2.1. Preparation Strategy of Silica Doped with Chitosan Gels
The samples were dried at ambient pressure, yielding compact and homogeneous cylindrical xerogels. Samples containing 0.5 and 1 mL of chitosan required mild heating in a water bath at 50 °C to induce gelation, whereas all other samples formed homogeneous sols that gelled spontaneously at room temperature. In other words, at low chitosan concentrations, gelation required mild heating, while at higher concentrations, the sols underwent spontaneous gelation at room temperature. This behavior indicates that chitosan acts as a structure-directing and gel-promoting component, influencing both the kinetics of network formation and the final textural properties of the composites. The resulting xerogels exhibited uniform, monolithic structures without visible cracking, suggesting good compatibility between the silica and biopolymer phases. The preparation scheme is shown in
Figure 1.
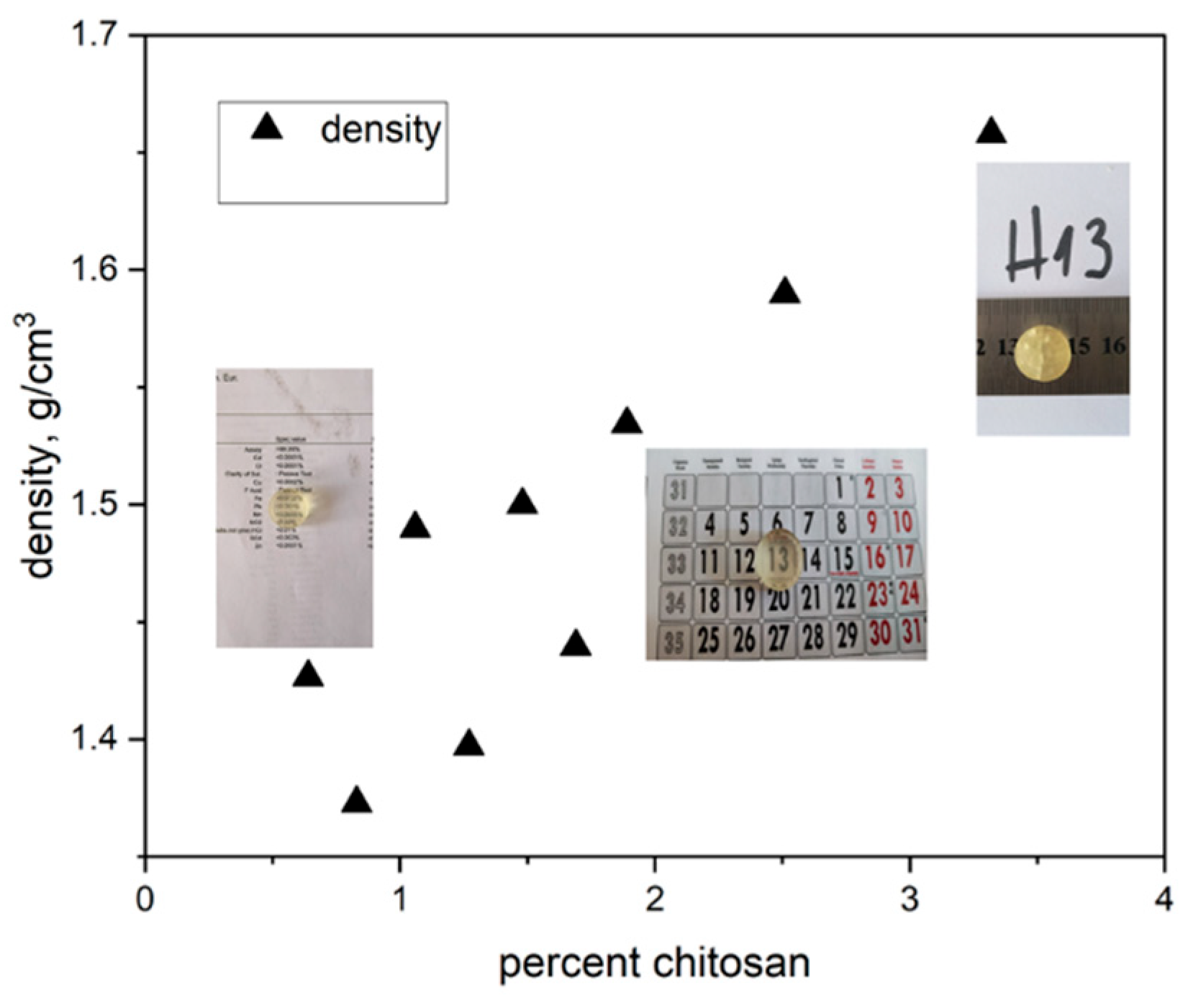
The prepared gels exhibit high density and well-defined cylindrical shapes with uniform size distribution: diameter 1.65–1.80 cm and height 0.5–0.6 cm. The gel densities range from 1.40 to 1.66 g·cm⁻³, increasing with the amount of chitosan added (
Figure 2). Low-doped samples are transparent, whereas gels with higher chitosan content become increasingly opaque, while all retain high density and well-defined shapes.
