Submitted:
04 December 2025
Posted:
05 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Compounds and Reagents
2.2. Cells and Culture Medium
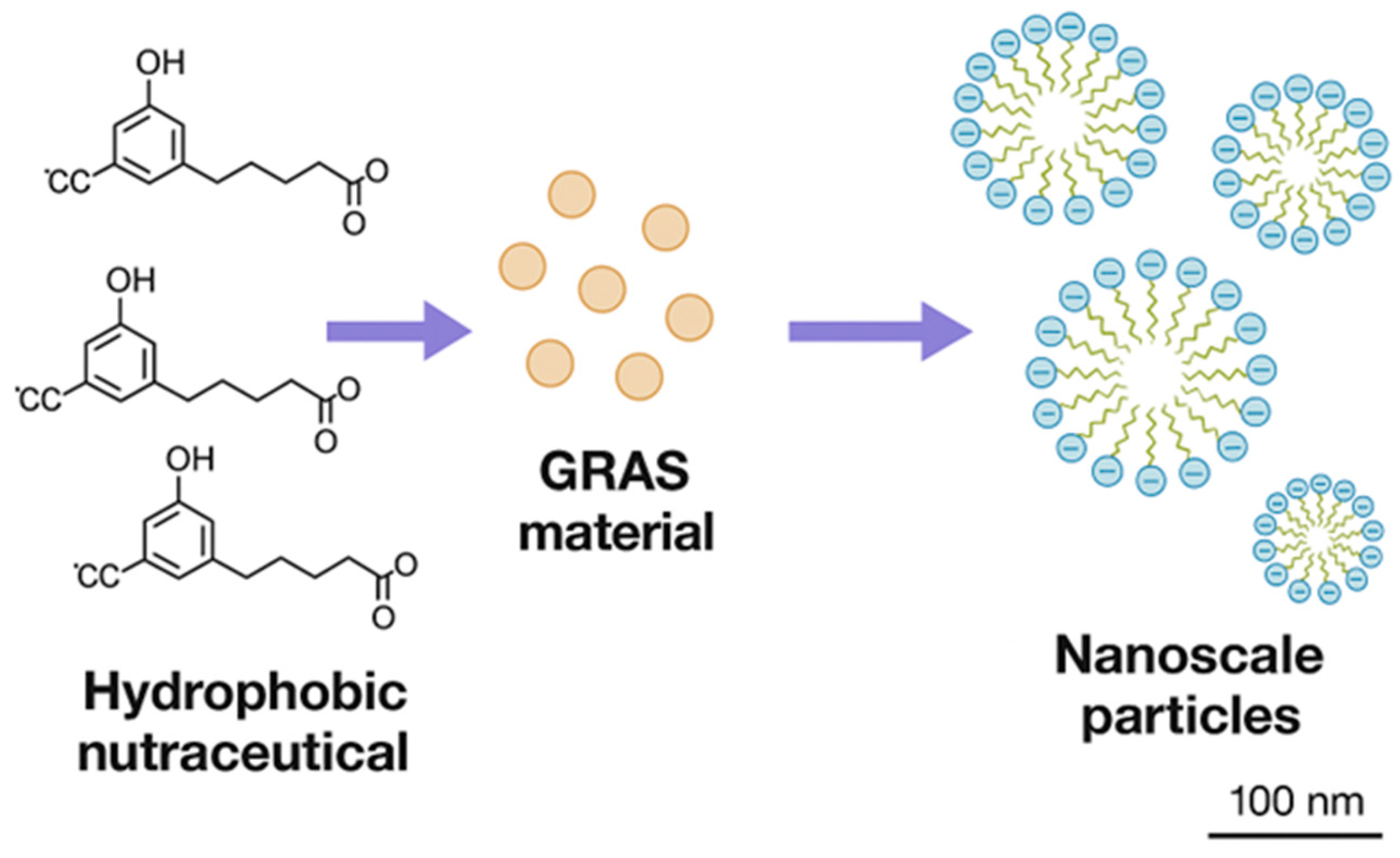
2.3. Preparation of Nanoparticles
2.4. Dynamic Light Scattering (DLS) Analysis
2.5. X-Ray Diffraction
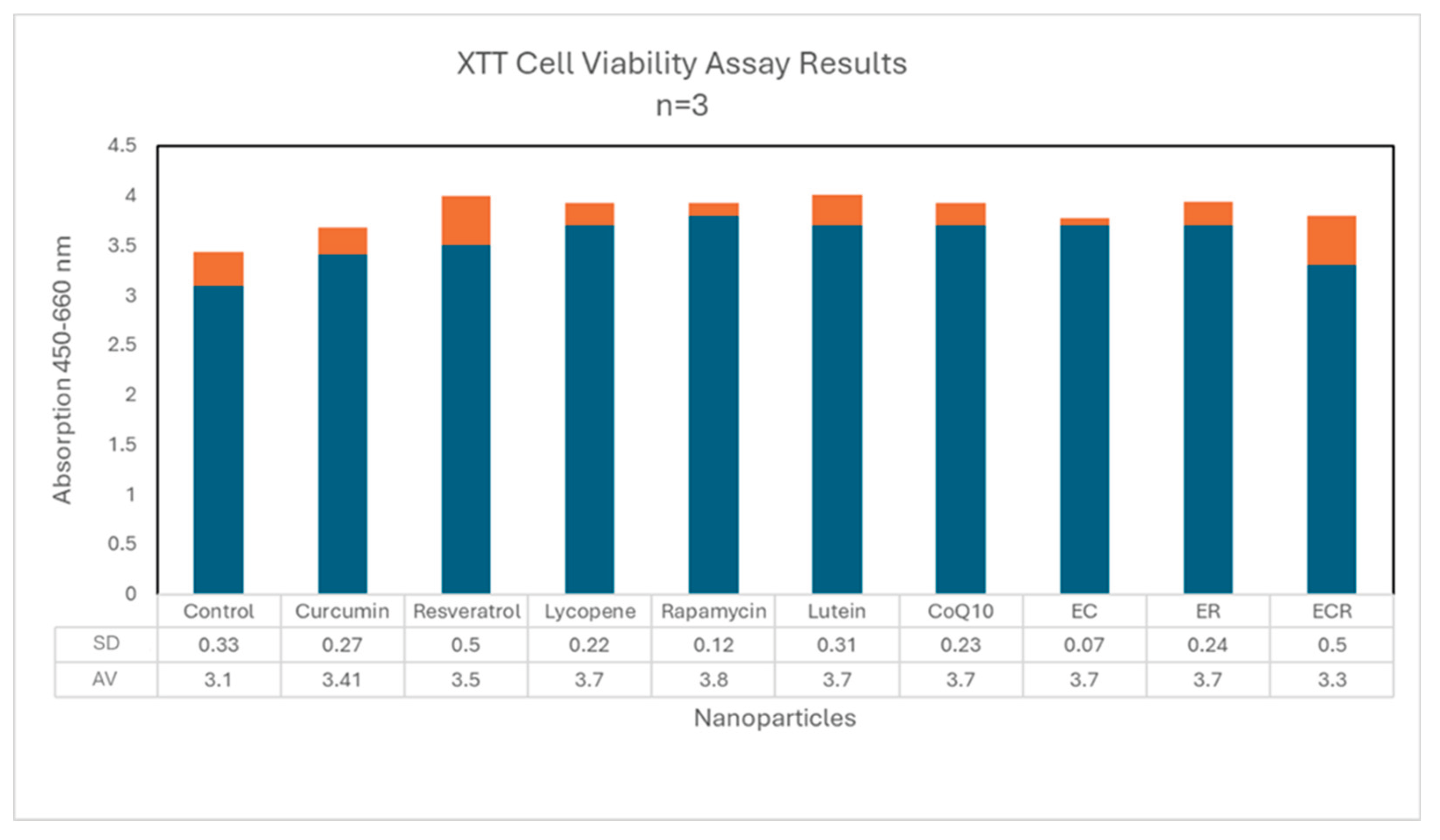
2.6. Cell Viability (XTT) Assay
2.7. Stability Under Acidic Conditions
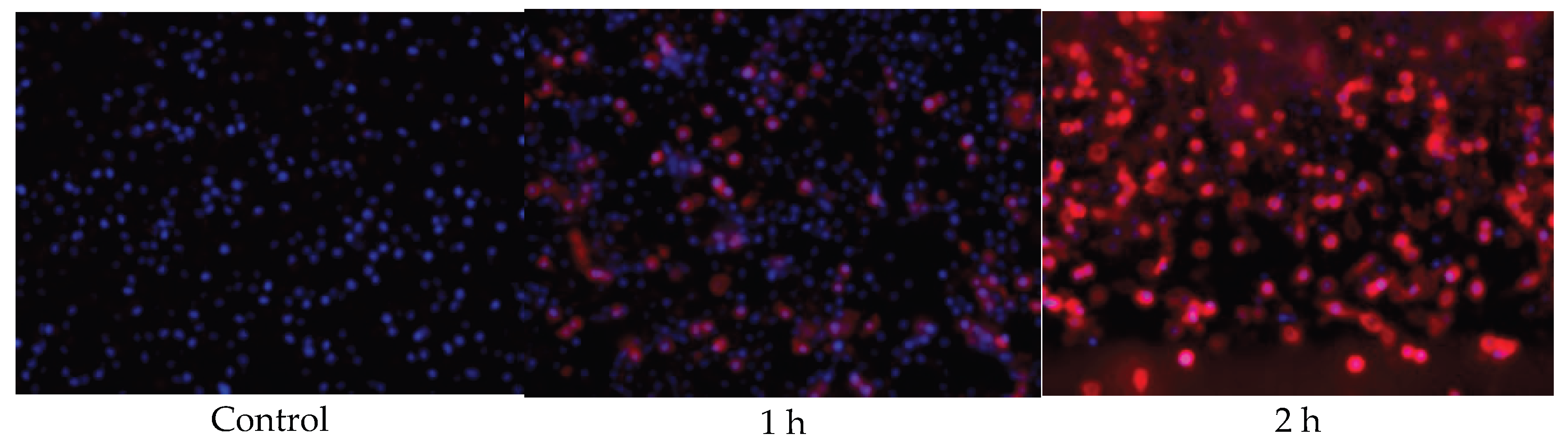
2.8. Fluorescent Imaging of EC16/Cy5 Nanoparticle-Cell Interaction
3. Results
3.1. Nanoparticles Formation
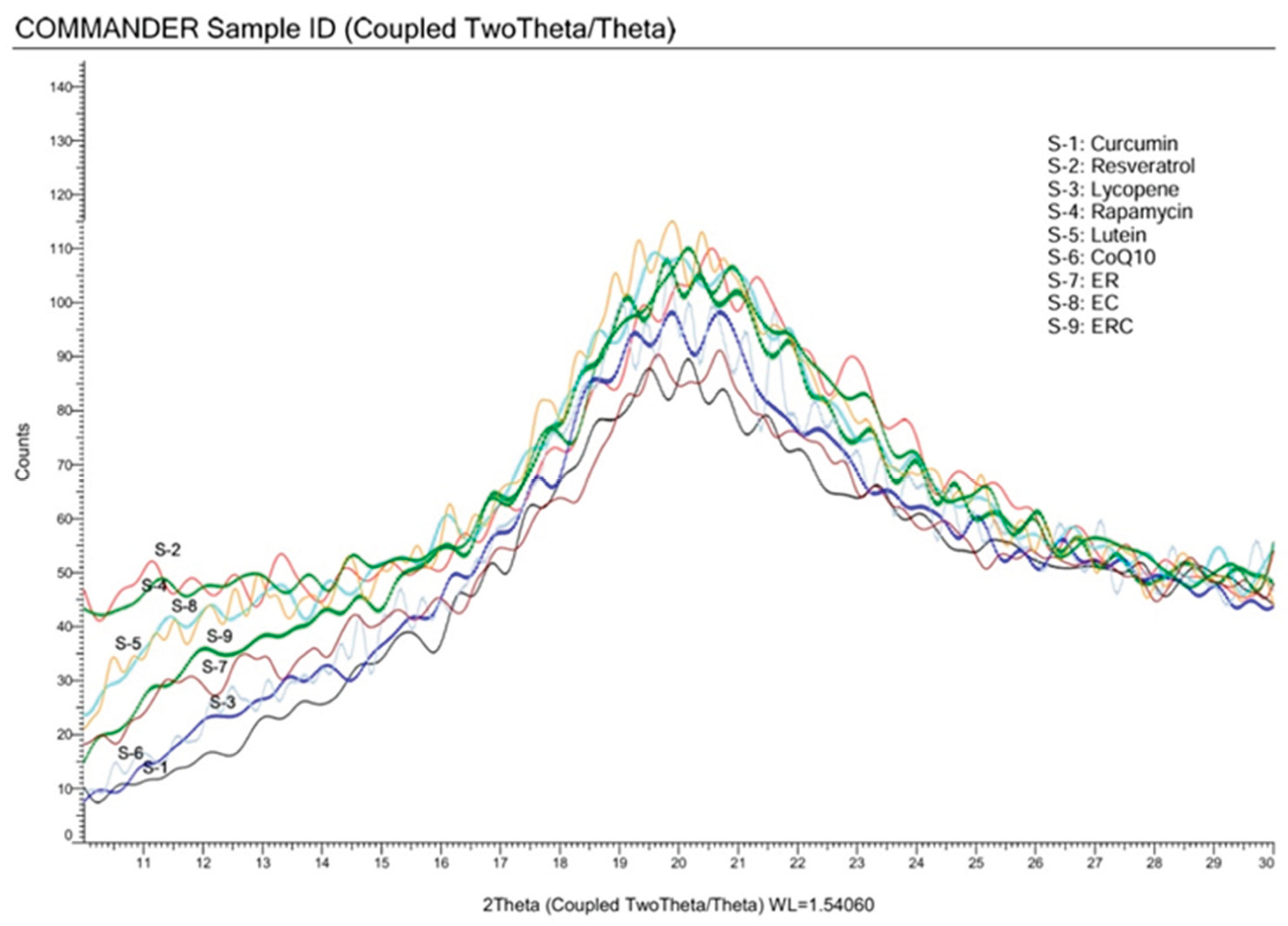
3.2. X-ray Diffraction (XRD)
3.3. Cell Viability
3.4. Stability under Acidic Conditions
3.5. EC16/Cy5 and Cy5 Nanoparticles
3.6. EC16/Cy5 Hybrid Nanoparticles Incubated with RAW264.7 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| GRAS FDA |
Generally Recognized as Safe Food and Drug Administration |
| FAST | Facilitated Self-assembling Technology |
| EC16 | Epigallocatechin-3-Gallate-Palmitate |
| CoQ10 Cy5 SLN NLC DLS EFSA DMSO HCl EDC DMAP |
Coenzyme Q10 Cy5 hydrazide (non-sulfonated) Solid lipid nanoparticles Nanostructured lipid carriers Dynamic Light Scattering European Food Safety Authority Dimethyl Sulfoxide Hydrochloric Acid 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide 4-Dimethylaminopyridine |
References
