Submitted:
05 December 2025
Posted:
08 December 2025
You are already at the latest version
Abstract
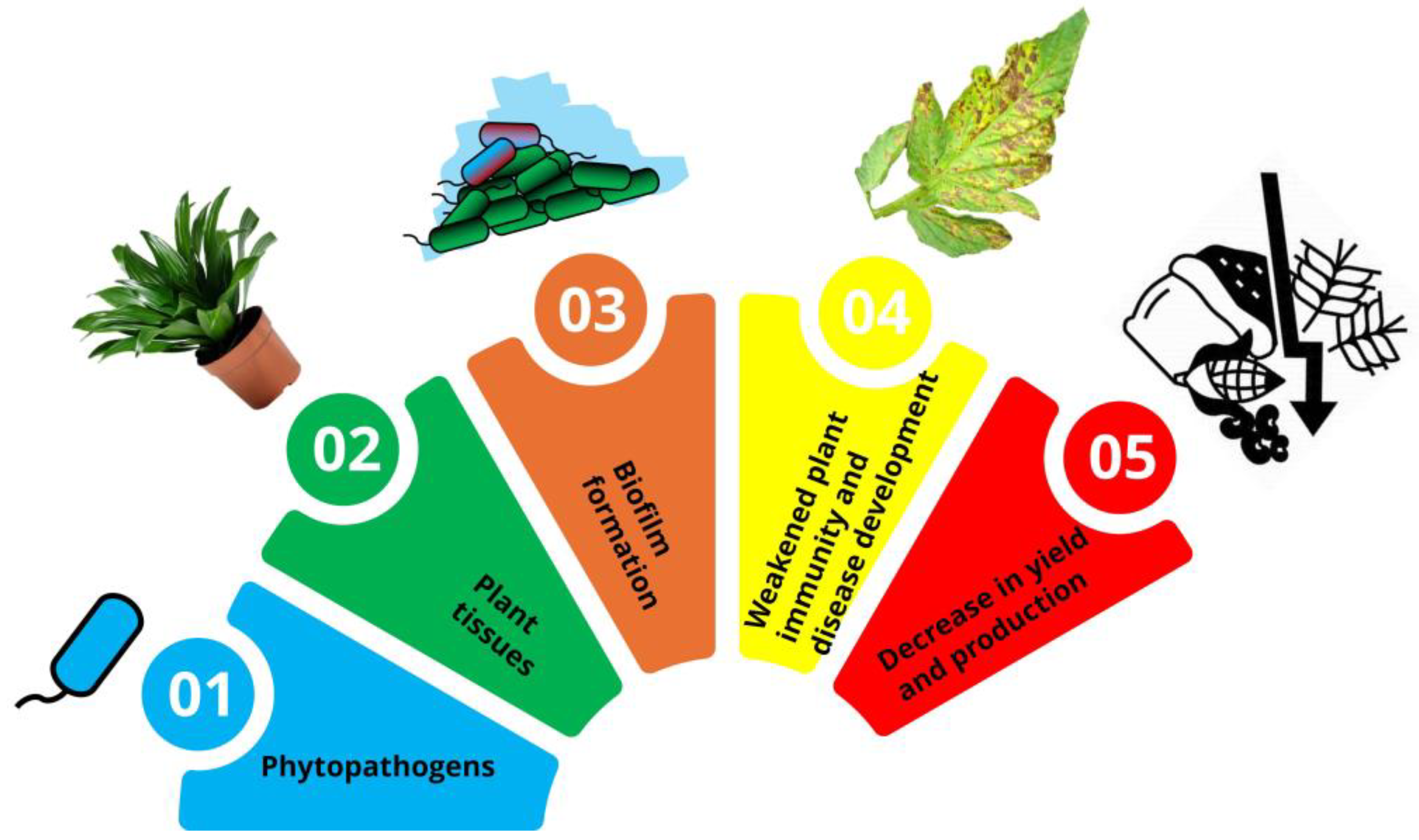
Biofilm-forming phytopathogens pose significant threats to global agriculture by causing persistent diseases in crops, leading to substantial economic losses and challenges in disease management. This review article aims to explore the mechanisms underlying biofilm formation by phytopathogens, with a particular focus on the role of the bacterial Type III secretion system (T3SS) in facilitating biofilm development and pathogenicity. Biofilms, which are structured communities of microorganisms encased in a self-produced matrix, enhance the survival and virulence of pathogens, making them resistant to conventional control methods. The article delves into the impacts of biofilm-associated infections on crop health, including weakened plant immunity and reduced yield. It also highlights the challenges faced by the agricultural industry, such as the limited efficacy of chemical treatments and the emergence of resistant strains. Innovative solutions, including biocontrol agents, quorum sensing inhibitors, nanotechnology-based approaches and edible coatings, are discussed as promising strategies to combat biofilm-related diseases. There is a need for a deeper understanding of biofilm dynamics, the development of sustainable management practices, and the integration of advanced technologies to mitigate the impact of biofilm-forming phytopathogens on agriculture.
