Submitted:
03 December 2025
Posted:
05 December 2025
You are already at the latest version
Abstract
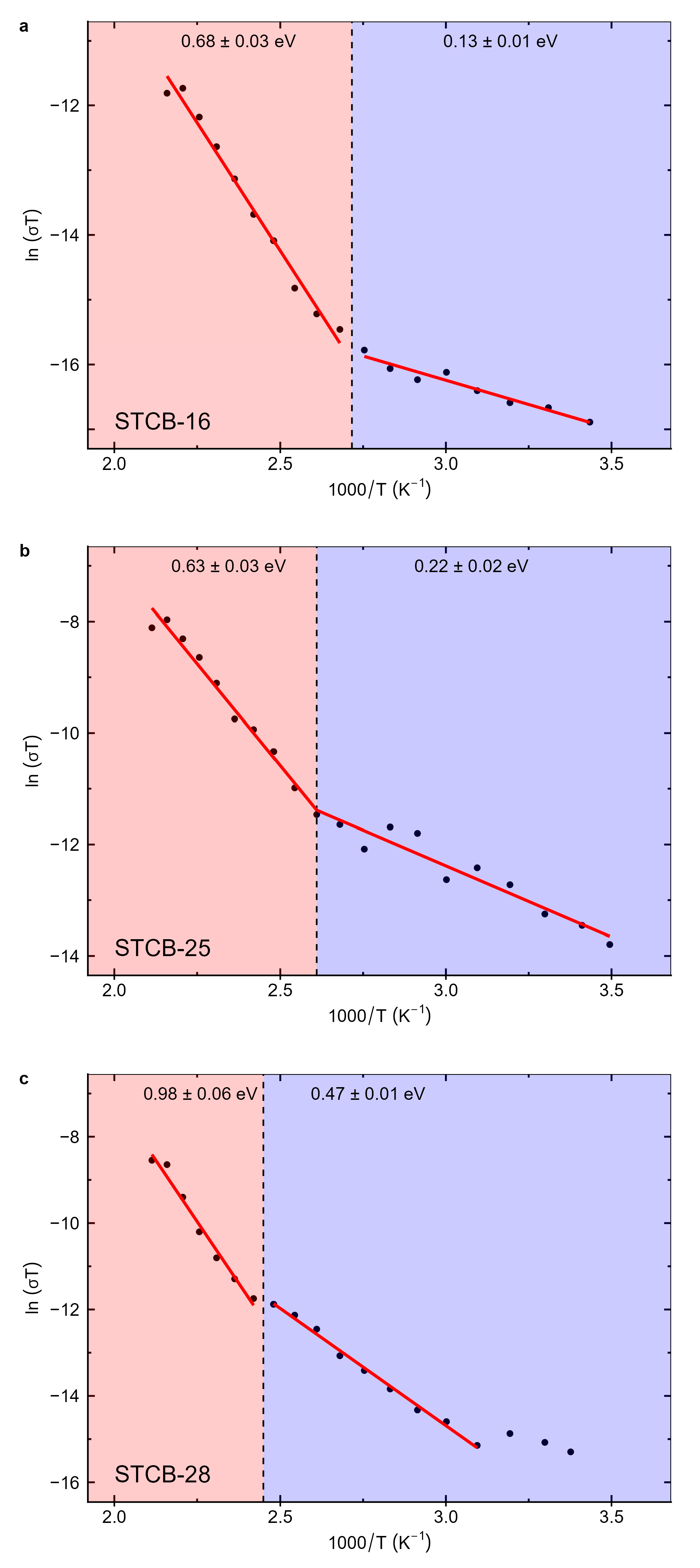
Multiferroic BaTiO3 (BTO, piezoelectric)/CoFe2O4 (CFO, magnetostrictive) bilayer thin films were prepared by laser ablation on conductive Nb-doped SrTiO3 (100) substrates to investigate the influence of BTO layer thickness on their structural, microstructural, dielectric, and electrical (DC and AC) properties. X-ray diffraction confirmed the coexistence of the cubic spinel CoFe2O4 phase and the tetragonal ferroelectric BaTiO3 phase. The films exhibit preferred orientation, with CFO showing the [400] direction along the growth axis and BTO displaying (100)/(001) planes stacked parallel to it. The CFO unit cell is compressed along the growth direction, while BTO presents the ferroelectric distortion with a tetragonality ratio (c/a) slightly below, but close to, the bulk value. Second harmonic generation studies further verified the non-centrosymmetric ferroelectric nature of BTO at room temperature. The temperature-dependent dielectric permittivity was modeled using the Havriliak–Negami function with an additional conductivity term to extract relaxation dynamics, DC conductivity, Curie temperature (Tc), and activation energies. The Curie temperature increases with BTO thickness, approaching the bulk value for thicker layers. DC conductivity activation energies exhibit a change at Tc, from below 0.5 eV for T < Tc to above 0.5 eV for T > Tc, consistent with small-polaron tunneling. The AC conductivity follows a Jonscher-type frequency dependence with two power-law contributions reflecting the behavior of both layers. Temperature-dependent analysis of the power-law exponents reveals that small-polaron tunneling dominates conduction in BTO, while ion hopping between octahedral sites governs conduction in CFO. Underoxidation leads to a more complex transport regime in BTO, showing a transition from quantum-mechanical tunneling below Tc to correlated barrier hopping above it. By revealing how transport processes operate within multiferroic oriented bilayer systems, these findings advance our understanding of material interactions and pave the way for the design of innovative multifunctional platforms optimized for spintronic technologies.
Keywords:
1. Introduction
2. Materials and Methods
3. Results and Discussion
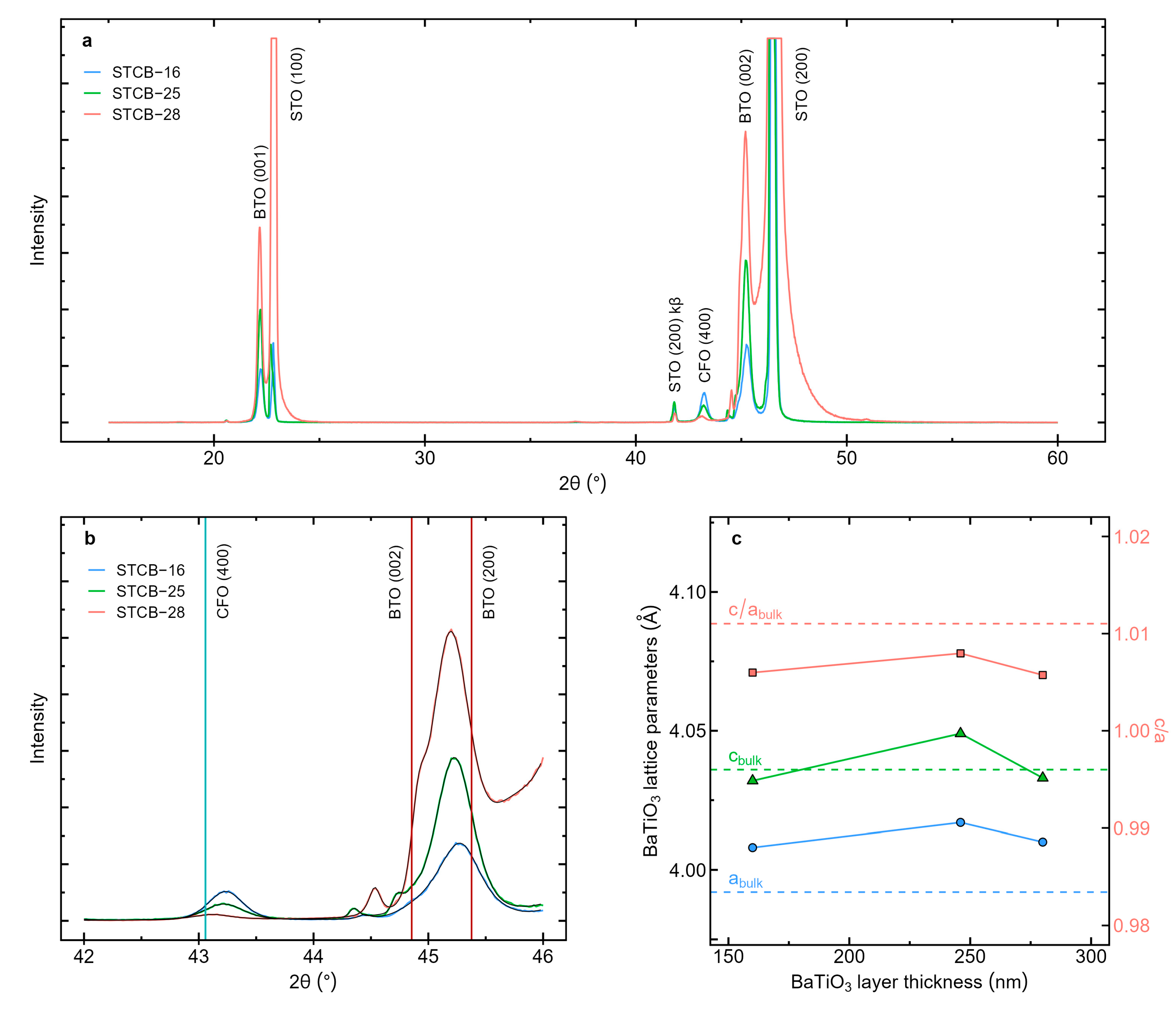
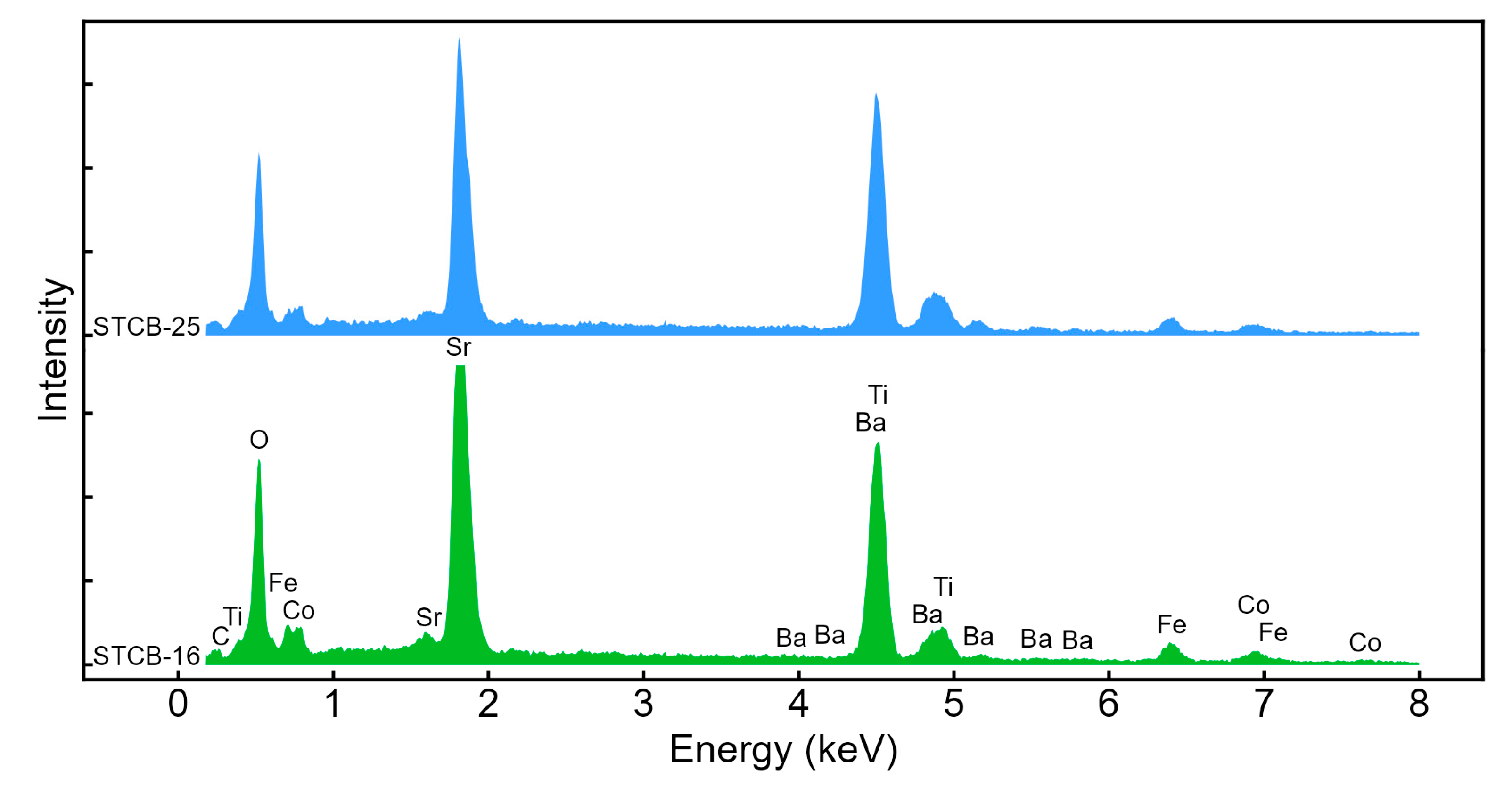
3.1. Structural and Morphological Properties
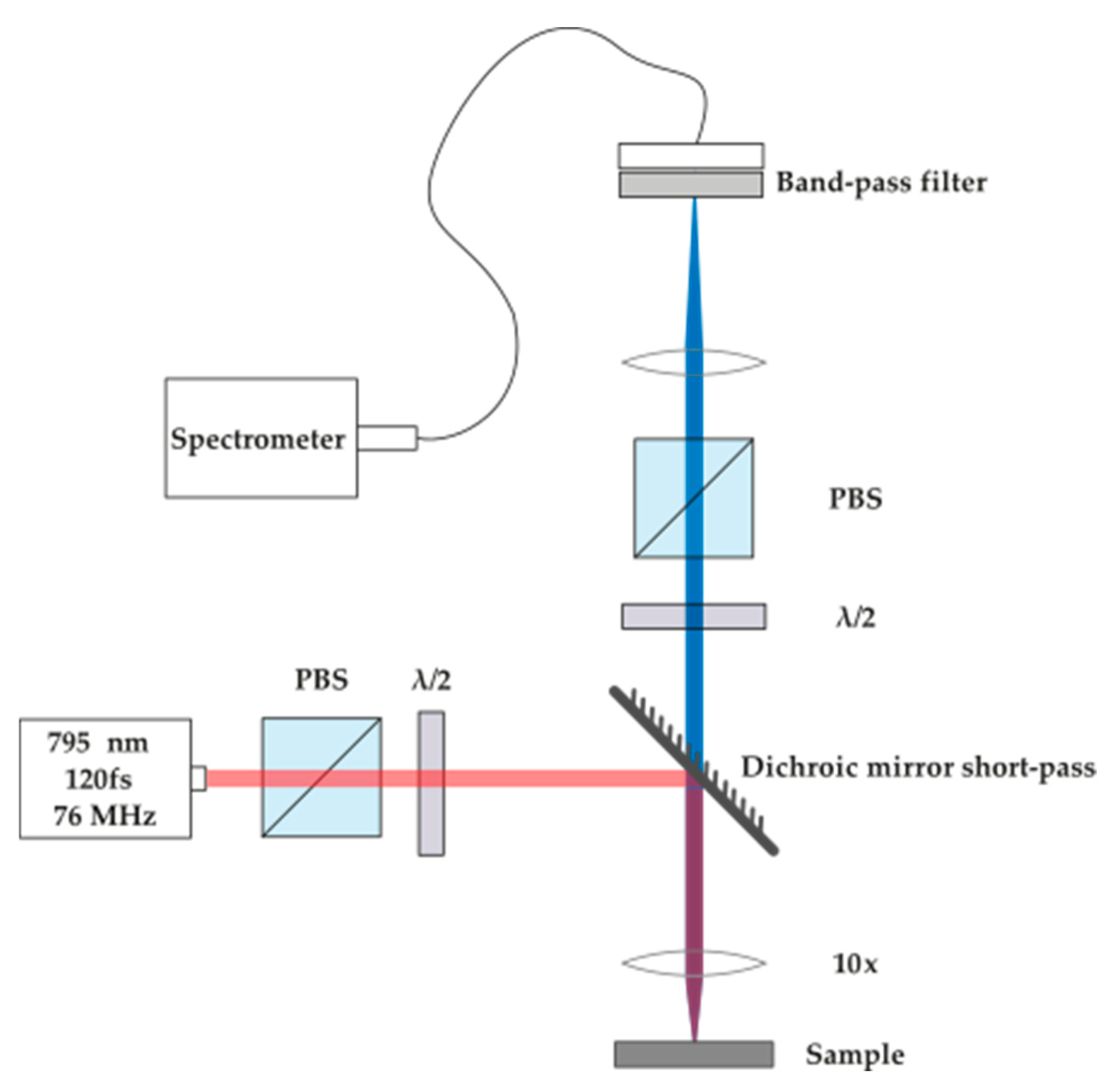
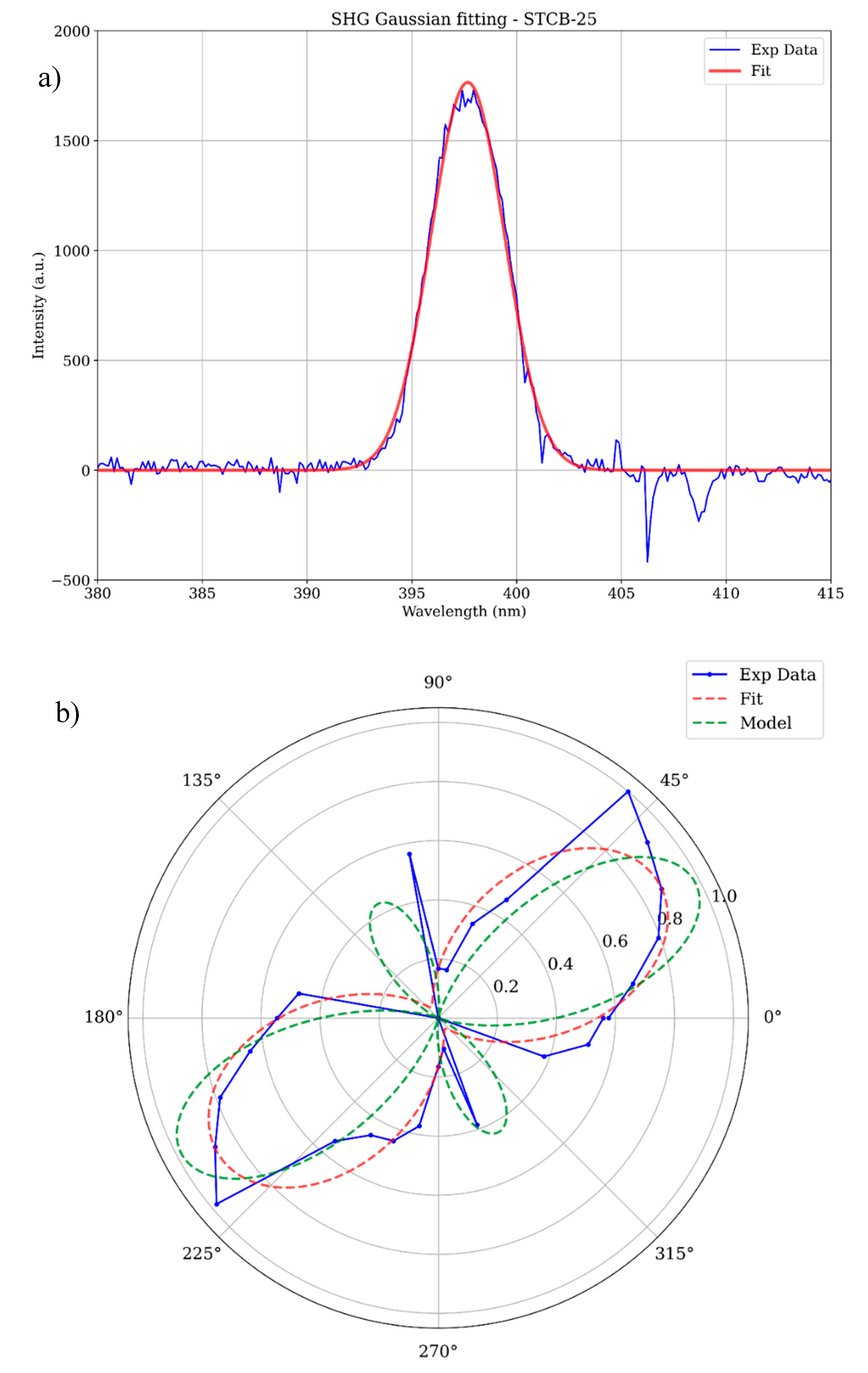