- Jampilek J, Kos J, Kralova K. Potential of nanomaterial applications in dietary supplements and foods for special medical purposes. Nanomaterials. 2019, 9, 296. [Google Scholar] [CrossRef]
- Vesely O, Baldovska S, Kolesarova A. Enhancing bioavailability of nutraceutically used resveratrol and other stilbenoids. Nutrients. 2021, 13, 3095. [Google Scholar] [CrossRef]
- Puri V, Nagpal M, Singh I, et al. A comprehensive review on nutraceuticals: therapy support and formulation challenges. Nutrients. 2022, 14, 4637. [Google Scholar] [CrossRef]
- Ashfaq R, Rasul A, Asghar S, et al. Lipid nanoparticles: an effective tool to improve the bioavailability of nutraceuticals. Int J Mol Sci. 2023, 24, 15764. [Google Scholar] [CrossRef] [PubMed]
- Hoti G, Matencio A, Rubin Pedrazzo A, et al. Nutraceutical concepts and dextrin-based delivery systems. Int J Mol Sci. 2022, 23, 4102. [Google Scholar] [CrossRef] [PubMed]
- Almeida L, Vaz-da-Silva M, Falcão A, et al. Pharmacokinetic and safety profile of trans-resveratrol in a rising multiple-dose study in healthy volunteers. Mol Nutr Food Res. 2009, 53, S7–S15. [Google Scholar]
- Vaz-da-Silva M, Loureiro AI, Falcão A, et al. Effect of food on the pharmacokinetic profile of trans-resveratrol. Int J Clin Pharmacol Ther. 2008, 46, 564–570. [Google Scholar] [CrossRef]