Keywords:
Introduction
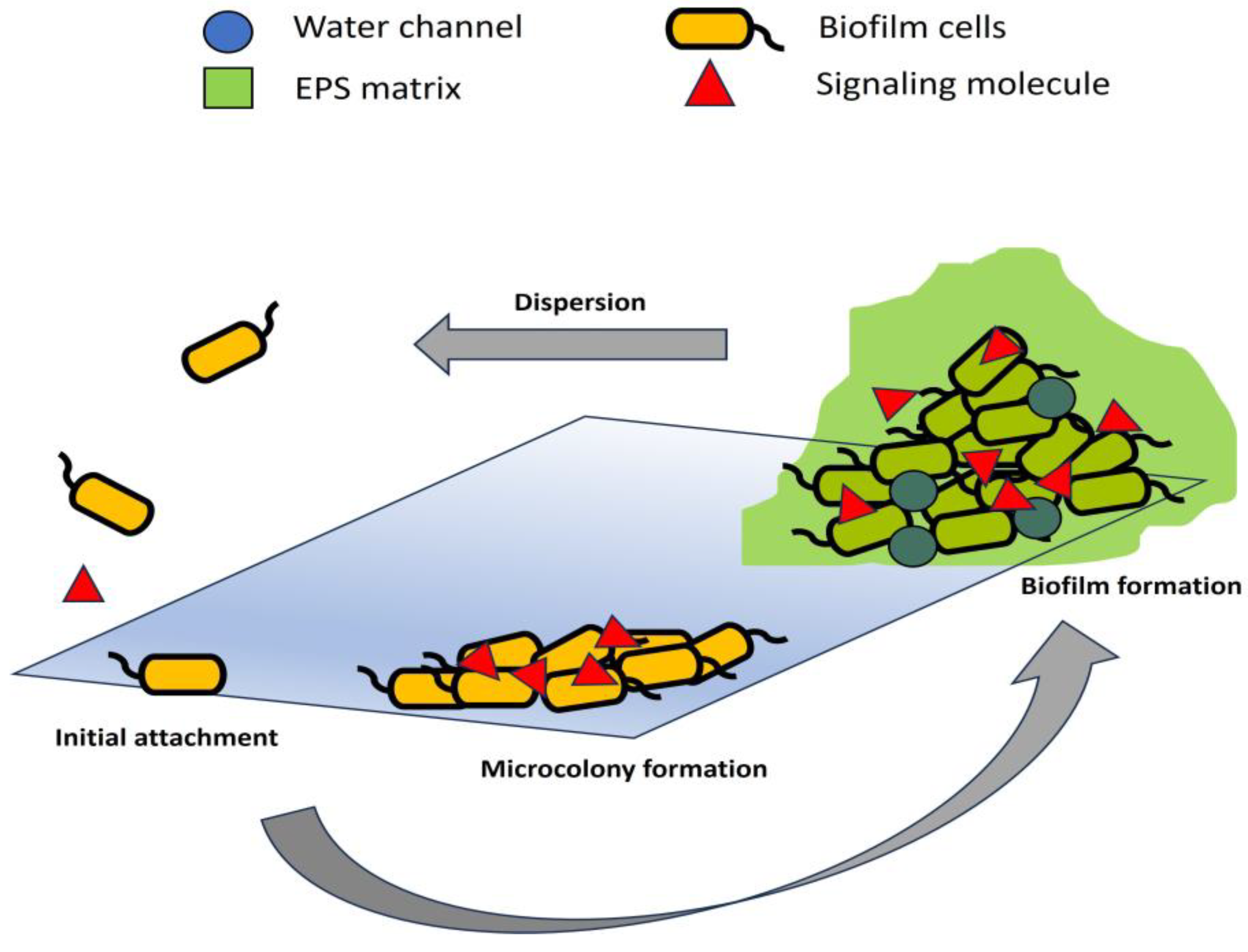
Mechanisms of Biofilm Formation in Agricultural Pathogens
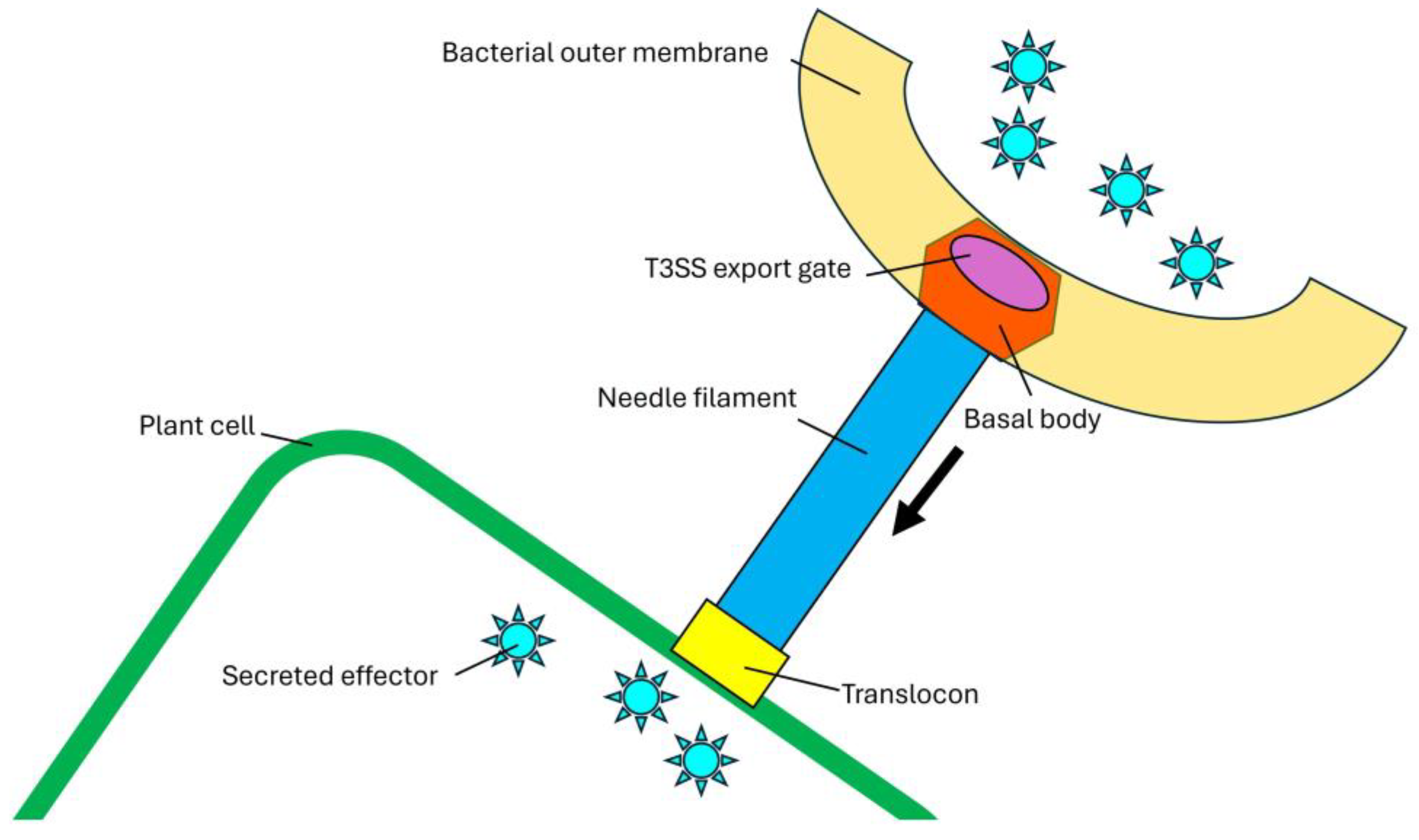
Type III Secretion System
Impact of Biofilm-Forming Phytopathogens on Crop Health
Phytopathogenic Biofilms in Soil and Water Systems
Challenges in Managing Biofilm-Forming Phytopathogens
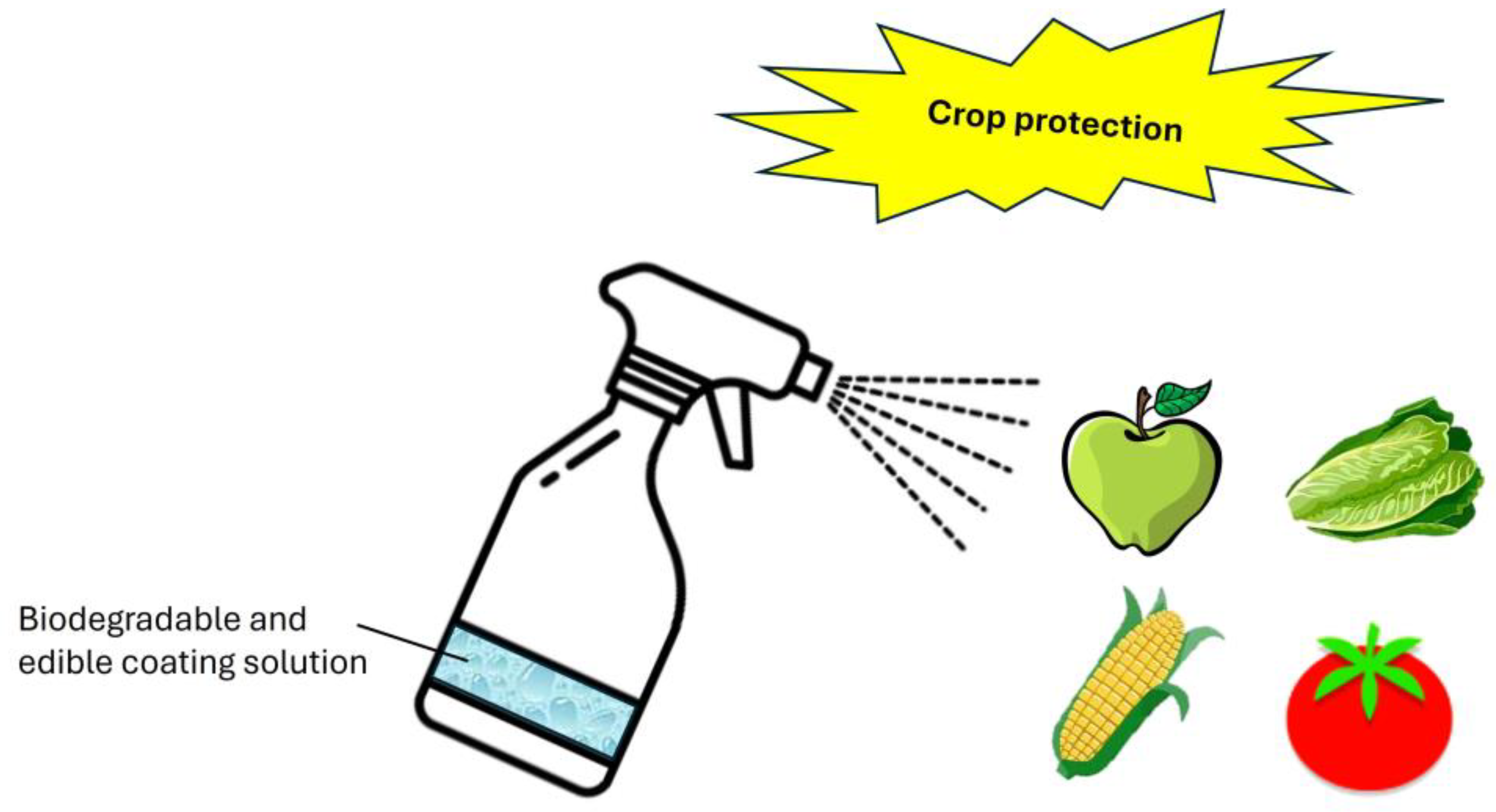
Innovative Strategies for Controlling Biofilm-Forming Phytopathogens
Future Directions
Conclusion
Author Contributions
Acknowledgments
Conflicts of Interest statement
References
- Acharya, A.; Pal, P. K. Agriculture nanotechnology: Translating research outcome to field applications by influencing environmental sustainability. NanoImpact 2020, 19, 100232. [Google Scholar] [CrossRef]
- Alshammari, M.; Ahmad, A.; AlKhulaifi, M.; Al Farraj, D.; Alsudir, S.; Alarawi, M.; Alyamani, E. Reduction of biofilm formation of Escherichia coli by targeting quorum sensing and adhesion genes using the CRISPR/Cas9-HDR approach, and its clinical application on urinary catheter. Journal of Infection and Public Health 2023, 16(8), 1174–1183. [Google Scholar] [CrossRef]
- Angel, P. A. S. R. Y.; Raghul, M.; Gowsalya, S.; Paulkumar, K.; Murugan, K. CRISPR interference system: a potential strategy to inhibit pathogenic biofilm in the agri-food sector. In CRISPR and RNAi Systems; Elsevier, 2021; pp. 387–403. [Google Scholar]
- Ariani, M.; Hervani, A.; Setyanto, P. Climate smart agriculture to increase productivity and reduce greenhouse gas emission–a preliminary study. In IOP Conference Series: Earth and Environmental Science; IOP Publishing, November 2018; Vol. 200, No. 1, p. 012024. [Google Scholar]
- Bergeron, J. R.; Worrall, L. J.; Sgourakis, N. G.; DiMaio, F.; Pfuetzner, R. A.; Felise, H. B.; Strynadka, N. C. A refined model of the prototypical Salmonella SPI-1 T3SS basal body reveals the molecular basis for its assembly. PLoS pathogens 2013, 9(4), e1003307. [Google Scholar] [CrossRef]
