Submitted:
08 December 2025
Posted:
09 December 2025
You are already at the latest version
Abstract
Keywords:
Introduction
Materials and Methods
1. Literature Search for Figure 2-5 and S1-S2
2. Data Analysis
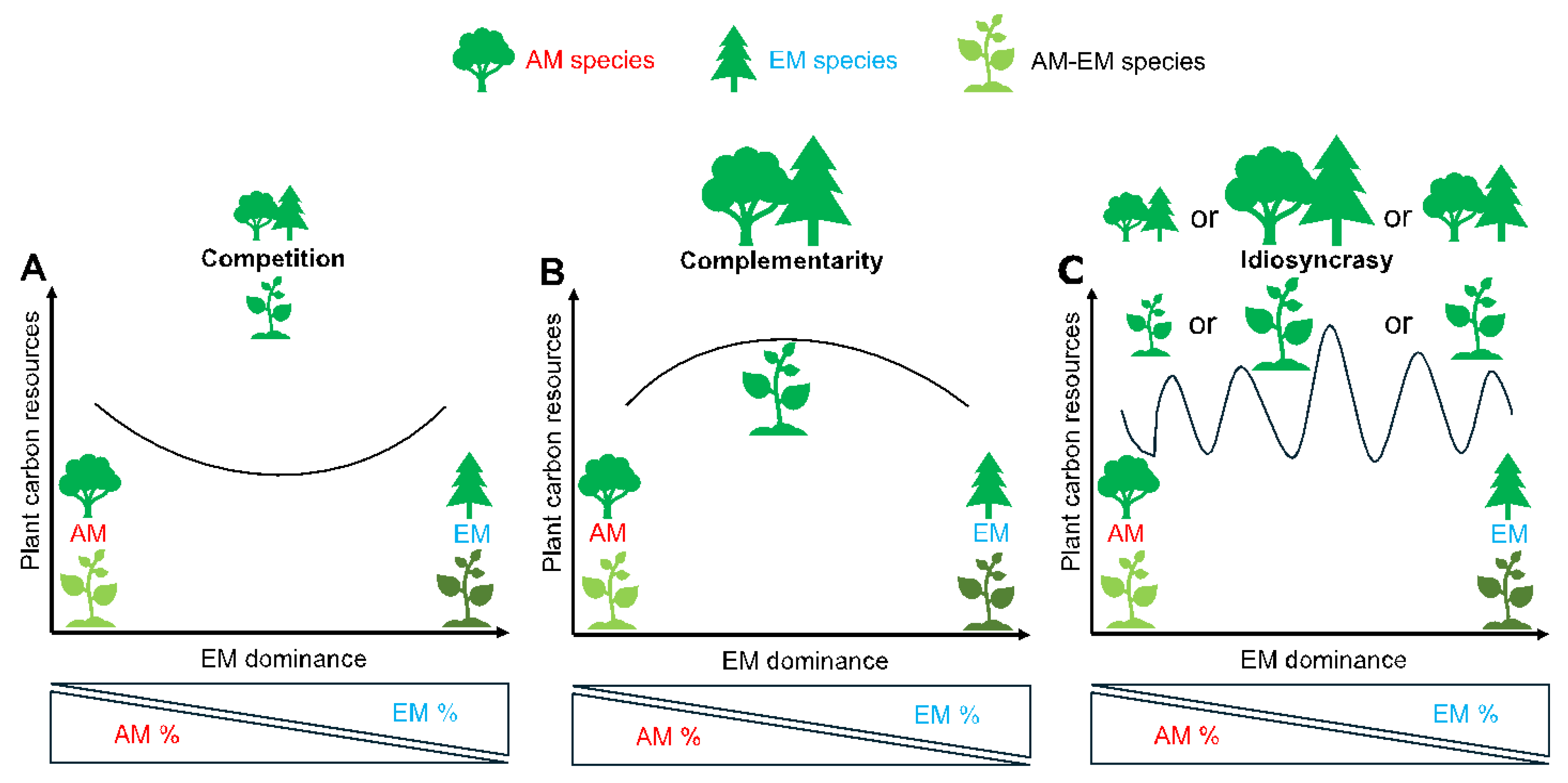
The Competition in AM and EM Interaction
Ecological Background
Physiological Factors
The Complementarity in AM and EM Interaction
Ecological Background
Physiological Factors
The Idiosyncratic Pattern of AM and EM Interaction
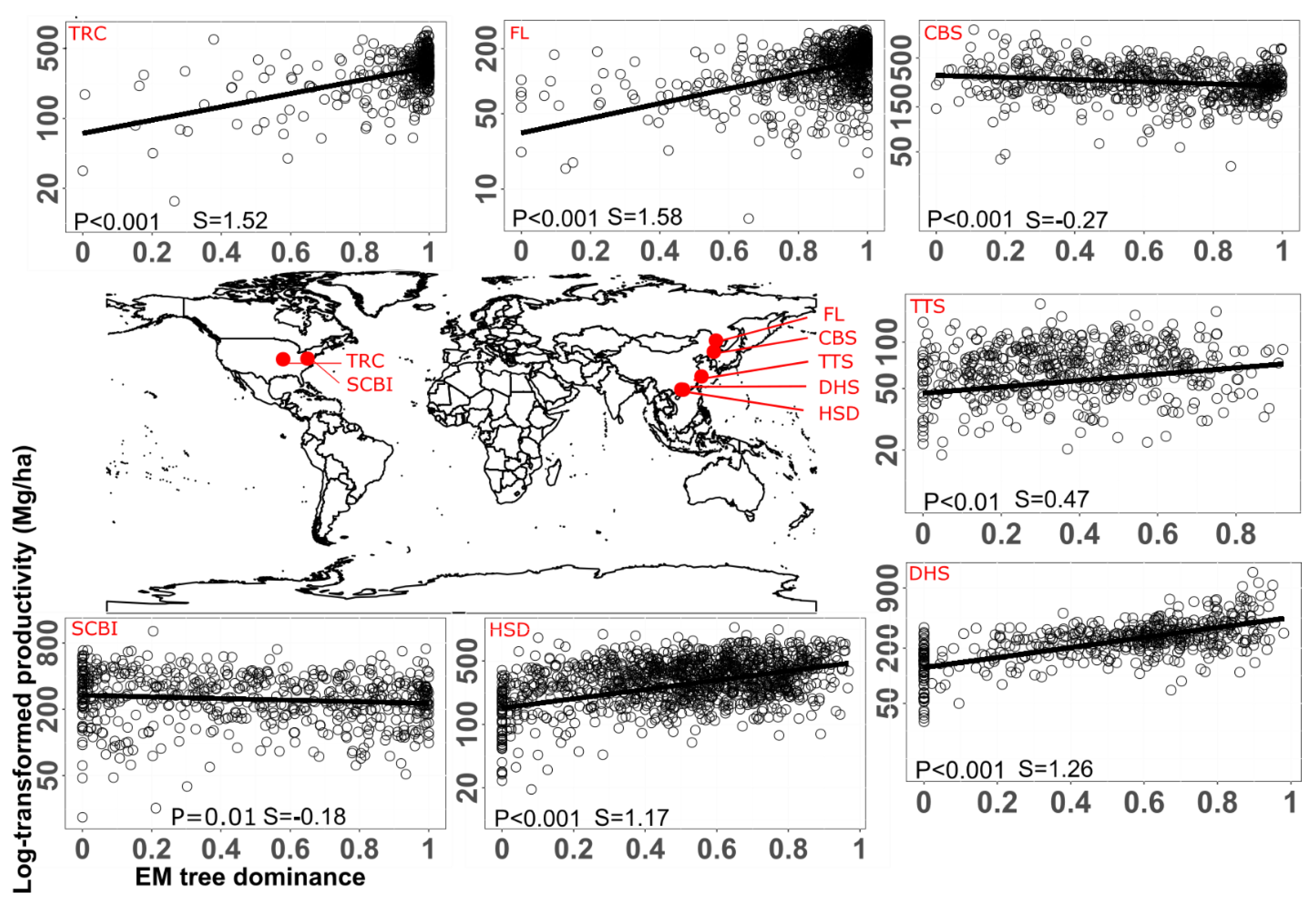
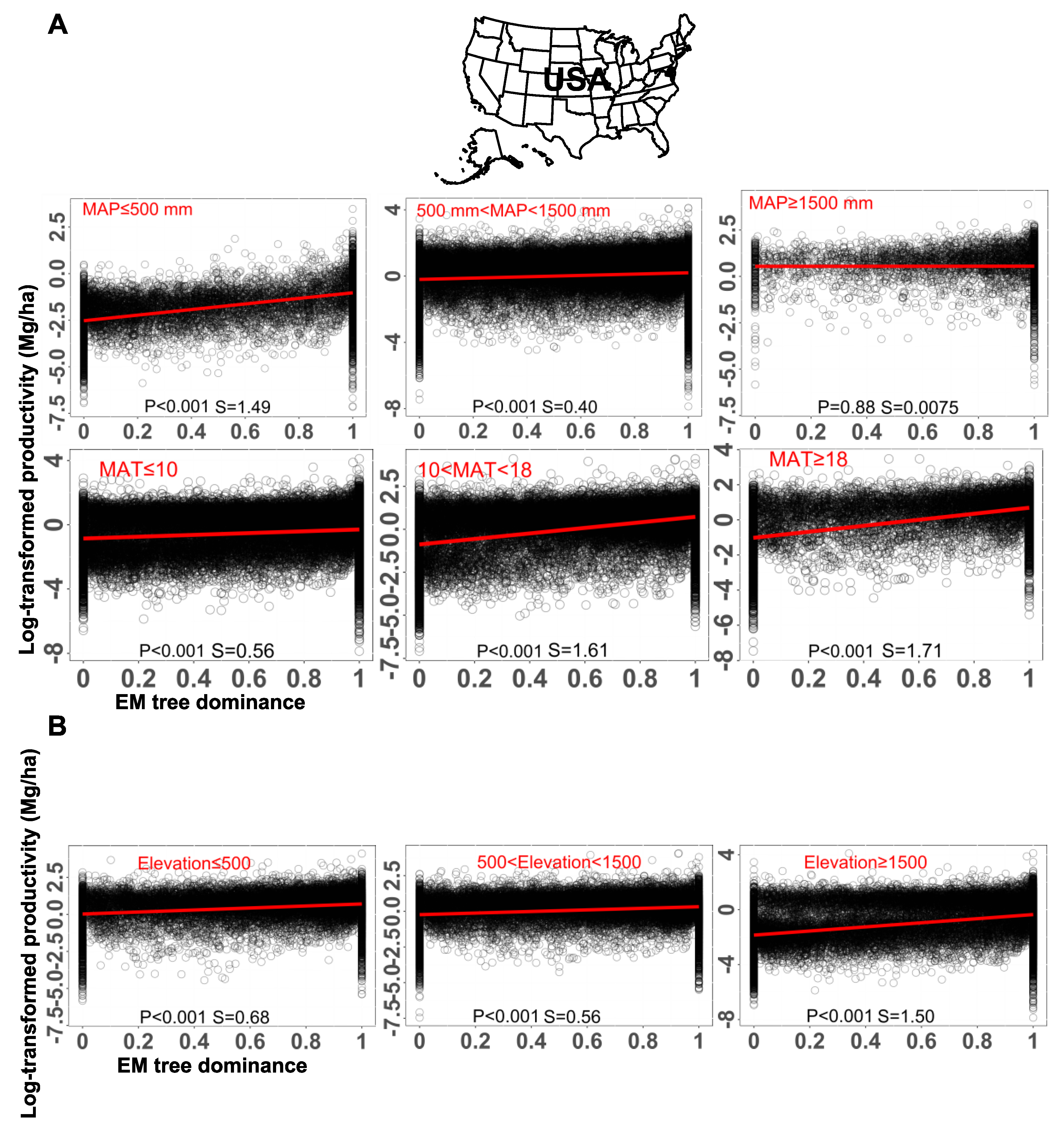
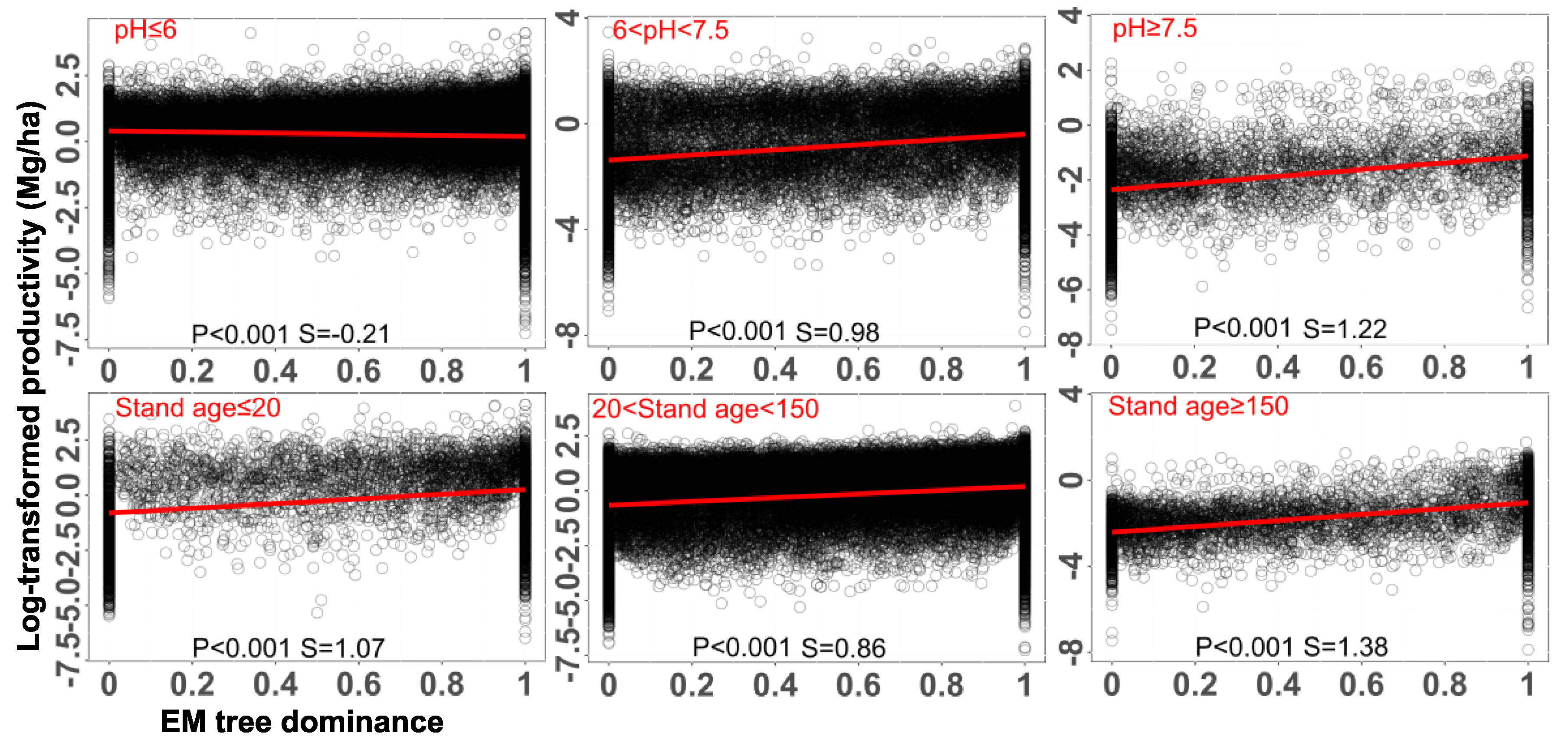
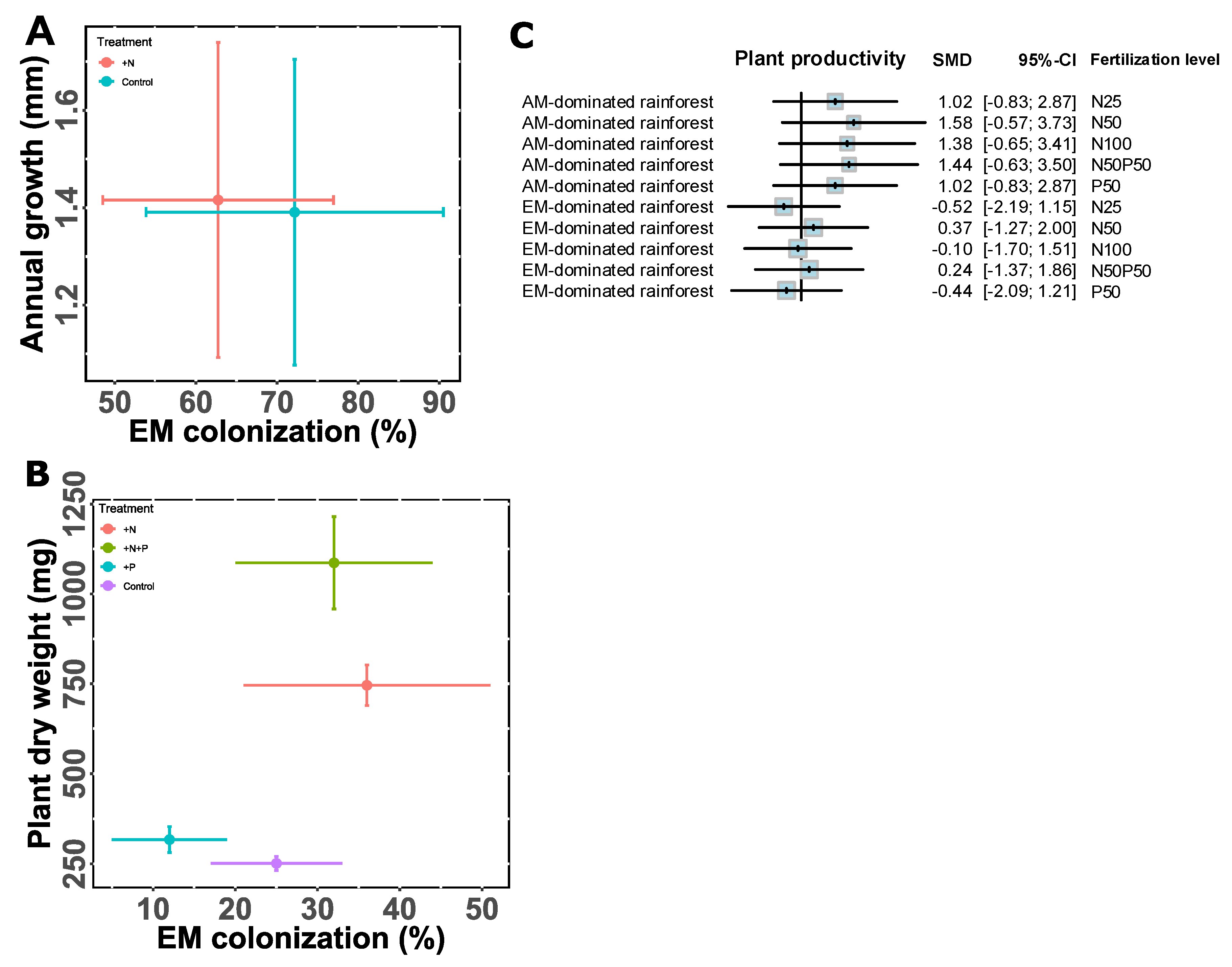
Metabolic Adjustments for the Idiosyncratic Pattern of AM and EM Interaction
Molecular Mechanisms of the Coexistence of AM and EM Symbiosis
| Plant common regulators | Plant taxa | Fungus taxa | Mycorrhizal type | Effect | Reference |
|---|---|---|---|---|---|
| TIFY10b (JAZ protein) | Musa acuminate Cavendish cv. Brail | Rhizophagus irregularis | AM | Regulatory roles in fungal infection | Lin et al.; 2021 |
| PtJAZ6 | Populus trichocarpa | Laccaria bicolor | EM | A co-repressor of JA-signaling | Daguerre et al.; 2020; Plett et al.; 2014 |
| MYC2 | Medicago truncatula | Rhizophagus irregularis | AM | The master regulator of JA responses | Adolfsson et al.; 2017 |
| MYC2 | Populus | Laccaria bicolor | EM | Controlling root fungal colonization | Marqués-Gálvez et al.; 2024 |
| SP7 | Medicago truncatula | Glomus intraradices | AM | Promoting symbiotic biotrophy | Kloppholz et al.; 2011 |
| MiSSP7 | Populus tremula × alba | Laccaria bicolor | EM | Promoting colonization | Plett et al.; 2011 |
| IAA, abscisic acid (ABA), JA, SA, CK | Olea europaea | Rhizophagus irregularis | AM | Water deficit tolerance | Tekaya et al.; 2022 |
| JA, ET | Populus | Laccaria bicolor | EM | Negative modulators | Plett et al.; 2014 |
| ABA, JA | Camellia sinensis | Glomus etunicatum | AM | Enhancing plant stress resistance and increasing nutrient uptake | Sun et al.; 2020 |
| JA, SA, GA, ET | Populus tremula × alba | Laccaria bicolor | EM | Multifaceted roles in EM development | Basso et al.; 2020 |
| LCOs | Medicago truncatula | Glomus intraradices | AM | Facilitating symbiosis | Maillet et al.; 2011 |
| LCOs | Populus | Laccaria bicolor | EM | Enhancing the colonization | Cope et al.; 2019 |
| Sesquiterpenes | Lotus japonicus | Gigaspora margarita | AM | Inducing hyphal branching | Akiyama et al.; 2005 |
| Sesquiterpenes | Eucalyptus grandis | Pisolithus microcarpus | EM | Altering root growth; promoting host colonization | Plett et al.; 2024 |
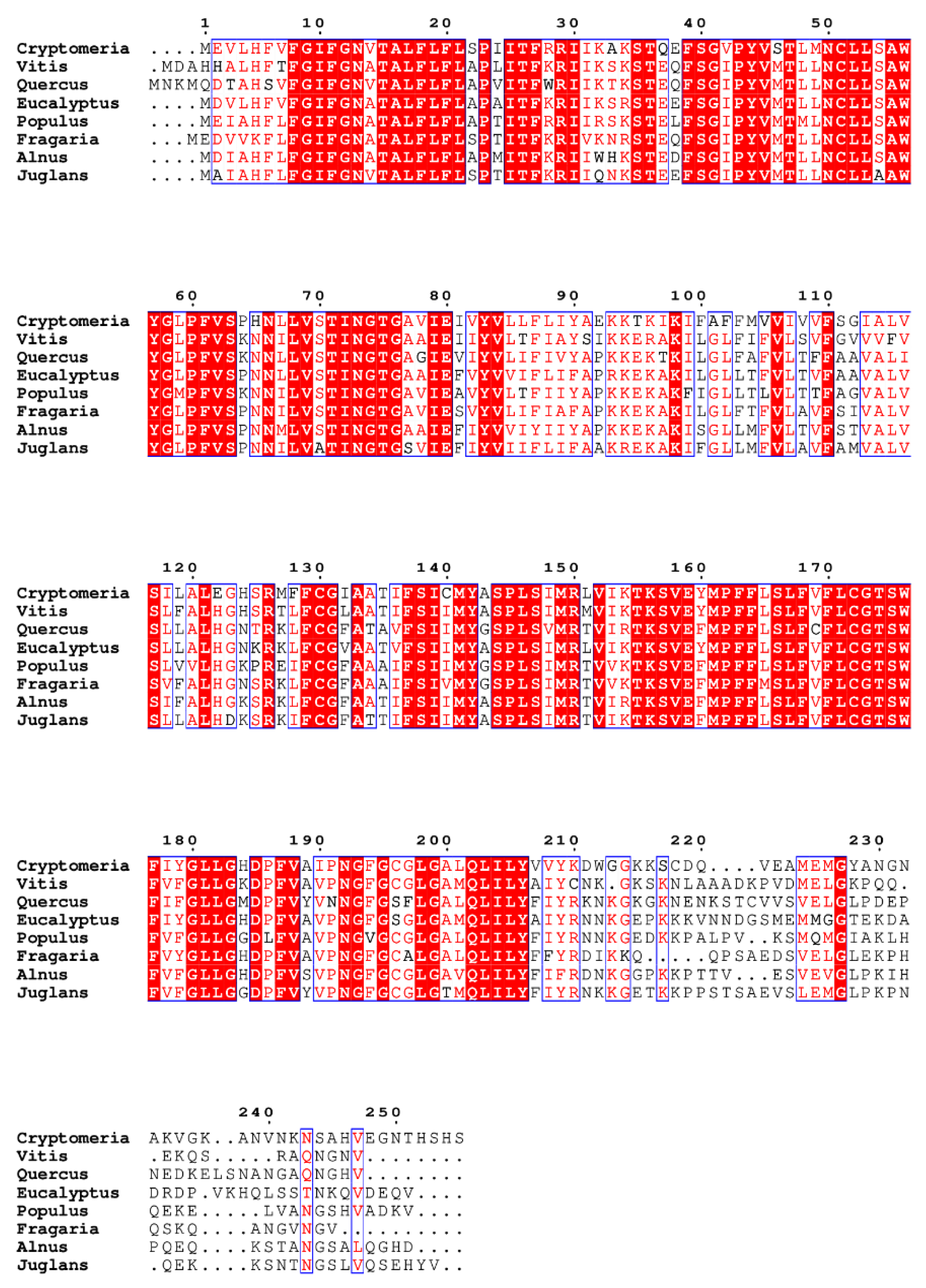
| SWEET1b | Medicago truncatula | Rhizophagus irregularis | AM | Maintaining arbuscules for a healthy mutually beneficial symbiosis | An et al.; 2019 |
| SWEET1c | Populus tremula × alba | Laccaria bicolor | EM | Facilitating glucose and sucrose transport |
Li et al.; 2024 |
Conclusions and Future Perspectives
Competing Interests
Supplementary Materials
Author Contributions
Data availability statement
Acknowledgments
References
- Adolfsson, L.; Nziengui, H.; Abreu, I.N.; Šimura, J.; Beebo, A.; Herdean, A.; Aboalizadeh, J.; Široká, J.; Moritz, T.; Novák, O.; et al. Enhanced secondary- and hormone metabolism in leaves of arbuscular mycorrhizal medicago truncatula. Plant Physiology 2017, 175, 392–411. [Google Scholar] [CrossRef]
- Agerer, R. 2 Characterization of Ectomycorrhiza. In Techniques for the Study of Mycorrhiza; Norris, J. R., Read, D.J., Varma, A. K., Eds.; Academic Press, 1991; Volume 23, pp. 25–73. [Google Scholar] [CrossRef]
- Akiyama, K.; Matsuzaki, K.I.; Hayashi, H. Plant sesquiterpenes induce hyphal branching in arbuscular mycorrhizal fungi. Nature 2005, 435, 824–827. [Google Scholar] [CrossRef]
- Albornoz, F.E.; Lambers, H.; Turner, B.L.; Teste, F.P.; Laliberté, E. Shifts in symbiotic associations in plants capable of forming multiple root symbioses across a long-term soil chronosequence. Ecology and Evolution 2016a, 6, 2368–2377. [Google Scholar] [CrossRef]
