Submitted:
08 December 2025
Posted:
09 December 2025
You are already at the latest version
Abstract
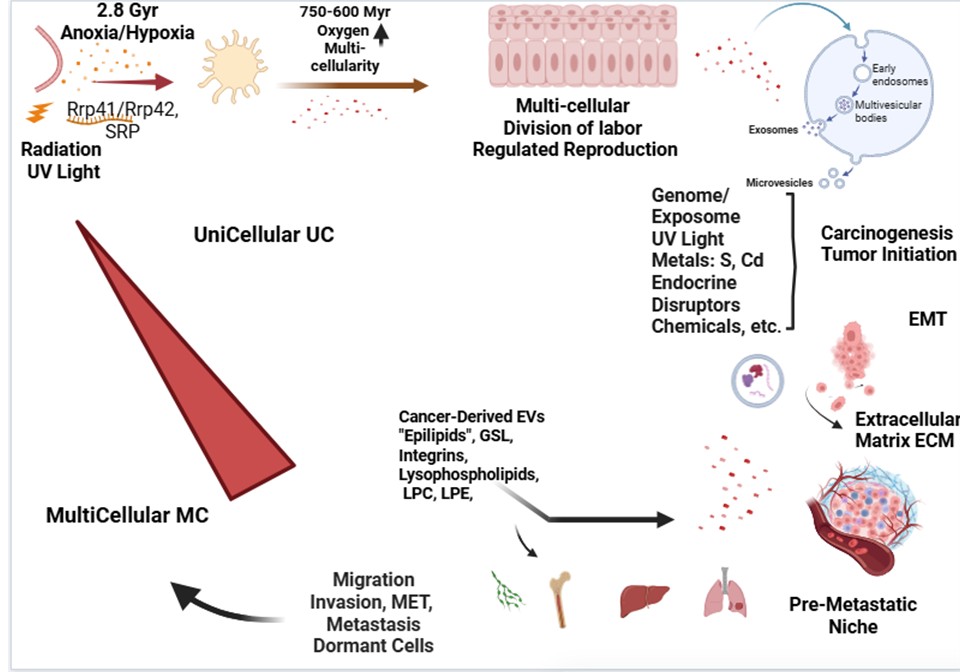
Keywords:
1. Introduction
2. The Evolutionary Context of EVs and Lipid Signaling
2.1. Cholesterol
2.2. Sphingolipids
2.3. Phospholipids
2.4. Glycerolipids
2.5. Fatty Acids
3. Mechanisms of Lipid-Mediated Communications
3.1. Direct Transfer of Lipids
3.2. Activation of Lipid Receptors
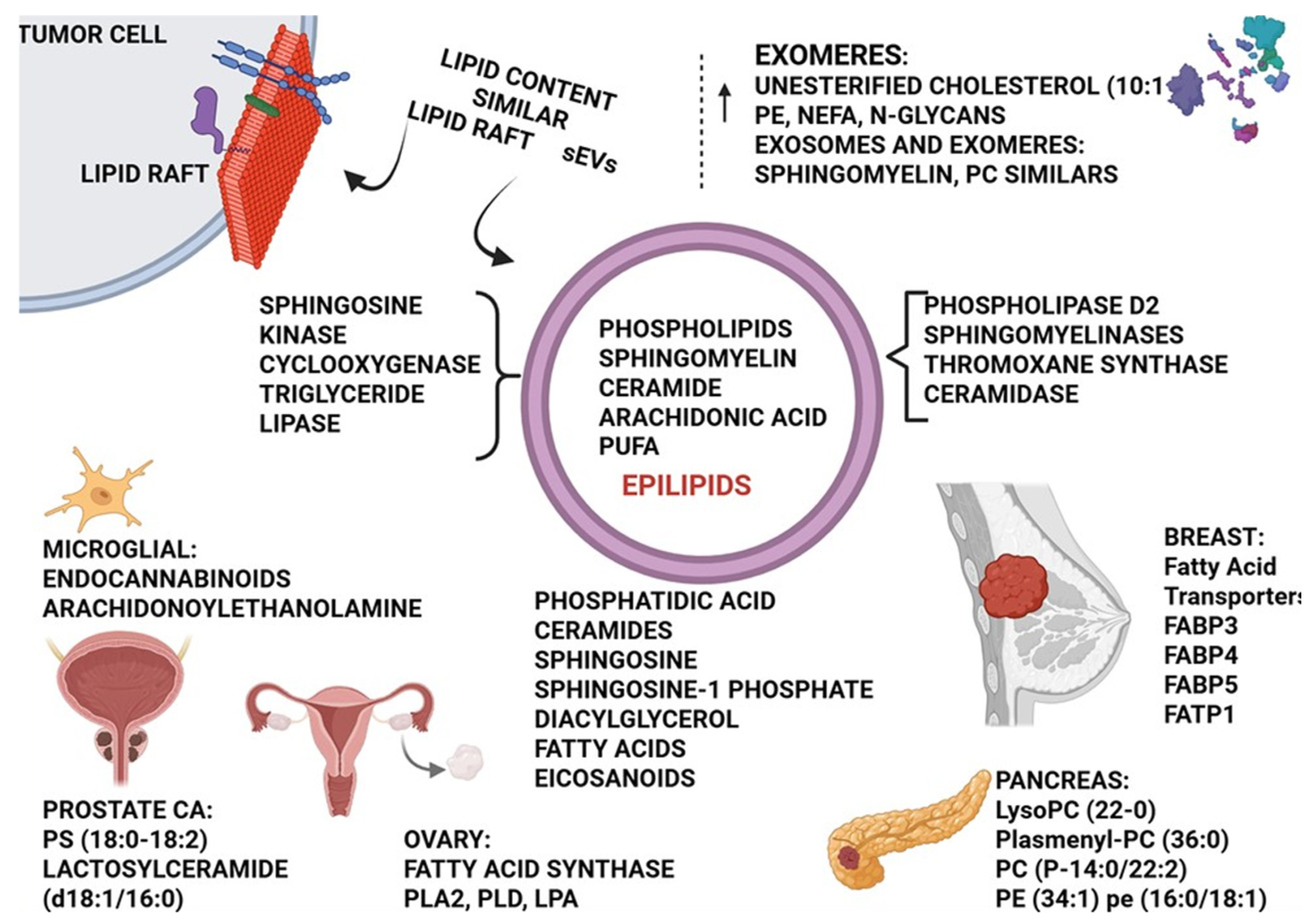
3.3. Epilipids
4. Lipid-Mediated Communication in Cancer Progression and Metastasis
4.1. Tumor Microenvironment Modulation
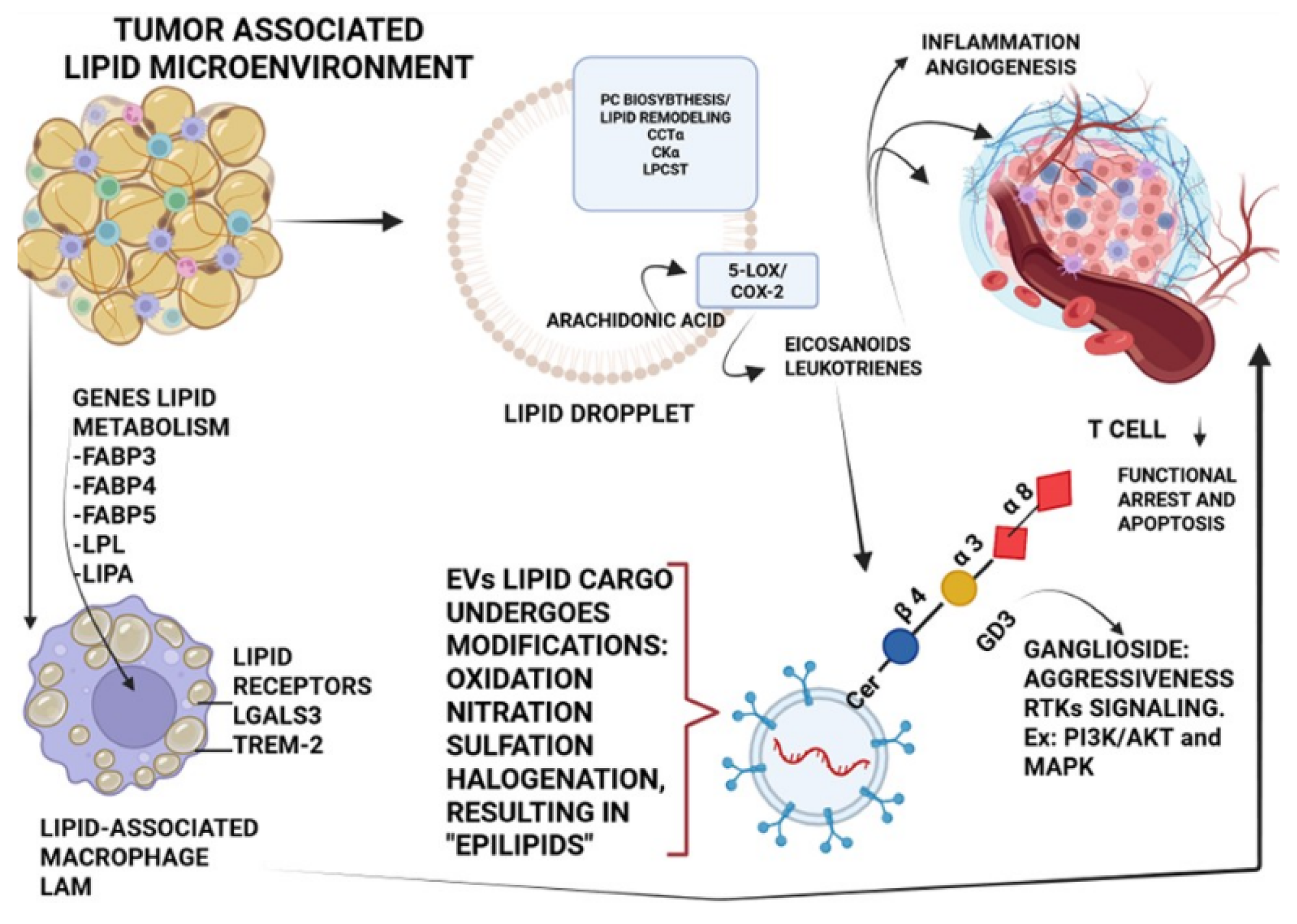
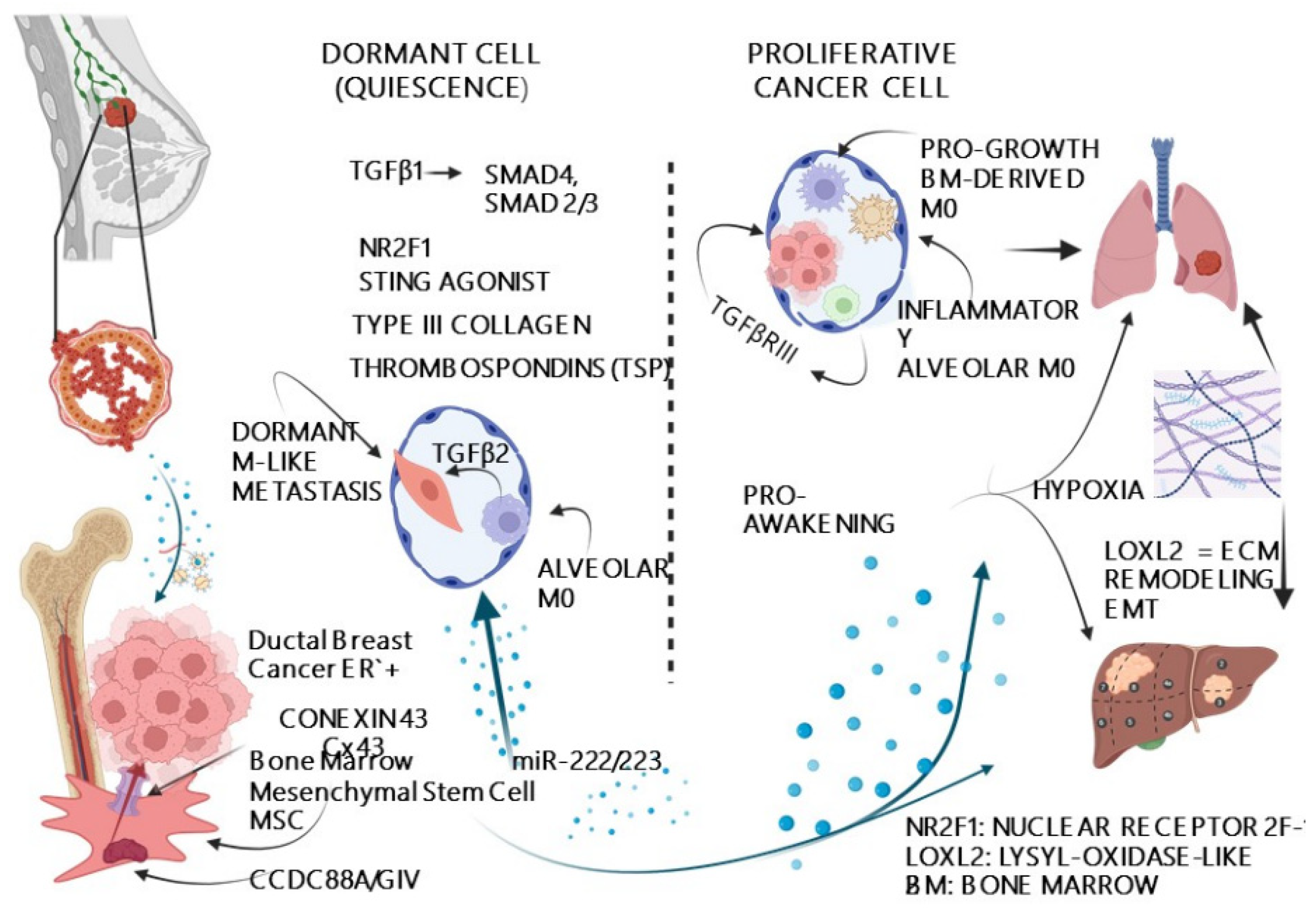
4.2. Metastatic Niche Formation and Organotropism
4.3. Lipid Rafts and Integrins
4.4. Signaling Molecules
4.5. Cancer Cell Homing
4.5.1. Chemokine Signaling
- Tumor progression: metastasis, proliferation, and cancer cell-induced angiogenesis
- Survival and resistance: anti-apoptotic effects on monocytes, cell survival, drug resistance
- Signaling pathways: AKT kinase activation, oncogenic signaling
4.5.2. Adhesion Molecules
5.5.2. Immune Modulation
5.6. Drug Resistance
5.7. Immune Evasion
5.8. Cell Dormancy
5.9. Signaling Pathway Modulation
5.10. Metabolic Alterations
5.11. Niche Interactions
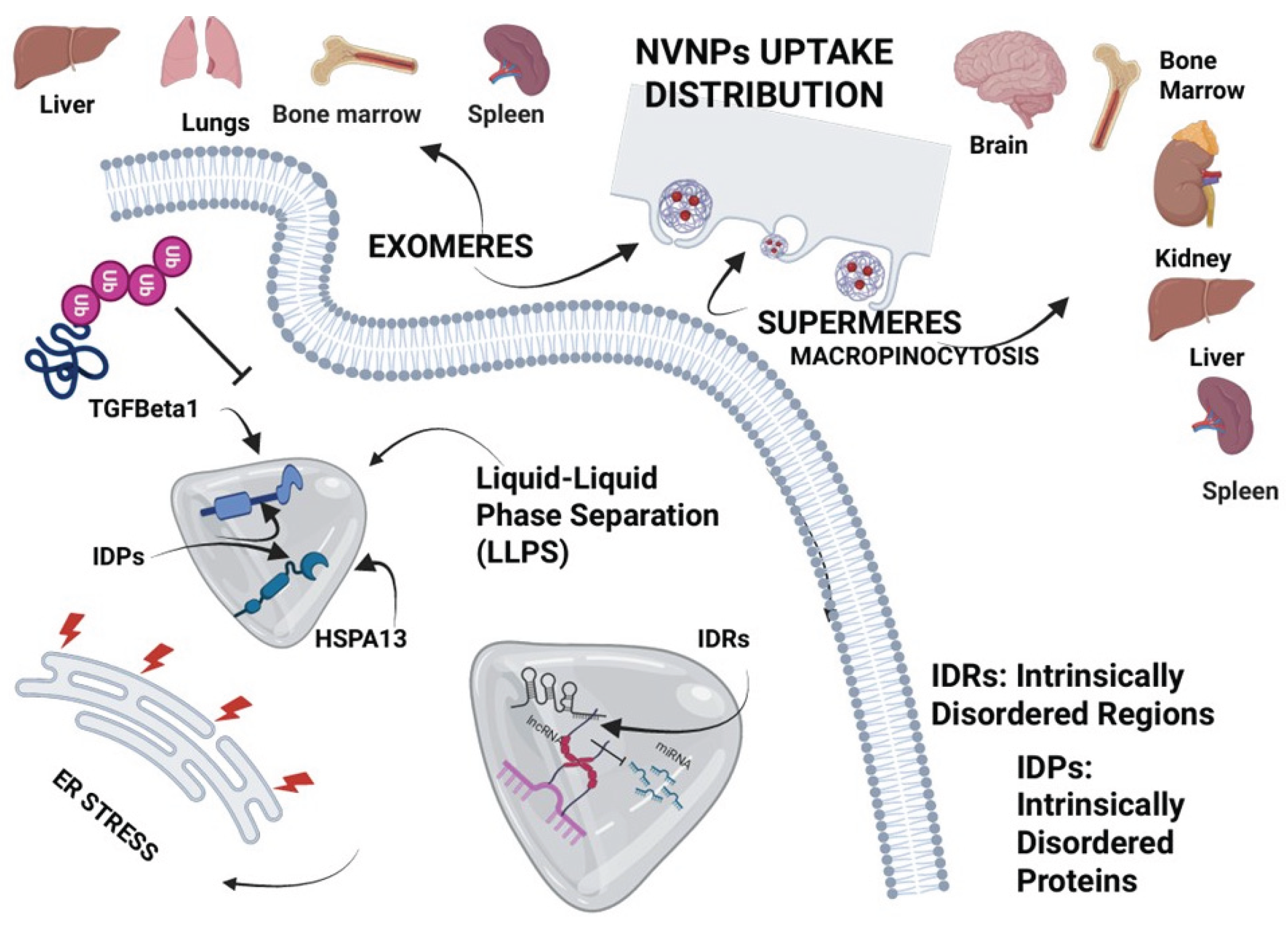
6. Post-Secretion Processes and Modification of Extracellular Vesicles (PSPMs)
6.1. Exomeres and Supermeres
7. Reverse Microevolutionary Process and Ancestral Nature of EVs
8. Discussion
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Baghban, R.; Roshangar, L.; Jahanban-Esfahlan, R.; Seidi, K.; Ebrahimi-Kalan, A.; Jaymand, M.; Kolahian, S.; Javaheri, T.; Zare, P. Tumor Microenvironment Complexity and Therapeutic Implications at a Glance. Cell Commun Signal 2020, 18, 59. [Google Scholar] [CrossRef]
- El-Tanani, M.; Rabbani, S.A.; Babiker, R.; Rangraze, I.; Kapre, S.; Palakurthi, S.S.; Alnuqaydan, A.M.; Aljabali, A.A.; Rizzo, M.; El-Tanani, Y.; et al. Unraveling the Tumor Microenvironment: Insights into Cancer Metastasis and Therapeutic Strategies. Cancer Letters 2024, 591, 216894. [Google Scholar] [CrossRef]
- Kalluri, R.; McAndrews, K.M. The Role of Extracellular Vesicles in Cancer. Cell 2023, 186, 1610–1626. [Google Scholar] [CrossRef]
- Clancy, J.W.; Boomgarden, A.C.; D’Souza-Schorey, C. Profiling and Promise of Supermeres. Nat Cell Biol 2021, 23, 1217–1219. [Google Scholar] [CrossRef]
- Wortzel, I.; Dror, S.; Kenific, C.M.; Lyden, D. Exosome-Mediated Metastasis: Communication from a Distance. Developmental Cell 2019, 49, 347–360. [Google Scholar] [CrossRef]
- Shirani, N.; Abdi, N.; Chehelgerdi, M.; Yaghoobi, H.; Chehelgerdi, M. Investigating the Role of Exosomal Long Non-Coding RNAs in Drug Resistance within Female Reproductive System Cancers. Front. Cell Dev. Biol. 2025, 13, 1485422. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, W.; Wang, Y.; Wang, H.; Liu, S. Extracellular Vesicle-Mediated Crosstalk between Pancreatic Cancer and Stromal Cells in the Tumor Microenvironment. J Nanobiotechnol 2022, 20, 208. [Google Scholar] [CrossRef]
- Fyfe, J.; Casari, I.; Manfredi, M.; Falasca, M. Role of Lipid Signalling in Extracellular Vesicles-Mediated Cell-to-Cell Communication. Cytokine & Growth Factor Reviews 2023, 73, 20–26. [Google Scholar] [CrossRef]
- Nishida-Aoki, N.; Izumi, Y.; Takeda, H.; Takahashi, M.; Ochiya, T.; Bamba, T. Lipidomic Analysis of Cells and Extracellular Vesicles from High- and Low-Metastatic Triple-Negative Breast Cancer. Metabolites 2020, 10, 67. [Google Scholar] [CrossRef]