The chitosan-doped silica composites (0.2–3.3 % chitosan) prepared via the sol–gel method in this study exhibit a high degree of homogeneity and reproducible structural integrity, demonstrating the robustness of the materials. The resulting materials combine a unique set of properties: the mechanical stability of SiO₂ is complemented by the biocompatibility and functional groups of chitosan. This synergy broadens the potential application range of the composites, including pollutant adsorption, controlled drug delivery, catalytic supports, antibacterial coatings, and chemical or biosensing platforms.
2.2. Thermal Analysis
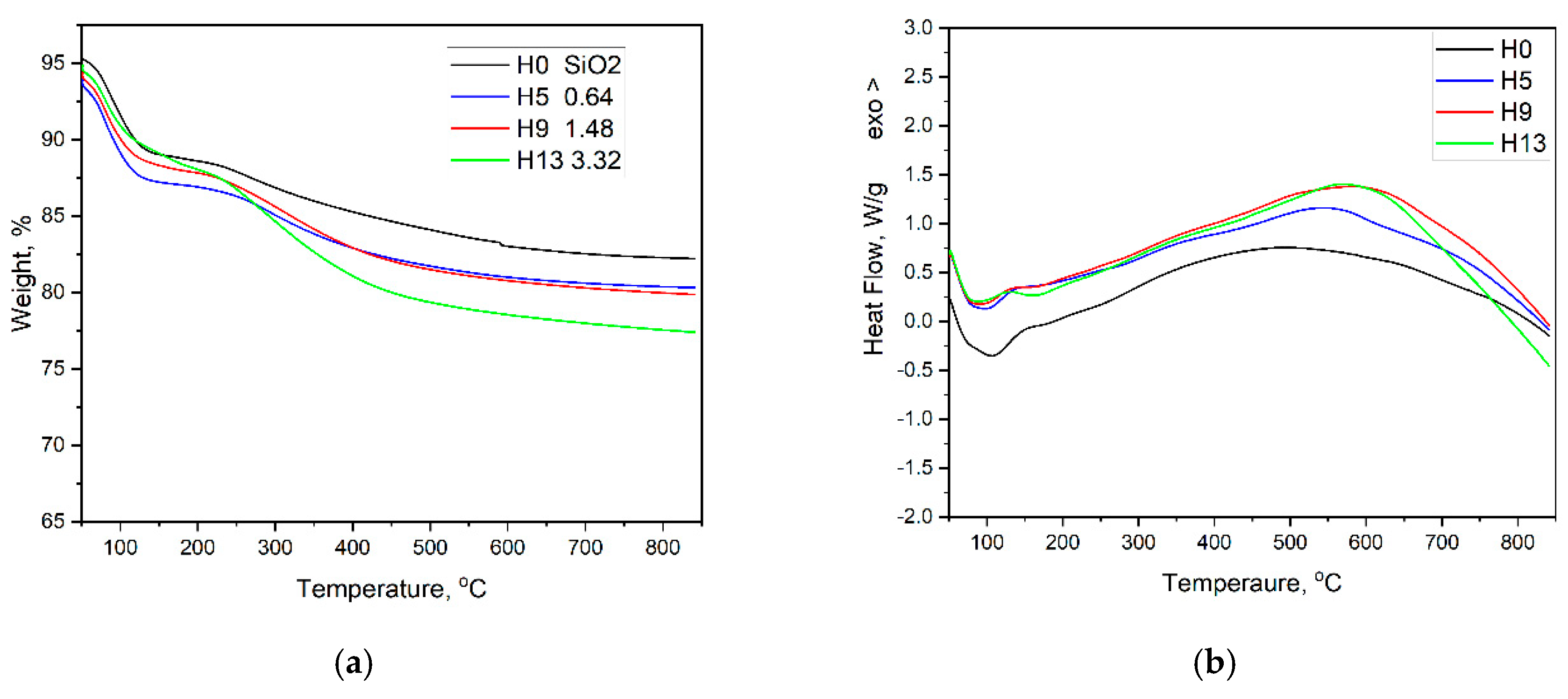
The thermal behavior of a series of chitosan-doped silica composites with different chitosan contents are also investigated. Thermogravimetric analysis (TGA) shows that chitosan decomposition occurs between 200 °C and 300 °C, which is confirmed by differential thermal analysis (DTA). In the pure SiO₂ gel, an endothermic peak appears at 100–150 °C, corresponding to the loss of physically adsorbed water (about 15% mass release), followed by a small endothermic peak at 200–300 °C, associated with the decomposition of chitosan in doped samples. Between 500 °C and 700 °C, the already degraded chitosan undergoes further decomposition and carbonization, (
Figure 3). The broad DTA peak observed in this temperature range is most likely due to the gradual nature of this process, including partial oxidation of the residual organic material and the formation of carbonaceous char [
12,
13].
