Submitted:
03 December 2025
Posted:
04 December 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material & Methods
2.1. Preparation of Epithelial Cell Monolayers (Cell Culture)
2.2. Preparation of Bacterial Cultures and Zgougou Juices
2.3. Exposure of Caco-2 Cell Line to Bacterial Biomass, CFS, and Zgougou Juices
2.4. Gene Level Expression Related to Intestinal Barrier Integrity
2.5. Quantification of TNFα
2.6. Statistical Analysis
3. Results & Discussions
3.1. Impact of Probiotics and Fermentation on Gut Integrity
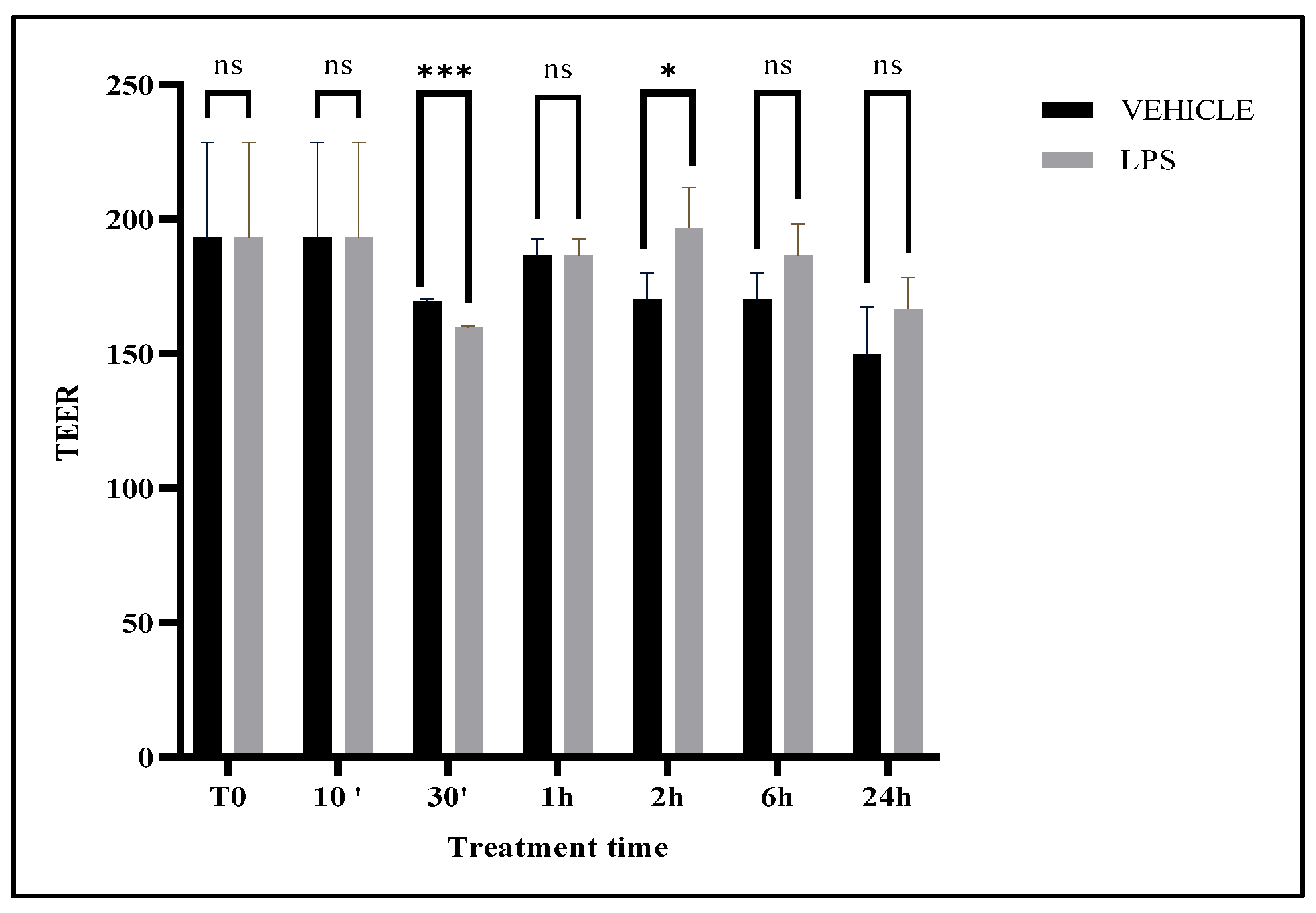
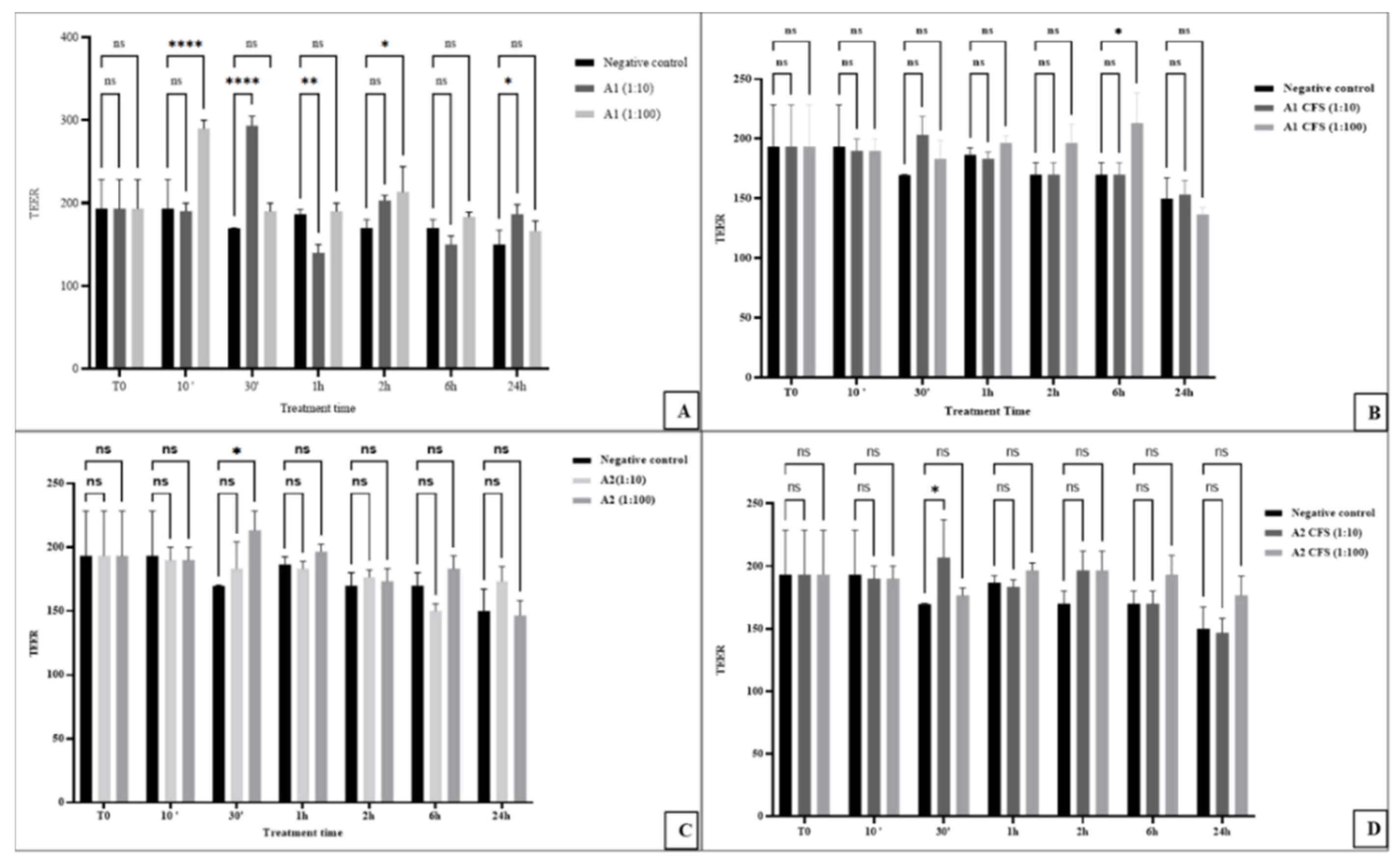
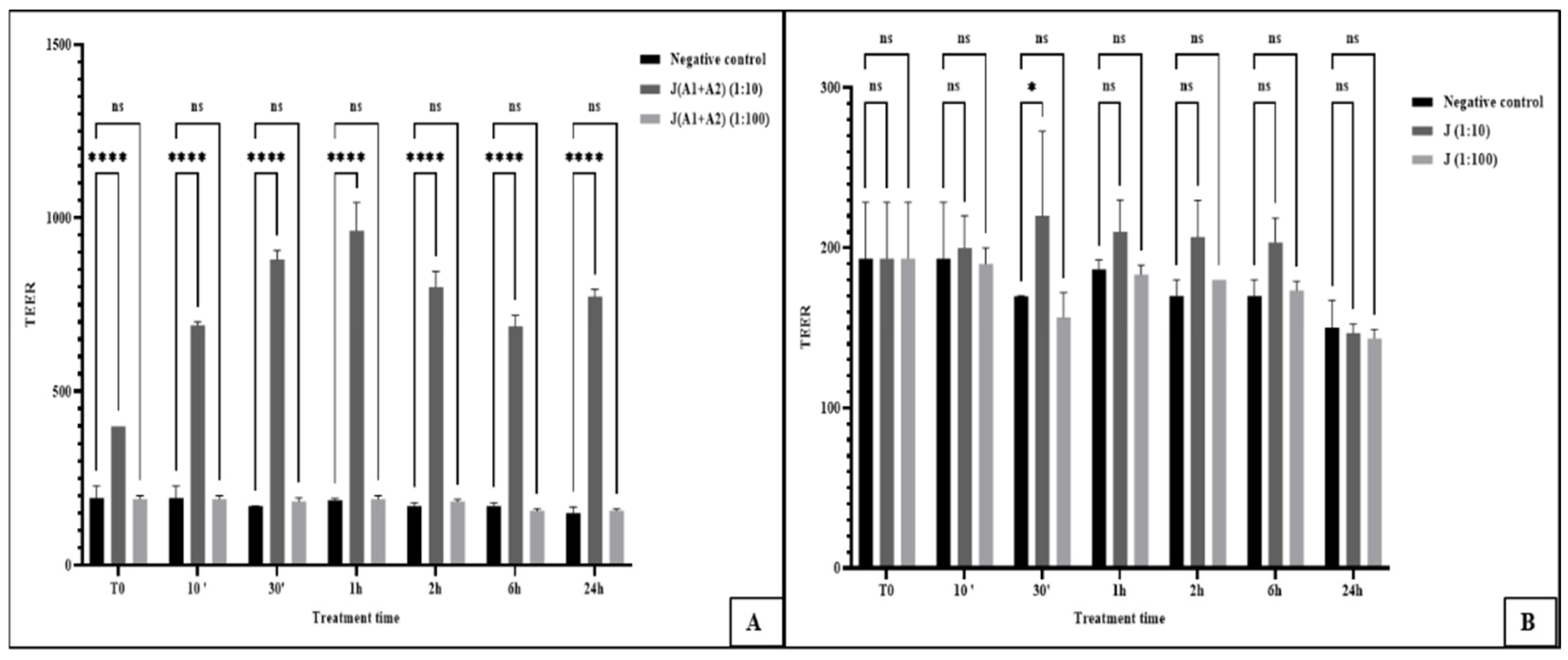
3.2. Differential Modulation of Tight Junction Genes (CLDN2 and OCLN)
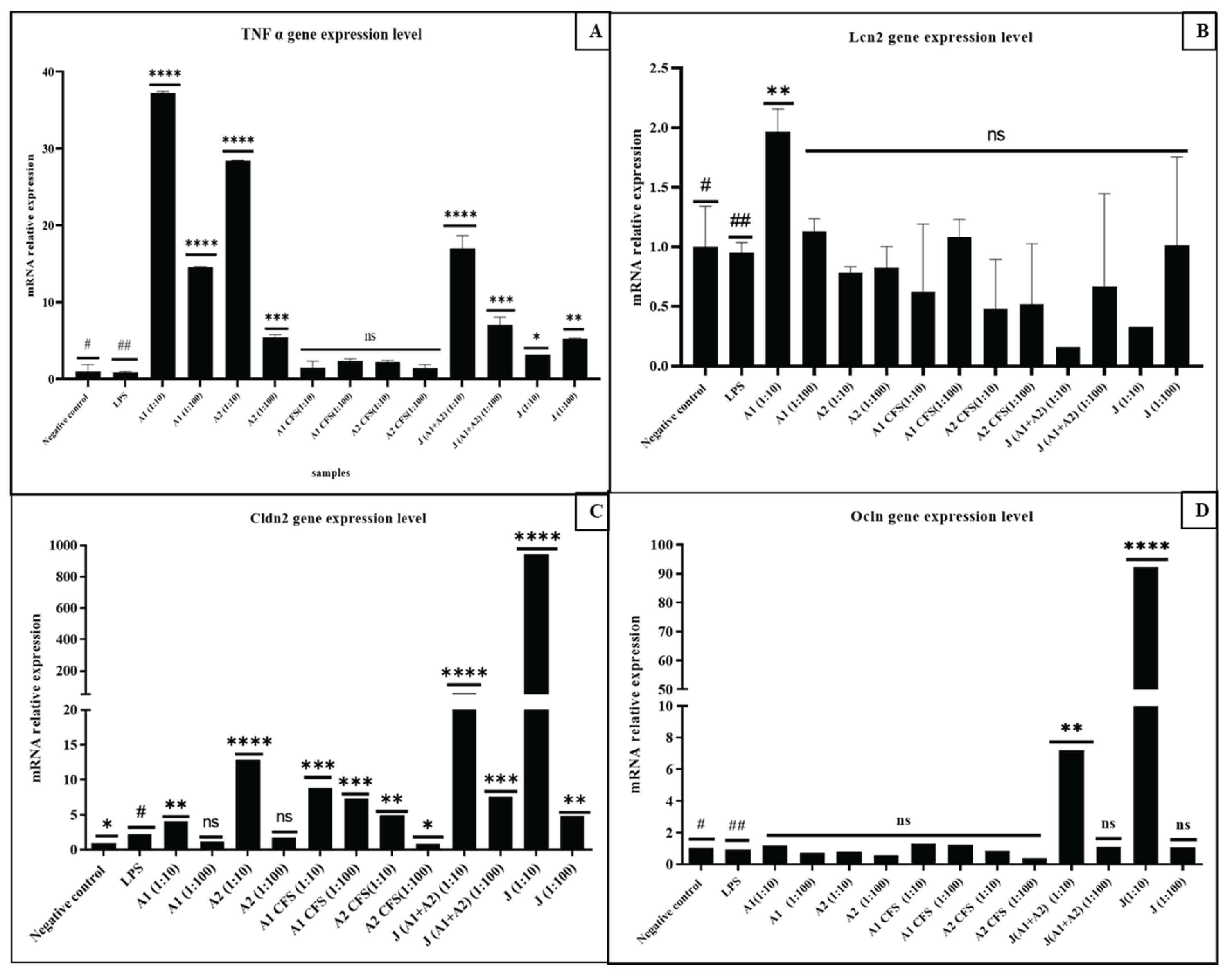
3.3. Modulation of TNFalpha Gene Expression in Caco-2 Monolayers
4. Conclusions and Perspectives
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| Caco-2 cell | Caucasian Colon Adenoarcinoma Cells |
| CFS | Cell-Free Supernatant |
| CLDN2 | Claudin-2 |
| HT-29 | Human Colon Carcinoma Cell Line |
| IBD | Inflammatory Bowel Disease |
| IECs | Intestinal Epithelial Cells |
| JAMS | Junction Adhesion Molecules |
| J | Zgougou Juice Obtained Upon Both Spontaneous |
| J(A1+A2) | Zgougou Juice Driven Fermentation With Autochthonous Probiotic Bacteria |
| LAB | Lactic Acid Bacteria |
| LCN2 | Lipocalin-2 |
| LP | LactiplantibacillusPlantarum |
| LPS | Escherchia Coli Lipo Poly Saccharide |
| LTA | Lipoteichoic Acid |
| OCLN | Occludin |
| RT-PCR | Real Time- Polymorphism Chain Reaction |
| TEER | Trans Epithelial Electrical Resistance |
| TJs | Tight Junctions |
| TJP | Tight Junction Proteins |
| TLR2 | Toll Like Receptor |
| TNFα | Tumour Necrosis Factor Alpha |
References
- Ait Atmane, S.; Aksoylu Özbek, Z.; Günç Ergönül, P.; Khettal, B. Valorization of Pinus halepensis Mill. seed oil: Physicochemical characteristics, bioactive compounds, and antioxidant activity as affected by location and extraction method. J. Food Process. Preserv. 2021, 45, e15548. [Google Scholar] [CrossRef]