- Albornoz, F.E.; Teste, F.P.; Lambers, H.; Bunce, M.; Murray, D.C.; White, N.E.; Laliberté, E. Changes in ectomycorrhizal fungal community composition and declining diversity along a 2-million-year soil chronosequence. Molecular Ecology 2016b, 25, 4919–4929. [Google Scholar] [CrossRef] [PubMed]
- An, J.; Zeng, T.; Ji, C.; de Graaf, S.; Zheng, Z.; Xiao, T.T.; Deng, X.; Xiao, S.; Bisseling, T.; Limpens, E.; Pan, Z. A Medicago truncatula SWEET transporter implicated in arbuscule maintenance during arbuscular mycorrhizal symbiosis. New Phytologist 2019, 224, 396–408. [Google Scholar] [CrossRef] [PubMed]
- Anthony, M.A.; Crowther, T.W.; van der Linde, S.; Suz, L.M.; Bidartondo, M.I.; Cox, F.; Schaub, M.; Rautio, P.; Ferretti, M.; Vesterdal, L.; et al. Forest tree growth is linked to mycorrhizal fungal composition and function across Europe. ISME Journal 2022, 16, 1327–1336. [Google Scholar] [CrossRef] [PubMed]
- Azizoglu, U.; Argentel-Martínez, L.; Peñuelas-Rubio, O.; Meriño-Hernández, Y.; Rodríguez-Yon, Y.; Dell Amico-Rodríguez, J.M.; González-Aguilera, J.; Shin, J.H. Arbuscular mycorrhizal fungus Rhizophagus irregularis enhances antioxidant system mechanisms in chickpea plants under saline conditions. Plant Growth Regulation 2025, 105, 883–894. [Google Scholar] [CrossRef]
- Balduzzi, S.; Rücker, G.; Schwarzer, G. How to perform a meta-analysis with R: a practical tutorial. 2019, 153–160. [Google Scholar] [CrossRef] [PubMed]
- Bakker, M.R.; Garbaye, J.; Nys, C. Effect of liming on the ectomycorrhizal status of oak. Forest Ecology and Management 2000, 126, 121–131. [Google Scholar] [CrossRef]
- Basso, V.; Kohler, A.; Miyauchi, S.; Singan, V.; Guinet, F.; Šimura, J.; Novák, O.; Barry, K.W.; Amirebrahimi, M.; Block, J.; et al. An ectomycorrhizal fungus alters sensitivity to jasmonate, salicylate, gibberellin, and ethylene in host roots. Plant Cell and Environment 2020, 43, 1047–1068. [Google Scholar] [CrossRef]
- Becerra, A.; Zak, M.R.; Horton, T.R.; Micolini, J. Ectomycorrhizal and arbuscular mycorrhizal colonization of Alnus acuminata from Calilegua National Park (Argentina). Mycorrhiza 2005, 15, 525–531. [Google Scholar] [CrossRef]
- Begum, N.; Akhtar, K.; Ahanger, M.A.; Iqbal, M.; Wang, P.; Mustafa, N.S.; Zhang, L. Arbuscular mycorrhizal fungi improve growth, essential oil, secondary metabolism, and yield of tobacco (Nicotiana tabacum L.) under drought stress conditions. Environmental Science and Pollution Research 2021, 28, 45276–45295. [Google Scholar] [CrossRef]
- Benabdellah, K.; Azcón-Aguilar, C.; Valderas, A.; Speziga, D.; Fitzpatrick, T.B.; Ferrol, N. GintPDX1 encodes a protein involved in vitamin B6 biosynthesis that is up-regulated by oxidative stress in the arbuscular mycorrhizal fungus Glomus intraradices. New Phytologist 2009, 184, 682–693. [Google Scholar] [CrossRef]
- Bennett, J.A.; Maherali, H.; Reinhart, K.O.; Lekberg, Y.; Hart, M.M.; Klironomos, J. Plant-soil feedbacks and mycorrhizal type influence temperate forest population dynamics. Science 2017, 355, 181–184. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, L.; Carletti, P.; Badeck, F.W.; Rizza, F.; Morcia, C.; Ghizzoni, R.; Rouphael, Y.; Colla, G.; Terzi, V.; Lucini, L. Metabolomic responses triggered by arbuscular mycorrhiza enhance tolerance to water stress in wheat cultivars. Plant Physiology and Biochemistry 2019, 137, 203–212. [Google Scholar] [CrossRef]
- Boivin, S.; Fonouni-Farde, C.; Frugier, F. How auxin and cytokinin phytohormones modulate root microbe interactions. Frontiers in Plant Science 2016, 7, 1240. [Google Scholar] [CrossRef]
- Booth, M.G. Mycorrhizal networks mediate overstorey-understorey competition in a temperate forest. Ecology Letters 2004, 7, 538–546. [Google Scholar] [CrossRef]