3.2. Second Harmonic Generation
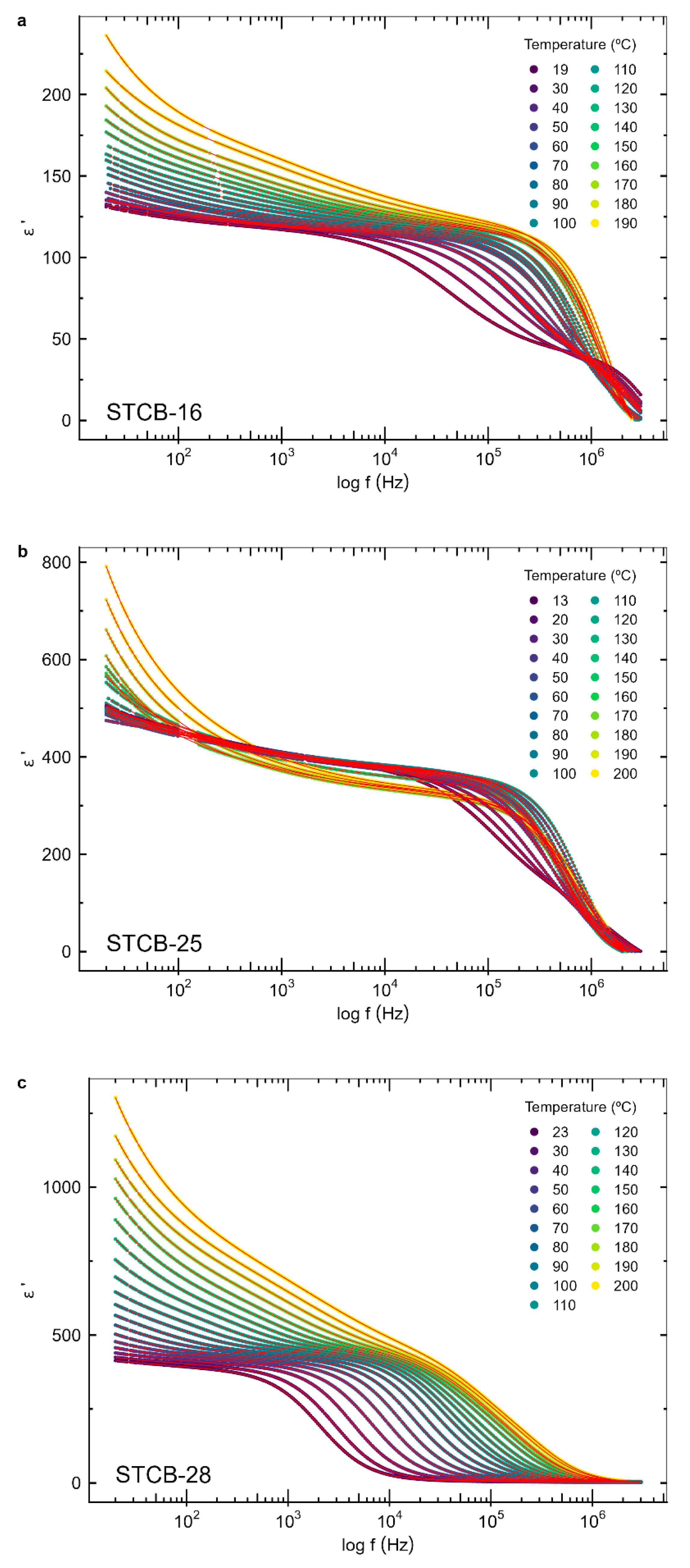
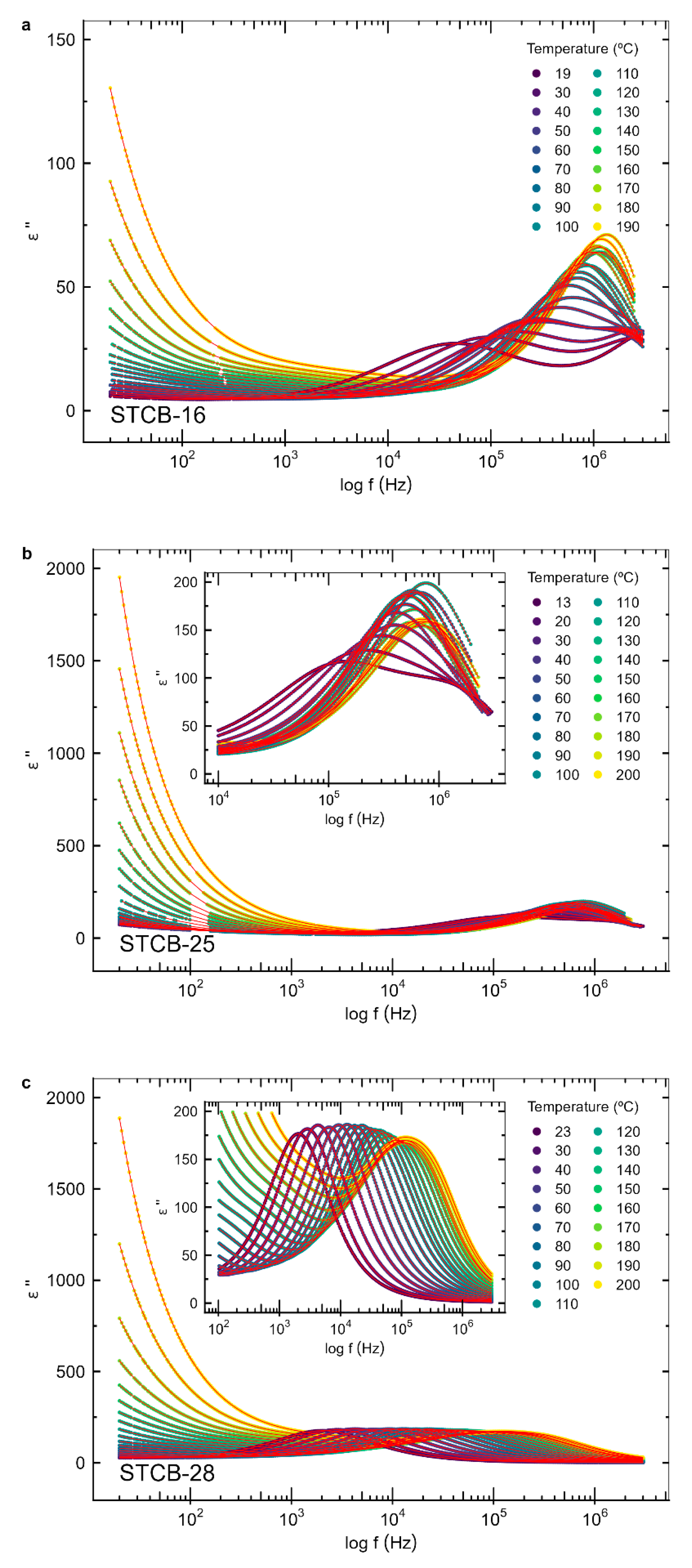
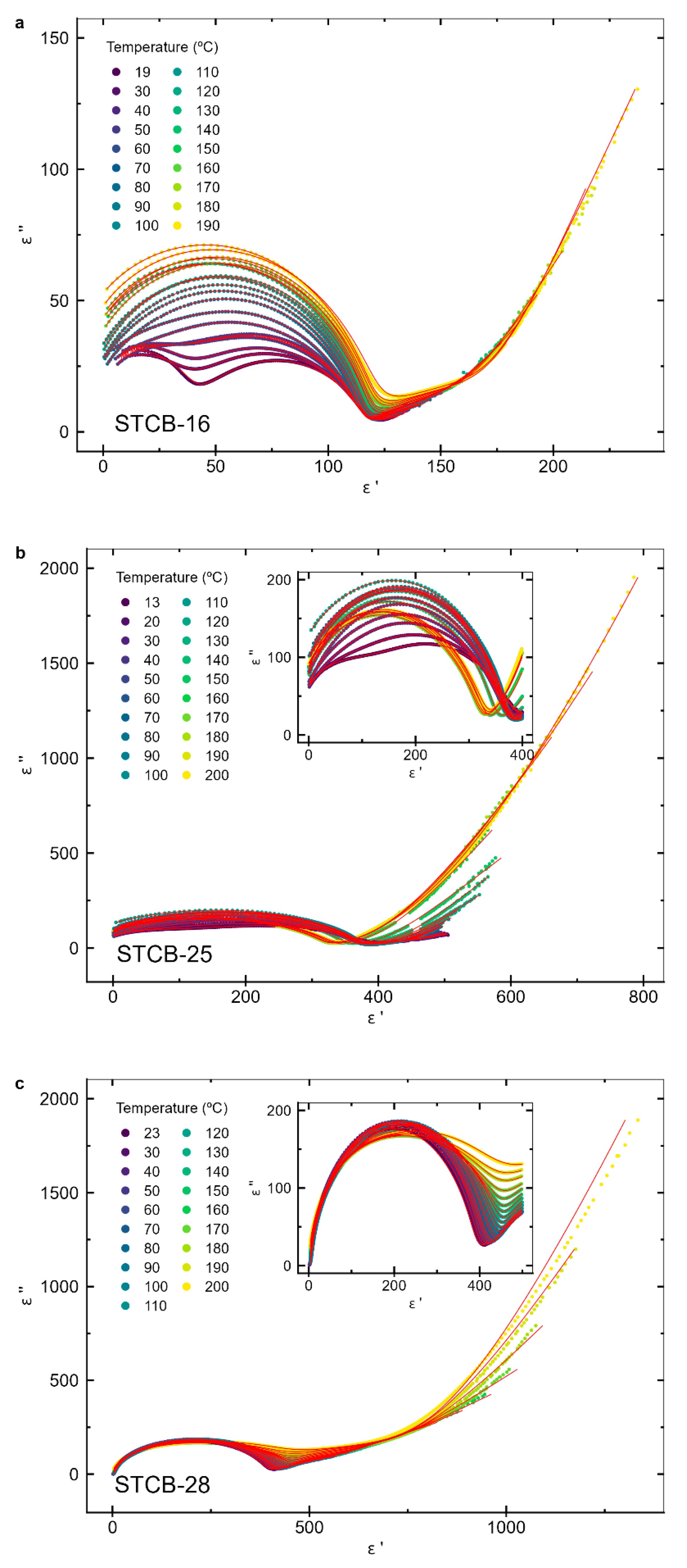
3.3. Dielectric Properties
3.4. AC Conductivity
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Taniyama, T.; Gohda, Y.; Hamaya, K.; Kimura, T. Artificial Multiferroic Heterostructures—Electric Field Effects and Their Perspectives. Sci. Technol. Adv. Mater. 2024, 25, 2412970. [Google Scholar] [CrossRef]
- Spaldin, N.A.; Ramesh, R. Advances in Magnetoelectric Multiferroics. Nat. Mater. 2019, 18, 203–212. [Google Scholar] [CrossRef] [PubMed]
- Leung, C.M.; Li, J.; Viehland, D.; Zhuang, X. A Review on Applications of Magnetoelectric Composites: From Heterostructural Uncooled Magnetic Sensors, Energy Harvesters to Highly Efficient Power Converters. J. Phys. D. Appl. Phys. 2018, 51, 263002. [Google Scholar] [CrossRef]
- Wang, J.; Chen, A.; Li, P.; Zhang, S. Magnetoelectric Memory Based on Ferromagnetic/Ferroelectric Multiferroic Heterostructure. Materials (Basel) 2021, 14. [Google Scholar] [CrossRef]
- Yu, P. Revolutions of Multiferroic Materials. npj Spintron. 2025, 3, 9. [Google Scholar] [CrossRef]
- Manipatruni, S.; Nikonov, D.E.; Young, I.A. Beyond CMOS Computing with Spin and Polarization. Nat. Phys. 2018, 14, 338–343. [Google Scholar] [CrossRef]
- Ghidini, M.; Mansell, R.; Maccherozzi, F.; Moya, X.; Phillips, L.C.; Yan, W.; Pesquera, D.; Barnes, C.H.W.; Cowburn, R.P.; Hu, J.-M.; et al. Shear-Strain-Mediated Magnetoelectric Effects Revealed by Imaging. Nat. Mater. 2019, 18, 840–845. [Google Scholar] [CrossRef]
- Chu, Z.; Pourhosseiniasl, M.; Dong, S. Review of Multi-Layered Magnetoelectric Composite Materials and Devices Applications. J. Phys. D. Appl. Phys. 2018, 51, 243001. [Google Scholar] [CrossRef]
- Gupta, R.; Kotnala, R.K. A Review on Current Status and Mechanisms of Room-Temperature Magnetoelectric Coupling in Multiferroics for Device Applications. J. Mater. Sci. 2022, 57, 12710–12737. [Google Scholar] [CrossRef]
- Barbosa, J.; Almeida, B.; Mendes, J.A.; Rolo, A.G.; Araújo, J.P. X-Ray Diffraction and Raman Study of Nanogranular BaTiO3–CoFe2O4 Thin Films Deposited by Laser Ablation on Si/Pt Substrates. Phys. status solidi 2007, 204, 1731–1737. [Google Scholar] [CrossRef]
- Juraschek, D.M.; Fechner, M.; Balatsky, A. V; Spaldin, N.A. Dynamical Multiferroicity. Phys. Rev. Mater. 2017, 1, 14401. [Google Scholar] [CrossRef]
- Sayedaghaee, S.O.; Xu, B.; Prosandeev, S.; Paillard, C.; Bellaiche, L. Novel Dynamical Magnetoelectric Effects in Multiferroic BiFeO3. Phys. Rev. Lett. 2019, 122, 97601. [Google Scholar] [CrossRef] [PubMed]