- Ait Atmane, S.; Batır, M. B.; Özbek, Z. A.; et al. Cold pressed Pinus halepensis Mill. seed oil for potential health applications: Analgesic, anti-inflammatory effects, and assessment of inflammatory mediators by RT-qPCR in skin wound healing. J. Ethnopharmacol. 2024, 319, 117157. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.C.; Cookson, A.L.; McNabb, W.C.; et al. Lactobacillus plantarum MB452 enhances the function of the intestinal barrier by increasing the expression levels of genes involved in tight junction formation. BMC Microbiol. 2010, 10, 316. [Google Scholar] [CrossRef]
- Ashaolu, T. J. Immune boosting functional foods and their mechanisms: A critical evaluation of probiotics and prebiotics. Biomed. Pharmacother. 2020, 130, 110625. [Google Scholar] [CrossRef]
- Ashida, N.; Yanagihara, S.; Shinoda, T.; Yamamoto, N. Characterization of adhesive molecule with affinity to Caco-2 cells in Lactobacillus acidophilus by proteome analysis. J. Biosci. Bioeng. 2011, 112, 333–337. [Google Scholar] [CrossRef]
- Aslam, H.; Green, J.; Jacka, F. N.; et al. Fermented foods, the gut and mental health: a mechanistic overview with implications for depression and anxiety. Nutr. Neurosci. 2020, 23, 659–671. [Google Scholar] [CrossRef]
- Bae, W.Y.; Jung, W.H.; Shin, S.L.; et al. Investigation of Immunostimulatory Effects of Heat-Treated Lacti-plantibacillus plantarum LM1004 and Its Underlying Molecular Mechanism. Food Sci. Anim. Resour. 2022, 2, 42–1031. [Google Scholar]
- Balda, M. S.; Matter, K. Transmembrane proteins of tight junctions. Semin. Cell Dev. Biol. 2000, 11, 281–289. [Google Scholar] [CrossRef]
- Bamola, V.D.; Dubey, D.; Samanta, P.; et al. Role of a probiotic strain in the modulation of gut microbiota and cytokines in inflammatory bowel disease. Anaerobe 2022, 78, 102652. [Google Scholar] [CrossRef] [PubMed]