- Brzozowski, J.S.; Jankowski, H.; Bond, D.R.; McCague, S.B.; Munro, B.R.; Predebon, M.J.; Scarlett, C.J.; Skelding, K.A.; Weidenhofer, J. Lipidomic Profiling of Extracellular Vesicles Derived from Prostate and Prostate Cancer Cell Lines—Lipids Health Dis. 2018, 17, 211. [Google Scholar] [CrossRef]
- Trigos, A.S.; Pearson, R.B.; Papenfuss, A.T.; Goode, D.L. How the Evolution of Multicellularity Set the Stage for Cancer. Br J Cancer 2018, 118, 145–152. [Google Scholar] [CrossRef]
- Bussey, K.J.; Cisneros, L.H.; Lineweaver, C.H.; Davies, P.C.W. Ancestral Gene Regulatory Networks Drive Cancer. Proc. Natl. Acad. Sci. U.S.A 2017, 114, 6160–6162. [Google Scholar] [CrossRef]
- Trigos, A.S.; Bongiovanni, F.; Zhang, Y.; Zethoven, M.; Tothill, R.; Pearson, R.; Papenfuss, A.T.; Goode, D.L. Disruption of Metazoan Gene Regulatory Networks in Cancer Alters the Balance of Co-Expression between Genes of Unicellular and Multicellular Origins. Genome Biol 2024, 25, 110. [Google Scholar] [CrossRef]
- Jordan, S.F.; Nee, E.; Lane, N. Isoprenoids Enhance the Stability of Fatty Acid Membranes at the Emergence of Life Potentially Leading to an Early Lipid Divide. Interface Focus. 2019, 9, 20190067. [Google Scholar] [CrossRef]
- Albani, A.E.; Bengtson, S.; Canfield, D.E.; Bekker, A.; Macchiarelli, R.; Mazurier, A.; Hammarlund, E.U.; Boulvais, P.; Dupuy, J.-J.; Fontaine, C.; et al. Large Colonial Organisms with Coordinated Growth in Oxygenated Environments 2.1 Gyr Ago. Nature 2010, 466, 100–104. [Google Scholar] [CrossRef] [PubMed]
- Feller, S.M. Early Beginnings - the Emergence of Complex Signaling Systems and Cell-to-Cell Communication. Cell Commun Signal 2010, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Ros-Rocher, N.; Pérez-Posada, A.; Leger, M.M.; Ruiz-Trillo, I. The Origin of Animals: An Ancestral Reconstruction of the Unicellular-to-Multicellular Transition. Open Biol. 2021, 11, 200359. [Google Scholar] [CrossRef]
- Brunet, T.; King, N. The Origin of Animal Multicellularity and Cell Differentiation. Developmental Cell 2017, 43, 124–140. [Google Scholar] [CrossRef] [PubMed]
- Sebé-Pedrós, A.; Degnan, B.M.; Ruiz-Trillo, I. The Origin of Metazoa: A Unicellular Perspective. Nat Rev Genet 2017, 18, 498–512. [Google Scholar] [CrossRef]
- Crawford, M.A.; Schmidt, W.F.; Broadhurst, C.L.; Wang, Y. Lipids in the Origin of Intracellular Detail and Speciation in the Cambrian Epoch and the Significance of the Last Double Bond of Docosahexaenoic Acid in Cell Signaling. Prostaglandins, Leukotrienes and Essential Fatty Acids 2021, 166, 102230. [Google Scholar] [CrossRef]
- Bader, J.; Brigger, F.; Leroux, J.-C. Extracellular Vesicles versus Lipid Nanoparticles for the Delivery of Nucleic Acids. Advanced Drug Delivery Reviews 2024, 215, 115461. [Google Scholar] [CrossRef]
- Espiau-Romera, P.; Gordo-Ortiz, A.; Ortiz-de-Solórzano, I.; Sancho, P. Metabolic Features of Tumor-Derived Extracellular Vesicles: Challenges and Opportunities. Extracell Vesicles Circ Nucleic Acids 2024, 5, 555–570. [Google Scholar] [CrossRef]
- Akoto, T.; Saini, S. Role of Exosomes in Prostate Cancer Metastasis. IJMS 2021, 22, 3528. [Google Scholar] [CrossRef] [PubMed]
- Pfrieger, F.W.; Vitale, N. Thematic Review Series: Exosomes and Microvesicles: Lipids as Key Components of Their Biogenesis and Functions, Cholesterol and the Journey of Extracellular Vesicles. Journal of Lipid Research 2018, 59, 2255–2261. [Google Scholar] [CrossRef]
- Gomez-Larrauri, A.; Larrea-Sebal, A.; Martín, C.; Gomez-Muñoz, A. The Critical Roles of Bioactive Sphingolipids in Inflammation. Journal of Biological Chemistry 2025, 301, 110475. [Google Scholar] [CrossRef]