- Nunes T, Almeida L, Rocha JF, et al. Pharmacokinetics of trans-resveratrol following repeated administration in healthy elderly and young subjects. J Clin Pharmacol. 2009, 49, 1477–1482. [Google Scholar] [CrossRef]
- Ramírez-Garza SL, Laveriano-Santos EP, Marhuenda-Munguía D, et al. Health effects of resveratrol: results from human intervention trials. Nutrients. 2018, 10, 1892. [Google Scholar] [CrossRef] [PubMed]
- Brown K, Deighton N, Moncayo-Calero M, et al. Resveratrol for the management of human health. Int J Mol Sci. 2024, 25, 747. [Google Scholar] [CrossRef]
- Tabanelli R, Brogi S, Calderone V, et al. Improving curcumin bioavailability: current strategies and future perspectives. Pharmaceutics. 2021, 13, 1715. [Google Scholar]
- Bertoncini-Silva C, Vlad A, Ricciarelli R, et al. Enhancing the bioavailability and bioactivity of curcumin: promises and challenges. Antioxidants (Basel). 2024, 13, 331. [Google Scholar]
- Shoba G, Joy D, Joseph T, et al. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Klickovic U, Doberer D, Gouya G, et al. Human pharmacokinetics of high-dose oral curcumin and its effect on heme oxygenase-1 expression in healthy male subjects. Biomed Res Int. 2014, 2014, 458592. [Google Scholar]
- Kapała A, Lange E, Piórecka B. The anti-cancer activity of lycopene: a systematic review of human and animal studies. Nutrients. 2022, 14, 5152. [Google Scholar] [CrossRef]