- Bermudez-Brito, M.; Muñoz-Quezada, S.; Gómez-Llorente, C.; et al. Lactobacillus paracasei CNCM I-4034 and its culture supernatant modulate Salmonella-induced inflammation in a novel transwell co-culture of human intestinal-like dendritic and Caco-2 cells. BMC Microbiol. 2015, 15, 79. [Google Scholar] [CrossRef]
- Botella, L.; Diez, J. J. Phylogenic diversity of fungal endophytes in Spanish stands of Pinus halepensis. Fungal Divers. 2011, 47, 9–18. [Google Scholar] [CrossRef]
- Campbell, H. K.; Maiers, J. L.; DeMali, K. A. Interplay between tight junctions & adherens junctions. Exp. Cell Res. 2017, 358, 39–44. [Google Scholar] [CrossRef]
- Cheikh-Rouhou, S.; Hentati, B.; Besbes, S.; et al. Chemical composition and lipid fraction characteristics of Aleppo pine (Pinus halepensis Mill.) seeds cultivated in Tunisia. Food Sci. Technol. Int. 2006, 12, 407–415. [Google Scholar] [CrossRef]
- Chopra, R.; Mathur, S. Probiotics in dentistry: A boon or sham. Dent. Res. J. 2013, 10, 302. [Google Scholar]
- Colegio, O. R.; Van Itallie, C.; Rahner, C.; Anderson, J. M. Claudin extracellular domains determine paracellular charge selectivity and resistance but not tight junction fibril architecture. Am. J. Physiol. Cell Physiol. 2003, 284, C1346–C1354. [Google Scholar] [CrossRef] [PubMed]
- Dasriya, V. L.; Samtiya, M.; Ranveer, S.; et al. Modulation of gut--microbiota through probiotics and dietary interventions to improve host health. J. Sci. Food Agric. 2024, 104, 6359–6375. [Google Scholar] [CrossRef]
- Di Cagno, R.; Filannino, P.; Gobbetti, M. Fermented foods: fermented vegetables and other products. In Encyclopedia of Food and Health; Elsevier Inc.: 2015; pp. 668–674.
- Escamilla, J.; Lane, M. A.; Maitin, V. Cell-free supernatants from probiotic Lactobacillus casei and Lactobacillus rhamnosus GG decrease colon cancer cell invasion in vitro. Nutr. Cancer 2012, 64, 871–878. [Google Scholar] [CrossRef]
- Guo, S.; Li, L.; Xu, B.; et al. A simple and novel fecal biomarker for colorectal cancer: ratio of Fusobacterium nucleatum to probiotics populations, based on their antagonistic effect. Clin. Chem. 2018, 64, 1327–1337. [Google Scholar] [CrossRef]