- Paiva, C.; Fechner, M.; Juraschek, D.M. Dynamically Induced Multiferroic Polarization. Phys. Rev. Lett. 2025, 135, 66702. [Google Scholar] [CrossRef] [PubMed]
- Bai, W.; Chen, C.; Yang, J.; Zhang, Y.; Qi, R.; Huang, R.; Tang, X.; Duan, C.G.; Chu, J. Dielectric Behaviors of Aurivillius Bi5 Ti3Fe0.5Cr0.5O15 Multiferroic Polycrystals: Determining the Intrinsic Magnetoelectric Responses by Impedance Spectroscopy. Sci. Rep. 2015, 5, 17846. [Google Scholar] [CrossRef]
- Kumar, M.; Arora, M.; Chauhan, S.; Pandey, H. Structural, Magnetic, Dielectric, Vibrational and Optical Properties of Zr Substituted Bi0.90Gd0.10FeO3 Multiferroics. J. Alloys Compd. 2018, 735, 684–691. [Google Scholar] [CrossRef]
- Sayedaghaee, S.O.; Paillard, C.; Prosandeev, S.; Xu, B.; Bellaiche, L. Strain-Induced Resonances in the Dynamical Quadratic Magnetoelectric Response of Multiferroics. npj Comput. Mater. 2020, 6, 60. [Google Scholar] [CrossRef]
- Li, Y.; Liao, Z.; Fang, F.; Wang, X.; Li, L.; Zhu, J. Significant Increase of Curie Temperature in Nano-Scale BaTiO3. Appl. Phys. Lett. 2014, 105, 182901. [Google Scholar] [CrossRef]
- Acosta, M.; Novak, N.; Rojas, V.; Patel, S.; Vaish, R.; Koruza, J.; Rossetti, G. A.; Rödel, J.J. BaTiO3-Based Piezoelectrics: Fundamentals, Current Status, and Perspectives. Appl. Phys. Rev. 2017, 4, 041305. [Google Scholar] [CrossRef]
- Buscaglia, V.; Randall, C.A. Size and Scaling Effects in Barium Titanate. An Overview. J. Eur. Ceram. Soc. 2020, 40, 3744–3758. [Google Scholar] [CrossRef]
- Jasrotia, R.; Prakash, J.; Saddeek, Y.B.; Alluhayb, A.H.; Younis, A.M.; Lakshmaiya, N.; Prakash, C.; Aly, K.A.; Sillanpää, M.; Ismail, Y.A.M.; et al. Cobalt Ferrites: Structural Insights with Potential Applications in Magnetics, Dielectrics, and Catalysis. Coord. Chem. Rev. 2025, 522, 216198. [Google Scholar] [CrossRef]
- Buscaglia, V.; Buscaglia, M.T. Core-Shell Heterostructures: From Particle Synthesis to Bulk Dielectric, Ferroelectric, and Multiferroic Composite Materials. Nanoscale Ferroelectr. Multiferroics Key Process. Charact. Issues, Nanoscale Eff. 2016, 72–99. [Google Scholar] [CrossRef]
- Shankar, S.; Thakur, O.P.; Jayasimhadri, M. Conductivity Behavior and Impedance Studies in BaTiO3–CoFe2O4 Magnetoelectric Composites. Mater. Chem. Phys. 2019, 234, 110–121. [Google Scholar] [CrossRef]
- Raza, S.A.; Awan, S.U.; Hussain, S.; Shah, S.A.; Iqbal, A.M.; Khurshid Hasanain, S. Structural, Ferromagnetic, Electrical, and Dielectric Relaxor Properties of BaTiO3 and CoFe2O4 Bulk, Nanoparticles, and Nanocomposites Materials for Electronic Devices. J. Appl. Phys. 2020, 128, 124101. [Google Scholar] [CrossRef]
