Submitted:
08 December 2025
Posted:
09 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Experimental animals and treatment
2.2. Micro-CT imaging
2.3. Hematology and biochemical analysis
2.4. CTX1 and P1NP Western blot
2.5. Statistical analysis
3. Results
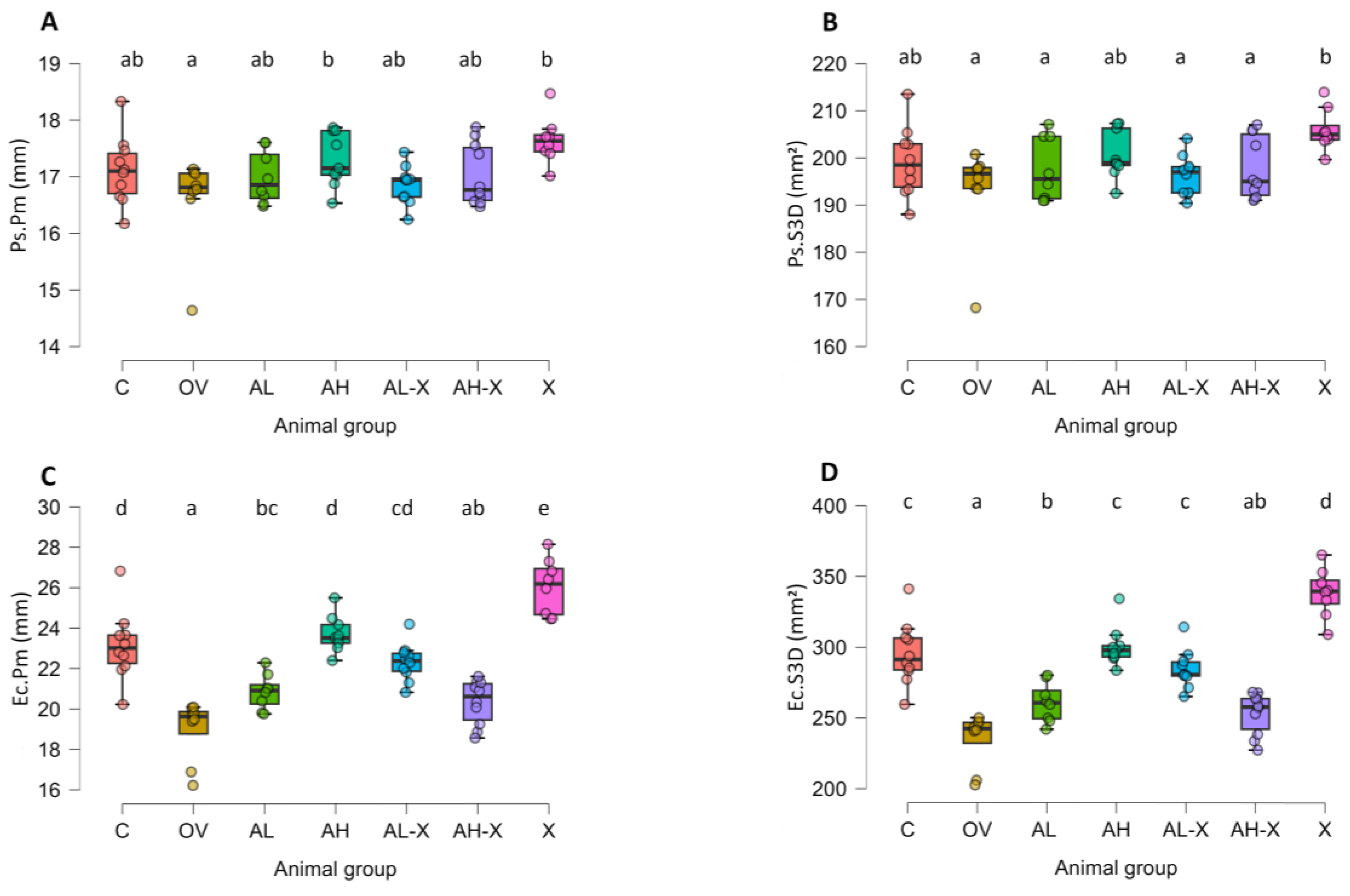
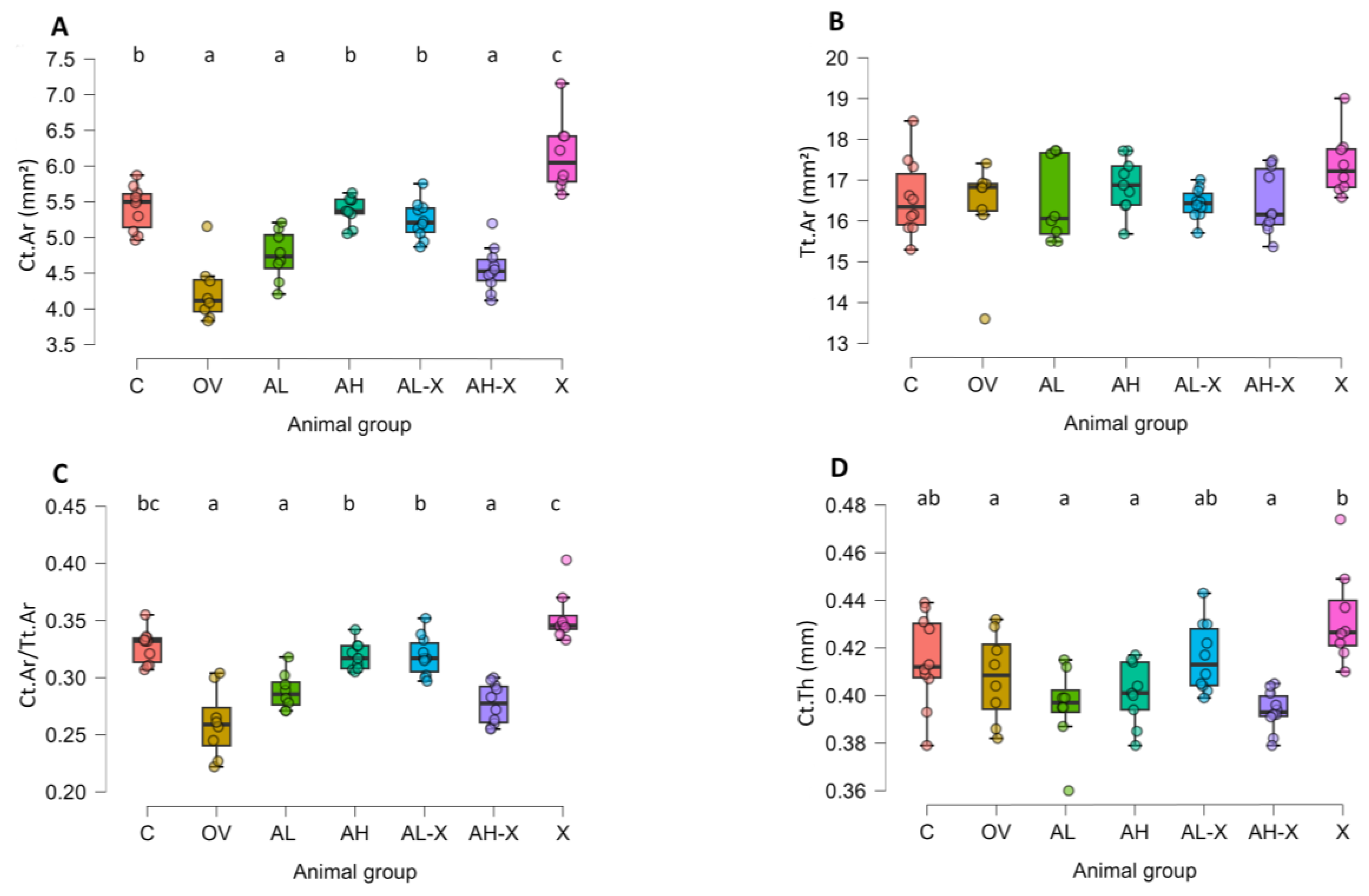
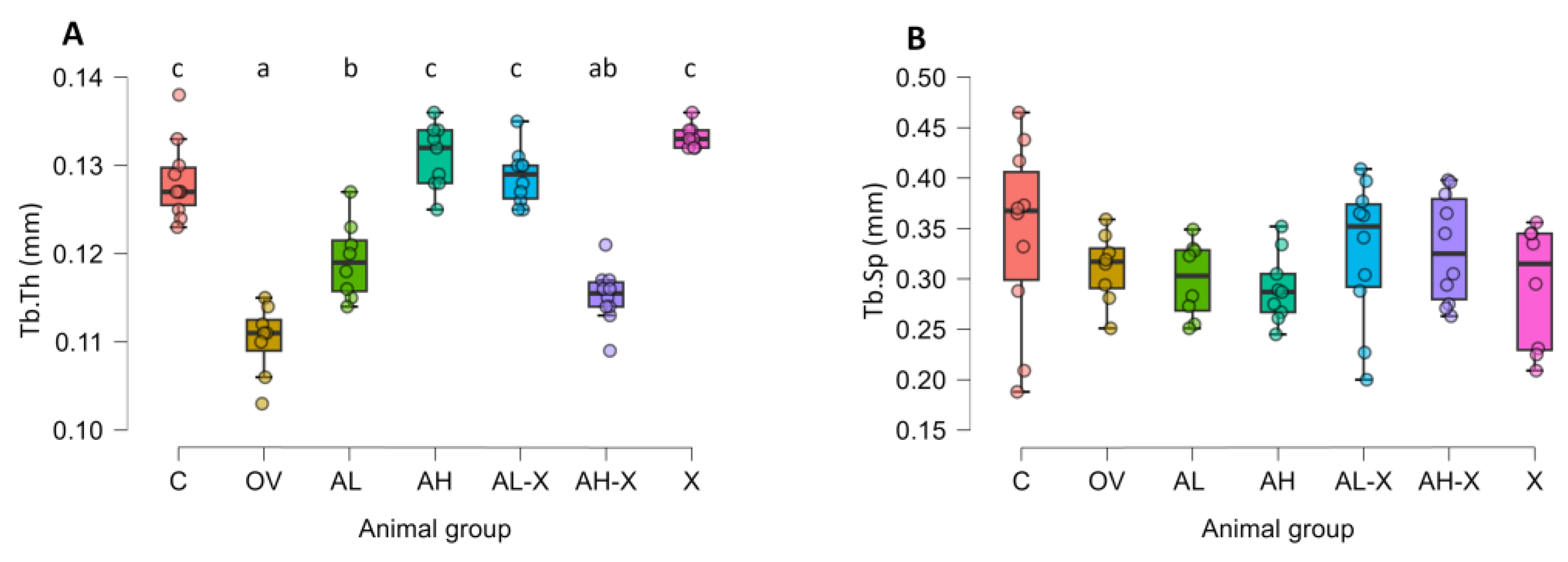
3.1. Micro-CT analysis
3.2. Hematology and serum biochemistry
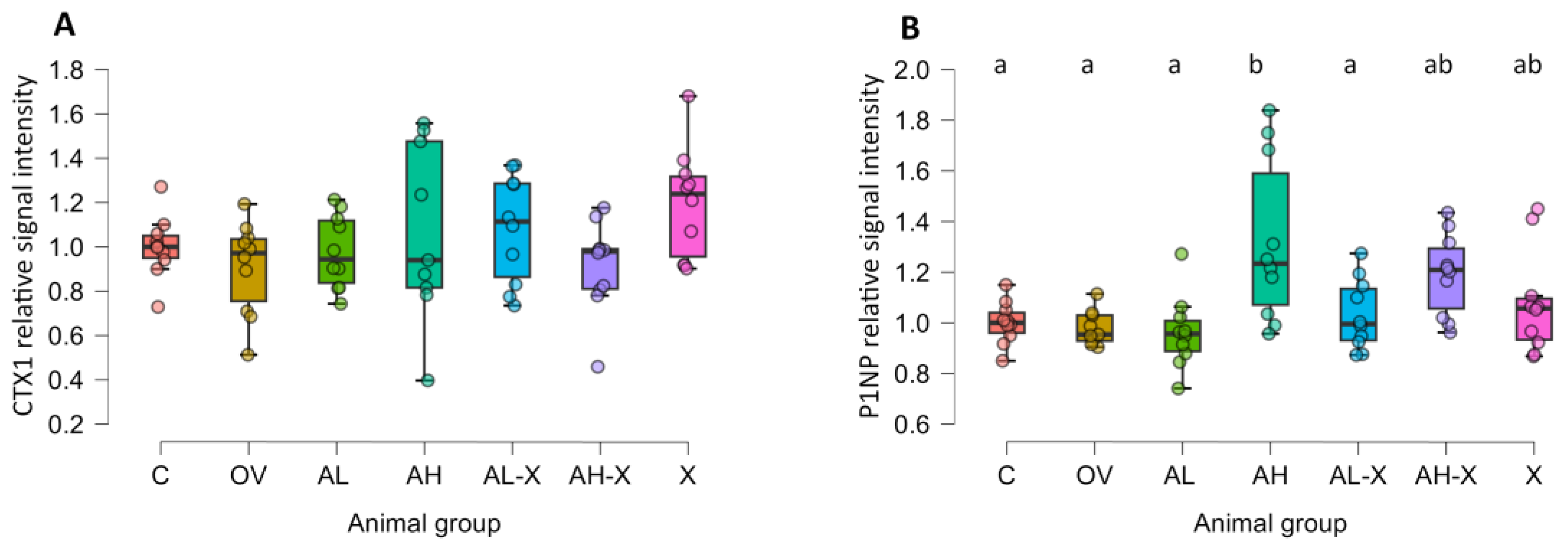
3.3. CTX1 and P1NP Western blot analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Rossini, M.; Adami, S.; Bertoldo, F.; Diacinti, D.; Gatti, D.; Giannini, S.; Giusti, A.; Malavolta, N.; Minisola, S.; Osella, G.; et al. Guidelines for the Diagnosis, Prevention and Management of Osteoporosis. Reumatismo 2016, 68, 1–39. [CrossRef]
- Bhatnagar, A.; Kekatpure, A.L. Postmenopausal Osteoporosis: A Literature Review. Cureus 14, e29367. [CrossRef]
- Kanis, J.A.; Norton, N.; Harvey, N.C.; Jacobson, T.; Johansson, H.; Lorentzon, M.; McCloskey, E. V.; Willers, C.; Borgström, F. SCOPE 2021: A New Scorecard for Osteoporosis in Europe. Arch. Osteoporos. 2021, 16, 1–82. [CrossRef]
- Rashki Kemmak, A.; Rezapour, A.; Jahangiri, R.; Nikjoo, S.; Farabi, H.; Soleimanpour, S. Economic Burden of Osteoporosis in the World: A Systematic Review. Med. J. Islam. Repub. Iran 2020, 34, 154. [CrossRef]