- Borenstein, M. Effect sizes for studies with continuous data. In The handbook of research synthesis and meta-analysis, 2nd ed.; Cooper, H., Hedges, L.V., Valentine, J.C., Eds.; SAGE: New York, NY, USA, 2009; pp. 221–252. [Google Scholar]
- Brundrett, M.C. Coevolution of roots and mycorrhizas of land plants. New Phytologist 2002, 154, 275–304. [Google Scholar] [CrossRef] [PubMed]
- Brundrett, M.C. Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant and Soil 2009, 320, 37–77. [Google Scholar] [CrossRef]
- Campo, S.; San Segundo, B. Systemic induction of phosphatidylinositol-based signaling in leaves of arbuscular mycorrhizal rice plants. Scientific Reports 2020, 10, 1–17. [Google Scholar] [CrossRef]
- Castaño, C.; Suarez-vidal, E.; Zas, R.; Bonet, Jose Antonio; Oliva, J.; Sampedro, L. Ectomycorrhizal fungi with hydrophobic mycelia and rhizomorphs dominate in young pine trees surviving experimental drought stress. Soil Biology and Biochemistry 2023, 178, 108932. [Google Scholar] [CrossRef]
- Chaudhury, R.; Chakraborty, A.; Rahaman, F.; Sarkar, T.; Dey, S.; Das, M. Mycorrhization in trees: ecology, physiology, emerging technologies and beyond. Plant Biology 2024, 26, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Checker, V.G.; Kushwaha, H.R.; Kumari, P.; Yadav, S. Role of Phytohormones in Plant Defense: Signaling and Cross Talk BT - Molecular Aspects of Plant-Pathogen Interaction; Singh, A., Singh, I. K., Eds.; Springer Singapore, 2018; pp. 159–184. [Google Scholar] [CrossRef]
- Chen, Y.L.; Brundrett, M.C.; Dell, B. Effects of ectomycorrhizas and vesicular-arbuscular mycorrhizas, alone or in competition, on root colonization and growth of Eucalyptus globulus and E. urophylla. New Phytologist 2000, 146, 545–555. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, W.; Lang, X.; Wang, M.; Liu, J.; Xu, C. Arbuscular mycorrhizal and ectomycorrhizal plants together shape seedling diversity in a subtropical forest. Frontiers in Forests and Global Change 2023, 6, 1304897. [Google Scholar] [CrossRef]
- Cheng, H.Q.; Ding, Y.E.; Shu, B.; Zou, Y.N.; Wu, Q.S.; Kuca, K. Plant Aquaporin responses to mycorrhizal symbiosis under abiotic stress. International Journal of Agriculture and Biology 2020, 23, 786–794. [Google Scholar]
- Chilvers, G.A.; Lapeyrie, F.F.; Horan, D.P. Ectomycorrhizal Vs Endomycorrhizal Fungi Within the Same Root System. New Phytologist 1987, 107, 441–448. [Google Scholar] [CrossRef]
- Clemmensen, K.E. Roots and Associated Fungi Drive Long-Term Carbon Sequestration in Boreal Forest. Science 2013, 339, 1615. [Google Scholar] [CrossRef] [PubMed]
- Cope, K.R.; Bascaules, A.; Irving, T.B.; Venkateshwaran, M.; Maeda, J.; Garcia, K.; Rush, T.A.; Ma, C.; Labbé, J.; Jawdy, S.; et al. The ectomycorrhizal fungus laccaria bicolor produces lipochitooligosaccharides and uses the common symbiosis pathway to colonize populus roots. Plant Cell 2019, 31, 2386–2410. [Google Scholar] [CrossRef]
- Courty, P.E.; Pritsch, K.; Schloter, M.; Hartmann, A.; Garbaye, J. Activity profiling of ectomycorrhiza communities in two forest soils using multiple enzymatic tests. New Phytologist 2005, 167, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Daguerre, Y.; Basso, V.; Hartmann-Wittulski, S.; Schellenberger, R.; Meyer, L.; Bailly, J.; Kohler, A.; Plett, J.M.; Martin, F.; Veneault-Fourrey, C. The mutualism effector MiSSP7 of Laccaria bicolor alters the interactions between the poplar JAZ6 protein and its associated proteins. Scientific Reports 2020, 10, 20362. [Google Scholar] [CrossRef]
- Dey, M.; Ghosh, S. Arbuscular mycorrhizae in plant immunity and crop pathogen control. Rhizosphere 2022, 22, 100524. [Google Scholar] [CrossRef]
- Dreischhoff, S.; Das, I.S.; Jakobi, M.; Kasper, K.; Polle, A. Local Responses and Systemic Induced Resistance Mediated by Ectomycorrhizal Fungi. Frontiers in Plant Science 2020, 11, 590063. [Google Scholar] [CrossRef]