- Yang, K.; Fu, W.; Deng, M.; Li, X.; Wu, M.; Wang, Y. The Sphingolipids Change in Exosomes from Cancer Patients and Association between Exosome Release and Sphingolipids Level Based on a Pseudotargeted Lipidomics Method. Analytica Chimica Acta 2024, 1305, 342527. [Google Scholar] [CrossRef]
- Stace, C.; Ktistakis, N. Phosphatidic Acid- and Phosphatidylserine-Binding Proteins. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 2006, 1761, 913–926. [Google Scholar] [CrossRef]
- Morita, S.; Ikeda, Y. Regulation of Membrane Phospholipid Biosynthesis in Mammalian Cells. Biochemical Pharmacology 2022, 206, 115296. [Google Scholar] [CrossRef] [PubMed]
- Cooke, M.; Kazanietz, M.G. Overarching Roles of Diacylglycerol Signaling in Cancer Development and Antitumor Immunity. Sci. Signal. 2022, 15, eabo0264. [Google Scholar] [CrossRef] [PubMed]
- Sundram, V.; Chauhan, S.C.; Jaggi, M. Emerging Roles of Protein Kinase D1 in Cancer. Molecular Cancer Research 2011, 9, 985–996. [Google Scholar] [CrossRef]
- Durand, N.; Borges, S.; Storz, P. Functional and Therapeutic Significance of Protein Kinase D Enzymes in Invasive Breast Cancer. Cell. Mol. Life Sci. 2015, 72, 4369–4382. [Google Scholar] [CrossRef]
- Zhang, Q.; Higginbotham, J.N.; Jeppesen, D.K.; Yang, Y.-P.; Li, W.; McKinley, E.T.; Graves-Deal, R.; Ping, J.; Britain, C.M.; Dorsett, K.A.; et al. Transfer of Functional Cargo in Exomeres. Cell Reports 2019, 27, 940–954.e6. [Google Scholar] [CrossRef]
- Record, M.; Silvente-Poirot, S.; Poirot, M.; Wakelam, MichaelJ.O. Extracellular Vesicles: Lipids as Key Components of Their Biogenesis and Functions. Journal of Lipid Research 2018, 59, 1316–1324. [Google Scholar] [CrossRef]
- Ghadami, S.; Dellinger, K. The Lipid Composition of Extracellular Vesicles: Applications in Diagnostics and Therapeutic Delivery. Front. Mol. Biosci. 2023, 10, 1198044. [Google Scholar] [CrossRef]
- Ribovski, L.; Joshi, B.S.; Gao, J.; Zuhorn, I.S. Breaking Free: Endocytosis and Endosomal Escape of Extracellular Vesicles. Extracell Vesicles Circ Nucleic Acids 2023, 4, 283–305. [Google Scholar] [CrossRef]
- Ma, Y.; Brocchini, S.; Williams, G.R. Extracellular Vesicle-Embedded Materials. Journal of Controlled Release 2023, 361, 280–296. [Google Scholar] [CrossRef]
- Kwok, Z.H.; Wang, C.; Jin, Y. Extracellular Vesicle Transportation and Uptake by Recipient Cells: A Critical Process to Regulate Human Diseases. Processes 2021, 9, 273. [Google Scholar] [CrossRef]
- Ragni, E. Extracellular Vesicles: Recent Advances and Perspectives. Front. Biosci. (Landmark Ed) 2025, 30, 36405. [Google Scholar] [CrossRef]
- Clement, E.; Lazar, I.; Attané, C.; Carrié, L.; Dauvillier, S.; Ducoux--Petit, M.; Esteve, D.; Menneteau, T.; Moutahir, M.; Le Gonidec, S.; et al. Adipocyte Extracellular Vesicles Carry Enzymes and Fatty Acids That Stimulate Mitochondrial Metabolism and Remodeling in Tumor Cells. The EMBO Journal 2020, 39, e102525. [Google Scholar] [CrossRef]
- Wang, Y.; Li, X.; Liu, D.; Wang, Z.; Xia, J.; Wang, L.; Zhang, X. Research Progress on the Role of Adipocyte Exosomes in Cancer Progression. OR 2024, 32, 1649–1660. [Google Scholar] [CrossRef]
- Lazar, I.; Clement, E.; Attane, C.; Muller, C.; Nieto, L. A New Role for Extracellular Vesicles: How Small Vesicles Can Feed Tumors’ Big Appetite. Journal of Lipid Research 2018, 59, 1793–1804. [Google Scholar] [CrossRef]
- Wang, M.; Wang, W.; Chopp, M.; Zhang, Z.G.; Zhang, Y. Therapeutic and Diagnostic Potential of Extracellular Vesicle (EV)-Mediated Intercellular Transfer of Mitochondria and Mitochondrial Components. J Cereb Blood Flow Metab 2025, 0271678X251338971. [Google Scholar] [CrossRef]