- Ji, M.-X.; Yu, Q. Primary Osteoporosis in Postmenopausal Women. Chronic Dis. Transl. Med. 2015, 1, 9–13. [CrossRef]
- Khosla, S.; Oursler, M.J.; Monroe, D.G. Estrogen and the Skeleton. Trends Endocrinol. Metab. 2012, 23, 576–581. [CrossRef]
- Prentice, R.L.; Anderson, G.L. The Women’s Health Initiative: Lessons Learned. Annu. Rev. Public Health 2008, 29, 131–150. [CrossRef]
- Liang, J.; Shang, Y. Estrogen and Cancer. Annu. Rev. Physiol. 2013, 75, 225–240. [CrossRef]
- LeBoff, M.S.; Greenspan, S.L.; Insogna, K.L.; Lewiecki, E.M.; Saag, K.G.; Singer, A.J.; Siris, E.S. The Clinician’s Guide to Prevention and Treatment of Osteoporosis. Osteoporos. Int. J. Establ. Result Coop. Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 2022, 1–54. [CrossRef]
- Langdahl, B.L. Overview of Treatment Approaches to Osteoporosis. Br. J. Pharmacol. 2021, 178, 1891–1906. [CrossRef]
- Li, H.; Xiao, Z.; Quarles, L.D.; Li, W. Osteoporosis: Mechanism, Molecular Target and Current Status on Drug Development. Curr. Med. Chem. 2021, 28, 1489–1507. [CrossRef]
- Pharmacology of Bisphosphonates - UpToDate Available online: https://www.uptodate.com/contents/pharmacology-of-bisphosphonates (accessed on 15 August 2023).