- Carezzano, M. E.; Paletti Rovey, M. F.; Cappellari, L. D. R.; Gallarato, L. A.; Bogino, P.; Oliva, M. D. L. M.; Giordano, W. Biofilm-forming ability of phytopathogenic bacteria: a review of its involvement in plant stress. Plants 2023, 12(11), 2207. [Google Scholar] [CrossRef]
- Carlier, A.; Burbank, L.; Von Bodman, S. B. Identification and characterization of three novel EsaI/EsaR quorum-sensing controlled stewartan exopolysaccharide biosynthetic genes in Pantoea stewartii ssp. stewartii. Molecular microbiology 2009, 74(4). [Google Scholar] [CrossRef]
- Castiblanco, L. F.; Sundin, G. W. New insights on molecular regulation of biofilm formation in plant-associated bacteria. Journal of integrative plant biology 2016, 58(4), 362–372. [Google Scholar] [CrossRef]
- Cerutti, A.; Jauneau, A.; Auriac, M. C.; Lauber, E.; Martinez, Y.; Chiarenza, S.; Noël, L. D. Immunity at cauliflower hydathodes controls systemic infection by Xanthomonas campestris pv campestris. Plant Physiology 2017, 174(2), 700–716. [Google Scholar] [CrossRef]
- Chalupowicz, L.; Zellermann, E. M.; Fluegel, M.; Dror, O.; Eichenlaub, R.; Gartemann, K. H.; Manulis-Sasson, S. Colonization and movement of GFP-labeled Clavibacter michiganensis subsp. michiganensis during tomato infection. Phytopathology 2012, 102(1), 23–31. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Chaudhury, S.; McShan, A. C.; Kaur, K.; De Guzman, R. N. Structure and biophysics of type III secretion in bacteria. Biochemistry 2013, 52(15), 2508–2517. [Google Scholar] [CrossRef] [PubMed]
- Coll, N. S.; Valls, M. Current knowledge on the R alstonia solanacearum type III secretion system. Microbial biotechnology 2013, 6(6), 614–620. [Google Scholar] [CrossRef] [PubMed]
- Danhorn, T.; Fuqua, C. Biofilm formation by plant-associated bacteria. Annu. Rev. Microbiol. 2007, 61(1), 401–422. [Google Scholar] [CrossRef]
- De Jonge, R.; Bolton, M. D.; Thomma, B. P. How filamentous pathogens co-opt plants: the ins and outs of fungal effectors. Current opinion in plant biology 2011, 14(4), 400–406. [Google Scholar] [CrossRef]