- Han, H.; You, Y.; Cha, S.; et al. Multi-Species Probiotic Strain Mixture Enhances Intestinal Barrier Function by Regulating Inflammation and Tight Junctions in Lipopolysaccharides Stimulated Caco-2 Cells. Microorganisms 2023, 11, 656. [Google Scholar] [CrossRef]
- Haque, M.; Kaminsky, L.; Abdulqadir, R.; et al. Lactobacillus acidophilus inhibits the TNF-α-induced increase in intestinal epithelial tight junction permeability via a TLR-2 and PI3K-dependent inhibition of NF-κB activation. Front. Immunol. 2024, 15, 1348010. [Google Scholar] [CrossRef]
- Huang, F.C. Upregulation of Salmonella-Induced IL-6 Production in Caco-2 Cells by PJ-34, PARP-1 Inhibitor: Involvement of PI3K, p38MAPK, ERK, JNK, and NF-κB. Mediators Inflamm. 2009, 103890. [Google Scholar] [CrossRef]
- Ilango, S.; Antony, U. Probiotic microorganisms from non-dairy traditional fermented foods. Trends Food Sci. Technol. 2021, 118, 617–638. [Google Scholar] [CrossRef]
- Kadar, A.D.; Astawan, M.; Putri, S.P.; Fukusaki, E.J.M. Metabolomics-Based Study of the Effect of Raw Materials to the End Product of Tempe—An Indonesian Fermented Soybean. Metabolites 2020, 10, 367. [Google Scholar] [CrossRef]
- Kadri, N.; Khettal, B.; Aid, Y.; Kherfellah, S.; Sobhi, W.; Barragan-Montero, V. Some physicochemical characteristics of pinus (Pinus halepensis Mill., Pinus pinea L., Pinus pinaster and Pinus canariensis) seeds from North Algeria, their lipid profiles and volatile contents. Food Chem. 2015, 188, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Karczewski, J.; Troost, F. J.; Konings, I.; et al. Regulation of human epithelial tight junction proteins by Lactobacillus plantarum in vivo and protective effects on the epithelial barrier. Am. J. Physiol. Gastrointest. Liver Physiol. 2010, 298, G851–G859. [Google Scholar] [CrossRef]