- Ekblad, A.; Wallander, H.; Näsholm, T. Chitin and ergosterol combined to measure total and living fungal biomass in ectomycorrhizas. New Phytologist 1998, 138, 143–149. [Google Scholar] [CrossRef]
- Fernandez, C.W.; Kennedy, P.G. Revisiting the “Gadgil effect”: Do interguild fungal interactions control carbon cycling in forest soils? New Phytologist 2016, 209, 1382–1394. [Google Scholar] [CrossRef]
- Fernández, N.; Knoblochová, T.; Kohout, P.; Janoušková, M.; Cajthaml, T.; Frouz, J.; Rydlová, J. Asymmetric Interaction Between Two Mycorrhizal Fungal Guilds and Consequences for the Establishment of Their Host Plants. Frontiers in Plant Science 2022, 13, 873204. [Google Scholar] [CrossRef] [PubMed]
- Gehring, C.A.; Mueller, R.C.; Whitham, T.G. Environmental and genetic effects on the formation of ectomycorrhizal and arbuscular mycorrhizal associations in cottonwoods. Oecologia 2006, 149, 158–164. [Google Scholar] [CrossRef]
- Genre, A.; Lanfranco, L.; Perotto, S.; Bonfante, P. Unique and common traits in mycorrhizal symbioses. Nature Reviews Microbiology 2020, 18, 649–660. [Google Scholar] [CrossRef]
- Gerke, J. The acquisition of phosphate by higher plants: Effect of carboxylate release by the roots. A critical review. Journal of Plant Nutrition and Soil Science 2015, 178, 351–364. [Google Scholar] [CrossRef]
- González-Guerrero, M.; Oger, E.; Benabdellah, K.; Azcón-Aguilar, C.; Lanfranco, L.; Ferrol, N. Characterization of a CuZn superoxide dismutase gene in the arbuscular mycorrhizal fungus Glomus intraradices. Current Genetics 2010, 56, 265–274. [Google Scholar] [CrossRef] [PubMed]
- Hawkins, H.J.; Cargill, R.I.M.; Van Nuland, M.E.; Hagen, S.C.; Field, K.J.; Sheldrake, M.; Soudzilovskaia, N.A.; Kiers, E.T. Mycorrhizal mycelium as a global carbon pool. Current Biology 2023, 33, 560–573. [Google Scholar] [CrossRef] [PubMed]
- He, X.H.; Critchley, C.; Bledsoe, C. Nitrogen transfer within and between plants through common mycorrhizal networks (CMNs). Critical Reviews in Plant Sciences 2003, 22, 531–567. [Google Scholar] [CrossRef]
- Heklau, H.; Schindler, N.; Buscot, F.; Eisenhauer, N.; Ferlian, O.; Prada Salcedo, L.D.; Bruelheide, H. Mixing tree species associated with arbuscular or ectotrophic mycorrhizae reveals dual mycorrhization and interactive effects on the fungal partners. Ecology and Evolution 2021, 11, 5424–5440. [Google Scholar] [CrossRef]
- Ho-Plágaro, T.; Usman, M.; Swinnen, J.; Ruytinx, J.; Gosti, F.; Gaillard, I.; Zimmermann, S.D. HcZnT2 is a highly mycorrhiza-induced zinc transporter from Hebeloma cylindrosporum in association with pine. Frontiers in Plant Science 2024, 15, 1466279. [Google Scholar] [CrossRef]
- Högberg, M.N.; Briones, M.J.I.; Keel, S.G.; Metcalfe, D.B.; Campbell, C.; Midwood, A.J.; Thornton, B.; Hurry, V.; Linder, S.; Näsholm, T.; Högberg, P. Quantification of effects of season and nitrogen supply on tree below-ground carbon transfer to ectomycorrhizal fungi and other soil organisms in a boreal pine forest. New Phytologist 2010, 187, 485–493. [Google Scholar] [CrossRef]
- Holste, E.K.; Kobe, R.K.; Gehring, C.A. Plant species differ in early seedling growth and tissue nutrient responses to arbuscular and ectomycorrhizal fungi. Mycorrhiza 2017, 27, 211–223. [Google Scholar] [CrossRef]
- Hortal, S.; Plett, K.L.; Plett, J.M.; Cresswell, T.; Johansen, M.; Pendall, E.; Anderson, I.C. Role of plant-fungal nutrient trading and host control in determining the competitive success of ectomycorrhizal fungi. ISME Journal 2017, 11, 2666–2676. [Google Scholar] [CrossRef]
- Janeeshma, E.; Puthur, J.T. Direct and indirect influence of arbuscular mycorrhizae on enhancing metal tolerance of plants. Archives of Microbiology 2020, 202, 1–16. [Google Scholar] [CrossRef]
- Janos, D.P.; Scott, J.; Aristizábal, C.; Bowman, D.M.J.S. Arbuscular-Mycorrhizal Networks Inhibit Eucalyptus tetrodonta Seedlings in Rain Forest Soil Microcosms. PLoS ONE 2013, 8. [Google Scholar] [CrossRef]