- Dönmez, D.; Isak, M. A.; İzgü, T.; Şimşek, Ö. Green Horizons: Navigating the future of agriculture through sustainable practices. Sustainability 2024, 16(8), 3505. [Google Scholar] [CrossRef]
- Flemming, H. C.; Percival, S. L.; Walker, J. T. Contamination potential of biofilms in water distribution systems. Water science and technology: water supply 2002, 2(1), 271–280. [Google Scholar] [CrossRef]
- Foroud, N. A.; Chatterton, S.; Reid, L. M.; Turkington, T. K.; Tittlemier, S. A.; Gräfenhan, T. Fusarium diseases of Canadian grain crops: impact and disease management strategies. In Future challenges in crop protection against fungal pathogens; New York, NY; Springer New York, 2014; pp. 267–316. [Google Scholar]
- Frederick, R. D.; Ahmad, M.; Majerczak, D. R.; Arroyo-Rodríguez, A. S.; Manulis, S.; Coplin, D. L. Genetic organization of the Pantoea stewartii subsp. stewartii hrp gene cluster and sequence analysis of the hrpA, hrpC, hrpN, and wtsE operons. Molecular plant-microbe interactions 2001, 14(10), 1213–1222. [Google Scholar] [CrossRef]
- Getahun, S.; Kefale, H.; Gelaye, Y. Application of precision agriculture technologies for sustainable crop production and environmental sustainability: A systematic review. The Scientific World Journal 2024, 2024(1), 2126734. [Google Scholar] [CrossRef]
- Ghiasian, M. Microbial biofilms: Beneficial applications for sustainable agriculture. In New and future developments in microbial biotechnology and bioengineering; Elsevier, 2020; pp. 145–155. [Google Scholar]
- Hamdan, H. F.; Ross, E. E. R.; Jalil, M. T. M.; Hashim, M. A.; Yahya, M. F. Z. R. Antibiofilm efficacy and mode of action of Etlingera elatior extracts against Staphylococcus aureus. Malaysian Applied Biology 2024, 53(1), 27–34. [Google Scholar] [CrossRef]
- Horna, G.; Ruiz, J. Type 3 secretion system of Pseudomonas aeruginosa. Microbiological Research 2021, 246, 126719. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Worrall, L. J.; Strynadka, N. C. Towards capture of dynamic assembly and action of the T3SS at near atomic resolution. Current Opinion in Structural Biology 2020, 61, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Hussein, A. H. A.; Jabbar, K. A.; Mohammed, A.; Al-Jawahry, H. M. AI and IoT in farming: A sustainable approach. In E3S Web of Conferences; EDP Sciences, 2024; Vol. 491, p. 01020. [Google Scholar]
