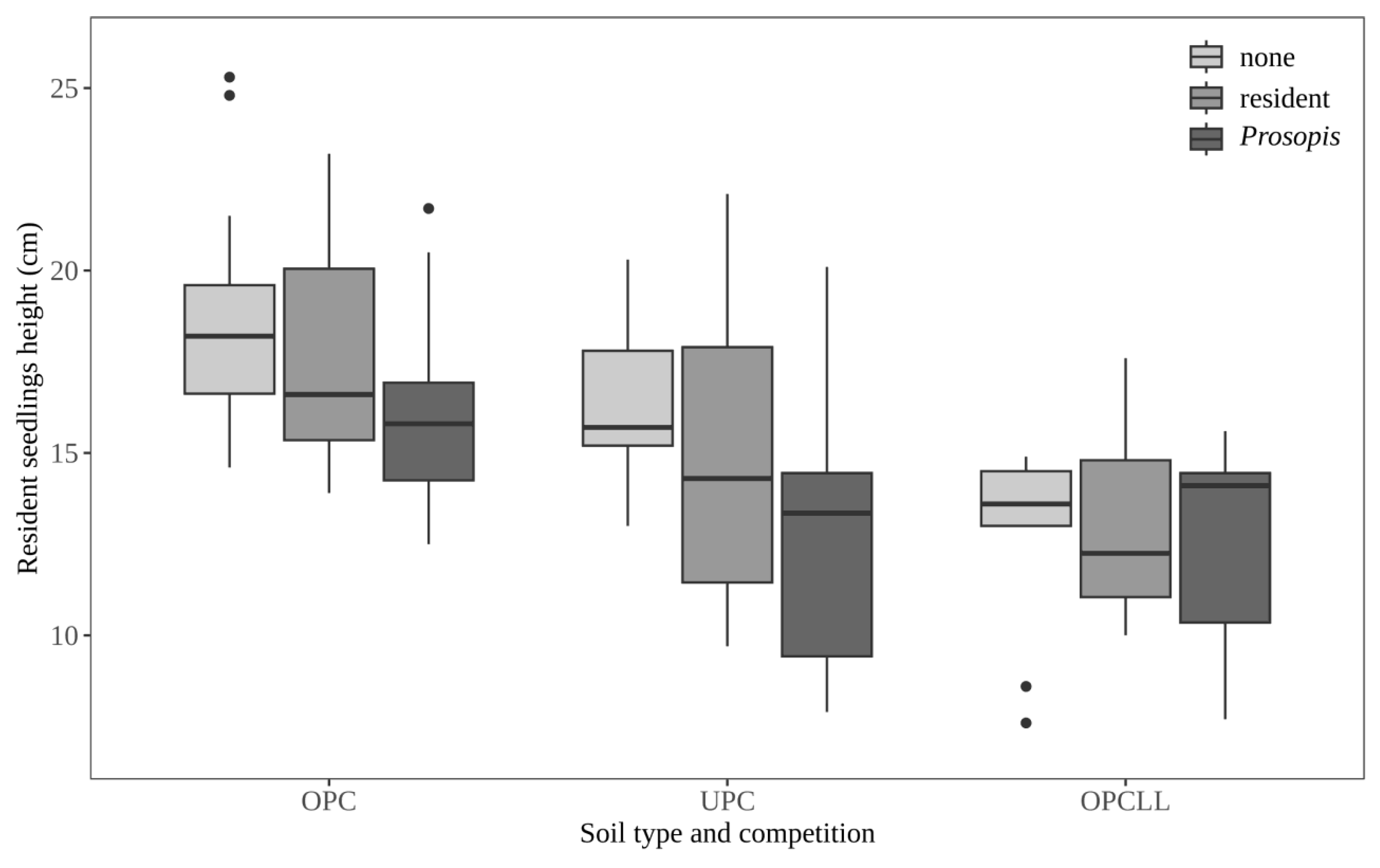
3.3. Effect of Soil Origin and Competition on Seedling Height
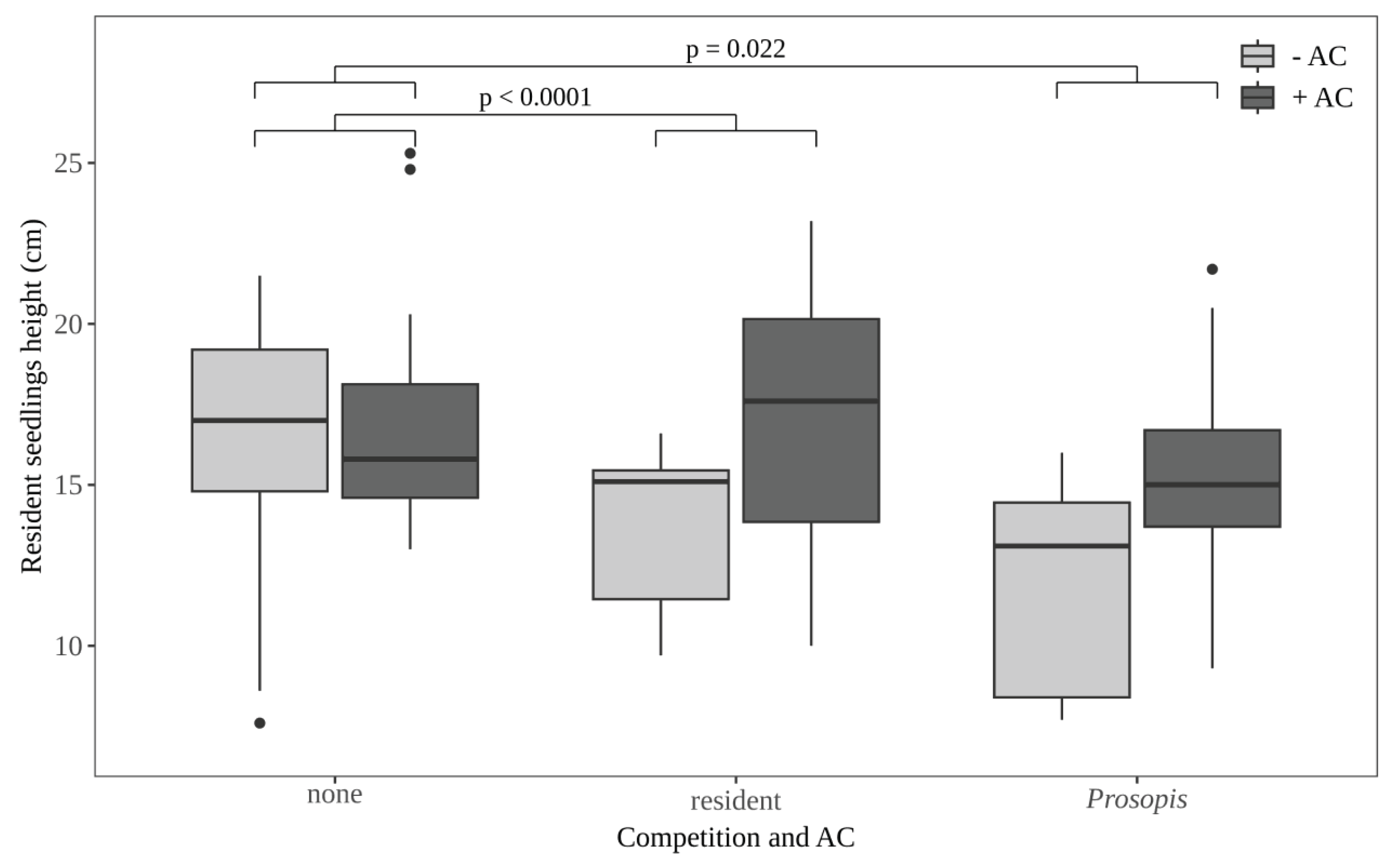
3.4. Effect of Competition and Activated Carbon (AC) on Seedling Height
4. Discussion
4.1. The Effect of Soil Origin
4.2. Leaf Litter as a Source of Allelopathic Substances
4.3. Assessing Allelopathy of P. juliflora Using Multiple Techniques Under Semi-Natural Conditions
5. Conclusion
Supplementary Materials
Acknowledgments
ACRONYMN and Abbreviation
References
- R. M. Callaway and W. M. Ridenour, “Novel weapons: Invasive success and the evolution of increased competitive ability,” Front Ecol Environ, vol. 2, no. 8, pp. 436–443, 2004. [CrossRef]
- T. Qu, X. Du, Y. Peng, W. Guo, C. Zhao, and G. Losapio, “Invasive species allelopathy decreases plant growth and soil microbial activity,” PLoS One, vol. 16, no. 2 February, pp. 1–12, 2021. [CrossRef]
- S. W. Choudhari, T. Chopde, V. P. Mane, and V. B. Shambharkar, “Allelopathic effects of Acacia nilotica (L.) Leaf leachate with emphasis on Trigonella foenum graceum L. (fenugreek),” vol. 8, no. 1, pp. 500–506, 2019.