- Zheng, H.; Wang, J.; Lofland, S.E.; Ma, Z.; Mohaddes-Ardabili, L.; Zhao, T.; Salamanca-Riba, L.; Shinde, S.R.; Ogale, S.B.; Bai, F.; et al. Multiferroic BaTiO3-CoFe2O4 Nanostructures. Science (80-. ) 2004, 303, 661–663. [Google Scholar] [CrossRef]
- Barbosa, J.; Almeida, B.; Pereira, A.M.; Araújo, J.P.; Gomes, I.; Mendes, J. Stress Induced Magnetic Anisotropy on BaTiO3-CoFe2O4 Nanogranular Composite Thin Films. J. Non. Cryst. Solids 2008, 354, 5250–5252. [Google Scholar] [CrossRef]
- Kim, D.; Rossell, M.D.; Campanini, M.; Erni, R.; Puigmartí-Luis, J.; Chen, X.-Z.; Pané, S. Magnetoelectric Coupling in Micropatterned BaTiO3/CoFe2O4 Epitaxial Thin Film Structures: Augmentation and Site-Dependency. Appl. Phys. Lett. 2021, 119, 12901. [Google Scholar] [CrossRef]
- Zhang, W.; Cheng, H.; Yang, Q.; Hu, F.; Ouyang, J. Crystallographic Orientation Dependent Dielectric Properties of Epitaxial BaTiO3 Thin Films. Ceram. Int. 2016, 42, 4400–4405. [Google Scholar] [CrossRef]
- Barbosa, J.G.; Gomes, I.T.; Pereira, M.R.; Moura, C.; Mendes, J.A.; Almeida, B.G. Structural and Dielectric Properties of Laser Ablated BaTiO3 Films Deposited over Electrophoretically Dispersed CoFe2O4 Grains. J. Appl. Phys. 2014, 116, 164112. [Google Scholar] [CrossRef]
- Araújo, C.; Almeida, B.G.; Aguiar, M.; Mendes, J.A. Structural and Magnetic Properties of CoFe2O4 Thin Films Deposited by Laser Ablation on Si (001) Substrates. Vacuum 2008, 82, 1437–1440. [Google Scholar] [CrossRef]
- Gomes, I.T.; Almeida, B.G.; Lopes, A.M.L.; Araújo, J.P.; Barbosa, J.; Mendes, J.A. Structural and Magnetic Characterization of LaSrMnO3 Thin Films Deposited by Laser Ablation on MgO Substrates. J. Magn. Magn. Mater. 2010, 322, 1174–1177. [Google Scholar] [CrossRef]
- Baghizadeh, A.; Vaghefi, P.M.; Huang, X.; Borme, J.; Almeida, B.; Salak, A.N.; Willinger, M.G.; Amaral, V.B.; Vieira, J.M. Interplay of Magnetic Properties and Doping in Epitaxial Films of H-REFeO3 Multiferroic Oxides. Small 2021, 17, 2005700. [Google Scholar] [CrossRef] [PubMed]
- Silva, B.M.; Oliveira, J.; Rebelo, T.; Isfahani, V.B.; Rocha-Rodrigues, P.; Lekshmi, N.; Belo, J.H.; Deepak, F.L.; Lopes, A.M.L.; Araújo, J.P.; et al. Synthesis, Structural and Dielectric Properties of Ca3Mn2O7 Thin Films Prepared by Pulsed Laser Deposition. Mater. Res. Bull. 2023, 158, 112066. [Google Scholar] [CrossRef]
- Cullity, B.D. Elements of X-Ray Diffraction; Addison Wesley Publishing Company: Reading, Massachusetts, 1978; Vol. 1, ISBN 0-201-01174-3. [Google Scholar]