- Johari, N. A.; Aazmi, M. S.; Yahya, M. F. Z. R. FTIR spectroscopic study of inhibition of chloroxylenol-based disinfectant against Salmonella enterica serovar Thyphimurium biofilm. Malaysian Applied Biology 2023, 52(2), 97–107. [Google Scholar] [CrossRef]
- Kan, Y.; Zhang, Y.; Lin, W.; Dong, T. Differential plant cell responses to Acidovorax citrulli T3SS and T6SS reveal an effective strategy for controlling plant-associated pathogens. MBio 2023, 14(4), e00459-23. [Google Scholar] [CrossRef]
- Kanarek, P.; Breza-Boruta, B.; Rolbiecki, R. Microbial composition and formation of biofilms in agricultural irrigation systems-a review. Ecohydrology & Hydrobiology 2024, 24(3), 583–590. [Google Scholar]
- Kamaruzzaman, A.N.A.; Mulok, T.E.T.Z.; Nor, N.H.M.; Yahya, M.F.Z.R. FTIR spectral changes in Candida albicans biofilm following exposure to antifungals. Malaysian Applied Biology 2022, 51(4), 57–66. [Google Scholar] [CrossRef]
- Koczan, J. M.; McGrath, M. J.; Zhao, Y.; Sundin, G. W. Contribution of Erwinia amylovora exopolysaccharides amylovoran and levan to biofilm formation: implications in pathogenicity. Phytopathology 2009, 99(11), 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Narayanan, M.; Shi, X.; Chen, X.; Li, Z.; Ma, Y. Biofilms formation in plant growth-promoting bacteria for alleviating agro-environmental stress. Science of the total environment 2024, 907, 167774. [Google Scholar] [CrossRef]
- Mhedbi-Hajri, N.; Jacques, M. A.; Koebnik, R. Adhesion mechanisms of plant-pathogenic Xanthomonadaceae. In Bacterial adhesion: Chemistry, biology and physics; 2011; pp. 71–89. [Google Scholar]
- Miletic, S.; Goessweiner-Mohr, N.; Marlovits, T. C. The structure of the type III secretion system needle complex. Bacterial Type III Protein Secretion Systems 2020, 67–90. [Google Scholar]