The thermal analysis results presented in this paper are consistent with our previous data concerning sol–gel silica-based nanocomposites [
1,
21]. While the mass loss at low temperatures is associated with dehydration processes of the matrix, it is clearly evident that the thermal effects observed at elevated temperatures are related to structural transformations and decomposition of the dopant—chitosan—and depend on the doping level. The conducted studies indicate that the obtained composites are suitable for applications at low temperatures, T < 50 °C. This low thermal stability could be improved in future work through the use of hydrophobizing additives, as in the case of optical composites based on hydrophobic aerogels. Such a preparative strategy, however, would lead to interactions between the dopant and the hydrophobizing agent.
2.3. Optical Properties of Chitosan Solutions and Chitosan-Doped Materials
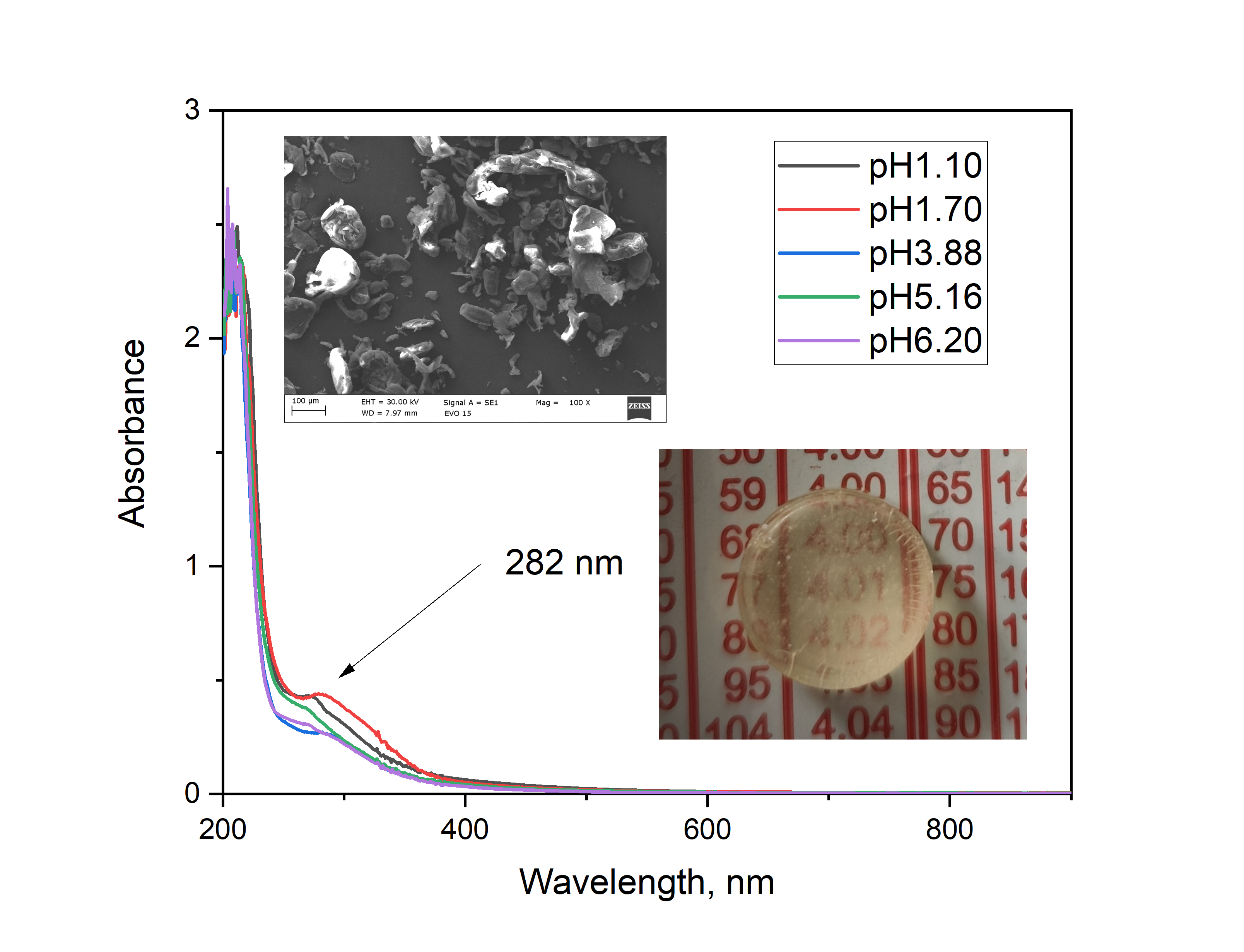