- JCPDS-ICDD PDF: 22-1086. Natl. Bur. Stand. (U.S.) Monogr. 1971, 25, 9 22.
- Wang, J.J.; Meng, F.Y.; Ma, X.Q.; Xu, M.X.; Chen, L.Q. Lattice, Elastic, Polarization, and Electrostrictive Properties of BaTiO3 from First-Principles. J. Appl. Phys. 2010, 108, 034107. [Google Scholar] [CrossRef]
- Wang, J.J.; Meng, F.Y.; Ma, X.Q.; Xu, M.X.; Chen, L.Q. Lattice, Elastic, Polarization, and Electrostrictive Properties of BaTiO3 from First-Principles. J. Appl. Phys. 2010, 108, 034107. [Google Scholar] [CrossRef]
- Boyd, R.W. Nonlinear Optics, 2nd ed.; Academic Press: San Diego, 2003; ISBN 978-0-12-121682-5. [Google Scholar]
- Wang, H.; Weiner, A.M. Efficiency of Short-Pulse Type-I Second-Harmonic Generation with Simultaneous Spatial Walk-off, Temporal Walk-off, and Pump Depletion. IEEE J. Quantum Electron. 2003, 39, 1600–1618. [Google Scholar] [CrossRef]
- Smith, A. V SNLO Nonlinear Optics Code Available from AS-Photonics; Albuquerque, NM.
- Shen, Y.-R. Principles of Nonlinear Optics, 1st ed.; Wiley-Interscience: New York, NY, USA, 1984; ISBN 9780471889984. [Google Scholar]
- Chernova, E.; Brooks, C.; Chvostova, D.; Bryknar, Z.; Dejneka, A.; Tyunina, M. Optical NIR-VIS-VUV Constants of Advanced Substrates for Thin-Film Devices. Opt. Mater. Express 2017, 7, 3844–3862. [Google Scholar] [CrossRef]
- Kekesi, R.; Royer, F.; Jamon, D.; Blanc Mignon, M.F.; Abou-Diwan, E.; Chatelon, J.P.; Neveu, S.; Tombacz, E. 3D Magneto-Photonic Crystal Made with Cobalt Ferrite Nanoparticles Silica Composite Structured as Inverse Opal. Opt. Mater. Express 2013, 3, 935–947. [Google Scholar] [CrossRef]
- Kremer, F.; Schönhals, A. Broadband Dielectric Spectroscopy, 1st ed.; Kremer, F., Schönhals, A., Eds.; Springer Berlin: Heidelberg, 2002; ISBN 978-3-642-62809-2. [Google Scholar]
- Silva, B.M.; Oliveira, J.; Rebelo, T.; Calianguila, A.; Silva, D.; Mendes, J.; Almeida, B.G. Influence of LiNbO3 Layer Thickness on Structural and Dielectric Properties of CoFe2O4\LiNbO3 Multiferroic Bilayers Prepared by Laser Ablation. Mater. Chem. Phys. 2023, 307, 128198. [Google Scholar] [CrossRef]
- Górska, K.; Horzela, A.; Penson, K.A. The Havriliak-Negami and Jurlewicz-Weron-Stanislavsky Relaxation Models Revisited: Memory Functions Based Study. J. Phys. A Math. Theor. 2023, 56, 313001. [Google Scholar] [CrossRef]
- Nuernberg, R.B. Numerical Comparison of Usual Arrhenius-Type Equations for Modeling Ionic Transport in Solids. Ionics (Kiel) 2020, 26, 2405–2412. [Google Scholar] [CrossRef]
- Almahmoud, E.; Kornev, I.; Bellaiche, L. Dependence of Curie Temperature on the Thickness of an Ultrathin Ferroelectric Film. Phys. Rev. B 2010, 81, 64105. [Google Scholar] [CrossRef]