- Cummings, S.R.; Santora, A.C.; Black, D.M.; Russell, R.G.G. History of Alendronate. Bone 2020, 137, 115411. [CrossRef]
- Bauer, D.C.; Abrahamsen, B. Bisphosphonate Drug Holidays in Primary Care: When and What to Do Next? Curr. Osteoporos. Rep. 2021, 19, 182–188. [CrossRef]
- Park, J.-H.; Ko, H.-J. The Association between Treatment with Bisphosphonates and the Risk of Atrial Fibrillation: A Meta-Analysis of Observational Studies. Korean J. Fam. Med. 2022, 43, 69–76. [CrossRef]
- Drake, M.T.; Clarke, B.L.; Khosla, S. Bisphosphonates: Mechanism of Action and Role in Clinical Practice. Mayo Clin. Proc. 2008, 83, 1032–1045. [CrossRef]
- Aki, S.; Eskiyurt, N.; Akarirmak, U.; Tüzün, F.; Eryavuz, M.; Alper, S.; Arpacioğlu, O.; Atalay, F.; Kavuncu, V.; Kokino, S.; et al. Gastrointestinal Side Effect Profile Due to the Use of Alendronate in the Treatment of Osteoporosis. Yonsei Med. J. 2003, 44, 961–967. [CrossRef]
- Cremers, S.; Drake, M.T.; Ebetino, F.H.; Bilezikian, J.P.; Russell, R.G.G. Pharmacology of Bisphosphonates. Br. J. Clin. Pharmacol. 2019, 85, 1052–1062. [CrossRef]
- Kousteni, S.; Han, L.; Chen, J.-R.; Almeida, M.; Plotkin, L.I.; Bellido, T.; Manolagas, S.C. Kinase-Mediated Regulation of Common Transcription Factors Accounts for the Bone-Protective Effects of Sex Steroids. J. Clin. Invest. 2003, 111, 1651–1664. [CrossRef]
- Kousteni, S.; Bellido, T.; Plotkin, L.I.; O’Brien, C.A.; Bodenner, D.L.; Han, L.; Han, K.; DiGregorio, G.B.; Katzenellenbogen, J.A.; Katzenellenbogen, B.S.; et al. Nongenotropic, Sex-Nonspecific Signaling through the Estrogen or Androgen Receptors. Cell 2001, 104, 719–730. [CrossRef]
- Oršolić, N.; Nemrava, J.; Jeleč, Ž.; Kukolj, M.; Odeh, D.; Terzić, S.; Fureš, R.; Bagatin, T.; Bagatin, D. The Beneficial Effect of Proanthocyanidins and Icariin on Biochemical Markers of Bone Turnover in Rats. Int. J. Mol. Sci. 2018, 19, 2746. [CrossRef]
- Oršolić, N.; Nemrava, J.; Jeleč, Ž.; Kukolj, M.; Odeh, D.; Jakopović, B.; Jazvinšćak Jembrek, M.; Bagatin, T.; Fureš, R.; Bagatin, D. Antioxidative and Anti-Inflammatory Activities of Chrysin and Naringenin in a Drug-Induced Bone Loss Model in Rats. Int. J. Mol. Sci. 2022, 23, 2872. [CrossRef]
- Nie, X.; Jin, H.; Wen, G.; Xu, J.; An, J.; Liu, X.; Xie, R.; Tuo, B. Estrogen Regulates Duodenal Calcium Absorption Through Differential Role of Estrogen Receptor on Calcium Transport Proteins. Dig. Dis. Sci. 2020, 65, 3502–3513. [CrossRef]
- Perrone, P.; De Rosa, C.; D’Angelo, S. Polyphenols and Bone Health: A Comprehensive Review of Their Role in Osteoporosis Prevention and Treatment. Molecules 2025, 30, 4154. [CrossRef]
- Oršolić, N.; Nemrava, J.; Jeleč, Ž.; Kukolj, M.; Odeh, D.; Terzić, S.; Fureš, R.; Bagatin, T.; Bagatin, D. The Beneficial Effect of Proanthocyanidins and Icariin on Biochemical Markers of Bone Turnover in Rats. Int. J. Mol. Sci. 2018, 19, 2746. [CrossRef]
- Roseti, L.; Borciani, G.; Grassi, F.; Desando, G.; Gambari, L.; Grigolo, B. Nutraceuticals in Osteoporosis Prevention. Front. Nutr. 2024, 11. [CrossRef]
- Faienza, M.F.; Giardinelli, S.; Annicchiarico, A.; Chiarito, M.; Barile, B.; Corbo, F.; Brunetti, G. Nutraceuticals and Functional Foods: A Comprehensive Review of Their Role in Bone Health. Int. J. Mol. Sci. 2024, 25, 5873. [CrossRef]
- Li, Y.; Li, L.; Li, X.; Luo, B.; Ye, Q.; Wang, H.; Yang, L.; Zhu, X.; Han, L.; Zhang, R.; et al. A Mechanistic Review of Chinese Medicine Polyphenols on Bone Formation and Resorption. Front. Pharmacol. 2022, 13. [CrossRef]
- Tomczyk-Warunek, A.; Winiarska-Mieczan, A.; Blicharski, T.; Blicharski, R.; Kowal, F.; Pano, I.T.; Tomaszewska, E.; Muszyński, S. Consumption of Phytoestrogens Affects Bone Health by Regulating Estrogen Metabolism. J. Nutr. 2024, 154, 2611–2627. [CrossRef]
- Domínguez-López, I.; Yago-Aragón, M.; Salas-Huetos, A.; Tresserra-Rimbau, A.; Hurtado-Barroso, S. Effects of Dietary Phytoestrogens on Hormones throughout a Human Lifespan: A Review. Nutrients 2020, 12, 2456. [CrossRef]
- Viggiani, M.T.; Polimeno, L.; Di Leo, A.; Barone, M. Phytoestrogens: Dietary Intake, Bioavailability, and Protective Mechanisms against Colorectal Neoproliferative Lesions. Nutrients 2019, 11, 1709. [CrossRef]
- Desmawati, D.; Sulastri, D. Phytoestrogens and Their Health Effect. Open Access Maced. J. Med. Sci. 2019, 7, 495–499. [CrossRef]
- Keiler, A.M.; Helle, J.; Bader, M.I.; Ehrhardt, T.; Nestler, K.; Kretzschmar, G.; Bernhardt, R.; Vollmer, G.; Nikolić, D.; Bolton, J.L.; et al. A Standardized Humulus Lupulus (L.) Ethanol Extract Partially Prevents Ovariectomy-Induced Bone Loss in the Rat without Induction of Adverse Effects in the Uterus. Phytomedicine 2017, 34, 50–58. [CrossRef]
- Hümpel, M.; Isaksson, P.; Schaefer, O.; Kaufmann, U.; Ciana, P.; Maggi, A.; Schleuning, W.-D. Tissue Specificity of 8-Prenylnaringenin: Protection from Ovariectomy Induced Bone Loss with Minimal Trophic Effects on the Uterus. J. Steroid Biochem. Mol. Biol. 2005, 97, 299–305. [CrossRef]
- van Breemen, R.B.; Yuan, Y.; Banuvar, S.; Shulman, L.P.; Qiu, X.; Alvarenga, R.F.R.; Chen, S.-N.; Dietz, B.M.; Bolton, J.L.; Pauli, G.F.; et al. Pharmacokinetics of Prenylated Hop Phenols in Women Following Oral Administration of a Standardized Extract of Hops. Mol. Nutr. Food Res. 2014, 58, 1962–1969. [CrossRef]
- Paraiso, I.L.; Plagmann, L.S.; Yang, L.; Zielke, R.; Gombart, A.F.; Maier, C.S.; Sikora, A.E.; Blakemore, P.R.; Stevens, J.F. Reductive Metabolism of Xanthohumol and 8-Prenylnaringenin by the Intestinal Bacterium Eubacterium Ramulus. Mol. Nutr. Food Res. 2019, 63, e1800923. [CrossRef]
- Effect of a Hop Extract Standardized in 8-Prenylnaringenin on Bone Health and Gut Microbiome in Postmenopausal Women with Osteopenia: A One-Year Randomized, Double-Blind, Placebo-Controlled Trial Available online: https://www.mdpi.com/2072-6643/15/12/2688 (accessed on 6 October 2025).