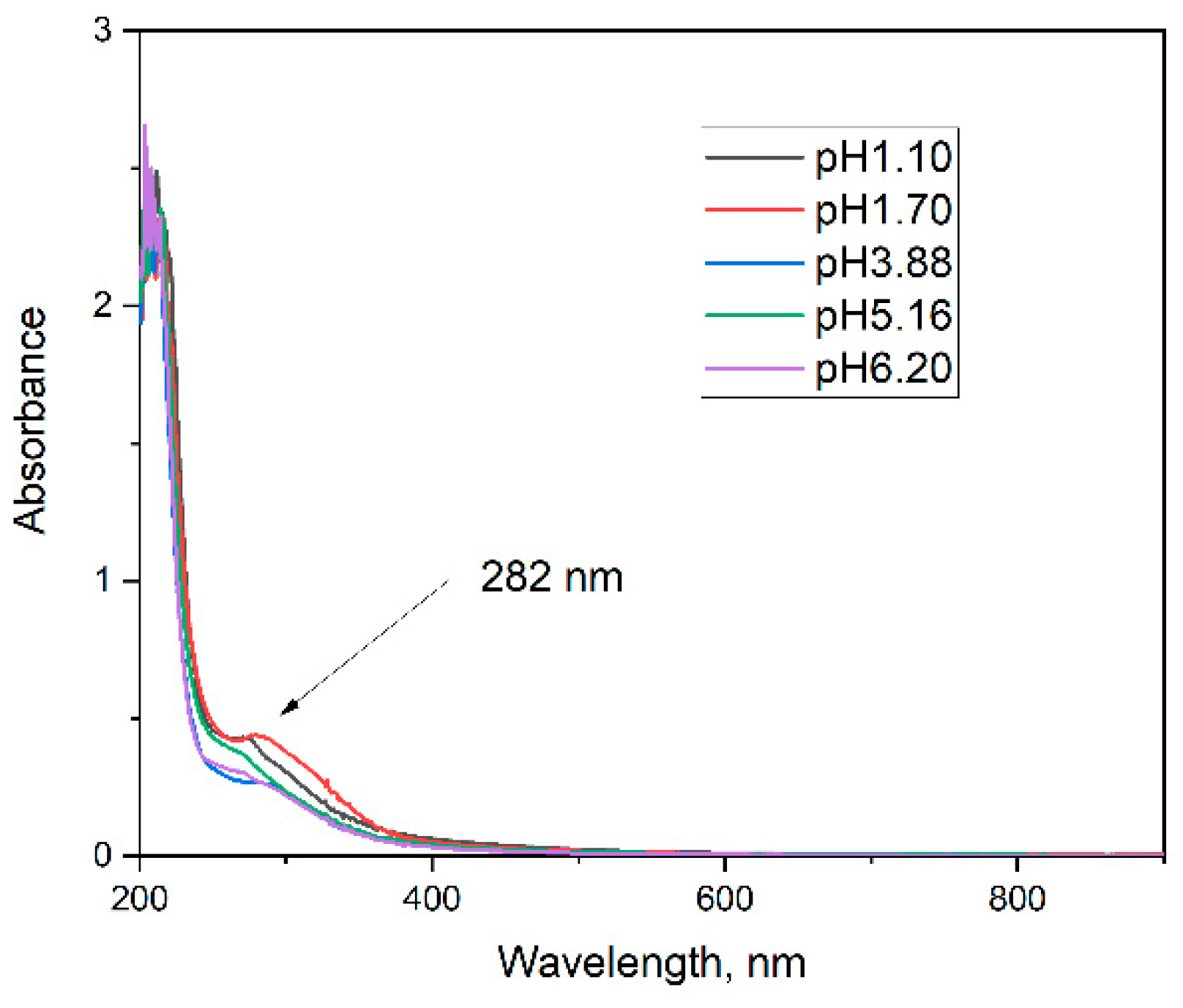
Chitosan solutions exhibit characteristic optical properties related to their polysaccharide nature, as well as to the degree of deacetylation, molecular weight, and pH of the medium. In aqueous or mildly acidic solutions, chitosan is optically transparent in the visible light range (400–800 nm), showing absorption in the UV region below 300 nm, which is primarily attributed to the n→π* transitions of the amine groups. Increasing the polymer concentration enhances light scattering, particularly in the presence of aggregation or pH-induced partial precipitation (
Figure 4). Such changes in optical behavior can provide useful insights into polymer–solvent interactions, conformational changes, and the stability of chitosan solutions. According to Ghazy, [
14] the optical properties of chitosan films extracted from shrimp depend on the degree of deacetylation and the molecular structure of the biopolymer.