- Penkov, S.; Fedorova, M. Membrane Epilipidome—Lipid Modifications, Their Dynamics, and Functional Significance. Cold Spring Harb Perspect Biol 2024, 16, a041417. [Google Scholar] [CrossRef]
- Wölk, M.; Prabutzki, P.; Fedorova, M. Analytical Toolbox to Unlock the Diversity of Oxidized Lipids. Acc. Chem. Res. 2023, 56, 835–845. [Google Scholar] [CrossRef]
- Duché, G.; Sanderson, J.M. The Chemical Reactivity of Membrane Lipids. Chem. Rev. 2024, 124, 3284–3330. [Google Scholar] [CrossRef]
- Li, J.; Peng, J.; Wang, J.; Chen, Z. The Dual Role of Exosomes in the Tumor Microenvironment: From Pro--Tumorigenic Signaling to Immune Modulation. Med Research 2025, 1, 257–284. [Google Scholar] [CrossRef]
- Hasnat, M.A.; Ohmi, Y.; Yesmin, F.; Kaneko, K.; Kambe, M.; Kitaura, Y.; Ito, T.; Imao, Y.; Kano, K.; Mishiro-Sato, E.; et al. Action Mechanisms of Exosomes Derived from GD3/GD2-Positive Glioma Cells in the Regulation of Phenotypes and Intracellular Signaling: Roles of Integrins. IJMS 2024, 25, 12752. [Google Scholar] [CrossRef]
- Shao, N.; Qiu, H.; Liu, J.; Xiao, D.; Zhao, J.; Chen, C.; Wan, J.; Guo, M.; Liang, G.; Zhao, X.; et al. Targeting Lipid Metabolism of Macrophages: A New Strategy for Tumor Therapy. Journal of Advanced Research 2025, 68, 99–114. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; He, Y.; Zhao, P.; Hu, Y.; Tao, J.; Chen, J.; Huang, Y. Targeting Lipid Metabolism to Overcome EMT-Associated Drug Resistance via Integrin Β3/FAK Pathway and Tumor-Associated Macrophage Repolarization Using Legumain-Activatable Delivery. Theranostics 2019, 9, 265–278. [Google Scholar] [CrossRef]
- Hoshino, A.; Costa-Silva, B.; Shen, T.-L.; Rodrigues, G.; Hashimoto, A.; Tesic Mark, M.; Molina, H.; Kohsaka, S.; Di Giannatale, A.; Ceder, S.; et al. Tumour Exosome Integrins Determine Organotropic Metastasis. Nature 2015, 527, 329–335. [Google Scholar] [CrossRef]
- Li, B.; Qin, Y.; Yu, X.; Xu, X.; Yu, W. Lipid Raft Involvement in Signal Transduction in Cancer Cell Survival, Cell Death and Metastasis. Cell Proliferation 2022, 55, e13167. [Google Scholar] [CrossRef]
- Lietha, D.; Izard, T. Roles of Membrane Domains in Integrin-Mediated Cell Adhesion. IJMS 2020, 21, 5531. [Google Scholar] [CrossRef]
- Kudo, K.; Miki, Y.; Carreras, J.; Nakayama, S.; Nakamoto, Y.; Ito, M.; Nagashima, E.; Yamamoto, K.; Higuchi, H.; Morita, S.; et al. Secreted Phospholipase A2 Modifies Extracellular Vesicles and Accelerates B Cell Lymphoma. Cell Metabolism 2022, 34, 615–633.e8. [Google Scholar] [CrossRef]
- Haraszti, R.A.; Didiot, M.; Sapp, E.; Leszyk, J.; Shaffer, S.A.; Rockwell, H.E.; Gao, F.; Narain, N.R.; DiFiglia, M.; Kiebish, M.A.; et al. High--resolution Proteomic and Lipidomic Analysis of Exosomes and Microvesicles from Different Cell Sources. J of Extracellular Vesicle 2016, 5, 32570. [Google Scholar] [CrossRef]
- Sarvaiya, P.J.; Guo, D.; Ulasov, I.; Gabikian, P.; Lesniak, M.S. Chemokines in Tumor Progression and Metastasis. Oncotarget 2013, 4, 2171–2185. [Google Scholar] [CrossRef]
- Wang, J.; Qu, C.; Xiao, P.; Liu, S.; Sun, J.-P.; Ping, Y.-Q. Progress in Structure-Based Drug Development Targeting Chemokine Receptors. Front. Pharmacol. 2025, 16, 1603950. [Google Scholar] [CrossRef]
- Marcuzzi, E.; Angioni, R.; Molon, B.; Calì, B. Chemokines and Chemokine Receptors: Orchestrating Tumor Metastasization. IJMS 2018, 20, 96. [Google Scholar] [CrossRef]
- Tvaroška, I.; Selvaraj, C.; Koča, J. Selectins—The Two Dr. Jekyll and Mr. Hyde Faces of Adhesion Molecules—A Review. Molecules 2020, 25, 2835. [Google Scholar] [CrossRef]