- Li, Y.; Baberschke, K. Dimensional Crossover in Ultrathin Ni(111) Films on W(110). Phys. Rev. Lett. 1992, 68, 1208–1211. [Google Scholar] [CrossRef]
- Deger, A.; Flindt, C. Determination of Universal Critical Exponents Using Lee-Yang Theory. Phys. Rev. Res. 2019, 1, 23004. [Google Scholar] [CrossRef]
- Almahmoud, E.; Kornev, I.; Bellaiche, L. Critical Behavior in Ferroelectrics from First Principles. Phys. Rev. Lett. 2009, 102, 105701. [Google Scholar] [CrossRef]
- Oliveira, J.; Silva, B.M.; Rebelo, T.; Rodrigues, P. V.; Baptista, R.M.F.; Rodrigues, M.J.L.F.; Belsley, M.; Lekshmi, N.; Araújo, J.P.; Mendes, J.A.; et al. Structural, Magnetic, and Dielectric Properties of Laser-Ablated CoFe2O4/BaTiO3 Bilayers Deposited over Highly Doped Si(100). Materials (Basel) 2024, 17, 5707. [Google Scholar] [CrossRef]
- Li, M.D.; Tang, X.G.; Zeng, S.M.; Jiang, Y.P.; Liu, Q.X.; Zhang, T.F.; Li, W.H. Oxygen-Vacancy-Related Dielectric Relaxation Behaviours and Impedance Spectroscopy of Bi(Mg1/2Ti1/2)O3 Modified BaTiO3 Ferroelectric Ceramics. J. Mater. 2018, 4, 194–201. [Google Scholar] [CrossRef]
- Cha, S.H.; Han, Y.H. Effects of Oxygen Vacancies on Relaxation Behavior of Mg-Doped BaTiO3. Japanese J. Appl. Physics, Part 1 Regul. Pap. Short Notes Rev. Pap. 2006, 45, 7797–7800. [Google Scholar] [CrossRef]
- Choi, M.; Oba, F.; Tanaka, I. Electronic and Structural Properties of the Oxygen Vacancy in BaTiO3. Appl. Phys. Lett. 2011, 98, 172901. [Google Scholar] [CrossRef]
- Hwang, J.; Kolodiazhnyi, T.; Yang, J.; Couillard, M. Doping and Temperature-Dependent Optical Properties of Oxygen-Reduced BaTiO3-δ. Phys. Rev. B - Condens. Matter Mater. Phys. 2010, 82, 214109. [Google Scholar] [CrossRef]
- Schrader, M.; Mienert, D.; Oh, T.S.; Yoo, H.I.; Becker, K.D. An Optical, EPR and Electrical Conductivity Study of Blue Barium Titanate, BaTiO3-δ. Solid State Sci. 2008, 10, 768–775. [Google Scholar] [CrossRef]
- Jing, X.; Xu, W.; Yang, C.; Feng, J.; Zhang, A.; Zeng, Y.; Qin, M.; Zeng, M.; Fan, Z.; Gao, J.; et al. Tuning Electrical Conductivity, Charge Transport, and Ferroelectricity in Epitaxial BaTiO3 Films by Nb-Doping. Appl. Phys. Lett. 2017, 110, 182903. [Google Scholar] [CrossRef]
- Tyunina, M.; Savinov, M.; Dejneka, A. Small-Polaron Conductivity in Perovskite Ferroelectric BaTiO3 Films. Appl. Phys. Lett. 2022, 121, 202901. [Google Scholar] [CrossRef]
- Tyunina, M.; Savinov, M.; Dejneka, A. Small-Polaron Conductivity in Perovskite Ferroelectric BaTiO3 Films. Appl. Phys. Lett. 2022, 121, 202901. [Google Scholar] [CrossRef]