- Arballo J, Amengual J, Erdman JW Jr. Lycopene: a critical review of digestion, absorption, metabolism, and excretion. Antioxidants (Basel). 2021, 10, 342. [Google Scholar] [CrossRef]
- Kotagiri SR, Morde A, Rai D, et al. Superior bioavailability of a novel lutein and zeaxanthin formulation in healthy human subjects. Ophthalmol Ther. 2022, 11, 1463–1477. [Google Scholar] [CrossRef]
- Thürmann PA, Schalch W, Aebischer JC, et al. Plasma kinetics of lutein, zeaxanthin, and 3-dehydro-lutein after multiple oral doses of a lutein supplement. Am J Clin Nutr. 2005, 82, 88–97. [Google Scholar] [CrossRef]
- Mantle D, Dybring A. Bioavailability of coenzyme Q10: an overview of the absorption process and subsequent metabolism. Antioxidants (Basel). 2020, 9, 386. [Google Scholar] [CrossRef]
- Maciejewska-Stupska K, Czarnecka K, Szymański P. Bioavailability enhancement of coenzyme Q10: an update of novel approaches. Arch Pharm (Weinheim). 2024, 357, e2300676. [Google Scholar] [CrossRef]
- López-Lluch G, del Pozo-Cruz J, Sánchez-Cadena M, et al. Bioavailability of coenzyme Q10 supplements depends on carrier lipids and solubilization. Nutr Hosp. 2019, 36, 996–1002. [Google Scholar]