- Hugonnet, M.; Singh, P.; Haas, Q.; Von Gunten, S. The Distinct Roles of Sialyltransferases in Cancer Biology and Onco-Immunology. Front. Immunol. 2021, 12, 799861. [Google Scholar] [CrossRef]
- Pietrobono, S.; Stecca, B. Aberrant Sialylation in Cancer: Biomarker and Potential Target for Therapeutic Intervention? Cancers 2021, 13, 2014. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Xu, X.; Zhou, X.; Feng, H.; Wang, R.; Yang, Y.; Li, J.; Fan, N.; Jiang, Y.; Li, X.; et al. Sialylation on Vesicular Integrin Β1 Determined Endocytic Entry of Small Extracellular Vesicles into Recipient Cells. Cell Mol Biol Lett 2024, 29, 46. [Google Scholar] [CrossRef]
- Burčík, D.; Macko, J.; Podrojková, N.; Demeterová, J.; Stano, M.; Oriňak, A. Role of Cell Adhesion in Cancer Metastasis Formation: A Review. ACS Omega 2025, 10, 5193–5213. [Google Scholar] [CrossRef]
- Zhao, L.; Ma, X.; Yu, J. Exosomes and Organ-Specific Metastasis. Molecular Therapy - Methods & Clinical Development 2021, 22, 133–147. [Google Scholar] [CrossRef]
- Su, C.; Li, J.; Zhang, L.; Wang, H.; Wang, F.; Tao, Y.; Wang, Y.; Guo, Q.; Li, J.; Liu, Y.; et al. The Biological Functions and Clinical Applications of Integrins in Cancers. Front. Pharmacol. 2020, 11, 579068. [Google Scholar] [CrossRef]
- Cong, F.; Huang, J.; Wu, C.; Zhong, H.; Qiu, G.; Luo, T.; Tang, W. Integrin A6 and Integrin Β4 in Exosomes Promote Lung Metastasis of Colorectal Cancer. Journal of Cancer Research and Therapeutics 2024, 20, 2082–2093. [Google Scholar] [CrossRef]
- Nannan, L.; Oudart, J.-B.; Monboisse, J.C.; Ramont, L.; Brassart-Pasco, S.; Brassart, B. Extracellular Vesicle-Dependent Cross-Talk in Cancer—Focus on Pancreatic Cancer. Front. Oncol. 2020, 10, 1456. [Google Scholar] [CrossRef]
- Sung, B.H.; Emmanuel, M.; Gari, M.K.; Guerrero, J.F.; Virumbrales-Muñoz, M.; Inman, D.; Krystofiak, E.; Rapraeger, A.C.; Ponik, S.M.; Weaver, A.M. Exosomes Are Specialized Vehicles to Induce Fibronectin Assembly 2025.
- Hirosawa, K.M.; Sato, Y.; Kasai, R.S.; Yamaguchi, E.; Komura, N.; Ando, H.; Hoshino, A.; Yokota, Y.; Suzuki, K.G.N. Uptake of Small Extracellular Vesicles by Recipient Cells Is Facilitated by Paracrine Adhesion Signaling. Nat Commun 2025, 16, 2419. [Google Scholar] [CrossRef]
- Ciobanasu, C.; Le Clainche, C. Integrins from Extracellular Vesicles as Players in Tumor Microenvironment and Metastasis. Cancer Metastasis Rev 2025, 44, 68. [Google Scholar] [CrossRef]
- Shen, Y.-Q.; Sun, L.; Wang, S.-M.; Zheng, X.-Y.; Xu, R. Exosomal Integrins in Tumor Progression, Treatment and Clinical Prediction (Review). Int J Oncol 2024, 65, 118. [Google Scholar] [CrossRef]
- Burčík, D.; Macko, J.; Podrojková, N.; Demeterová, J.; Stano, M.; Oriňak, A. Role of Cell Adhesion in Cancer Metastasis Formation: A Review. ACS Omega 2025, 10, 5193–5213. [Google Scholar] [CrossRef] [PubMed]
- Nidhi, G.; Yadav, V.; Singh, T.; Sharma, D.; Bohot, M.; Satapathy, S.R. Beyond Boundaries: Exploring the Role of Extracellular Vesicles in Organ-Specific Metastasis in Solid Tumors. Front. Immunol. 2025, 16, 1593834. [Google Scholar] [CrossRef] [PubMed]
- Kuang, L.; Wu, L.; Li, Y. Extracellular Vesicles in Tumor Immunity: Mechanisms and Novel Insights. Mol Cancer 2025, 24, 45. [Google Scholar] [CrossRef]
- Jurj, A.; Paul, D.; Calin, G.A. Extracellular Vesicles in Cancer: From Isolation and Characterization to Metastasis, Drug Resistance, and Clinical Applications. BMC Cancer 2025, 25, 1154. [Google Scholar] [CrossRef]
- Bucci-Muñoz, M.; Gola, A.M.; Rigalli, J.P.; Ceballos, M.P.; Ruiz, M.L. Extracellular Vesicles and Cancer Multidrug Resistance: Undesirable Intercellular Messengers? Life 2023, 13, 1633. [Google Scholar] [CrossRef]