- Panday, D.; Bhusal, N.; Das, S.; Ghalehgolabbehbahani, A. Rooted in nature: the rise, challenges, and potential of organic farming and fertilizers in agroecosystems. Sustainability 2024, 16(4), 1530. [Google Scholar] [CrossRef]
- Pinto, R. M.; Soares, F. A.; Reis, S.; Nunes, C.; Van Dijck, P. Innovative strategies toward the disassembly of the EPS matrix in bacterial biofilms. Frontiers in Microbiology 2020, 11, 952. [Google Scholar] [CrossRef]
- Priller, J. P. R.; Reid, S.; Konein, P.; Dietrich, P.; Sonnewald, S. The Xanthomonas campestris pv. vesicatoria type-3 effector XopB inhibits plant defence responses by interfering with ROS production. PLoS One 2016, 11(7), e0159107. [Google Scholar] [CrossRef]
- Quan, K.; Hou, J.; Zhang, Z.; Ren, Y.; Peterson, B. W.; Flemming, H. C.; van der Mei, H. C. Water in bacterial biofilms: pores and channels, storage and transport functions. Critical Reviews in Microbiology 2021, 48(3), 283–302. [Google Scholar] [CrossRef]
- Rinaudi, L. V.; Giordano, W. An integrated view of biofilm formation in rhizobia. FEMS microbiology letters 2010, 304(1), 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rather, M. A.; Gupta, K.; Mandal, M. Impact of microbial biofilm on crop productivity and agricultural sustainability. In Microbes in land use change management; Elsevier, 2021; pp. 451–469. [Google Scholar]
- Sabino, Y. N. V.; de Araújo Domingues, K. C.; Mathur, H.; Gómez-Mascaraque, L. G.; Drouin, G.; Martínez-Abad, A.; Mantovani, H. C. Exopolysaccharides produced by Bacillus spp. inhibit biofilm formation by Staphylococcus aureus strains associated with bovine mastitis. International Journal of Biological Macromolecules 2023, 253, 126689. [Google Scholar] [CrossRef]
- Schumann, A.; Mungofa, P.; Waldo, L.; Oswalt, C. Smartphone App Under Development for Diagnosing Citrus Leaf Symptoms. In EDIS; 2020. [Google Scholar]
- Sen, Y.; van der Wolf, J.; Visser, R. G.; van Heusden, S. Bacterial canker of tomato: current knowledge of detection, management, resistance, and interactions. Plant Disease 2015, 99(1), 4–13. [Google Scholar] [CrossRef]