- Choi SJ, McClements DJ. Nanoemulsions as delivery systems for lipophilic nutraceuticals: strategies for improving their formulation, stability, functionality, and bioavailability. Food Sci Biotechnol. 2020, 29, 149–168. [Google Scholar] [CrossRef]
- Subramanian P, Nandhini M, Balusamy SR, et al. Lipid-based nanocarrier system for the effective delivery of nutraceuticals. Nanomaterials (Basel). 2021, 11, 2439. [Google Scholar]
- Blanco-Llamero C, Porto-Sánchez A, Vicente-Díez I, et al. Nutraceuticals and food-grade lipid nanoparticles. Foods. 2022, 11, 2318. [Google Scholar] [CrossRef]
- Teixé-Roig J, Flores-Talavera A, Megías-Romero J, et al. Emulsion-based delivery systems to enhance the bioavailability of plant bioactives in functional foods. Foods. 2023, 12, 1502. [Google Scholar] [CrossRef] [PubMed]
- Frank N, Dickinson D, Liu Y, Yu H, Cai J, Hsu S. Facilitated Self-Assembling Technology (FAST) for the preparation of nanoparticles to increase the solubility and bioavailability of hydrophobic molecules. Fortune J Health Sci. 2025, 8, 283–295. [Google Scholar]
- Helwa I, Cai J, Drewry MD, et al. A comparative study of serum exosome isolation using differential ultracentrifugation and three commercial reagents. PLoS One. 2017, 12, e0170628. [Google Scholar]
- Cuffari, B. Nanotechnology and the future of the beverage industry. AZO Nano. Oct 22, 2020.Available at: https://www.azonano.com/article.aspx?ArticleID=5573.