- Chu, X.; Xing, H.; Chao, M.; Xie, P.; Jiang, L. Gut Microbiota Modulation in Osteoporosis: Probiotics, Prebiotics, and Natural Compounds. Metabolites 2025, 15, 301. [CrossRef]
- Štulíková, K.; Karabín, M.; Nešpor, J.; Dostálek, P. Therapeutic Perspectives of 8-Prenylnaringenin, a Potent Phytoestrogen from Hops. Molecules 2018, 23, 660. [CrossRef]
- Wanionok, N.E.; Colareda, G.A.; Fernandez, J.M. In Vitro Effects and Mechanisms of Humulus Lupulus Extract on Bone Marrow Progenitor Cells and Endothelial Cells. Mol. Cell. Endocrinol. 2024, 592, 112328. [CrossRef]
- Wanionok, N.E.; Colareda, G.A.; Fernandez, J.M. Humulus Lupulus Promoting Osteoblast Activity and Bone Integrity: Effects and Mechanisms. Biology 2025, 14, 582. [CrossRef]
- National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals; National Academies Press: Washington, D.C., 2011; ISBN 978-0-309-15400-0.
- Zhang, R.; Yang, M.; Li, Y.; Liu, H.; Ren, M.; Tao, Z.-S. Effect of Alendronate on the Femoral Metaphyseal Defect under Carbamazepine in Ovariectomized Rats. J. Orthop. Surg. 2021, 16, 14. [CrossRef]
- Ban, Y.-H.; Yon, J.-M.; Cha, Y.; Choi, J.; An, E.S.; Guo, H.; Seo, D.W.; Kim, T.-S.; Lee, S.-P.; Kim, J.-C.; et al. A Hop Extract Lifenol® Improves Postmenopausal Overweight, Osteoporosis, and Hot Flash in Ovariectomized Rats. Evid.-Based Complement. Altern. Med. ECAM 2018, 2018, 2929107. [CrossRef]
- Fu, L.; Tang, T.; Hao, Y.; Dai, K. Long-Term Effects of Alendronate on Fracture Healing and Bone Remodeling of Femoral Shaft in Ovariectomized Rats. Acta Pharmacol. Sin. 2013, 34, 387–392. [CrossRef]
- Tokmak Özşahin, E.T.; Çam, B.; Dere, F.; Kürkçü, M.; Evrüke, C.; Soames, R.; Oğuz, Ö. The Effect of Alendronate Sodium on Trabecular Bone Structure in an Osteoporotic Rat Model. Turk. J. Phys. Med. Rehabil. 2017, 63, 165–173. [CrossRef]
- Nowak, B.; Poźniak, B.; Popłoński, J.; Bobak, Ł.; Matuszewska, A.; Kwiatkowska, J.; Dziewiszek, W.; Huszcza, E.; Szeląg, A. Pharmacokinetics of Xanthohumol in Rats of Both Sexes after Oral and Intravenous Administration of Pure Xanthohumol and Prenylflavonoid Extract. Adv. Clin. Exp. Med. Off. Organ Wroclaw Med. Univ. 2020, 29, 1101–1109. [CrossRef]
- Mukai, R.; Hata, N. Tissue Distribution and Pharmacokinetics of Isoxanthohumol from Hops in Rodents. Food Sci. Nutr. 2024, 12, 2210–2219. [CrossRef]
- Ronneberger, O.; Fischer, P.; Brox, T. U-Net: Convolutional Networks for Biomedical Image Segmentation. In Proceedings of the Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics); Springer Verlag, 2015; Vol. 9351, pp. 234–241.
- Buie, H.R.; Campbell, G.M.; Klinck, R.J.; MacNeil, J.A.; Boyd, S.K. Automatic Segmentation of Cortical and Trabecular Compartments Based on a Dual Threshold Technique for in Vivo Micro-CT Bone Analysis. Bone 2007, 41, 505–515. [CrossRef]
- Bouxsein, M.L.; Boyd, S.K.; Christiansen, B.A.; Guldberg, R.E.; Jepsen, K.J.; Müller, R. Guidelines for Assessment of Bone Microstructure in Rodents Using Micro-Computed Tomography. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2010, 25, 1468–1486. [CrossRef]
- JASP Team JASP (Version 0.95.2) [Computer Software] 2025.
- Callewaert, F.; Sinnesael, M.; Gielen, E.; Boonen, S.; Vanderschueren, D. Skeletal Sexual Dimorphism: Relative Contribution of Sex Steroids, GH–IGF1, and Mechanical Loading. J. Endocrinol. 2010, 207, 127–134. [CrossRef]
- Chen, X.; Wang, Z.; Duan, N.; Zhu, G.; Schwarz, E.M.; Xie, C. Osteoblast–Osteoclast Interactions. Connect. Tissue Res. 2018, 59, 99–107. [CrossRef]
- Luo, D.; Kang, L.; Ma, Y.; Chen, H.; Kuang, H.; Huang, Q.; He, M.; Peng, W. Effects and Mechanisms of 8-Prenylnaringenin on Osteoblast MC3T3-E1 and Osteoclast-like Cells RAW264.7. Food Sci. Nutr. 2014, 2, 341–350. [CrossRef]
- Kim, H.-N.; Ponte, F.; Nookaew, I.; Ucer Ozgurel, S.; Marques-Carvalho, A.; Iyer, S.; Warren, A.; Aykin-Burns, N.; Krager, K.; Sardao, V.A.; et al. Estrogens Decrease Osteoclast Number by Attenuating Mitochondria Oxidative Phosphorylation and ATP Production in Early Osteoclast Precursors. Sci. Rep. 2020, 10, 11933. [CrossRef]
- Weinstein, R.S.; Roberson, P.K.; Manolagas, S.C. Giant Osteoclast Formation and Long-Term Oral Bisphosphonate Therapy. N. Engl. J. Med. 2009, 360, 53–62. [CrossRef]
- Norton, J.T.; Hayashi, T.; Crain, B.; Cho, J.S.; Miller, L.S.; Corr, M.; Carson, D.A. Cutting Edge: Nitrogen Bisphosphonate-Induced Inflammation Is Dependent upon Mast Cells and IL-1. J. Immunol. Baltim. Md 1950 2012, 188, 2977–2980. [CrossRef]
- Oršolić, N.; Jazvinšćak Jembrek, M. Molecular and Cellular Mechanisms of Propolis and Its Polyphenolic Compounds against Cancer. Int. J. Mol. Sci. 2022, 23, 10479. [CrossRef]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and Prooxidant Properties of Flavonoids. Fitoterapia 2011, 82, 513–523. [CrossRef]
- Simunkova, M.; Barbierikova, Z.; Jomova, K.; Hudecova, L.; Lauro, P.; Alwasel, S.H.; Alhazza, I.; Rhodes, C.J.; Valko, M. Antioxidant vs. Prooxidant Properties of the Flavonoid, Kaempferol, in the Presence of Cu(II) Ions: A ROS-Scavenging Activity, Fenton Reaction and DNA Damage Study. Int. J. Mol. Sci. 2021, 22, 1619. [CrossRef]
- Vona, R.; Gambardella, L.; Ortona, E.; Santulli, M.; Malorni, W.; Carè, A.; Pietraforte, D.; Straface, E. Functional Estrogen Receptors of Red Blood Cells. Do They Influence Intracellular Signaling? Cell. Physiol. Biochem. 2019, 53, 186–199. [CrossRef]
- Sánchez-Aguilera, A.; Arranz, L.; Martín-Pérez, D.; García-García, A.; Stavropoulou, V.; Kubovcakova, L.; Isern, J.; Martín-Salamanca, S.; Langa, X.; Skoda, R.C.; et al. Estrogen Signaling Selectively Induces Apoptosis of Hematopoietic Progenitors and Myeloid Neoplasms without Harming Steady-State Hematopoiesis. Cell Stem Cell 2014, 15, 791–804. [CrossRef]
- Roma, A.; Spagnuolo, P.A. Estrogen Receptors Alpha and Beta in Acute Myeloid Leukemia. Cancers 2020, 12, 907. [CrossRef]
- Butkiewicz, A.M.; Kemona, H.; Dymicka-Piekarska, V.; Matowicka-Karna, J. Does Menopause Affect Thrombocytopoiesis and Platelet Activation? Przegl. Lek. 2006, 63, 1291–1293.