- Kim, H.; Hur, S.; Lim, J.; et al. Enhancement of the phenolic compounds and antioxidant activities of Kombucha prepared using specific bacterial and yeast. Food Biosci. 2023, 56, 103431. [Google Scholar] [CrossRef]
- Kita, M.; Ano, Y.; Inoue, A.; Aoki, J. Identification of P2Y receptors involved in oleamide-suppressing inflammatory responses in murine microglia and human dendritic cells. Sci. Rep. 2019, 9, 3135. [Google Scholar] [CrossRef]
- Klingberg, T. D.; Pedersen, M. H.; Cencic, A.; Budde, B. B. Application of measurements of transepithelial electrical resistance of intestinal epithelial cell monolayers to evaluate probiotic activity. Appl. Environ. Microbiol. 2005, 71, 7528–7530. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, K.; Imai, Y.; Shimizu, Y.; et al. Lactobacillus delbrueckii subsp. bulgaricus 2038 and Streptococcus thermophilus 1131 ameliorate barrier dysfunction in human induced pluripotent stem cell-derived crypt-villus structural small intestine. Front. Immunol. 2025, 16, 1585007. [Google Scholar] [CrossRef]
- Kobayashi, K.; Mochizuki, J.; Yamazaki, F.; Sashihara, T. Yogurt starter strains ameliorate intestinal barrier dysfunction via activating AMPK in Caco-2 cells. Tissue Barriers 2023, 12. [Google Scholar] [CrossRef]
- La Fata, G.; Weber, P.; Mohajeri, M. H. Probiotics and the gut immune system: indirect regulation. Probiotics Antimicrob. Proteins 2018, 10, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Lavermicocca, P. Highlights on new food research. Dig. Liver Dis. 2006, 38, S295–S299. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Kim, D. S.; Cho, J.; et al. Perilla frutescens Britton: A comprehensive study on flavor/taste and chemical properties during the roasting process. Molecules 2019, 24, 1374. [Google Scholar] [CrossRef]
- Lee, S.Y.; Sekhon, S.S.; Kim, H.C.; et al. Anti-inflammatory effect of lactic acid bacteria isolated from kimchi on acid-induced acute colitis in model mice. Toxicol. Environ. Health Sc. 2017, 9, 279–283. [Google Scholar] [CrossRef]