- L. Yuan, J. M. Li, F. H. Yu, A. M. O. Oduor, and M. van Kleunen, “Allelopathic and competitive interactions between native and alien plants,” Biol Invasions, vol. 23, no. 10, pp. 3077–3090, 2021. [CrossRef]
- Y. Xu, X. Chen, L. Ding, and C. H. Kong, “Allelopathy and Allelochemicals in Grasslands and Forests,” Mar. 01, 2023, MDPI. [CrossRef]
- R. M. U. K. Gunarathne and G. A. D. Perera, “Does the invasion of Prosopis juliflora cause the die-back of the native Manilkara hexandra in seasonally dry tropical forests of Sri Lanka?,” Trop Ecol, vol. 57, no. 3, pp. 475–488, 2016.
- S. Getachew, S. Demissew, and T. Woldemariam, “Allelopathic effects of the invasive Prosopis juliflora (Sw.) DC. on selected native plant species in mIddle Awash, Southern Afar Rift of Ethiopia,” Management of Biological Invasions, vol. 3, no. 2, pp. 105–114, 2012. [CrossRef]
- J. Mungoche, O. V. Wasonga, D. Ikiror, H. Akala, C. Gachuiri, and G. Gitau, “Prosopis juliflora (sw.) DC in the drylands: A review of invasion, impacts and management in Eastern Africa,” 2025, Taylor and Francis Ltd. [CrossRef]
- A. Noha, L. Nigatu, and R. Manikandan, “Allelopathy effect of prosopis juliflora on selected grass species (Cenchrus ciliaris, paspalidium desertorum and lintonia nutans),” Journal of Phytology, vol. 13, pp. 75–78, 2021. [CrossRef]
- R. M. C. S. Ratnayake, “Allelopathic effects of Prosopis juliflora (Sw.) Dc. on selected crops and native plants,” no. November, 2021.
- A. Noha, L. Nigatu, and R. Manikandan, “Allelopathy effect of prosopis juliflora on selected grass species (Cenchrus ciliaris, paspalidium desertorum and lintonia nutans),” Journal of Phytology, vol. 13, pp. 75–78, 2021. [CrossRef]
- S. Endris, “Gene Conserve - Articles - Articles - - Articles - - Articles - Gene Conserve - Articles - Articles - Volume 7 - Issue 30 - October / December, 2008.,” Production, vol. 7, no. 27, pp. 2008–2010, 2008. [CrossRef]
- Z. H. Mehari, “The invasion of Prosopis juliflora and Afar pastoral livelihoods in the Middle Awash area of Ethiopia,” Ecol Process, vol. 4, no. 1, pp. 1–9, 2015. [CrossRef]
- A. El-Keblawy and A. Al-Rawai, “Impacts of the invasive exotic Prosopis juliflora (Sw.) D.C. on the native flora and soils of the UAE,” Plant Ecol, vol. 190, no. 1, pp. 23–35, 2007. [CrossRef]
- G. Asrat and A. Seid, “Allelopathic effect of meskit (Prosopis juliflora (Sw.) DC) aqueous extracts on tropical crops tested under laboratory conditions,” Momona Ethiopian Journal of Science, vol. 9, no. 1, p. 32, 2017. [CrossRef]
- S. Bibi, A. Bibi, M. A. Al-Ghouti, and M. H. Abu-Dieyeh, “Allelopathic Effects of the Invasive Prosopis juliflora (Sw.) DC. on Native Plants: Perspectives toward Agrosystems,” Agronomy, vol. 13, no. 2, 2023. [CrossRef]
- R. M. U. K. Gunarathne and G. A. D. Perera, “Does the invasion of Prosopis juliflora cause the die-back of the native Manilkara hexandra in seasonally dry tropical forests of Sri Lanka?,” Trop Ecol, vol. 57, no. 3, pp. 475–488, 2016.