- Kim, J.; Kim, H.S.; Lee, H.S.; Kwon, Y.-J. The Relationship between Platelet Count and Bone Mineral Density: Results from Two Independent Population-Based Studies. Arch. Osteoporos. 2020, 15, 43. [CrossRef]
- Kristjansdottir, H.L.; Mellström, D.; Johansson, P.; Karlsson, M.; Vandenput, L.; Lorentzon, M.; Herlitz, H.; Ohlsson, C.; Lerner, U.H.; Lewerin, C. High Platelet Count Is Associated with Low Bone Mineral Density: The MrOS Sweden Cohort. Osteoporos. Int. 2021, 32, 865–871. [CrossRef]
- Kim, H.-L.; Cho, H.Y.; Park, I.Y.; Choi, J.M.; Kim, M.; Jang, H.J.; Hwang, S.-M. The Positive Association between Peripheral Blood Cell Counts and Bone Mineral Density in Postmenopausal Women. Yonsei Med. J. 2011, 52, 739. [CrossRef]
- Dupuis, M.; Severin, S.; Noirrit-Esclassan, E.; Arnal, J.-F.; Payrastre, B.; Valéra, M.-C. Effects of Estrogens on Platelets and Megakaryocytes. Int. J. Mol. Sci. 2019, 20, 3111. [CrossRef]
- Do, H.J.; Shin, J.-S.; Lee, J.; Lee, Y.J.; Kim, M.; Nam, D.; Kim, E.-J.; Park, Y.; Suhr, K.; Ha, I.-H. Association between Liver Enzymes and Bone Mineral Density in Koreans: A Cross-Sectional Study. BMC Musculoskelet. Disord. 2018, 19, 410. [CrossRef]
- Breitling, L.P. Liver Enzymes and Bone Mineral Density in the General Population. J. Clin. Endocrinol. Metab. 2015, 100, 3832–3840. [CrossRef]
- Bisphosphonates; National Institute of Diabetes and Digestive and Kidney Diseases, 2012;
- Turner, A.S.; Alvis, M.; Myers, W.; Stevens, M.L.; Lundy, M.W. Changes in Bone Mineral Density and Bone-Specific Alkaline Phosphatase in Ovariectomized Ewes. Bone 1995, 17, 395–402. [CrossRef]
- Afshinnia, F.; Wong, K.K.; Sundaram, B.; Ackermann, R.J.; Pennathur, S. Hypoalbuminemia and Osteoporosis: Reappraisal of a Controversy. J. Clin. Endocrinol. Metab. 2016, 101, 167–175. [CrossRef]
- El-Kafoury, B.; Mohamed, F.; Bahgat, N.; El Samad, A.A.; Shawky, M.; Abdel-Hady, E.A. Failure of Subcutaneous Lipectomy to Combat Metabolic Dysregulations in Ovariectomy-Induced Obesity in Young Female Rats. Hormones 2022, 21, 421–436. [CrossRef]
- Park, S.; Sim, K.-S.; Hwangbo, Y.; Park, S.-J.; Kim, Y.-J.; Kim, J.-H. Naringenin and Phytoestrogen 8-Prenylnaringenin Protect against Islet Dysfunction and Inhibit Apoptotic Signaling in Insulin-Deficient Diabetic Mice. Molecules 2022, 27, 4227. [CrossRef]
- Pohjanvirta, R.; Nasri, A. The Potent Phytoestrogen 8-Prenylnaringenin: A Friend or a Foe? Int. J. Mol. Sci. 2022, 23, 3168. [CrossRef]
- Yoon, S.; Lee, J.; Lee, S.-M. Combined Treatment of Isoflavone Supplementation and Exercise Restores the Changes in Hepatic Protein Expression in Ovariectomized Rats - a Proteomics Approach. J. Int. Soc. Sports Nutr. 2014, 11, 29. [CrossRef]
- Papadaki, H.A.; Tsatsanis, C.; Christoforidou, A.; Malliaraki, N.; Psyllaki, M.; Pontikoglou, C.; Miliaki, M.; Margioris, A.N.; Eliopoulos, G.D. Alendronate Reduces Serum TNFalpha and IL-1beta, Increases Neutrophil Counts, and Improves Bone Mineral Density and Bone Metabolism Indices in Patients with Chronic Idiopathic Neutropenia (CIN)-Associated Osteopenia/Osteoporosis. J. Bone Miner. Metab. 2004, 22, 577–587. [CrossRef]
- Duque, G.; Rivas, D. Alendronate Has an Anabolic Effect on Bone Through the Differentiation of Mesenchymal Stem Cells. J. Bone Miner. Res. 2007, 22, 1603–1611. [CrossRef]
- Qvist, P.; Christgau, S.; Pedersen, B.J.; Schlemmer, A.; Christiansen, C. Circadian Variation in the Serum Concentration of C-Terminal Telopeptide of Type I Collagen (Serum CTx): Effects of Gender, Age, Menopausal Status, Posture, Daylight, Serum Cortisol, and Fasting. Bone 2002, 31, 57–61. [CrossRef]
- Song, L.; Bi, Y.; Zhang, P.; Yuan, X.; Liu, Y.; Zhang, Y.; Huang, J.; Zhou, K. Optimization of the Time Window of Interest in Ovariectomized Imprinting Control Region Mice for Antiosteoporosis Research. BioMed Res. Int. 2017, 2017, 1–10. [CrossRef]
- Kim, M.R.; Kim, H.J.; Yu, S.H.; Lee, B.S.; Jeon, S.Y.; Lee, J.J.; Lee, Y.C. Combination of Red Clover and Hops Extract Improved Menopause Symptoms in an Ovariectomized Rat Model. Evid. Based Complement. Alternat. Med. 2020, 2020, 1–9. [CrossRef]
- Sathyapalan, T.; Aye, M.; Rigby, A.S.; Fraser, W.D.; Thatcher, N.J.; Kilpatrick, E.S.; Atkin, S.L. Soy Reduces Bone Turnover Markers in Women During Early Menopause: A Randomized Controlled Trial. J. Bone Miner. Res. 2017, 32, 157–164. [CrossRef]
- Ming, L.-G.; Lv, X.; Ma, X.-N.; Ge, B.-F.; Zhen, P.; Song, P.; Zhou, J.; Ma, H.-P.; Xian, C.J.; Chen, K.-M. The Prenyl Group Contributes to Activities of Phytoestrogen 8-Prenynaringenin in Enhancing Bone Formation and Inhibiting Bone Resorption in Vitro. Endocrinology 2013, 154, 1202–1214. [CrossRef]
- Štulíková, K.; Karabín, M.; Nešpor, J.; Dostálek, P. Therapeutic Perspectives of 8-Prenylnaringenin, a Potent Phytoestrogen from Hops. Molecules 2018, 23, 660. [CrossRef]
- Schaefer, O.; Hümpel, M.; Fritzemeier, K.-H.; Bohlmann, R.; Schleuning, W.-D. 8-Prenyl Naringenin Is a Potent ERα Selective Phytoestrogen Present in Hops and Beer. J. Steroid Biochem. Mol. Biol. 2003, 84, 359–360. [CrossRef]
- Rođak, E.; Oršolić, N.; Grgac, R.; Rajc, J.; Bakula, M.; Bijelić, N. Analysis of Uterine Morphology in Ovariectomized Rats Treated With Alendronate and Hop Extract Using Open-Source Software. Image Anal. Stereol. 2022, 41, 193–202. [CrossRef]
- Ts, X.; Ly, L.; Qy, Z.; Yp, J.; Ch, L.; Xy, L.; Lp, Q.; Hl, X. Humulus Lupulus L. Extract Prevents Ovariectomy-Induced Osteoporosis in Mice and Regulates Activities of Osteoblasts and Osteoclasts. Chin. J. Integr. Med. 2021, 27. [CrossRef]
- Nowak, B.; Matuszewska, A.; Popłoński, J.; Nikodem, A.; Filipiak, J.; Tomanik, M.; Dziewiszek, W.; Danielewski, M.; Belowska-Bień, K.; Kłobucki, M.; et al. Prenylflavonoids Counteract Ovariectomy-Induced Disturbances in Rats. J. Funct. Foods 2021, 86, 104742. [CrossRef]
- Lecomte, M.; Tomassi, D.; Rizzoli, R.; Tenon, M.; Berton, T.; Harney, S.; Fança-Berthon, P. Effect of a Hop Extract Standardized in 8-Prenylnaringenin on Bone Health and Gut Microbiome in Postmenopausal Women with Osteopenia: A One-Year Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2023, 15, 2688. [CrossRef]
- Sansai, K.; Na Takuathung, M.; Khatsri, R.; Teekachunhatean, S.; Hanprasertpong, N.; Koonrungsesomboon, N. Effects of Isoflavone Interventions on Bone Mineral Density in Postmenopausal Women: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Osteoporos. Int. J. Establ. Result Coop. Eur. Found. Osteoporos. Natl. Osteoporos. Found. USA 2020, 31, 1853–1864. [CrossRef]
- Tomczyk-Warunek, A.; Winiarska-Mieczan, A.; Blicharski, T.; Blicharski, R.; Kowal, F.; Pano, I.T.; Tomaszewska, E.; Muszyński, S. Consumption of Phytoestrogens Affects Bone Health by Regulating Estrogen Metabolism. J. Nutr. 2024, 154, 2611–2627. [CrossRef]
- Pecyna, P.; Wargula, J.; Murias, M.; Kucinska, M. More Than Resveratrol: New Insights into Stilbene-Based Compounds. Biomolecules 2020, 10, 1111. [CrossRef]
- Xu, S.; Xia, T.; Zhang, J.; Jiang, Y.; Wang, N.; Xin, H. Protective Effects of Bitter Acids from Humulus Lupulus L. against Senile Osteoporosis via Activating Nrf2/HO-1/NQO1 Pathway in D-Galactose Induced Aging Mice. J. Funct. Foods 2022, 94, 105099. [CrossRef]
- Hümpel, M.; Isaksson, P.; Schaefer, O.; Kaufmann, U.; Ciana, P.; Maggi, A.; Schleuning, W.-D. Tissue Specificity of 8-Prenylnaringenin: Protection from Ovariectomy Induced Bone Loss with Minimal Trophic Effects on the Uterus. J. Steroid Biochem. Mol. Biol. 2005, 97, 299–305. [CrossRef]
- Aghamiri, V.; Mirghafourvand, M.; Mohammad-Alizadeh-Charandabi, S.; Nazemiyeh, H. The Effect of Hop (Humulus Lupulus L.) on Early Menopausal Symptoms and Hot Flashes: A Randomized Placebo-Controlled Trial. Complement. Ther. Clin. Pract. 2016, 23, 130–135. [CrossRef]
- Sengupta, P. The Laboratory Rat: Relating Its Age With Human’s. Int. J. Prev. Med. 2013, 4, 624–630.