- Xavier, C.P.R.; Belisario, D.C.; Rebelo, R.; Assaraf, Y.G.; Giovannetti, E.; Kopecka, J.; Vasconcelos, M.H. The Role of Extracellular Vesicles in the Transfer of Drug Resistance Competences to Cancer Cells. Drug Resistance Updates 2022, 62, 100833. [Google Scholar] [CrossRef]
- Musi, A.; Bongiovanni, L. Extracellular Vesicles in Cancer Drug Resistance: Implications on Melanoma Therapy. Cancers 2023, 15, 1074. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, J.; Liu, S.; Yu, L.; Liu, S.; Li, M.; Jin, F. Extracellular Vesicles in Oral Squamous Cell Carcinoma: Current Progress and Future Prospect. Front. Bioeng. Biotechnol. 2023, 11, 1149662. [Google Scholar] [CrossRef]
- Chen, S.; Iannotta, D.; O’Mara, M.L.; Goncalves, J.P.; Wolfram, J. Extracellular Vesicle Lipids in Cancer Immunoevasion. Trends in Cancer 2023, 9, 883–886. [Google Scholar] [CrossRef]
- Gupta, P.; Kadamberi, I.P.; Mittal, S.; Tsaih, S.; George, J.; Kumar, S.; Vijayan, D.K.; Geethadevi, A.; Parashar, D.; Topchyan, P.; et al. Tumor Derived Extracellular Vesicles Drive T Cell Exhaustion in Tumor Microenvironment through Sphingosine Mediated Signaling and Impacting Immunotherapy Outcomes in Ovarian Cancer. Advanced Science 2022, 9, 2104452. [Google Scholar] [CrossRef]
- Shenoy, G.N.; Loyall, J.; Berenson, C.S.; Kelleher, R.J.; Iyer, V.; Balu-Iyer, S.V.; Odunsi, K.; Bankert, R.B. Sialic Acid–Dependent Inhibition of T Cells by Exosomal Ganglioside GD3 in Ovarian Tumor Microenvironments. The Journal of Immunology 2018, 201, 3750–3758. [Google Scholar] [CrossRef]
- Yin, X.; Zeng, W.; Wu, B.; Wang, L.; Wang, Z.; Tian, H.; Wang, L.; Jiang, Y.; Clay, R.; Wei, X.; et al. PPARα Inhibition Overcomes Tumor-Derived Exosomal Lipid-Induced Dendritic Cell Dysfunction. Cell Reports 2020, 33, 108278. [Google Scholar] [CrossRef]
- Nasrollahzadeh, E.; Rezaei, N. Time to Sleep: Immunologic Niche Switches Tumor Dormancy at Metastatic Sites. In Cancerous Cells; Rezaei, N., Ed.; Handbook of Cancer and Immunology; Springer Nature Switzerland: Cham, 2025; Vol. 2, pp. 635–661. ISBN 978-3-032-00758-2. [Google Scholar]
- Pathak, A.; Pal, A.K.; Roy, S.; Nandave, M.; Jain, K. Role of Angiogenesis and Its Biomarkers in Development of Targeted Tumor Therapies. Stem Cells International 2024, 2024, 9077926. [Google Scholar] [CrossRef]
- Tufail, M.; Jiang, C.-H.; Li, N. Tumor Dormancy and Relapse: Understanding the Molecular Mechanisms of Cancer Recurrence. Military Med Res 2025, 12, 7. [Google Scholar] [CrossRef]
- Walker, N.D.; Elias, M.; Guiro, K.; Bhatia, R.; Greco, S.J.; Bryan, M.; Gergues, M.; Sandiford, O.A.; Ponzio, N.M.; Leibovich, S.J.; et al. Exosomes from Differentially Activated Macrophages Influence Dormancy or Resurgence of Breast Cancer Cells within Bone Marrow Stroma. Cell Death Dis 2019, 10, 59. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, T.; Sano, T.; Oshimo, Y.; Kawada, C.; Kasai, M.; Yamamoto, S.; Fukuhara, H.; Inoue, K.; Ogura, S. Enhanced Lipid Metabolism Induces the Sensitivity of Dormant Cancer Cells to 5-Aminolevulinic Acid-Based Photodynamic Therapy. Sci Rep 2021, 11, 7290. [Google Scholar] [CrossRef] [PubMed]
- Dioufa, N.; Clark, A.M.; Ma, B.; Beckwitt, C.H.; Wells, A. Bi-Directional Exosome-Driven Intercommunication between the Hepatic Niche and Cancer Cells. Mol Cancer 2017, 16, 172. [Google Scholar] [CrossRef]
- Ten, A.; Yudintceva, N.; Samochernykh, K.; Combs, S.E.; Jha, H.C.; Gao, H.; Shevtsov, M. Post-Secretion Processes and Modification of Extracellular Vesicles. Cells 2025, 14, 408. [Google Scholar] [CrossRef]