- Jiang, D.; Tan, M.; Wu, S.; Zheng, L.; Wang, Q.; Wang, G.; Yan, S. Defense responses of arbuscular mycorrhizal fungus-colonized poplar seedlings against gypsy moth larvae: a multiomics study. Horticulture Research 2021, 8. [Google Scholar] [CrossRef]
- Johnson, N.C.; Wilson, G.W.T.; Bowker, M.A.; Wilson, J.A.; Miller, R.M. Resource limitation is a driver of local adaptation in mycorrhizal symbioses. Proceedings of the National Academy of Sciences 2010, 107, 2093–2098. [Google Scholar] [CrossRef]
- Kariman, K.; Barker, S.J.; Jost, R.; Finnegan, P.M.; Tibbett, M. A novel plant-fungus symbiosis benefits the host without forming mycorrhizal structures. New Phytologist 2014, 201, 1413–1422. [Google Scholar] [CrossRef] [PubMed]
- Kebert, M.; Kostić, S.; Čapelja, E.; Vuksanović, V.; Stojnić, S.; Markić, A.G.; Zlatković, M.; Milović, M.; Galović, V.; Orlović, S. Ectomycorrhizal Fungi Modulate Pedunculate Oak’s Heat Stress Responses through the Alternation of Polyamines, Phenolics, and Osmotica Content. Plants 2022, 11, 3360. [Google Scholar] [CrossRef]
- Kilpeläinen, J.; Aphalo, P.J.; Lehto, T. Temperature affected the formation of arbuscular mycorrhizas and ectomycorrhizas in Populus angustifolia seedlings more than a mild drought. Soil Biology and Biochemistry 2020, 146, 107798. [Google Scholar] [CrossRef]
- Kloppholz, S.; Kuhn, H.; Requena, N. A secreted fungal effector of glomus intraradices promotes symbiotic biotrophy. Current Biology 2011, 21, 1204–1209. [Google Scholar] [CrossRef]
- Kluber, L.A.; Smith, J.E.; Myrold, D.D. Distinctive fungal and bacterial communities are associated with mats formed by ectomycorrhizal fungi. Soil Biology and Biochemistry 2011, 43, 1042–1050. [Google Scholar] [CrossRef]
- Knoblochová, T.; Kohout, P.; Püschel, D.; Doubková, P.; Frouz, J.; Cajthaml, T.; Kukla, J.; Vosátka, M.; Rydlová, J. Asymmetric response of root-associated fungal communities of an arbuscular mycorrhizal grass and an ectomycorrhizal tree to their coexistence in primary succession. Mycorrhiza 2017, 27, 775–789. [Google Scholar] [CrossRef]
- Kuyper, T.W.; Suz, L.M. Do Ectomycorrhizal Trees Select Ectomycorrhizal Fungi That Enhance Phosphorus Uptake under Nitrogen Enrichment? Forests 2023, 14, 467. [Google Scholar] [CrossRef]
- Lambers, H.; Raven, J.A.; Shaver, G.R.; Smith, S.E. Plant nutrient-acquisition strategies change with soil age. Trends in Ecology and Evolution 2008, 23, 95–103. [Google Scholar] [CrossRef]
- Li, R.; Shi, W.; Zhang, P.; Ma, J.; Zou, R.; Zhang, X.; Kohler, A.; Martin, F.M.; Zhang, F. The poplar SWEET1c glucose transporter plays a key role in the ectomycorrhizal symbiosis. New Phytologist 2024, 244, 2518–2535. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Liu, C.; Gao, Y.; Han, L.; Chu, H. Arbuscular mycorrhizal fungi contribute to reactive oxygen species homeostasis of Bombax ceiba L. under drought stress. Frontiers in Microbiology 2022, 13, 991781. [Google Scholar] [CrossRef]
- Lin, G.; McCormack, M.L.; Ma, C.; Guo, D. Similar below-ground carbon cycling dynamics but contrasting modes of nitrogen cycling between arbuscular mycorrhizal and ectomycorrhizal forests. New Phytologist 2017, 213, 1440–1451. [Google Scholar] [CrossRef]
- Lin, P.; Zhang, M.; Wang, M.; Li, Y.; Liu, J.; Chen, Y. Inoculation with arbuscular mycorrhizal fungus modulates defense-related genes expression in banana seedlings susceptible to wilt disease. Plant Signaling and Behavior 2021, 16. [Google Scholar] [CrossRef]
- Lindahl, B.D.; Tunlid, A. Ectomycorrhizal fungi - potential organic matter decomposers, yet not saprotrophs. New Phytologist 2015, 205, 1443–1447. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Burslem, D.F.R.P.; Taylor, J.D.; Taylor, A.F.S.; Khoo, E.; Majalap-Lee, N.; Helgason, T.; Johnson, D. Partitioning of soil phosphorus among arbuscular and ectomycorrhizal trees in tropical and subtropical forests. Ecology Letters 2018, 21, 713–723. [Google Scholar] [CrossRef]