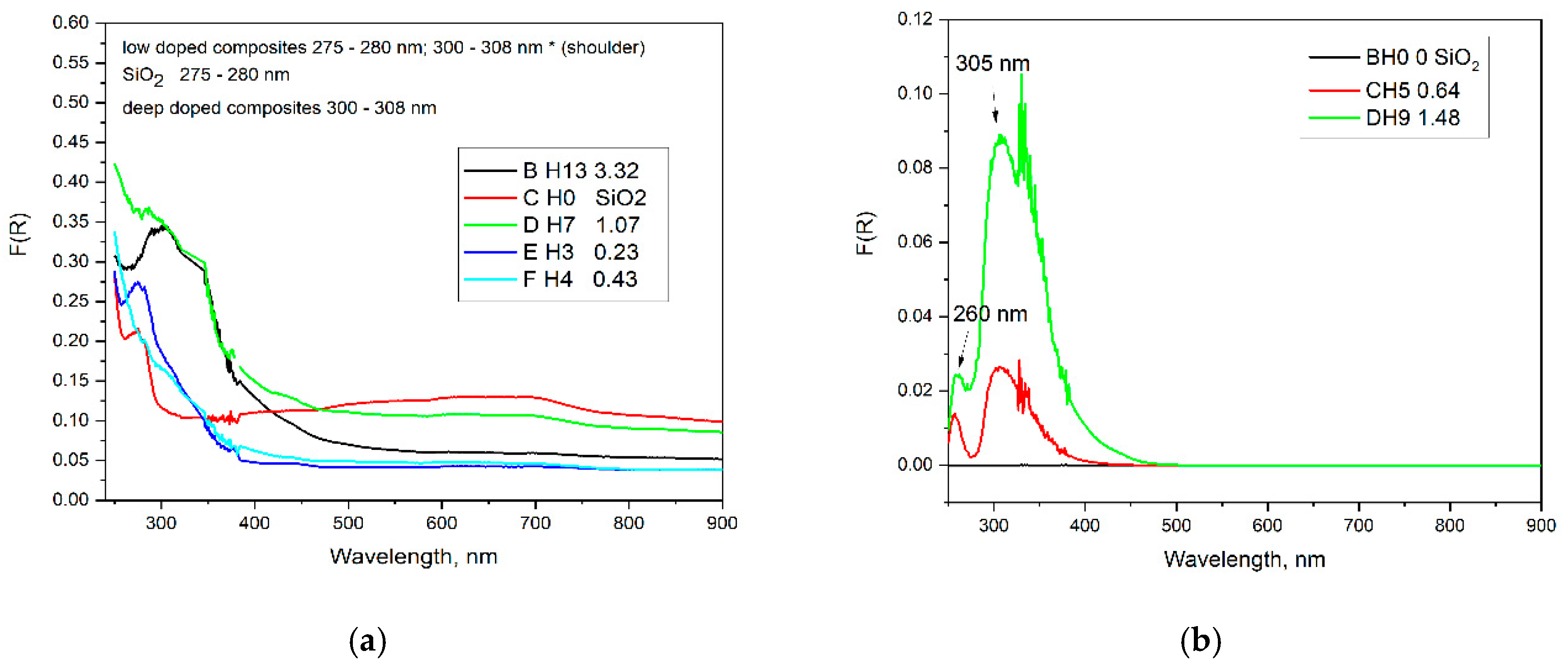
The optical properties of chitosan solutions here strongly depend on the pH of the medium (
Figure 4) and correlates well with data about the pK
b of the amino group of the chitosan. Above pH 6.5 the chitosan molecules become deprotonated and non-charged and the solutions turns cloudy because chitosan precipitates. Below pH 6.5 the amino groups are mostly protonated and the molecule is soluble. Two characteristic absorption bands are typically observed — a main peak around 280 nm and a shoulder near 300 nm — with the relative intensity of the 300 nm peak increasing as the pH decreases in agreement with theoretical explanations and measurements in [
14]. This behavior was confirmed by diffuse reflectance spectra and differential spectra, where the same absorption features are visible but with varying relative intensities. In the spectra of highly doped silica gels, the relative intensity of the 300 nm peak further increases with the doping level, as clearly seen in the differential spectra (
Figure 5b).
From a general spectroscopic perspective, the electronic transitions in chitosan can be classified as follows: transitions observed at 214–228 nm are assigned to π–π* or n–π* transitions associated with amine linkages and/or residual acetylated groups [
14]. Several studies report peaks or shoulders in the 250–283 nm region, sometimes depending on the molecular weight of the polymer or the formation of nanoparticles [
15]. Optical transitions in the 305–310 nm region are possibly related to π–π* transitions of conjugated groups or chitosan acetate particles [
16,
17,
18]. Additionally, chemical bonding between the amino groups of chitosan and the silanol groups of silica leads to changes in local electron density, which may induce slight shifts in the UV–Vis spectra within the 250–300 nm region (
Figure 5).