- Tóth, E.Á.; Turiák, L.; Visnovitz, T.; Cserép, C.; Mázló, A.; Sódar, B.W.; Försönits, A.I.; Petővári, G.; Sebestyén, A.; Komlósi, Z.; et al. Formation of a Protein Corona on the Surface of Extracellular Vesicles in Blood Plasma. J of Extracellular Vesicle 2021, 10, e12140. [Google Scholar] [CrossRef]
- Beit--Yannai, E.; Tabak, S.; Stamer, W.D. Physical Exosome:Exosome Interactions. J Cellular Molecular Medi 2018, 22, 2001–2006. [Google Scholar] [CrossRef]
- Ogungbemi, D.; Shorter, S.; Asadollahi, R.; Boussios, S.; Ovsepian, S.V. Molecular Engines Driving Biogenesis, Trafficking and Release of Exosomes: SNARE Proteins. Extracellular Vesicle 2025, 6, 100089. [Google Scholar] [CrossRef]
- Shin, W.; Arpino, G.; Thiyagarajan, S.; Su, R.; Ge, L.; McDargh, Z.; Guo, X.; Wei, L.; Shupliakov, O.; Jin, A.; et al. Vesicle Shrinking and Enlargement Play Opposing Roles in the Release of Exocytotic Contents. Cell Reports 2020, 30, 421–431.e7. [Google Scholar] [CrossRef]
- Thompson, W.; Papoutsakis, E.T. The Role of Biomechanical Stress in Extracellular Vesicle Formation, Composition and Activity. Biotechnology Advances 2023, 66, 108158. [Google Scholar] [CrossRef]
- Wang, L.; Wang, D.; Ye, Z.; Xu, J. Engineering Extracellular Vesicles as Delivery Systems in Therapeutic Applications. Advanced Science 2023, 10, 2300552. [Google Scholar] [CrossRef]
- Matsuzaka, Y.; Yashiro, R. Classification and Molecular Functions of Heparan Sulfate Proteoglycans and Their Molecular Mechanisms with the Receptor. Biologics 2024, 4, 105–129. [Google Scholar] [CrossRef]
- Murakami, M. Extracellular Vesicles as a Hydrolytic Platform of Secreted Phospholipase A2. Biochimica et Biophysica Acta (BBA) - Molecular and Cell Biology of Lipids 2024, 1869, 159536. [Google Scholar] [CrossRef]
- Jeppesen, D.K.; Zhang, Q.; Franklin, J.L.; Coffey, R.J. Extracellular Vesicles and Nanoparticles: Emerging Complexities. Trends in Cell Biology 2023, 33, 667–681. [Google Scholar] [CrossRef]
- Zhang, Q.; Jeppesen, D.K.; Higginbotham, J.N.; Graves-Deal, R.; Trinh, V.Q.; Ramirez, M.A.; Sohn, Y.; Neininger, A.C.; Taneja, N.; McKinley, E.T.; et al. Supermeres Are Functional Extracellular Nanoparticles Replete with Disease Biomarkers and Therapeutic Targets. Nat Cell Biol 2021, 23, 1240–1254. [Google Scholar] [CrossRef]
- Domazet-Lošo, T.; Tautz, D. Phylostratigraphic Tracking of Cancer Genes Suggests a Link to the Emergence of Multicellularity in Metazoa. BMC Biol 2010, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Bussey, K.J.; Davies, P.C.W. Reverting to Single-Cell Biology: The Predictions of the Atavism Theory of Cancer. Progress in Biophysics and Molecular Biology 2021, 165, 49–55. [Google Scholar] [CrossRef]
- Carvalho, J. Evidence for the Atavistic Reversal to a Unicellular--like State as a Central Hallmark of Cancer. Intl Journal of Cancer 2025, 156, 1671–1671. [Google Scholar] [CrossRef]
- Daignan--Fornier, B.; Pradeu, T. Critically Assessing Atavism, an Evolution--centered and Deterministic Hypothesis on Cancer. BioEssays 2024, 46, 2300221. [Google Scholar] [CrossRef]
- Proietto, M.; Crippa, M.; Damiani, C.; Pasquale, V.; Sacco, E.; Vanoni, M.; Gilardi, M. Tumor Heterogeneity: Preclinical Models, Emerging Technologies, and Future Applications. Front. Oncol. 2023, 13, 1164535. [Google Scholar] [CrossRef]
- Al Lawati, Z.; Al Lawati, A. Short Communication | Hallmarks of Cancer; A Summarized Overview of Sustained Proliferative Signalling Component. Gulf J Oncolog 2025, 1, 36–42. [Google Scholar]
- Bhat, G.R.; Sethi, I.; Sadida, H.Q.; Rah, B.; Mir, R.; Algehainy, N.; Albalawi, I.A.; Masoodi, T.; Subbaraj, G.K.; Jamal, F.; et al. Cancer Cell Plasticity: From Cellular, Molecular, and Genetic Mechanisms to Tumor Heterogeneity and Drug Resistance. Cancer Metastasis Rev 2024, 43, 197–228. [Google Scholar] [CrossRef]