- El Bachiri, A.; F., B.; Bousselamti, M. Dielectric and Electrical Properties of LiNbO3 Ceramics. J. Asian Ceram. Soc. 2016, 4, 46–54. [Google Scholar] [CrossRef]
- Ortega, N.; Kumar, A.; Bhattacharya, P.; Majumder, S.B.; Katiyar, R.S. Impedance Spectroscopy of Multiferroic PbZrxTi1−xO3/CoFe2O4 Layered Thin Films. Phys. Rev. B 2008, 77, 14111. [Google Scholar] [CrossRef]
- Dam, T.; Jena, S.S.; Pradhan, D.K. The Ionic Transport Mechanism and Coupling between the Ion Conduction and Segmental Relaxation Processes of PEO20-LiCF3SO3 Based Ion Conducting Polymer Clay Composites. Phys. Chem. Chem. Phys. 2016, 18, 19955–19965. [Google Scholar] [CrossRef]
- Macdonald, J.R. Comparison of the Universal Dynamic Response Power-Law Fitting Model for Conducting Systems with Superior Alternative Models. Solid State Ionics 2000, 133, 79–97. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Müller, M.; Khemani, V.; Knap, M.; Demler, E.; Huse, D.A. Low-Frequency Conductivity in Many-Body Localized Systems. Phys. Rev. B 2015, 92, 104202. [Google Scholar] [CrossRef]
- Gillot, B.; Jemmali, F. Dependence of Electrical Properties in Iron-Cobalt, Iron-Zinc Ferrites near Stoichiometry on Firing Temperature and Atmosphere. Phys. Status Solidi 1983, 76, 601–608. [Google Scholar] [CrossRef]
- George, M.; Nair, S.S.; Malini, K.A.; Joy, P.A.; Anantharaman, M.R. Finite Size Effects on the Electrical Properties of Sol–Gel Synthesized CoFe2O4 Powders: Deviation from Maxwell–Wagner Theory and Evidence of Surface Polarization Effect. J. Phys. D. Appl. Phys. 2007, 40, 1593–1602. [Google Scholar] [CrossRef]
- Zhang, R.; Sun, L.; Wang, Z.; Hao, W.; Cao, E.; Zhang, Y. Dielectric and Magnetic Properties of CoFe2O4 Prepared by Sol-Gel Auto-Combustion Method. Mater. Res. Bull. 2018, 98, 133–138. [Google Scholar] [CrossRef]
- Ponpandian, N.; Balaya, P.; Narayanasamy, A. Electrical Conductivity and Dielectric Behaviour of Nanocrystalline NiFe2O4 Spinel. J. Phys. Condens. Matter 2002, 14, 3221. [Google Scholar] [CrossRef]
- Elliott, S.R. A.c. Conduction in Amorphous Chalcogenide and Pnictide Semiconductors. Adv. Phys. 1987, 36, 135–217. [Google Scholar] [CrossRef]
- Karmakar, S.; Behera, D. Non-Overlapping Small Polaron Tunneling Conduction Coupled Dielectric Relaxation in Weak Ferromagnetic NiAl2O4. J. Phys. Condens. Matter 2019, 31, 245701. [Google Scholar] [CrossRef] [PubMed]
- Long, A.R. Frequency-Dependent Loss in Amorphous Semiconductors. Adv. Phys. 1982, 31, 553–637. [Google Scholar] [CrossRef]