Owing to its absorption behavior in the near-UV region and high transparency in the visible range, chitosan shows potential as a natural, biocompatible optical UV filter. When incorporated into a silica sol–gel matrix, small but significant changes in the optical properties are observed, arising from interactions between the organic biopolymer and the inorganic network. The resulting silica–chitosan composites generally maintain good optical transparency, particularly at low chitosan content and when the polymer is homogeneously distributed within the matrix. Following the preparation scheme used here (
Figure 1), the most probable reasons for the observed changes in the optical spectra are protonation of chitosan due to the low pH of the solution and the formation of chitosan acetate.
The optical transparency of the composite gels is commonly evaluated through the transmittance coefficient and the refractive index, which depend on the SiO₂/chitosan ratio, the drying method, and the degree of condensation of the silica network. At low chitosan content (up to ~1 %), the materials maintain high transparency, whereas higher contents lead to increased light scattering due to the formation of microphases or polymer agglomerates. Furthermore, incorporation of chitosan into the inorganic network may influence the fluorescence characteristics of the system. As demonstrated in a recent study, chemical modification of chitosan with heterocyclic aromatic dyes resulted in composites with pronounced fluorescence even at low substitution levels, indicating that functionalization of chitosan can effectively impart strong fluorescent properties [
19]. Although chitosan itself exhibits only weak intrinsic fluorescence, the presence of functional groups with donor–acceptor character can facilitate local energy transfer within the composite network, potentially influencing the optical behavior of incorporated species. Thus, silica–chitosan gels provide a suitable platform for the incorporation of optically active species or nanoparticles, which can be stabilized within the matrix without significant loss of transparency and potentially enhance its luminescent properties [
18]. The composite samples obtained in this work, however, do not exhibit visible luminescence under UV excitation.
The diffuse reflectance spectra of the silica–chitosan composites, along with their differential spectra relative to pure silica, reveal characteristic features associated with the chitosan component. The spectral maxima are similar to those observed in chitosan solutions at different pH values but display distinct variations in relative intensity. These differences indicate structural modifications of the chitosan chains upon incorporation and immobilization within the silicate matrix, suggesting changes in the local electronic environment and interaction between the organic and inorganic phases. Overall, the optical behavior of chitosan and silica–chitosan composites reflects a delicate balance between molecular structure, pH-dependent structural conformation, and polymer–matrix interactions. Their high transparency and UV absorption capability render these hybrid systems promising candidates for optical UV filters, photonic coatings, and sensing applications, particularly for environmentally friendly and biocompatible materials.
2.4. Thermal Conductivity Measurements
The obtained data provide insight into how chitosan incorporation affects the heat transfer characteristics of the silica gel network, reflecting the interplay between polymer content, water retention, and microstructural organization within the hybrid gels. The obtained values of the thermal conductivity and thermal effusivity of the investigated composites are presented in
Table 1. From the data in (