- D. Biológica et al., “TESIS DOCTORAL CONGENERIC APPROACH FOR UNDERSTANDING INVASIVE ABILITY OF THE EXOTIC PROSOPIS JULIFLORA UNDER THE HYPER-ARID DESERT OF THE UNITED ARAB EMIRATES (UAE) Programa de Doctorado,” 2022. [Online]. Available: https://orcid.org/0000-0001-9740-894X.
- H. Shiferaw, T. Alamirew, S. Dzikiti, W. Bewket, G. Zeleke, and U. Schaffner, “Water use of Prosopis juliflora and its impacts on catchment water budget and rural livelihoods in Afar Region, Ethiopia,” Sci Rep, vol. 11, no. 1, pp. 1–14, 2021. [CrossRef]
- Z. Zhang, Y. Liu, L. Yuan, E. Weber, and M. van Kleunen, “Effect of allelopathy on plant performance: a meta-analysis,” Ecol Lett, vol. 24, no. 2, pp. 348–362, 2021. [CrossRef]
- Q. Wang, M. Kong, J. Wang, B. Gao, and X. Ping, “The Specific Impacts of Allelopathy and Resource Competition from Artemisia frigida on the Growth of Three Plant Species in Northern China,” Plants, vol. 13, no. 23, 2024. [CrossRef]
- D. Cipollini and K. Cipollini, “A review of garlic mustard (Alliaria petiolata, Brassicaceae) as an allelopathic plant,” Journal of the Torrey Botanical Society, vol. 143, no. 4, pp. 339–348, 2016. [CrossRef]
- M. Parepa and O. Bossdorf, “Testing for allelopathy in invasive plants: it all depends on the substrate!,” Biol Invasions, vol. 18, no. 10, pp. 2975–2982, 2016. [CrossRef]
- M. Parepa and O. Bossdorf, “Testing for allelopathy in invasive plants: it all depends on the substrate!,” Biol Invasions, vol. 18, no. 10, pp. 2975–2982, 2016. [CrossRef]
- Z. Zhang, Y. Liu, L. Yuan, E. Weber, and M. van Kleunen, “Effect of allelopathy on plant performance: a meta-analysis,” Ecol Lett, vol. 24, no. 2, pp. 348–362, 2021. [CrossRef] [PubMed]
- J. A. Lau et al., “Inference of allelopathy is complicated by effects of activated carbon on plant growth,” pp. 412–423, 2003.
- G. Asrat and A. Seid, “Allelopathic effect of meskit (Prosopis juliflora (Sw.) DC) aqueous extracts on tropical crops tested under laboratory conditions,” Momona Ethiopian Journal of Science, vol. 9, no. 1, p. 32, 2017. [CrossRef]
- S. Getachew, S. Demissew, and T. Woldemariam, “Allelopathic effects of the invasive Prosopis juliflora (Sw.) DC. on selected native plant species in mIddle Awash, Southern Afar Rift of Ethiopia,” Management of Biological Invasions, vol. 3, no. 2, pp. 105–114, 2012. [CrossRef]
- C. Murrell, E. Gerber, C. Krebs, M. Parepa, U. Schaffner, and O. Bossdorf, “Nvasive knotweed affects native plants through allelopathy 1,” vol. 98, no. 1, pp. 38–43, 2011. [CrossRef]
- A. Pechci’nskapechci’nska, “The Robustness of Heteroscedasticity Tests,” 2023.
- A. F. Hayes and L. I. Cai, “Using heteroskedasticity-consistent standard error estimators in OLS regression: An introduction and software implementation,” 2007.