- Yoo, Y.-S.; Kim, M.-G.; Park, H.-J.; Chae, M.-Y.; Choi, Y.-J.; Oh, C.-K.; Son, C.-G.; Lee, E.-J. Additional Effects of Herbal Medicine Combined with Bisphosphonates for Primary Osteoporosis: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2024, 15, 1413515. [CrossRef]
- Dc, C.; K, R.; Kt, K.; Jk, S. The Therapeutic Effects of Combination Therapy with Curcumin and Alendronate on Spine Fusion Surgery in the Ovariectomized Rats. Korean J. Spine 2017, 14. [CrossRef]
- S, M.; S, V.; F, Z.; A, S.; M, J.; S, S.; D, R.; A, V.; O, A.; M, S.; et al. Quercetin Potentiates the Anti-Osteoporotic Effects of Alendronate through Modulation of Autophagy and Apoptosis Mechanisms in Ovariectomy-Induced Bone Loss Rat Model. Mol. Biol. Rep. 2023, 50. [CrossRef]
- Cho, D.-C.; Ryu, K.; Kim, K.-T.; Sung, J.-K. The Therapeutic Effects of Combination Therapy with Curcumin and Alendronate on Spine Fusion Surgery in the Ovariectomized Rats. Korean J. Spine 2017, 14, 35–40. [CrossRef]
- Yamaguchi, M.; Levy, R.M. Combination of Alendronate and Genistein Synergistically Suppresses Osteoclastic Differentiation of RAW267.4 Cells in Vitro. Exp. Ther. Med. 2017, 14, 1769. [CrossRef]
- Kim, M.R.; Kim, H.J.; Yu, S.H.; Lee, B.S.; Jeon, S.Y.; Lee, J.J.; Lee, Y.C. Combination of Red Clover and Hops Extract Improved Menopause Symptoms in an Ovariectomized Rat Model. Evid.-Based Complement. Altern. Med. ECAM 2020, 2020, 7941391. [CrossRef]
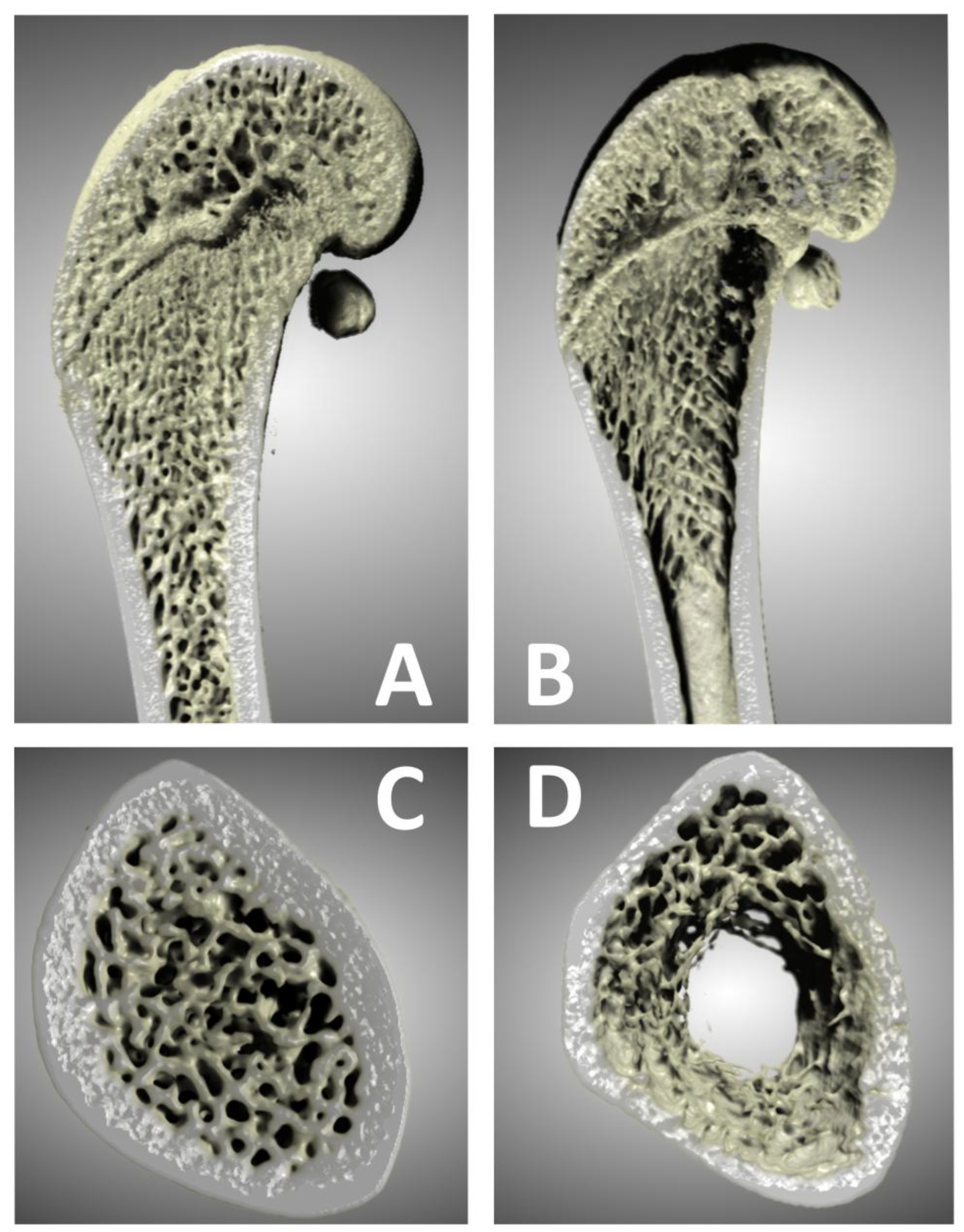
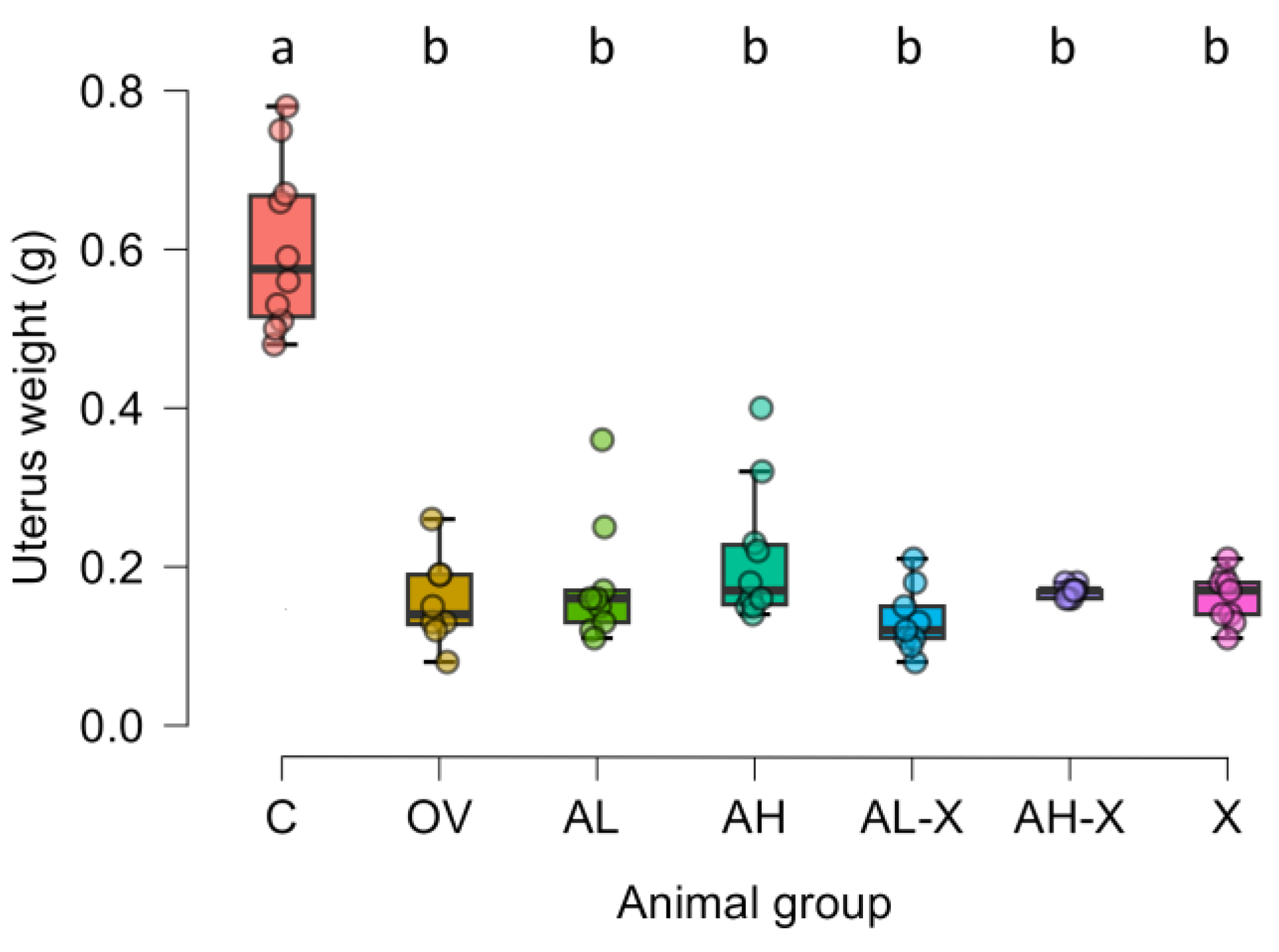