Table 1) a mean specific heat capacities at constant pressure C
p = 510.47
58.6 J/kg·K for a mean powder density of 1.52
0.1 g / cm
3 is calculated. The deviation from the mean values is most probable connected with variations of the density and contact area in the doped composites. As a reference, the thermal conductivity and effusivity of pure chitosan powders are 0.067 W/m·K and 164.9 Wm
−2s
1/2K
−1, respectively [
20].
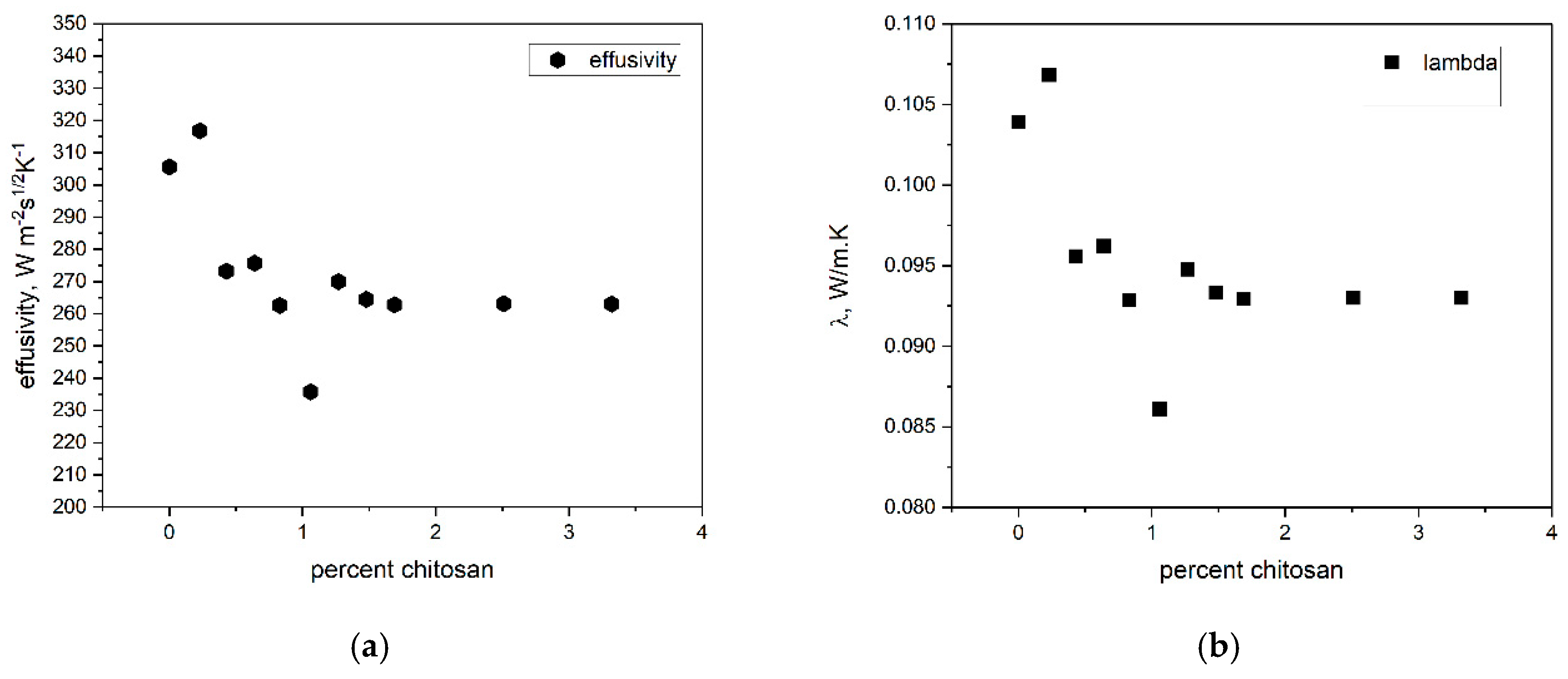
Based on the obtained measurements, a quantitative relationship was established between the thermal conductivity and thermal effusivity as a function of the chitosan content in the sol,
Figure 6a,b.
At low chitosan doping levels, a noticeable increase in thermal conductivity and effusivity is observed, most likely associated with a local increase in the density of the composite matrix (
Figure 2). As a reference, the thermal conductivity and effusivity of pure chitosan powders are 0.067 W/m·K and 164.9 W·m⁻²·s¹
/²·K⁻¹, respectively. The addition of chitosan to silica results in a moderate decrease in both thermal conductivity and effusivity with increasing polymer content. Consequently, the lower specific heat capacity (C
p) of the composites compared to pure silica (700–750 J/kg·K) can be explained. Similar observations, as the detected here changes in C
p of silica–chitosan composites relative to pure silica have been discussed in the literature and are attributed to chemical or structural modifications of the matrix [
21,
22]. It is visible, that chitosan addition decreases the thermal capacity, thermal conductivity and effusivity of silica. Variations in the thermal properties of these composites may indicate chemical or structural changes within the material [
23]. As expected, the thermal conductivity of the composites increases slightly with their density (
Table 1).
The thermal properties of the composites are found to depend on the chitosan content, resulting in a moderate decrease in thermal conductivity and effusivity compared to pure silica gels. Thermogravimetric (TGA) and differential thermal analyses (DTA) reveal that the thermal stability and decomposition behavior of the composites are strongly influenced by the amount of chitosan. In particular, the presence of chitosan introduces additional mass-loss steps between 200–300 °C, corresponding to biopolymer decomposition, while higher temperatures (500–700 °C) lead to further degradation and carbonization of residual organic components. These results indicate that chitosan not only modulates the mechanical and optical properties of the silica network but also plays a key role in defining its thermal response.
2.5. SEM Investigations and X-Ray Diffraction
SEM micrographs (
Figure 7) show the characteristic morphology of amorphous silicate gels. The presence of chitosan is not clearly discernible due to its low concentration. The images reveal a porous three-dimensional network formed by interconnected particles, with some regions exhibiting variations in density, most probable responsible for variations of the thermal effusivity of the composites. These observations are consistent with the typical gelation behavior of sol-gel-derived silica composites and corroborate the morphological modifications induced by chitosan incorporation.
An influence of chitosan included in the silicate matrix on its morphology was also observed. The incorporation of chitosan leads to a noticeable modification of the morphology, manifested as densification of the gel network (
Figure 1). These effects are most likely associated with hydrogen bonding and electrostatic interactions between the amino groups of chitosan and the silanol groups of SiO₂. Such structural rearrangements are expected to enhance the mechanical stability and overall integrity of the composites.