- Su Q, Zhao X, Zhang X, et al. Nano functional food: opportunities, development, and future perspectives. Int J Mol Sci. 2022, 24, 234. [Google Scholar] [CrossRef]
- Frank N, Dickinson D, Dudish C, et al. Potential therapeutic use of EGCG-palmitate nanoparticles for norovirus infection. Biomed J Sci Tech Res. 2024; 59.
- Garcia-Becerra C, Méndez-García LA, González-Santiago O, et al. Characterization and bioavailability of a novel coenzyme Q10 oil-in-water nanoemulsion designed for infant formula. Int J Pharm. 2023, 631, 122576. [Google Scholar]
- Kroon MAGM, van Laarhoven HWM, Swart EL, et al. A pharmacokinetic study and critical reappraisal of curcumin formulations enhancing bioavailability. iScience. 2025, 28, 112575. [Google Scholar] [CrossRef] [PubMed]
- Chen S, Wu J, Tang Q, et al. Nano-micelles based on hydroxyethyl starch–curcumin conjugates for improved stability, antioxidant and anticancer activity of curcumin. Carbohydr Polym. 2020; 228.
- Gonçalves RFS, Vicente AA, Pinheiro AC. Incorporation of curcumin-loaded lipid-based nano delivery systems into food: release behavior in food simulants and a case study in a beverage. Food Chem. 2023;405(Pt A):134740.
- Frank N, Dickinson D, Garcia W, et al. Evaluation of aqueous nanoformulations of epigallocatechin-3-gallate–palmitate (EC16) against human coronavirus as a potential intervention drug. Biomed J Sci Tech Res. 2023;50(1.
- Frank N, Dickinson D, Garcia W, et al. Feasibility study of developing a saline-based antiviral nanoformulation containing lipid-soluble EGCG: a potential nasal drug to treat long COVID. Viruses. 2024, 16, 196. [Google Scholar] [CrossRef] [PubMed]
- Frank N, Dickinson D, Lovett G, et al. Evaluation of novel nasal mucoadhesive nanoformulations containing lipid-soluble EGCG for long COVID treatment. Pharmaceutics. 2024, 16, 791. [Google Scholar] [CrossRef] [PubMed]
| Compound | Median Size nm | Zeta Potential mV | Density /ml, 0.01% |
|---|---|---|---|
| Curcumin | 136.6 ± 104.9 | -33.72 ± 0.93 | 4.4x 108 |
| Resveratrol | 174.0 ± 76.76 | -31.75 ± 1.86 | 0.8x108 |
| Lycopene | 184.9 ± 156.2 | -20.50 ± 0.87 | 1.7x107 |
| Lutein | 141.7 ± 53.4 | -24.54 ± 1.18 | 1.3x109 |
| CoQ10 | 315.3 ± 131.8 | -26.8 ± 1.11 | 1.2x107 |
| Procyanidin B2 | 144.2 ± 120.4 | -71.59 ± 0 | 4.5x108 |
| Quercetin | 163.8 ± 102.2 | -62 ± 1.0 mV | 1.6 x109 |
| EGCG-palmitate (EC16) | 152.5 ± 78.8 | -60.11± 0.59 mV | 3.2 x109 |
| Rapamycin | 147.7 ± 89.1 | -44.28 ± 1.79 mV | 2.8 x 109 |
| Compound | Median Size nm | Zeta Potential mV | Density /ml, 0.01% |
|---|---|---|---|
| Curcumin | 166.4 ± 57.3 | -45.71 ± 1.53 | 4.2x 108 |
| Resveratrol | 134.2 ± 55.8 | -44.43 ± 1.89 | 1.3x108 |
| Lycopene | 149 ± 74.9 | -38.86 ± 0.87 | 2.7x108 |
| Lutein | 154.2 ± 66.7 | -32.63 ± 0.13 | 3.0x108 |
| CoQ10 | 232.3 ± 151.3 | -42.81 ± 1.71 | 2.3x107 |
| Compounds | Type of NPs | Median Size nm | Zeta Potential mV | Density /ml, 0.01% |
|---|---|---|---|---|
| EC16-Resveratrol | Hybrid | 96.2 ± 64.1 | -53.29 ± 0.67 | 1x 1010 |
| EC16-Curcumin | Hybrid | 85.0 ± 36.9 | -57.22 ± 0.87 | 2.5x1010 |
| EC16-Res-Cur | Hybrid | 99.9 ± 79.8 | -51.23 ± 1.25 | 5.7x109 |
| EC16/Resveratrol | Mix | 180.0 ± 155.2 | -57.02 ± 1.37 | 2.3x109 |