- Lightfoot, Y. L.; Yang, T.; Sahay, B.; Mohamadzadeh, M. Targeting aberrant colon cancer-specific DNA methylation with lipoteichoic acid-deficient Lactobacillus acidophilus. Gut Microbes 2013, 4, 84–88. [Google Scholar] [CrossRef]
- Lin, Q.; Mathieu, O.; Tompkins, T. A.; et al. Modulation of the TNF$\alpha$-induced gene expression profile of intestinal epithelial cells by soy fermented with lactic acid bacteria. J. Funct. Foods 2016, 23, 400–411. [Google Scholar] [CrossRef]
- Ma, Y.; Chen, X.; Xu, R.; et al. Lactiplantibacillus plantarum fermentation enhanced the protective effect of kiwifruit on intestinal injury in rats: Based on mitochondrial morphology and function. Food Chem. X 2023, 20, 101025. [Google Scholar] [CrossRef]
- Minervini, F.; Missaoui, J.; Celano, G.; et al. Use of autochthonous lactobacilli to increase the safety of zgougou. Microorganisms 2020, 8, 29. [Google Scholar] [CrossRef]
- Missaoui, J.; Saidane, D.; Mzoughi, R.; Minervini, F. Fermented Seeds (“Zgougou”) From Aleppo Pine as Novel Source of Potentially Probiotic Lactic Acid Bacteria. Microorganisms 2019, 7, 709. [Google Scholar] [CrossRef]
- Mosiej, W.; Dorota, Z.; Marcin, K.; et al. The Impact of food-derived lactic acid bacteria (LAB) Supplementation on Gut Microbiota Modulation in Elderly Individuals With Cognitive Impairment: Insights from the SHIME® Model. Int. J. Psychophysiol. 2025, 213, 113147. [Google Scholar] [CrossRef]
- Nasri, N.; Triki, S. Analyse des lipides des graines de pins de Tunisie (Pinus halepensis Mill. et Pinus pinea L.). Riv. Ital. Sostanze Grasse 2004, 81, 244–247. [Google Scholar]
- Nithya, A.; Misra, S.; Panigrahi, C.; et al. Probiotic potential of fermented foods and their role in non-communicable diseases management: An understanding through recent clinical evidences. Food Chem. Adv. 2023, 3, 100381. [Google Scholar] [CrossRef]
- Nouri, Z.; Karami, F.; Neyazi, N.; et al. Dual anti-metastatic and anti-proliferative activity assessment of two probiotics on HeLa and HT-29 cell lines. Cell J. (Yakhteh) 2016, 18, 127. [Google Scholar]