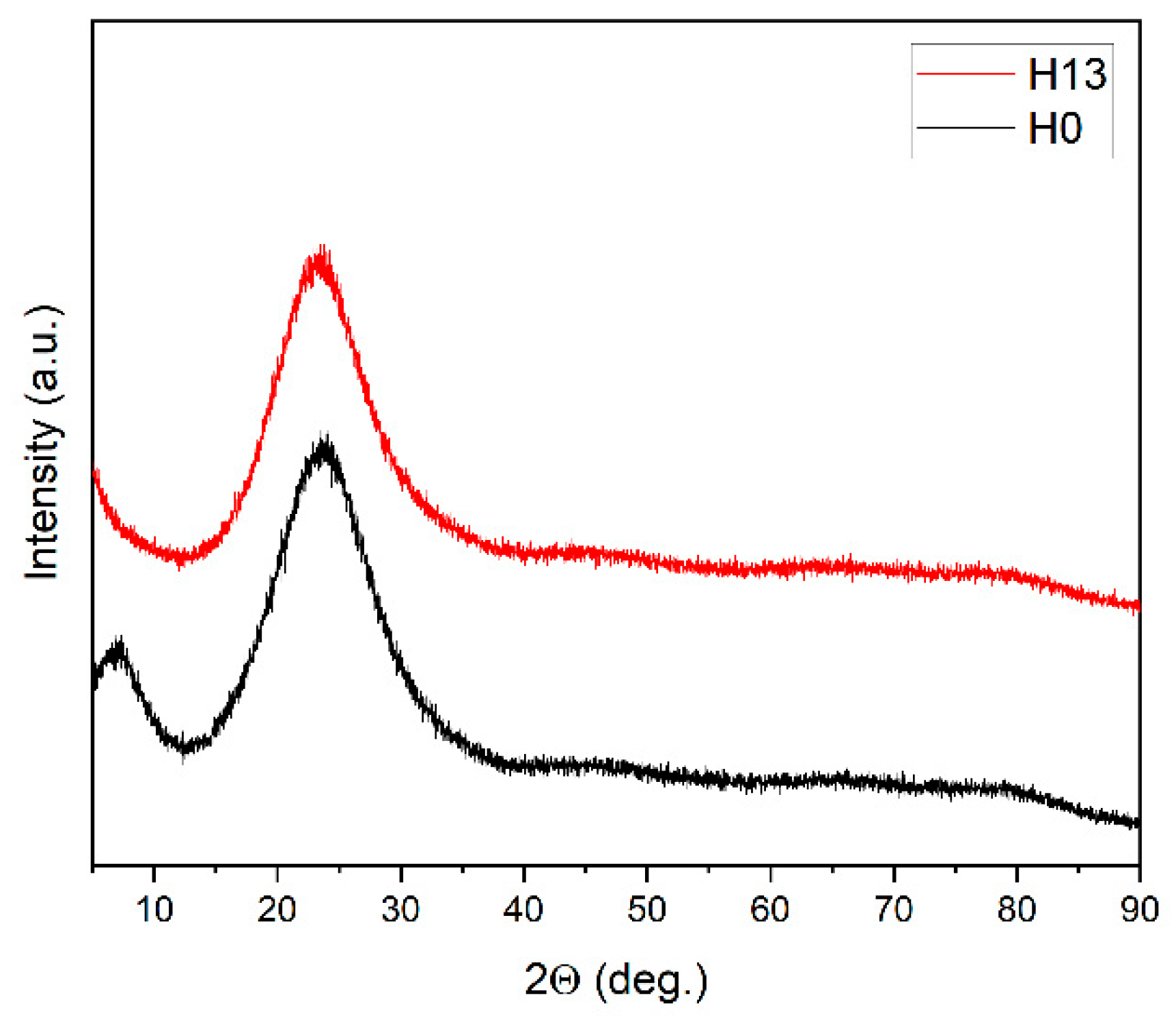
The SEM observations are supported by powder X-ray diffractograms, which demonstrated the amorphous state of the investigated composites (S1). Peaks corresponding to crystalline chitosan are not detected [
24]. Both SEM and X-ray analyses confirmed that the chitosan dopant is homogeneously distributed within the silica matrix.
Representative X-ray diffraction data of composites with a different doping level are shown on
Figure 8.
The figure shows that the amorphous character of the composites is preserved even at relatively high doping levels. For comparison [
7], in silica nanocomposites doped with 1% Eu(phen)₂(NO₃)₃ or Tb(phen)₂(NO₃)₃, the nanocrystalline nature of the dopant is clearly visible, and the appearance of a defective crystal structure associated with the interaction between the dopant and the silicate matrix has been discussed.
The reason for the observed amorphous structure in the present study is the easy amorphization of chitosan, evident from its low glass-transition temperature, Tg = 130–150 °C [
25]. Changes in the conditions of the sol–gel synthesis—such as composition, temperature, and water content—can further facilitate the amorphization of chitosan and thus affect the homogeneity of the resulting composites without altering their functionality. More about the glass-transition temperature of chitosan-related systems, depending on doping and chemical composition can be found in [
26,
27,
28], while in [
29] a review of the thermodynamics and kinetics of glass crystallization is given.