- Sharma, G.; Karnwal, A. Biological strategies against biofilms. In Microbial Biotechnology: Basic Research and Applications; 2020; pp. 205–232. [Google Scholar]
- Slater, H.; Alvarez-Morales, A.; Barber, C. E.; Daniels, M. J.; Dow, J. M. A two-component system involving an HD-GYP domain protein links cell–cell signalling to pathogenicity gene expression in Xanthomonas campestris. Molecular microbiology 2000, 38(5), 986–1003. [Google Scholar] [CrossRef]
- Syahputri, D.; Lubis, S.; Anggraini, B. Analisis Peran Sektor Pertanian Dalam Pengurangan Kemiskinan dan Peningkatan Kesejahteraan di Negara-Negara Berkembang. Jurnal Ekonomi, Bisnis Dan Manajemen 2024, 3(1), 93–103. [Google Scholar] [CrossRef]
- Van Tilburg Bernardes, E.; Charron-Mazenod, L.; Reading, D. J.; Reckseidler-Zenteno, S. L.; Lewenza, S. Exopolysaccharide-repressing small molecules with antibiofilm and antivirulence activity against Pseudomonas aeruginosa. Antimicrobial agents and chemotherapy 2017, 61(5), 10–1128. [Google Scholar] [CrossRef]
- Wei, H.; Xu, Q.; Taylor, L. E., II; Baker, J. O.; Tucker, M. P.; Ding, S. Y. Natural paradigms of plant cell wall degradation. Current Opinion in Biotechnology 2009, 20(3), 330–338. [Google Scholar] [CrossRef]
- Yaacob, M. F.; Murata, A.; Nor, N. H. M.; Jesse, F. F. A.; Yahya, M. F. Z. R. Biochemical composition, morphology and antimicrobial susceptibility pattern of Corynebacterium pseudotuberculosis biofilm. Journal of King Saud University-Science 2021, 33(1), 101225. [Google Scholar] [CrossRef]
- Yahya, M. F. Z. R.; Alias, Z.; Karsani, S. A. Subtractive protein profiling of Salmonella typhimurium biofilm treated with DMSO. The Protein Journal 36 2017, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Yahya, M. F. Z. R.; Nor, N. H. M.; Mahat, M. M.; Siburian, R. Edible coating, food-contact surface coating, and nanosensor for biofilm mitigation plans in food industry. Food Materials Research 2024, 4(1). [Google Scholar] [CrossRef]