- G. Peguero, O. R. Lanuza, R. Savé, and J. M. Espelta, “Allelopathic potential of the neotropical dry-forest tree Acacia pennatula Benth.: Inhibition of seedling establishment exceeds facilitation under tree canopies,” Plant Ecol, vol. 213, no. 12, pp. 1945–1953, 2012. [CrossRef]
- P. Lesica and T. H. Deluca, “Is tamarisk allelopathic?,” 2004.
- M. Dorning and D. Cipollini, “Leaf and root extracts of the invasive shrub, Lonicera maackii, inhibit seed germination of three herbs with no autotoxic effects,” Plant Ecol, vol. 184, no. 2, pp. 287–296, Jun. 2006. [CrossRef]
- “packer2000”.
- H. Kato-Noguchi, “Allelopathy and Allelochemicals of Imperata cylindrica as an Invasive Plant Species,” Oct. 01, 2022, MDPI. [CrossRef]
- Z. Gul, A. Akbar, and S. K. Leghari, “Elucidating Therapeutic and Biological Potential of Berberis baluchistanica Ahrendt Bark, Leaf, and Root Extracts,” Front Microbiol, vol. 13, no. March, pp. 1–14, 2022. [CrossRef]
- D. Zhang et al., “The Dynamics of Allelochemicals and Phytotoxicity in Eisenia fetida during the Decomposition of Eucalyptus grandis Litter,” Plants, vol. 13, no. 17, Sep. 2024. [CrossRef]
- M. Parepa, U. Schaffner, and O. Bossdorf, “Sources and modes of action of invasive knotweed allelopathy: the effects of leaf litter and trained soil on the germination and growth of native plants,” NeoBiota, vol. 13, pp. 15–30, 2012. [CrossRef]
- W. Huang, G. V. P. Reddy, P. Shi, J. Huang, H. Hu, and T. Hu, “Allelopathic effects of Cinnamomum septentrionale leaf litter on Eucalyptus grandis saplings,” Glob Ecol Conserv, vol. 21, p. e00872, 2020. [CrossRef]
- M. Parepa and O. Bossdorf, “Testing for allelopathy in invasive plants: it all depends on the substrate!,” Biol Invasions, vol. 18, no. 10, pp. 2975–2982, 2016. [CrossRef]
- D. Cipollini and K. Cipollini, “A review of garlic mustard (Alliaria petiolata, Brassicaceae) as an allelopathic plant,” Journal of the Torrey Botanical Society, vol. 143, no. 4, pp. 339–348, 2016. [CrossRef]
- A. I. Arroyo et al., “Evidence for chemical interference effect of an allelopathic plant on neighboring plant species: A field study,” PLoS One, vol. 13, no. 2, 2018. [CrossRef]
- S. Shannon-Firestone and J. Firestone, “Allelopathic potential of invasive species is determined by plant and soil community context,” Plant Ecol, vol. 216, no. 3, pp. 491–502, 2015. [CrossRef]
- Z. Noumi and M. Chaieb, “Allelopathic effects of Acacia tortilis (Forssk.) hayne subsp. Raddiana (Savi) brenan in North Africa,” Pak J Bot, vol. 43, no. 6, pp. 2801–2805, 2011.
- R. Kaur et al., “Community Impacts of Prosopis juliflora Invasion: Biogeographic and Congeneric Comparisons,” PLoS One, vol. 7, no. 9, 2012. [CrossRef]
- C. Wang et al., “Effects of autotoxicity and allelopathy on seed germination and seedling growth in Medicago truncatula,” Front Plant Sci, vol. 13, no. July, pp. 1–11, 2022. [CrossRef]
- C. Murrell, E. Gerber, C. Krebs, M. Parepa, U. Schaffner, and O. Bossdorf, “Nvasive knotweed affects native plants through allelopathy 1,” vol. 98, no. 1, pp. 38–43, 2011. [CrossRef]
- S. Shannon-Firestone and J. Firestone, “Allelopathic potential of invasive species is determined by plant and soil community context,” Plant Ecol, vol. 216, no. 3, pp. 491–502, 2015. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