- Luo, S.; Phillips, R.P.; Jo, I.; Fei, S.; Liang, J.; Schmid, B.; Eisenhauer, N. Higher productivity in forests with mixed mycorrhizal strategies. Nature Communications 2023, 14, 1–10. [Google Scholar] [CrossRef]
- Madeira, F.; Madhusoodanan, N.; Lee, J.; Eusebi, A.; Niewielska, A.; Tivey, A.R.N.; Lopez, R.; Butcher, S. The EMBL-EBI Job Dispatcher sequence analysis tools framework in 2024. Nucleic Acids Research 2024, 52, 521–525. [Google Scholar] [CrossRef]
- Maillet, F.; Poinsot, V.; André, O.; Puech-Pagés, V.; Haouy, A.; Gueunier, M.; Cromer, L.; Giraudet, D.; Formey, D.; Niebel, A.; et al. Fungal lipochitooligosaccharide symbiotic signals in arbuscular mycorrhiza. Nature 2011, 469, 58–64. [Google Scholar] [CrossRef]
- Mao, Z.; van der Plas, F.; Corrales, A.; Anderson-Teixeira, K.J.; Bourg, N.A.; Chu, C.; Hao, Z.; Jin, G.; Lian, J.; Lin, F.; et al. Scale-dependent diversity–biomass relationships can be driven by tree mycorrhizal association and soil fertility. Ecological Monographs 2023, 93, e1568. [Google Scholar] [CrossRef]
- Mao, Z.; Wiegand, T.; Corrales, A.; Fang, S.; Hao, Z.; Lin, F.; Ye, J.; Yuan, Z.; Wang, X. Mycorrhizal Types Regulate Tree Spatial Associations in Temperate Forests: Ectomycorrhizal Trees Might Favour Species Coexistence. Ecology Letters 2024, 27, e70005. [Google Scholar] [CrossRef] [PubMed]
- Marqués-Gálvez, J.E.; Pandharikar, G.; Basso, V.; Kohler, A.; Lackus, N.D.; Barry, K.; Keymanesh, K.; Johnson, J.; Singan, V.; Grigoriev, I.V.; et al. Populus MYC2 orchestrates root transcriptional reprogramming of defence pathway to impair Laccaria bicolor ectomycorrhizal development. New Phytologist 2024, 242, 658–674. [Google Scholar] [CrossRef] [PubMed]
- McHugh, T.A.; Gehring, C.A. Below-ground interactions with arbuscular mycorrhizal shrubs decrease the performance of pinyon pine and the abundance of its ectomycorrhizas. New Phytologist 2006, 171, 171–178. [Google Scholar] [CrossRef]
- Miao, F.; Wang, S.; Yuan, Y.; Chen, Y.; Guo, E.; Li, Y. The Addition of a High Concentration of Phosphorus Reduces the Diversity of Arbuscular Mycorrhizal Fungi in Temperate Agroecosystems. Diversity 2023, 15, 1045. [Google Scholar] [CrossRef]
- Midgley, M.G.; Phillips, R.P. Mycorrhizal associations of dominant trees influence nitrate leaching responses to N deposition. Biogeochemistry 2014, 117, 241–253. [Google Scholar] [CrossRef]
- Miyamoto, Y.; Maximov, T.C.; Bryanin, S.V.; Kononov, A.; Sugimoto, A. Host phylogeny is the primary determinant of ectomycorrhizal fungal community composition in the permafrost ecosystem of eastern Siberia at a regional scale. Fungal Ecology 2022, 55, 101117. [Google Scholar] [CrossRef]
- Nave, L.E.; Nadelhoffer, K.J.; Le Moine, J.M.; van Diepen, L.T.A.; Cooch, J.K.; Van Dyke, N.J. Nitrogen Uptake by Trees and Mycorrhizal Fungi in a Successional Northern Temperate Forest: Insights from Multiple Isotopic Methods. Ecosystems 2013, 16, 590–603. [Google Scholar] [CrossRef]
- Nehls, U.; Hampp, R. Carbon allocation in ectomycorrhizas. Physiological and Molecular Plant Pathology 2000, 57, 95–100. [Google Scholar] [CrossRef]
- Neville, J.; Tessier, J.L.; Morrison, I.; Scarratt, J.; Canning, B.; Klironomos, J.N. Soil depth distribution of ecto- and arbuscular mycorrhizal fungi associated with Populus tremuloides within a 3-year-old boreal forest clear-cut. Applied Soil Ecology 2002, 19, 209–216. [Google Scholar] [CrossRef]
- Parasquive, V.; Brisson, J.; Laliberté, E.; Chagnon, P.L. Arbuscular and ectomycorrhizal tree seedling growth is inhibited by competition from neighboring roots and associated fungal hyphae. Plant and Soil 2025, 507, 571–584. [Google Scholar] [CrossRef]
- Parasquive, V.; Brisson, J.; Laliberté, E.; Chagnon, P.L. Limited impact of soil inocula from arbuscular and ectomycorrhizal-dominated sites on root morphology and growth of four tree seedling species from a temperate deciduous forest. Plant and Soil 2025b, 511, 1137–1149. [Google Scholar] [CrossRef]