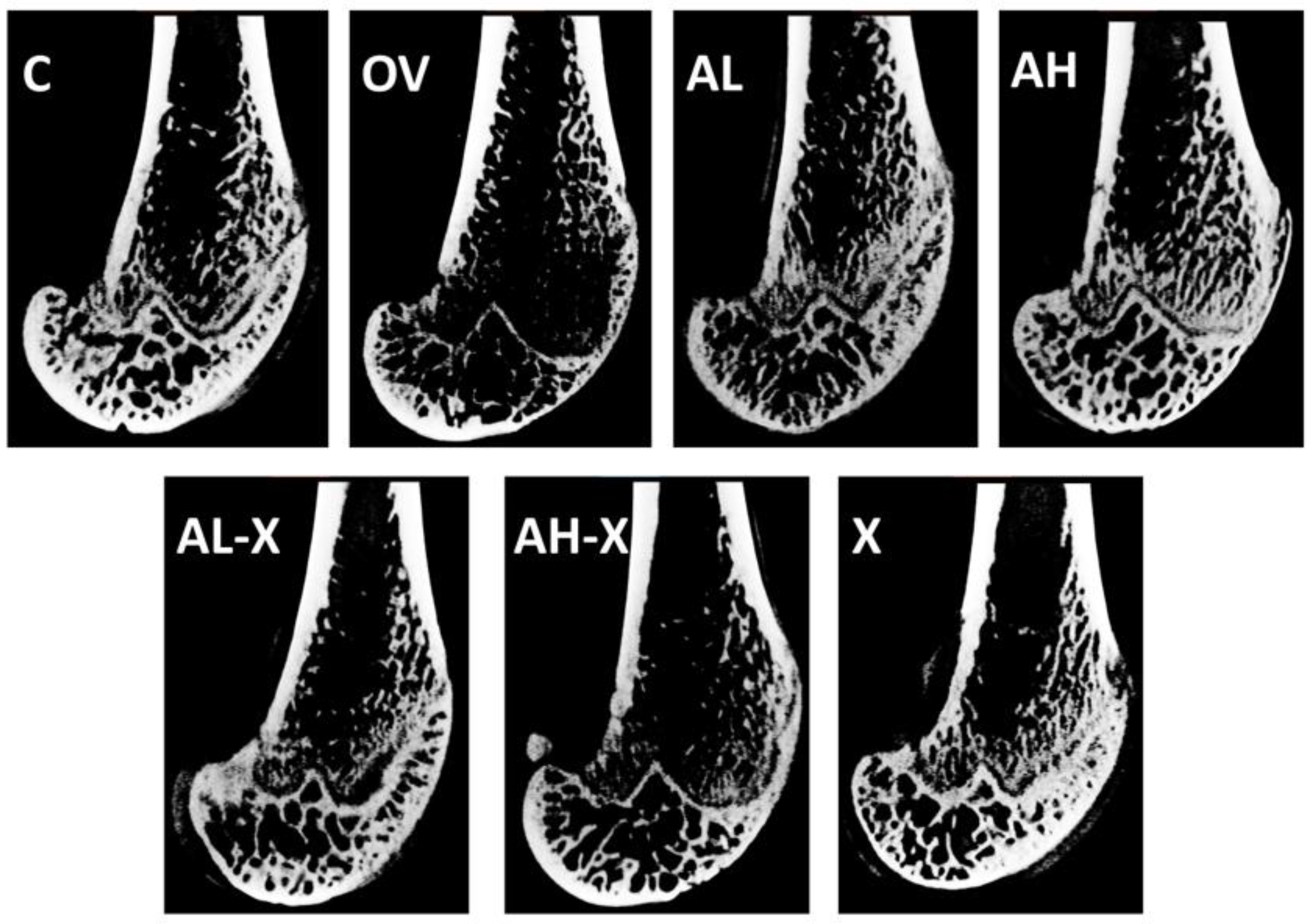
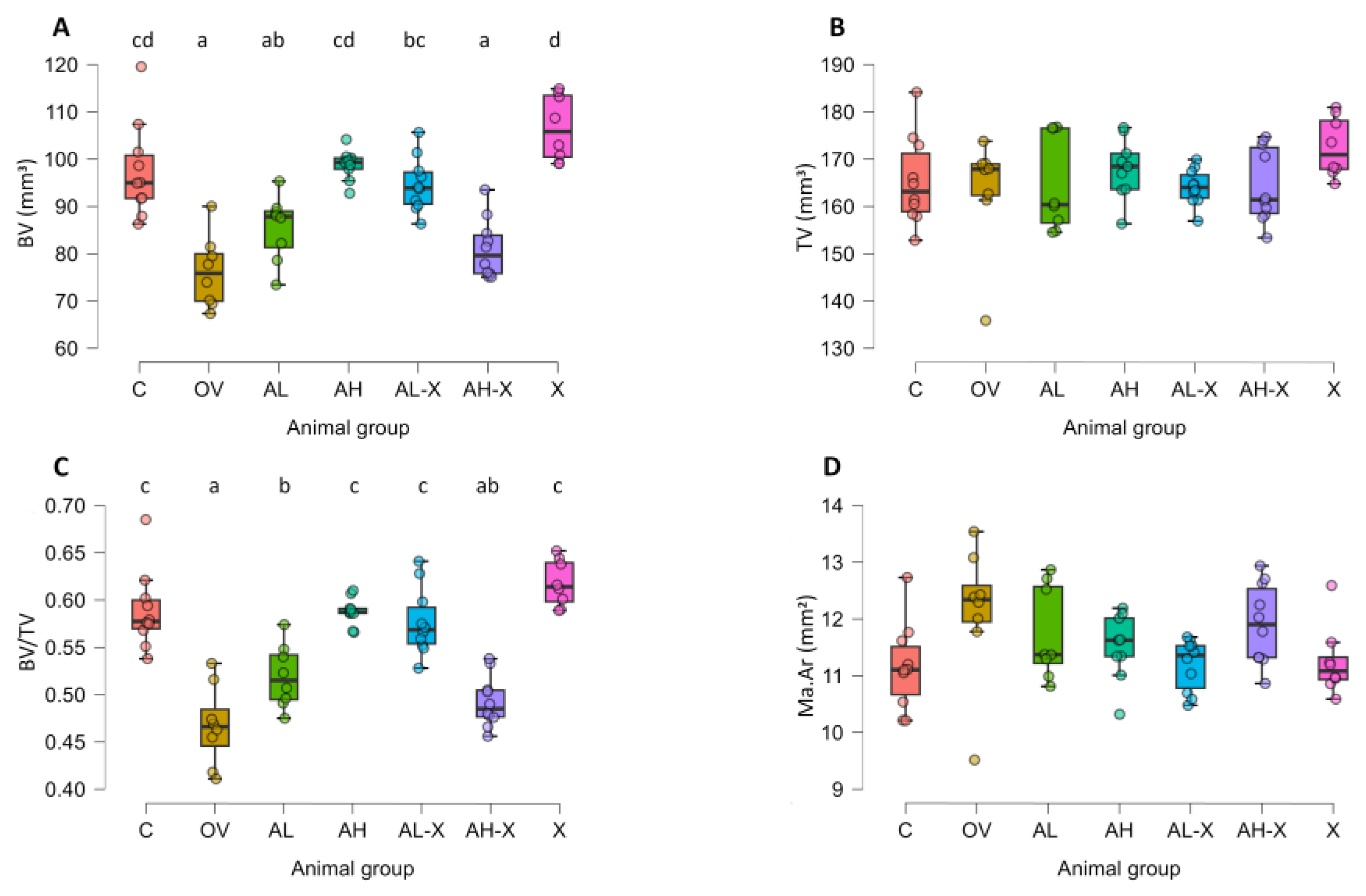

| Group | RBC (×1012/L) | WBC (×109/L) | PLT (×109/L) | Hb (g/L) | Hct (L/L) | MCV (fL) | MCH (pg) | MCHC (g/L) |
| C | 6.96 (0.26) | 3.52 (0.9) | 716.5 (37.73) | 152.2 (5.2) | 0.41 (0.01) | 58.4 (1.96) | 21.89 (0.68) | 375.1 (8.03) |
| OV | 7.82 (0.51) | 5.3 (2.79) | 623 (64.2) | 170.14 (11.33) | 0.46 (0.03) | 58.29 (2.63) | 21.8 (1.07) | 373.29 (4.82) |
| AL | 7.79 (0.81) | 4.31 (1.14) | 594.38 (104.03) | 175.88 (17.87) | 0.46 (0.05) | 58.75 (1.04) | 22.56 (0.73) | 384.63 (10.68) |
| AH | 7.27 (0.63) | 5.22 (1.21) | 543.44 (94.71) | 167.89 (10.35) | 0.43 (0.03) | 58.89 (1.36) | 23.21 (1.95) | 394.89 (29.17) |
| AL-X | 7.99 (0.32) | 3.92 (1.17) | 556.2 (98.3) | 176.7 (17.73) | 0.48 (0.05) | 59.4 (3.95) | 22.37 (1.34) | 376.22 (6.3) |
| AH-X | 7.32 (0.81) | 5.22 (1.31) | 507.2 (90.08) | 166.8 (11.61) | 0.43 (0.04) | 59.5 (1.9) | 22.88 (1.22) | 384.6 (10.74) |
| X | 7.02 (0.49) | 5.57 (0.9) | 620.83 (29.71) | 161.5 (9.2) | 0.41 (0.02) | 59.17 (1.17) | 23.02 (0.57) | 390 (8.83) |
| p | <0.001 | 0.005 | <0.001 | <0.001 | 0.001 | 0.88 | 0.04 | 0.005 |
| Group | Ca (mmol/L) | P (mmol/L) | Na (mmol/L) | K (mmol/L) | Glc (mmol/L) | BUN (mmol/L) | CRE (µmol/L) |
| C | 2.73 (0.09) | 2.13 (0.27) | 131.3 (6.02) | 4.52 (0.39) | 13.7 (2.49) | 5.66 (0.91) | 39.7 (12.4) |
| OV | 2.62 (0.05) | 2.11 (0.24) | 131.57 (4.43) | 4.5 (0.45) | 15.4 (2.95) | 5.79 (1.05) | 36.14 (8.15) |