- Askenase, P.W. Ancient Evolutionary Origin and Properties of Universally Produced Natural Exosomes Contribute to Their Therapeutic Superiority Compared to Artificial Nanoparticles. IJMS 2021, 22, 1429. [Google Scholar] [CrossRef]
- Ashouri, A.; Zhang, C.; Gaiti, F. Decoding Cancer Evolution: Integrating Genetic and Non-Genetic Insights. Genes 2023, 14, 1856. [Google Scholar] [CrossRef]
- Kasperski, A. Life Entrapped in a Network of Atavistic Attractors: How to Find a Rescue. IJMS 2022, 23, 4017. [Google Scholar] [CrossRef] [PubMed]
- Brinster, R.L. The effect of cells transferred into the mouse blastocyst on subsequent development. J Exp Med 1974, 140, 1049–1056. [Google Scholar] [CrossRef] [PubMed]
- Proietti, S.; Cucina, A.; Pensotti, A.; Biava, P.M.; Minini, M.; Monti, N.; Catizone, A.; Ricci, G.; Leonetti, E.; Harrath, A.H.; et al. Active Fraction from Embryo Fish Extracts Induces Reversion of the Malignant Invasive Phenotype in Breast Cancer through Down-Regulation of TCTP and Modulation of E-Cadherin/β-Catenin Pathway. IJMS 2019, 20, 2151. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, M.J.; Kweh, M.F.; Biava, P.M.; Olalde, J.; Toro, A.P.; Goldschmidt-Clermont, P.J.; White, I.A. Evaluation of Exosome Derivatives as Bio-Informational Reprogramming Therapy for Cancer. J Transl Med 2021, 19, 103. [Google Scholar] [CrossRef]
- Pensotti, A.; Bizzarri, M.; Bertolaso, M. The Phenotypic Reversion of Cancer: Experimental Evidences on Cancer Reversibility through Epigenetic Mechanisms (Review). Oncol Rep 2024, 51, 48. [Google Scholar] [CrossRef]
- Fan, X.; Xie, Y.; Cao, S.; Zhu, L.; Wang, X. VPS35-Retromer: Multifunctional Roles in Various Biological Processes – A Focus on Neurodegenerative Diseases and Cancer. JIR 2025, 18, 4665–4680. [Google Scholar] [CrossRef]
- Yu, J.; Feng, H.; Sang, Q.; Li, F.; Chen, M.; Yu, B.; Xu, Z.; Pan, T.; Wu, X.; Hou, J.; et al. VPS35 Promotes Cell Proliferation via EGFR Recycling and Enhances EGFR Inhibitors Response in Gastric Cancer. eBioMedicine 2023, 89, 104451. [Google Scholar] [CrossRef] [PubMed]
- Hao, Q.; Bai, Y.; Guan, R.; Dong, R.; Bai, W.; Hamdy, H.; Wang, L.; Meng, M.; Sun, Y.; Shen, J.; et al. VPS35/Retromer-Dependent MT1-MMP Regulation Confers Melanoma Metastasis. Sci. China Life Sci. 2025, 68, 1996–2009. [Google Scholar] [CrossRef]
- Semeradtova, A.; Liegertova, M.; Herma, R.; Capkova, M.; Brignole, C.; Del Zotto, G. Extracellular Vesicles in Cancer’s Communication: Messages We Can Read and How to Answer. Mol Cancer 2025, 24, 86. [Google Scholar] [CrossRef]
- Zhang, H.; Freitas, D.; Kim, H.S.; Fabijanic, K.; Li, Z.; Chen, H.; Mark, M.T.; Molina, H.; Martin, A.B.; Bojmar, L.; et al. Identification of Distinct Nanoparticles and Subsets of Extracellular Vesicles by Asymmetric Flow Field-Flow Fractionation. Nat Cell Biol 2018, 20, 332–343. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Sinclair, J.A.; Shi, T.; Kim, G.; Zhu, R.; Gasper, G.; Wang, Y.; Higginbotham, J.N.; Zhang, Q.; Jeppesen, D.K.; et al. Surface Markers on Supermeres Outperform Extracellular Vesicles in Cancer Diagnosis 2025.
- Yu, L.; Shi, H.; Gao, T.; Xu, W.; Qian, H.; Jiang, J.; Yang, X.; Zhang, X. Exomeres and Supermeres: Current Advances and Perspectives. Bioactive Materials 2025, 50, 322–343. [Google Scholar] [CrossRef] [PubMed]
- Jeppesen, D.K.; Zhang, Q.; Franklin, J.L.; Coffey, R.J. Are Supermeres a Distinct Nanoparticle? J of Extracellular Bio 2022, 1, e44. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