- Yakup, N. F.; Nor, N. H. M.; Mahat, M. M.; Siburian, R.; Yahya, M. F. Z. R. Unveiling the Antibiofilm Arsenal: A Mini Review on Nanoparticles’ Mechanisms and Efficacy in Biofilm Inhibition. International Conference on Science Technology and Social Sciences–Physics, Material and Industrial Technology (ICONSTAS-PMIT 2023); Atlantis Press, 2024; pp. 177–185. [Google Scholar]
- Yan, D.; Yang, P.; Rowan, M.; Ren, S.; Pitts, D. Biofilm accumulation and structure in the flow path of drip emitters using reclaimed wastewater. Transactions of the ASABE 2010, 53(3), 751–758. [Google Scholar] [CrossRef]
- Yang, H.; Ma, R.; Chen, J.; Xie, Q.; Luo, W.; Sun, P.; Guo, J. Discovery of melittin as triple-action agent: broad-spectrum antibacterial, anti-biofilm, and potential anti-quorum sensing activities. Molecules 2024, 29(3), 558. [Google Scholar] [CrossRef]
- Yao, Y.; Habimana, O. Biofilm research within irrigation water distribution systems: Trends, knowledge gaps, and future perspectives. Science of the total environment 2019, 673, 254–265. [Google Scholar] [CrossRef] [PubMed]
- Yaron, S.; Römling, U. Biofilm formation by enteric pathogens and its role in plant colonization and persistence. Microbial biotechnology 2014, 7(6), 496–516. [Google Scholar] [CrossRef] [PubMed]
- Yusri, M. H.; Nor, N. H. M.; Mahat, M. M.; Siburian, R.; Yahya, M. F. Z. R. Insights into Antibiofilm Mode of Actions of Natural and Synthetic Polymers: A Mini Review. In Proceedings of the International Conference on Science Technology and Social Sciences-Physics, Material and Industrial Technology (ICONSTAS-PMIT 2023). Springer Nature, 2024; p. 134. [Google Scholar]
- Zheng, H.; Ma, W.; He, Q. Climate-smart agricultural practices for enhanced farm productivity, income, resilience, and greenhouse gas mitigation: a comprehensive review. Mitigation and Adaptation Strategies for Global Change 2024, 29(4), 28. [Google Scholar] [CrossRef]
- Zimaro, T.; Thomas, L.; Marondedze, C.; Sgro, G. G.; Garofalo, C. G.; Ficarra, F. A.; Gottig, N. The type III protein secretion system contributes to Xanthomonas citri subsp. citri biofilm formation. Bmc Microbiology 2014, 14, 1–15. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