- Yadav, A.; Jha, P.A.; Jha, P.K.; Jha, N.; Singh, P. Overlapping Large Polaron Tunnelling in Lanthanum Silicate Oxyapatite. J. Phys. Condens. Matter 2023, 35, 95702. [Google Scholar] [CrossRef]
- Sahu, S.; Dobbidi, P. Unlocking the Impact of Layer Configuration on Dielectric Relaxation and Conduction Mechanisms in Bilayer Ni0.5Co0.5Fe2O4/BaTiO3 and BaTiO3/Ni0.5Co0.5Fe2O4 Multiferroic Films. Appl. Surf. Sci. 2026, 715, 164591. [Google Scholar] [CrossRef]
- Ghosh, A. Frequency-Dependent Conductivity in Bismuth-Vanadate Glassy Semiconductors. Phys. Rev. B 1990, 41, 1479–1488. [Google Scholar] [CrossRef]
- Karoui, S.; Kamoun, S. Study of Dielectric Relaxation and Polaron Conductivity Mechanism in Sodium Nitroprusside (SNP): Na2[Fe(CN)5(NO)]·2H2O. Phys. E Low-dimensional Syst. Nanostructures 2021, 132, 114771. [Google Scholar] [CrossRef]
- Lisenkov, S.; Ladera, A.; Ponomareva, I. Ba(Ti_1-x,Zr_x)O3 Relaxors: Dynamic Ferroelectrics in the Gigahertz Frequency Range. Phys. Rev. B 2020, 102, 224109. [Google Scholar] [CrossRef]
- Meena, R.; Dhaka, R.S. Dielectric Properties and Impedance Spectroscopy of NASICON Type Na3Zr2Si2PO12. Ceram. Int. 2022, 48, 35150–35159. [Google Scholar] [CrossRef]
- Chun, J.; Heo, J.; Lee, K.; Ye, B.U.; Kang, B.S.; Yoon, S.-H. Thermal Activation Energy on Electrical Degradation Process in BaTiO3 Based Multilayer Ceramic Capacitors for Lifetime Reliability. Sci. Rep. 2024, 14, 616. [Google Scholar] [CrossRef]

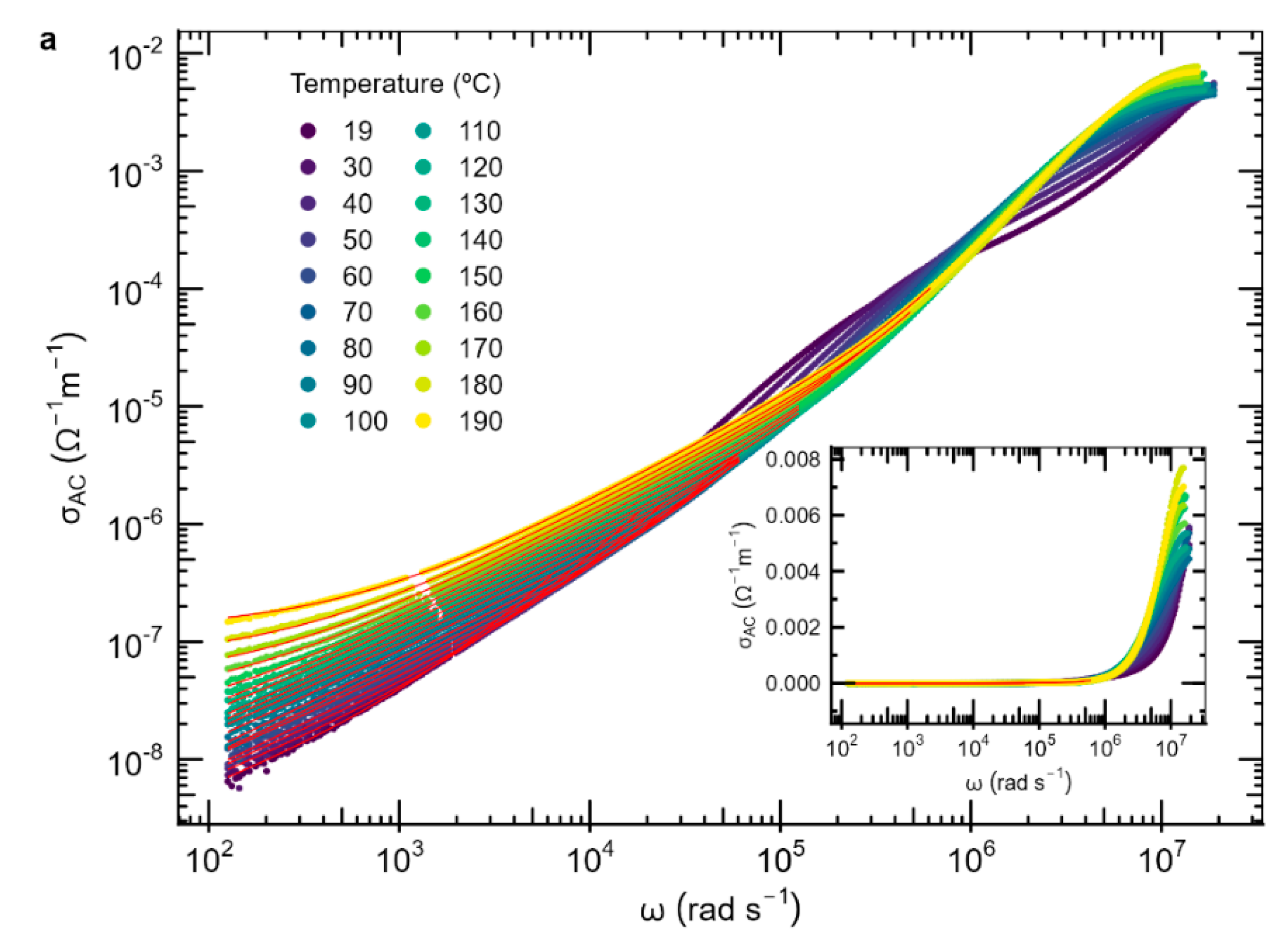
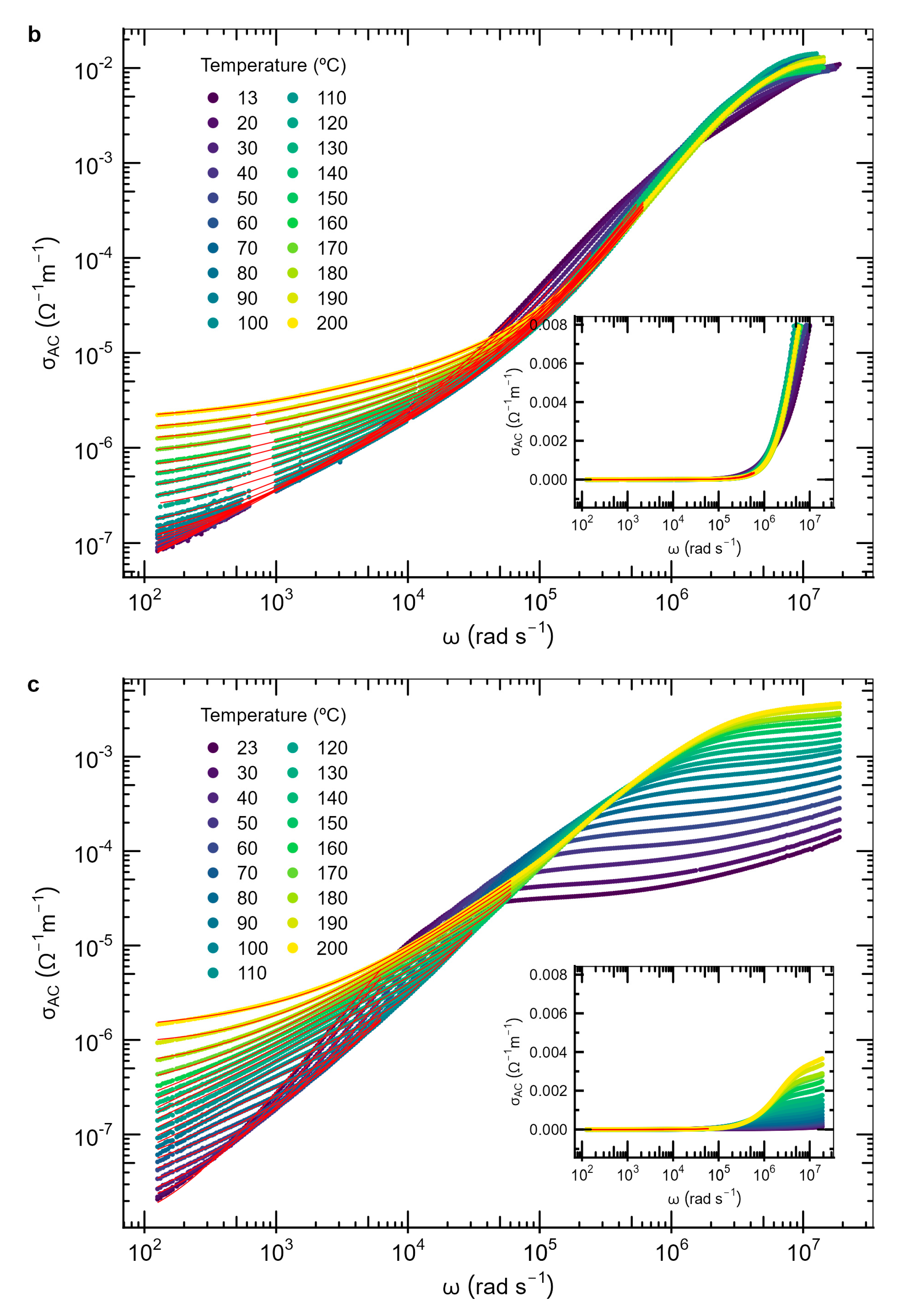
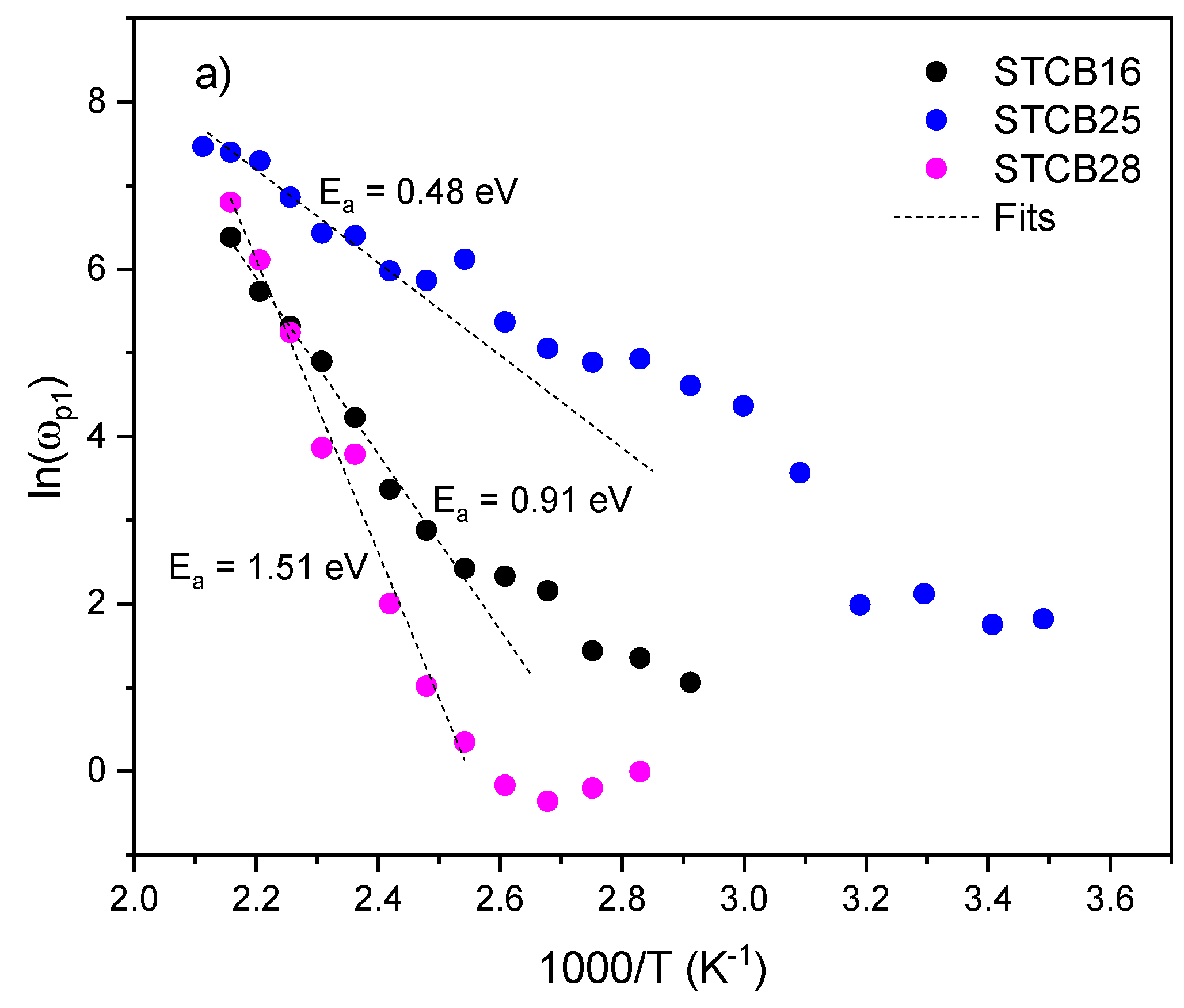
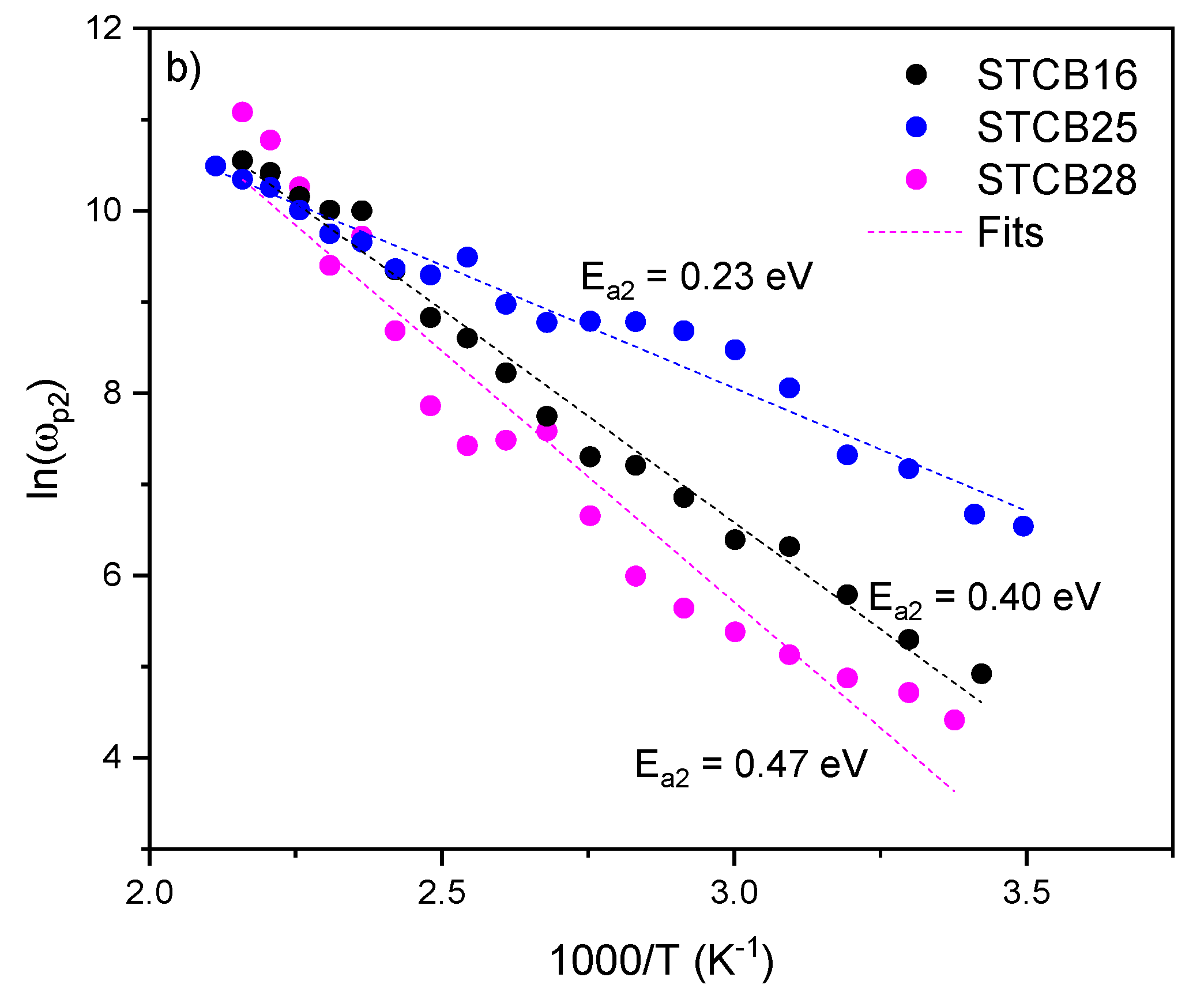
| Material | P (mbar) | f (Hz) | d (cm) | T (°C) |
|---|---|---|---|---|
| CoFe2O4 | 0.1 | 10 | 3.5 | 650 |
| BaTiO3 | 0.03 | 5 | 5.0 | 700 |
| Sample | Thickness CoFe2O4 (nm) |
Thickness BaTiO3 (nm) |
Latt. Param. CoFe2O4 (Å) | Grain Size CoFe2O4 (nm) | Grain Size BaTiO3 (nm) |
|---|---|---|---|---|---|
| STCB-16 | 72 | 160 | 8.377 | 32 | 32 |
| STCB-25 | 82 | 246 | 8.391 | 32 | 30 |
| STCB-28 | 79 | 280 | 8.386 | 28 | 33 |
| Sample |
Eσ LT (eV) |
Eσ HT (eV) |
TC (°C) |
|---|---|---|---|
| STCB-16 | 0.13 | 0.68 | 95 |
| STCB-25 | 0.22 | 0.63 | 110 |
| STCB-28 | 0.47 | 0.98 | 135 |
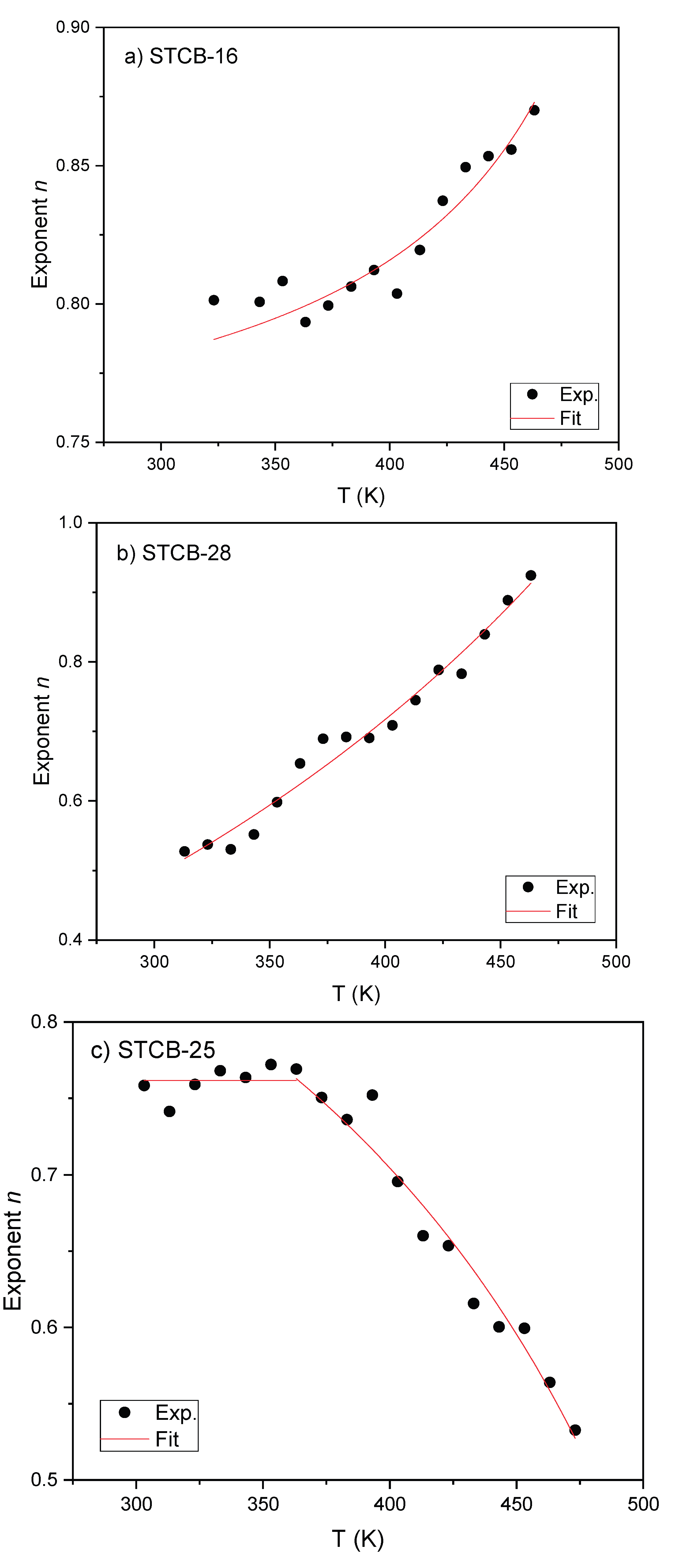
| Sample | WH (eV) NSTP |
τ0 (s) NSTP |
N(EF) (eV-1cm-3) NSTP | RT (Å) NSPT |
τ0 (s) QMT |
N(EF) (eV-1cm-3) QMT | Rω (Å) QMT |
W (eV) CBH |
|---|---|---|---|---|---|---|---|---|
| STCB-16 | 0.27 | 7.0×10−12 | 2.00×1024 | 7.3 | ||||
| STCB-25 | 8.3×10−13 | 1.14×1022 | 4.2 | 1.39 | ||||
| STCB-28 | 0.35 | 1.4×10−8 | 3.25×1025 | 1.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