- Ohland, C.L.; Mac Naughton, W.K. Probiotic bacteria and intestinal epithelial barrier function. Am. J. Physiol. Ganstrointest. Liver Physiol. 2010, 298, G807–G819. [Google Scholar] [CrossRef]
- Oladejo, B.O.; Akingbade, G.T.; Adeboboye, C.F.; Adeoye, S.B. Lactobacillus species modulates TNF-α and Nrf2 expression in carrageenan-induced rat paw oedema. The Microbe 2024, 2, 100050. [Google Scholar] [CrossRef]
- Oudat, Q.; Okour, A. The Role of Probiotics in Modulating Gut Microbiota and Metabolic Health for Weight Management: A Mini Review. Acta Microbiol. Hell. 2025, 70, 5. [Google Scholar] [CrossRef]
- Paradis, T.; Bègue, H.; Basmaciyan, L.; Dalle, F.; Bon, F. Tight junctions as a key for pathogens invasion in intestinal epithelial cells. Int. J. Mol. Sci. 2021, 22, 2506. [Google Scholar] [CrossRef]
- Parlesak, A.; Haller, D.; Brinz, S.; et al. Modulation of cytokine release by differentiated CACO-2 cells in a compartmentalized coculture model with mononuclear leucocytes and nonpathogenic bacteria. Scand. J. Immunol. 2004, 60, 477–485. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.; Butani, K.; Kumar, A.; et al. Effects of fermented food consumption on non-communicable diseases. Foods 2023, 12, 687. [Google Scholar] [CrossRef]
- Qin, H.; Zhang, Z.; Hang, X.; Jiang, Y. L. L. plantarum prevents enteroinvasive Escherichia coli-induced tight junction proteins changes in intestinal epithelial cells. BMC Microbiol. 2009, 9, 63. [Google Scholar] [CrossRef] [PubMed]
- Saha, P.; Chassaing, B.; Yeoh, B.S.; et al. Ectopic expression of innate immune protein, lipocalin-2, in Lactococcus lactis protects against gut and environmental stressors. Inflamm. Bowel Dis. 2017, 23, 1120–1132. [Google Scholar] [CrossRef] [PubMed]
- Sahu, B.; Johnson, L.M.; Sohrabi, M.; et al. Effects of Probiotics on Colitis-Induced Exacerbation of Alzheimer’s Disease in AppNL-G-F Mice. 2023, 24, 11551.