- Pellitier, P.T.; Zak, D.R. Ectomycorrhizal fungi and the enzymatic liberation of nitrogen from soil organic matter: why evolutionary history matters. New Phytologist 2018, 217, 68–73. [Google Scholar] [CrossRef]
- Phillips, R.P.; Brzostek, E.; Midgley, M.G. The mycorrhizal-associated nutrient economy: A new framework for predicting carbon-nutrient couplings in temperate forests. New Phytologist 2013, 199, 41–51. [Google Scholar] [CrossRef]
- Pierre-Emmanuel, C.; Karin, P.; Michael, S.; Anton, H.; Jean, G. Activity profiling of ectomycorrhiza communities in two forest soils using multiple enzymatic tests. New Phytologist 2005, 167, 309–319. [Google Scholar] [CrossRef]
- Plett, J.M.; Daguerre, Y.; Wittulsky, S.; Vayssier̀es, A.; Deveau, A.; Melton, S.J.; Kohler, A.; Morrell-Falvey, J.L.; Brun, A.; Veneault-Fourrey, C.; Martin, F. Effector MiSSP7 of the mutualistic fungus Laccaria bicolor stabilizes the Populus JAZ6 protein and represses jasmonic acid (JA) responsive genes. Proceedings of the National Academy of Sciences of the United States of America 2014, 111, 8299–8304. [Google Scholar] [CrossRef]
- Plett, J.M.; Kemppainen, M.; Kale, S.D.; Kohler, A.; Legué, V.; Brun, A.; Tyler, B.M.; Pardo, A.G.; Martin, F. A secreted effector protein of laccaria bicolor is required for symbiosis development. Current Biology 2011, 21, 1197–1203. [Google Scholar] [CrossRef]
- Plett, J.M.; Khachane, A.; Ouassou, M.; Sundberg, B.; Kohler, A.; Martin, F. Ethylene and jasmonic acid act as negative modulators during mutualistic symbiosis between Laccaria bicolor and Populus roots. New Phytologist 2014, 202, 270–286. [Google Scholar] [CrossRef]
- Plett, J.M.; Plett, K.L.; Wong-Bajracharya, J.; de Freitas Pereira, M.; Costa, M.D.; Kohler, A.; Martin, F.; Anderson, I.C. Mycorrhizal effector PaMiSSP10b alters polyamine biosynthesis in Eucalyptus root cells and promotes root colonization. New Phytologist 2020, 228, 712–727. [Google Scholar] [CrossRef]
- Posit Software, PBC. RStudio: Integrated development environment for R; Posit Software, PBC: Boston, MA, USA, 2024; Available online: https://posit.co/.
- R Core Team. R: A language and environment for statistical computing; R Foundation for Statistical Computing: Vienna, Austria, 2024; Available online: https://www.R-project.org/.
- Qin, F.; Yu, S. Compatible Mycorrhizal Types Contribute to a Better Design for Mixed Eucalyptus Plantations. Frontiers in Plant Science 2021, 12, 616726. [Google Scholar] [CrossRef] [PubMed]
- Queralt, M.; Walker, J.K.M.; De Miguel, A.M.; Parladé, J.; Anderson, I.C.; Hortal, S. The ability of a host plant to associate with different symbiotic partners affects ectomycorrhizal functioning. FEMS Microbiology Ecology 2019, 95, 1–11. [Google Scholar] [CrossRef]
- Rapaport, A.; Livne-Luzon, S.; Fox, H.; Oppenheimer-Shaanan, Y.; Klein, T. Rapid and chemically diverse C transfer from trees to mycorrhizal fruit bodies in the forest. Functional Ecology 2024, 2024, 1–15. [Google Scholar] [CrossRef]
- Read, D.J. Mycorrhizas in ecosystems. Experientia 1991, 47, 376–391. [Google Scholar] [CrossRef]
- Read, D.J.; Perez-Moreno, J. Mycorrhizas and nutrient cycling in ecosystems - A journey towards relevance? New Phytologist 2003, 157, 475–492. [Google Scholar] [CrossRef] [PubMed]
- Rillig, M.C.; Wright, S.F.; Eviner, V.T. The role of arbuscular mycorrhizal fungi and glomalin in soil aggregation. Plant and Soil 2002, 238, 325–333. Available online: https://link.springer.com/content/pdf/10.1023/A:1014483303813.pdf. [CrossRef]
- Robert, X.; Gouet, P. Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Research 2014, 42, 320–324. [Google Scholar] [CrossRef]
- Rog, I.; Lerner, D.; Bender, S.F.; van der Heijden, M.G.A. The Increased Environmental Niche of Dual-Mycorrhizal Woody Species. Ecology Letters 2025, 28. [Google Scholar] [CrossRef]
- Schwarzer G meta: An R package for meta-analysis. R news 2007, 7, 40–45.