| EC16/Curcumin | Mix | 198.8 ± 115.8 | -67.45 ± 1.17 | 5.0x109 |
| EC16/Res/Cur | Mix | 201.4 ± 117.7 | -62.32 ± 0.92 | 4.7x109 |
| EC16-Cy5 (Fluorescent) | Hybrid | 141.9 ± 86.8 | -43.1 ± 1.13 mV | 6.1 x1010 |
| NPs/Incubation time (min) | Density/ml | Median Size (nm) |
| Curcumin 0 | 2.0 x 107 | 165 ± 111 |
| Curcumin 60 | 8.4 x 106 | 130 ± 78 |
| Resveratrol 0 | 1.1 x 109 | 189 ± 647 |
| Resveratrol 60 | 5.6 x 108 | 182 ± 810 |
| Hybrid 0 | 3.0 x 108 | 158 ± 165 |
| Hybrid 60 | 2.6 x 108 | 154 ± 361 |
| Mix 0 | 1.3 x 108 | 160 ± 125 |
| Mix 60 | 3.1 x 107 | 156 ± 220 |
| Parameter | HES–CUR–COOH Chemical Conjugation [33] | Lipid-Based Nano Delivery Systems (NE, SLN, NLC) [34] | FAST (Facilitated Self-Assembling Technology, Current Study) |
| Main Purpose | Covalent synthesis of curcumin–HES micelles for drug delivery | Encapsulation of curcumin in lipid nanoparticles for beverage and food applications | Physical self-assembly of curcumin and natural amphiphiles for nutraceutical and beverage delivery |
| Chemicals Used | Pyridine, glutaric anhydride, EDC, DMAP, DMSO, HCl, ethyl acetate | Beeswax, lecithin, Tween 80, MCT oil, ethanol, acetic acid, sodium benzoate, ascorbic acid | Only GRAS ingredients |
| Reaction Type | Multi-step covalent conjugation (ester and amide bond formation) | Physical emulsification, high-pressure homogenization, and ultrasonication | Spontaneous self-assembly through hydrophobic and hydrogen-bond interactions |
| Processing Time | > 1 week including synthesis and purification | 4–6 h (multiple homogenization and cooling cycles) | < 60 min from powder to nanoparticle stock readily for use |
| Energy Requirement | High—solvent reflux, evaporation, and lyophilization | Very high—ultrasonication and homogenization at ~20,000 psi | Low |
| Byproducts / Waste | Pyridine and DMSO residues; requires dialysis and lyophilization | Residual surfactants and organic solvents | None (zero waste) |
| Potential Toxicity | High — due to pyridine, DMSO, and EDC residues; not suitable for ingestion without rigorous removal | Low to moderate; Tween 80 and lecithin are food-grade but may affect gut permeability | Minimal—fully GRAS and biodegradable materials |
| Particle Composition | Curcumin–HES conjugate polymers, irregular particles | SLN and NLC (~150 nm) lipid carriers, curcumin encapsulated in wax/oil core | Native curcumin nanoparticles (median 150 nm), amorphous and colloidally stable |
| Manufacturing Complexity | Multi-step, laboratory scale | Multi-stage homogenization; high-energy and temperature sensitive | One-step scalable process; compatible with beverage manufacturing lines |
| Scalability / Industrial Feasibility | Low—requires organic solvent handling | Moderate—food-grade but high-cost energy equipment | Excellent—simple, aqueous, eco-friendly, and low-cost |
| Regulatory Classification | Pharmaceutical; requires toxicology approval | Food-grade but limited by surfactant and process energy requirements | Nutraceutical / food-grade, GRAS-compliant, and clean-label |
| Environmental Impact | High solvent use and waste | Moderate—requires heating and surfactant disposal | Near-zero waste, solvent-free, and energy-efficient |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