- Sharma, R.; Young, C.; Neu, J. Molecular modulation of intestinal epithelial barrier: contribution of microbiota. BioMed Res. Int. 2010, 2010, 305879. [Google Scholar] [CrossRef]
- Şengün, İ. Y.; Güney, D. Probiotic potential of fermented foods and their effects on immune system. Turk. J. Agric. Food Sci. Technol. 2021, 9, 1744–1750. [Google Scholar] [CrossRef]
- Schiller, G. Therapeutic Use of Aleppo Pine (Pinus halepensis Mill.). In Medicinal and Aromatic Plants of the Middle-East; Yaniv, Z., Dudai, N., Eds.; Springer Netherlands: Dordrecht, 2014; pp. 215–224. [Google Scholar]
- Suzuki, T. Regulation of the intestinal barrier by nutrients: The role of tight junctions. Anim. Sci. J. 2020, 91, e13357. [Google Scholar] [CrossRef]
- Talà, A.; Guerra, F.; Resta, S. C.; et al. Phenotyping of fecal microbiota of Winnie, a rodent model of spontaneous chronic colitis, reveals specific metabolic, genotoxic, and pro-inflammatory properties. Inflammation 2022, 45, 2477–2497. [Google Scholar] [CrossRef]
- Tiptiri-Kourpeti, A.; Spyridopoulou, K.; Santarmaki, V.; et al. Lactobacillus casei exerts anti-proliferative effects accompanied by apoptotic cell death and up-regulation of TRAIL in colon carcinoma cells. PloS One 2016, 11, e0147960. [Google Scholar] [CrossRef]
- Tiss, M.; Souiy, Z.; Ben Abdeljelil, N.; et al. Fermented soy milk prepared using kefir grains prevents and ameliorates obesity, type 2 diabetes, hyperlipidemia and Liver-Kidney toxicities in HFFD-rats. J. Funct. Foods 2020, 67, 103869. [Google Scholar] [CrossRef]
- Tukan, S. K.; Al-Ismail, K.; Ajo, R. Y.; Al-Dabbas, M. M. Seeds and seed oil compositions of Aleppo pine (Pinus halepensis Mill.) grown in Jordan. Riv. Ital. Delle Sostanze Grasse 2013, 90, 87–93. [Google Scholar]
- Tunisi, L.; Forte, N.; Fernández-Rilo, A. C.; et al. Orexin-A prevents lipopolysaccharide-induced neuroinflammation at the level of the intestinal barrier. Front. Endocrinol. 2019, 10, 219. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.Y.; Lin, P.R.; Ng, C.C.; Shyu, Y.T. Probiotic properties of Lactobacillus strains isolated from the feces of breast-fed infants and Taiwanese pickled cabbage. Anaerobe 2010, 16, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Wang, I.-K.; Wu, Y.-Y.; Yang, Y.-F.; et al. The effect of probiotics on serum levels of cytokine and endotoxin in peritoneal dialysis patients: a randomised, double blind, placebo-controlled trial. Benef. Microbes 2015, 6, 423–430. [Google Scholar] [CrossRef]
- Wang, I.K.; Yen, T.H.; Hsieh, P.S.; et al. Effect of a probiotic combination in an experimental mouse model and clinical patients with chronic kidney disease: a pilot study. Front. Nutr. 2021, 8, 661794. [Google Scholar] [CrossRef] [PubMed]
- Weber, K.T.; Alipui, D.O.; Sison, C.P.; et al. Serum levels of the proinflammatory cytokine interleukin-6 vary based on diagnoses in individuals with lumbar intervertebral disc diseases. Arthritis Res. Ther. 2016, 18, 3. [Google Scholar] [CrossRef]
- Wei, C.X.; Wu, J.H.; Huang, Y.H.; et al. Lactobacillus plantarum improves LPS-induced Caco2 cell line intestinal barrier damage via cyclic AMP-PKA signaling. PLoS One 2022, 17, e0267831. [Google Scholar] [CrossRef] [PubMed]
- Wierzbicka, A.; Ma’nkowska Wierzbicka, D.; Mardas, M.; Stelmach Mardas, M. Role of Probiotics in Modulating Human Gut Microbiota Populations and Activities in Patients with Colorectal Cancer—A Systematic Review of Clinical Trials. Nutrients 2021, 13, 1160. [Google Scholar] [CrossRef] [PubMed]
- Woo, J. K.; Choi, S.; Kang, J. H.; et al. Fermented barley and soybean (BS) mixture enhances intestinal barrier function in dextran sulfate sodium (DSS)-induced colitis mouse model. BMC Complement. Altern. Med. 2016, 16, 498. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, X.; Le, M.H.; et al. Reutericyclin producing Lactobacillus reuteri modulates development of fecal microbiota in weanling pigs. Front. Microbiol. 2015, 6, 762. [Google Scholar] [CrossRef]
- Yuki, T.; Haratake, A.; Koishikawa, H.; et al. Tight junction proteins in keratinocytes: localization and contribution to barrier function. Exp. Dermatol. 2007, 16, 324–330. [Google Scholar] [CrossRef]
- Zaroual, H.; Boughattas, F.; Karoui, R. Traditional foods in Maghreb: Production and research progress. In Traditional Foods: History, Preparation, Processing and Safety; 2019; pp. 51–113.
- Zhang, X. F.; Guan, X. X.; Tang, Y. J.; et al. Clinical effects and gut microbiota changes of using probiotics, prebiotics or synbiotics in inflammatory bowel disease: a systematic review and meta-analysis. Eur. J. Nutr. 2021, 60, 2855–2875. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Zhang, C.; et al. Postbiotics derived from Lactobacillus plantarum 1.0386 ameliorate lipopolysaccharide-induced tight junction injury via MicroRNA-200c-3p mediated activation of the MLCK-MLC pathway in Caco-2 cells. Food Funct. 2022, 13, 11008–11020. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