- Sebastiana, M.; Duarte, B.; Monteiro, F.; Malhó, R.; Caçador, I.; Matos, A.R. The leaf lipid composition of ectomycorrhizal oak plants shows a drought-tolerance signature. Plant Physiology and Biochemistry 2019, 144, 157–165. [Google Scholar] [CrossRef]
- Shuting, Z.; Hongwei, D.; Qing, M.; Rui, H.; Huarong, T.; Lianyu, Y. Identification and expression analysis of the ZRT, IRT-like protein (ZIP) gene family in Camellia sinensis (L.) O. Kuntze. Plant Physiology and Biochemistry 2022, 172, 87–100. [Google Scholar] [CrossRef]
- Smith, S.E.; Jakobsen, I.; Grønlund, M.; Smith, F.A. Roles of arbuscular mycorrhizas in plant phosphorus nutrition: Interactions between pathways of phosphorus uptake in arbuscular mycorrhizal roots have important implications for understanding and manipulating plant phosphorus acquisition. Plant Physiology 2011, 156, 1050–1057. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D.J. Mycorrhizal symbiosis; Academic Press and Elsevier: London, UK, 2008. [Google Scholar]
- Soudzilovskaia, N.A.; van Bodegom, P.M.; Terrer, C.; Zelfde; van’t, M.; McCallum, I.; Luke McCormack, M.; Fisher, J.B.; Brundrett, M.C.; de Sá, N.C.; Tedersoo, L. Global mycorrhizal plant distribution linked to terrestrial carbon stocks. Nature Communications 2019, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Steidinger, B.S.; Crowther, T.W.; Liang, J.; Van Nuland, M.E.; Werner, G.D.A.; Reich, P.B.; Nabuurs, G.; de-Miguel, S.; Zhou, M.; Picard, N.; et al. Climatic controls of decomposition drive the global biogeography of forest-tree symbioses. Nature 2019, 569, 404–408. [Google Scholar] [CrossRef]
- Sun, M.; Yuan, D.; Hu, X.; Zhang, D.; Li, Y. Effects of mycorrhizal fungi on plant growth, nutrient absorption and phytohormones levels in tea under shading condition. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2020, 48, 2006–2020. [Google Scholar] [CrossRef]
- Tedersoo, L.; Bahram, M. Mycorrhizal types differ in ecophysiology and alter plant nutrition and soil processes. Biological Reviews 2019, 94, 1857–1880. [Google Scholar] [CrossRef]
- Tedersoo, L.; Bahram, M.; Põlme, S.; Kõljalg, U.; Yorou, N.S.; Wijesundera, R.; Ruiz, L.V.; Vasco-palacios, A.M.; Thu, P.Q.; Suija, A.; et al. Global diversity and geography of soil fungi. Science 2014, 346, 1052–1053. [Google Scholar] [CrossRef] [PubMed]
- Tedersoo, L.; Bahram, M.; Zobel, M. How mycorrhizal associations drive plant population and community biology. Science 2020, 367. [Google Scholar] [CrossRef] [PubMed]
- Tekaya, M.; Dabbaghi, O.; Guesmi, A.; Attia, F.; Chehab, H.; Khezami, L.; Algathami, F.K.; Ben Hamadi, N.; Hammami, M.; Prinsen, E.; Mechri, B. Arbuscular mycorrhizas modulate carbohydrate, phenolic compounds and hormonal metabolism to enhance water deficit tolerance of olive trees (Olea europaea). Agricultural Water Management 2022, 274, 107947. [Google Scholar] [CrossRef]
- Terrer, C.; Jackson, R.B.; Prentice, I.C.; Keenan, T.F.; Kaiser, C.; Vicca, S.; Fisher, J.B.; Reich, P.B.; Stocker, B.D.; Hungate, B.A.; et al. Nitrogen and phosphorus constrain the CO 2 fertilization of global plant biomass. Nature Climate Change 2019, 9, 684–689. [Google Scholar] [CrossRef]
- Teste, F.P.; Jones, M.D.; Dickie, I.A. Dual-mycorrhizal plants: their ecology and relevance. New Phytologist 2020, 225, 1835–1851. [Google Scholar] [CrossRef]
- Teste, F.P.; Laliberté, E. Plasticity in root symbioses following shifts in soil nutrient availability during long-term ecosystem development. Journal of Ecology 2019, 107, 633–649. [Google Scholar] [CrossRef]
- Teste, F.P.; Veneklaas, E.J.; Dixon, K.W.; Lambers, H. Complementary plant nutrient-acquisition strategies promote growth of neighbour species. Functional Ecology 2014, 28, 819–828. [Google Scholar] [CrossRef]
- Van Nuland, M.E.; Ke, P.J.; Wan, J.; Peay, K.G. Mycorrhizal nutrient acquisition strategies shape tree competition and coexistence dynamics. Journal of Ecology 2023, 111, 564–577. [Google Scholar] [CrossRef]
- Wahab, A.; Muhammad, M.; Munir, A.; Abdi, G.; Zaman, W.; Ayaz, A.; Khizar, C.; Reddy, S.P.P. Ecosystems under Abiotic and Biotic Stresses. Plants 2023, 12, 1–40. [Google Scholar] [CrossRef]
- Watts-Williams, S.J.; Wege, S.; Ramesh, S.A.; Berkowitz, O.; Xu, B.; Gilliham, M.; Whelan, J.; Tyerman, S.D. The function of the Medicago truncatula ZIP transporter MtZIP14 is linked to arbuscular mycorrhizal fungal colonization. Plant Cell and Environment 2023, 46, 1691–1704. [Google Scholar] [CrossRef] [PubMed]
- Wright, S.F.; Upadhyaya, A. A survey of soils for aggregate stability and glomalin, a glycoprotein produced by hyphae of arbuscular mycorrhizal fungi. Plant and Soil 1998, 198, 97–107. [Google Scholar] [CrossRef]
- Xia, M.; Suseela, V.; McCormack, M.L.; Kennedy, P.G.; Tharayil, N. Common and lifestyle-specific traits of mycorrhizal root metabolome reflect ecological strategies of plant–mycorrhizal interactions. Journal of Ecology 2023, 111, 601–616. [Google Scholar] [CrossRef]
- Yang, B.; Liang, Y.; Schmid, B.; Baruffol, M.; Li, Y.; He, L.; Salmon, Y.; Tian, Q.; Niklaus, P.A.; Ma, K. Soil Fungi Promote Biodiversity–Productivity Relationships in Experimental Communities of Young Trees. Ecosystems 2022, 25, 858–871. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, X.; Hartley, I.P.; Dungait, J.A.J.; Wen, X.; Li, D.; Guo, Z.; Quine, T.A. Contrasting rhizosphere soil nutrient economy of plants associated with arbuscular mycorrhizal and ectomycorrhizal fungi in karst forests. Plant and Soil 2022, 470, 81–93. [Google Scholar] [CrossRef]
- Ye, Q.; Wang, H.; Li, H. Arbuscular Mycorrhizal Fungi Enhance Drought Stress Tolerance by Regulating Osmotic Balance, the Antioxidant System, and the Expression of Drought-Responsive Genes in Vitis vinifera L. Australian Journal of Grape and Wine Research 2023, 2023, 7208341. [Google Scholar] [CrossRef]
- Zou, Y.N.; Wu, Q.S.; Kuča, K. Unravelling the role of arbuscular mycorrhizal fungi in mitigating the oxidative burst of plants under drought stress. Plant Biology 2021, 23, 50–57. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