| AL | 2.66 (0.14) | 1.98 (0.3) | 131.5 (4.9) | 4.59 (0.31) | 15.35 (2.31) | 5.95 (0.64) | 38.13 (7) |
| AH | 2.58 (0.1) | 1.97 (0.5) | 127.67 (7.31) | 4.7 (0.64) | 15.86 (1.94) | 6 (0.93) | 27 (14.59) |
| AL-X | 2.67 (0.09) | 2.06 (0.2) | 132.3 (3.86) | 4.67 (0.57) | 16.25 (1.59) | 5.64 (0.47) | 45.7 (16.61) |
| AH-X | 2.66 (0.05) | 2.08 (0.21) | 132.5 (4.74) | 4.81 (0.5) | 15.68 (1.87) | 6.47 (0.57) | 42.2 (19.05) |
| X | 2.52 (0.31) | 2.1 (0.24) | 132 (4.86) | 4.7 (0.89) | 15.19 (1.34) | 6.49 (1) | 56.67 (14.54) |
| p | 0.06 | 0.94 | 0.81 | 0.85 | 0.34 | 0.08 | 0.09 |
| Group* | ALP (U/L) | ALT (U/L) | TBIL (mmol/L) | AMY (U/L) | ALB (g/L) | GLO (g/L) | TP (g/L) |
| C | 66.1 (29.34) | 24.1 (10.72) | 4.5 (0.53) | 508.2 (100.11) | 52.6 (3.95) | 12 (3.27) | 64.6 (2.63) |
| OV | 97.57 (40.48) | 21.86 (7.34) | 4.71 (1.5) | 454.14 (59.68) | 46.29 (4.5) | 12.29 (4.72) | 58.57 (3.51) |
| AL | 68.13 (19.42) | 28.25 (19.25) | 5 (0.93) | 477.63 (63.88) | 46.75 (4.5) | 14 (2.27) | 60.75 (3.73) |
| AH | 92 (33.99) | 21.33 (9.68) | 4.78 (0.44) | 512.33 (53.84) | 50.11 (5.06) | 10.44 (1.42) | 60.67 (4.44) |
| AL-X | 90.9 (43.46) | 32.7 (18.3) | 4.8 (0.79) | 468.5 (71.53) | 49.4 (5.17) | 12.3 (2.31) | 61.6 (4.48) |
| AH-X | 102.2 (49.17) | 44.4 (33.24) | 4.8 (0.63) | 492.3 (60.41) | 47.1 (2.51) | 11.4 (2.5) | 58.6 (2.01) |
| X | 112.5 (36.83) | 24.43 (7.93) | 5 (0.89) | 466.43 (58.69) | 48 (2.37) | 11.67 (1.86) | 61.14 (3.53) |
| p | 0.11 | 0.38 | 0.81 | 0.51 | 0.04 | 0.07 | 0.002 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




